Submitted:
27 March 2025
Posted:
28 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Biosynthesis and Metabolism of ZiN
3. Toxicity and Safety Profile of ZiN
4. Pharmacological Properties of ZiN: Insights from Pre-Clinical Studies
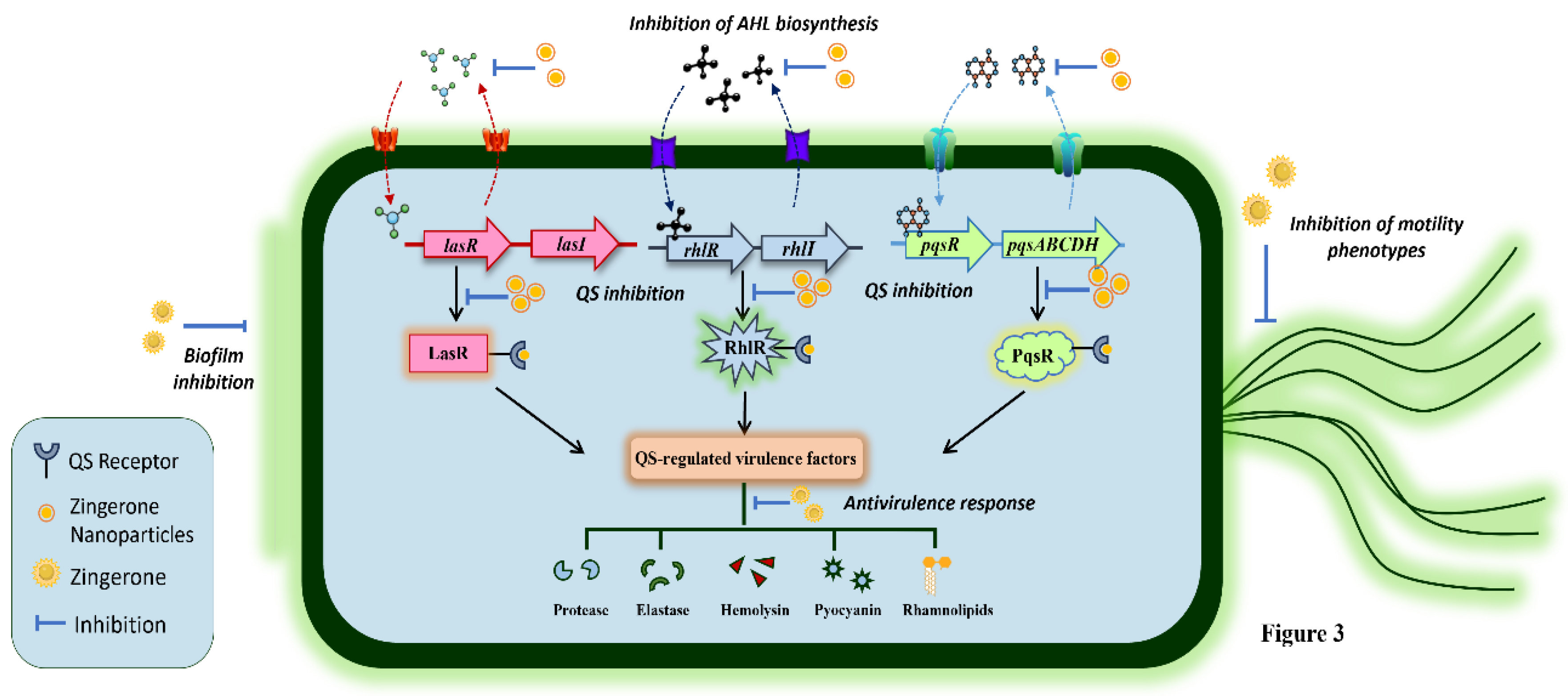
4.1. Beyond the Antimicrobial Spectrum: Anti-Quorum Sensing and Antivirulence Prospects of ZiN
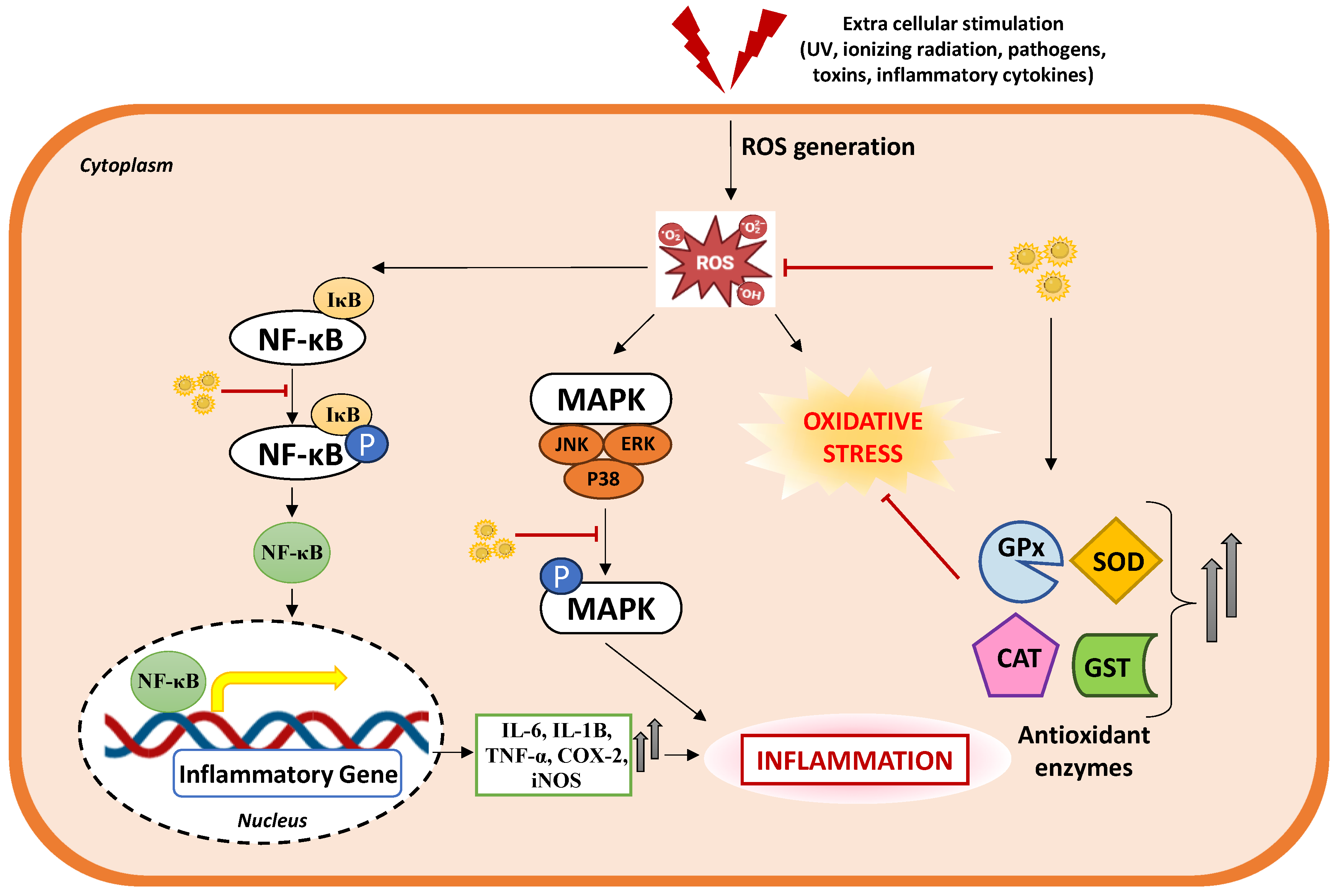
4.2. Antioxidant Potential: Radical Scavenging Activity in Focus
4.3. Anti-Inflammatory Property of ZiN: Subsiding the Inflammatory Responses
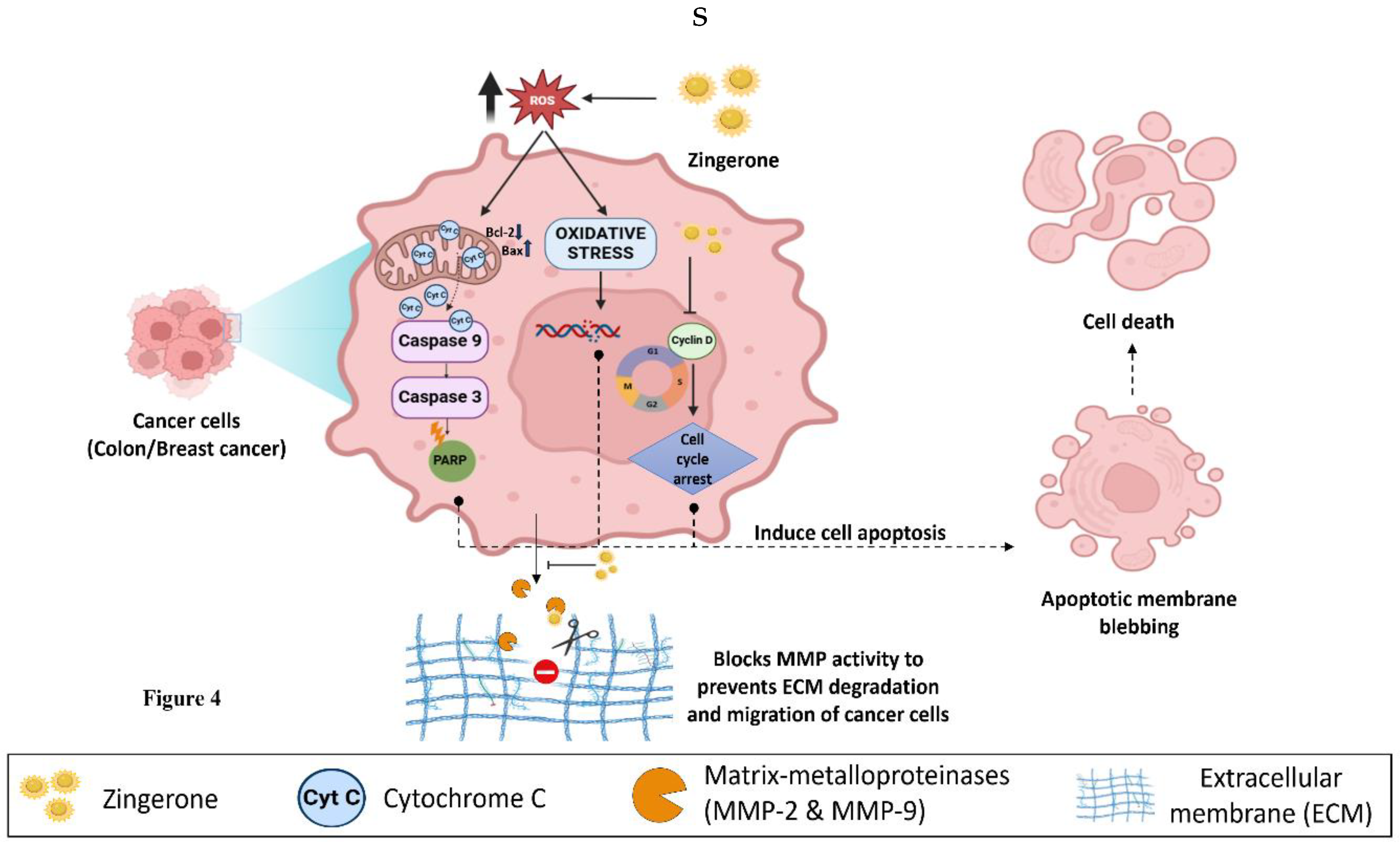
4.4. Anticancer Potential: Far-Reaching Effects of ZiN
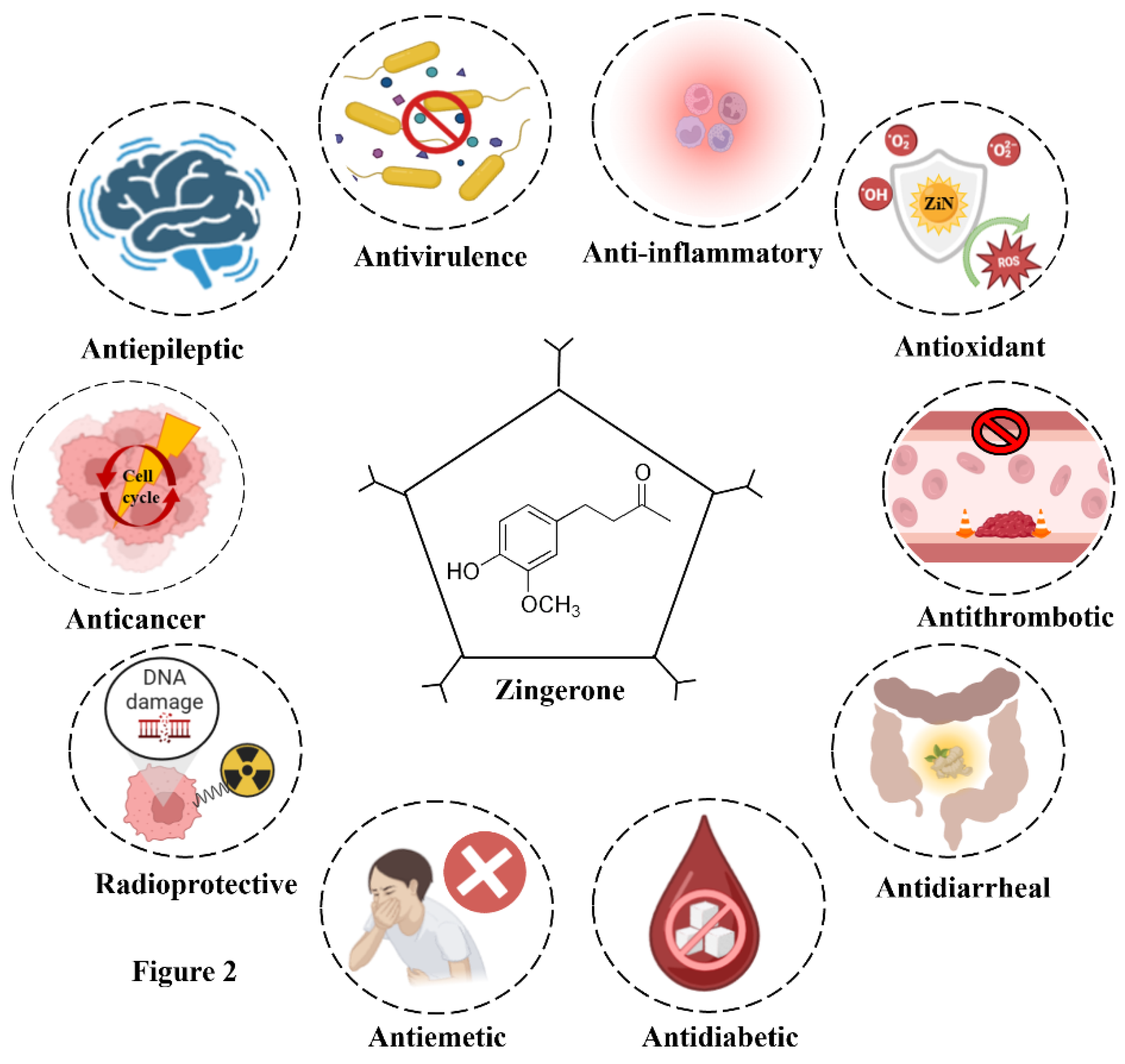
4.5. Other Biological Properties of ZiN: Extending Beyond Boundaries
5. Clinical Trials on ZiN: The Ongoing Journey from Lab to Market
6. Conclusion and Future Prospects
Acknowledgements
Author Contributions
Conflict of Interest
Data availability
Funding
Abbreviations
References
- Aeschbach, R.; Löliger, J.; Scott, B.C.; Murcia, A.; Butler, J.; Halliwell, B.; Aruoma, O.I. Antioxidant actions of thymol, carvacrol, 6-gingerol, zingerone and hydroxytyrosol. Food Chem Toxicol 1994, 32, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Afzal, N.; Nguyen, N.; Min, M.; Egli, C.; Afzal, S.; Chaudhuri, R.K.; Burney, W.A.; Sivamani, R.K. Prospective randomized double-blind comparative study of topical acetyl zingerone with tetrahexyldecyl ascorbate versus tetrahexyldecyl ascorbate alone on facial photoaging. J Cosmet Dermatol 2024, 23, 2467–2477. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B.; Rehman, M.U.; Amin, I.; Arif, A.; Rasool, S.; Bhat, S.A.; Afzal, I.; Hussain, I.; Bilal, S.; Mir, M.u.R.; Efferth, T. A Review on Pharmacological Properties of Zingerone (4-(4-Hydroxy-3-methoxyphenyl)-2-butanone). Sci World J 2015, 2015, 816364. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B.; Rehman, M.U.; Amin, I.; Mir, M.u.R.; Ahmad, S.B.; Farooq, A.; Muzamil, S.; Hussain, I.; Masoodi, M.; Fatima, B. Zingerone (4-(4-hydroxy-3-methylphenyl) butan-2-one) protects against alloxan-induced diabetes via alleviation of oxidative stress and inflammation: Probable role of NF-kB activation. Saudi Pharm J 2018, 26, 1137–1145. [Google Scholar] [CrossRef]
- Al-Daghri, N.M.; Alokail, M.S.; Alkharfy, K.M.; Mohammed, A.K.; Abd-Alrahman, S.H.; Yakout, S.M.; Amer, O.E.; Krishnaswamy, S. Fenugreek extract as an inducer of cellular death via autophagy in human T lymphoma Jurkat cells. BMC Complement Altern Med 2012, 12, 1–8. [Google Scholar] [CrossRef]
- Alam, M.F.; Hijri, S.I.; Alshahrani, S.; Alqahtani, S.S.; Jali, A.M.; Ahmed, R.A.; Adawi, M.M.; Algassmi, S.M.; Shaheen, E.S.; Moni, S.S.; Anwer, T. Zingerone Attenuates Carfilzomib-Induced Cardiotoxicity in Rats through Oxidative Stress and Inflammatory Cytokine Network. Int J Mol Sci 2022, 23, 15617. [Google Scholar] [CrossRef]
- Alharbi, H.M.; Alaghaz, A.-N.M.A.; Al Hujran, T.A.; Eldin, Z.E.; Elbeltagi, S. A novel zingerone-loaded zinc MOF coated by niosome nanocomposites to enhance antimicrobial properties and apoptosis in breast cancer cells. Mater Today Commun 2024, 41, 110245. [Google Scholar] [CrossRef]
- Alibakhshi, T.; Khodayar, M.J.; Khorsandi, L.; Rashno, M.; Zeidooni, L. Protective effects of zingerone on oxidative stress and inflammation in cisplatin-induced rat nephrotoxicity. Biomed Pharmacother 2018, 105, 225–232. [Google Scholar] [CrossRef]
- ANZCTR (2024) A phase 1 study to evaluate the safety, tolerability and pharmacokinetics of a ginger tincture extract in healthy volunteers. Available online: https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=387127 (accessed on 25 February 2025).
- Bae, W.-Y.; Choi, J.-S.; Kim, J.-E.; Park, C.; Jeong, J.-W. Zingerone suppresses angiogenesisviainhibition of matrix metalloproteinases during tumor development. Oncotarget 2016, 7, 47232–47241. [Google Scholar] [CrossRef]
- Cao, X.; Zhu, Q.; Wang, Q.-L.; Adu-Frimpong, M.; Wei, C.-M.; Weng, W.; Bao, R.; Wang, Y.-P.; Yu, J.-N.; Xu, X.M. Improvement of Oral Bioavailability and Anti-Tumor Effect of Zingerone Self-Microemulsion Drug Delivery System. J Pharm Sci 2021, 110, 2718–2727. [Google Scholar] [CrossRef]
