Submitted:
29 March 2025
Posted:
31 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Ceramide Biosynthesis and Metabolism
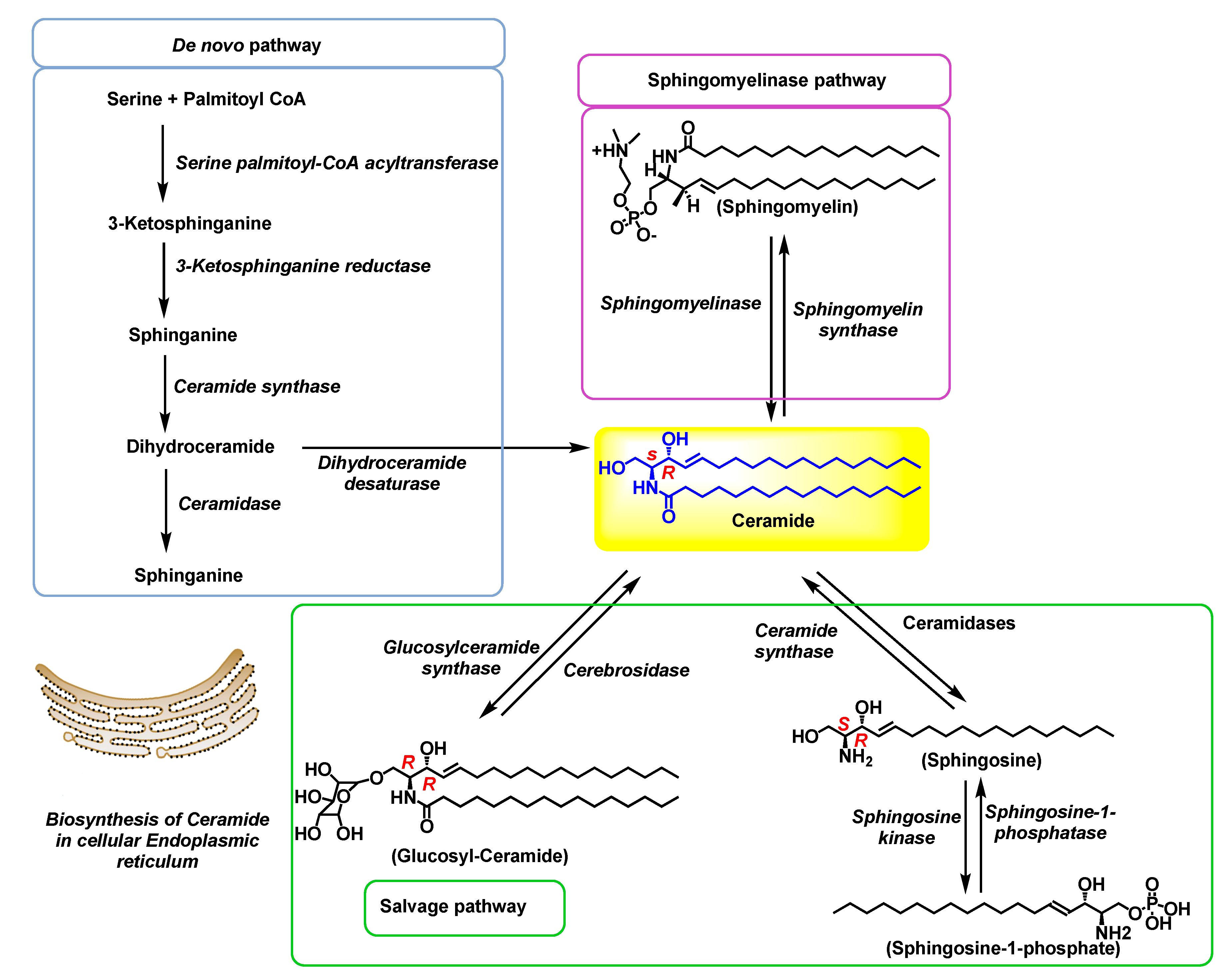
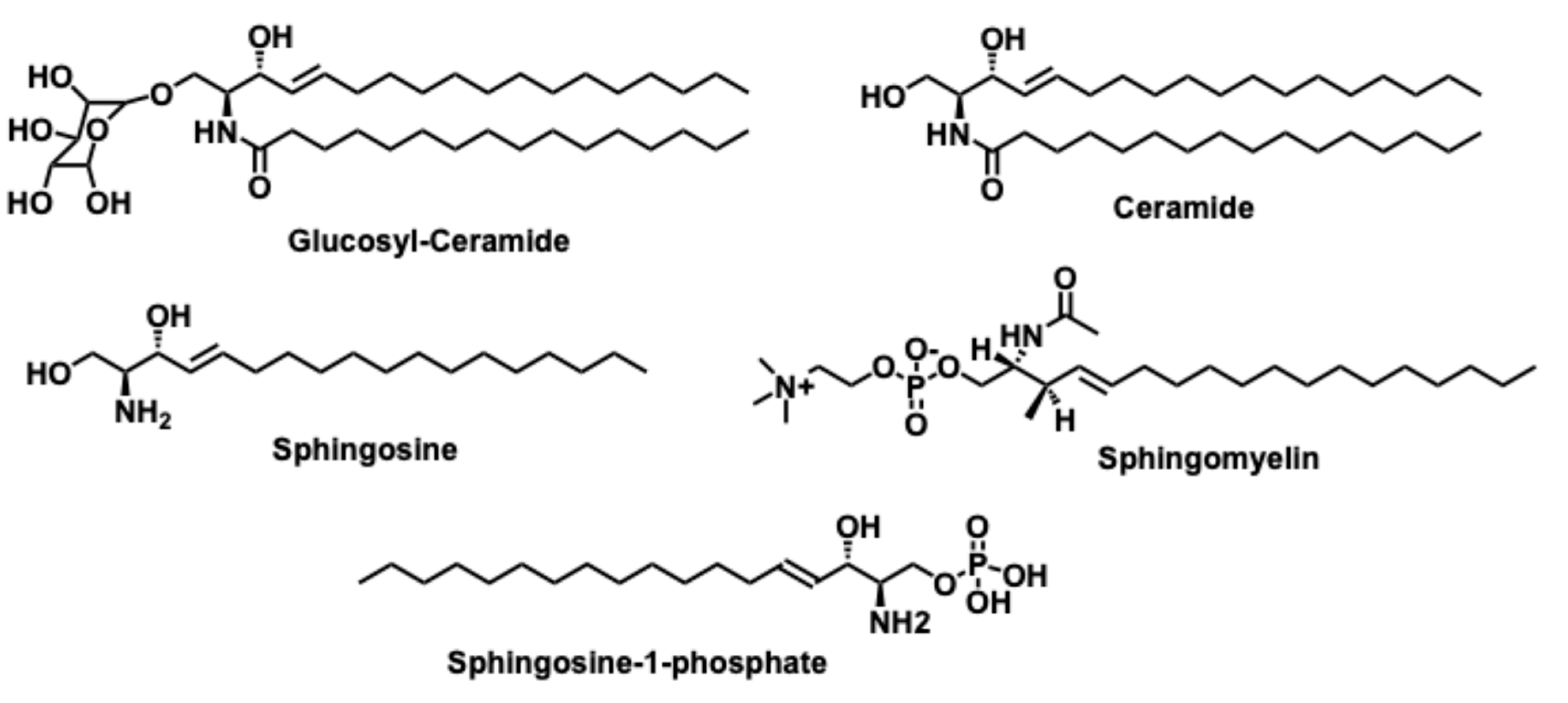
2.1. De novo biosynthesis pathway
2.2. Sphingomyelinase (SMase) pathway
2.3. The salvage pathway
3. Biological significance of Sphingolipids and derivatized sphingolipids
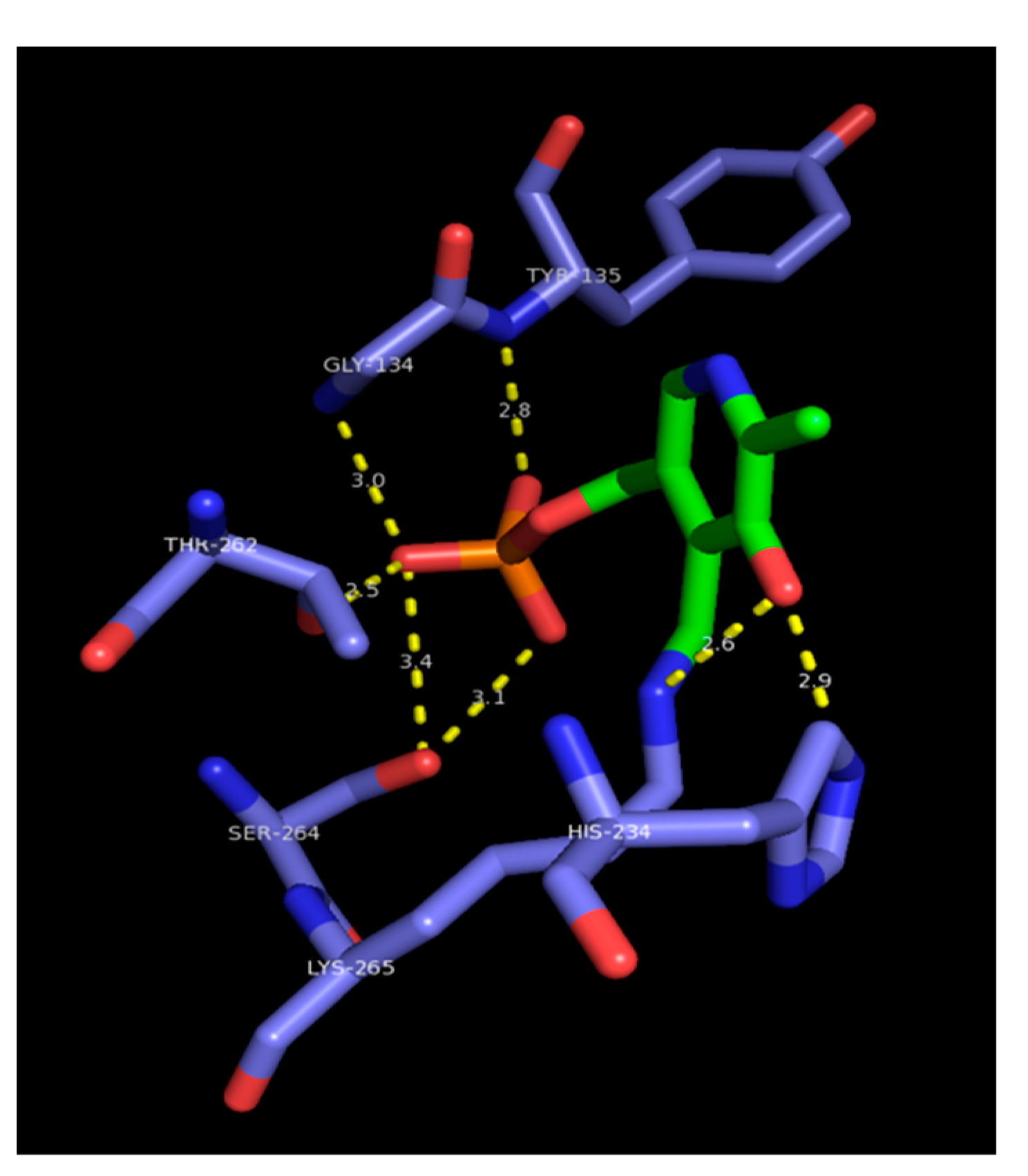
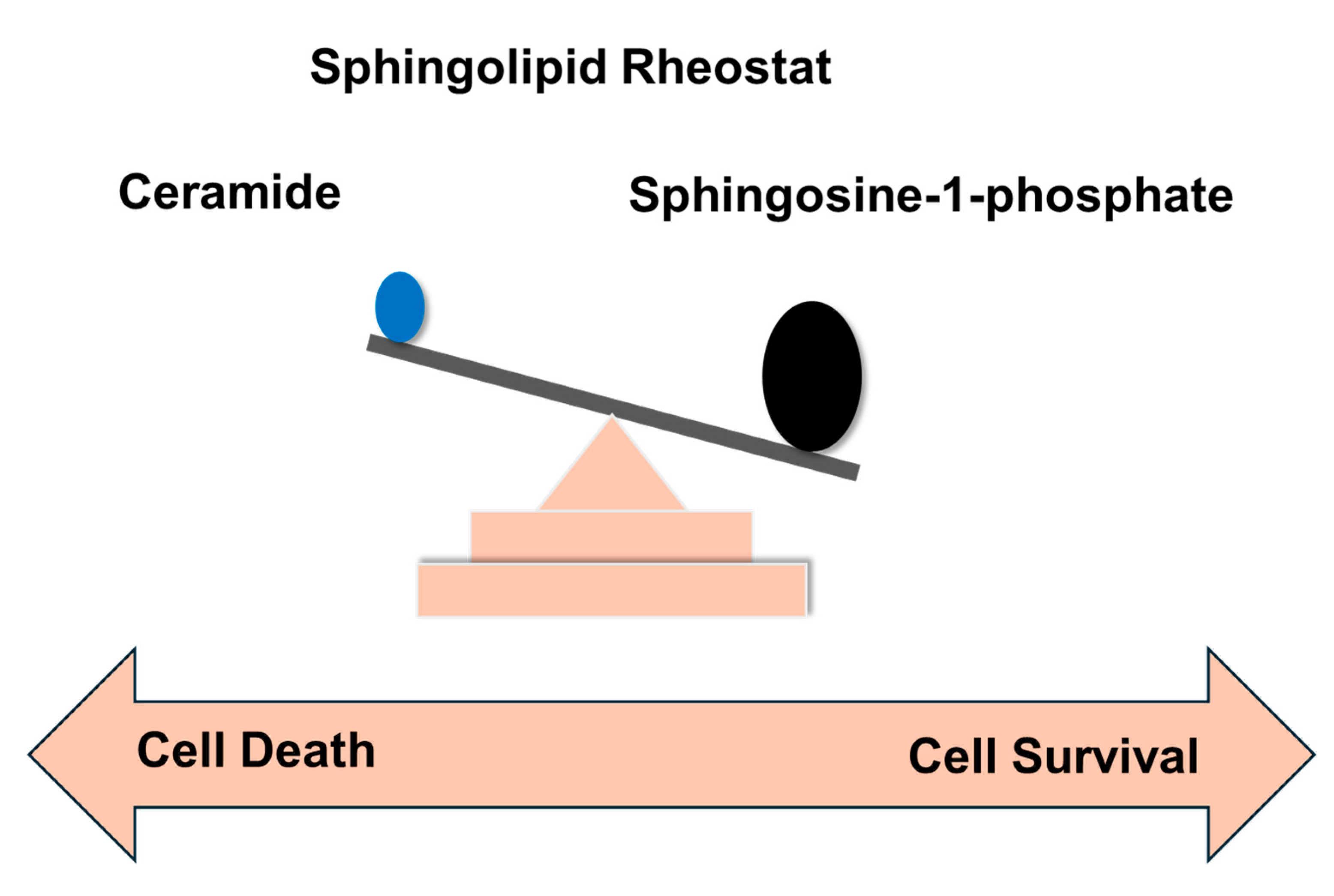
4. Sphingolipid rheostat, and significance in cancer
5. Sphingolipid-based therapeutic agents
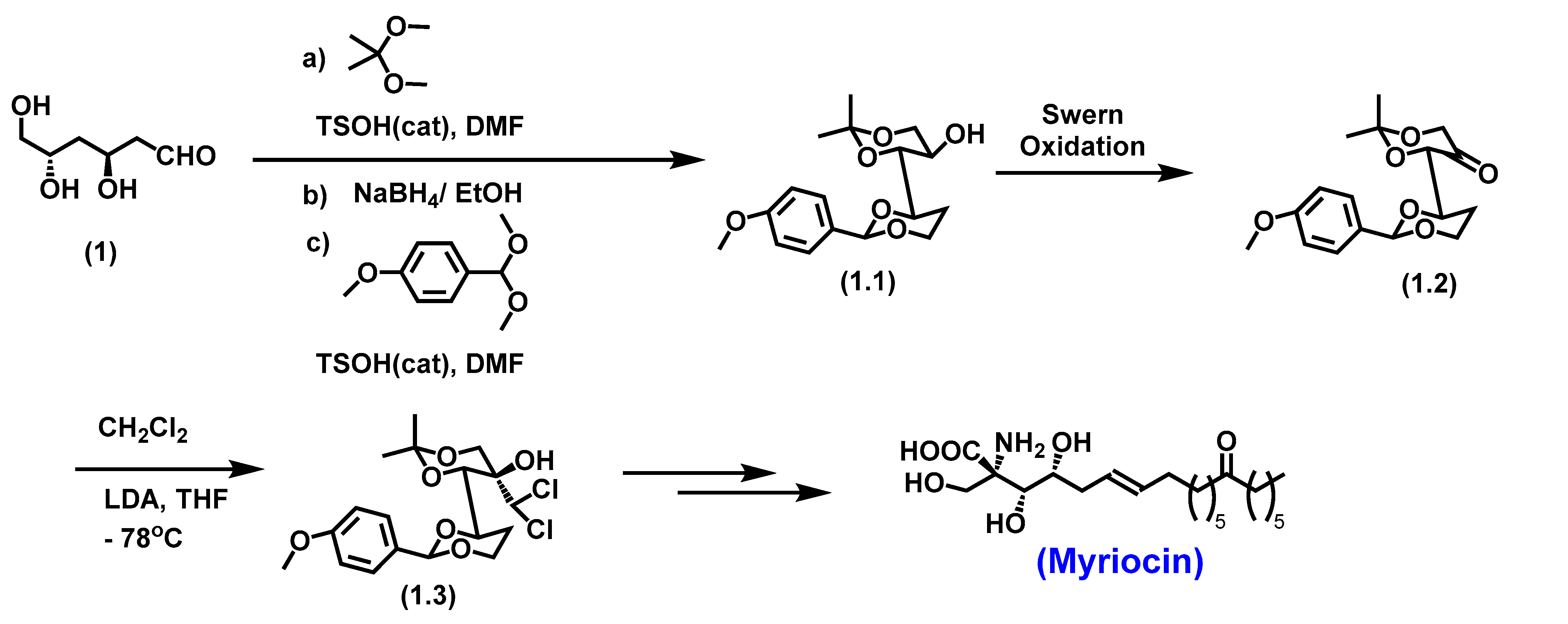
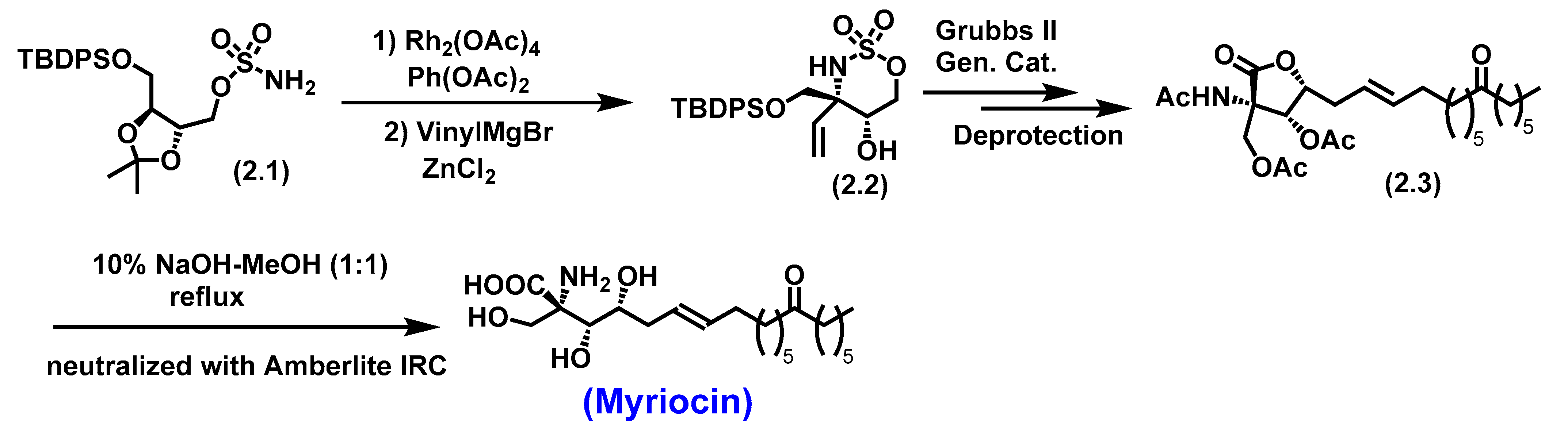
5.1. Myriocin
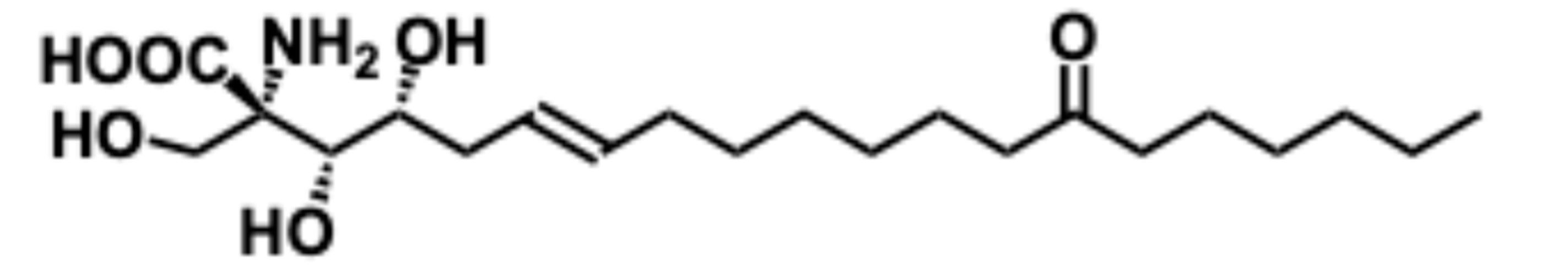
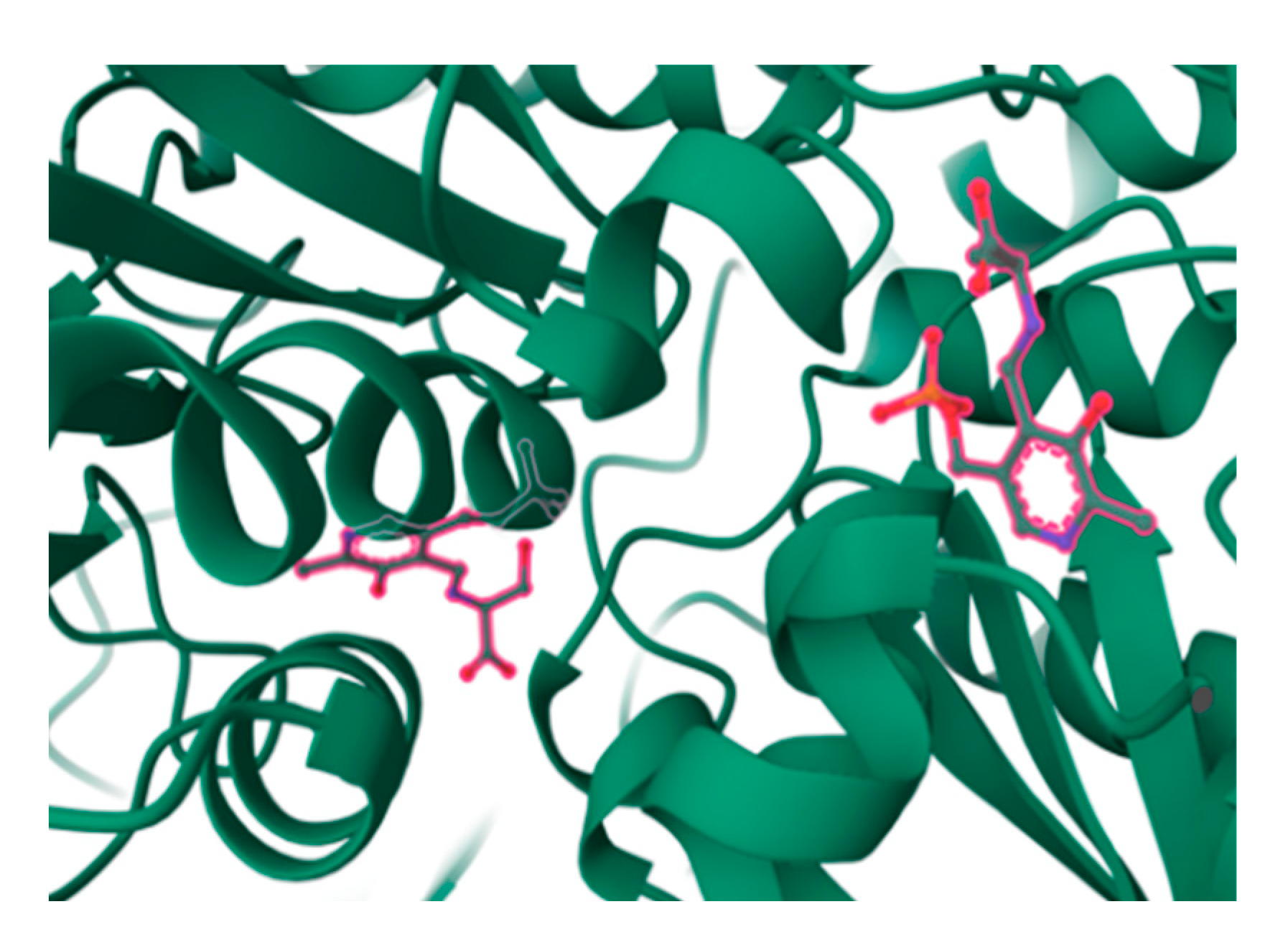
5.1.1. Biological Significance of Myriocin
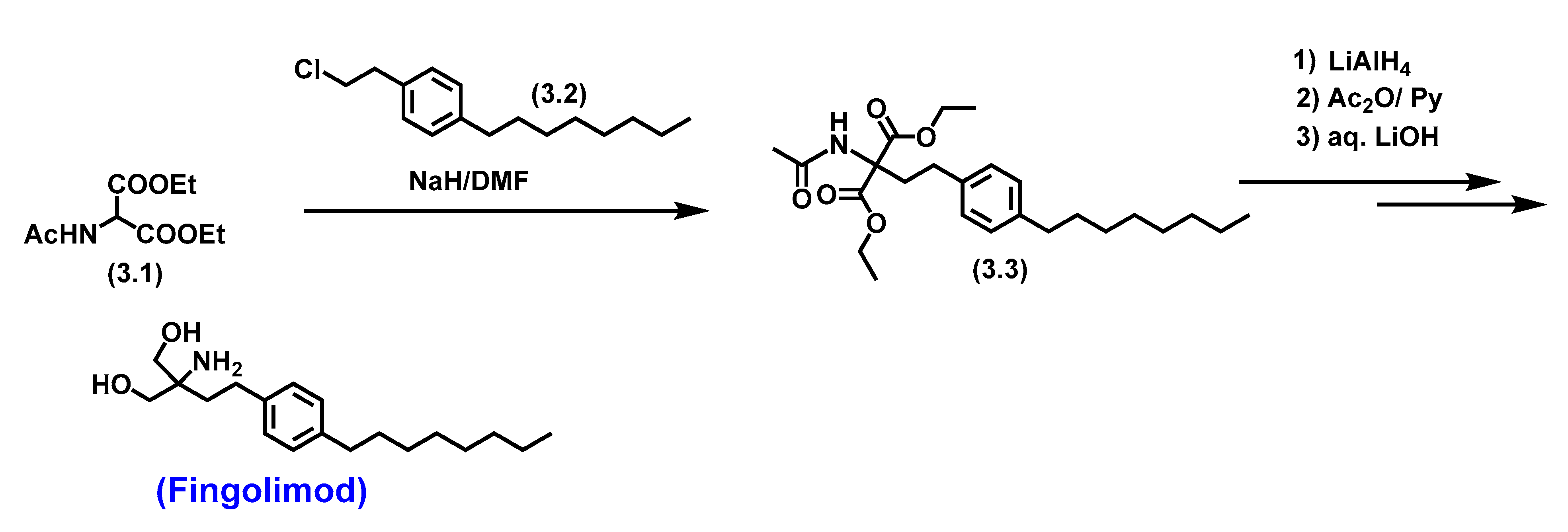
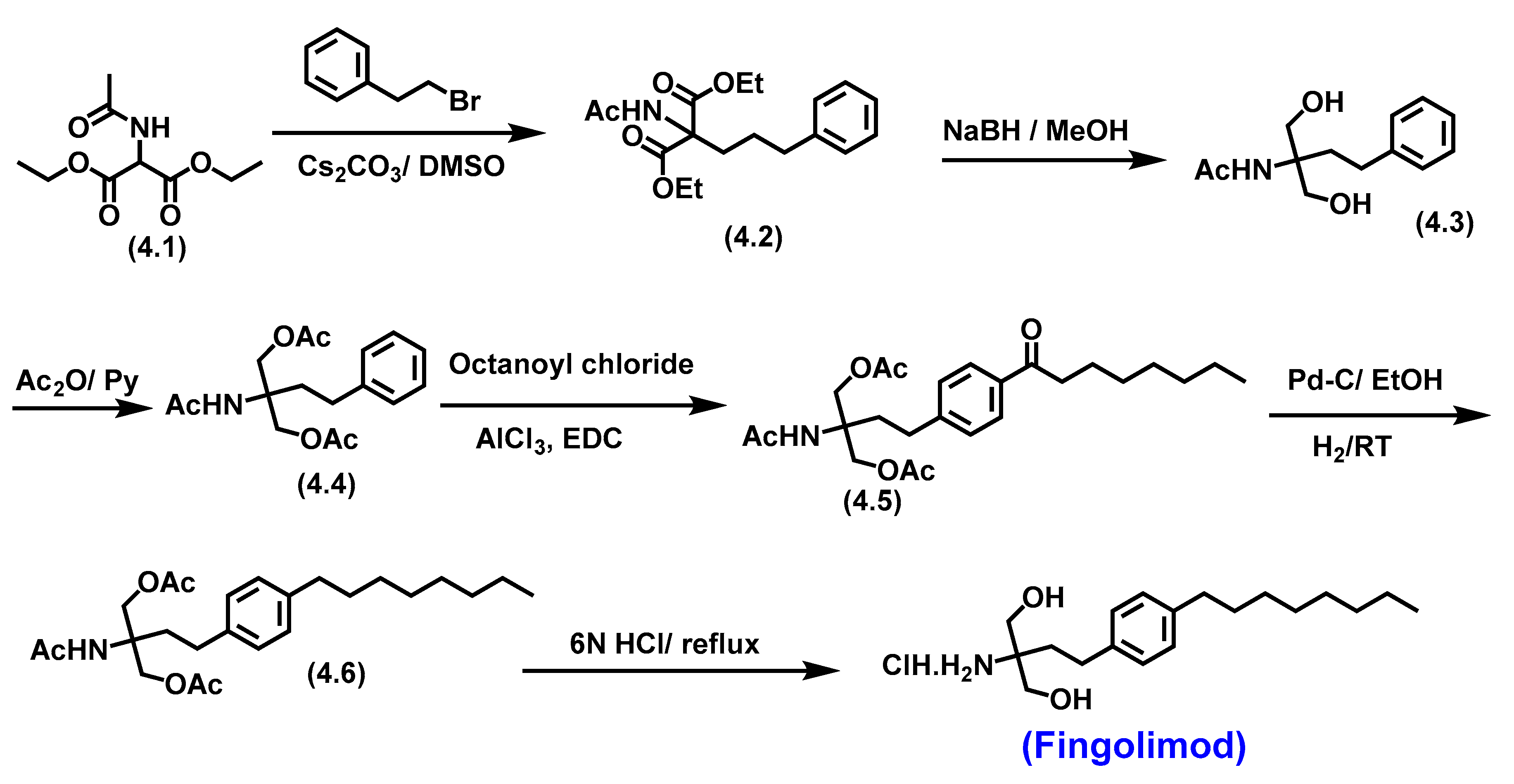
5.2. Fingolimod
5.2.1. Pharmacological significance of Fingolimod
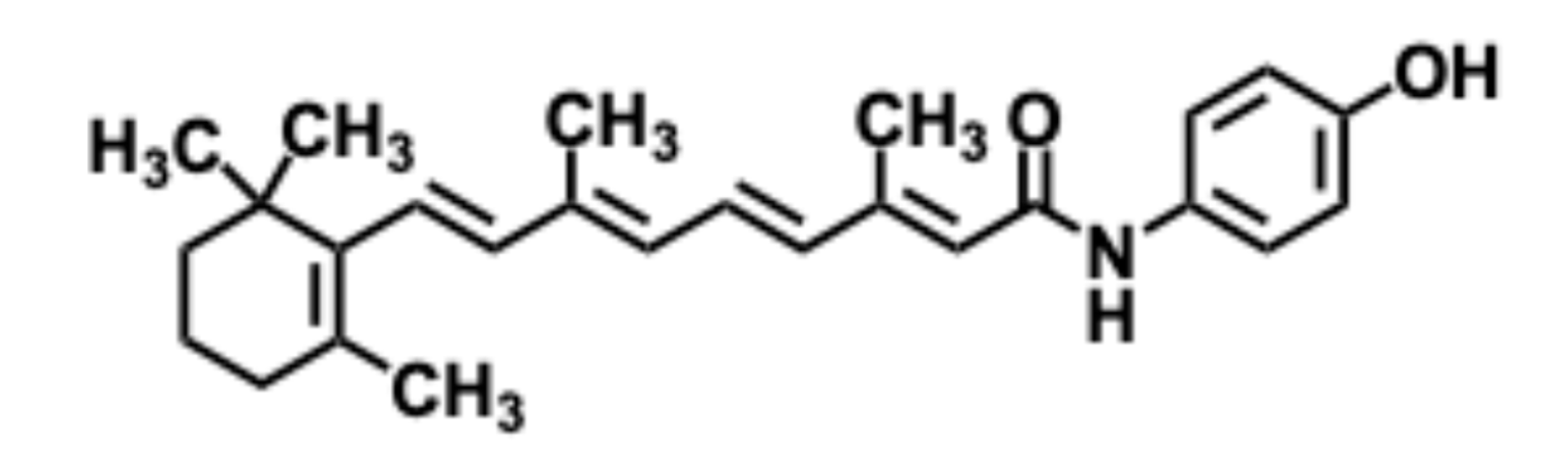
5.3. Fenretinide
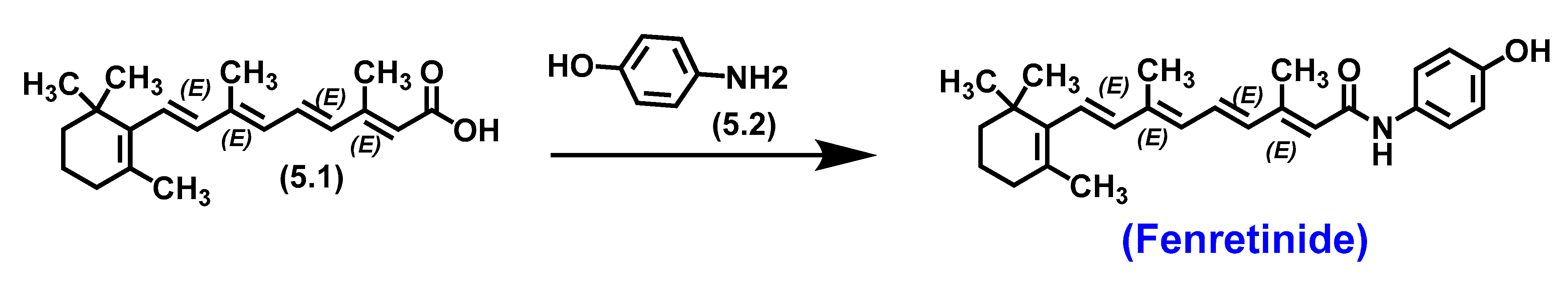
5.3.1. Pharmacological significance of Fenretinide
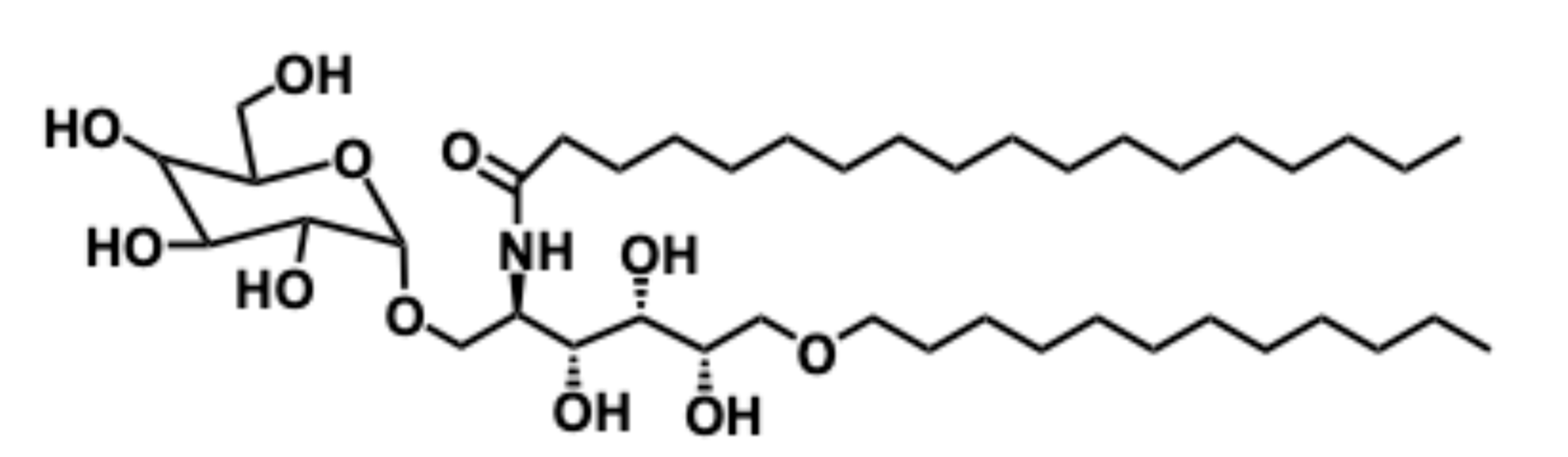
