Submitted:
09 April 2025
Posted:
10 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
- Group 1: Patients diagnosed with pulmonary tuberculosis (PTB) and SARS-CoV-2 coinfection (n=32)
- Group 2: Patients diagnosed with extrapulmonary tuberculosis (extraPTB) and SARS-CoV-2 coinfection (n=23)
2.2. Inclusion Criteria
- ✓
- Confirmed PTB or extraPTB diagnosis within one month prior to SARS-CoV-2 infection, verified through solid or liquid cultures or GeneXpert test at the TB ambulatory service in Timișoara [19].
- ✓
- SARS-CoV-2 infection confirmed by RT-PCR from nasopharyngeal swabs at an accredited laboratory [20]. Moderate or severe SARS-CoV-2 infection at hospital admission. A moderate case was defined by clinical signs of lower respiratory disease and SpO₂ ≥ 94% on room air, while severe cases presented SpO₂ < 94%, a respiratory rate > 30 breaths/min, or lung infiltrates > 50% [20].
- ✓
- Normal renal function (diureseis, eGFR, creatinine, and urea within normal limits) [21].
- ✓
- History of BCG vaccination [22].
2.3. Exclusion Criteria
2.4. Ethical Considerations
2.5. Data Collection
- BCG vaccination status.
- Comorbidities (COPD, T2DM).
- Smoking status (never smoked/smoker).
- History of previous TB treatment and cure status.
- Body Mass Index, calculated as weight (kg) / height² (m²) [28].
- SpO₂ (%) at SARS-CoV-2 diagnosis and lowest recorded value during hospitalization [29].
- Peripheral systolic and diastolic blood pressure (SBP, DBP, mmHg) at diagnosis
- COVID-19 symptoms, including fever, cough, dyspnea, fatigue, abdominal pain, chest pain, myalgia, vomiting, diarrhea, headache, and anosmia/ageusia. Symptoms were classified as severe if associated with tachypnea (respiratory rate ≥ 30 breaths/min) [30].
- C-reactive protein (CRP, mg/L)
- Procalcitonin (PCT, ng/mL)
- Aspartate aminotransferase (AST, U/L) and alanine aminotransferase (ALT, U/L)
- Lactate dehydrogenase (LDH, U/L)
- Interleukin-6 (IL-6, pg/mL)
- D-dimer levels (mg/L)
- Neutrophil, lymphocyte, and platelet counts (/µL)
- CT scan interpretation: an experienced radiologist (≥10 years of experience) analyzed chest CT scans at the time of SARS-CoV-2 infection confirmation. Lung involvement was assessed using a semi-quantitative CT severity score (0–25 points), based on the extent of ground-glass opacifications, consolidations, crazy paving patterns, and other abnormalities [34,35,36,37].
- Hospitalization duration (days)
- Symptoms severity
- Resolution (PCR-negative discharge) or mortality
2.6. Statistical Analysis
2.7. Study Limitations
- The retrospective, cross-sectional design limits causal inference.
- The small sample size may impact the statistical power of subgroup analyses
- The exclusion of patients with obesity and chronic diseases may reduce generalizability but ensures a focused comparison of COVID-19 manifestations in PTB vs. extraPTB.
3. Results
- a significantly lower BMI;
- a significantly lower SpO2 both at diagnosis, and with regard the lowest value registered, and a significantly higher chest CT involvement median score than the extraPTB group, revealing a clear higher pulmonary involvement;
- significantly higher SBP and DBP median values.
4. Discussion
- Demographic characteristics
- Key findings regarding clinical and inflammatory parameters
- Hospitalizationand Mortality
- Implications for Clinical Practice
- Study Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALT | Alanine aminotransferase |
| ANCOVA | one-way analysis of covariance |
| Apr | adjusted prevalence ratio |
| AST | Aspartate aminotransferase |
| AUC | Area under the receiver operating characteristic curve |
| BMI | Body mass index |
| COPD | Chronic Obstructive Pulmonary Disease |
| COVID-19 | Coronavirus Disease 2019 |
| CRP | C-reactive protein |
| CT | Computer tomography |
| DM | Diabetes Mellitus |
| extraPTB | Extrapulmonary tuberculosis |
| HIV | Human Deficiency Virus |
| IL-6 | Interleukin 6 |
| LASSO regression | Least Absolute Shrinkage and Selection Operator regression |
| LDH | Lactate Dehydrogenase |
| n | number of subjects |
| MTB | Mycobacterium tuberculosis |
| OR | odds ratio |
| p | p-value |
| PCT | Procalcitonin |
| PTB | pulmonary tuberculosis |
| ROC | Receiver operating characteristic |
| RT-PCR | Reverse Transcription Polymerase Chain Reaction |
| SpO2 | Saturation of peripheral oxygen |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| TB | Tuberculosis |
| WHO | World Health Organization |
References
- World Health Organization. Global Tuberculosis Report 2024; World Health Organization: Geneva, Switzerland. Available online: https://www.who.int/teams/global-tuberculosis-programme/tb-reports/global-tuberculosis-report-2024 (accessed on 29 October 2024).
- Houben, R.M.G.J.; Dodd, P.J. The Global Burden of Latent Tuberculosis Infection: A Re-estimation Using Mathematical Modelling. Eur. Respir. J. 2016, 48, 889–900. [Google Scholar] [CrossRef]
- Ramírez-Lapausa, M.; Menéndez-Saldaña, A.; Noguerado-Asensio, A. Extrapulmonary Tuberculosis: An Overview. Rev. Esp. Sanid. Penit. 2015, 17, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Bastián, M.; Díaz-Pollán, B.; Falces-Romero, I.; Toro-Rueda, C.; García-Rodríguez, J.; SARS-CoV-2 Working Group. Impact on Tuberculosis Diagnosis during the COVID-19 Pandemic in a Tertiary Care Hospital. Enferm. Infecc. Microbiol. Clin. 2024, 42, 135–139.
- Lee, J.Y. Diagnosis and Treatment of Extrapulmonary Tuberculosis. Tuberc. Respir. Dis. 2015, 78, 47–55. [Google Scholar] [CrossRef]
- Rodriguez-Takeuchi, S.Y.; Renjifo, M.E.; Medina, F.J. Extrapulmonary Tuberculosis: Pathophysiology and Imaging Findings. Radiographics 2019, 39, 2023–2037. [Google Scholar] [CrossRef]
- Jeong-Han, K.; Eu-Suk, K.; Kang-Il, J. Delayed Diagnosis of Extrapulmonary Tuberculosis Presenting as Fever of Unknown Origin in an Intermediate-Burden Country. BMC Infect. Dis. 2018, 18, 426. [Google Scholar] [CrossRef]
- World Health Organization. Global Tuberculosis Report 2023; World Health Organization: Geneva, Switzerland. Available online: https://www.who.int/teams/global-tuberculosis-programme/tb-reports/global-tuberculosis-report-2023 (accessed on 29 October 2024).