- Caval, M.; Dettori, M.A.; Carta, P.; Dallocchio, R.; Dessì, A.; Marceddu, S.; Serra, P.A.; Fabbri, D.; Rocchitta, G. Sustainable Electropolymerization of Zingerone and Its C2 Symmetric Dimer for Amperometric Biosensor Films. Molecules 2023, 28, 6017. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, R.K.; Meyer, T.; Premi, S.; Brash, D. Acetyl zingerone: An efficacious multifunctional ingredient for continued protection against ongoing DNA damage in melanocytes after sun exposure ends. Int J Cosmet Sci 2019, 42, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Cheong, K.O.; Shin, D.-S.; Bak, J.; Lee, C.; Kim, K.W.; Je, N.K.; Chung, H.Y.; Yoon, S.; Moon, J.-O. Hepatoprotective effects of zingerone on carbon tetrachloride- and dimethylnitrosamine-induced liver injuries in rats. Arch Pharm Res 2015, 39, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-S.; Ryu, J.; Bae, W.-Y.; Park, A.; Nam, S.; Kim, J.-E.; Jeong, J.-W. Zingerone Suppresses Tumor Development through Decreasing Cyclin D1 Expression and Inducing Mitotic Arrest. Int J Mol Sci 2018, 19, 2832. [Google Scholar] [CrossRef]
- Chougule, S.; Basrani, S.; Gavandi, T.; Patil, S.; Yankanchi, S.; Jadhav, A.; Karuppayil, S.M. Zingerone effect against Candida albicans growth and biofilm production. J Med Mycol 2025, 35, 101527. [Google Scholar] [CrossRef]
- Cs, P.; Vincent, S. Antidiabetic, hypolipidemic, and histopathological analysis of zingerone in streptozotocin-induced diabetic rats. Asian J Pharm Clin Res 2016, 220–224. [Google Scholar] [CrossRef]
- Darekar, S.U.; Nagrale, D.S.N.; Babar, D.V.B.; Pondkule, A. Review on ginger: Chemical constituents & biological effects. J Pharmacogn Phytochem 2023, 12, 267–271. [Google Scholar] [CrossRef]
- Dhaliwal, S.; Rybak, I.; Pourang, A.; Burney, W.; Haas, K.; Sandhu, S.; Crawford, R.; KSivamani, R. Randomized double-blind vehicle controlled study of the effects of topical acetyl zingerone on photoaging. J Cosmet Dermatol 2020, 20, 166–173. [Google Scholar] [CrossRef]
- El Adawy, M.A.A.E.A.; Abd El-Latif, A.; Farrag, F.; Shukry, M.; Braiji, S.H.B.H.; Hanafy, A. Effect of Zingerone and/or Vitamin C on the Immune System of Albino Rats, Hematological, Biochemical, Gene Expression Biomarkers and Histological Study. Egypt J Vet Sci 2025, 56, 1393–1404. [Google Scholar] [CrossRef]
- Elshopakey, G.E.; Almeer, R.; Alfaraj, S.; Albasher, G.; Abdelgawad, M.E.; Abdel Moneim, A.E.; Essawy, E.A. Zingerone mitigates inflammation, apoptosis and oxidative injuries associated with renal impairment in adriamycin-intoxicated mice. Toxin Rev 2021, 41, 731–742. [Google Scholar] [CrossRef]
- Fahmi, A.; Hassanen, N.; Abdur-Rahman, M.; Shams-Eldin, E. Phytochemicals, antioxidant activity and hepatoprotective effect of ginger (Zingiber officinale) on diethylnitrosamine toxicity in rats. Biomarkers 2019, 24, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Garza-Cadena, C.; Ortega-Rivera, D.M.; Machorro-García, G.; Gonzalez-Zermeño, E.M.; Homma-Dueñas, D.; Plata-Gryl, M.; Castro-Muñoz, R. A comprehensive review on Ginger (Zingiber officinale) as a potential source of nutraceuticals for food formulations: Towards the polishing of gingerol and other present biomolecules. Food Chem 2023, 413, 135629. [Google Scholar] [CrossRef] [PubMed]
- Ghafoor, K.; Al Juhaimi, F.; Özcan, M.M.; Uslu, N.; Babiker, E.E.; Mohamed Ahmed, I.A. Total phenolics, total carotenoids, individual phenolics and antioxidant activity of ginger (Zingiber officinale) rhizome as affected by drying methods. Lwt 2020, 126, 109354. [Google Scholar] [CrossRef]
- Ghareib, S.A.; El-Bassossy, H.M.; Elberry, A.A.; Azhar, A.; Watson, M.L.; Banjar, Z.M.; Alahdal, A.M. Protective effect of zingerone on increased vascular contractility in diabetic rat aorta. Eur J Pharmacol 2016, 780, 174–179. [Google Scholar] [CrossRef]