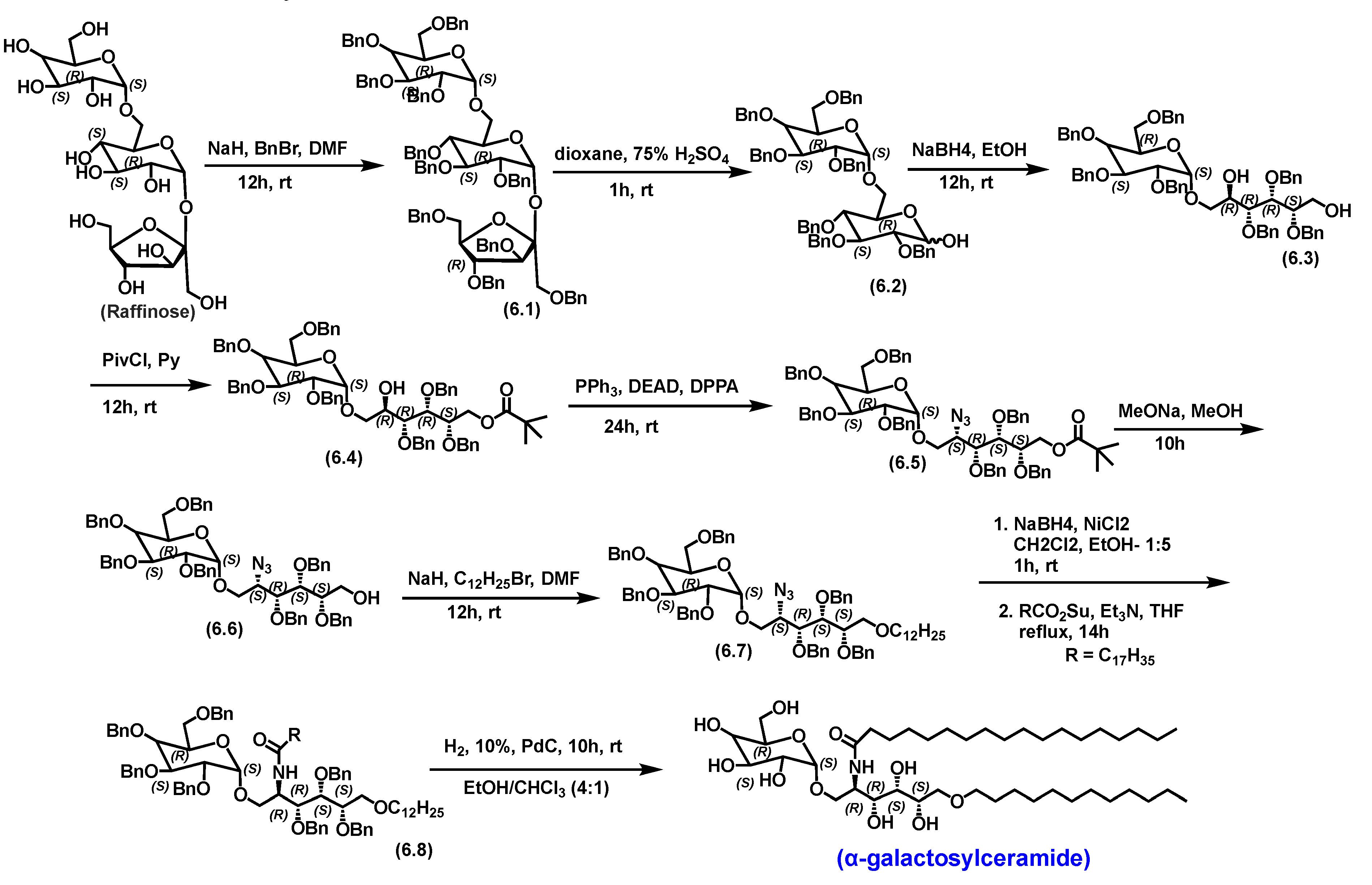
5.4.1. Pharmacological relevance and mode of action of α-galactosyl ceramide
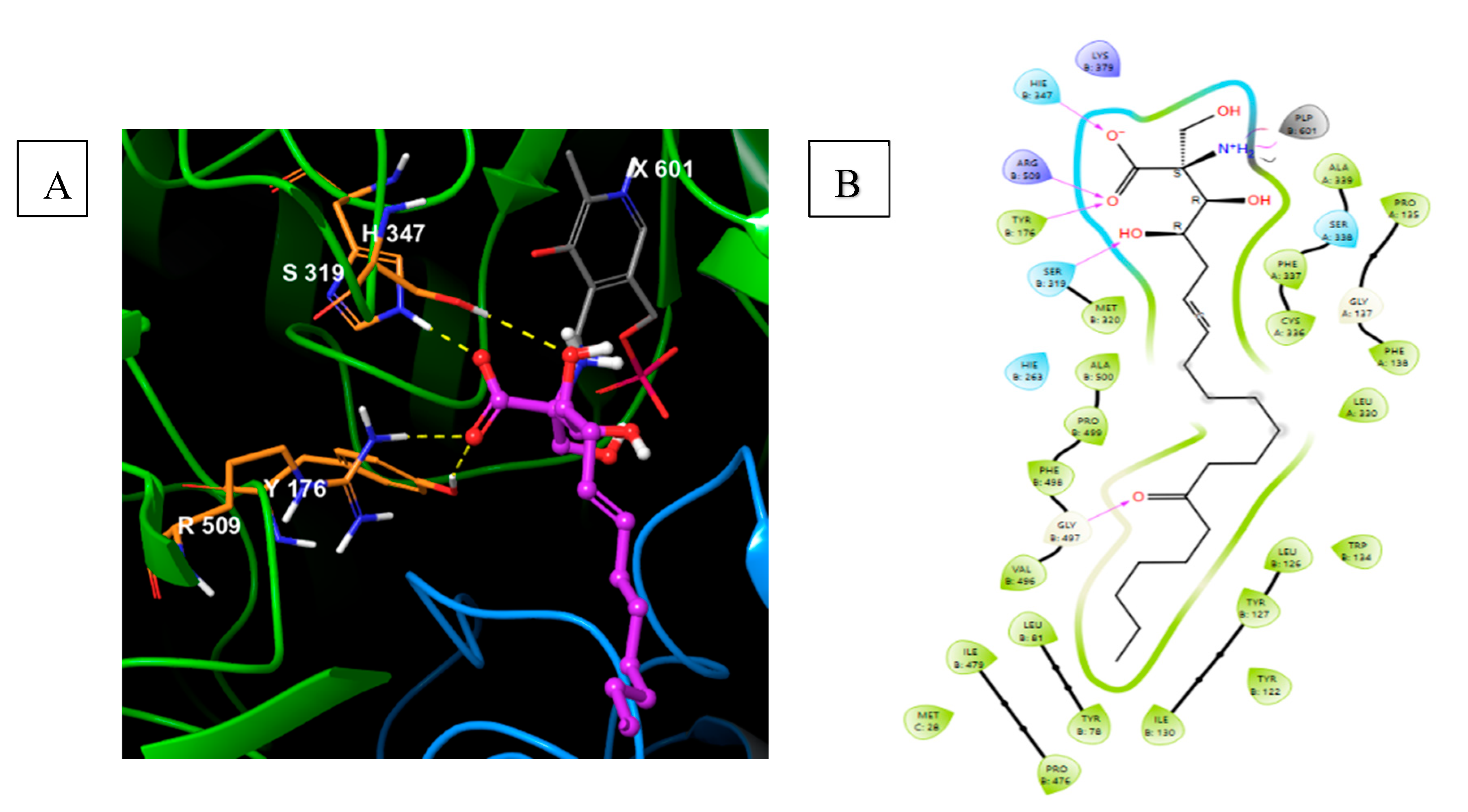
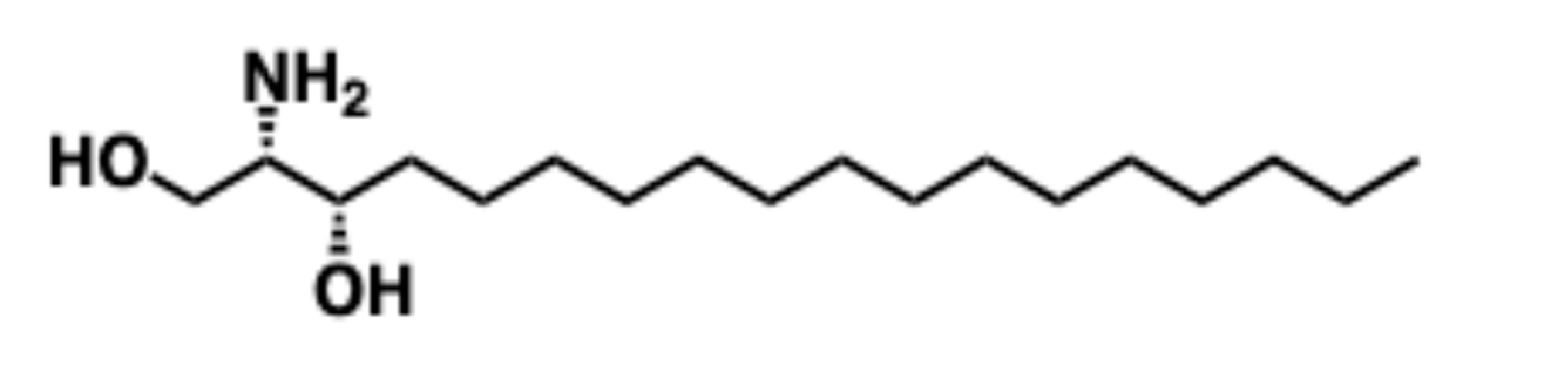
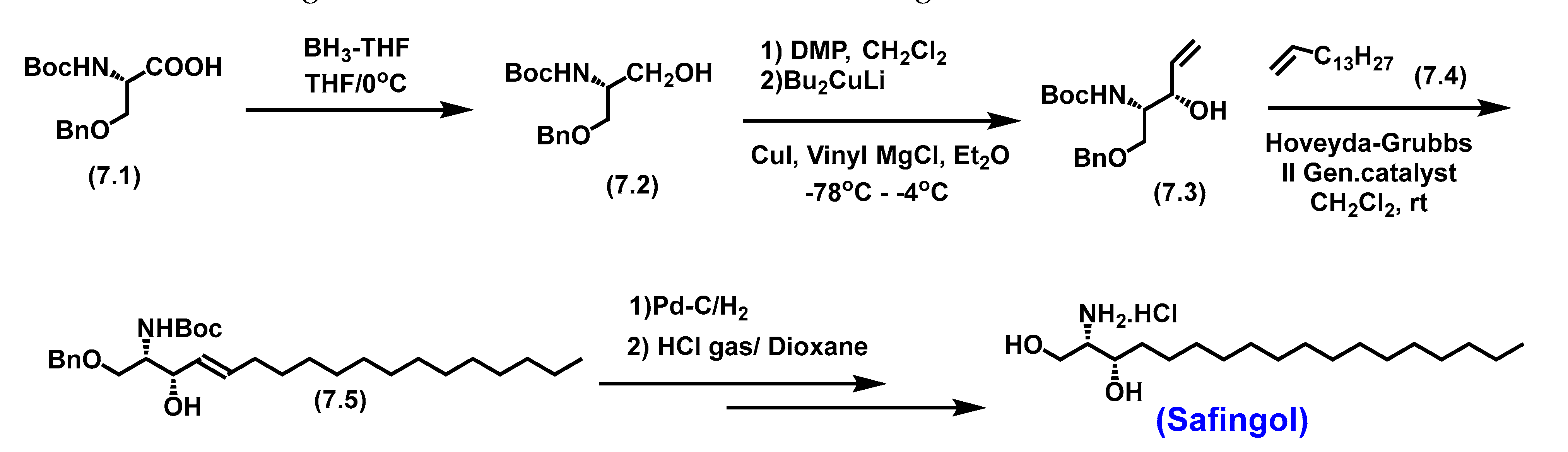
5.5. Safingol
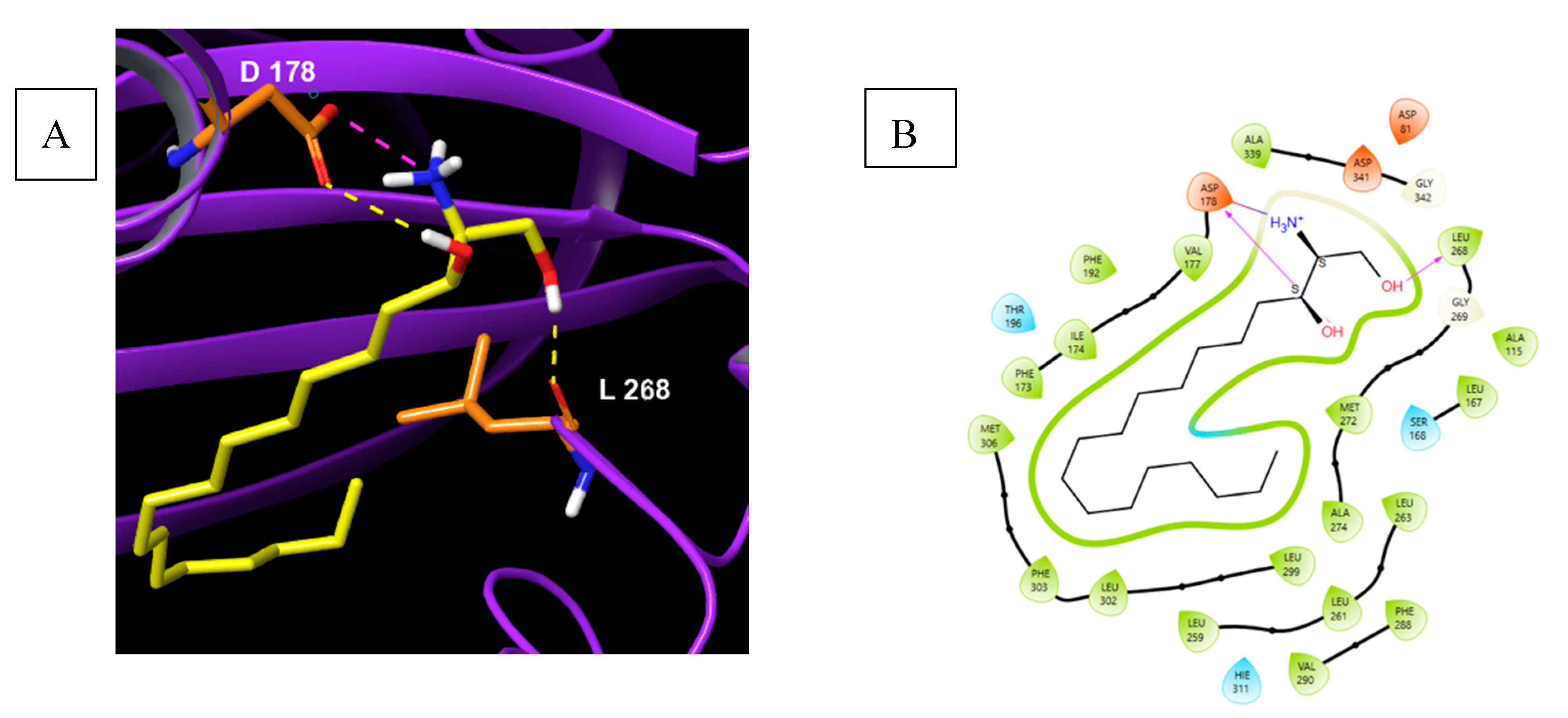
5.5.1. Pharmacological relevance and Mode of action of Safingol
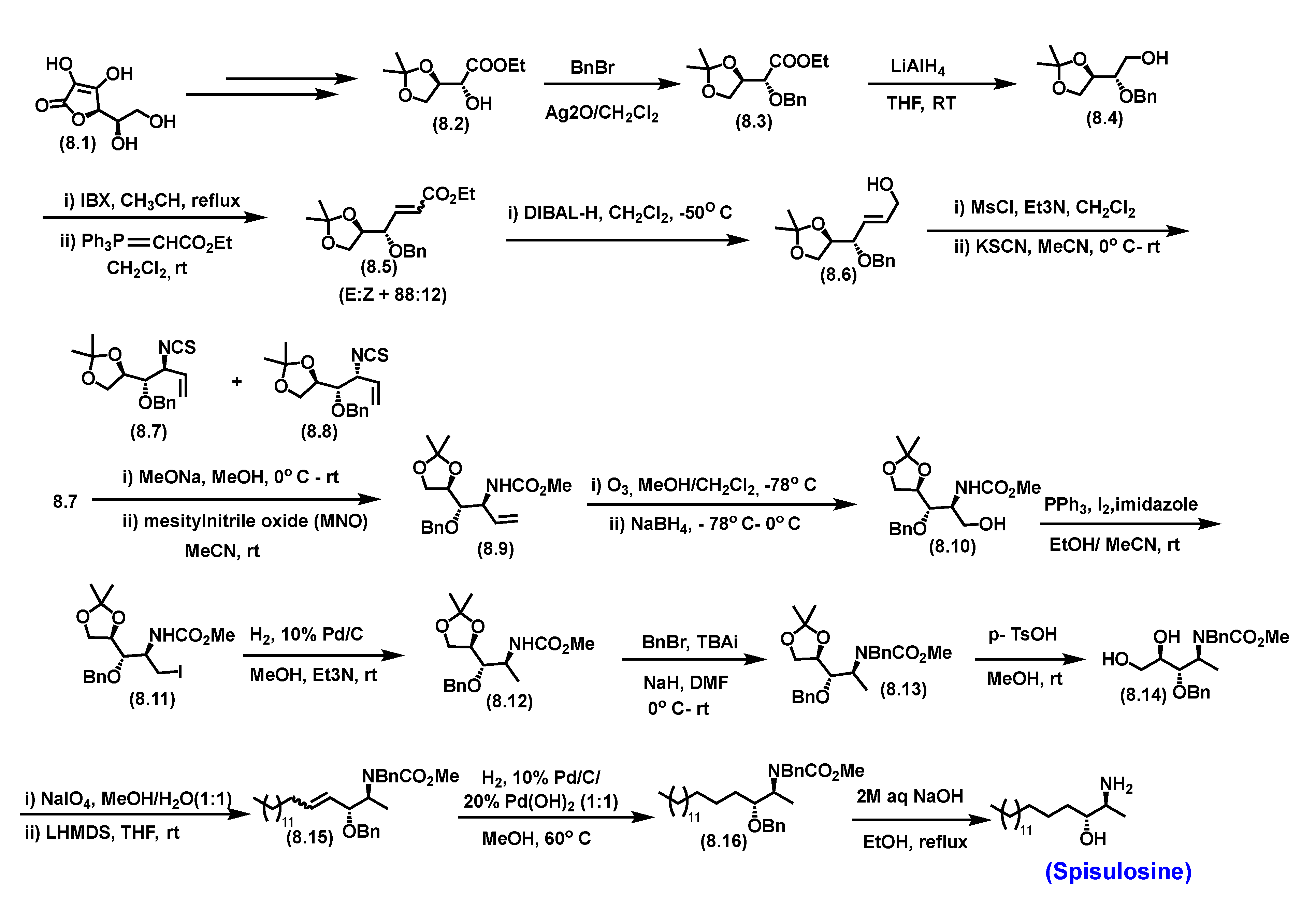
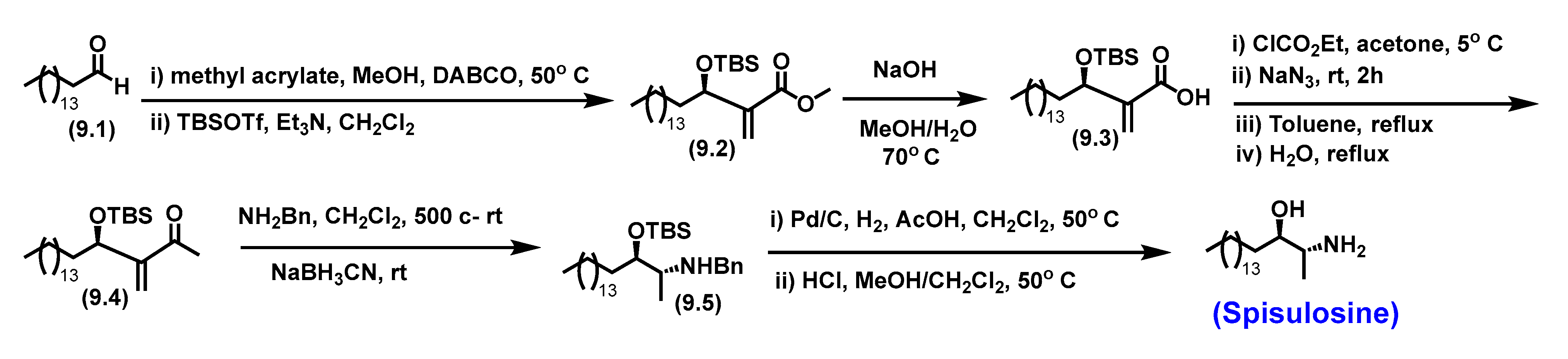
5.6. Spisulosine
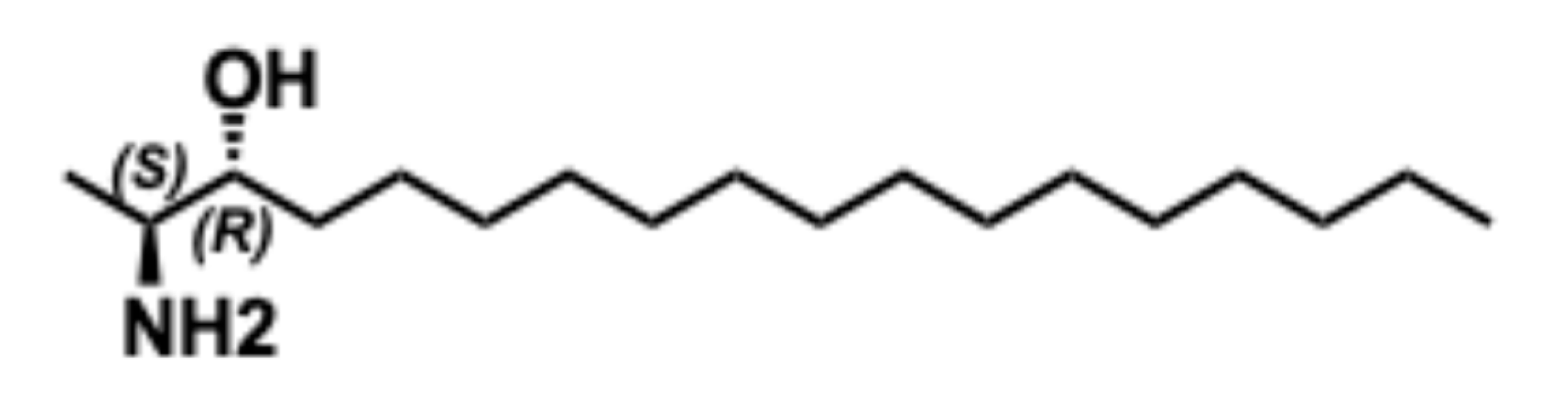
5.6.1. Synthesis of Spisulosine
5.6.2. Pharmacological relevance and mode of action of Spisulosine
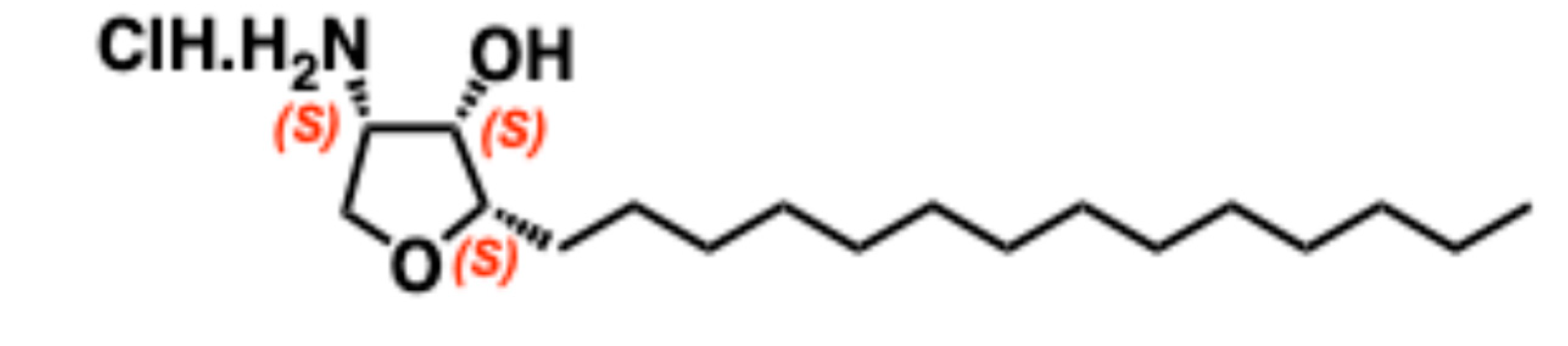
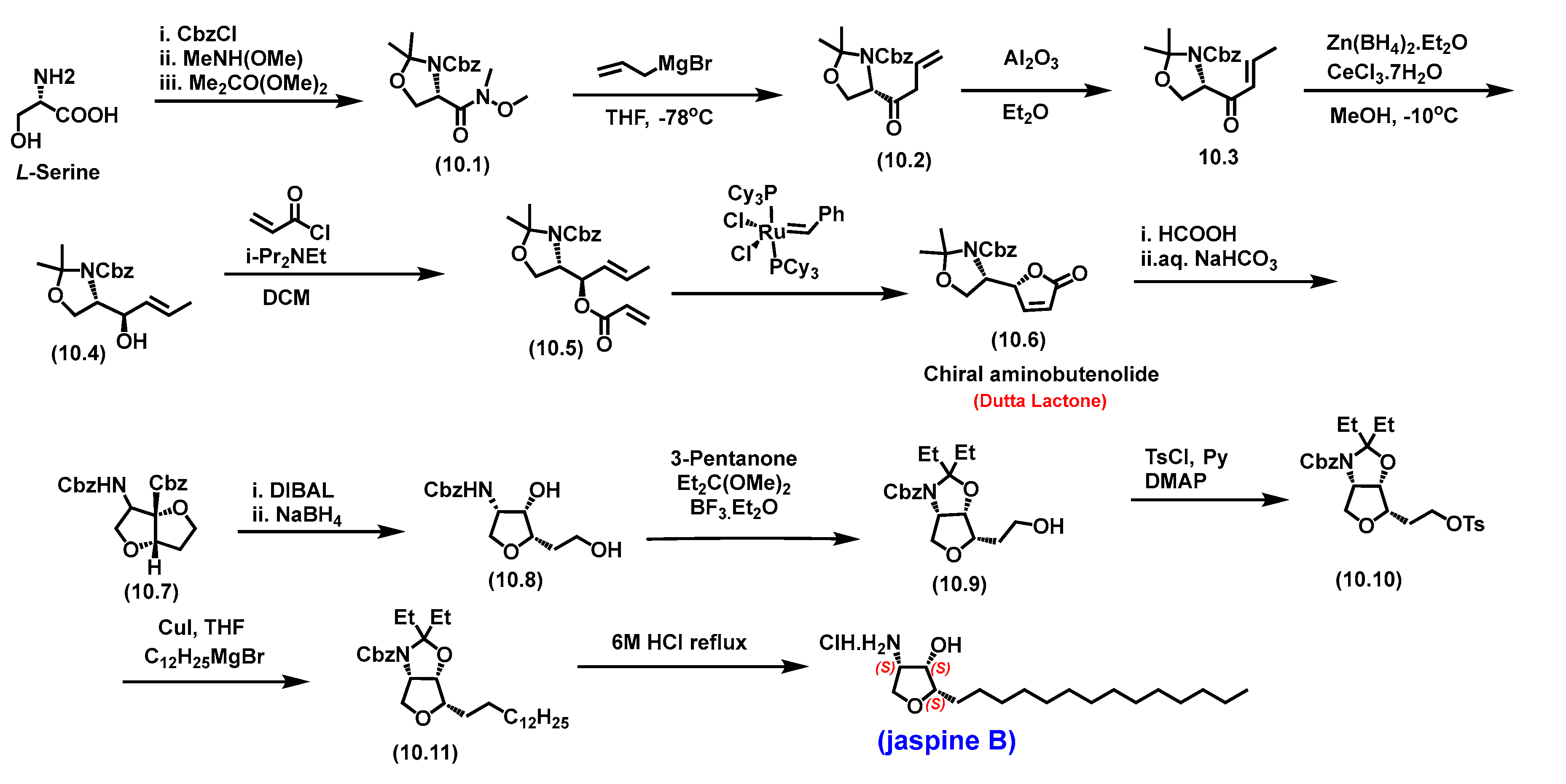
5.7. Jaspine B
5.7.1. Synthesis of Jaspine B
5.7.2. Pharmacological relevance and mode of action of Jaspine B
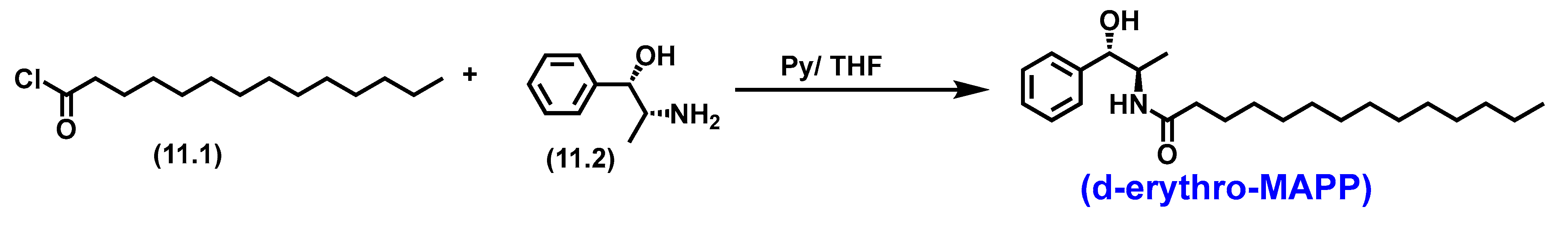
5.8.1. Pharmacological significance of d-erythro-MAPP
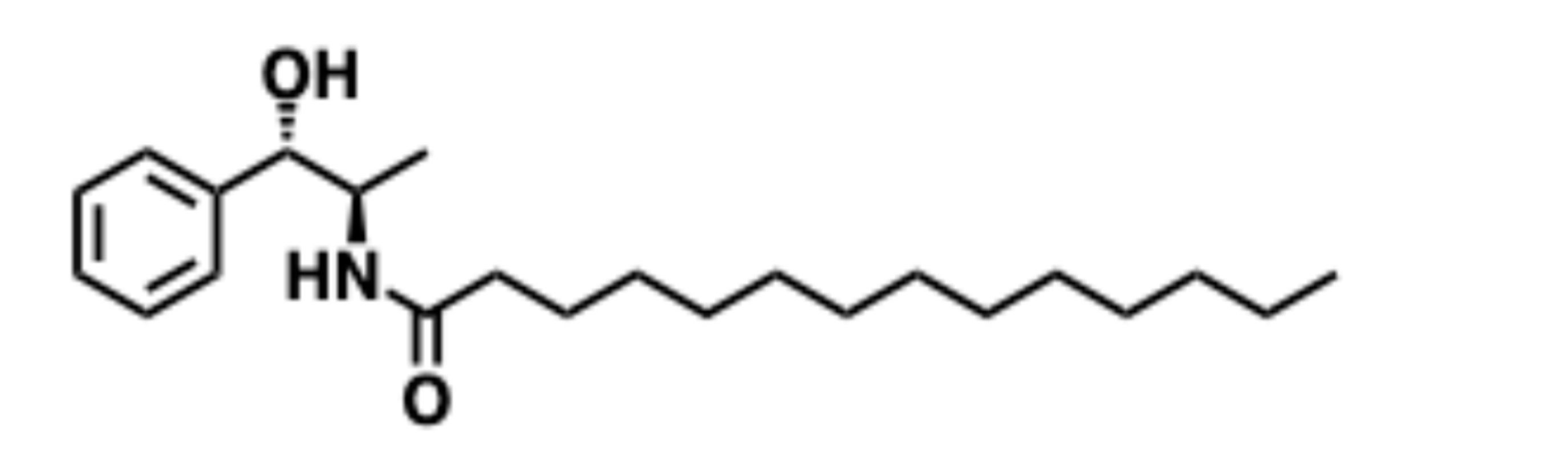
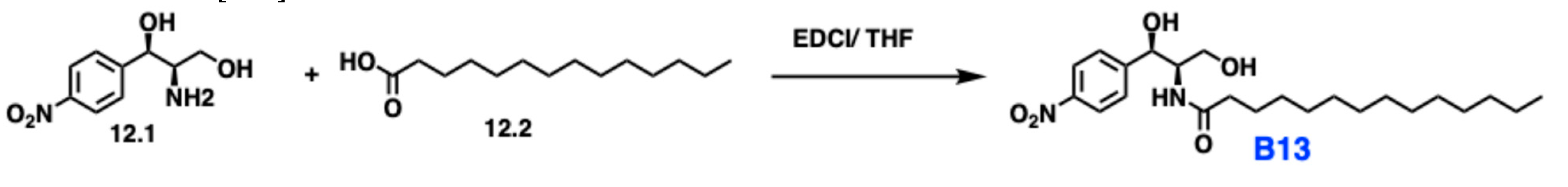
5.9. B13
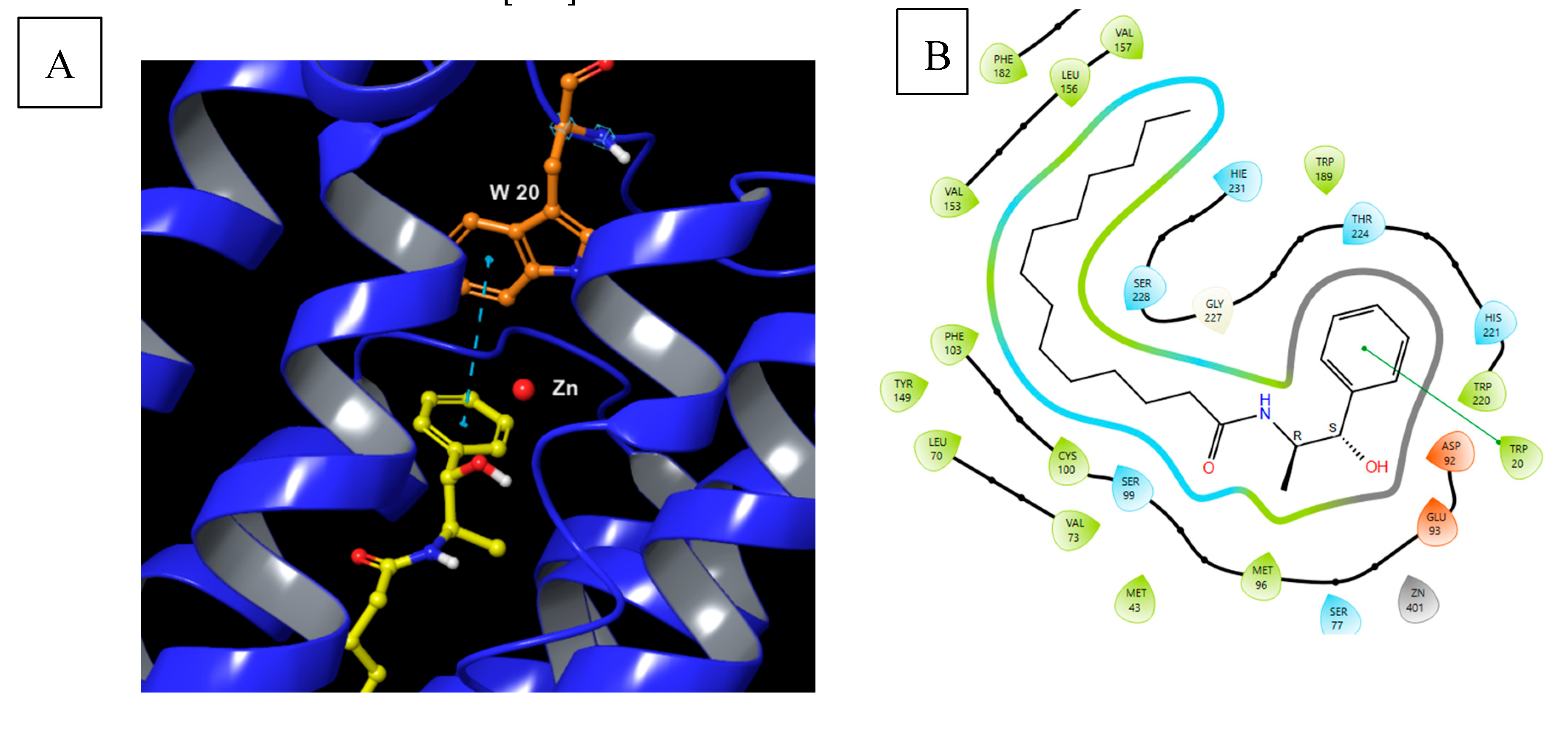
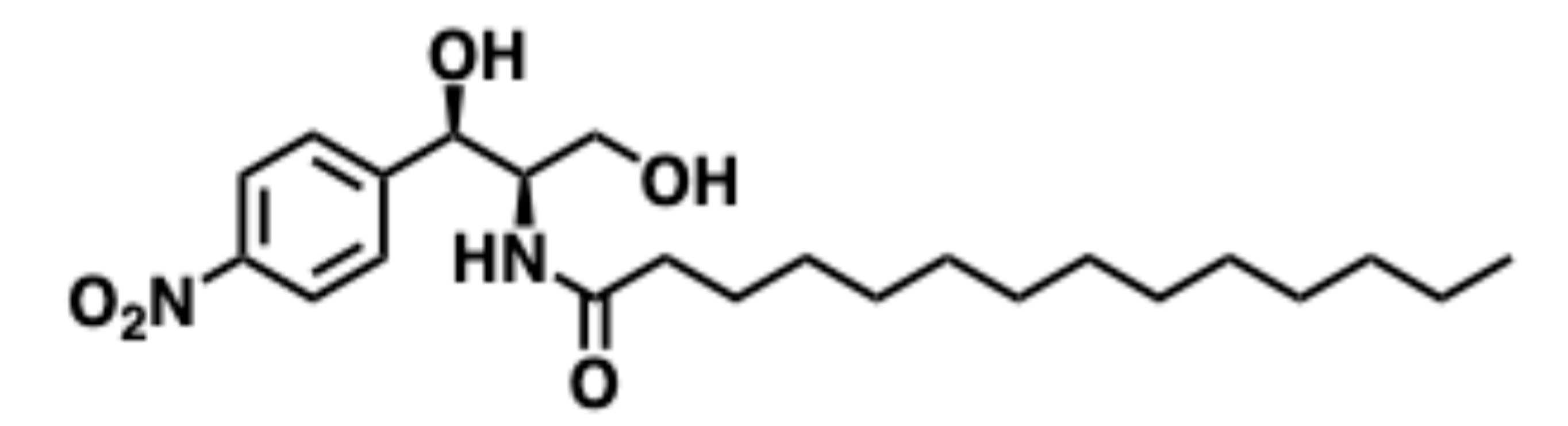
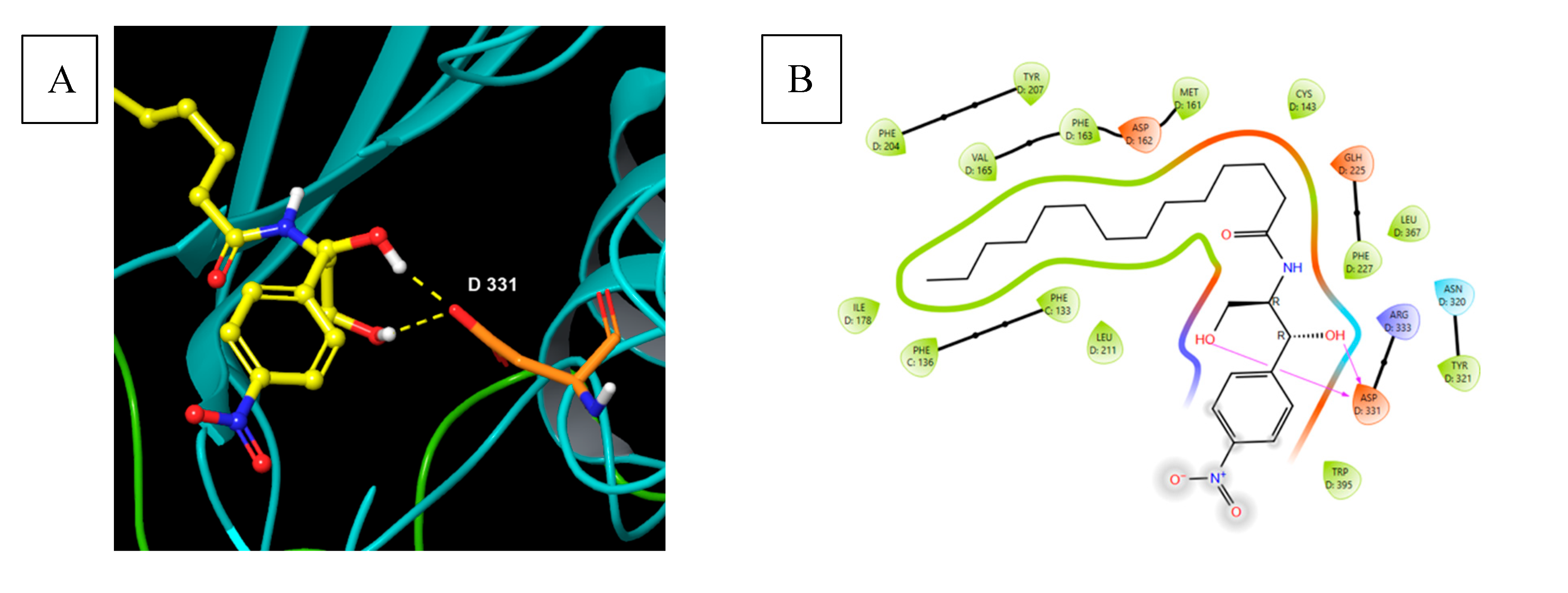
