Submitted:
08 April 2025
Posted:
09 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Antioxidant and Anti-Inflammatory Effects and Cellular Protection
2.1. Antioxidant and Anti-Inflammatory Properties
2.2. Cellular Protection Mechanisms
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Antioxidant Properties | Diabetic patients | Significant reduction in malondialdehyde and interleukin-6 levels, highlighting potent antioxidant effects | Feng et al. (2018) [1] |
| Overweight/obese individuals | Effective lowering of malondialdehyde and isoprostane levels, confirming oxidative stress reduction | Choi et al. (2011) [2] | |
| Cellular Protection |
Muscle cells | Activation of AMPK/Sirtuins/PGC-1α pathway, upregulation of antioxidant enzymes | Lewis et al. (2022) [4] |
| RAW 264.7 macrophages | Mitochondrial-targeted astaxanthin nanoparticles reduce ROS levels, enhance mitochondrial integrity | Mei et al. (2019) [5] | |
| Anti-inflammatory Effects | LPS-induced dendritic cells | Suppression of oxidative stress and inflammatory factor production via HO-1/Nrf2 pathway | Yin et al. (2021) [6] |
3. Immune Regulatory Effect
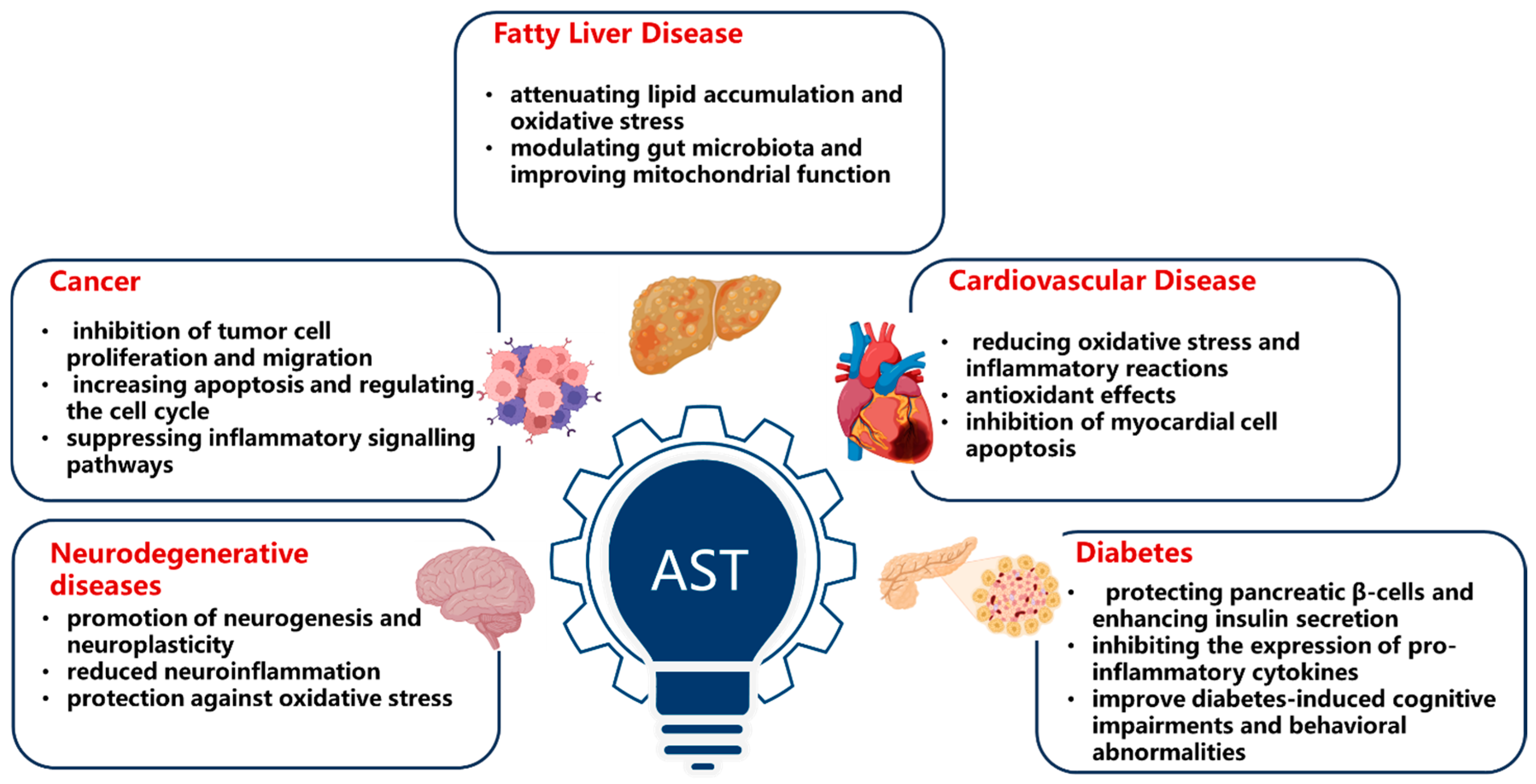
4. Anti-Apoptotic Effect and Nervous System Protection
5. Anti-Tumor Effect
6. Liver Protection
7. Anti-Fibrotic Effect
8. Cardiovascular Health Improvement
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Antioxidant Properties | Human umbilical vein endothelial cell | Activating the Nrf2/HO-1 pathway to mitigate oxidative stress and inflammatory responses | Niu et al. (2018) [34] |
| Anti-apoptotic Effect | H9c2 cell and primary cardiomyocyte | Protection of the heart from alcoholic cardiomyopathy partially by attenuating ER stress | Wang et al. (2021) [36] |
9. Anti-Diabetes Effect
10. Discussion and Conclusion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| AFLD | Alcoholic fatty liver disease |
| COPD | Chronic obstructive pulmonary disease |
| EMT | Epithelial-mesenchymal transition |
| FGF21 | Fibroblast growth factor 21 |
| IL-6 | Interleukin-6 |
| MCP-1 | Monocyte chemoattractant protein-1 |
| NAFLD | Non-alcoholic fatty liver disease |
| PD | Parkinson’s disease |
| PGC-1α | Peroxisome proliferator-activated receptor gamma coactivator 1α |
| ROS | Reactive oxygen species |
| TNF-α | Tumor necrosis factor-alpha |
| VE-cadherin | Vascular endothelial cadherin |
References
- Feng, Y.; Chu, A.; Luo, Q.; Wu, M.; Shi, X.; Chen, Y. The Protective Effect of Astaxanthin on Cognitive Function via Inhibition of Oxidative Stress and Inflammation in the Brains of Chronic T2DM Rats. Front Pharmacol 2018, 9, 748. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.D.; Kim, J.H.; Chang, M.J.; Kyu-Youn, Y.; Shin, W.G. Effects of astaxanthin on oxidative stress in overweight and obese adults. Phytother Res 2011, 25, 1813–1818. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Xu, N.; Qin, T.; Zhou, B.; Shi, Y.; Zhao, X.; Ma, B.; Xu, Z.; Li, C. Astaxanthin Provides Antioxidant Protection in LPS-Induced Dendritic Cells for Inflammatory Control. Mar Drugs 2021, 19. [Google Scholar] [CrossRef]