- Grivennikov, S.; Kumar, L.; Chhibber, S.; Harjai, K. Zingerone Suppresses Liver Inflammation Induced by Antibiotic Mediated Endotoxemia through Down Regulating Hepatic mRNA Expression of Inflammatory Markers in Pseudomonas aeruginosa Peritonitis Mouse Model. PLoS ONE 2014, 9, e106536. [Google Scholar] [CrossRef]
- Han, L.-K.; Morimoto, C.; Zheng, Y.-N.; Li, W.; Asami, E.; Okuda, H.; Saito, M. Effects of Zingerone on Fat Storage in Ovariectomized Rats. Yakugaku Zasshi 2008, 128, 1195–1201. [Google Scholar] [CrossRef]
- Hemalatha, K.L.; Stanely Mainzen Prince, P. Antihyperlipidaemic, Antihypertrophic, and Reducing Effects of Zingerone on Experimentally Induced Myocardial Infarcted Rats. J Biochem Mol Toxicol 2015, 29, 182–188. [Google Scholar] [CrossRef]
- Hemalatha, K.L.; Stanely Mainzen Prince, P. Anti-inflammatory and anti-thrombotic effects of zingerone in a rat model of myocardial infarction. Eur J Pharmacol 2016, 791, 595–602. [Google Scholar] [CrossRef]
- Heo, K.T.; Park, K.W.; Won, J.; Lee, B.; Jang, J.-H.; Ahn, J.-O.; Hwang, B.Y.; Hong, Y.-S. Construction of an Artificial Biosynthetic Pathway for Zingerone Production in Escherichia coli Using Benzalacetone Synthase from Piper methysticum. J Agric Food Chem 2021, 69, 14620–14629. [Google Scholar] [CrossRef]
- Hsiang, C.-Y.; Lo, H.-Y.; Huang, H.-C.; Li, C.-C.; Wu, S.-L.; Ho, T.-Y. Ginger extract and zingerone ameliorated trinitrobenzene sulphonic acid-induced colitis in mice via modulation of nuclear factor-κB activity and interleukin-1β signalling pathway. Food Chem 2013, 136, 170–177. [Google Scholar] [CrossRef]
- Huang, S.; Yao, X.; Cao, B.; Zhang, N.; Soladoye, O.P.; Zhang, Y.; Fu, Y. Encapsulation of zingerone by self-assembling peptides derived from fish viscera: Characterization, interaction and effects on colon epithelial cells. Food Chem X 2024, 22, 101506. [Google Scholar] [CrossRef] [PubMed]
- Iwami, M.; Shiina, T.; Hirayama, H.; Shima, T.; Takewaki, T.; Shimizu, Y. Inhibitory effects of zingerone, a pungent component of Zingiber officinale Roscoe, on colonic motility in rats. J Nat Med 2010, 65, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Jesudoss VAS, Victor Antony Santiago S, Venkatachalam K, Subramanian P (2017) Zingerone (Ginger Extract): antioxidant potential for efficacy in gastrointestinal and liver disease. Gastrointest Tissue pp. 289–297. [CrossRef]
- Jin, Z.; Lee, G.; Kim, S.; Park, C.-S.; Park, Y.S.; Jin, Y.-H. Ginger and Its Pungent Constituents Non-Competitively Inhibit Serotonin Currents on Visceral Afferent Neurons. The Korean J Physiol Pharmacol 2014, 18, 149. [Google Scholar] [CrossRef] [PubMed]
- Jindal, S.; Ghosal, K.; Khamaisi, B.; Kana'an, N.; Nassar-Marjiya, E.; Farah, S. Facile Green Synthesis of Zingerone Based Tissue-Like Biodegradable Polyester with Shape-Memory Features for Regenerative Medicine. Adv Funct Mater 2024, 34. [Google Scholar] [CrossRef]
- Kabuto, H.; Nishizawa, M.; Tada, M.; Higashio, C.; Shishibori, T.; Kohno, M. Zingerone [4-(4-hydroxy-3-methoxyphenyl)-2-butanone] Prevents 6-Hydroxydopamine-induced Dopamine Depression in Mouse Striatum and Increases Superoxide Scavenging Activity in Serum. Neurochem Res 2005, 30, 325–332. [Google Scholar] [CrossRef]
- Kazemi, M.; Jafarzadeh, A.; Nemati, M.; Taghipour, F.; Oladpour, O.; Rezayati, M.T.; Khorramdelazad, H.; Hassan, Z.M. Zingerone improves the immune responses in an animal model of breast cancer. J Altern Complement Med 2021, 18, 303–310. [Google Scholar] [CrossRef]
- Kharga, K.; Dhar, I.; Kashyap, S.; Sengupta, S.; Kumar, D.; Kumar, L. Zingerone inhibits biofilm formation and enhances antibiotic efficacy against Salmonella biofilm. World J Microbiol Biotechnol 2023, 39. [Google Scholar] [CrossRef]
- Kim Jung, N.; Kim Hyun, J.; Kim, I.; Kim Yun, T.; Kim Byung, J. The Mechanism of Action of Zingerone in the Pacemaker Potentials of Interstitial Cells of Cajal Isolated from Murine Small Intestine. Cell Physiol Biochem 2018, 46, 2127–2137. [Google Scholar] [CrossRef]