- World Health Organization. COVID-19 Epidemiological Update—15 March 2024; World Health Organization: Geneva, Switzerland, 2024. Available online: https://www.who.int/publications/m/item/covid-19-epidemiological-update-15-march-2024 (accessed on 29 October 2024).
- Lopes-Pacheco, M.; Leme Silva, P.; Ferreira Cruz, F. Pathogenesis of Multiple Organ Injury in COVID-19 and Potential Therapeutic Strategies. Front. Physiol. 2021, 12, 593223. [Google Scholar] [CrossRef]
- Petro, B.; Katalin, A.W.; Dylan, S. Immune Interaction between SARS-CoV-2 and Mycobacterium tuberculosis. Front. Immunol. 2023, 14, 1254206. [Google Scholar]
- Lerner, T.R.; Borel, S.; Gutierrez, M.G. The Innate Immune Response in Human Tuberculosis. Cell Microbiol. 2015, 17, 1277–1285. [Google Scholar] [CrossRef]
- Pozdnyakov, A.; Jin, A.; Bader, M. Reactivation of Pulmonary Tuberculosis in a Patient with COVID-19. Case Report and Review of Literature. Infect. Dis. Clin. (Baltim MD) 2021, 29, e468–e470. [Google Scholar] [CrossRef]
- Kaftan, A.N.; Hussain, M.K.; Algenabi, A.A.; Naser, F.H.; Enaya, M.A. Predictive Value of C-Reactive Protein, Lactate Dehydrogenase, Ferritin and D-Dimer Levels in Diagnosing COVID-19 Patients: A Retrospective Study. Acta Inform. Med. 2021, 29, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Dheda, K.; Booth, H.; Huggett, J.F.; Johnson, M.A.; Zumla, A.; Rook, G.A.W. Lung Remodeling in Pulmonary Tuberculosis. J. Infect. Dis. 2005, 192, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- Colby, S.; Shah, R. TB Reactivation Following COVID-19 Infection. Chest 2022, 162, A329. [Google Scholar] [CrossRef]
- Mihuta, C.; Socaci, A.; Hogea, P.; Tudorache, E.; Mihuta, M.S.; Oancea, C. Colliding Challenges: An Analysis of SARS-CoV-2 Infection in Patients with Pulmonary Tuberculosis versus SARS-CoV-2 Infection Alone. Medicina 2024, 60, 823. [Google Scholar] [CrossRef]
- Mihuta, C.; Socaci, A.; Oancea, C. Colliding Challenges Part 2: An Analysis of SARS-CoV-2 Infection in Patients with Extrapulmonary Tuberculosis Versus SARS-CoV-2 Infection Alone. Medicina 2024, 60, 2071. [Google Scholar] [CrossRef]
- Prakash, A.K.; Datta, B.; Goyal, P.; Chatterjee, P.; Gupta, G. GENE-XPERT gives early diagnosis in early tuberculosis. Eur. Respir. J. 2016, 48 (Suppl. S60), PA2775. [Google Scholar]
- COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available online: https://www.covid19treatmentguidelines.nih.gov/ (accessed on 1 June 2024).
- Gounden, V.; Bhatt, H.; Jialal, I. Renal Function Tests. [Updated 2023 Jul 17]. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024; Available online: https://www.ncbi.nlm.nih.gov/books/NBK507821/ (accessed on 13 June 2024).
- World Health Organization. BCG vaccines: WHO position paper—February 2018. Wkly. Epidemiol. Rec. 2018, 93, 73–96 (accessed on 13 June 2024).
- Purnell, J.Q. Purnell, J.Q. Definitions, Classification, and Epidemiology of Obesity. [Updated 4 May 2023]. In Endotext; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK279167/ (accessed on 13 June 2024).
- Akpek, M. Does COVID-19 Cause Hypertension? Angiology 2022, 73, 682–687. [Google Scholar] [CrossRef]
- Metra, M.; Dinatolo, E.; Dasseni, N. The New Heart Failure Association Definition of Advanced Heart Failure. Card. Fail. Rev. 2019, 5, 5–8. [Google Scholar] [CrossRef]
- Parker, C.S.; Siracuse, C.G.; Litle, V.R. Identifying lung cancer in patients with active pulmonary tuberculosis. JTD 2018, 10 (Suppl. S28), S3392–S3397. [Google Scholar] [CrossRef]
- Ramamoorthy, S.; Srinivas, B.H.; Badhe, B.A.; Jinkala, S.; Ganesh, R.N. Coexistence of malignancy and tuberculosis: Is it double disease or double hit related to COVID-19?—Experience from a tertiary care center. Int. J. Clin. Exp. Pathol. 2023, 16, 1–7. [Google Scholar] [PubMed]
- NHLBI Obesity Education Initiative Expert Panel on the Identification E and T of O in A (US). Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: The Evidence Report. In Obesity Prevention and Management; National Heart, Lung, and Blood Institute, Ed.; NHLBI Obesity Education Initiative Expert Panel on the Identification, Evaluation, and Treatment of Obesity in Adults (US): Bethesda, MD, USA, 1998.
