Submitted:
10 April 2025
Posted:
10 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Characteristics of Potato Varieties
2.2. Characteristics of Effective Microorganisms
2.3. Field Tests
2.4. Examination of the Darkening of the Tuber Flesh

2.4.1. Darkening of Cooked Potato Flesh
2.5. Potato Tuber Texture Analysis
2.6. Soil Conditions
2.7. Meteorological Conditions
2.8. Statistical Calculations
3. Results
3.1. Darkening of Raw of Tubers
3.2. Darkening of the Tuber Flesh After Cooking
3.3. Rheological Studies
3.4. Statistical Description and Relationships Between Flesh Darkening and Biotic and Abiotic Factors
3.5. Pearson Correlation Coefficients Between Darkening Characteristics of Raw and Cooked Potato Tuber Flesh and Rheological Properties of Cooked Flesh
4. Discussion
4.1. Benefits of Using EMFarming Plus
4.2. Factors Influencing the Intensity of Darkening of the Flesh of Raw and Cooked Tubers
4.3. The Influence of Cultivars on the Darkening and Rheological Evaluation of Tuber Flesh
4.4. The Influence of External and Internal Factors on the Structure of Tuber Flesh
4.5. Practical Recommendations
4.6. Limitations of the Research
4.6.1. Limitations of the Use of Effective Microorganisms (EM) Technology in Potato Cultivation
4.6.2. Texture
4.7. Potato Tuber Flesh Darkening: Influencing Factors, Research Limitations, and Future Perspectives
4.8. Statistical Characterization of Variability and Distribution of Flesh Darkening Characteristics and Rheological Properties
4.8.1. Comparison of the Distributions of Darkening Features and Rheological Properties Between Varieties
5. Implications and Perspectives
- -
- Impact on the biodiversity of soil microflora and the stability of agroecosystems.
- -
- Impact on soil fertility and its retention capacity.
- -
- Mechanisms of impact on biogeochemical processes:
- -
- The role of EM in nitrogen and carbon cycles, including the potential for CO₂ sequestration in soil.
- -
- Impact on the availability of nutrients for plants.
- -
- Effectiveness of EM under abiotic stress conditions (drought, extreme temperatures).
- -
- Adaptation of technology to diverse environmental conditions.
- -
- Optimization of agrotechnical practices:
- -
- Determination of effective doses, formulations (e.g. aqueous suspensions, biofilms) and application frequency.
- -
- Develop molecular research - identify specific EM strains responsible for inhibiting enzymatic browning and the exact mechanisms of DC-CIRHT action.
- -
- Field tests in different soil and climate conditions – verification of effectiveness in real agronomic conditions.
- -
- Integration with other methods – e.g. balanced fertilization or biological plant protection.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Arjmandi, B. , & Ellouze, I. Let food be medicine. Academia Nutrition and Dietetics, 2024, 1(1). [CrossRef]
- Wszelaczyńska E, Pobereżny J, Gościnna K, Szczepanek M, Tomaszewska-Sowa M, Lemańczyk G, Lisiecki K, Trawczyński C, Boguszewska-Mańkowska D, Pietraszko M. Determination of the effect of abiotic stress on the oxidative potential of edible potato tubers. Sci Rep. 2023;13(1): 9999. [CrossRef] [PubMed]
- FAO (2023). Statistical Yearbook. World Food and Agriculture. Food and Agriculture Organization of the United Nations Rome, 2023 ISSN 2225-7373; ISBN 978-92-5-138262-2, pp. 384.
- Wang-Pruski G and J Nowak. Potato after-cooking darkening. Amer J Potato Res 2004, 81: 7-16.
- Sawicka B., Kuś J., Barbaś P. Ciemnienie miąższu bulw ziemniaka w warunkach ekologicznego i integrowanego systemu uprawy. Pamiętnik Puławski 2006, 142, 445-457. (in Polish).
- Grudzińska, M. Czynniki wpływające na ciemnienie miąższu bulw surowych i po ugotowaniu. Ziem. Pol. (In Polish). 2009, 4, 33–36. [Google Scholar]
- Bienia, B. , Sawicka, B., & Krochmal-Marczak, B. The effect of foliar fertilization on the darkening of tuber flesh of selected potato cultivars. Agronomy Science, 2019, 74(4), 61-71. [CrossRef]
- Gunko, S. , Vakuliuk, P., Naumenko, Bober, А., Boroday, V., Nasikovskyi, V., & Muliar, О. The mineral composition of potatoes and its influence on the darkening of tubers pulp. Food science and Technology, 2023, 17(1), 21-28.
- Ali, H. M., El-Gizawy, A. M., El-Bassiouny, R. E., & Saleh, M. A. The role of various amino acids in enzymatic browning process in potato tubers and identifying the browning products. Food Chemistry, 2016, 192, 879-885.
- Wang-Pruski G. Digital Imaging for evaluation of potato after-cooking darkening and its comparison with other methods. Inter J Food Sci and Tech 2006, 41(8):885-891.
- Wang-Pruski, G., Zebarth, B. J., Leclerc, Y., Arsenault, W. J., Botha, E. J., Moorehead, S., & Ronis, D. Effect of soil type and nutrient management on potato after-cooking darkening. American Journal of Potato Research, 2007, 84, 291-299.
- LeRiche, E. L. , & Wang-Pruski, G. Digital imaging for the evaluation of potato after-cooking darkening: correcting the effect of flesh color. International Journal of Food Science and Technology, 2009, 44(12), 2669-2671. [CrossRef]
- Calder, B. L. , Cowles, E. A., Davis-Dentici, K., & Bushway, A. A. The effectiveness of antibrowning dip treatments to reduce after-cooking darkening in potatoes. Journal of Food Science, 2012, 77(10), 342-347. [CrossRef]
- Mystkowska, I. , Baranowska, A., Zarzecka, K., Gugała, M., & Sikorska, A. The effect of biostimulators on the tastiness and darkening of the pulp of raw and cooked potato tubers. Journal of Ecological Engineering, 2018, 19(5), 116–121. [CrossRef]
- Wang-Pruski G, T Astatkie, H De Jong and Y Leclerc. Genetic and environmental interactions affecting potato after-cooking darkening. Acta Hort (ISHS), 2003, 619:45-52.