- Kim, M.K.; Chung, S.W.; Kim, D.H.; Kim, J.M.; Lee, E.K.; Kim, J.Y.; Ha, Y.M.; Kim, Y.H.; No, J.-K.; Chung, H.S.; Park, K.-Y.; Rhee, S.H.; Choi, J.S.; Yu, B.P.; Yokozawa, T.; Kim, Y.J.; Chung, H.Y. Modulation of age-related NF-κB activation by dietary zingerone via MAPK pathway. Exp Gerontol 2010, 45, 419–426. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Jeon, Y.; Kim, T.; Lim, W.-C.; Ham, J.; Park, Y.N.; Kim, T.-J.; Ko, H. Combined treatment with zingerone and its novel derivative synergistically inhibits TGF-β1 induced epithelial-mesenchymal transition, migration and invasion of human hepatocellular carcinoma cells. Bioorg Med Chem Lett 2017, 27, 1081–1088. [Google Scholar] [CrossRef]
- Kumar, L.; Chhibber, S.; Harjai, K. Zingerone inhibit biofilm formation and improve antibiofilm efficacy of ciprofloxacin against Pseudomonas aeruginosa PAO1. Fitoterapia 2013, 90, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Kumar, L.; Chhibber, S.; Harjai, K. Structural alterations in Pseudomonas aeruginosa by zingerone contribute to enhanced susceptibility to antibiotics, serum and phagocytes. Life Sci 2014, 117, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Kumar, L.; Chhibber, S.; Kumar, R.; Kumar, M.; Harjai, K. Zingerone silences quorum sensing and attenuates virulence of Pseudomonas aeruginosa. Fitoterapia 2015, 102, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Kung, M.-L.; Lin, P.-Y.; Huang, S.-T.; Tai, M.-H.; Hsieh, S.-L.; Wu, C.-C.; Yeh, B.-W.; Wu, W.-J.; Hsieh, S. Zingerone Nanotetramer Strengthened the Polypharmacological Efficacy of Zingerone on Human Hepatoma Cell Lines. ACS Appl Mater Interfaces 2018, 11, 137–150. [Google Scholar] [CrossRef]
- Larijanian L, Shafie M, Pirbalouti AG, Ferdousi A, Chiani M (2024) Antibacterial and antibiofilm activities of zingerone and niosomal zingerone against methicillin-resistant Staphylococcus aureus (MRSA)s. Iran J Microbiol. [CrossRef]
- Lee, J.; Oh, S.W.; Shin, S.W.; Lee, K.-W.; Cho, J.-Y.; Lee, J. Zingerone protects keratinocyte stem cells from UVB-induced damage. Chem Biol Interact 2018, 279, 27–33. [Google Scholar] [CrossRef]
- Lee, W.; Ku, S.-K.; Kim, M.-A.; Bae, J.-S. Anti-factor Xa activities of zingerone with anti-platelet aggregation activity. Food and Chemical Toxicology 2017, 105, 186–193. [Google Scholar] [CrossRef]
- Li, L.-L.; Cui, Y.; Guo, X.-H.; Ma, K.; Tian, P.; Feng, J.; Wang, J.-M. Pharmacokinetics and Tissue Distribution of Gingerols and Shogaols from Ginger (Zingiber officinale Rosc.) in Rats by UPLC–Q-Exactive–HRMS. Molecules 2019, 24. [Google Scholar] [CrossRef]
- Li, Y.; Li, D.; Fan, S.; Wang, M.; Li, H.; Dong, Y.; Cui, J.; Duan, Y.; Wu, J. Protective effects of zingerone derivate on ionizing radiation-induced intestinal injury. J Radiat Res 2019, 60, 740–746. [Google Scholar] [CrossRef]
- Liu, L.; Simon, S.A. Similarities and differences in the currents activated by capsaicin, piperine, and zingerone in rat trigeminal ganglion cells. J Neurophysiol 1996, 76, 1858–1869. [Google Scholar] [CrossRef]
- Liu, L.; Welch, J.M.; Erickson, R.P.; Reinhart, P.H.; Simon, S.A. Different responses to repeated applications of zingerone in behavioral studies, recordings from intact and cultured TG neurons, and from VR1 receptors. Physiol Behav 2000, 69, 177–186. [Google Scholar] [CrossRef]
- Mani, V.; Arivalagan, S.; Islam Siddique, A.; Namasivayam, N. Antihyperlipidemic and antiapoptotic potential of zingerone on alcohol induced hepatotoxicity in experimental rats. Chem Biol Interact 2017, 272, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Mani, V.; Arivalagan, S.; Siddique, A.I.; Namasivayam, N. Antioxidant and anti-inflammatory role of zingerone in ethanol-induced hepatotoxicity. Molecular and Cellular Biochemistry 2016, 421, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinale Roscoe). Foods 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Monge P, Scheline R, Solheim E. The Metabolism of Zingerone, a Pungent Principle of Ginger. Xenobiotica 2009, 6, 411–423. [Google Scholar] [CrossRef]
- Nageshwar Rao, B.; Satish Rao, B.S. Antagonistic effects of Zingerone, a phenolic alkanone against radiation-induced cytotoxicity, genotoxicity, apoptosis and oxidative stress in Chinese hamster lung fibroblast cells growing in vitro. Mutagenesis 2010, 25, 577–587. [Google Scholar] [CrossRef]