- Seifi, S.; Khatony, A.; Moradi, G.; Abdi, A.; Najafi, F. Accuracy of pulse oximetry in detection of oxygen saturation in patients admitted to the intensive care unit of heart surgery: Comparison of finger, toe, forehead, and earlobe probes. BMC Nurs. 2018, 17, 15. [Google Scholar] [CrossRef] [PubMed]
- Fukui, S.; Ikeda, K.; Kobayashi, M.; Nishida, K.; Yamada, K.; Horie, S.; Shimada, Y.; Miki, H.; Goto, H.; Hayashi, K.; et al. Predictive prognostic biomarkers in patients with COVID 19 infection. Mol. Med. Rep. 2023, 27, 15. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.T.; Jabbar, A.S.; Al Ali, H.S.; Hamadi, S.S.; Jabir, M.S.; Albukhaty, S. Extensive Study on Hematological, Immunological, Inflammatory Markers, and Biochemical Profile to Identify the Risk Factors in COVID-19 Patients. Int. J. Inflam 2022, 2022, 5735546. [Google Scholar] [CrossRef]
- Ravindra, R.; Ramamurthy, P.; Aslam, S.S.M.; Kulkarni, A.; Suhail, K.; Ramamurthy, P.S. Platelet Indices and Platelet to Lymphocyte Ratio (PLR) as Markers for Predicting COVID-19 Infection Severity. Cureus 2022, 14, e28206. [Google Scholar] [CrossRef]
- Mangoni, A.A.; Zinellu, A. Systemic inflammation index, disease severity, and mortality in patients with COVID-19: A systematic review and meta-analysis. Front. Immunol. 2023, 14, 1212998. [Google Scholar] [CrossRef]
- Hansell, D.M.; Bankier, A.A.; MacMahon, H.; McLoud, T.C.; Müller, N.L.; Remy, J. Fleischner society: Glossary of terms for thoracic imaging. Radiology 2008, 246, 697–722. [Google Scholar] [CrossRef]
- Hernandez-Romieu, A.C.; Little, B.P.; Bernheim, A.; Schechter, M.C.; Ray, S.M.; Bizune, D.; Kempker, R. Increasing Number and Volume of Cavitary Lesions on Chest Computed Tomography Are Associated With Prolonged Time to Culture Conversion in Pulmonary Tuberculosis. Open Forum Infect Dis. 2019, 6, ofz232. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Yu, C.-J.; Chang, S.-C.; Galvin, J.R.; Liu, H.-M.; Hsiao, C.-H.; Kuo, P.-H.; Chen, K.-Y.; Franks, T.J.; Huang, K.-M.; et al. Pulmonary sequelae in convalescent patients after severe acute respiratory syndrome: Evaluation with thin-section CT. Radiology 2005, 236, 1067–1075. [Google Scholar] [CrossRef]
- Yazdi, N.A.; Ghadery, A.H.; SeyedAlinaghi, S.; Jafari, F.; Jafari, S.; Hasannezad, M.; Koochak, H.E.; Salehi, M.; Manshadi, S.A.D.; Meidani, M.; et al. Predictors of the chest CT score in COVID-19 patients: A cross-sectional study. Virol. J. 2021, 18, 225. [Google Scholar] [CrossRef]
- Borgdorff, M.W.; Nagelkerke, N.J.; Dye, C.; Nunn, P. Gender and tuberculosis: a comparison of prevalence surveys with notification data to explore sex differences in case detection. Int. J. Tuberc. Lung Dis. 2000, 4, 123–132. [Google Scholar] [PubMed]
- Uplekar, M.W.; Rangan, S.; Weiss, M.G.; et al. Attention to gender issues in tuberculosis control. Int. J. Tuberc. Lung Dis. 2001, 5, 220–224. [Google Scholar] [PubMed]
- Secretaría de Salud, Subsecretaría de Prevención y Control de Enfermedades, Instituto Nacional de Salud Pública. Encuesta Nacional de Salud 2000; 1st ed.; Secretaría de Salud: Mexico City, Mexico, 2000; pp. 41–50.
- Balasubramanian, R.; Garg, R.; Santha, T.; et al. Gender disparities in tuberculosis: Report from a rural DOTS programme in South India. Int. J. Tuberc. Lung Dis. 2004, 8, 323–332. [Google Scholar] [PubMed]
- Fine, P.E.M. Immunities in and to tuberculosis: Implications for pathogenesis and vaccination. In Tuberculosis Back to the Future; Porter, J.D.H., McAdam, K.P.W.J., Eds.; Wiley and Sons: Chichester, UK, 1993; pp. 53–78. [Google Scholar]
- Murray, C.J.L. Social, economic and operational research on tuberculosis: Recent studies and some priority questions. Bull. Int. Union Tuberc. Lung Dis. 1991, 66, 149–156. [Google Scholar]
- Bates, I.; Fenton, C.; Gruber, J.; et al. Vulnerability to malaria, tuberculosis, and HIV/AIDS infection and disease. Part 1: Determinants operating at individual and household level. Lancet Infect. Dis. 2004, 4, 267–277. [Google Scholar] [CrossRef]