- Lewis Luján, L.M.; McCarty, M.F.; Di Nicolantonio, J.J.; Gálvez Ruiz, J.C.; Rosas-Burgos, E.C.; Plascencia-Jatomea, M.; Iloki Assanga, S.B. Nutraceuticals/Drugs Promoting Mitophagy and Mitochondrial Biogenesis May Combat the Mitochondrial Dysfunction Driving Progression of Dry Age-Related Macular Degeneration. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Mei, S.; Song, X.; Wang, Y.; Wang, J.; Su, S.; Zhu, J.; Geng, Y. Studies on Protection of Astaxanthin from Oxidative Damage Induced by H(2)O(2) in RAW 264.7 Cells Based on (1)H NMR Metabolomics. J Agric Food Chem 2019, 67, 13568–13576. [Google Scholar] [CrossRef]
- Yin, Y.; Xu, N.; Shi, Y.; Zhou, B.; Sun, D.; Ma, B.; Xu, Z.; Yang, J.; Li, C. Astaxanthin Protects Dendritic Cells from Lipopolysaccharide-Induced Immune Dysfunction. Mar Drugs 2021, 19. [Google Scholar] [CrossRef]
- Speranza, L.; Pesce, M.; Patruno, A.; Franceschelli, S.; Lutiis, M.A.; Grilli, A.; Felaco, M. Astaxanthin treatment reduced oxidative induced pro-inflammatory cytokines secretion in U937: SHP-1 as a novel biological target. Mar Drugs 2012, 10, 890–899. [Google Scholar] [CrossRef]
- Jeong, S.M.; Kim, Y.J. Astaxanthin Treatment Induces Maturation and Functional Change of Myeloid-Derived Suppressor Cells in Tumor-Bearing Mice. Antioxidants (Basel) 2020, 9. [Google Scholar] [CrossRef]
- Fan, Q.; Chen, Z.; Wu, Y.; Zhu, J.; Yu, Z. Study on the Enhancement of Immune Function of Astaxanthin from Haematococcus pluvialis. Foods 2021, 10. [Google Scholar] [CrossRef]
- Li, Q.; Jia, M.; Song, H.; Peng, J.; Zhao, W.; Zhang, W. Astaxanthin Inhibits STING Carbonylation and Enhances Antiviral Responses. J Immunol 2024, 212, 1188–1195. [Google Scholar] [CrossRef]
- He, Y.; Ding, M.; Zhang, J.; Huang, C.; Shi, J.; Wang, Y.; Tao, R.; Wu, Z.; Guo, W. Astaxanthin Alleviates Autoimmune Hepatitis by Modulating CD8(+) T Cells: Insights From Mass Cytometry and Single-Cell RNA Sequencing Analyses. Adv Sci (Weinh) 2024, 11, e2403148. [Google Scholar] [CrossRef]
- Wu, Y.; Xiao, Y.; Li, W.; Yang, C.; Ma, W.; Pang, Z.; Zhang, J.; Xiao, Z.; Li, J. Tea polyphenols, astaxanthin, and melittin can significantly enhance the immune response of juvenile spotted knifejaw (Oplegnathus punctatus). Fish Shellfish Immunol 2023, 138, 108817. [Google Scholar] [CrossRef]
- Fu, M.; Liang, X.; Zhang, X.; Yang, M.; Ye, Q.; Qi, Y.; Liu, H.; Zhang, X. Astaxanthin delays brain aging in senescence-accelerated mouse prone 10: inducing autophagy as a potential mechanism. Nutr Neurosci 2023, 26, 445–455. [Google Scholar] [CrossRef]
- Yan, T.; Ding, F.; Zhang, Y.; Wang, Y.; Wang, Y.; Zhang, Y.; Zhu, F.; Zhang, G.; Zheng, X.; Jia, G.; et al. Astaxanthin Inhibits H(2)O(2)-Induced Excessive Mitophagy and Apoptosis in SH-SY5Y Cells by Regulation of Akt/mTOR Activation. Mar Drugs 2024, 22. [Google Scholar] [CrossRef]