- Jimenez, M. E. Changes during cooking processes in 6 varieties of Andean potatoes (Solanum tuberosum ssp. Andinum). American Journal of Plant Sciences 2015, 6, 725–736. [Google Scholar] [CrossRef]
- Bordoloi, A. , Kaur, L., & Singh, J. Parenchyma cell microstructure and textural characteristics of raw and cooked potatoes. Food Chemistry, 2012, 133(4), 1092-1100. [CrossRef]
- Pszczółkowski, P.; Krochmal-Marczak, B.; Sawicka, B.; Pszczółkowski, M. The Impact of Effective Microorganisms on Flesh Color and Chemical Composition of Raw Potato Tubers. Appl. Sci. 2021, 11, 8959. [Google Scholar] [CrossRef]
- Lenartowicz, T. Descriptive List of Agricultural Cultivars; COBORU Publishing House: Słupia Wielka, Poland, 2018; (In Polish). ISSN 1641-7003. [Google Scholar]
- Peeten, M.G.H. , Schipper E., Schipper K.J., Baarveld R.H. Netherlands catalogue of potato varieties. Wyd. NIVAP, Den Haag, 2007.
- EUPVP - Common Catalogue Information System. 2024. EUPVP - Common Catalogue. https://ec.europa.eu/food/plant-variety-portal/.
- AlHdadidi N., Hasan H., Pap Z., Ferenc T., Papp O., Drexler D., Ganszky D., and Kappel N. Beneficial Effects of Microbial Inoculation to Improve Organic Potato Production under Irrigated and Non-Irrigated Conditions. International Journal of Agriculture & Biology, 2024. ISSN Print: 1560–8530; ISSN Online: 1814–9596 23–0220/2024/31–1–57–64. [CrossRef]
- Philippot, L. , Raaijmakers, J. M., Lemanceau, P., & van der Putten, W.H. Going back to the roots: the microbial ecology of root-soil interactions. Nature Reviews Microbiology, 2013, 11(12), 789-799.
- Berendsen, R. L. , Pieterse, C. M., & Bakker, P. A. The rhizosphere microbiome is a key component in plant health. Trends in Microbiology, 2012, 20(6), 278-283.
- Piotrowska, A., & Boruszko, D. Potential of Effective Microorganisms in the Aspect of Sustainable Development. Rocznik Ochrona Środowiska, 2024, 26, 106-114. [CrossRef]
- Bashash, M. , Wang-Pruski, G., He, Q. S., & Sun, X. The emulsifying capacity and stability of potato proteins and peptides: A comprehensive review. Comprehensive Reviews in Food Science and Food Safety, 2024, 23(5), e70007. [CrossRef]
- Pszczółkowski P, Sawicka B, Barbaś P, Skiba D, Krochmal-Marczak B. The Use of Effective Microorganisms as a Sustainable Alternative to Improve the Quality of Potatoes in Food Processing. Applied Sciences. 2023; 13(12):7062. [CrossRef]
- Rana, A., & Jhilta, P. Improved practices through biological means for sustainable potato production. In Kaushal, M., Prasad, R. (eds.), Microbial Biotechnology in Crop Protection (pp. 189-207). Singapore: Springer Singapore, 2021. [CrossRef]
- Roztropowicz S., Czerko Z., Głuska A., Goliszewski W., Gruczek T., Lis B., Lutomirska B., Nowacki W. Rykaczewska K., Sowa-Niedziałkowska G., Szutkowska M., Wierzejska-Bujakowska A., Zarzyńska K. 1999. Metodyka obserwacji, pomiarów i pobierania prób w agrotechnicznych doświadczeniach z ziemniakiem. Red. S. Roztropowicz. Wyd. IHAR, Jadwisin. (In Polish).
- Trinh T.K., Glasgow S. On the texture profile analysis test. Conference: Chemeca, Wellington, New Zealand, September 2012, 1-12.
- Luyten J.P., Corveleyn S.J., Blatt J. Chapter 3. The convergence among psychodynamic and cognitive-behavioral theories of depression: A critical overview of empirical research. January 2005, 1-88https://www.researchgate.net/publication/292408464_The_convergence_among_psychodynamic_and_cognitive-behavioral_theories_of_depression_A_critical_overview_of_empirical_research#fullTextFileContent.
- Klosek, V. Crystallographic textures. EPJ Web Conf., 2017, 155, 24 00005. [Google Scholar] [CrossRef]
- Nawrocki, S.; Nawozowe, Z.; Część, I. Liczby Graniczne do Wyceny Zawartości w Glebach Makro-i Mikroelementów; Wyd. IUNG: Puławy, Poland, 1985; p. 38. (in Polish) [Google Scholar]
- Gliński J., Debicki R. Użytkowanie ziemi oraz jakość gleb Polesia Lubelskiego. Acta Agrophysica, 2002, 66: 77-87 (in Polish).
- Skowera, B. , Kopcińska J., Kopeć B. Changes in thermal and precipitation conditions in Poland in 1971-2010. Ann. Warsaw Univ. of Life Sci. - SGGW, Land Reclaim., 2014, 46 (2), 153-162.
- SAS Institute Inc. (2008). SAS/STAT®9.2 User’s Guide. Cary, NC: SAS Institute Inc.
- Rymuza, K. Multi-trait evaluation of value for cultivation and use of early maturing edible potato cultivars registered in Poland. J. Ecol. Eng. 2015; 16(1), 50–56. [CrossRef]
- Field, A.P. Discovering Statistics Using IBM SPSS Statistics. 5th Edition, Sage, Newbury Park, 2018.