- Ponce-de-Leon, A.; Garcia-Garcia, M.L.; Garcia-Sancho, M.C.; et al. Tuberculosis and diabetes in southern Mexico. Diabetes Care 2004, 27, 1584–1590. [Google Scholar] [CrossRef]
- Thulstrup, A.M.; Molle, I.; Svendsen, N.; et al. Incidence and prognosis of tuberculosis in patients with cirrhosis of the liver: A Danish nationwide population-based study. Epidemiol. Infect. 2000, 124, 221–225. [Google Scholar] [CrossRef]
- Caracta, C.F. Gender differences in pulmonary disease. Mt Sinai J. Med. 2003, 70, 215–224. [Google Scholar]
- Tekkel, M.; Rahu, M.; Loit, H.M.; et al. Risk factors for pulmonary tuberculosis in Estonia. Int. J. Tuberc. Lung Dis. 2002, 6, 887–894. [Google Scholar]
- Curtis, A.B.; Ridzon, R.; Novick, L.F.; et al. Analysis of Mycobacterium tuberculosis transmission patterns in a homeless shelter outbreak. Int. J. Tuberc. Lung Dis. 2000, 4, 308–313. [Google Scholar]
- García-García, M.; Palacios-Martinez, M.; Ponce-de-León, A.; et al. The role of core groups in transmitting Mycobacterium tuberculosis in a high prevalence community in Southern Mexico. Int. J. Tuberc. Lung Dis. 2000, 4, 12–17. [Google Scholar] [PubMed]
- Godoy, P.; Nogues, A.; Alseda, M.; et al. Risk factors associated with tuberculosis patients with positive sputum microscopy. Gac Sanit 2001, 15, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Kolappan, C.; Gopi, P.G. Tobacco smoking and pulmonary tuberculosis. Thorax 2002, 57, 964–966. [Google Scholar] [CrossRef] [PubMed]
- Baya, B.; Sanogo, I.; Kone, M.; Soumare, D.; Ouattara, K.; Somboro, A.; Wague, M.; Coulibaly, N.; Koloma, I.; Coulibaly, M.; Nantoume, M.; Perou, M.; Kone, K.; Coulibaly, D.; Boukary Diarra, H.; Kone, B.; Diarra, A.; Coulibaly, M.D.; Sanogo, M.; Diarra, B.; Diakite, M.; Achenbach, C.J.; Doumbia, S.; Bishai, W.R.; Klein, S.L.; Holl, J.L.; Diallo, S.; Murphy, R.L.; Toloba, Y.; Dabitao, D. Relationship between patient sex and anatomical sites of extrapulmonary tuberculosis in Mali. J. Clin. Tuberc. Other Mycobact. Dis. 2023, 33, 100389. J. Clin. Tuberc. Other Mycobact. Dis. 2023, 33, 100389. [Google Scholar] [CrossRef]
- Min, J.; Lee, J.H.; Kim, S.Y.; Park, J.S.; Cho, Y.J.; Yoon, H.I.; Lee, C.T.; Lee, J.H. Differential effects of sex on tuberculosis location and severity across the lifespan. Sci. Rep. 2023, 13, 33245. [Google Scholar] [CrossRef]
- Ramírez-Lapausa, M.; Menéndez-Saldaña, A.; Noguerado-Asensio, A. Extrapulmonary Tuberculosis: An Overview. Rev. Esp. Sanid. Penit. 2015, 17, 3–11. [Google Scholar] [CrossRef]
- Caraux-Paz, P.; Diamantis, S.; de Wazières, B.; Gallien, S. Tuberculosis in the Elderly. J. Clin. Med. 2021, 10, 5888. [Google Scholar] [CrossRef]
- Bartleson, J.M.; Radenkovic, D.; Covarrubias, A.J.; Furman, D.; Winer, D.A.; Verdin, E. SARS-CoV-2, COVID-19 and the Ageing Immune System. Nat. Aging. 2021, 1, 769–782. [Google Scholar] [CrossRef]
- Daneshvar, P.; Hajikhani, B.; Sameni, F.; Noorisepehr, N.; Zare, F.; Bostanshirin, N.; Yazdani, S.; Goudarzi, M.; Sayyari, S.; Dadashi, M. COVID-19 and Tuberculosis Coinfection: An Overview of Case Reports/Case Series and Meta-Analysis of Prevalence Studies. Heliyon 2023, 9, e13637. [Google Scholar] [CrossRef]
- Nuwagira, E.; Mpagama, S.G.; Katusiime, A.; Natamba, B.; Baluku, J.B.; Lai, P.S. Coinfection of COVID-19 and Tuberculosis in Uganda. Am. J. Trop. Med. Hyg. 2023, 108, 1240–1243. [Google Scholar] [CrossRef]
- Nabity, S.A.; Marks, S.M.; Goswami, N.D.; et al. Characteristics of and Deaths among 333 Persons with Tuberculosis and COVID-19 in Cross-Sectional Sample from 25 Jurisdictions, United States. Emerg. Infect. Dis. 2023, 29, 2016–2023. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, Y.; Gu, L.; Lou, L.; Zhang, J.; Zhang, K. The Clinical Characteristics and Risk Factors for Severe COVID-19 in Patients with COVID-19 and Tuberculosis Coinfection. Front. Microbiol. 2022, 13, 1061879. [Google Scholar] [CrossRef] [PubMed]