- Mohaghegh Shalmani, L.; Valian, N.; Pournajaf, S.; Abbaszadeh, F.; Dargahi, L.; Jorjani, M. Combination therapy with astaxanthin and epidermal neural crest stem cells improves motor impairments and activates mitochondrial biogenesis in a rat model of spinal cord injury. Mitochondrion 2020, 52, 125–134. [Google Scholar] [CrossRef]
- Liu, N.; Zeng, L.; Zhang, Y.M.; Pan, W.; Lai, H. Astaxanthin alleviates pathological brain aging through the upregulation of hippocampal synaptic proteins. Neural Regen Res 2021, 16, 1062–1067. [Google Scholar] [CrossRef]
- Nakagawa, K.; Kiko, T.; Miyazawa, T.; Carpentero Burdeos, G.; Kimura, F.; Satoh, A.; Miyazawa, T. Antioxidant effect of astaxanthin on phospholipid peroxidation in human erythrocytes. Br J Nutr 2011, 105, 1563–1571. [Google Scholar] [CrossRef]
- Wang, L.; Lu, K.; Lou, X.; Zhang, S.; Song, W.; Li, R.; Geng, L.; Cheng, B. Astaxanthin ameliorates dopaminergic neuron damage in paraquat-induced SH-SY5Y cells and mouse models of Parkinson’s disease. Brain Res Bull 2023, 202, 110762. [Google Scholar] [CrossRef]
- Wang, C.M.; Cai, X.L.; Wen, Q.P. Astaxanthin reduces isoflurane-induced neuroapoptosis via the PI3K/Akt pathway. Mol Med Rep 2016, 13, 4073–4078. [Google Scholar] [CrossRef]
- Sun, S.Q.; Zhao, Y.X.; Li, S.Y.; Qiang, J.W.; Ji, Y.Z. Anti-Tumor Effects of Astaxanthin by Inhibition of the Expression of STAT3 in Prostate Cancer. Mar Drugs 2020, 18. [Google Scholar] [CrossRef]
- Faraone, I.; Sinisgalli, C.; Ostuni, A.; Armentano, M.F.; Carmosino, M.; Milella, L.; Russo, D.; Labanca, F.; Khan, H. Astaxanthin anticancer effects are mediated through multiple molecular mechanisms: A systematic review. Pharmacol Res 2020, 155, 104689. [Google Scholar] [CrossRef]
- Ni, X.; Yu, H.; Wang, S.; Zhang, C.; Shen, S. Astaxanthin Inhibits PC-3 Xenograft Prostate Tumor Growth in Nude Mice. Mar Drugs 2017, 15. [Google Scholar] [CrossRef]
- Maoka, T.; Tokuda, H.; Suzuki, N.; Kato, H.; Etoh, H. Anti-oxidative, anti-tumor-promoting, and anti-carcinogensis activities of nitroastaxanthin and nitrolutein, the reaction products of astaxanthin and lutein with peroxynitrite. Mar Drugs 2012, 10, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, M.; Zhou, Y.; Bao, S.; Wang, F.; Li, C. Protective Effects of Astaxanthin against Oxidative Stress: Attenuation of TNF-α-Induced Oxidative Damage in SW480 Cells and Azoxymethane/Dextran Sulfate Sodium-Induced Colitis-Associated Cancer in C57BL/6 Mice. Mar Drugs 2024, 22. [Google Scholar] [CrossRef]
- Xu, Y.; Jiang, C. Astaxanthin suppresses the malignant behaviors of nasopharyngeal carcinoma cells by blocking PI3K/AKT and NF-κB pathways via miR-29a-3p. Genes Environ 2024, 46, 10. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Li, Z.; Xu, F.; Tian, Y.; Chen, T.; Li, J.; Guo, Y.; Lyu, Q. Antitumor Effects of Astaxanthin on Esophageal Squamous Cell Carcinoma by up-Regulation of PPARγ. Nutr Cancer 2022, 74, 1399–1410. [Google Scholar] [CrossRef]