5.9.1. Pharmacological significance of B13
6. Discussion
Author Contributions
Funding
Acknowledgements
References
- Merrill, A.H., Jr.; Schmelz Em Fau - Dillehay, D.L.; Dillehay Dl Fau - Spiegel, S.; Spiegel S Fau - Shayman, J.A.; Shayman Ja Fau - Schroeder, J.J.; Schroeder Jj Fau - Riley, R.T.; Riley Rt Fau - Voss, K.A.; Voss Ka Fau - Wang, E.; Wang, E. Sphingolipids--the enigmatic lipid class: biochemistry, physiology, and pathophysiology.
- Schöffski, P.; Dumez, H.; Ruijter, R.; Miguel-Lillo, B.; Soto-Matos, A.; Alfaro, V.; Giaccone, G. Spisulosine (ES-285) given as a weekly three-hour intravenous infusion: results of a phase I dose-escalating study in patients with advanced solid malignancies. Cancer Chemotherapy and Pharmacology 2011, 68, 1397–1403. [Google Scholar] [CrossRef] [PubMed]
- Ogretmen, B. Sphingolipid metabolism in cancer signalling and therapy. (1474-1768 (Electronic)).
- Bouscary, A.; Quessada, C.; René, F.; Spedding, M.; Turner, B.J.; Henriques, A.; Ngo, S.T.; Loeffler, J.P. Sphingolipids metabolism alteration in the central nervous system: Amyotrophic lateral sclerosis (ALS) and other neurodegenerative diseases.