- Ayed, H.B.; Koubaa, M.; Marrakchi, C.; Rekik, K.; Hammami, F.; Smaoui, F.; Ben Hmida, M.; Yaich, S.; Maaloul, I.; Damak, J.; Ben Jemaa, M. Extrapulmonary Tuberculosis: Update on the Epidemiology, Risk Factors and Prevention Strategies. Int. J. Trop. Dis. 2018, 1, 006. [Google Scholar] [CrossRef]
- Wang, Q.; Cao, Y.; Liu, X.; Fu, Y.; Zhang, J.; Zhang, Y.; Zhang, L.; Wei, X.; Yang, L. Systematic Review and Meta-Analysis of Tuberculosis and COVID-19 Co-Infection: Prevalence, Fatality, and Treatment Considerations. PLoS Negl. Trop. Dis. 2024, 18, e0012136. [Google Scholar] [CrossRef]
- Booysen, P.; Wilkinson, K.A.; Sheerin, D.; Waters, R.; Coussens, A.K.; Wilkinson, R.J. Immune Interaction between SARS-CoV-2 and Mycobacterium tuberculosis. Front. Immunol. 2023, 14, 1254206. [Google Scholar] [CrossRef]
- Nathella Pavan, K.; Arul, P.N.; Kadar, M.; Pradeep, A.M.; Vaithilingam, V.B.; Nair, D.; Sujatha, N.; Babu, S. Low Body Mass Index Is Associated with Diminished Plasma Cytokines and Chemokines in Both Active and Latent Tuberculosis. Front. Nutr. 2023, 10, 1194682. [Google Scholar] [CrossRef]
- Edwards, L.B.; Livesay, V.T.; Acquaviva, F.A. , et al. Height, Weight, Tuberculous Infection, and Tuberculous Disease. Arch. Environ. Health. 1971, 22, 106–112. [Google Scholar] [CrossRef]
- Tverdal, A. Body Mass Index and Incidence of Tuberculosis. Eur. J. Respir. Dis. 1986, 69, 355–362. [Google Scholar]
- Oda, G.; Lucero-Obusan, C.; Schirmer, P.; Chung, J.; Holodniy, M. Risk Factors for Extrapulmonary Tuberculosis Among US Veterans, 1990–2022. Open Forum Infect. Dis. 2024, 11, ofae698. [Google Scholar] [CrossRef]
- Kornfeld, H.; Sahukar, S.B.; Procter-Gray, E.; Kumar, N.P.; West, K.; Kane, K.; Natarajan, M.; Li, W.; Babu, S.; Viswanathan, V. Impact of Diabetes and Low Body Mass Index on Tuberculosis Treatment Outcomes. Clin. Infect. Dis. 2020, 71, e392–e398. [Google Scholar] [CrossRef]
- Slane, V.H.; Unakal, C.G. Tuberculous Meningitis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025; Available online: https://www.ncbi.nlm.nih.gov/books/NBK541015/ (accessed on February 2, 2025).
- Gao, M.; Piernas, C.; Astbury, N.M.; et al. Associations between Body-Mass Index and COVID-19 Severity in 6.9 Million People in England: A Prospective, Community-Based, Cohort Study. Lancet Diabetes Endocrinol. 2021, 9, 350–359. [CrossRef]
- Lockhart, S.M.; O'Rahilly, S. When Two Pandemics Meet: Why Is Obesity Associated with Increased COVID-19 Mortality? Med (N. Y.) 2020, 1, 33–42. [Google Scholar] [CrossRef]
- Hewitt, J.; Carter, B.; Vilches-Moraga, A.; et al. The Effect of Frailty on Survival in Patients with COVID-19 (COPE): A Multicentre, European, Observational Cohort Study. Lancet Public Health 2020, 5, e444–e451. [Google Scholar] [CrossRef] [PubMed]
- Jayanama, K.; Theou, O.; Godin, J.; et al. Relationship of Body Mass Index with Frailty and All-Cause Mortality Among Middle-Aged and Older Adults. BMC Med. 2022, 20, 404. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, D.; Yoshida, T.; Watanabe, Y.; Yamada, Y.; Kimura, M. A U-Shaped Relationship between the Prevalence of Frailty and Body Mass Index in Community-Dwelling Japanese Older Adults: The Kyoto–Kameoka Study. J. Clin. Med. 2020, 9, 1367. [Google Scholar] [CrossRef] [PubMed]
- de Martino, M.; Lodi, L.; Galli, L.; Chiappini, E. Immune Response to Mycobacterium Tuberculosis: A Narrative Review. Front. Pediatr. 2019, 7, 350. [Google Scholar] [CrossRef]
- Flores-Lovon, K.; Ortiz-Saavedra, B.; Cueva-Chicaña, L.A.; Aperrigue-Lira, S.; Montes-Madariaga, E.S.; Soriano-Moreno, D.R.; Bell, B.; Macedo, R. Immune Responses in COVID-19 and Tuberculosis Coinfection: A Scoping Review. Front. Immunol. 2022, 13, 992743. [Google Scholar] [CrossRef]