- Tsuji, S.; Nakamura, S.; Maoka, T.; Yamada, T.; Imai, T.; Ohba, T.; Yako, T.; Hayashi, M.; Endo, K.; Saio, M.; et al. Antitumour Effects of Astaxanthin and Adonixanthin on Glioblastoma. Mar Drugs 2020, 18. [Google Scholar] [CrossRef]
- Wu, L.; Mo, W.; Feng, J.; Li, J.; Yu, Q.; Li, S.; Zhang, J.; Chen, K.; Ji, J.; Dai, W.; et al. Astaxanthin attenuates hepatic damage and mitochondrial dysfunction in non-alcoholic fatty liver disease by up-regulating the FGF21/PGC-1α pathway. Br J Pharmacol 2020, 177, 3760–3777. [Google Scholar] [CrossRef]
- Liu, H.; Liu, M.; Fu, X.; Zhang, Z.; Zhu, L.; Zheng, X.; Liu, J. Astaxanthin Prevents Alcoholic Fatty Liver Disease by Modulating Mouse Gut Microbiota. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Krestinina, O.; Odinokova, I.; Sotnikova, L.; Krestinin, R.; Zvyagina, A.; Baburina, Y. Astaxanthin Is Able to Prevent Alcohol-Induced Dysfunction of Liver Mitochondria. Antioxidants (Basel) 2022, 11. [Google Scholar] [CrossRef]
- Diao, W.; Chen, W.; Cao, W.; Yuan, H.; Ji, H.; Wang, T.; Chen, W.; Zhu, X.; Zhou, H.; Guo, H.; et al. Astaxanthin protects against renal fibrosis through inhibiting myofibroblast activation and promoting CD8(+) T cell recruitment. Biochim Biophys Acta Gen Subj 2019, 1863, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Meng, M.; Zhang, J.; Li, L.; Zhu, X.; Zhang, L.; Wang, C.; Gao, M. Astaxanthin ameliorates renal interstitial fibrosis and peritubular capillary rarefaction in unilateral ureteral obstruction. Mol Med Rep 2019, 19, 3168–3178. [Google Scholar] [CrossRef]
- Chen, H.; Wang, J.; Li, R.; Lv, C.; Xu, P.; Wang, Y.; Song, X.; Zhang, J. Astaxanthin attenuates pulmonary fibrosis through lncITPF and mitochondria-mediated signal pathways. J Cell Mol Med 2020, 24, 10245–10250. [Google Scholar] [CrossRef] [PubMed]
- Niu, T.; Xuan, R.; Jiang, L.; Wu, W.; Zhen, Z.; Song, Y.; Hong, L.; Zheng, K.; Zhang, J.; Xu, Q.; et al. Astaxanthin Induces the Nrf2/HO-1 Antioxidant Pathway in Human Umbilical Vein Endothelial Cells by Generating Trace Amounts of ROS. J Agric Food Chem 2018, 66, 1551–1559. [Google Scholar] [CrossRef]
- Bjørklund, G.; Gasmi, A.; Lenchyk, L.; Shanaida, M.; Zafar, S.; Mujawdiya, P.K.; Lysiuk, R.; Antonyak, H.; Noor, S.; Akram, M.; et al. The Role of Astaxanthin as a Nutraceutical in Health and Age-Related Conditions. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Wang, W.; Liu, T.; Liu, Y.; Yu, L.; Yan, X.; Weng, W.; Lu, X.; Zhang, C. Astaxanthin attenuates alcoholic cardiomyopathy via inhibition of endoplasmic reticulum stress-mediated cardiac apoptosis. Toxicol Appl Pharmacol 2021, 412, 115378. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Tang, N.; Kord-Varkaneh, H.; Low, T.Y.; Tan, S.C.; Wu, X.; Zhu, Y. The effects of astaxanthin supplementation on obesity, blood pressure, CRP, glycemic biomarkers, and lipid profile: A meta-analysis of randomized controlled trials. Pharmacol Res 2020, 161, 105113. [Google Scholar] [CrossRef]
