Submitted:
02 April 2025
Posted:
03 April 2025
You are already at the latest version
Abstract
Keywords:
Neosynthesis of Collagen and Extracellular Matrix: The Long-Dominant Foreign Body Reaction Paradigm
Is It Conceivable to Progress Beyond the Foreign Body Response in Skin Connective tissue Regeneration?
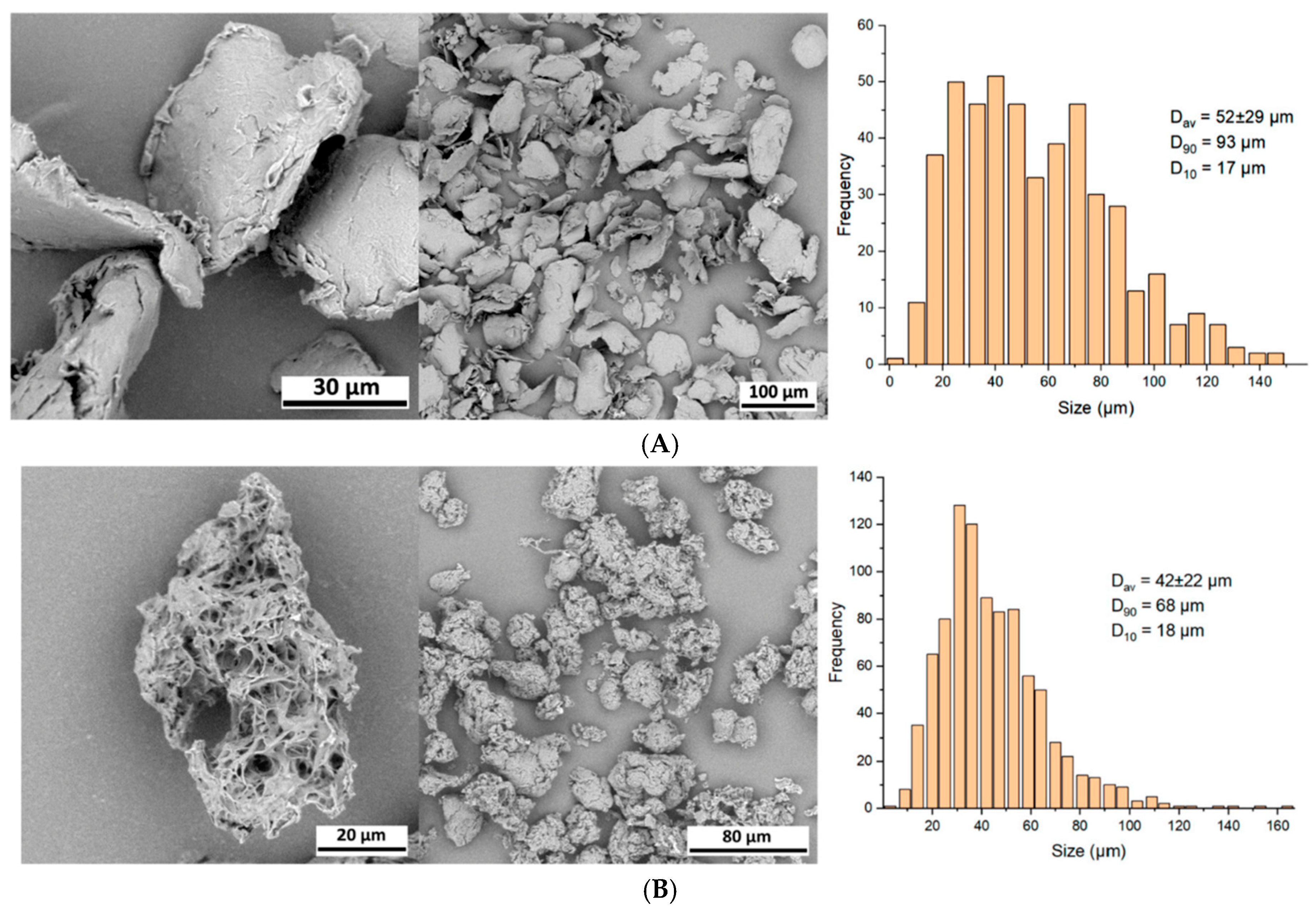