- Hiti, L.; Markovič, T.; Lainscak, M.; Farkaš Lainščak, J.; Pal, E.; Mlinarič-Raščan, I. The Immunopathogenesis of a Cytokine Storm: The Key Mechanisms Underlying Severe COVID-19. Cytokine Growth Factor Rev. 2025, 82, 1–17. [Google Scholar] [CrossRef]
- Wang, H.; Wang, Z.; Cao, W.; Wu, Q.; Yuan, Y.; Zhang, X. Regulatory T Cells in COVID-19. Aging Dis. 2021, 12, 1545. [Google Scholar] [CrossRef]
- Galván-Peña, S.; Leon, J.; Chowdhary, K.; Michelson, D.A.; Vijaykumar, B.; Yang, L.; Magnuson, A.M.; Chen, F.; Manickas-Hill, Z.; Piechocka-Trocha, A.; et al. Profound Treg Perturbations Correlate with COVID-19 Severity. Proc. Natl. Acad. Sci. USA. 2021, 118, e2111315118. [Google Scholar] [CrossRef]
- Tan, L.; Wang, Q.; Zhang, D.; et al. Lymphopenia Predicts Disease Severity of COVID-19: A Descriptive and Predictive Study. Signal Transduct. Target Ther. 2020, 5, 33. [Google Scholar] [CrossRef]
- Cioboata, R.; Vasile, C.M.; Bălteanu, M.A.; Georgescu, D.E.; Toma, C.; Dracea, A.S.; Nicolosu, D. Evaluating Serum Calcium and Magnesium Levels as Predictive Biomarkers for Tuberculosis and COVID-19 Severity: A Romanian Prospective Study. Int. J. Mol. Sci. 2024, 25, 418. [Google Scholar] [CrossRef] [PubMed]
- TB/COVID-19 Global Study Group. Tuberculosis and COVID-19 Co-Infection: Description of the Global Cohort. Eur. Respir. J, 2102; 59. [CrossRef]
- Bostanghadiri, N.; Jazi, F.M.; Razavi, S.; Fattorini, L.; Darban-Sarokhalil, D. Mycobacterium tuberculosis and SARS-CoV-2 Coinfections: A Review. Front. Microbiol. 2022, 12, 747827. [Google Scholar] [CrossRef] [PubMed]
- Cioboata, R.; Biciusca, V.; Olteanu, M.; Vasile, C.M. COVID-19 and Tuberculosis: Unveiling the Dual Threat and Shared Solutions Perspective. J. Clin. Med. 2023, 12, 4784. [Google Scholar] [CrossRef] [PubMed]
- Maritescu, A.; Crisan, A.F.; Pescaru, C.C.; Stoicescu, E.R.; Oancea, C.; Iacob, D. Effectiveness of Combined Pulmonary Rehabilitation and Progressive Muscle Relaxation in Treating Long-Term COVID-19 Symptoms: A Randomized Controlled Trial. J. Clin. Med. 2024, 13, 6237. [Google Scholar] [CrossRef]
| PTB-SARS-CoV-2 n=32 |
extraPTB-SARS-CoV-2 n=23 |
||
| Sex | F | 11 (34%) | 10 (43.5%) |
| M | 21 (65%) | 13 (56.5%) | |
| Age | 19-40 y | 2 (6.2%) | 15 (65.2%) |
| 41-65 y | 16 (50%) | 6 (26.1%) | |
| >66 y | 14 (43.8%) | 2 (8.7%) |
| Parameter | PTB-SARS-CoV-2 median value |
extraPTB-SARS-CoV-2 median value |
p |
| BMI (kg/m2) | 21.88 | 24.45 | 0.01 |
| SpO2 at diagnosis (%) | 90 | 96 | <0.001 |
| Lowest SpO2 (%) | 83.5 | 92 | 0.001 |
| Peripheral SBP at diagnosis (mmHg) | 138 | 129 | 0.03 |
| Peripheral DBP at diagnosis (mmHg) | 92 | 85 | 0.04 |
| CRP (mg/dL) | 89.5 | 66.1 | 0.01 |
| LDH (UI/L) | 288 | 203 | 0.14 |
| IL-6 (pg/mL) | 4.2 | 8.8 | 0.009 |
| AST (UI/L) | 36 | 38 | 0.28 |
| ALT (UI/L) | 38.5 | 42 | 0.74 |
| D-dimer (mg/L) | 1.91 | 1.65 | 0.55 |
| Neutrophils/uL* | 1539.69 (SD=933.29) | 6194.8 (SD=1837.6) | <0.001 |
| Lymphocytes/uL | 2210 | 1460 | 0.04 |
| Thrombocytes/uL | 242500 | 351000 | 0.02 |
| NLR | 2 | 3.73 | <0.001 |
| PLR | 128.85 | 235.54 | 0.01 |
| SII | 134549.68 | 1255888.88 | <0.001 |
| Chest CT Involvement score | 16 | 6 | <0.001 |
| Parameter | p |
|---|---|
| Sex | 0.7 |
| Smoking | 0.4 |
| Associating COPD | 0.86 |
| Associatiang T2MD | 0.82 |
| Severity of symptoms | 0.7 |
| Outcome | 0.54 |
| Feature | Importance |
| Lowest SpO2 | 0.20 |
| SpO2 at diagnosis | 0.15 |
| Age | 0.08 |
| CT involvement score | 0.07 |
| Lymphocyte count | 0.07 |
| LDH | 0.06 |
| CRP | 0.06 |
| D-dimer | 0.05 |
| IL6 | 0.05 |
| Neutrophil count | 0.04 |
| AST | 0.03 |
| Thrombocyte count | 0.03 |
| ALT | 0.03 |
| BMI | 0.02 |
| PLR | 0.02 |
| SII | 0.02 |
| NLR | 0.02 |
| Type of TB | 0.01 |
| Precision | Recall | F1-score | Support | |
| Mild symptoms | 1 | 1 | 1 | 38 |
| Severe symptoms | 1 | 1 | 1 | 17 |
| Accuracy | 1 | 1 | 1 | 1 |
| Macro Average | 1 | 1 | 1 | 55 |
| Weighted Average | 1 | 1 | 1 | 55 |
