Submitted:
08 April 2025
Posted:
11 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. L. capitata: Polyphenol Profile and Antioxidant Activity
2.1. Extraction Methods
2.2. Polyphenol Content
3. L. cuneata: Polyphenol Profile and Antioxidant Activity
3.1. Extraction and Fractionation
3.2. Polyphenol Content
4. L. bicolor: Polyphenol Profile and Antioxidant Activity
4.1. Extraction Techniques
4.2. Polyphenol Composition
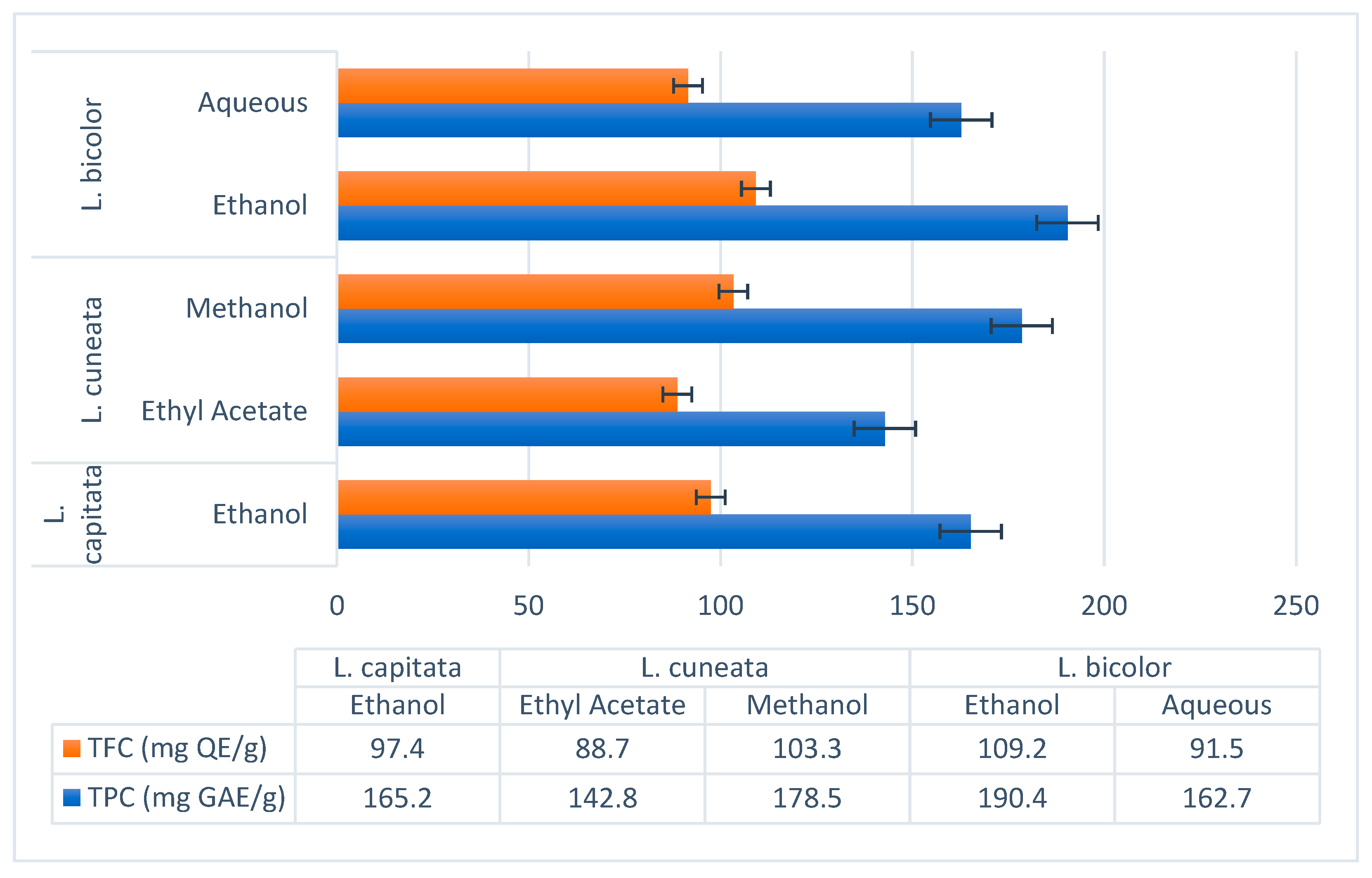
- Ethanol Extract (Ren et al., 2023): TPC: 190.4 mg GAE/g, TFC: 109.2 mg QE/g. Major polyphenols included rutin (22.1 mg/g), hyperoside (19.3 mg/g), and kaempferol-3-O-rutinoside (14.6 mg/g) [55].