- Gault, C.R.; Obeid, L.M.; Hannun, Y.A. An Overview of Sphingolipid Metabolism: From Synthesis to Breakdown. In Sphingolipids as Signaling and Regulatory Molecules, Chalfant, C., Poeta, M.D., Eds.; Springer New York: New York, NY, 2010; pp. 1–23. [Google Scholar]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nature Reviews Molecular Cell Biology 2018, 19, 175–191. [Google Scholar] [CrossRef] [PubMed]
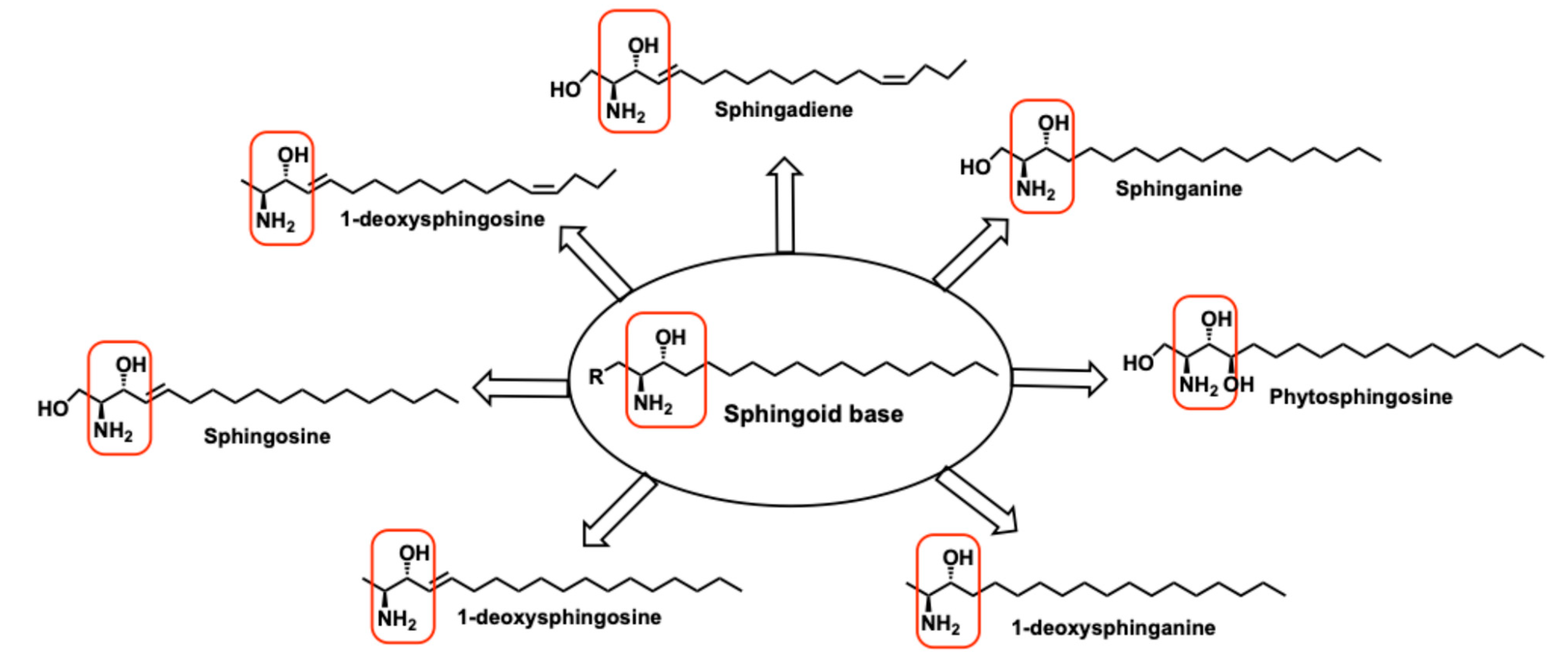
- Pruett, S.T.; Bushnev, A.; Hagedorn, K.; Adiga, M.; Haynes, C.A.; Sullards, M.C.; Liotta, D.C.; Merrill, A.H., Jr. Biodiversity of sphingoid bases ("sphingosines") and related amino alcohols. J Lipid Res 2008, 49, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.; Myers, S.; McGowan, C.; Henstridge, D.; Eri, R.; Sonda, S.; Caruso, V. 1-Deoxysphingolipids, Early Predictors of Type 2 Diabetes, Compromise the Functionality of Skeletal Myoblasts. Front Endocrinol (Lausanne) 2021, 12, 772925. [Google Scholar] [CrossRef]