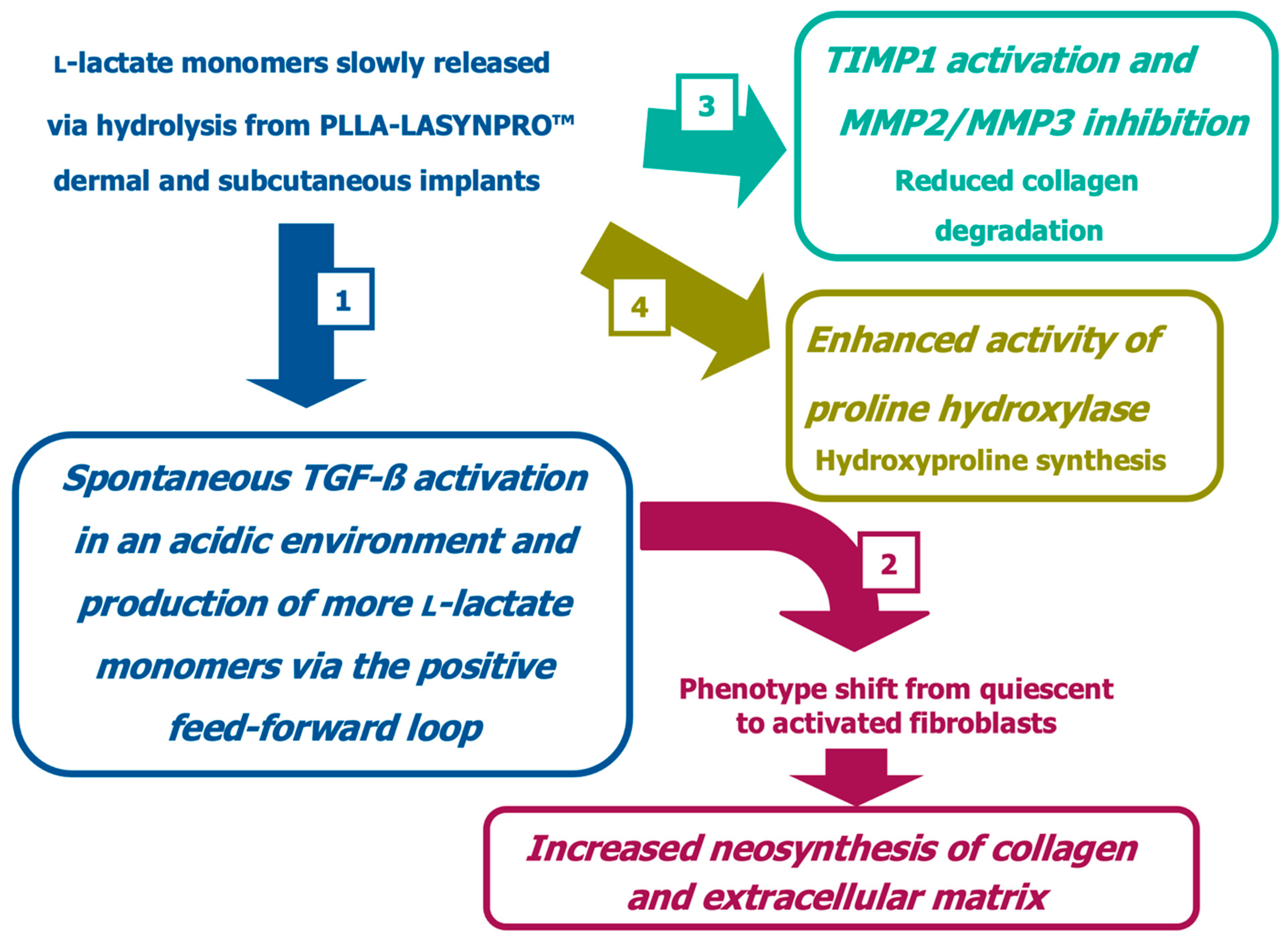
The PLLA-LASYNPRO™ Rationale Beyond the FBR Paradigm
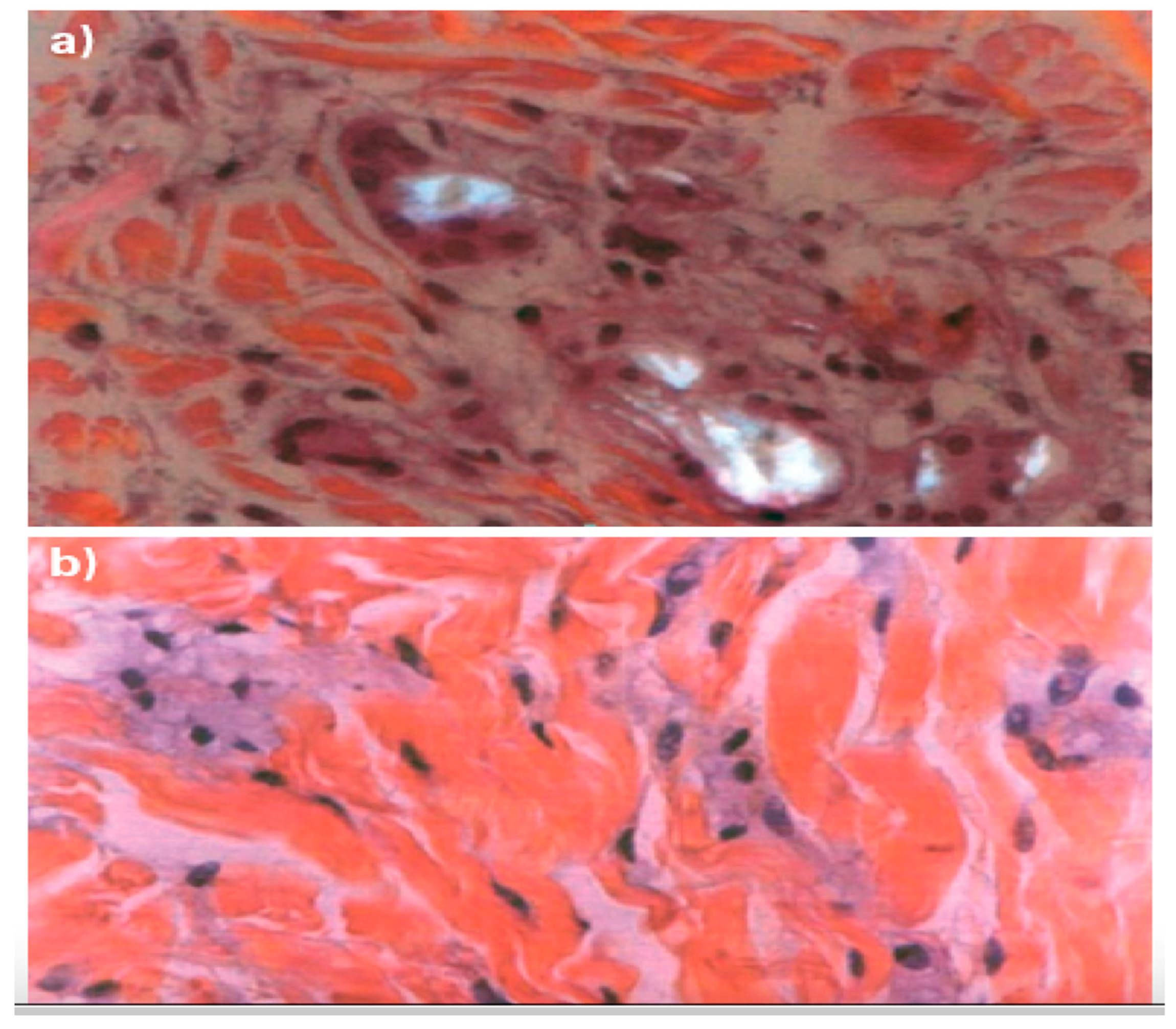
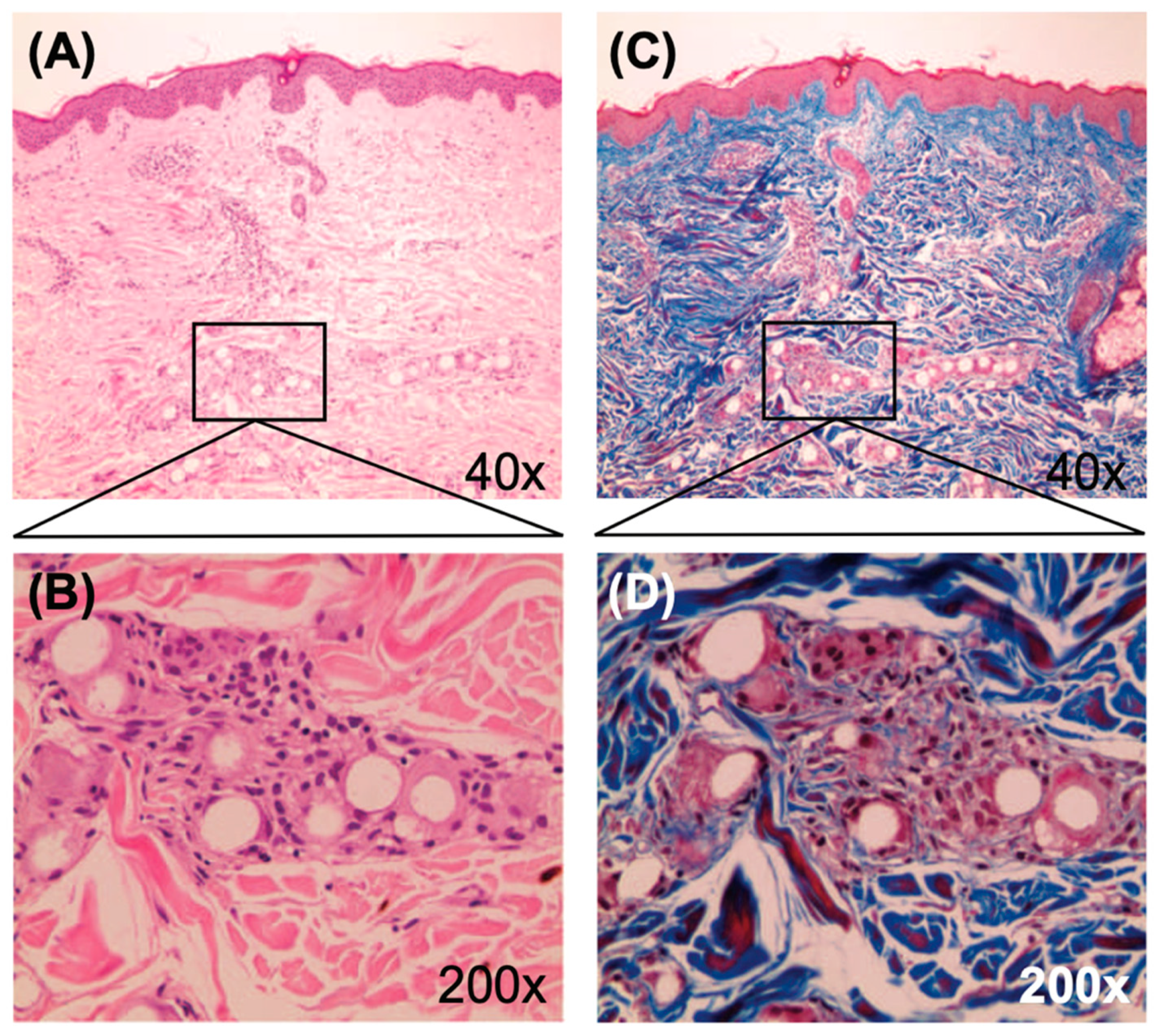
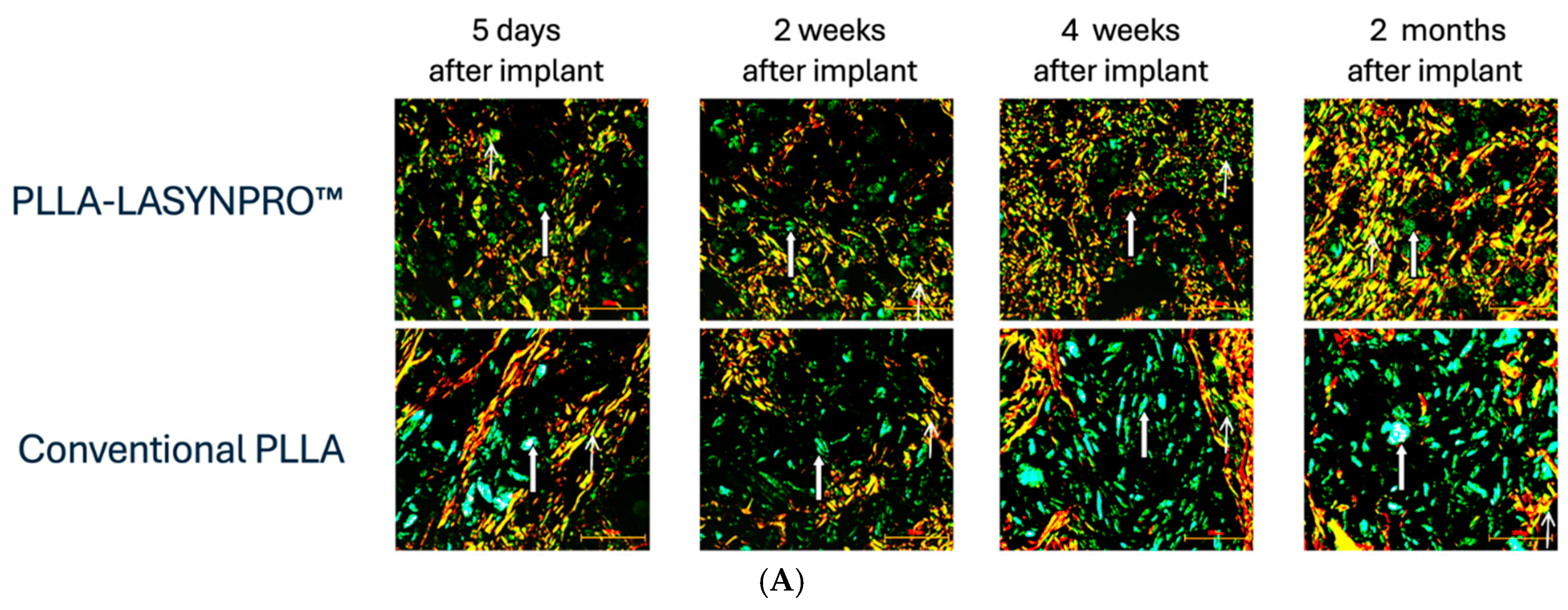
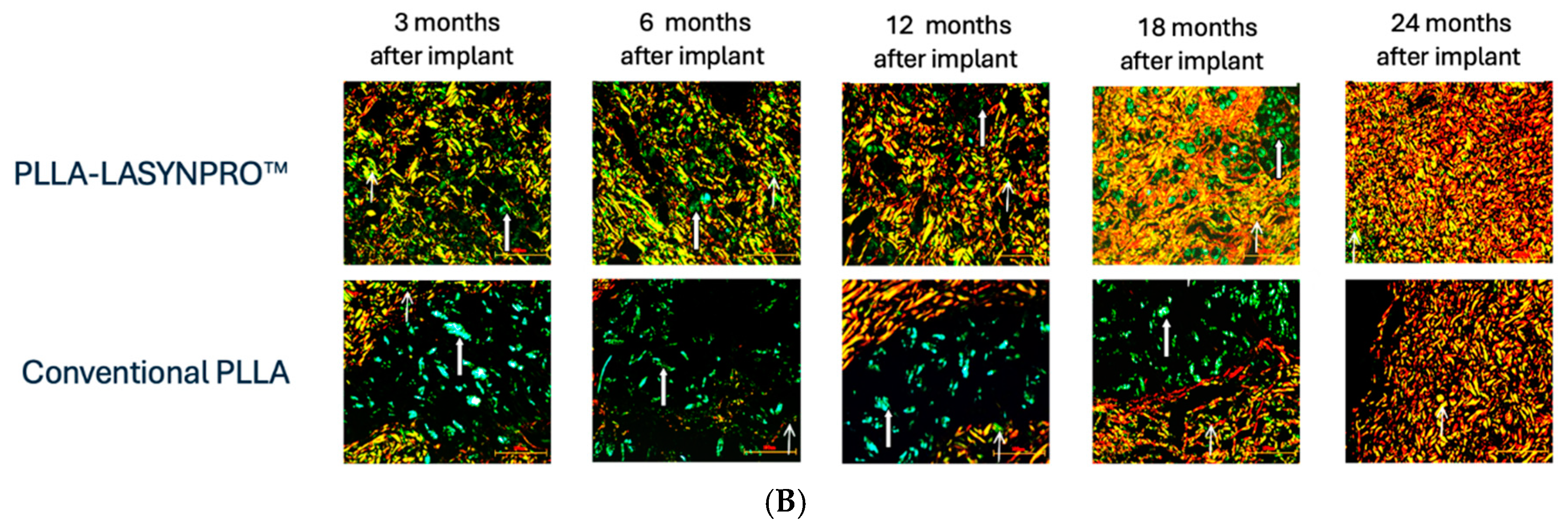
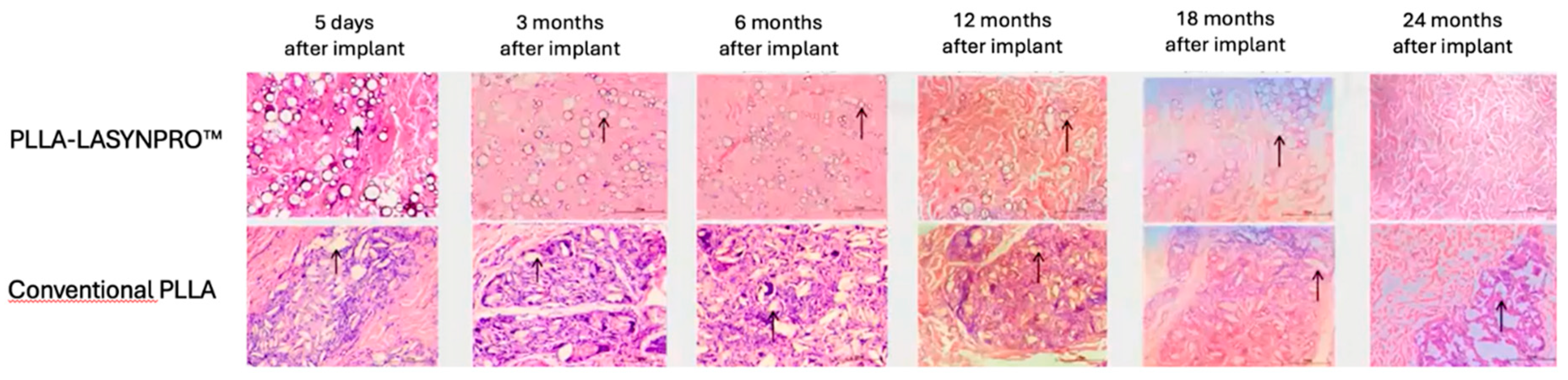
The Non-Inflammatory Action of the New-Technology Microspheres