- Aqueous Extract (Tarbeeva et al., 2019): TPC: 162.7 mg GAE/g, TFC: 91.5 mg QE/g. Key compounds included apigenin (10.8 mg/g) and luteolin (8.4 mg/g) [54].
5. Comparative Analysis of L. Species
5.1. Antioxidant Activity
5.2. Anti-Inflammatory Effects
5.3. Antidiabetic Activity (α-Glucosidase Inhibition)
5.4. Anticancer and Neuroprotective Effects
6. Literature Review Process
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- The Plant Family Fabaceae: Biology and Physiological Responses to Environmental Stresses; Hasanuzzaman, M., Araújo, S., Gill, S.S., Eds.; Springer: Singapore, 2020; ISBN 978-981-15-4751-5. [Google Scholar]
- Rüping, B.; Ernst, A.M.; Jekat, S.B.; Nordzieke, S.; Reineke, A.R.; Müller, B.; Bornberg-Bauer, E.; Prüfer, D.; Noll, G.A. Molecular and Phylogenetic Characterization of the Sieve Element Occlusion Gene Family in Fabaceae and Non-Fabaceaeplants. BMC Plant Biol 2010, 10, 219. [Google Scholar] [CrossRef] [PubMed]
- Shavanov, M.V. The Role of Food Crops within the Poaceae and Fabaceae Families as Nutritional Plants. IOP Conf. Ser.: Earth Environ. Sci. 2021, 624, 012111. [Google Scholar] [CrossRef]
- Ciotir, C.; Applequist, W.; Crews, T.E.; Cristea, N.; DeHaan, L.R.; Frawley, E.; Herron, S.; Magill, R.; Miller, J.; Roskov, Y.; et al. Building a Botanical Foundation for Perennial Agriculture: Global Inventory of Wild, Perennial Herbaceous Fabaceae Species. PLANTS, PEOPLE, PLANET 2019, 1, 375–386. [Google Scholar] [CrossRef]
- Maroyi, A. Medicinal Uses of the Fabaceae Family in Zimbabwe: A Review. Plants 2023, 12, 1255. [Google Scholar] [CrossRef]
- Asfaw, M.M.; Abebe, F.B. Traditional Medicinal Plant Species Belonging to Fabaceae Family in Ethiopia: A Systematic Review. International Journal of Plant Biology 2021, 12, 8473. [Google Scholar] [CrossRef]
- Souleymane, F.; Charlemagne, G.; Moussa, O.; Eloi, P.; Hc, N.R.; Baptiste, N.J.; Pierre, G.I.; Jacques, S. DPPH Radical Scavenging and Lipoxygenase Inhibitory Effects in Extracts from Erythrina Senegalensis (Fabaceae) DC. AJPP 2016, 10, 185–191. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Drouet, S.; Lorenzo, J.M.; Hano, C. Characterization of Bioactive Phenolics and Antioxidant Capacity of Edible Bean Extracts of 50 Fabaceae Populations Grown in Thailand. Foods 2021, 10, 3118. [Google Scholar] [CrossRef]
- Xu, Z.; Deng, M. Fabaceae or Leguminosae. In Identification and Control of Common Weeds: Volume 2; Xu, Z., Deng, M., Eds.; Springer Netherlands: Dordrecht, 2017; pp. 547–615. ISBN 978-94-024-1157-7. [Google Scholar]
- Das, S.; Sharangi, A.B.; Egbuna, C.; Jeevanandam, J.; Ezzat, S.M.; Adetunji, C.O.; Tijjani, H.; Olisah, M.C.; Patrick-Iwuanyanwu, K.C.; Adetunji, J.B.; et al. Health Benefits of Isoflavones Found Exclusively of Plants of the Fabaceae Family. In Functional Foods and Nutraceuticals: Bioactive Components, Formulations and Innovations; Egbuna, C., Dable Tupas, G., Eds.; Springer International Publishing: Cham, 2020; pp. 473–508. ISBN 978-3-030-42319-3. [Google Scholar]