- González-Ramírez, E.J.; García-Arribas, A.B.; Artetxe, I.; Shaw, W.A.; Goñi, F.M.; Alonso, A.; Jiménez-Rojo, N. (1-Deoxy)ceramides in bilayers containing sphingomyelin and cholesterol.
- Carreira, A.C.; Santos, T.C.; Lone, M.A.; Zupančič, E.; Lloyd-Evans, E.; de Almeida, R.F.M.; Hornemann, T.; Silva, L.C. Mammalian sphingoid bases: Biophysical, physiological and pathological properties. Progress in Lipid Research 2019, 75, 100988. [Google Scholar] [CrossRef]
- Stith, J.L.; Velazquez, F.N.; Obeid, L.M. Advances in determining signaling mechanisms of ceramide and role in disease. Journal of Lipid Research 2019, 60, 913–918. [Google Scholar] [CrossRef]
- Hernández-Corbacho, M.J.; Canals, D.; Adada, M.M.; Liu, M.; Senkal, C.E.; Yi, J.K.; Mao, C.; Luberto, C.; Hannun, Y.A.; Obeid, L.M. Tumor Necrosis Factor-α (TNFα)-induced Ceramide Generation via Ceramide Synthases Regulates Loss of Focal Adhesion Kinase (FAK) and Programmed Cell Death *. Journal of Biological Chemistry 2015, 290, 25356–25373. [Google Scholar] [CrossRef]
- Green, C.D.; Maceyka, M.; Cowart, L.A.; Spiegel, S. Sphingolipids in metabolic disease: The good, the bad, and the unknown.