| Type of TB | Outcome | Smo-king | COPD | T2DM | Prior TB | PCT | NLR | PLR | SII |
D -dimer |
Hospitalization | SpO2 at dg | Lowest SpO2 | CT score | |
| Type of TB | 1 | -0.28 | 0.1 | -0.02 | -0.27 | -0.90 | -0.07 | 0.66 | 0.35 | 0.66 | 0.08 | 0.73 | 0.53 | 0.48 | -0.77 |
| p | 0 | 0.04 | 0.45 | 0.9 | 0.05 | <0.001 | 0.6 | <0.001 | 0.008 | <0.001 | 0.56 | <0.001 | <0.001 | 0.0002 | <0.001 |
| Outcome | -0.28 | 1 | 0.12 | 0.17 | 0.39 | 0.32 | 0.31 | 0.17 | 0.34 | 0.13 | 0.28 | 0.001 | -0.45 | -0.50 | 0.61 |
| p | 0.04 | 0 | 0.37 | 0.2 | 0.003 | 0.02 | 0.02 | 0.22 | 0.01 | 0.34 | 0.04 | 0.99 | <0.001 | <0.001 | <0.001 |
| Smoking | 0.10 | 0.12 | 1 | 0.36 | -0.11 | -0.16 | -0.07 | 0.13 | 0.08 | 0.08 | 0.14 | 0.21 | -0.12 | -0.03 | -0.04 |
| p | 0.45 | 0.37 | 0 | 0.006 | 0.44 | 0.25 | 0.6 | 0.33 | 0.57 | 0.57 | 0.32 | 0.12 | 0.39 | 0.8 | 0.79 |
| COPD | -0.02 | 0.17 | 0.36 | 1 | 0.13 | 0.07 | -0.06 | 0.10 | 0.02 | 0.04 | 0.22 | 0.14 | -0.17 | -0.22 | 0.04 |
| p | 0.9 | 0.2 | 0.006 | 0 | 0.33 | 0.62 | 0.67 | 0.48 | 0.88 | 0.76 | 0.11 | 0.31 | 0.21 | 0.1 | 0.78 |
| T2DM | -0.27 | 0.39 | -0.11 | 0.13 | 1 | 0.33 | 0.39 | -0.01 | 0.11 | -0.04 | 0.23 | -0.06 | -0.47 | -0.46 | 0.36 |
| p | 0.05 | 0.003 | 0.44 | 0.33 | 0 | 0.01 | 0.003 | 0.94 | 0.42 | 0.79 | 0.09 | 0.65 | <0.001 | <0.001 | 0.007 |
| Prior TB | -0.90 | 0.32 | -0.16 | 0.07 | 0.33 | 1 | 0.14 | -0.52 | -0.22 | -0.53 | -0.006 | -0.66 | -0.54 | -0.51 | 0.75 |
| p | <0.001 | 0.02 | 0.25 | 0.62 | 0.01 | 0 | 0.32 | <0.001 | 0.11 | <0.001 | 0.96 | <0.001 | <0.001 | <0.001 | <0.001 |
| PCT | -0.07 | 0.31 | -0.07 | -0.06 | 0.39 | 0.14 | 1 | 0.33 | 0.48 | 0.37 | 0.44 | 0.04 | -0.47 | -0.53 | 0.19 |
| p | 0.60 | 0.02 | 0.60 | 0.67 | 0.003 | 0.32 | 0 | 0.01 | <0.001 | 0.005 | <0.001 | 0.79 | <0.001 | <0.001 | 0.16 |
| NLR | 0.66 | 0.17 | 0.13 | 0.10 | -0.01 | -0.52 | 0.33 | 1 | 0.77 | 0.95 | 0.45 | 0.62 | -0.0016 | -0.05 | -0.29 |
| p | <0.001 | 0.22 | 0.33 | 0.48 | 0.94 | <0.001 | 0.01 | 0 | <0.001 | <0.001 | <0.001 | <0.001 | 0.99 | 0.72 | 0.03 |
| PLR | 0.35 | 0.34 | 0.08 | 0.02 | 0.11 | -0.22 | 0.48 | 0.77 | 1 | 0.86 | 0.66 | 0.26 | -0.38 | -0.42 | -0.05 |
| p | 0.008 | 0.01 | 0.57 | 0.88 | 0.42 | 0.11 | <0.001 | <0.001 | 0 | <0.001 | <0.001 | 0.06 | 0.004 | 0.001 | 0.73 |
| SII | 0.66 | 0.13 | 0.08 | 0.04 | -0.04 | -0.53 | 0.37 | 0.95 | 0.86 | 1 | 0.54 | 0.54 | -0.05 | -0.11 | -0.32 |
| p | <0.001 | 0.34 | 0.57 | 0.76 | 0.79 | <0.001 | 0.005 | <0.001 | <0.001 | 0 | <0.001 | <0.001 | 0.70 | 0.41 | 0.02 |
| D-dimer | 0.08 | 0.28 | 0.14 | 0.22 | 0.23 | -0.006 | 0.44 | 0.45 | 0.66 | 0.54 | 1 | 0.14 | -0.42 | -0.48 | 0.07 |
| p | 0.56 | 0.04 | 0.32 | 0.11 | 0.09 | 0.96 | <0.001 | <0.001 | <0.001 | <0.001 | 0 | 0.31 | 0.001 | <0.001 | 0.62 |
| Hospitalization | 0.73 | 0.001 | 0.21 | 0.14 | -0.06 | -0.66 | 0.04 | 0.62 | 0.26 | 0.54 | 0.14 | 1 | 0.35 | 0.29 | -0.36 |
| p | <0.001 | 0.99 | 0.12 | 0.31 | 0.65 | <0.001 | 0.79 | <0.001 | 0.06 | <0.001 | 0.31 | 0 | 0.009 | 0.03 | 0.007 |
|
SpO2 at dg |
0.53 | -0.45 | -0.12 | -0.17 | -0.47 | -0.54 | -0.47 | -0.0016 | -0.38 | -0.05 | -0.42 | 0.35 | 1 | 0.95 | -0.57 |
| p | <0.001 | <0.001 | 0.39 | 0.21 | <0.001 | <0.001 | <0.001 | 0.99 | 0.004 | 0.70 | 0.001 | 0.009 | 0 | <0.001 | <0.001 |
| Lowest SpO2 | 0.48 | -0.50 | -0.03 | -0.22 | -0.46 | -0.51 | -0.53 | -0.05 | -0.42 | -0.11 | -0.48 | 0.29 | 0.95 | 1 | -0.52 |
| p | <0.001 | <0.001 | 0.8 | 0.1 | <0.001 | <0.001 | <0.001 | 0.72 | 0.001 | 0.41 | <0.001 | 0.03 | <0.001 | 0 | <0.001 |
| CT score | -0.77 | 0.61 | -0.04 | 0.04 | 0.36 | 0.75 | 0.19 | -0.29 | -0.05 | -0.32 | 0.07 | -0.36 | -0.57 | -0.52 | 1 |
| p | <0.001 | <0.001 | 0.79 | 0.78 | 0.007 | <0.001 | 0.16 | 0.03 | 0.73 | 0.02 | 0.62 | 0.007 | <0.001 | <0.001 | 0 |
| Parameter | AUC | Criterion (cut-off) | p-value | Sensitivity % | Specificity % | PPV | NPV |
| BMI | 0.7 | 23.45 | 0.06 | 65.22 | 84.38 | 0.75 | 0.77 |
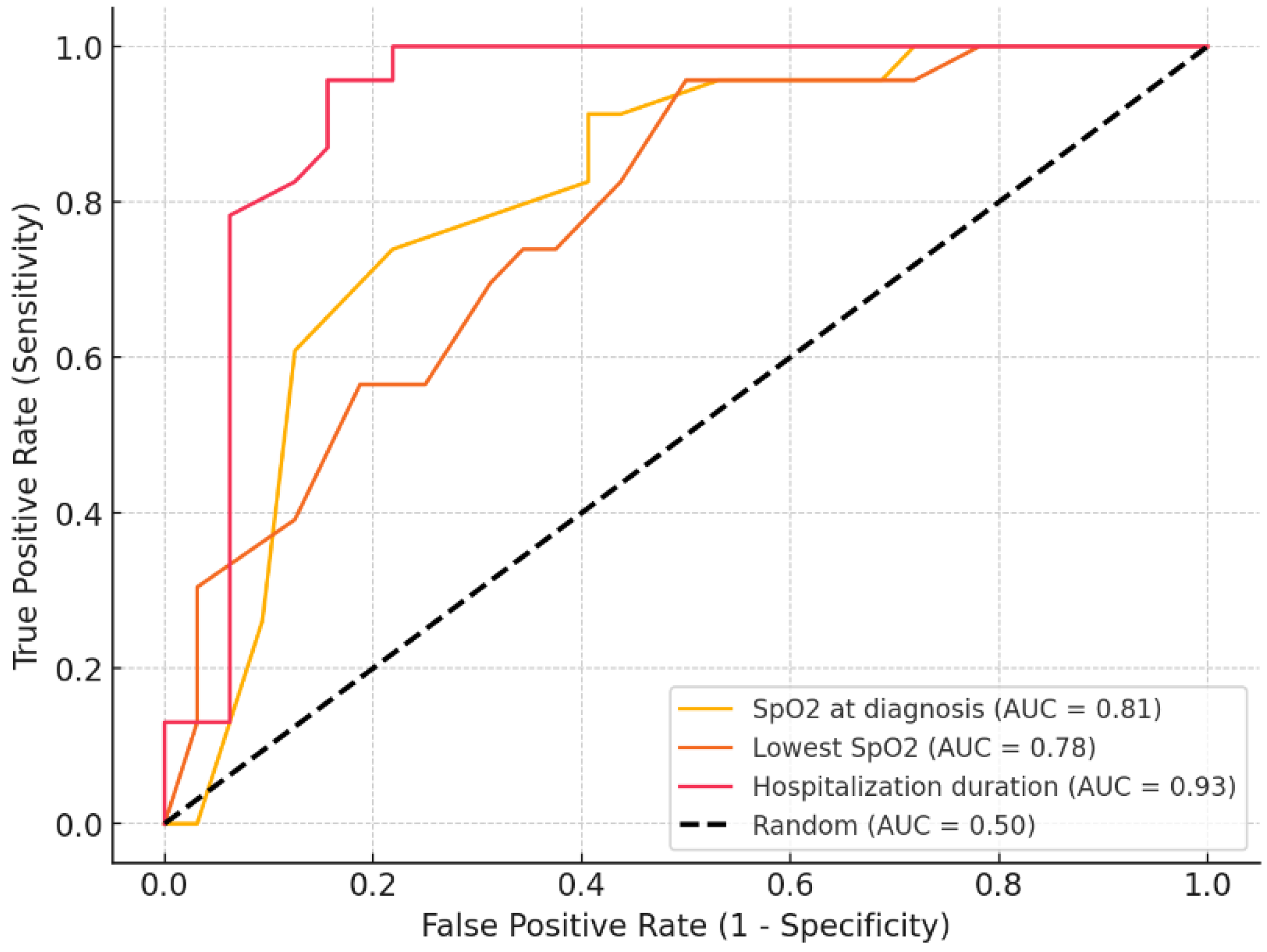