- Raj, S.P.; Solomon, P.R.; Thangaraj, B. Fabaceae. In Biodiesel from Flowering Plants; Raj, S.P., Solomon, P.R., Thangaraj, B., Eds.; Springer: Singapore, 2022; pp. 291–363. ISBN 978-981-16-4775-8. [Google Scholar]
- Schweingruber, F.H.; Dvorský, M.; Börner, A.; Doležal, J. Fabaceae. In Atlas of Stem Anatomy of Arctic and Alpine Plants Around the Globe; Schweingruber, F.H., Dvorský, M., Börner, A., Doležal, J., Eds.; Springer International Publishing: Cham, 2020; pp. 194–206. ISBN 978-3-030-53976-4. [Google Scholar]
- Zou, P. Traditional Chinese Medicine, Food Therapy, and Hypertension Control: A Narrative Review of Chinese Literature. Am. J. Chin. Med. 2016, 44, 1579–1594. [Google Scholar] [CrossRef]
- Lou, D.; Li, Y.; Yan, G.; Bu, J.; Wang, H. Soy Consumption with Risk of Coronary Heart Disease and Stroke: A Meta-Analysis of Observational Studies. Neuroepidemiology 2016, 46, 242–252. [Google Scholar] [CrossRef]
- Li, L.; Lv, Y.; Xu, L.; Zheng, Q. Quantitative Efficacy of Soy Isoflavones on Menopausal Hot Flashes. British Journal of Clinical Pharmacology 2015, 79, 593–604. [Google Scholar] [CrossRef]
- Xuan, X.; Zhang, J.; Fan, J.; Zhang, S. Research Progress of Traditional Chinese Medicine (TCM) in Targeting Inflammation and Lipid Metabolism Disorder for Arteriosclerosis Intervention: A Review. Medicine 2023, 102, e33748. [Google Scholar] [CrossRef] [PubMed]
- Singla, N.; Gupta, G.; Kulshrestha, R.; Sharma, K.; Bhat, A.A.; Mishra, R.; Patel, N.; Thapa, R.; Ali, H.; Mishra, A.; et al. Daidzein in Traditional Chinese Medicine: A Deep Dive into Its Ethnomedicinal and Therapeutic Applications. Pharmacological Research - Modern Chinese Medicine 2024, 12, 100460. [Google Scholar] [CrossRef]
- Ren, Y.; Qiao, W.; Fu, D.; Han, Z.; Liu, W.; Ye, W.; Liu, Z. Traditional Chinese Medicine Protects against Cytokine Production as the Potential Immunosuppressive Agents in Atherosclerosis. Journal of Immunology Research 2017, 2017, 7424307. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wang, Y.; Liu, Y.; Ruan, R. Bioactive Peptides Derived from Traditional Chinese Medicine and Traditional Chinese Food: A Review. Food Research International 2016, 89, 63–73. [Google Scholar] [CrossRef]
- Kolodziejczyk-Czepas, J. Trifolium Species-Derived Substances and Extracts—Biological Activity and Prospects for Medicinal Applications. Journal of Ethnopharmacology 2012, 143, 14–23. [Google Scholar] [CrossRef]
- Kolodziejczyk-Czepas, J. Trifolium Species – the Latest Findings on Chemical Profile, Ethnomedicinal Use and Pharmacological Properties. Journal of Pharmacy and Pharmacology 2016, 68, 845–861. [Google Scholar] [CrossRef]
- Sabudak, T.; Guler, N. Trifolium L. – A Review on Its Phytochemical and Pharmacological Profile. Phytotherapy Research 2009, 23, 439–446. [Google Scholar] [CrossRef]
- Çölgeçen, H.; Koca, U.; Büyükkartal, H.N. Use of Red Clover (Trifolium Pratense L.) Seeds in Human Therapeutics. In Nuts and Seeds in Health and Disease Prevention (Second Edition); Preedy, V.R., Watson, R.R., Eds.; Academic Press, 2020; pp. 421–427 ISBN 978-0-12-818553-7.