- Sakayanathan, P.; Loganathan, C.; Thayumanavan, P. Astaxanthin-S-Allyl Cysteine Ester Protects Pancreatic β-Cell From Glucolipotoxicity by Suppressing Oxidative Stress, Endoplasmic Reticulum Stress and mTOR Pathway Dysregulation. J Biochem Mol Toxicol 2024, 38, e70058. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, L.; Guo, Y.; Zhang, T.; Qiao, X.; Wang, J.; Xu, J.; Xue, C. Hydrophilic Astaxanthin: PEGylated Astaxanthin Fights Diabetes by Enhancing the Solubility and Oral Absorbability. J Agric Food Chem 2020, 68, 3649–3655. [Google Scholar] [CrossRef]
- Ying, C.J.; Zhang, F.; Zhou, X.Y.; Hu, X.T.; Chen, J.; Wen, X.R.; Sun, Y.; Zheng, K.Y.; Tang, R.X.; Song, Y.J. Anti-inflammatory Effect of Astaxanthin on the Sickness Behavior Induced by Diabetes Mellitus. Cell Mol Neurobiol 2015, 35, 1027–1037. [Google Scholar] [CrossRef]
- Kanwugu, O.N.; Glukhareva, T.V.; Danilova, I.G.; Kovaleva, E.G. Natural antioxidants in diabetes treatment and management: prospects of astaxanthin. Crit Rev Food Sci Nutr 2022, 62, 5005–5028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.S.; Zhang, X.; Wu, Q.; Li, W.; Wang, C.X.; Xie, G.B.; Zhou, X.M.; Shi, J.X.; Zhou, M.L. Astaxanthin offers neuroprotection and reduces neuroinflammation in experimental subarachnoid hemorrhage. J Surg Res 2014, 192, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Qi, X. The Putative Role of Astaxanthin in Neuroinflammation Modulation: Mechanisms and Therapeutic Potential. Front Pharmacol 2022, 13, 916653. [Google Scholar] [CrossRef]
- Zhao, T.; Ma, D.; Mulati, A.; Zhao, B.; Liu, F.; Liu, X. Development of astaxanthin-loaded layer-by-layer emulsions: physicochemical properties and improvement of LPS-induced neuroinflammation in mice. Food Funct 2021, 12, 5333–5350. [Google Scholar] [CrossRef] [PubMed]
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Enhancement of both cellular and humoral immunity | SPF Kunming female mice | Significantly improvement in delayed allergy reaction and NK cell activity | Fan et al. (2021) [9] |
| Inhibition in STING carbonylation to enhance antiviral responses | HSV-1-induced mouse primary peritoneal macrophages | Mitigating lipid peroxidation and inflammation, augments type I interferon production, restricting viral replication | Li et al. (2024) [10] |
| Downregulation of pro-inflammatory cytokines in autoimmune hepatitis | Concanavalin A-induced mouse model | Alleviating liver damage, downregulates pro-inflammatory cytokines, increases CD8+ T cells | He et al. (2024) [11] |
| Counteracting post-exercise decline in immune-related plasma proteins | Oplegnathus punctatus | Effective in counteracting the post-exercise decline in immune-related plasma proteins, particularly immunoglobulin IgM | Wu et al. (2023) [12] |
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Improvement of brain aging | 6-month-old SAMP10 mice | Induction of autophagy by regulating IGF-1/Akt/mTOR and IGF-1/Akt/FoxO3a signaling pathways. | Fu et al. (2023) [13,14] |