- Xu, H. , Wang, X., Deng, F., Wang, H., & Xu, W. Effects of beneficial microbial agents on antioxidant system in plants. Plant Physiology and Biochemistry, 2012, 58, 171–177. [Google Scholar]
- Sawicka, B. , Pszczółkowski P., Noaema A.H., Krochmal-Marczak B., Kiełtyka-Dadasiewicz A. Efektywne Mikroorganizmy w Rolnictwie i Przetwórstwie Spożywczym [In book:] Contemporary research on the state of the environment/Współczesne badania nad stanem środowiska i leczniczym wykorzystaniem roślin. Publisher: University of Life Science in Lublin, 2016: 43-63. (in Polish).
- Baranowska A., Zarzecka K., Gugała M., Mystkowska I. The effect of fertilizer on UGmaxsoil on the presence of Streptomyces scabies on edible potato tubers. J. Ecol. Eng. 2018, 3, 68–73. [CrossRef]
- Mendes, R. , Raaijmakers, J. M., de Hollander, M., van Bruggen, A. H., & Paul, N. D. Functional roles of microbiomes in plant health and disease. Proceedings of the International Symposium on Soil and Plant Microbiomes, 2013, 1-10.
- Faturrahman, L. , Meryandini A., Junior M.Z., Rusmana I. The Role of Agaro lytic Bacteria in Enhancing Physiological Function for Digestive System of Abalone (Haliotis asinine). J Appl. Environ. Biol. Sci. 2015, 5(5), 49–56, ISSN: 2090-4274.
- Lancaster S.H., Haney R.L., Senseman S.A., Hons F.M., Handler J.M. Soil microbial activity is affected by Roundup Weather Max and pesticides applied to cotton (Gossypium hirsutum). J. Agric. Food Chem. 2006, 54, 7221–7226.
- Han L., Ganjurjav H., Hu G., Wu Y., Wang X., Yan Y. et al. Warming enhances the effects of nitrogen addition on fungal but not on bacterial diversity in an alpine meadow, Basic and Applied Ecology, 2025, 84: 110-120, ISSN 1439-1791. [CrossRef]
- Lee, K. H. Effect of organic amendments and EM on the growth and yield of crops and on soil properties. In: Proceedings of the Second International Conference on Kyusei Nature Farming. US Department of Agriculture, Washington, DC, USA 1994, p. 142-147.
- Xu, H. , Wang, X., Deng, F., Wang, H., & Xu, W. Effects of beneficial microbial agents on antioxidant system in plants. Plant Physiology and Biochemistry, 2012, 58, 171–177. [Google Scholar]
- Xu, H. , Wang, X., Deng, F., Wang, H., & Xu, W. Effects of beneficial microbial agents on antioxidant system in plants. Plant Physiology and Biochemistry, 2012, 58, 171–177. [Google Scholar]
- Hussain, R., Sanabil, Huali, X. et al. Correction: Discoloration of Raw and Cooked Potatoes: Fundamentals of Nature, Mechanisms, Causes, Measurements, and Controls. Am. J. Potato Res. 99, 307 (2022. [CrossRef]
- Krochmal-Marczak, B. Krochmal-Marczak, B., & Sawicka, B. Darking tuber flesh sweet potato (Ipomoea batatas L. [Lam.]) [Ciemniejący miąższ bulw słodkiego ziemniaka (Ipomoea batatas L. [Lam.]) w Polsce w południowo-wschodniej]. Polish in a South-Eastern. Rural Development 2013, 153-155.
- LeRiche, E. A Study of Potato (Solanum tuberosum L.) Tuber Element Distribution and Its Implication for After-cooking Darkening. 2007, Dalhousie University and Nova Scotia Agricultural College.
- LeRiche, E. L. , & Wang-Pruski, G. Digital imaging for the evaluation of potato after-cooking darkening: correcting the effect of flesh color. International Journal of Food Science and Technology, 2009, 44(12), 2669-2671. [CrossRef]
- Ouyang N, Guo L, Hong Ch, Zhang H, Liu Y, Guo Y, Wang Y, Ma H. Effects of delayed tuber cutting after catalytic infrared heat treatment on browning of fresh-cut potatoes and its potential mechanisms, Food Chemistry, 2025, 480: 143834, ISSN 0308-8146. [CrossRef]
- Dourado C., Pinto C., Barba FJ., Jose M. Delgadillo L.I., Saraiva JA. Innovative non-thermal technologies affecting potato tuber and fried potato quality, Trends in Food Science & Technology, 88, 2019, 274-289, ISSN 0924-2244.
- Zavadska, O. , Iliuk, N., Ivanytska, A., Semenenko, S., & Mykhailyn, V. Suitability of potato tubers of different varieties for processing. Scientific Reports of the National University of Life and Environmental Sciences of Ukraine, 2024, 20(4), 20-30. [CrossRef]
- Gao H, Pei X, Song X, Wang S, Yang Z, Zhu J, Lin Q, Zhu Q and Yang X/ Application and development of CRISPR technology in the secondary metabolic pathway of the active ingredients of phytopharmaceuticals. Front. Plant Sci. 2025, 15:1477894. [CrossRef]
- Kołodziejczyk, M. Effectiveness of nitrogen fertilization and application of microbial preparations in potato cultivation. Turkish Journal of Agriculture and Forestry, 2014, 38: 299–310. [CrossRef]
- Li, G. , Wang, X., Zhu, H., Li, G., Du, J., Song, X., & Erihemu. Use of different food additives to control browning in fresh-cut potatoes. Food Science & Nutrition, 2023, 11(12), 7967-7973. [CrossRef]
- Anonimous. Food Chemistry (2024) – PPO inhibitor review, https://www.sciencedirect.com/journal/food-chemistry/vol/446/suppl/C.
- Krochmal-Marczak, B. , & Sawicka, B. Darkening tuber Flesch sweet potato (Ipomoea batatas L. [Lam.]), Polish in a South-Eastern. Rural Development 2013, 153-155.