- Quinville, B.M.; Deschenes, N.M.; Ryckman, A.E.; Walia, J.S. A Comprehensive Review: Sphingolipid Metabolism and Implications of Disruption in Sphingolipid Homeostasis. LID - 10.3390/ijms22115793 [doi] LID - 5793.
- Foran, D.A.-O.; Antoniades, C.; Akoumianakis, I. Emerging Roles for Sphingolipids in Cardiometabolic Disease: A Rational Therapeutic Target? LID - 10.3390/nu16193296 [doi] LID - 3296.
- Šakić, Z.; Atić, A.A.-O.; Potočki, S.; Bašić-Jukić, N.A.-O. Sphingolipids and Chronic Kidney Disease. LID - 10.3390/jcm13175050 [doi] LID - 5050.
- Doll, C.A.-O.; Snider, A.A.-O. The diverse roles of sphingolipids in inflammatory bowel disease.
- Vona, R.; Iessi, E.; Matarrese, P. Role of Cholesterol and Lipid Rafts in Cancer Signaling: A Promising Therapeutic Opportunity? Frontiers in Cell and Developmental Biology 2021, 9. [Google Scholar] [CrossRef]
- Dietrich, C.; Bagatolli, L.A.; Volovyk, Z.N.; Thompson, N.L.; Levi, M.; Jacobson, K.; Gratton, E. Lipid Rafts Reconstituted in Model Membranes. Biophysical Journal 2001, 80, 1417–1428. [Google Scholar] [CrossRef]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cellular Signalling 2008, 20, 1010–1018. [Google Scholar] [CrossRef]
- Mandon, E.C.; Ehses I Fau - Rother, J.; Rother J Fau - van Echten, G.; van Echten G Fau - Sandhoff, K.; Sandhoff, K. Subcellular localization and membrane topology of serine palmitoyltransferase, 3-dehydrosphinganine reductase, and sphinganine N-acyltransferase in mouse liver.
- Moskot, M.; Bocheńska, K.; Jakóbkiewicz-Banecka, J.; Banecki, B.; Gabig-Cimińska, M. Abnormal Sphingolipid World in Inflammation Specific for Lysosomal Storage Diseases and Skin Disorders. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Perry, D.K. Serine palmitoyltransferase: role in apoptotic de novo ceramide synthesis and other stress responses. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2002, 1585, 146–152. [Google Scholar] [CrossRef]
- Yard, B.A.; Carter, L.G.; Johnson, K.A.; Overton, I.M.; Dorward, M.; Liu, H.; McMahon, S.A.; Oke, M.; Puech, D.; Barton, G.J.; et al. The Structure of Serine Palmitoyltransferase; Gateway to Sphingolipid Biosynthesis. Journal of Molecular Biology 2007, 370, 870–886. [Google Scholar] [CrossRef] [PubMed]
- Zemski Berry, K.A.-O.X.; Garfield, A.A.-O.; Jambal, P.; Zarini, S.A.-O.; Perreault, L.A.-O.; Bergman, B.A.-O. Oxidised phosphatidylcholine induces sarcolemmal ceramide accumulation and insulin resistance in skeletal muscle.
- Tahia, F.; Ma, D.A.-O.; Stephenson, D.J.; Basu, S.K.; Del Mar, N.A.; Lenchik, N.; Kochat, H.; Brown, K.; Chalfant, C.A.-O.; Mandal, N.A.-O. Inhibiting De Novo Biosynthesis of Ceramide by L-Cycloserine Can Prevent Light-Induced Retinal Degeneration in Albino BALB/c Mice. LID - 13389 [pii] LID - 10.3390/ijms252413389 [doi].
- Hengst, J.A.; Ruiz-Velasco, V.J.; Raup-Konsavage, W.A.-O.; Vrana, K.A.-O.; Yun, J.A.-O. Cannabinoid-Induced Immunogenic Cell Death of Colorectal Cancer Cells Through De Novo Synthesis of Ceramide Is Partially Mediated by CB2 Receptor. LID - 10.3390/cancers16233973 [doi] LID - 3973.
- Jenkins, G.M.; Cowart, L.A.; Signorelli, P.; Pettus, B.J.; Chalfant, C.E.; Hannun, Y.A. Acute Activation of de Novo Sphingolipid Biosynthesis upon Heat Shock Causes an Accumulation of Ceramide and Subsequent Dephosphorylation of SR Proteins*. Journal of Biological Chemistry 2002, 277, 42572–42578. [Google Scholar] [CrossRef]
- Jamjoum, R.; Majumder, S.; Issleny, B.; Stiban, J. Mysterious sphingolipids: metabolic interrelationships at the center of pathophysiology. Front Physiol 2023, 14, 1229108. [Google Scholar] [CrossRef] [PubMed]
- Thurm, A.; Chlebowski, C.; Joseph, L.; Farmer, C.; Adedipe, D.; Weiss, M.; Wiggs, E.; Farhat, N.; Bianconi, S.; Berry-Kravis, E.; et al. Neurodevelopmental Characterization of Young Children Diagnosed with Niemann-Pick Disease, Type C1. Journal of Developmental & Behavioral Pediatrics 2020, 41. [Google Scholar]
- Kim, M.Y.; Linardic, C.; Obeid, L.; Hannun, Y. Identification of sphingomyelin turnover as an effector mechanism for the action of tumor necrosis factor alpha and gamma-interferon. Specific role in cell differentiation. Journal of Biological Chemistry 1991, 266, 484–489. [Google Scholar] [CrossRef]
- Yi, J.; Qi, B.; Yin, J.; Li, R.; Chen, X.; Hu, J.; Li, G.; Zhang, S.; Zhang, Y.; Yang, M. Molecular basis for the catalytic mechanism of human neutral sphingomyelinases 1 (hSMPD2). Nature Communications 2023, 14, 7755. [Google Scholar] [CrossRef]
- Tomiuk, S.; Hofmann K Fau - Nix, M.; Nix M Fau - Zumbansen, M.; Zumbansen M Fau - Stoffel, W.; Stoffel, W. Cloned mammalian neutral sphingomyelinase: functions in sphingolipid signaling?
- Monturiol-Gross, L.; Villalta-Romero, F.; Flores-Díaz, M.; Alape-Girón, A. Bacterial phospholipases C with dual activity: phosphatidylcholinesterase and sphingomyelinase. FEBS Open Bio 2021, 11, 3262–3275. [Google Scholar] [CrossRef]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell Signal 2008, 20, 1010–1018. [Google Scholar] [CrossRef]
- Bikman, B.T.; Summers, S.A. Ceramides as modulators of cellular and whole-body metabolism.
- Maceyka, M.; Spiegel, S. Sphingolipid metabolites in inflammatory disease. Nature 2014, 510, 58–67. [Google Scholar] [CrossRef]
- Zaibaq, F.A.-O.; Dowdy, T.; Larion, M.A.-O. Targeting the Sphingolipid Rheostat in Gliomas. LID - 10.3390/ijms23169255 [doi] LID - 9255.
- Karmelić, I.; Jurilj Sajko, M.; Sajko, T.; Rotim, K.; Fabris, D. The role of sphingolipid rheostat in the adult-type diffuse glioma pathogenesis.
- Moro, K.; Ichikawa, H.; Koyama, Y.; Abe, S.; Uchida, H.; Naruse, K.; Obata, Y.; Tsuchida, J.; Toshikawa, C.; Ikarashi, M.; et al. Oral Administration of Glucosylceramide Suppresses Tumor Growth by Affecting the Ceramide/Sphingosine-1-Phosphate Balance in Breast Cancer Tissue.
- Piccoli, M.A.-O.; Cirillo, F.A.-O.; Ghiroldi, A.A.-O.; Rota, P.A.-O.; Coviello, S.; Tarantino, A.A.-O.X.; La Rocca, P.A.-O.; Lavota, I.; Creo, P.; Signorelli, P.A.-O.; et al. Sphingolipids and Atherosclerosis: The Dual Role of Ceramide and Sphingosine-1-Phosphate. LID - 10.3390/antiox12010143 [doi] LID - 143.
- Li, R.Z.; Wang, X.R.; Wang, J.; Xie, C.; Wang, X.X.; Pan, H.D.; Meng, W.Y.; Liang, T.L.; Li, J.X.; Yan, P.Y.; et al. The key role of sphingolipid metabolism in cancer: New therapeutic targets, diagnostic and prognostic values, and anti-tumor immunotherapy resistance. Front Oncol 2022, 12, 941643. [Google Scholar] [CrossRef] [PubMed]
- Wajapeyee, N.; Beamon, T.C.; Gupta, R. Roles and therapeutic targeting of ceramide metabolism in cancer. Molecular Metabolism 2024, 83, 101936. [Google Scholar] [CrossRef]
- Santos, W.L.; Lynch, K.R. Drugging sphingosine kinases.
- O'Brien, M.A.; Kirby, R. Apoptosis: A review of pro-apoptotic and anti-apoptotic pathways and dysregulation in disease. Journal of Veterinary Emergency and Critical Care 2008, 18, 572–585. [Google Scholar] [CrossRef]
- Hait, N.C.; Oskeritzian, C.A.; Paugh, S.W.; Milstien, S.; Spiegel, S. Sphingosine kinases, sphingosine 1-phosphate, apoptosis and diseases. Biochimica et Biophysica Acta (BBA) - Biomembranes 2006, 1758, 2016–2026. [Google Scholar] [CrossRef]
- Chakraborty, P.; Vaena, S.G.; Thyagarajan, K.; Chatterjee, S.; Al-Khami, A.; Selvam, S.P.; Nguyen, H.; Kang, I.; Wyatt, M.W.; Baliga, U.; et al. Pro-Survival Lipid Sphingosine-1-Phosphate Metabolically Programs T Cells to Limit Anti-tumor Activity. Cell Reports 2019, 28, 1879–1893.e1877. [Google Scholar] [CrossRef] [PubMed]
- Giussani, P.; Tringali, C.; Riboni, L.; Viani, P.; Venerando, B. Sphingolipids: Key Regulators of Apoptosis and Pivotal Players in Cancer Drug Resistance. International Journal of Molecular Sciences 2014, 15, 4356–4392. [Google Scholar] [CrossRef]
- Van Brocklyn, J.R.; Williams, J.B. The control of the balance between ceramide and sphingosine-1-phosphate by sphingosine kinase: Oxidative stress and the seesaw of cell survival and death. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology 2012, 163, 26–36. [Google Scholar] [CrossRef]
- Torres, O.; Matute, J.; Gelineau-van Waes, J.; Maddox, J.R.; Gregory, S.G.; Ashley-Koch, A.E.; Showker, J.L.; Voss, K.A.; Riley, R.T. Human health implications from co-exposure to aflatoxins and fumonisins in maize-based foods in Latin America: Guatemala as a case study. World Mycotoxin Journal 2015, 8, 143–160. [Google Scholar] [CrossRef]
- Oskouian, B.; Saba, J.D. Cancer Treatment Strategies Targeting Sphingolipid Metabolism. In Sphingolipids as Signaling and Regulatory Molecules, Chalfant, C., Poeta, M.D., Eds.; Springer New York: New York, NY, 2010; pp. 185–205. [Google Scholar]
- Zaibaq, F.; Dowdy, T.; Larion, M. Targeting the Sphingolipid Rheostat in Gliomas. International Journal of Molecular Sciences 2022, 23. [Google Scholar] [CrossRef]
- Lee, Y.S.; Choi, K.M.; Choi, M.H.; Ji, S.Y.; Lee, S.; Sin, D.M.; Oh, K.W.; Lee, Y.M.; Hong, J.T.; Yun, Y.P.; et al. Serine palmitoyltransferase inhibitor myriocin induces growth inhibition of B16F10 melanoma cells through G2/M phase arrest. Cell Proliferation 2011, 44, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Hada, N.; Katsume, A.; Kenichi, K.; Endo, C.; Horiba, N.; Sudoh, M. Novel oral SPT inhibitor CH5169356 inhibits hepatic stellate cell activation and ameliorates hepatic fibrosis in mouse models of non-alcoholic steatohepatitis (NASH). Pharmacology Research & Perspectives 2023, 11, e01094. [Google Scholar] [CrossRef]
- Tahia, F.; Basu, S.K.; Prislovsky, A.; Mondal, K.; Ma, D.; Kochat, H.; Brown, K.; Stephenson, D.J.; Chalfant, C.E.; Mandal, N. Sphingolipid biosynthetic inhibitor L-Cycloserine prevents oxidative-stress-mediated death in an in vitro model of photoreceptor-derived 661W cells. Experimental Eye Research 2024, 242, 109852. [Google Scholar] [CrossRef]
- De Vita, T.; Albani, C.; Realini, N.; Migliore, M.; Basit, A.; Ottonello, G.; Cavalli, A. Inhibition of Serine Palmitoyltransferase by a Small Organic Molecule Promotes Neuronal Survival after Astrocyte Amyloid Beta 1–42 Injury. ACS Chemical Neuroscience 2019, 10, 1627–1635. [Google Scholar] [CrossRef]
- Lowther, J.; Beattie, A.E.; Langridge-Smith, P.R.R.; Clarke, D.J.; Campopiano, D.J. l-Penicillamine is a mechanism-based inhibitor of serine palmitoyltransferase by forming a pyridoxal-5′-phosphate-thiazolidine adduct. MedChemComm 2012, 3, 1003–1008. [Google Scholar] [CrossRef]
- Hanada, K.; Nishijima, M.; Fujita, T.; Kobayashi, S. Specificity of inhibitors of serine palmitoyltransferase (SPT), a key enzyme in sphingolipid biosynthesis, in intact cells: A novel evaluation system using an SPT-defective mammalian cell mutant. Biochemical Pharmacology 2000, 59, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Ji, C.; Zhou, Y.; Huang, W.; Ni, W.; Tong, X.; Wei, J.-F. Sphingosine kinase inhibitors: A patent review. Int J Mol Med 2018, 41, 2450–2460. [Google Scholar] [CrossRef] [PubMed]
- Companioni, O.; Mir, C.; Garcia-Mayea, Y.; ME, L.L. Targeting Sphingolipids for Cancer Therapy.
- Adams, D.R.; Pyne, S.; Pyne, N.J. Structure-function analysis of lipid substrates and inhibitors of sphingosine kinases. Cellular Signalling 2020, 76, 109806. [Google Scholar] [CrossRef] [PubMed]
- Bonica, J.; Clarke, C.J.; Obeid, L.M.; Luberto, C.; Hannun, Y.A. Upregulation of sphingosine kinase 1 in response to doxorubicin generates an angiogenic response via stabilization of Snail. The FASEB Journal 2023, 37, e22787. [Google Scholar] [CrossRef]
- Hammer, S.; Sauer, B.; Spika, I.; Schraut, C.; Kleuser, B.; Schäfer-Korting, M. Glucocorticoids mediate differential anti-apoptotic effects in human fibroblasts and keratinocytes via sphingosine-1-phosphate formation. Journal of Cellular Biochemistry 2004, 91, 840–851. [Google Scholar] [CrossRef]
- Hara-Yokoyama, M.; Terasawa, K.; Ichinose, S.; Watanabe, A.; Podyma-Inoue, K.A.; Akiyoshi, K.; Igarashi, Y.; Yanagishita, M. Sphingosine kinase 2 inhibitor SG-12 induces apoptosis via phosphorylation by sphingosine kinase 2. Bioorganic & Medicinal Chemistry Letters 2013, 23, 2220–2224. [Google Scholar] [CrossRef]
- Britten, C.D.; Garrett-Mayer, E.; Chin, S.H.; Shirai, K.; Ogretmen, B.; Bentz, T.A.; Brisendine, A.; Anderton, K.; Cusack, S.L.; Maines, L.W.; et al. A Phase I Study of ABC294640, a First-in-Class Sphingosine Kinase-2 Inhibitor, in Patients with Advanced Solid Tumors. Clinical Cancer Research 2017, 23, 4642–4650. [Google Scholar] [CrossRef]
- Yi, X.; Tang, X.; Li, T.; Chen, L.; He, H.; Wu, X.; Xiang, C.; Cao, M.; Wang, Z.; Wang, Y.; et al. Therapeutic potential of the sphingosine kinase 1 inhibitor, PF-543. Biomedicine & Pharmacotherapy 2023, 163, 114401. [Google Scholar] [CrossRef]
- Gehin, M.; Melchior, M.; Welford, R.W.D.; Sidharta, P.N.; Dingemanse, J. Assessment of Target Engagement in a First-in-Human Trial with Sinbaglustat, an Iminosugar to Treat Lysosomal Storage Disorders. Clinical and Translational Science 2021, 14, 558–567. [Google Scholar] [CrossRef]
- Dong, G.; Deng, Y.; Yasgar, A.; Yadav, R.; Talley, D.; Zakharov, A.V.; Jain, S.; Rai, G.; Noinaj, N.; Simeonov, A.; et al. Venglustat Inhibits Protein N-Terminal Methyltransferase 1 in a Substrate-Competitive Manner. Journal of Medicinal Chemistry 2022, 65, 12334–12345. [Google Scholar] [CrossRef]
- Shayman, J.A.; Lee, L.; Abe, A.; Shu, L. [38] Inhibitors of glucosylceramide synthase. In Methods in Enzymology; Academic Press: 2000; Volume 311, pp. 373–387.