- Kubasova, E.D.; Дмитриевна, К.Е.; Kubasova, E.D.; Krylov, I.A.; Альбертoвич, К.И.; Krylov, I.A.; Kubasov, R.V.; Виктoрoвич, К.Р.; Kubasov, R.V.; Sukhanov, A.E.; et al. Pharmaceutical potential of red clover ( Trifolium pratense L. ). Ekologiya cheloveka (Human Ecology) 2024, 31, 179–190. [Google Scholar] [CrossRef]
- Costin, A.D.; Muntean, L.; Butaș, A.A.; Mariș, R. Medicinal and Therapeutic Uses of Red Clover (Trifolium Pratense L.) - Review. Hop and Medicinal Plants 2021, 29, 73–81. [Google Scholar] [CrossRef]
- Kaurinovic, B.; Popovic, M.; Vlaisavljevic, S.; Schwartsova, H.; Vojinovic-Miloradov, M. Antioxidant Profile of Trifolium Pratense L. Molecules 2012, 17, 11156–11172. [Google Scholar] [CrossRef]
- Nemoto, T.; Ohashi, H.; Itoh, T. A New Species of Lespedeza (Leguminosae) from China and Japan. 2007, 82, 222–231. [Google Scholar]
- Ohashi, H.; Nemoto, T. A New System of Lespedeza (Leguminosae Tribe Desmodieae). Journal of Japanese Botany 2014, 89, 1–11. [Google Scholar]
- Ohashi, H.; Nemoto, T.; Ohashi, K. A Revision of Lespedeza Subgenus Lespedeza (Leguminosae) of China. Journal of Japanese Botany 2009, 84, 143–166. [Google Scholar]
- Kang, H.-K.; Yi, J.-Y.; Song, H.-S. Germination Characteristics and Maturity by Production Time of Chamaecrista nomame, Lespedeza cuneata and Lespedeza bicolor Seed in Fabaceae Plant. Korean Journal of Plant Resources 2014, 27, 359–364. [Google Scholar] [CrossRef]
- Baek, J.; Lee, D.; Lee, T.K.; Song, J.H.; Lee, J.S.; Lee, S.; Yoo, S.-W.; Kang, K.S.; Moon, E.; Lee, S.; et al. (−)-9′-O-(α-l-Rhamnopyranosyl)Lyoniresinol from Lespedeza Cuneata Suppresses Ovarian Cancer Cell Proliferation through Induction of Apoptosis. Bioorganic & Medicinal Chemistry Letters 2018, 28, 122–128. [Google Scholar] [CrossRef]
- Wang, W.; Patra, A.K.; Puchala, R.; Ribeiro, L.; Gipson, T.A.; Goetsch, A.L. Effects of Dietary Inclusion of Sericea Lespedeza Hay on Feed Intake, Digestion, Nutrient Utilization, Growth Performance, and Ruminal Fermentation and Methane Emission of Alpine Doelings and Katahdin Ewe Lambs. Animals 2022, 12, 2064. [Google Scholar] [CrossRef]
- Wang, W.; Patra, A.K.; Puchala, R.; Ribeiro, L.; Gipson, T.A.; Goetsch, A.L. Effects of Dietary Inclusion of Tannin-Rich Sericea Lespedeza Hay on Relationships among Linear Body Measurements, Body Condition Score, Body Mass Indexes, and Performance of Growing Alpine Doelings and Katahdin Ewe Lambs. Animals 2022, 12, 3183. [Google Scholar] [CrossRef]