- Sawicka B.H., Pszczółkowski P., Messoudi M., Skiba D., Barbaś P., Krochmal-Marczak B., Noaema A.H. Genotype, Environment and Heavy Metals: Variability of The Content of Elements in 40 Potato Varieties from Central-Eastern Poland. Chemistry and Biodiversity 2025. ISSN 1612-1872. [CrossRef]
- Krochmal-Marczak, B. , Sawicka, B., & Kiełtyka-Dadasiewicz, A. Culinary properties of selected potato cultivars in relation to storage. Pol. J. of Commodity Sci, 2017, 4(53), 72-81.
- Ouyang N, Guo L, Hong Ch, Zhang H, Liu Y, Guo Y, Wang Y, Ma H. Effects of delayed tuber cutting after catalytic infrared heat treatment on browning of fresh-cut potatoes and its potential mechanisms, Food Chemistry, 2025, 480: 143834, ISSN 0308-8146. [CrossRef]
- Li, Y., Huang, X., Yang, Y., Mulati, A., Hong, J., Wang, J. The Effects of Cold-Plasma Technology on the Quality Properties of Fresh-Cut Produce: A Review. Foods, 2025, 14, 149. [CrossRef]
- Lupwayi, N.Z. , Soon, Y.K., & Clayton, G.W. Meta-analysis of field experiments on effective microorganisms (EM) effects on crop yields. Canadian Journal of Soil Science, 2021, 101(4), 633-644.
- Chen Y, Gustafsson J, Tafur Rangel, Anton M, Domenzain I, Kittikunapong C, Li F, Yuan L, Nielsen J, Kerkhoven EJ. Reconstruction, simulation and analysis of enzyme-constrained metabolic models using GECKO Toolbox 3.0. Nat Protoc, 2024, 19(3):629-667.
- Backer, R. , Saldanha, R., De Vleesschauwer, D., Höfte, M., & Goethals, P. The rhizosphere microbiota is a key player in sustainable plant production. Reviews in Environmental Science and Biotechnology, 2018, 17(2), 263-294.
- Compant, S. , Clément, C., & Sessitsch, A. Plant growth-promoting bacteria in the rhizo-and endosphere of plants: main determinants of their effects and future perspectives. Journal of Applied Microbiology, 2019, 127(4), 930-949.



| Years | The content of available forms (mg.100 g-1 in dry mass of soil) | pH (1M KCl) |
||
| P2O5 | K2O | Mg | ||
| 2017 | 20.2 | 118 | 44 | 5.8 |
| 2018 | 21.4 | 12.6 | 82 | 62 |
| 2019 | 16,0 | 134 | 73 | 61 |
| Mean | 192 | 126 | 66 | 60 |
| Year | Month | Monthly rainfall (mm) |
Average monthly air temperature (°C) | Coefficient hydrothermal* |
| 2017 | April | 17.2 | 9.1 | 0.6 |
| May | 93.0 | 14.9 | 2.1 | |
| June | 63.7 | 18.3 | 1.1 | |
| July | 63.2 | 22.0 | 0.9 | |
| August | 141.0 | 20.1 | 2.2 | |
| September | 77.4 | 12.1 | 2.1 | |
| Total | 4555 | |||
| 2018 | April | 39.8 | 9.5 | 13 |
| May | 46.3 | 14.1 | 1.1 | |
| June | 117.3 | 18.5 | 2.2 | |
| July | 169.6 | 18.6 | 2.9 | |
| August | 429 | 18.0 | 0.9 | |
| September | 90 | 14.4 | 0.2 | |
| Total | 424.9 | |||
| 2019 | April | 30.1 | 9.3 | 1.1 |
| May | 38.0 | 15.1 | 0.9 | |
| June | 100.7 | 17.4 | 1.9 | |
| July | 53.2 | 219 | 0.7 | |
| August | 700 | 18.6 | 1.1 | |
| September | 34.0 | 14.4 | 0.9 | |
| Total | 326.0 |
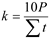 , [35] where: P - the sum of the monthly precipitation in mm. Σt – monthly total air temperature > 0ºC Ranges of values of this index were classified as follows: extremely dry − ≤k 0.4; very dry – 0.7≤k<0.4; dry − 1.0≤k<0.7; rather dry − 1.3≤k<1.0; optimal – 1.6≤k<1.,3; rather humid 2.0≤k<1.6; wet − 2.5≤k<2.,0; very humid −– 30≤k<2,5; extremely humid – 3.0>k2017:.