- Ounjaijean, S.; Somsak, V. Combination of zingerone and dihydroartemisinin presented synergistic antimalarial activity against Plasmodium berghei infection in BALB/c mice as in vivo model. Parasitol Int 2020, 76. [Google Scholar] [CrossRef]
- Pulbutr, P.; Thunchomna, K.; Lawa, K.; Mangkhalat, A.; Saenubol, P. Lipolytic Effects of Zingerone in Adipocytes Isolated from Normal Diet-Fed Rats and High Fat Diet-Fed Rats. International Journal of Pharmacology 2011, 7, 629–634. [Google Scholar] [CrossRef]
- Pulla Reddy, A.C.; Lokesh, B.R. Studies on spice principles as antioxidants in the inhibition of lipid peroxidation of rat liver microsomes. Mol Cell Biochem 1992, 111. [Google Scholar] [CrossRef]
- Rao, B.N.; Archana, P.R.; Aithal, B.K.; Rao, B.S.S. Protective effect of Zingerone, a dietary compound against radiation induced genetic damage and apoptosis in human lymphocytes. Eur J Pharmacol 2011, 657, 59–66. [Google Scholar] [CrossRef]
- Rao, B.N.; Rao, B.S.S.; Aithal, B.K.; Kumar, M.R.S. Radiomodifying and anticlastogenic effect of Zingerone on Swiss albino mice exposed to whole body gamma radiation. Mutat Res Genet Toxicol Environ Mutagen 2009, 677, 33–41. [Google Scholar] [CrossRef]
- Rashid, S.; Wali, A.F.; Rashid, S.M.; Alsaffar, R.M.; Ahmad, A.; Jan, B.L.; Paray, B.A.; Alqahtani, S.M.A.; Arafah, A.; Rehman, U. Zingerone Targets Status Epilepticus by Blocking Hippocampal Neurodegeneration via Regulation of Redox Imbalance, Inflammation and Apoptosis. Pharmaceuticals 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Rawat, S.; Singh, B.; Kumar, R.; Pendem, C.; Bhandari, S.; Natte, K.; Narani, A. Value addition of lignin to zingerone using recyclable AlPO4 and Ni/LRC catalysts. Chem Eng J 2022, 431. [Google Scholar] [CrossRef]
- Sharma, K.; Bose, S.K.; Chhibber, S.; Harjai, K. Exploring the Therapeutic Efficacy of Zingerone Nanoparticles in Treating Biofilm-Associated Pyelonephritis Caused by Pseudomonas aeruginosa in the Murine Model. Inflammation 2020, 43, 2344–2356. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Nirbhavane, P.; Chhibber, S.; Harjai, K. Sustained release of Zingerone from polymeric nanoparticles: An anti-virulence strategy against Pseudomonas aeruginosa. J Bioact Compat Polym 2020, 35, 538–553. [Google Scholar] [CrossRef]
- Shin, S.-G.; Kim, J.Y.; Chung, H.Y.; Jeong, J.-C. Zingerone as an Antioxidant against Peroxynitrite. J Agric Food Chem 2005, 53, 7617–7622. [Google Scholar] [CrossRef]
- Sistani Karampour, N.; Arzi, A.; Rezaie, A.; Pashmforoosh, M.; Kordi, F. Gastroprotective Effect of Zingerone on Ethanol-Induced Gastric Ulcers in Rats. Medicina 2019, 55. [Google Scholar] [CrossRef]
- Songvut, P.; Nakareangrit, W.; Cholpraipimolrat, W.; Kwangjai, J.; Worasuttayangkurn, L.; Watcharasit, P.; Satayavivad, J. Unraveling the interconversion pharmacokinetics and oral bioavailability of the major ginger constituents: [6]-gingerol, [6]-shogaol, and zingerone after single-dose administration in rats. Front Pharmacol 2024, 15, 1391019. [Google Scholar] [CrossRef]
- Stanely Mainzen Prince, P.; Hemalatha, K.L. A molecular mechanism on the antiapoptotic effects of zingerone in isoproterenol induced myocardial infarcted rats. Eur J Pharmacol 2018, 821, 105–111. [Google Scholar] [CrossRef]
- Su, P.; Veeraraghavan, V.P.; Krishna Mohan, S.; Lu, W. A ginger derivative, zingerone—a phenolic compound—induces ROS-mediated apoptosis in colon cancer cells (HCT-116). J Biochem Mol Toxicol 2019, 33, e22403. [Google Scholar] [CrossRef]
- Sunnap, O.; Subramanian, S.; Vemula, P.K.; Karuppannan, S. Zingerone-encapsulated Solid Lipid Nanoparticles as Oral Drug-delivery Systems to Potentially Target Inflammatory Diseases. Chem Nano Mat 2022, 8, e202200388. [Google Scholar] [CrossRef]
- Surh, Y.-J. Molecular mechanisms of chemopreventive effects of selected dietary and medicinal phenolic substances. Mutat Res 1999, 428, 305–327. [Google Scholar] [CrossRef] [PubMed]