- Lee, J.H.; Parveen, A.; Do, M.H.; Lim, Y.; Shim, S.H.; Kim, S.Y. Lespedeza Cuneata Protects the Endothelial Dysfunction via eNOS Phosphorylation of PI3K/Akt Signaling Pathway in HUVECs. Phytomedicine 2018, 48, 1–9. [Google Scholar] [CrossRef]
- Kim, S.M.; Kang, K.; Jho, E.H.; Jung, Y.-J.; Nho, C.W.; Um, B.-H.; Pan, C.-H. Hepatoprotective Effect of Flavonoid Glycosides from Lespedeza Cuneata against Oxidative Stress Induced by Tert-Butyl Hyperoxide. Phytotherapy Research 2011, 25, 1011–1017. [Google Scholar] [CrossRef]
- Ha, S.J.; Lee, J.; Song, K.-M.; Kim, Y.H.; Lee, N.H.; Kim, Y.-E.; Jung, S.K. Ultrasonicated Lespedeza Cuneata Extract Prevents TNF-α-Induced Early Atherosclerosis in Vitro and in Vivo. Food Funct. 2018, 9, 2090–2101. [Google Scholar] [CrossRef]
- Lungu, I.I. CATECHIN-ZINC-COMPLEX: SYNTHESIS, CHARACTERIZATION AND BIOLOGICAL ACTIVITY ASSESSMENT. FARMACIA 2023, 71, 755–763. [Google Scholar] [CrossRef]
- Springer, T.L.; McGraw, R.L.; Aiken, G.E. Variation of Condensed Tannins in Roundhead Lespedeza Germplasm. Crop Science 2002, 42, 2157–2160. [Google Scholar] [CrossRef]
- Kronberg, S.L.; Zeller, W.E.; Waghorn, G.C.; Grabber, J.H.; Terrill, T.H.; Liebig, M.A. Effects of Feeding Lespedeza Cuneata Pellets with Medicago Sativa Hay to Sheep: Nutritional Impact, Characterization and Degradation of Condensed Tannin during Digestion. Animal Feed Science and Technology 2018, 245, 41–47. [Google Scholar] [CrossRef]
- Terrill, T.H.; Windham, W.R.; Evans, J.J.; Hoveland, C.S. Condensed Tannin Concentraton in Sericea Lespedeza as Influenced by Preservation Method. Crop Science 1990, 30, cropsci1990.0011183X003000010047x. [Google Scholar] [CrossRef]
- Muir, J.P.; Terrill, T.H.; Mosjidis, J.A.; Luginbuhl, J.-M.; Miller, J.E.; Burke, J.M.; Coleman, S.W. Season Progression, Ontogenesis, and Environment Affect Lespedeza Cuneata Herbage Condensed Tannin, Fiber, and Crude Protein Concentrations. Crop Science 2017, 57, 515–524. [Google Scholar] [CrossRef]
- Obistioiu, D.; Cocan, I.; Tîrziu, E.; Herman, V.; Negrea, M.; Cucerzan, A.; Neacsu, A.-G.; Cozma, A.L.; Nichita, I.; Hulea, A.; et al. Phytochemical Profile and Microbiological Activity of Some Plants Belonging to the Fabaceae Family. Antibiotics 2021, 10, 662. [Google Scholar] [CrossRef]
- Šibul, F.; Orčić, D.; Vasić, M.; Anačkov, G.; Nađpal, J.; Savić, A.; Mimica-Dukić, N. Phenolic Profile, Antioxidant and Anti-Inflammatory Potential of Herb and Root Extracts of Seven Selected Legumes. Industrial Crops and Products 2016, 83, 641–653. [Google Scholar] [CrossRef]
- Gulewicz, P.; Martínez-Villaluenga, C.; Kasprowicz-Potocka, M.; Frías, J. Non-Nutritive Compounds in Fabaceae Family Seeds and the Improvement of Their Nutritional Quality by Traditional Processing - A Review. 2014. [Google Scholar] [CrossRef]