, [35] where: P - the sum of the monthly precipitation in mm. Σt – monthly total air temperature > 0ºC Ranges of values of this index were classified as follows: extremely dry − ≤k 0.4; very dry – 0.7≤k<0.4; dry − 1.0≤k<0.7; rather dry − 1.3≤k<1.0; optimal – 1.6≤k<1.,3; rather humid 2.0≤k<1.6; wet − 2.5≤k<2.,0; very humid −– 30≤k<2,5; extremely humid – 3.0>k2017:.| Experimental Factors | After 10 minutes | After 1 h | |||||||
| Years | Mean | Years | Mean | ||||||
| 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | ||||
| Traditional | 8.8a | 8.8a | 8.8a | 8.8a | 7.6a | 7.5a | 7.5a | 7.5c | |
| Technologies | EM I | 8.8a | 8.9a | 8.9a | 8.9a | 7.5b | 7.8a | 7.6a | 7.6b |
| EM II | 8.9a | 8.9a | 8.9a | 8.9a | 7.9a | 7.9a | 7.7a | 7.8a | |
| LSD p0.05 | ns**** | ns | 0.3 | 0.1 | |||||
| Krasa | 8.8a | 8.8a | 8.8a | 8.8a | 7.8a | 7.5a | 7.6a | 7.6a | |
| Nora | 8.5a | 8.5a | 8.5a | 8.5b | 7.9a | 8.0a | 7.8a | 7.9a | |
| Nicola | 8.9a | 8.9a | 8.9a | 8.9a | 7.5a | 7.6a | 7.4a | 7.5b | |
| Bellarosa | 9.0a | 9.0a | 9.0a | 9.0a | 7.4a | 7.7a | 7.4a | 7.5b | |
| Vineta | 8.9a | 8.9a | 8.9a | 8.9a | 7.4a | 7.6a | 7.3a | 7.4b | |
| Varieties | Roxana | 8.3a | 8.3a | 8.3a | 8.3b | 7.8a | 7.5a | 7.6a | 7.7a |
| Red Fantasy | 8.9a | 8.9a | 8.9a | 8.9a | 7.6a | 79a | 7.1a | 7.5b | |
| Czapla | 8.9a | 8.9a | 8.9a | 8.9a | 7.8a | 8.2a | 7.6a | 7.9a | |
| Oktan | 8.8a | 8.8a | 8.8a | 8.8a | 6.9a | 6.6a | 7.4a | 7.0c | |
| Ewelina | 9.0a | 9.0a | 90a | 9.0a | 7.8a | 7.6a | 7.7a | 7.7a | |
| Jelly | 8.8a | 8.8a | 8.8a | 8.8a | 7.8a | 7.7a | 7.8a | 7.8a | |
| Zuzanna | 9.0a | 9.0a | 9.0a | 9.0a | 8.1a | 8.0a | 8.0a | 80a | |
| Hinga | 8.9a | 8.9a | 8.9a | 8.9a | 7.9a | 7.7a | 8.1a | 7.9a | |
| Korona | 8.9a | 8.9a | 8.9a | 8.9a | 7.8a | 8.1a | 7.4a | 7.8a | |
| LSD p0.05 | ns | 0.5 | ns | 0.5 | |||||
| Years | 8.8b | 8.9a | 8,9 | 89 | 7.7a | 7.7a | 7.6b | 77 | |
| LSD p0.05 | 0.1 | 0.1 | |||||||
| Experimental Factors | After 10 minutes | After 2 h | ||||||||||
| Years | Mean | Years | Mean | |||||||||
| 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | |||||||
| Traditional* | 8.8a | 8.9a | 8.7a | 8.8a | 7.9a | 7.2b | 8.0a | 7.7b | ||||
| Technologies | EM I** | 8.9a | 8.9a | 8.9a | 8.9a | 8.0a | 7.3b | 7.9a | 7.7b | |||
| EM II*** | 8.9a | 8.9a | 8.7a | 8.8a | 8.1a | 7.6a | 8.1a | 7.9a | ||||
| LSD p0.05 | ns**** | ns | 0.6 | 0.2 | ||||||||
| Krasa | 9.0a | 8.9a | 8.9a | 8.9a | 8.1a | 7.0a | 8.8a | 8.0a | ||||