- Stocker, B.L.; Dangerfield, E.M.; Win-Mason, A.L.; Haslett, G.W.; Timmer, M.S.M. Recent Developments in the Synthesis of Pyrrolidine-Containing Iminosugars. European Journal of Organic Chemistry 2010, 2010, 1615–1637. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, W.; Sun, J.; Huang, Y.; Wang, P.; Venkataramanan, R.; Yang, D.; Ma, X.; Rana, A.; Li, S. Novel glucosylceramide synthase inhibitor based prodrug copolymer micelles for delivery of anticancer agents. Journal of Controlled Release 2018, 288, 212–226. [Google Scholar] [CrossRef]
- Tanaka, Y.; Seto, M.; Kakegawa, K.; Takami, K.; Kikuchi, F.; Yamamoto, T.; Nakamura, M.; Daini, M.; Murakami, M.; Ohashi, T.; et al. Discovery of Brain-Penetrant Glucosylceramide Synthase Inhibitors with a Novel Pharmacophore. Journal of Medicinal Chemistry 2022, 65, 4270–4290. [Google Scholar] [CrossRef] [PubMed]
- Delgado, A.; Casas, J.; Llebaria, A.; Abad, J.L.; Fabriás, G. Chemical Tools to Investigate Sphingolipid Metabolism and Functions. ChemMedChem 2007, 2, 580–606. [Google Scholar] [CrossRef]
- Aerts, J.M.; Ottenhoff, R.; Powlson, A.S.; Grefhorst, A.; van Eijk, M.; Dubbelhuis, P.F.; Aten, J.; Kuipers, F.; Serlie, M.J.; Wennekes, T.; et al. Pharmacological Inhibition of Glucosylceramide Synthase Enhances Insulin Sensitivity. Diabetes 2007, 56, 1341–1349. [Google Scholar] [CrossRef]
- Artola, M.; Kuo, C.-L.; Lelieveld, L.T.; Rowland, R.J.; van der Marel, G.A.; Codée, J.D.C.; Boot, R.G.; Davies, G.J.; Aerts, J.M.F.G.; Overkleeft, H.S. Functionalized Cyclophellitols Are Selective Glucocerebrosidase Inhibitors and Induce a Bona Fide Neuropathic Gaucher Model in Zebrafish. Journal of the American Chemical Society 2019, 141, 4214–4218. [Google Scholar] [CrossRef] [PubMed]
- Rempel, B.P.; Withers, S.G. Covalent inhibitors of glycosidases and their applications in biochemistry and biology. Glycobiology 2008, 18, 570–586. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-L.; Kallemeijn, W.W.; Lelieveld, L.T.; Mirzaian, M.; Zoutendijk, I.; Vardi, A.; Futerman, A.H.; Meijer, A.H.; Spaink, H.P.; Overkleeft, H.S.; et al. In vivo inactivation of glycosidases by conduritol B epoxide and cyclophellitol as revealed by activity-based protein profiling. The FEBS Journal 2019, 286, 584–600. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Oyama, M.; Iwai, T.; Tanabe, M. Glucosylceramide synthase inhibitor ameliorates chronic inflammatory pain. Journal of Pharmacological Sciences 2024, 156, 235–238. [Google Scholar] [CrossRef]
- Othman, M.A.; Yuyama, K.; Murai, Y.; Igarashi, Y.; Mikami, D.; Sivasothy, Y.; Awang, K.; Monde, K. Malabaricone C as Natural Sphingomyelin Synthase Inhibitor against Diet-Induced Obesity and Its Lipid Metabolism in Mice. ACS Medicinal Chemistry Letters 2019, 10, 1154–1158. [Google Scholar] [CrossRef]
- Meng, A.; Luberto, C.; Meier, P.; Bai, A.; Yang, X.; Hannun, Y.A.; Zhou, D. Sphingomyelin synthase as a potential target for D609-induced apoptosis in U937 human monocytic leukemia cells. Experimental Cell Research 2004, 292, 385–392. [Google Scholar] [CrossRef]
- Deng, X.; Lin, F.; Zhang, Y.; Li, Y.; Zhou, L.; Lou, B.; Li, Y.; Dong, J.; Ding, T.; Jiang, X.; et al. Identification of small molecule sphingomyelin synthase inhibitors. European Journal of Medicinal Chemistry 2014, 73, 1–7. [Google Scholar] [CrossRef]
- Lou, B.; Dong, J.; Li, Y.; Ding, T.; Bi, T.; Li, Y.; Deng, X.; Ye, D.; Jiang, X.-C. Pharmacologic Inhibition of Sphingomyelin Synthase (SMS) Activity Reduces Apolipoprotein-B Secretion from Hepatocytes and Attenuates Endotoxin-Mediated Macrophage Inflammation. PLOS ONE 2014, 9, e102641. [Google Scholar] [CrossRef]
- Pavek, A.; Afrin, F.; Meldrum, T.; Pashikanti, S.; Barrott, J. Sphingomyelin synthase inhibition in sarcomas results in ceramide accumulation and apoptosis. The FASEB Journal 2019, 33, 471.416–471.416. [Google Scholar] [CrossRef]
- JaffrÉZou, J.-P.; Bruno, A.P.; Moisand, A.; Levade, T.; Laurent, G.U.Y. Activation of a nuclear sphingomyelinase in radiation induced apoptosis. The FASEB Journal 2001, 15, 123–133. [Google Scholar] [CrossRef]
- Kakoi, H.; Maeda, S.; Shinohara, N.; Matsuyama, K.; Imamura, K.; Kawamura, I.; Nagano, S.; Setoguchi, T.; Yokouchi, M.; Ishidou, Y.; et al. Bone Morphogenic Protein (BMP) Signaling Up-regulates Neutral Sphingomyelinase 2 to Suppress Chondrocyte Maturation via the Akt Protein Signaling Pathway as a Negative Feedback Mechanism*. Journal of Biological Chemistry 2014, 289, 8135–8150. [Google Scholar] [CrossRef] [PubMed]
- Keresztes, A.; Streicher, J.M. Synergistic interaction of the cannabinoid and death receptor systems – a potential target for future cancer therapies? FEBS Letters 2017, 591, 3235–3251. [Google Scholar] [CrossRef]
- Klutzny, S.; Lesche, R.; Keck, M.; Kaulfuss, S.; Schlicker, A.; Christian, S.; Sperl, C.; Neuhaus, R.; Mowat, J.; Steckel, M.; et al. Functional inhibition of acid sphingomyelinase by Fluphenazine triggers hypoxia-specific tumor cell death. Cell Death & Disease 2017, 8, e2709–e2709. [Google Scholar] [CrossRef]
- Risner, M.L.; Ribeiro, M.; McGrady, N.R.; Kagitapalli, B.S.; Chamling, X.; Zack, D.J.; Calkins, D.J. Neutral sphingomyelinase inhibition promotes local and network degeneration in vitro and in vivo. Cell Communication and Signaling 2023, 21, 305. [Google Scholar] [CrossRef]
- Arenz, C. Small Molecule Inhibitors of Acid Sphingomyelinase. Cellular Physiology and Biochemistry 2010, 26, 1–8. [Google Scholar] [CrossRef]
- Skácel, J.; Slusher, B.S.; Tsukamoto, T. Small Molecule Inhibitors Targeting Biosynthesis of Ceramide, the Central Hub of the Sphingolipid Network. Journal of Medicinal Chemistry 2021, 64, 279–297. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Yokota, W.; Katoh, T. Enantioselective Total Synthesis of (+)-Scyphostatin, a Potent and Specific Inhibitor of Neutral Sphingomyelinase. Synthesis 2007, 2007, 622–637. [Google Scholar] [CrossRef]
- Cheng, J.C.; Bai, A.; Beckham, T.H.; Marrison, S.T.; Yount, C.L.; Young, K.; Lu, P.; Bartlett, A.M.; Wu, B.X.; Keane, B.J.; et al. Radiation-induced acid ceramidase confers prostate cancer resistance and tumor relapse. The Journal of Clinical Investigation 2013, 123, 4344–4358. [Google Scholar] [CrossRef] [PubMed]
- Ghandour, B.; Dbaibo, G.; Darwiche, N. The unfolding role of ceramide in coordinating retinoid-based cancer therapy. Biochemical Journal 2021, 478, 3621–3642. [Google Scholar] [CrossRef]
- El Bawab, S.; Birbes, H.; Roddy, P.; Szulc, Z.M.; Bielawska, A.; Hannun, Y.A. Biochemical Characterization of the Reverse Activity of Rat Brain Ceramidase: A CoA-INDEPENDENT AND FUMONISIN B1-INSENSITIVE CERAMIDE SYNTHASE*. Journal of Biological Chemistry 2001, 276, 16758–16766. [Google Scholar] [CrossRef]
- Draper, J.M.; Xia, Z.; Smith, R.A.; Zhuang, Y.; Wang, W.; Smith, C.D. Discovery and Evaluation of Inhibitors of Human Ceramidase. Molecular Cancer Therapeutics 2011, 10, 2052–2061. [Google Scholar] [CrossRef] [PubMed]
- Kus, G.; Kabadere, S.; Uyar, R.; Kutlu, H.M. Induction of apoptosis in prostate cancer cells by the novel ceramidase inhibitor ceranib-2. In Vitro Cellular & Developmental Biology - Animal 2015, 51, 1056–1063. [Google Scholar] [CrossRef]
- Saied, E.M.; Arenz, C. Small Molecule Inhibitors of Ceramidases. Cellular Physiology and Biochemistry 2014, 34, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Bielawska, A.; Bielawski, J.; Szulc, Z.M.; Mayroo, N.; Liu, X.; Bai, A.; Elojeimy, S.; Rembiesa, B.; Pierce, J.; Norris, J.S.; et al. Novel analogs of d-e-MAPP and B13. Part 2: Signature effects on bioactive sphingolipids. Bioorganic & Medicinal Chemistry 2008, 16, 1032–1045. [Google Scholar] [CrossRef]
- Saied, E.M.; Arenz, C. Inhibitors of Ceramidases. Chemistry and Physics of Lipids 2016, 197, 60–68. [Google Scholar] [CrossRef]
- Jarocki, M.; Turek, K.; Saczko, J.; Tarek, M.; Kulbacka, J.A.-O. Lipids associated with autophagy: mechanisms and therapeutic targets.
- Ruzzi, F.; Cappello, C.; Semprini, M.S.; Scalambra, L.; Angelicola, S.; Pittino, O.M.; Landuzzi, L.; Palladini, A.; Nanni, P.; Lollini, P.L. Lipid rafts, caveolae, and epidermal growth factor receptor family: friends or foes?
- Jia, W.; Yuan, J.; Zhang, J.; Li, S.; Lin, W.; Cheng, B. Bioactive sphingolipids as emerging targets for signal transduction in cancer development.
- Lai, M.K.P.; Chew, W.S.; Torta, F.; Rao, A.; Harris, G.L.; Chun, J.; Herr, D.R. Biological Effects of Naturally Occurring Sphingolipids, Uncommon Variants, and Their Analogs. NeuroMolecular Medicine 2016, 18, 396–414. [Google Scholar] [CrossRef]
- Campisi, G.M.; Signorelli, P.; Rizzo, J.; Ghilardi, C.; Antognetti, J.; Caretti, A.; Lazarević, J.S.; Strettoi, E.; Novelli, E.; Ghidoni, R.; et al. Determination of the serine palmitoyl transferase inhibitor myriocin by electrospray and Q-trap mass spectrometry. Biomedical Chromatography 2017, 31, e4026. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Yokokawa, Y.; Okuno, Y.; Murakami, N. ChemInform Abstract: Total Syntheses of Myriocin and Z-Myriocin, Two Potent Immunosuppressants, from 2-Deoxy-D-glucose. ChemInform 1995, 26. [Google Scholar] [CrossRef]
- Noda, N.; Nambu, H.; Ubukata, K.; Fujiwara, T.; Tsuge, K.; Yakura, T. Total synthesis of myriocin and mycestericin D employing Rh(II)-catalyzed CH amination followed by stereoselective alkylation. Tetrahedron 2017, 73, 868–878. [Google Scholar] [CrossRef]
- Miyake, Y.; Kozutsumi, Y.; Nakamura, S.; Fujita, T.; Kawasaki, T. Serine Palmitoyltransferase Is the Primary Target of a Sphingosine-like Immunosuppressant, ISP-1/Myriocin. Biochemical and Biophysical Research Communications 1995, 211, 396–403. [Google Scholar] [CrossRef]
- Choi, K.E.; Jung, Y.S.; Kim, D.H.; Song, J.K.; Kim, J.Y.; Jung, Y.Y.; Eum, S.Y.; Kim, J.H.; Yoon, N.Y.; Yoo, H.S.; et al. Myriocin induces apoptotic lung cancer cell death via activation of DR4 pathway. Archives of Pharmacal Research 2014, 37, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Kornienko, A.; Evidente, A.; Vurro, M.; Mathieu, V.; Cimmino, A.; Evidente, M.; van Otterlo, W.A.L.; Dasari, R.; Lefranc, F.; Kiss, R. Toward a Cancer Drug of Fungal Origin. Medicinal Research Reviews 2015, 35, 937–967. [Google Scholar] [CrossRef]
- Sharma, S.; Mathur, A.G.; Pradhan, S.; Singh, D.B.; Gupta, S. Fingolimod (FTY720): First approved oral therapy for multiple sclerosis. Journal of Pharmacology and Pharmacotherapeutics 2011, 2, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Adachi, K.; Kohara, T.; Nakao, N.; Arita, M.; Chiba, K.; Mishina, T.; Sasaki, S.; Fujita, T. Design, synthesis, and structure-activity relationships of 2-substituted-2-amino-1,3-propanediols: Discovery of a novel immunosuppressant, FTY720. Bioorganic & Medicinal Chemistry Letters 1995, 5, 853–856. [Google Scholar] [CrossRef]
- Kandagatla, B.; Prasada Raju, V.V.N.K.V.; Kumar, N.S.; Reddy, G.M.; Srinivas, K.; Iqbal, J.; Bandichhor, R.; Oruganti, S. Practical synthesis of fingolimod from diethyl acetamidomalonate. RSC Advances 2013, 3, 9687–9689. [Google Scholar] [CrossRef]
- Hunter, S.F.; Bowen, J.D.; Reder, A.T. The Direct Effects of Fingolimod in the Central Nervous System: Implications for Relapsing Multiple Sclerosis. CNS Drugs 2015, 30, 135–147. [Google Scholar] [CrossRef]
- Smith, G.P.; Chan, E.S.L. 87 - Immunomodulating Pharmaceuticals. In Clinical Immunology (Fifth Edition), Rich, R.R., Fleisher, T.A., Shearer, W.T., Schroeder, H.W., Frew, A.J., Weyand, C.M., Eds.; Elsevier: London, 2019; pp. 1177–1184. [Google Scholar]
- Rolland, W.B.; Lekic T Fau - Krafft, P.R.; Krafft Pr Fau - Hasegawa, Y.; Hasegawa Y Fau - Altay, O.; Altay O Fau - Hartman, R.; Hartman R Fau - Ostrowski, R.; Ostrowski R Fau - Manaenko, A.; Manaenko A Fau - Tang, J.; Tang J Fau - Zhang, J.H.; Zhang, J.H. Fingolimod reduces cerebral lymphocyte infiltration in experimental models of rodent intracerebral hemorrhage.
- Billich, A.; Bornancin, F.; Dévay, P.; Mechtcheriakova, D.; Urtz, N.; Baumruker, T. Phosphorylation of the Immunomodulatory Drug FTY720 by Sphingosine Kinases*. Journal of Biological Chemistry 2003, 278, 47408–47415. [Google Scholar] [CrossRef]
- Zhang, L.; Wang H Fau - Zhu, J.; Zhu J Fau - Ding, K.; Ding K Fau - Xu, J.; Xu, J. FTY720 reduces migration and invasion of human glioblastoma cell lines via inhibiting the PI3K/AKT/mTOR/p70S6K signaling pathway.
- Mahajan-Thakur, S.; Bien-Möller, S.; Marx, S.; Schroeder, H.; Rauch, B.H. Sphingosine 1-Phosphate (S1P) Signaling in Glioblastoma Multiforme—A Systematic Review. International Journal of Molecular Sciences 2017, 18. [Google Scholar] [CrossRef]
- Payne, S.G.; Oskeritzian Ca Fau - Griffiths, R.; Griffiths R Fau - Subramanian, P.; Subramanian P Fau - Barbour, S.E.; Barbour Se Fau - Chalfant, C.E.; Chalfant Ce Fau - Milstien, S.; Milstien S Fau - Spiegel, S.; Spiegel, S. The immunosuppressant drug FTY720 inhibits cytosolic phospholipase A2 independently of sphingosine-1-phosphate receptors.