- Alam, T.; Najam, L. Faba-Bean Antioxidant and Bioactive Composition: Biochemistry and Functionality. In Faba Bean: Chemistry, Properties and Functionality. Punia Bangar, S., Bala Dhull, S., Eds.; Springer International Publishing: Cham, 2022; pp. 123–162. ISBN 978-3-031-14587-2. [Google Scholar]
- Sharma, A.; Kaur, R.; Katnoria, J.K.; Kaur, R.; Nagpal, A.K. Family Fabaceae: A Boon for Cancer Therapy. In Biotechnology and Production of Anti-Cancer Compounds; Malik, S., Ed.; Springer International Publishing: Cham, 2017; pp. 157–175. ISBN 978-3-319-53880-8. [Google Scholar]
- Ebada, S.S.; Ayoub, N.A.; Singab, A.N.B.; Al-Azizi, M.M. PHCOG MAG.: Research Article Phytophenolics from Peltophorum Africanum Sond. (Fabaceae) with Promising Hepatoprotective Activity. Pharmacognosy Magazine 4.
- Pereira, D.L.; Cunha, A.P.S.D.; Cardoso, C.R.P.; Rocha, C.Q.D.; Vilegas, W.; Sinhorin, A.P.; Sinhorin, V.D.G. Antioxidant and Hepatoprotective Effects of Ethanolic and Ethyl Acetate Stem Bark Extracts of Copaifera Multijuga (Fabaceae) in Mice. Acta Amaz. 2018, 48, 347–357. [Google Scholar] [CrossRef]
- Donfack, J.H.; Nico, F.N.; Ngameni, B.; Tchana, A.; Chuisseu, D.; Finzi, P.V.; Ngadjui, B.T.; Moundipa, P.F. Activities of Diprenylated Isoflavonoids From. 2008.
- Obogwu, M.B.; Akindele, A.J.; Adeyemi, O.O. Hepatoprotective and in Vivo Antioxidant Activities of the Hydroethanolic Leaf Extract of Mucuna Pruriens (Fabaceae) in Antitubercular Drugs and Alcohol Models. Chinese Journal of Natural Medicines 2014, 12, 273–283. [Google Scholar] [CrossRef]
- Chitiala, R.-D.; Burlec, A.F.; Nistor, A.; Caba, I.C.; Mircea, C.; Hancianu, M.; Cioanca, O. CHEMICAL ASSESSMENT AND BIOLOGIC POTENTIAL OF A SPECIAL LESPEDEZA CAPITATA EXTRACT. The Medical-Surgical Journal 2023, 127, 474–479. [Google Scholar] [CrossRef]
- Kim, H.Y.; Ko, J.Y.; Song, S.B.; Kim, J.I.; Seo, H.I.; Lee, J.S.; Kwak, D.Y.; Jung, T.W.; Kim, K.Y.; Oh, I.S.; et al. Antioxidant and α-Glucosidase Inhibition Activities of Solvent Fractions from Methanolic Extract of Sericea Lespedeza (Lespedeza cuneata G. Don). Journal of the Korean Society of Food Science and Nutrition 2012, 41, 1508–1514. [Google Scholar] [CrossRef]
- Mariadoss, A.V.A.; Park, S.; Saravanakumar, K.; Sathiyaseelan, A.; Wang, M.-H. Phytochemical Profiling, in Vitro Antioxidants, and Antidiabetic Efficacy of Ethyl Acetate Fraction of Lespedeza Cuneata on Streptozotocin-Induced Diabetic Rats. Environ Sci Pollut Res 2023, 30, 60976–60993. [Google Scholar] [CrossRef] [PubMed]