| Nora | 8.6a | 8.9a | 8.8a | 8.8a | 8.2a | 6.8a | 8.3a | 7.8ab | ||||
| Nicola | 8.8a | 8.8a | 8.8a | 8.8a | 8.0a | 6.7a | 7.7a | 7.5b | ||||
| Bellarosa | 8.8a | 8.9a | 8.9a | 8.9a | 8.5a | 7.7a | 8.2a | 81a | ||||
| Vineta | 9.0a | 8.9a | 9.0a | 9.0a | 8.9a | 8.0a | 8.2a | 8.4a | ||||
| Varieties | Roxana | 9.0a | 89a | 8.9a | 8.9a | 8.5a | 7.1a | 7.9a | 7.9ab | |||
| Red Fantasy | 8.9a | 8.9a | 8.9a | 8.9a | 7.1a | 7.0a | 7.3a | 7.1bc | ||||
| Czapla | 8.5a | 8.9a | 8.7a | 8.7a | 7.9a | 7.7a | 8.0a | 7.9ab | ||||
| Oktan | 8.8a | 8.9a | 8.8a | 8.8a | 7.7a | 7.7a | 7.7a | 7.7ab | ||||
| Ewelina | 8.9a | 8.9a | 8.9a | 8.9a | 7.6a | 7.2a | 8.1a | 7.6ab | ||||
| Jelly | 9.0a | 9.0a | 9.0a | 9.0a | 8.0a | 7.8a | 8.3a | 8.0a | ||||
| Zuzanna | 9.0a | 9.0a | 9.0a | 9.0a | 7.8a | 7.8a | 7.7a | 7.8ab | ||||
| Hinga | 8.8a | 8.9a | 8.8a | 8.8a | 7.4a | 7.0a | 7.9a | 7.4b | ||||
| Korona | 8.8a | 8.8a | 8.8a | 8.8a | 7.9a | 7.9a | 7.9a | 7.9ab | ||||
| LSD p0.05 | ns | ns | ns | 0.9 | ||||||||
| Years | 8.8a | 8.9a | 8.8a | 8.8 | 8.0a | 7.4b | 8.0a | 7.8 | ||||
| LSD p0.05 | ns | 0.2 | ||||||||||
| Experimental Factors | After 10 minutes | After 2 h | |||||||
| Years | Mean | Years | Mean | ||||||
| 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | ||||
| Traditional* | 8.5a | 8.7a | 8.6a | 8.6a | 7.6a | 7.2a | 7.4a | 7.4b | |
| Technologies | EM I** | 8.7a | 8.7a | 8.6a | 8.7a | 7.6a | 7.1a | 7.7a | 7,5a |
| EM II*** | 8.7a | 8.7a | 8.9a | 8.8b | 7.8a | 7.5a | 7.6a | 7.6a | |
| LSD p0.05 | ns**** | 0.2 | ns | 0.2 | |||||
| Krasa | 8.9a | 8.8a | 8.5a | 8.8a | 7.9a | 7.0a | 7.7a | 7.5ab | |
| Nora | 8.2a | 8.8a | 8.9a | 8.6a | 7.7a | 6.8a | 7.7a | 7.4ab | |
| Nicola | 8.4a | 8.6a | 8.7a | 8.6a | 7.6a | 6.6a | 7.5a | 7.2b | |
| Bellarosa | 8.5a | 8.8a | 8.8a | 8.7a | 8.0a | 7.5a | 7.4a | 7.7a | |
| Vineta | 9.0a | 8.8a | 8.8a | 8.9a | 8.4a | 7.8a | 7.9a | 8.0a | |
| Varieties | Roxana | 9.0a | 8.7a | 8.4a | 8.7a | 8.2a | 7.7a | 7.6a | 7.8a |
| Red Fantasy | 8.8a | 8.9a | 8.8a | 8.8a | 7.1a | 6.9a | 7.3a | 7.1b | |
| Czapla | 8.6a | 8.8a | 84a | 8.6a | 7.5a | 7.6a | 7.5a | 7.5ab | |
| Oktan | 8.6a | 8.6a | 8.5a | 8.6a | 7.4a | 7.4a | 7.3a | 7.4ab | |
| Ewelina | 8.4a | 8.4a | 8.5a | 8.4a | 7.2a | 6.9a | 7.4a | 7.2b | |
| Jelly | 8.8a | 8.7a | 8.9a | 8.8a | 7.8a | 7.7a | 7.9a | 7.8a | |
| Zuzanna | 8.8a | 8.8a | 8.9a | 8.8a | 7.5a | 7.6a | 7.5a | 7.5a | |
| Hinga | 8.5a | 8.6a | 8.5a | 8.5a | 7.3a | 6.8a | 7.5a | 7.2b | |