| Prevention of neurotoxicity | H2O2-induced SH-SY5Y cells | Inhibition H2O2-induced apoptosis in SH-SY5Y cells by ameliorating mitochondrial damage and enhancing cell survival. | Yan et al. (2024) [13,14] |
| Promoting neurogenesis and neuroplasticity | Epidermal neural crest stem cells extracted from bulge hair follicle in adults | Enhances spatial memory performance, improves hippocampus-related spatial memory | Mohaghegh et al. (2020) [15] |
| Antioxidant Properties | A mouse model of brain aging | Improvements in the learning, cognitive, and memory abilities of mice. | Liu et al. (2021) [16] |
| Improvement of erythrocyte antioxidant status | Thirty middle-aged and senior subjects | Decreased PLOOH levels, which may contribute to the prevention of dementia. | Nakagawa et al. (2011) [17] |
| Suppression of oxidative stress and apoptosis in neurons | PQ-induced SH-SY5Y cells and mice Parkinson’s disease model | Inhibition PQ-induced activation of MAPK signaling pathway | Wang et al. (2023) [18] |
| Anti-neuroapoptosis Effects | Isofluorane-induced rat model | Reducing the isoflurane-induced neuroapoptosis via activation of the PI3K/Akt signaling pathway | Wang et al. (2016) [19] |
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Anti-tumor effect | Prostate cancer DU145 cell | Suppression of tumor cell proliferation and metastasis by inhibiting STAT3 expression | Sun et al. (2020) [20] |
| PC-3 prostate cancer xenograft mouse modelcells | Validated inhibitory effects on tumor growth |
Ni et al. (2017) [22] |
|
| Mouse skin papillomas | Antioxidant properties and capacity to scavenge peroxynitrite | Maoka et al. (2012) [23] |
|
| SW480 cell and colorectal cancer mice model | Curtails tumor cell proliferation and migration in colorectal cancer by regulating MAPK and NF-κB signaling pathways | Zhang et al. (2024) [24] |
|
| C666-1 cell | Inhibits proliferation, migration, and invasion in nasopharyngeal carcinoma by blocking PI3K/AKT and NF-κB pathways via miR-29a-3p | Xu et al. (2024) [25] |
|
| F344 rats | Inhibits proliferation and migration of esophageal cancer cells by upregulating PPARγ expression | Cui et al. (2022) [26] |
|
| U251MG cell | Hormetic effect in glioblastoma multiforme, where low concentrations promote cell proliferation, while high concentrations induce apoptosis | Tsuji et al. (2020) [27] |
| Mechanism | Study Population/Model | Key Findings | Reference |
|---|---|---|---|
| Antioxidant Properties | Pancreatic β-cell | Protection of pancreatic β-cells, enhances insulin secretion | Sakayanathan et al. (2024) [38] |
| Anti-inflammation Effect | Fat- and high-sucrose-diet-induced insulin-resistant mouse model | Strengthening endogenous antioxidant system, mitigates oxidative damage | Liu et al. (2020) [39] |
| Diabetic mice model | Decreasing GFAP-positive cells in the brain and down-regulating the cleaved caspase-3, IL-6, and IL-1β, and up-regulating CBS in the frontal cortex | Ying et al. (2015) [40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