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vleggaar, D. Facial volumetric correction with injectable poly-L-lactic acid. Dermatol Surg 2005, 31 (11, Pt 2), 1511–1518. [Google Scholar] [CrossRef]
- Kim, J.A.; Van Abel, D. Neocollagenesis in human tissue injected with a polycaprolactone-based dermal filler. J Cosm Laser Ther 2015, 17, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Palm, M.; Mayoral, F.; Rajani, A.; et al. Chart review presenting the safety of injectable PLLA used with alternative reconstitution volume for facial treatments. J Drugs Dermatol 2021, 20, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Fang, W.; Wang, F. Injectable fillers: current status, physicochemical properties, function mechanism, and perspectives. RSC Adv 2023, 13, 23841–23858. [Google Scholar] [CrossRef]
- Ianhez, M.; de Goés E Silva Freire, G.; Sigrist, R.M.S.; Colpas, P.T.; Alves de Faria, I.; Américo Brasil Parada, M.O.; Miot, H.A. Complications of collagen biostimulators in Brazil: Description of products, treatments, and evolution of 55 cases. J Cosmet Dermatol 2024, 23, 2829–2835. [Google Scholar] [CrossRef]
- U.S. FDA Center for Devices and Radiological Health. Polycaprolactone (PCL) Safety Profile. Published by ECRI, Plymouth Meeting, PA, USA. December 2, 2021. Downloaded on September 12, 2024, from www.fda.gov/media/158492/download.