- Svetaz, L.; Di Liberto, M.; Zanardi, M.; Suárez, A.; Zacchino, S. Efficient Production of the Flavoring Agent Zingerone and of both (R)- and (S)-Zingerols via Green Fungal Biocatalysis. Comparative Antifungal Activities between Enantiomers. Int J Mol Sci 2014, 15, 22042–22058. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.H.; Nishida, R. Zingerone in the floral synomone of Bulbophyllum baileyi (Orchidaceae) attracts Bactrocera fruit flies during pollination. Biochem Syst Ecol 2007, 35, 334–341. [Google Scholar] [CrossRef]
- Vaibhav, K.; Shrivastava, P.; Tabassum, R.; Khan, A.; Javed, H.; Ahmed, M.E.; Islam, F.; Safhi, M.M.; Islam, F. Delayed administration of zingerone mitigates the behavioral and histological alteration via repression of oxidative stress and intrinsic programmed cell death in focal transient ischemic rats. Pharmacol Biochem Behav 2013, 113, 53–62. [Google Scholar] [CrossRef]
- Vinothkumar, R.; Vinothkumar, R.; Sudha, M.; Nalini, N. Chemopreventive effect of zingerone against colon carcinogenesis induced by 1,2-dimethylhydrazine in rats. Eur J Cancer Prev 2014, 23, 361–371. [Google Scholar] [CrossRef]
- Yang, Z.; Guo, Z.; Yan, J.; Xie, J. Nutritional components, phytochemical compositions, biological properties, and potential food applications of ginger (Zingiber officinale): A comprehensive review. J Food Compos Anal 2024, 128, 106057. [Google Scholar] [CrossRef]
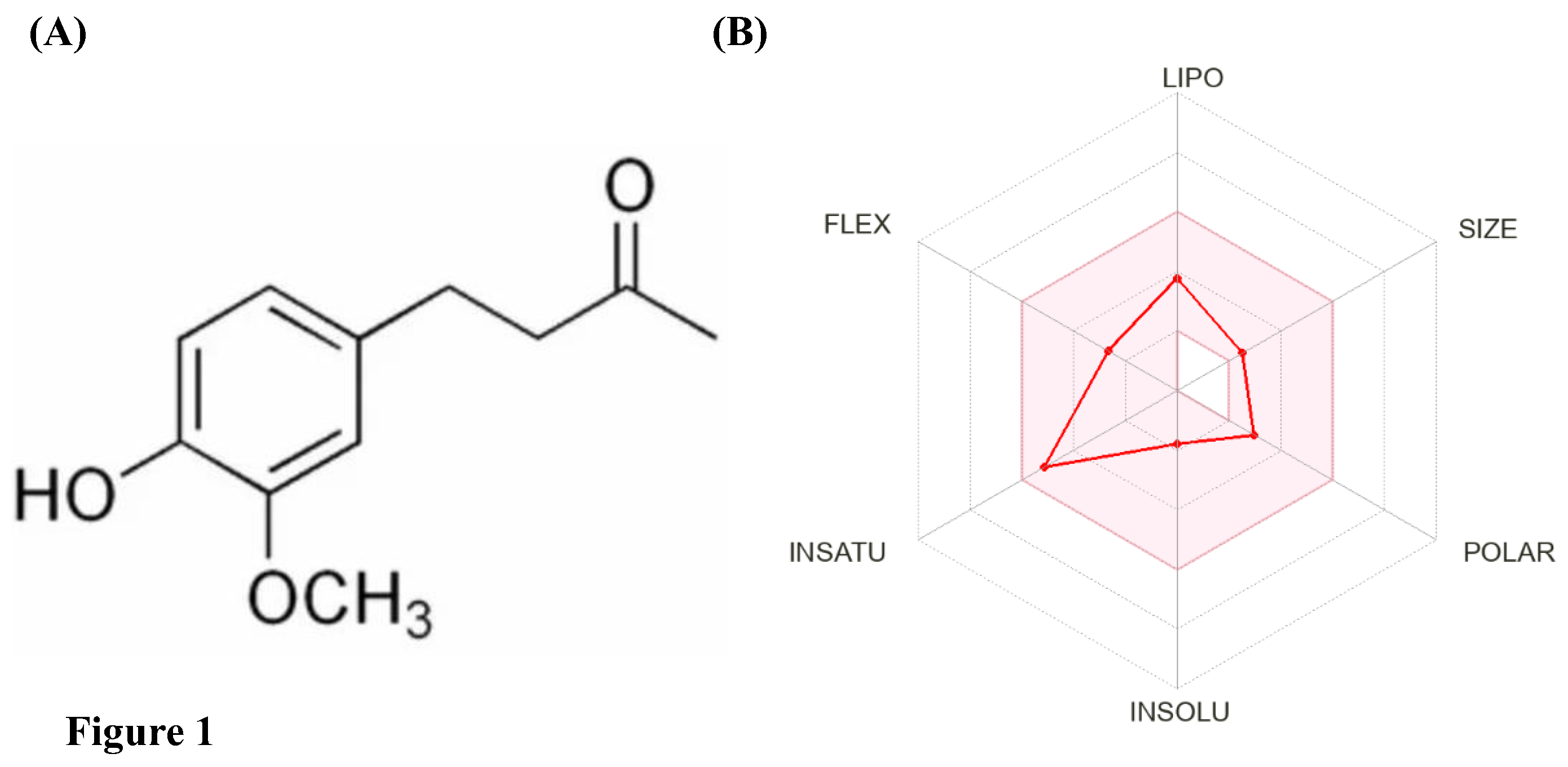
| S. No. | Descriptor | ZiN attributes |
|---|---|---|
| 1 | Molecular weight | 194.23 g/mol |
| 2 | Log P | 1.9224 |
| 3 | Rotatable bond count | 4 |
| 4 | # Acceptors | 3 |
| 5 | # Donors | 1 |
| 6 | Surface area | 83.325 |
| Pharmacokinetic property | Model Name | Predicted value | Measurement units |
|---|---|---|---|
| Absorption | Water solubility | -1.7 | Numeric (log mol/L) |
| Caco2 permeability | 1.233 | Numeric (log Papp in 10-6 cm/s) | |
| Intestinal absorption (human) | 94.103 | Numeric (% Absorbed) | |
| Skin Permeability | -2.653 | Numeric (log Kp) | |
| P-glycoprotein substrate | No | Categorical (Yes/No) | |
| P-glycoprotein I inhibitor | No | Categorical (Yes/No) | |
| P-glycoprotein II inhibitor | No | Categorical (Yes/No) | |
| Distribution | VDss (human) | 0.177 | Numeric (log L/kg) |
| Fraction unbound (human) | 0/407 | Numeric (Fu) | |
| BBB permeability | 0.006 | Numeric (log BB) | |
| CNS permeability | -2.175 | Numeric (log PS) | |
| Metabolism | CYP2D6 substrate | No | Categorical (Yes/No) |
| CYP3A4 substrate | No | Categorical (Yes/No) | |
| CYP1A2 inhibitor | No | Categorical (Yes/No) | |
| CYP2C19 inhibitor | No | Categorical (Yes/No) | |
| CYP2C9 inhibitor | No | Categorical (Yes/No) | |
| CYP2D6 inhibitor | No | Categorical (Yes/No) | |
| CYP3A4 inhibitor | No | Categorical (Yes/No) | |
| Excretion | Total Clearance | 0.307 | Numeric (log ml/min/kg) |
| Renal OCT2 substrate | No | Categorical (Yes/No) | |
| Toxicity | AMES toxicity | No | Categorical (Yes/No) |
| Max. tolerated dose (human) | 0.544 | Numeric (log mg/kg/day) | |
| hERG I inhibitor | No | Categorical (Yes/No) | |
| hERG II inhibitor | No | Categorical (Yes/No) | |
| Oral Rat Acute Toxicity (LD50) | 2.129 | Numeric (mol/kg) | |
| Oral Rat Chronic Toxicity (LOAEL) | 1.953 | Numeric (log mg/kg bw/day) | |
| Hepatotoxicity | Yes | Categorical (Yes/No) | |
| Skin Sensitisation | No | Categorical (Yes/No) | |
| T. pyriformis toxicity | 0.634 | Numeric (log ug/L) | |
| Minnow toxicity | 1.645 | Numeric (log mM) |
| Pharmacological/Biological property reported | Effects described | Reference |
|---|---|---|
| Antimalarial activity | Treatment with ZiN demonstrated potent antimalarial activity in Plasmodium berghei-infected mice, reducing parasitemia by 30.65% and 45.75% at doses of 50 mg/kg and 100 mg/kg, respectively. Additionally, ZiN exhibited a synergistic antimalarial effect when combined with dihydroartemisinin. | (Ounjaijean and Somsak, 2020) |
| Antidiarrheal activity | Intraluminal application of ZiN (30 mM and 50 mM) was reported to inhibit colonic movements via a direct action on smooth muscles in a dose-dependent manner in vitro. These effects were further confirmed by evaluating parameters such as the amplitude of intraluminal pressure changes and fluid output (control: 2.8 ± 0.8 mL/10 min vs. ZiN application: 0.8 ± 0.2 mL/10 min) in vivo. | (Iwami et al. 2010) |