| Korona | 8.7a | 87a | 8.8a | 8.7a | 7.5a | 7.6a | 73 | 7.5ab | |
| LSD p0.05 | ns | ns | ns | 0.8 | |||||
| Years | Mean | 8.7a | 8.7a | 8.7a | 8.7 | 7.6a | 7.3b | 7.5a | 7,5 |
| LSD p0.05 | ns | 0,2 | |||||||
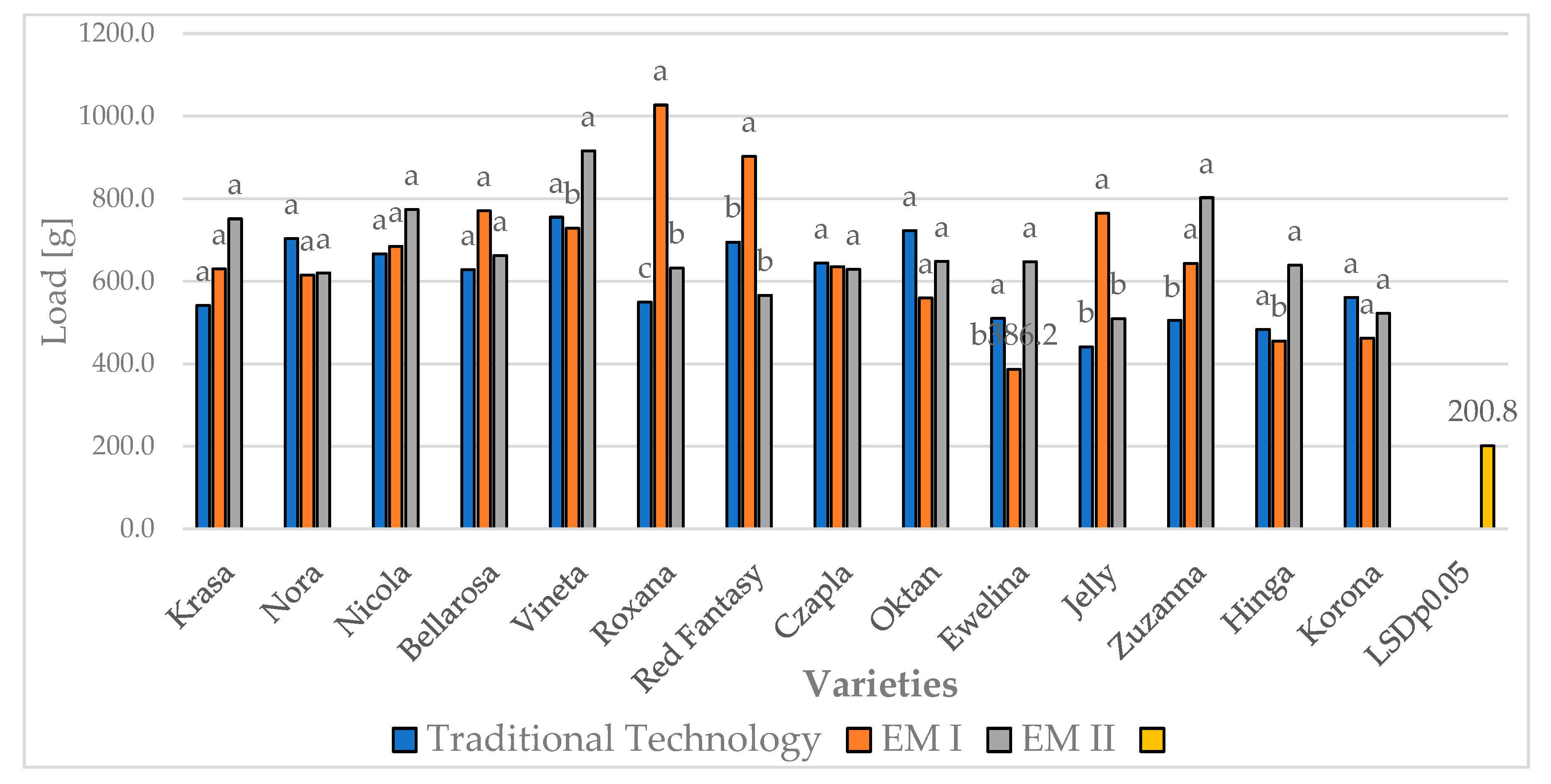
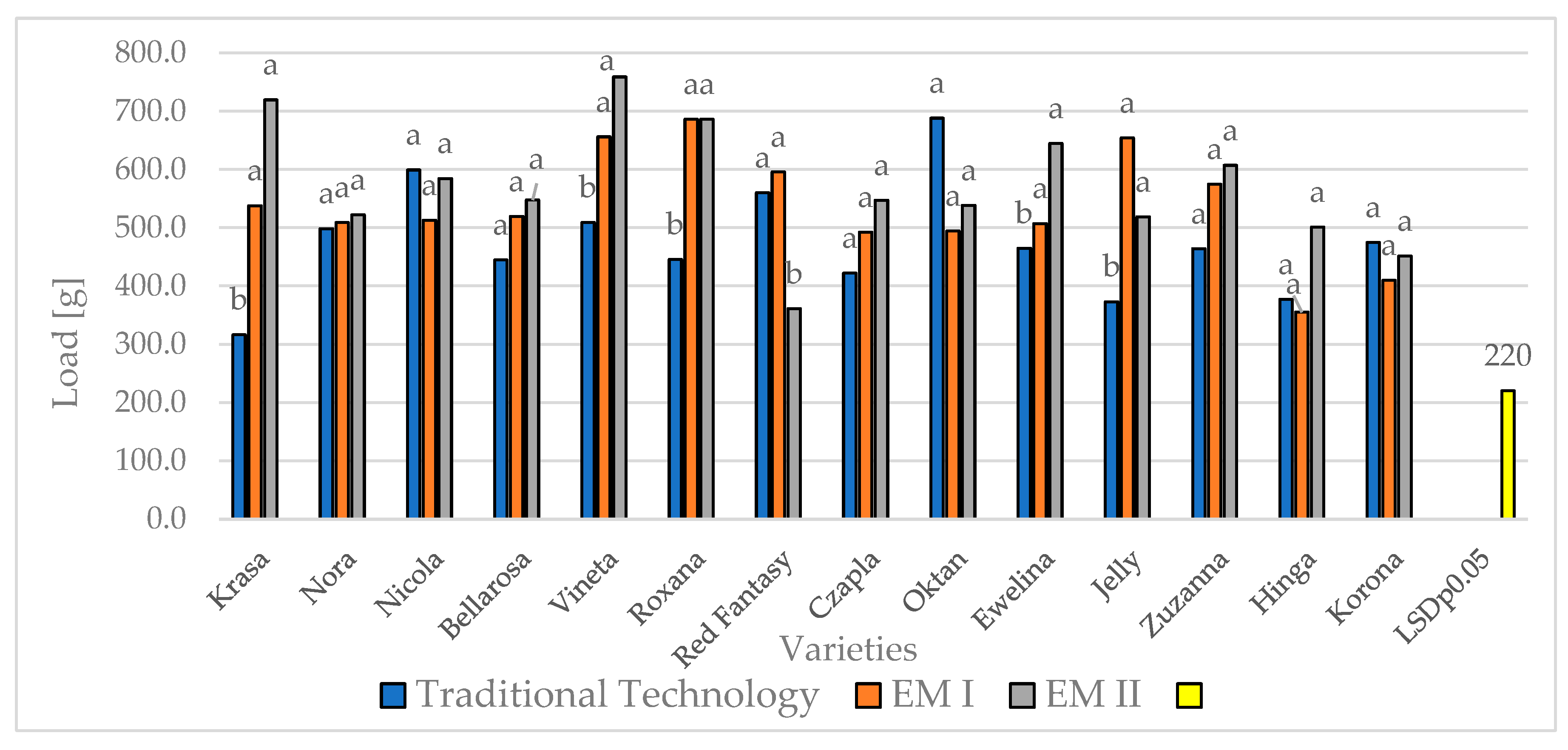
| Experimental Factors | Work [MJ] | Load [g] | Final load [g] | Load height [mm] | |||||||||||||
| Years | Mean | Years | Mean | years | Mean | Years | Mean | ||||||||||
| 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | ||||||
| Technolo gies |
Traditional | 22.2a | 21.7a | 13.1b | 19.0a | 721.6a | 772.7a | 306.9b | 600.4b | 541.8a | 577.4a | 302.7b | 474.0b | 3.1a | 2.8a | 2.8a | 2.9b |
| EM I* | 20.6a | 21.8a | 2.9b | 15.1b | 724,8a | 786,3a | 473,9b | 661,7a | 575.3a | 594.9a | 437.6b | 535.9a | 3.4a | 3.0b | 3.0b | 3.1a | |
| EM II** | 20.9a | 23.1a | 3.2b | 15.8b | 653,5b | 877,0a | 465,9c | 665,4a | 579.9a | 641.2a | 490.5b | 570.5a | 3.3a | 3.0a | 3.0a | 3.1a | |
| LSD p0.05 | 2.4 | 0.8 | 69.9 | 23.2 | 73,6 | 24,5 | 0.4 | 0.1 | |||||||||
| Krasa | 30.0a | 19.6a | 8.6b | 19.4a | 755.4a | 766.2a | 400.8a | 640.b | 607.8a | 488.7a | 476.9a | 524.4b | 3.0a | 2.5a | 2.5a | 2.7b | |
| Nora | 30.3a | 22.0a | 6.3b | 19.5a | 823.9a | 767.9a | 346.4b | 646.1b | 601.3a | 581.7a | 346.4a | 509.8b | 2.9a | 2.4a | 2.4a | 2.6b | |
| Nicola | 28.3a | 26.4a | 10.9b | 21.8a | 822.9a | 928.4a | 372.9b | 708.1a | 587.8a | 768.8a | 339.6a | 565.4a | 4.7a | 3.9a | 3.9a | 4.2a | |
| Bellarosa | 32.4a | 18.1b | 9.4bc | 20.0a | 875.7a | 660.6a | 524.7b | 687.0a | 595.2a | 457.1a | 459.3a | 503.8b | 3.0a | 2.5a | 2.5a | 2.7b | |
| Vineta | 34.2a | 23.7b | 7.6c | 21.8a | 841.4a | 911.1a | 646.4a | 799.6a | 680.9a | 615.8a | 626.7a | 641.1a | 2.4a | 2.0a | 2.0a | 2.1c | |
| Varieties | Roxana | 40.3a | 19.5bc | 6.3c | 22.0a | 902.8a | 772.4a | 532.9b | 736.0a | 821.1a | 584.4a | 411.9a | 605.8a | 3.6a | 3.0a | 3.0a | 3.2b |
| Red Fantasy | 15.1a | 25.2a | 5.7 b | 15.3b | 721.0a | 987.5a | 454.5b | 721.0a | 505.4 | 689.4a | 321.4a | 505.4b | 3.6a | 2.9a | 2.9a | 3.2b | |
| Czapla | 13.2b | 24.6a | 4.6c | 14.2b | 636.2a | 982.5a | 289.9b | 636.2b | 487.1a | 702.9a | 271.3a | 487.1b | 3.0a | 3.0a | 3.0a | 3.0b | |
| Oktan | 12.2a | 21.8a | 5.0b | 13.0b | 643.4a | 881.6a | 405.2b | 643.4b | 573.6a | 673.6a | 473.5a | 573.6a | 4.1a | 3.4a | 3.4a | 3.6a | |