- Christsen, M.-O.; Vercesi, F. Polycaprolactone: How a well-known and futuristic polymer has become an innovative collagen-stimulator in esthetics. Clin Cosmet Investig Dermatol 2020, 13, 31–48. [Google Scholar] [CrossRef]
- Nowag, B.; Schäfer, D.; Hengl, T.; Corduff, N.; Goldie, K. Biostimulating fillers and induction of inflammatory pathways: A preclinical investigation of macrophage response to calcium hydroxylapatite and poly-L-lactic acid. J Cosmet Dermatol 2024, 23, 99–106. [Google Scholar] [CrossRef]
- McCarthy, A.D.; Hartmann, C.; Durkin, A.; Shahriar, S.; Khalifian, S.; Xie, J. A morphological analysis of calcium hydroxylapatite and poly-L-lactic acid biostimulator particles. Skin Res Technol 2024, 30, e13764. [Google Scholar] [CrossRef] [PubMed]
- Waibel, J.; Nguyen, T.Q.; Le, J.H.T.D.; et al. Gene analysis of biostimulators: Poly-L-lactic acid triggers regeneration while calcium hydroxylapatite induces inflammation upon facial injection. J Drugs Dermatol 2025, 24, 34–40. [Google Scholar] [CrossRef]
- Mazzuco, R.; Evangelista, C.; Gobbato, D.O.; de Almeida, L.M. Clinical and histological comparative outcomes after injections of poly-L-lactic acid and calcium hydroxyapatite in arms: A split side study. J Cosmet Dermatol 2022, 21, 6727–6733. [Google Scholar] [CrossRef]
- McCarthy, A.D.; van Loghem, J.; Martinez, K.A.; Aguilera, S.B.; Funt, D. A structured approach for treating calcium hydroxylapatite focal accumulations. Aesthet Surg J 2024, 44, 869–879. [Google Scholar] [CrossRef] [PubMed]
- The effect of size and shape of PLLA particles: usability, technical and clinical perspectives. RISE Research Institutes of Sweden AB, Gothenburg, 2023.
- Baranov, M.V.; Kumar, M.; Sacanna, S.; Thutupalli, S.; van den Bogaart, G. Modulation of immune responses by particle size and shape. Front Immunol 2021, 11, 607945. [Google Scholar] [CrossRef]
- Ao, Y.J.; Yi, Y.; Wu, G.H. Application of PLLA (Poly-L-Lactic acid) for rejuvenation and reproduction of facial cutaneous tissue in aesthetics. A review. Medicine (Baltimore) 2024, 103, e37506. [Google Scholar] [CrossRef]
- Urdiales-Gálvez, F.; Benítez, P.A.; Díaz, I. Facial rejuvenation with an innovative poly-L-lactic acid (Juläine) for nasolabial folds: interim data analysis of a prospective, non-randomized, multicenter, open-label Spanish study. J Cosm Dermatol 2025, 24, e70137. [Google Scholar] [CrossRef] [PubMed]
- Dario Bertossi, Maurizio Cavallini, Alessandra Camporese on behalf of The Next-Generation PLLA-LASYNPRO™ Regenerative Medicine Expert Board. First insights on the upcoming role of next-generation PLLA-LASYNPRO™ in aesthetic and regenerative medicine. A survey of experts — Practical Suggestions. This accompanying manuscript is under contemporary review by J Clin Med.
- Sedush, N.G.; Kalinin, K.T.; Azarkevich, P.N.; Gorskaya, A.A. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: A comparative study. Cosmetics 2023, 10, 110. [Google Scholar] [CrossRef]
- McCarthy, A.D.; Hartmann, C.; Durkin, A.; Shahriar, S.; Khalifian, S.; Xie, J. A morphological analysis of calcium hydroxylapatite and poly-L-lactic acid biostimulator particles. Skin Res Technol 2024, 30, e13764. [Google Scholar] [CrossRef]
- Lemperle, G.; Neugebauer, P.; Kernke, R.; Lerche, K.-H.; Lemperle, S. Microspheres for cosmetic and medical injections must be free of phagocytosable microparticles under 20 microns. Biomed J Sci & Tech Res 2017, 1, 1682–1686. [Google Scholar]
- Nordberg Medical R&D internal reports available upon request.