| Antithrombotic activity | ZiN demonstrated anti-factor Xa activity by inhibiting its catalytic activity and platelet aggregation induced by adenosine diphosphate and U46619, leading to a significant reduction in platelet activation markers. Furthermore, it exhibited significant antithrombotic effects in a murine model of arterial thrombosis. | (Lee et al. 2017) |
| Immunomodulatory effect | Treatment with ZiN, in combination with vitamin C, showcased a synergistic influence on erythropoiesis due to their antioxidant activity, along with an increase in total leukocyte count. This combination also enhanced the immune system by promoting the expansion of CD4+ and CD8+ T-lymphocyte populations. | (El Adawy et al. 2025) |
| Anti-fungal activity | ZiN exhibited antifungal activity against Candida albicans and effectively suppressed biofilm formation at a MIC range of 2–4 mg/mL. Its efficacy was further confirmed by validating its antifungal potential in a silkworm model. | (Chougule et al. 2025) |
| Anti-epeliptic effect | Due to its antioxidant and anti-inflammatory potential, ZiN alleviated epilepsy in status epilepticus-induced by lithium chloride and pilocarpine, as well as in maximal electroshock and pentylenetetrazole-induced seizure models. ZiN exerted its protective effects by reducing seizure severity, mitigating oxidative stress, and modulating inflammatory and apoptotic pathways, thereby enhancing neuroprotection. | (Cs and Vincent, 2016; Rashid et al. 2021) |
| Anti-obesity activity | The anti-obesity action of ZiN was evaluated in ovariectomized rats through oral administration (170 mg/kg body weight), which was able to prevent fat storage through the activation of lipolysis. | (Han et al. 2008) |
| Lipolytic effect | ZiN was tested for its lipolytic activity in adipocytes from normal pellet diet-fed and high fat diet-fed rats. At 1000μM, ZiN increased lipolytic effect in normally-fed rats as compared to high fat-fed rats. | (Pulbutr et al. 2011) |
| Anti-emetic effect | ZiN was examined for its antiemetic effect along with other constituents of ginger, which acted as non-competitive antagonist of hydroxytryptamine receptors present in visceral afferent neurons. Despite its lower potency compared to the others, it contributed to the overall antiemetic effect of ginger. | (Jin et al. 2014) |
| Gastroprotective effect | The effects of ZiN on the gastrointestinal tract's interstitial cells of Cajal (ICCs) and its potential as a treatment for GI disorder have been investigated. Via MAPK signaling and NO/cGMP-dependent ATP-sensitive K+ channels, ZiN suppressed pacemaker potentials. Another study confirmed its protective effect against ethanol-induced gastric ulcers in rats. | (Kim et al. 2018; Sistani Karampour et al. 2019) |
| Anti-apoptotic activity | The study elucidated the mechanism of ZiN’s anti-apoptotic effect on a molecular level in rats with myocardial infarction. ZiN (6 mg/kg body weight) pretreatment helped prevent cardiomyocyte apoptosis by modulating genes linked to apoptosis and enhancing antioxidant systems. | (Stanely Mainzen Prince and Hemalatha, 2018) |
| Formulation employed | Effects reported | Reference |
|---|---|---|
| Biodegradable polyester | Synthesized a tissue-like polyester incorporating ZiN-OH (a reduced form of zingerone) with citric acid, sebacic acid, and xylitol. This polyester demonstrated potential applications in tissue engineering, exhibited antibacterial activity with good in vitro biocompatibility. Additionally, it promoted wound healing in mouse fibroblast cells (NIH/3T3). | (Jindal et al. 2024) |
| Solid Lipid Nanoparticles | ZiN was encapsulated into solid-lipid nanoparticles using the encapsulation method. The resulting nanoparticles exhibited remarkable cytocompatibility with sustained drug release, demonstrating a significant anti-inflammatory effect in vitro. | (Sunnap et al. 2022) |