- Tarbeeva, D.V.; Fedoreyev, S.A.; Veselova, M.V.; Blagodatski, A.S.; Klimenko, A.M.; Kalinovskiy, A.I.; Grigorchuk, V.P.; Berdyshev, D.V.; Gorovoy, P.G. Cytotoxic Polyphenolic Compounds from Lespedeza Bicolor Stem Bark. Fitoterapia 2019, 135, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Li, Q.; Luo, T.; Betti, M.; Wang, M.; Qi, S.; Wu, L.; Zhao, L. Antioxidant Polyphenols from Lespedeza Bicolor Turcz. Honey: Anti-Inflammatory Effects on Lipopolysaccharide-Treated RAW 264.7 Macrophages. Antioxidants 2023, 12, 1809. [Google Scholar] [CrossRef]
- Bae, Y.; Pyun, G.; Choi, Y.; Yun, H.-Y.; Kim, I.; Cheon, S.; Yin, M.; Choi, P.; Lee, S.J.; Lee, Y.; et al. Antioxidant activity of Lespedeza cuneata G. Don against H₂O₂-induced oxidative stress in HepG2 cells. 한국식품영양과학회 학술대회발표집 2016, 504–504.
- Wahab, A.; Sim, H.; Choi, K.; Kim, Y.; Lee, Y.; Kang, B.; No, Y.S.; Lee, D.; Lee, I.; Lee, J.; et al. Antioxidant and Anti-Inflammatory Activities of Lespedeza Cuneata in Coal Fly Ash-Induced Murine Alveolar Macrophage Cells. Korean Journal of Veterinary Research 2023, 63, 27.1–27.9. [Google Scholar] [CrossRef]
- Kim, Y.M.; Lee, J.; Park, S.-H.; Lee, C.; Lee, J.W.; Lee, D.; Kim, N.; Lee, D.; Kim, H.Y.; Lee, C.H. LC–MS-Based Chemotaxonomic Classification of Wild-Type Lespedeza Sp. and Its Correlation with Genotype. Plant Cell Rep 2012, 31, 2085–2097. [Google Scholar] [CrossRef]
- Do, M.H.; Lee, J.H.; Cho, K.; Kang, M.C.; Subedi, L.; Parveen, A.; Kim, S.Y. Therapeutic Potential of Lespedeza Bicolor to Prevent Methylglyoxal-Induced Glucotoxicity in Familiar Diabetic Nephropathy. Journal of Clinical Medicine 2019, 8, 1138. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Tarbeeva, D.; Fedoreyev, S.; Busenbender, T.; Kaune, M.; Veselova, M.; Kalinovskiy, A.; Hauschild, J.; Grigorchuk, V.; Kim, N.; et al. Polyphenolic Compounds from Lespedeza Bicolor Root Bark Inhibit Progression of Human Prostate Cancer Cells via Induction of Apoptosis and Cell Cycle Arrest. Biomolecules 2020, 10, 451. [Google Scholar] [CrossRef]
- Ko, Y.-H.; Shim, K.-Y.; Kim, S.-K.; Seo, J.-Y.; Lee, B.-R.; Hur, K.-H.; Kim, Y.-J.; Kim, S.-E.; Do, M.H.; Parveen, A.; et al. Lespedeza Bicolor Extract Improves Amyloid Beta25 – 35-Induced Memory Impairments by Upregulating BDNF and Activating Akt, ERK, and CREB Signaling in Mice. Planta Medica 2019, 85, 1363–1373. [Google Scholar] [CrossRef]
| Species | DPPH IC50 (µg/mL) |
| L. cuneata | 20-25 µg/mL (strong) |
| L. bicolor | 35-50 µg/mL (moderate) |
| L. capitata | 40-60 µg/mL (weak) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