- Berdyshev, E.V.; Gorshkova I Fau - Skobeleva, A.; Skobeleva A Fau - Bittman, R.; Bittman R Fau - Lu, X.; Lu X Fau - Dudek, S.M.; Dudek Sm Fau - Mirzapoiazova, T.; Mirzapoiazova T Fau - Garcia, J.G.N.; Garcia Jg Fau - Natarajan, V.; Natarajan, V. FTY720 inhibits ceramide synthases and up-regulates dihydrosphingosine 1-phosphate formation in human lung endothelial cells. J Biol Chem 2009, 284, 5467–5477. [Google Scholar] [CrossRef]
- Zununi Vahed, S.; Ghiyasvand, S.; Hosseiniyan Khatibi, S.M.; Patel, B.; Mohajel Shoja, M.; Tolouian, R.; Ardalan, M. Sphingosine 1 phosphate agonists (SPI); a potential agent to prevent acute lung injury in COVID-19. Immunopathol Persa 2021, 7, e03–e03. [Google Scholar] [CrossRef]
- Teymouri, S.; Pourbayram Kaleybar, S.; Hejazian, S.S.; Hejazian, S.M.; Ansarin, K.; Ardalan, M.; Zununi Vahed, S. The effect of Fingolimod on patients with moderate to severe COVID-19. Pharmacology Research & Perspectives 2023, 11, e01039. [Google Scholar] [CrossRef]
- Thompson, D.; Mahmood, S.; Morrice, N.; Kamli-Salino, S.; Dekeryte, R.; Hoffmann, P.A.; Doherty, M.K.; Whitfield, P.D.; Delibegović, M.; Mody, N. Fenretinide inhibits obesity and fatty liver disease but induces Smpd3 to increase serum ceramides and worsen atherosclerosis in LDLR−/− mice. Scientific Reports 2023, 13, 3937. [Google Scholar] [CrossRef] [PubMed]
- Hunsu, V.O.; Facey, C.O.B.; Fields, J.Z.; Boman, B.M. Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies. International Journal of Molecular Sciences 2021, 22. [Google Scholar] [CrossRef]
- Cuello, M.; Coats Ao Fau - Darko, I.; Darko I Fau - Ettenberg, S.A.; Ettenberg Sa Fau - Gardner, G.J.; Gardner Gj Fau - Nau, M.M.; Nau Mm Fau - Liu, J.R.; Liu Jr Fau - Birrer, M.J.; Birrer Mj Fau - Lipkowitz, S.; Lipkowitz, S. N-(4-hydroxyphenyl) retinamide (4HPR) enhances TRAIL-mediated apoptosis through enhancement of a mitochondrial-dependent amplification loop in ovarian cancer cell lines.
- Yang, X.; Hao, Y.; Ding, Z.; Pater, A. BAG-1 Promotes Apoptosis Induced by N-(4-hydroxyphenyl)retinamide in Human Cervical Carcinoma Cells. Experimental Cell Research 2000, 256, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Fields, A.L.; Soprano, D.R.; Soprano, K.J. Retinoids in biological control and cancer. Journal of Cellular Biochemistry 2007, 102, 886–898. [Google Scholar] [CrossRef]
- Veronesi, U.; Mariani, L.; Decensi, A.; Formelli, F.; Camerini, T.; Miceli, R.; Di Mauro, M.G.; Costa, A.; Marubini, E.; Sporn, M.B.; et al. Fifteen-year results of a randomized phase III trial of fenretinide to prevent second breast cancer. Annals of Oncology 2006, 17, 1065–1071. [Google Scholar] [CrossRef]
- Potenza, R.L.; Lodeserto, P.; Orienti, I. Fenretinide in Cancer and Neurological Disease: A Two-Face Janus Molecule. International Journal of Molecular Sciences 2022, 23. [Google Scholar] [CrossRef]
- Cazzaniga, M.; Varricchio, C.; Montefrancesco, C.; Feroce, I.; Guerrieri-Gonzaga, A. Fenretinide (4-HPR): A Preventive Chance for Women at Genetic and Familial Risk? BioMed Research International 2012, 2012, 172897. [Google Scholar] [CrossRef]
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2013. CA: A Cancer Journal for Clinicians 2013, 63, 11–30. [Google Scholar] [CrossRef]
- Orienti, I.; Francescangeli, F.; De Angelis, M.L.; Fecchi, K.; Bongiorno-Borbone, L.; Signore, M.; Peschiaroli, A.; Boe, A.; Bruselles, A.; Costantino, A.; et al. A new bioavailable fenretinide formulation with antiproliferative, antimetabolic, and cytotoxic effects on solid tumors. Cell Death & Disease 2019, 10, 529. [Google Scholar] [CrossRef]
- De Angelis, M.L.; Francescangeli, F.; Aricò, E.; Verachi, P.; Zucchetti, M.; Matteo, C.; Petricci, E.; Pilozzi, E.; Orienti, I.; Boe, A.; et al. A nanoencapsulated oral formulation of fenretinide promotes local and metastatic breast cancer dormancy in HER2/neu transgenic mice. Journal of Experimental & Clinical Cancer Research 2024, 43, 296. [Google Scholar] [CrossRef]
- Thomas, J.A.-O.X.; El-Khoueiry, A.B.; Maurer, B.J.; Groshen, S.; Pinski, J.K.; Cobos, E.; Gandara, D.R.; Lenz, H.J.; Kang, M.H.; Reynolds, C.P.; et al. A phase I study of intravenous fenretinide (4-HPR) for patients with malignant solid tumors.
- Zhang†, Y.; , R. S.;, S.C.;, X.L., Xiaotian Feng, Rosa Moshirian,; Yuan*, R.Y.a.W. α-GalCer and iNKT Cell-Based Cancer Immunotherapy: Realizing the Therapeutic Potentials. Frontiers in Immunology 2019, 10. [Google Scholar] [CrossRef]
- Wang*†‡§, Z.Z.W.Z.B.W.C.X.W.Z.P.G. The Total Synthesis of Immunostimulant α-Galactosylceramides from Naturally Configured α-Galactoside Raffinose. Organic Letters 2011. [Google Scholar] [CrossRef]
- Barbara, A. , sullivan. Activation or anergy: NKT cells are stunned by α- galactosylceramide. J Clin Invest. 2005, 115, 2328–2329. [Google Scholar] [CrossRef]
- A Kikuchi, M.N. , C Schmidt, Y Koezuka, S Ishihara, Y Ishikawa, K Tadokoro, S Durrant, A Boyd, T Juji & A Nicol In vitro anti-tumour activity of α-galactosylceramide-stimulated human invariant Vα24+NKT cells against melanoma. British Journal of Cancer 85, 741–746. [CrossRef]
- Takahide Toyoda, 2 Toshiko Kamata,1,2 Kazuhisa Tanaka,1; Fumie Ihara; Mariko Takami; Hidemi Suzuki; Takahiro Nakajima; Takayuki Ikeuchi; Yohei Kawasaki; Hideki Hanaoka; Toshinori Nakayama; Ichiro Yoshino; et al. Phase II study of α-galactosylceramidepulsed antigen-presenting cells in patients with advanced or recurrent non-small cell lung cancer. Journal forImmunoTherapy of Cancer 2020, 8. [CrossRef]
- Jesse Coward, G.A. , Elgilda Musi, Jean-Philip Truman, Adriana; Haimovitz-Friedman, J.C.A., Elaine Wang, Alfred H. Merrill, Jr. &; Schwartz, G.K. Safingol (l-threo-sphinganine) induces autophagy in solid tumor cells through inhibition of PKC and the PI3-kinase pathway. Autophagy 2009, 5, 184–193. [Google Scholar] [CrossRef]
- Michael, E. Jung, S.W.Y. Synthesis of threo-β-aminoalcohols from aminoaldehydes via chelation-controlled additions. Total synthesis of l-threo sphingosine and safingol2012. Tetrahedron Letters 2012, 53, 4216–4220. [Google Scholar] [CrossRef]
- Ling, L.U.; Tan, K.B.; Lin, H.; Chiu, G.N.C. The role of reactive oxygen species and autophagy in safingol-induced cell death. Cell Death & Disease 2011, 2, e129–e129. [Google Scholar] [CrossRef]
- Dickson, M.A.; Carvajal, R.D.; Merrill, A.H., Jr; Gonen, M.; Cane, L.M.; Schwartz, G.K. A Phase I Clinical Trial of Safingol in Combination with Cisplatin in Advanced Solid Tumors. Clinical Cancer Research 2011, 17, 2484–2492. [Google Scholar] [CrossRef]
- Kunkel, G.T.; Maceyka, M.; Milstien, S.; Spiegel, S. Targeting the sphingosine-1-phosphate axis in cancer, inflammation and beyond. Nature Reviews Drug Discovery 2013, 12, 688–702. [Google Scholar] [CrossRef]
- Fabišíková, M.; Martinková, M.; Hirková, S.; Gonda, J.; Pilátová, M.B.; Gönciová, G. Total synthesis and the anticancer activity of (+)-spisulosine. Carbohydrate Research 2016, 435, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Amarante, G.W.; Cavallaro, M.; Coelho, F. Highly diastereoselective total synthesis of the anti-tumoral agent (±)-Spisulosine (ES285) from a Morita–Baylis–Hillman adduct. Tetrahedron Letters 2010, 51, 2597–2599. [Google Scholar] [CrossRef]
- Salcedo, M.; Cuevas, C.; Alonso, J.L.; Otero, G.; Faircloth, G.; Fernandez-Sousa, J.M.; Avila, J.; Wandosell, F. The marine sphingolipid-derived compound ES 285 triggers an atypical cell death pathway. Apoptosis 2007, 12, 395–409. [Google Scholar] [CrossRef]
- Sánchez, A.M.; Malagarie-Cazenave, S.; Olea, N.; Vara, D.; Cuevas, C.; Díaz-Laviada, I. Spisulosine (ES-285) induces prostate tumor PC-3 and LNCaP cell death by de novo synthesis of ceramide and PKCζ activation. European Journal of Pharmacology 2008, 584, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Cuadros, R.; Montejo de Garcini, E.; Wandosell, F.; Faircloth, G.; Fernández-Sousa, J.M.; Avila, J. The marine compound spisulosine, an inhibitor of cell proliferation, promotes the disassembly of actin stress fibers. Cancer Letters 2000, 152, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Vilar, E.; Grünwald, V.; Schöffski, P.; Singer, H.; Salazar, R.; Iglesias, J.L.; Casado, E.; Cullell-Young, M.; Baselga, J.; Tabernero, J. A phase I dose-escalating study of ES-285, a marine sphingolipid-derived compound, with repeat dose administration in patients with advanced solid tumors. Investigational New Drugs 2012, 30, 299–305. [Google Scholar] [CrossRef]
- Massard, C.; Salazar, R.; Armand, J.P.; Majem, M.; Deutsch, E.; García, M.; Oaknin, A.; Fernández-García, E.M.; Soto, A.; Soria, J.C. Phase I dose-escalating study of ES-285 given as a three-hour intravenous infusion every three weeks in patients with advanced malignant solid tumors. Investigational New Drugs 2012, 30, 2318–2326. [Google Scholar] [CrossRef]
- Campo, M.W.J.D.B.N.H.B.M.D. P.
- Ledroit, V.; Debitus, C.; Lavaud, C.; Massiot, G. Jaspines A and B: two new cytotoxic sphingosine derivatives from the marine sponge Jaspis sp. Tetrahedron Letters 2003, 44, 225–228. [Google Scholar] [CrossRef]
- Cingolani, F.; Simbari, F.; Abad, J.L.; Casasampere, M.; Fabrias, G.; Futerman, A.H.; Casas, J. Jaspine B induces nonapoptotic cell death in gastric cancer cells independently of its inhibition of ceramide synthase. J Lipid Res 2017, 58, 1500–1513. [Google Scholar] [CrossRef]
- Srinath Pashikanti, R.U. , Sunil A. David, Apurba Datta. Total Synthesis and Structure-Activity Relationship Studies of the Cytotoxic Anhydrophytosphingosine Jaspine B (Pachastrissamine). Synthesis 2017, 49, 2088–2100. [Google Scholar] [CrossRef]
- Liu, X.; Jin, Y.; Zhang, M.; Jin, Y.; Cao, J.; Dong, H.; Fu, X.; Jin, C.Y. The RIP3 activator C8 regulates the autophagy flux mediated by p62 and promotes the immunogenic form of cell death in human gastric cancer cells.
- Bielsa, N.; Casasampere, M.; Abad, J.L.; Enrich, C.A.-O.; Delgado, A.; Fabriàs, G.; Lizcano, J.A.-O.; Casas, J. Methuosis Contributes to Jaspine-B-Induced Cell Death. International Journal of Molecular Sciences 2022. [Google Scholar] [CrossRef] [PubMed]
- Pashikanti, S.; Ukani, R.; David, S.A.; Datta, A. Total Synthesis and Structure–Activity Relationship Studies of the Cytotoxic Anhydrophytosphingosine Jaspine B (Pachastrissamine). Synthesis 2017, 49, 2088–2100. [Google Scholar] [CrossRef]
- Khajeh Pour, S.; Mateen, S.; Pashikanti, S.; Barrott, J.A.-O.; Aghazadeh-Habashi, A.A.-O. Formulation, Characterization, and In Vitro/In Vivo Efficacy Studies of a Novel Liposomal Drug Delivery System of Amphiphilic Jaspine B for Treatment of Synovial Sarcoma. LID - 10.3390/md20080509 [doi] LID - 509.
- Adada, M.; Luberto, C.; Canals, D. Inhibitors of the sphingomyelin cycle: Sphingomyelin synthases and sphingomyelinases. Chemistry and physics of lipids 2016, 197, 45–59. [Google Scholar] [CrossRef]
- Choi, M.-K.; Lee, J.; Nam, S.J.; Kang, Y.J.; Han, Y.; Choi, K.; Choi, Y.A.; Kwon, M.; Lee, D.; Song, I.-S. Pharmacokinetics of Jaspine B and Enhancement of Intestinal Absorption of Jaspine B in the Presence of Bile Acid in Rats. Marine Drugs 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Alicja Bielawska, M.S.G. , David Perry, Supriya Jayadev, James A. Shayman, Charles McKay, Yusuf A. Hannun,. (1S,2R)-D-erythro-2-(N-Myristoylamino)-1-phenyl-1-propanol as an Inhibitor of Ceramidase. Journal of Biological Chemistry 1996, 271, 12646–12654. [Google Scholar] [CrossRef]
- Chang, Y.-T.; Choi, J.; Ding, S.; Prieschl, E.E.; Baumruker, T.; Lee, J.-M.; Chung, S.-K.; Schultz, P.G. The Synthesis and Biological Characterization of a Ceramide Library. Journal of the American Chemical Society 2002, 124, 1856–1857. [Google Scholar] [CrossRef]
- Liu, B.; Xiao, J.; Dong, M.; Qiu, Z.; Jin, J. Human alkaline ceramidase 2 promotes the growth, invasion, and migration of hepatocellular carcinoma cells via sphingomyelin phosphodiesterase acid-like 3B. Cancer Science 2020, 111, 2259–2274. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Larrauri, A.; Das Adhikari, U.; Aramburu-Nuñez, M.; Custodia, A.; Ouro, A. Ceramide Metabolism Enzymes—Therapeutic Targets against Cancer. Medicina 2021, 57, 729. [Google Scholar] [CrossRef]
- Xu, R.; Sun, W.; Jin, J.; Obeid, L.M.; Mao, C. Role of alkaline ceramidases in the generation of sphingosine and its phosphate in erythrocytes. The FASEB Journal 2010, 24, 2507–2515. [Google Scholar] [CrossRef]
- Aiping Bai, Z.M.S. , Jacek Bielawski, Jason S. Pierce, Barbara Rembiesa, Silva Terzieva, Cungui Mao, Ruijuan Xu, Bill Wu, Christopher J. Clarke, Benjamin Newcomb, Xiang Liu, James Norris, Yusuf A. Hannun, Alicja Bielawska. Targeting (cellular) lysosomal acid ceramidase by B13: Design, synthesis and evaluation of novel DMG-B13 ester prodrugs. Bioorganic & Medicinal Chemistry Letters 2014, 22, 6933–6944. [Google Scholar] [CrossRef]
- Holman, D.H.; Turner, L.S.; El-Zawahry, A.; Elojeimy, S.; Liu, X.; Bielawski, J.; Szulc, Z.M.; Norris, K.; Zeidan, Y.H.; Hannun, Y.A. Lysosomotropic acid ceramidase inhibitor induces apoptosis in prostate cancer cells. Cancer chemotherapy and pharmacology 2008, 61, 231–242. [Google Scholar] [CrossRef] [PubMed]
| Enzyme | Small molecule | Function |
|
Serine palmitoyl Transferase Inhibitors |
Myriocin/ ISP-1 [53], NA808 [54], L-cycloserine [55], ARN14494 [56] ; L-penicillamine [57], β-halo-l-alanines [58]. | Inhibition |
| Sphingosine kinase Inhibitors | SKI-I-V [59],Safingol [60,61], Doxorubicin [62], Actinomycin D [63], SG-12 [64], ABC294640 [65],PF-543 [66] | Inhibition |
| Glucosylceramide synthase inhibitors | Sinbaglustat (OGT2378) [67], Ibiglustat HCl/Venglustat [68], PDMP.HCl [69], L-Malic acid [70], PPMP.HCl [71], T-036[72]. | Inhibition |
| Glucosylceramidase inhibitors | D-gluconolactone [73], AMP-DNM-(5′-adamantane-1′-yl-methoxy)-pentyl-1-deoxynojirimycin) [74], Cyclophellitol [75],Conduritol B aziridine (CBA), Conduritol B epoxide (CBE) [76,77], butyldeoxynojirimycin (NB-DNJ) [78]. |
Inhibition |
| Sphingomyelin synthase inhibitors | Malabaricone C [79] , D609 [80], MS-209 [81], D609 Prodrug [80], Dy105 [82], Jaspine B [83]. | Inhibition |
| Sphingomyelinase | Radiation[84], BMP[85] ,Win5[86], | Activation |
| Fluphenazine [87], GW4869 [88], AD2765 [89], Cowanin [90] Scyphostatin [91]. | Inhibition | |
| Ceramidase | Radiation [92], Daunorubicin [93] | Activation |
| Fumonisin B1 [94], Ceranib-1 [95], Ceranib-2 [96], Cystatin SA [97], D-e-MAP[98], Carmofur [99], SABRAC [99], B13 [99]. | Inhibition |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