| Ewelina | 11.4a | 19.7a | 5.5b | 12.2b | 514.6a | 651.6a | 377.6a | 514.6c | 538.8a | 588.5a | 489.1a | 538.8a | 3.5a | 3.5a | 3.5a | 3.5a | |
| Jelly | 13.1a | 23.9a | 4.9b | 14.0b | 571.5a | 788.1a | 354.9b | 571.5c | 515.2a | 665.9a | 364.5a | 515.2b | 3.0a | 3.0a | 3.0a | 3.0b | |
| Zuzanna | 13.5b | 24.2a | 4.4c | 14.0b | 650.4a | 841.3a | 459.0b | 650.4b | 548.4a | 658.6a | 438.3a | 548.4a | 3.1a | 3.1a | 3.1a | 3.1b | |
| Hinga | 11.5a | 21.3a | 4.7b | 12.5c | 525.6b | 784.4a | 266.9c | 525.6c | 411.2a | 543.7a | 278.6a | 411.2c | 2.7a | 2.7a | 2.7a | 2.7b | |
| Korona | 12.1a | 21.0a | 5.9b | 13.0c | 514.9a | 644.4a | 385.4a | 514.9c | 445.1a | 444.1a | 446.1a | 445.1b | 3.0a | 3.0a | 3.0a | 3.0b | |
| LSD p0.05 | 11.0 | 3.7 | 326.1 | 108.7 | ns | 114.4 | ns | 0.7 | |||||||||
| Years | 21.3a | 22.2a | 6.4b | 16.6 | 700.0b | 812.0a | 415.6c | 642.5 | 565.6b | 604.5a | 410.3c | 526.8 | 3.3a | 2.9a | 2.9a | 3.0 | |
| LSD p0.05 | 0.8 | 23.3 | 24.5 | ns | |||||||||||||
| Specification | y1 | x1 | x2 | x3 | x4 | x5 | x6 | x7 | x8 | x9 |
| Mean | 8.85 | 7.65 | 8.84 | 8.68 | 7.79 | 7.49 | 16.57 | 642.51 | 526.80 | 3.05 |
| Median | 8.90 | 7.70 | 9.00 | 8.80 | 7.83 | 7.45 | 15.22 | 642.87 | 530.70 | 2.80 |
| Standard deviation | 0.17 | 0.48 | 0.28 | 0.34 | 0.61 | 0.54 | 11.00 | 273.94 | 193.21 | 1.04 |
| Kurtosis | 5.65 | 1.37 | 8.44 | 1.62 | -0.31 | -0.23 | 0.02 | -0.83 | -0.12 | -0.06 |
| Skewness | -2.01 | -0.68 | -2.71 | -1.32 | -0.10 | -0.13 | 0.56 | 0.05 | 0.16 | 0.88 |
| Range | 1.00 | 2.90 | 1.60 | 1.60 | 2.70 | 2.80 | 58.11 | 1104.20 | 1101.80 | 4.60 |
| Minimum | 8.00 | 5.90 | 7.40 | 7.40 | 6.30 | 6.00 | 0.51 | 96.50 | 96.50 | 1.40 |
| Maximum | 9.00 | 8.80 | 9.00 | 9.00 | 9.00 | 8.80 | 58.62 | 1200.70 | 1198.30 | 6.00 |
| V* | 1.97 | 6.29 | 3.14 | 3.93 | 7.81 | 7.15 | 66.40 | 42.64 | 36.68 | 34.22 |
| Specification | y1 | x1 | x2 | x3 | x4 | x5 | x6 | x7 | x8 | x9 |
| y1 | 1.00 | |||||||||
| x1 | 0.20 | 1.00 | ||||||||
| x2 | 0.12 | 0.07 | 1.00 | |||||||
| x3 | 0.04 | 0.17 | 0.59 | 1.00 | ||||||
| x4 | -0.11 | 0.02 | 0.13 | 0.20 | 1.00 | |||||
| x5 | -0.10 | 0.09 | 0.27 | 0.39 | 0.80 | 1.00 | ||||
| x6 | -0.20 | 0.05 | 0.21 | 0.12 | 0.00 | 0.11 | 1.00 | |||
| x7 | -0.19 | -0.03 | 0.11 | -0.03 | -0.20 | -0.09 | 0.65 | 1.00 | ||
| x8 | -0.18 | -0.14 | 0.12 | -0.06 | -0.09 | -0.08 | 0.53 | 0.78 | 1.00 | |
| x9 | 0.01 | -0.18 | -0.16 | -0.28 | -0.18 | -0.20 | 0.08 | -0.02 | 0.14 | 1.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