| SpO2 at diagnosis | 0.81 | 95 | 0.01 | 73.91 | 78.12 | 0.71 | 0.81 |
| Lowest SpO2 | 0.78 | 84 | 0.03 | 95.65 | 50 | 0.58 | 0.94 |
| AST | 0.59 | 74 | 0.62 | 34.78 | 100 | 1 | 0.68 |
| ALT | 0.53 | 95 | 0.38 | 21.74 | 100 | 1 | 0.64 |
| LDH | 0.38 | 166 | 0.09 | 86.96 | 25 | 0.45 | 0.73 |
| IL6 | 0.71 | 8.8 | 0.07 | 52.17 | 84.38 | 0.71 | 0.71 |
| D-dimer | 0.55 | 4.86 | 0.12 | 26.09 | 100 | 1 | 0.65 |
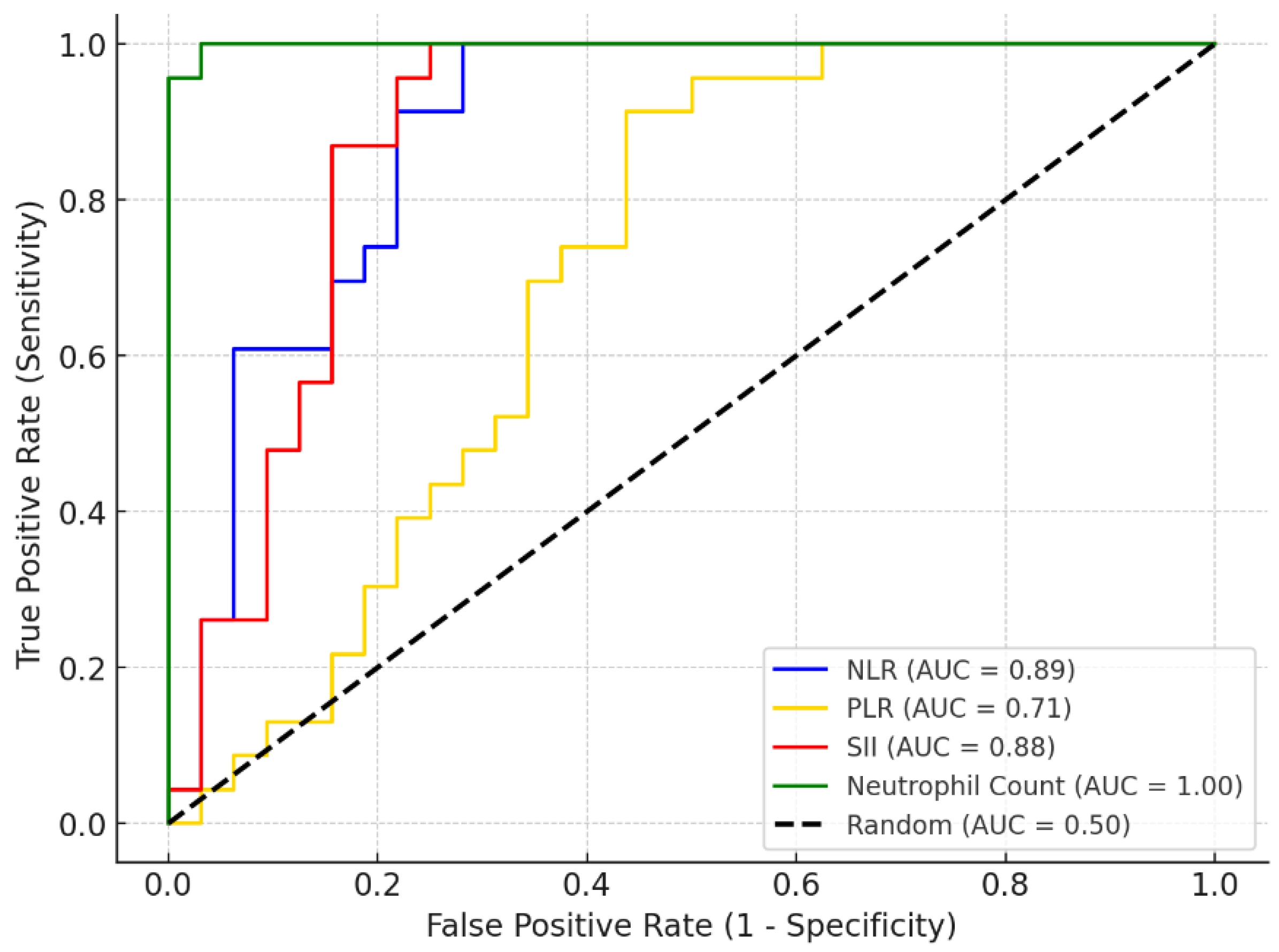
| Neutrophil count | 0.95 | 3300 | <0.001 | 100 | 96.88 | 0.96 | 1 |
| Lymphocyte count | 0.34 | 810 | 0.23 | 91.3 | 15.62 | 0.44 | 0.71 |
| Thrombocyte count | 0.68 | 242000 | 0.08 | 91.30 | 50.00 | 0.57 | 0.89 |
| NLR | 0.89 | 1.57 | 0.02 | 100 | 71.88 | 0.72 | 1 |
| PLR | 0.71 | 154.95 | 0.06 | 91.3 | 56.25 | 0.6 | 0.9 |
| SII | 0.88 | 437758.9 | 0.04 | 100 | 75 | 0.74 | 1 |
| Hospitalization | 0.93 | 20 | 0.02 | 95.65 | 84.38 | 0.81 | 0.96 |
| Sum of squares | df | F | p | |
| Type of TB | 50.62 | 1 | 1.53 | 0.22 |
| Age | 105.41 | 1 | 3.18 | 0.08 |
| BMI | 305.41 | 1 | 9.21 | 0.004 |
| SpO2 at diagnosis | 2.05 | 1 | 0.06 | 0.81 |
| Lowest SpO2 | 19.89 | 1 | 0.60 | 0.44 |
| CRP | 79.45 | 1 | 2.39 | 0.13 |
| AST | 9.57 | 1 | 0.29 | 0.59 |
| ALT | 1.08 | 1 | 0.03 | 0.86 |
| LDH | 4.76 | 1 | 0.14 | 0.71 |
| IL6 | 151.40 | 1 | 4.56 | 0.04 |
| D-dimer | 287.69 | 1 | 8.67 | 0.005 |
| Neutrophil count | 524.87 | 1 | 15.82 | 0.0003 |
| Thrombocyte count | 24.06 | 1 | 0.73 | 0.4 |
| Lymphocyte count | 102.70 | 1 | 3.10 | 0.08 |
| CT_involvement_score | 100.32 | 1 | 3.02 | 0.09 |
| NLR | 79.10 | 1 | 2.38 | 0.13 |
| PLR | 108.50 | 1 | 3.27 | 0.08 |
| SII | 181.05 | 1 | 5.46 | 0.03 |
| Residual | 1194.30 | 36 |
| Variable | β Coefficient |
| Neutrophil count | 6.526728 |
| Type of TB | 4.713506 |
| IL6 | 4.039135 |
| Thrombocyte count | -3.68729 |
| BMI | -2.59693 |
| CT involvement score | 2.152821 |
| D-dimer levels | 1.903791 |
| SII | -1.77909 |
| CRP | -1.45796 |
| LDH | 1.201368 |
| AGE | -0.74844 |
| AST | 0.699141 |
| PLR | -0.14857 |
| SpO2 at diagnosis | 0 |
| Lowest SpO2 | 0 |
| ALT | 0 |
| Lymphocyte count | 0 |
| NLR | 0 |
| Age | BMI | SpO2 at diagnosis | Lowest SpO2 |
| 61 | 21.22 | 89 | 79 |
| CRP | IL6 | LDH | D-dimer |
| 112 | 9.1 | 388 | 3.31 |
| Neutrophil count | Lymphocyte count | Thrombocyte count | CT involvement score |
| 2910 | 1000 | 380000 | 22 |
| NLR | PLR | SII | Hospitalization |
| 2.88 | 333.33 | 717250 | 18 |
| Variable | Coefficient | Odds Ratio | 95% CI Lower | 95% CI Upper |
| Type of TB | -0.01 | 0.99 | 0.76 | 1.28 |
| Age | -0.55 | 0.58 | 0.44 | 0.75 |
| BMI | -0.06 | 0.94 | 0.73 | 1.23 |
| SpO2 at diagnosis | 0.21 | 1.23 | 0.95 | 1.6 |
| Lowest SpO2 | -0.56 | 0.57 | 0.44 | 0.74 |
| CRP | -0.10 | 0.90 | 0.69 | 1.17 |
| AST | -0.22 | 0.80 | 0.61 | 1.04 |
| ALT | -0.07 | 0.93 | 0.71 | 1.21 |
| LDH | 0.39 | 1.47 | 1.13 | 1.91 |
| IL6 | 0.51 | 1.67 | 1.29 | 2.17 |
| D-dimer | 0.002 | 1 | 0.77 | 1.3 |
| Neutrophil count | 0.26 | 1.3 | 1 | 1.68 |
| Lymphocyte count | -0.76 | 0.47 | 0.36 | 0.61 |
| Thrombocyte count | -0.20 | 0.82 | 0.63 | 1.06 |
| CT involvement score | 1.88 | 6.57 | 5.06 | 8.54 |
| NLR | -0.23 | 0.79 | 0.61 | 1.03 |
| PLR | 0.15 | 1.16 | 0.89 | 1.51 |
| SII | -0.11 | 0.9 | 0.69 | 1.17 |
| Feature | Importance |
| CT involvement score | 0.22 |
| Lowest SpO2 | 0.13 |
| SpO2 at diagnosis | 0.09 |
| CRP | 0.08 |
| LDH | 0.06 |
| Lymphocyte count | 0.06 |
| IL6 | 0.06 |
| Age | 0.04 |
| BMI | 0.04 |
| PLR | 0.04 |
| Neutrophil count | 0.03 |
| NLR | 0.03 |
| D-dimer | 0.03 |
| SII | 0.03 |
| AST | 0.02 |
| Type of TB | 0.01 |
| Thrombocyte count | 0.01 |
| ALT | 0.01 |
| Precision | Recall | F1-score | Support | |
| Resolution | 0.9 | 1 | 0.94 | 9 |
| Fatality | 1 | 0.5 | 0.66 | 2 |
| Accuracy | 0.9 | 0.9 | 0.9 | 0.9 |
| Macro Average | 0.95 | 0.75 | 0.8 | 11 |
| Weighted Average | 0.91 | 0.9 | 0.89 | 11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