- Sung-Ae, K.; Hyo-Seon, K.; Jin-Woong, J.; Sung-Il, S.; Young-Wook, R. Poly-L-Lactic acid increases collagen gene expression and synthesis in cultured dermal fibroblast (Hs68) through the p38 MAPK pathway. Ann Dermatol 2019, 31, 97–100. [Google Scholar]
- Li, G.; Li, Y.-Y.; Sun, J.-E.; Lin, W.-H.; Zhou, R.-X. ILK-PI3K/AKT pathway participates in cutaneous wound contraction by regulating fibroblast migration and differentiation to myofibroblast. Lab Invest 2016, 96, 741–51. [Google Scholar] [CrossRef]
- Kottmann, R.M.; Kulkarni, A.A.; Smolnycki, K.A.; et al. Lactic acid is elevated in idiopathic pulmonary fibrosis and induces myofibroblast differentiation via pH-dependent activation of transforming growth factor-beta. Am J Respir Crit Care Med 2012, 186, 740–751. [Google Scholar] [CrossRef]
- Judge, J.L.; Owens, K.M.; Pollock, S.J.; et al. Ionizing radiation induces myofibroblast differentiation via lactate dehydrogenase. Am J Physiol Lung Cell Mol Physiol 2015, 309, L879–L887. [Google Scholar] [CrossRef]
- Meng, K.-M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol 2016, 12, 325–338. [Google Scholar] [CrossRef]
- Khalil, H.; Kanisicak, O.; Prasad, V.; et al. Fibroblast-specific TGF-beta-Smad2/3 signaling underlies cardiac fibrosis. J Clin Invest 2017, 127, 3770–3783. [Google Scholar] [CrossRef]
- Stein, P.; Vitavska, O.; Kind, P.; Hoppe, W.; Wieczorek, H.; Schürer, N.Y. The biological basis for poly-L-lactic acid-induced augmentation. J Dermatol Sci 2015, 78, 26–33. [Google Scholar] [CrossRef]
- Zhu, W.; Dong, C. Poly-L-Lactic acid increases collagen gene expression and synthesis in cultured dermal fibroblast (Hs68) through the TGF-beta/Smad pathway. J Cosmet Dermatol 2023, 22, 1213–1219. [Google Scholar] [CrossRef]
- Vavřička, J.; Brož, P.; Follprecht, D.; Novák, J.; Kroužecký, A. Modern perspective of lactate metabolism. Physiol Res 2024, 73, 499–514. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Lee, J.H.; Kim, H.M.; et al. Poly-L-lactic acid fillers improved dermal collagen synthesis by modulating M2 macrophage polarization in aged animal skin. Cells 2023, 12, 1320. [Google Scholar] [CrossRef] [PubMed]
- Kottmann, R.M.; Kulkarni, A.A.; Smolnycki, K.A.; Lyda, E.; Dahanayake, T.; Salibi, R.; Honnons, S.; et al. Lactic acid is elevated in idiopathic pulmonary fibrosis and induces myofibroblast differentiation via pH-dependent activation of transforming growth factor-beta. Am J Respir Crit Care Med 2012, 186, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Judge, J.L.; Owens, K.M.; Pollock, S.J.; et al. Ionizing radiation induces myofibroblast differentiation via lactate dehydrogenase. Am J Physiol Lung Cell Mol Physiol 2015, 309, L879–L887. [Google Scholar] [CrossRef]
- Meng, K.-M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol 2016, 12, 325–338. [Google Scholar] [CrossRef]
- Khalil, H.; Kanisicak, O.; Prasad, V.; Correll, R.N.; Fu, X.; Schips, T.; Vagnozzi, R.J.; et al. Fibroblast-specific TGF-beta-Smad2/3 signaling underlies cardiac fibrosis. J Clin Invest 2017, 127, 3770–3783. [Google Scholar] [CrossRef] [PubMed]
- Comstock, J.P.; Udenfriend, S. Effect of lactate on collagen proline hydroxylase activity in cultured L-929 fibroblasts. Proc Natl Acad Sci U S A. 1970, 66, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Jung, Y.A.; Yun, J.M.; Kim, Y.; Kim, S.A.; Suh, S.I.; Ryoo, Y.W. Effects of poly-L-lactic acid on adipogenesis and collagen gene expression in cultured adipocytes irradiated with ultraviolet B rays. Ann Dermatol 2023, 35, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Chirumbolo, S.; · Bertossi, D.; Magistretti, P. Insights on the role of l-lactate as a signaling molecule in skin aging. Biogerontology 2023, 24, 709–726. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