| Nanotetramer (nanoparticle) | One-pot synthesis was used for synthesizing ZiN-NPs with a particle size of 1.42 ± 0.67 nm, which were validated for their antitumor effects on human hepatoma cell lines (SK-Hep-1 and Huh7). ZiN-NPs suppressed Akt activity and NF-κB expression, thereby activating caspases, inciting DNA damage, and resulting in apoptosis. | (Kung et al. 2018) |
| Polymeric Films | Polymeric films prepared from PolyZiN and PolyZiNDimer through the electropolymerization of ZiN and its dimer, were used in construction of amperometric biosensors. These films are effective in shielding interfering species such as ascorbic acid and serve as sustainable alternatives to traditional material like polyphenylenediamine making them highly suitable for biosensor application. | (Caval et al. 2023) |
| Self-assembling peptides derived from fish viscera | ZiN was encapsulated with the help of self-assembling peptide, forming a complex with ZiN, which enhanced the drug release and showed significant antiproliferative effects against colon epithelial Caco-2 cells. | (Huang et al. 2024) |
| Zinc-metal organic framework (Zn-MOF) and noisome hybrid (ZiN-Zn-MOF@Nio) | A ZiN-loaded Zn-MOF@Nio hybrid nanocomposite was prepared with an encapsulation efficiency of 92.56% and a loading capacity of 11.55%. It demonstrated antibacterial activity against S. aureus and B. subtilis (MIC ~ 31.25 µg/mL) as well as E. coli and P. aeruginosa (MIC ~ 62.5 µg/mL). The hybrid demonstrated significant cytotoxicity against MCF-7 breast cancer cells in vitro, with an IC₅₀ value of 46.2 µg/mL, indicating effective anticancer activity. | (Alharbi et al. 2024) |
| Patent Title | Description | Patent No. | Status |
|---|---|---|---|
| Pharmaceutical composition for preventing or treating periodontitis comprising zingerone | The inventors prepared a pharmaceutical composition to prevent periodontitis caused by P. gingivalis. Oral administration of ZiN (40mg/kg) six times over two weeks suppressed periodontitis inflammation and prevented systemic infection. | KR20200013493A | Filed |
| Cosmetic or dermatological preparations containing combinations of zingerone and interface- or surface-active citric acid esters | Preparation of a skin-related formulation of ZiN (0.001- 10% by weight) combined with other surface-active ingredients such as glyceryl stearate citrate or glyceryl stearate tartrate to treat chronic skin aging by stimulating adipocyte differentiation and promoting synthesis and storage of triglycerides. | WO2011063865A2 | Filed |
| Ginger extract for the protection of stem cells | A ginger extract containing 0.001 to 1% b.w. ZiN along with other components was formulated for topical administration to protect stem cells of the hair follicle against UVB irradiation owing to its antioxidant, anti-inflammatory activity in vitro. | US9125936B2 | Granted |
| Methods of Inhibiting Neutrophil Recruitment to the Gingival Crevice | The inventors synthesized an oral care toothpaste incorporating zinc oxide, zinc citrate, stannous fluoride, and ZiN (0.01- 1%) for oral application which modulates proteins and controls neutrophil recruitment in the gums. | US20220071868A1 | Filed |
| A kind of gingerone compound micropowder preparation for reducing blood sugar in type II diabetes and preparation method thereof | The invention presents the method for preparation of micro powder formulations containing ZiN (0.75%-1.5%) which aimed at reducing blood sugar levels in individuals with type II diabetes. The formulation was tested in type Ⅱ diabetic mice, demonstrating a hypoglycemic effect with enhanced glucose tolerance. | CN108553551B | Granted |
| Use of zingerone or its derivatives to reduce or delay the signs of skin aging | The inventors utilized ZiN (2 μg/mL and 20 μg/mL) to activate keratinocyte differentiation by increasing filaggrin protein levels, thereby restoring skin thickness and delaying skin aging. It also helped in the reduction of dryness and restoring hydration. | JP6282582B2 | Granted |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




