Submitted:
28 March 2025
Posted:
31 March 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
Phosphate Homeostasis
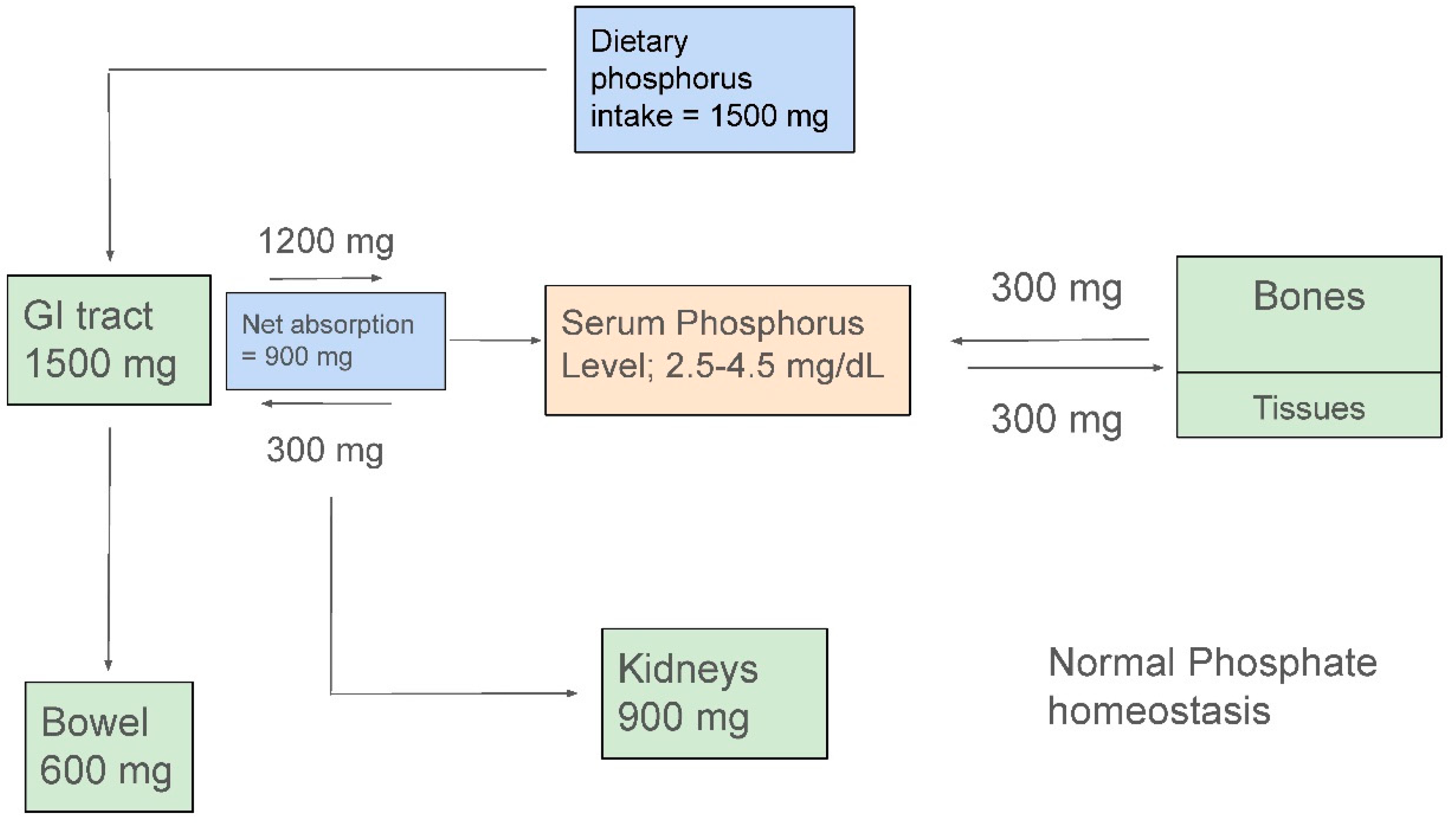
Phosphate Balance in Health:
Intestinal Phosphorus Handling
Renal Phosphate Handling
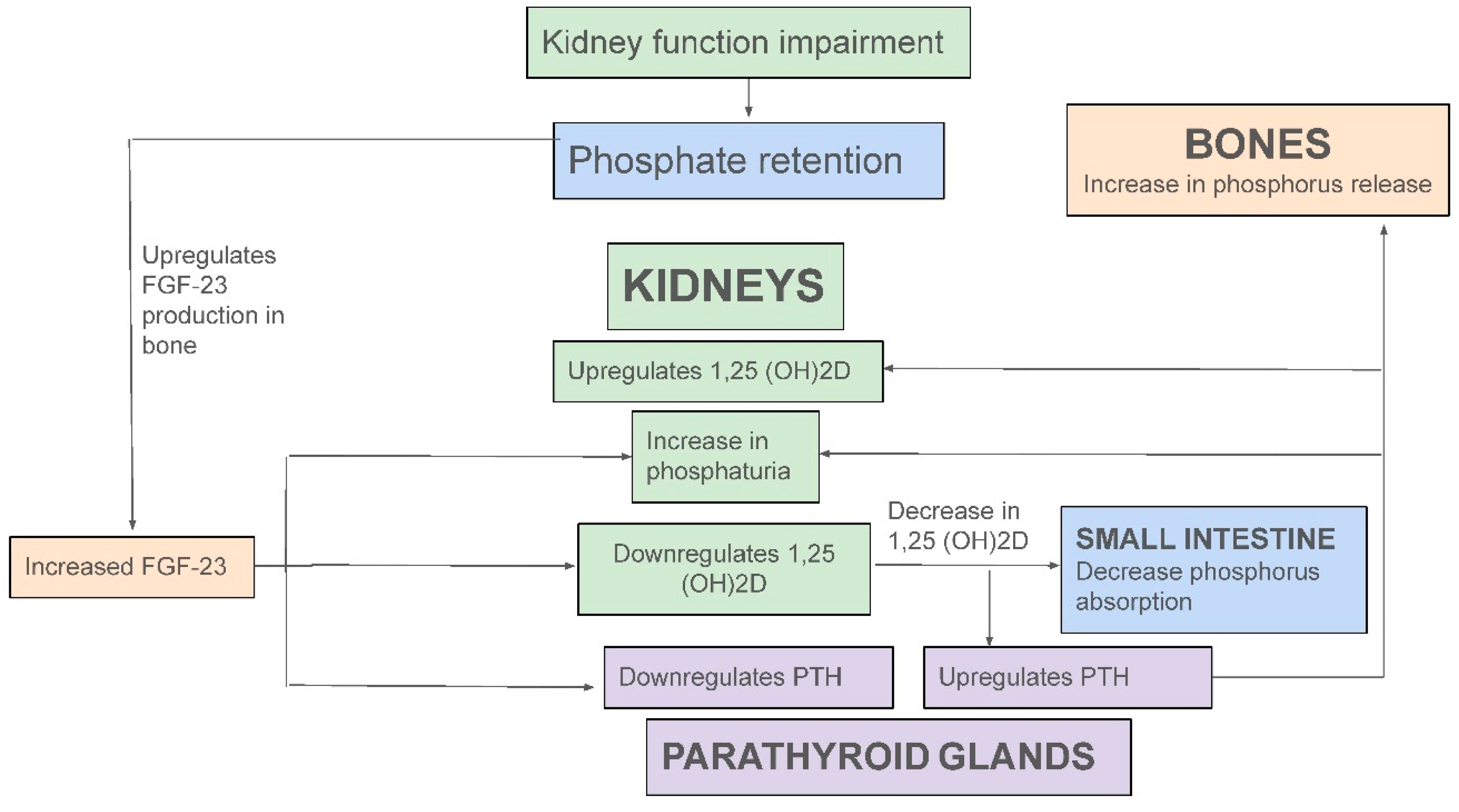
Endocrine Regulation of Phosphate Homeostasis in Health and Kidney Disease
Complications and Outcomes Associated with Hyperphosphatemia
Hyperphosphatemia: Cardiovascular Risks and Mortality
Hyperphosphatemia and Risk of Mortality and Progression of Renal Disease
Hyperphosphatemia and Metabolic Bone Disease
Management of Hyperphosphatemia
Removal of Phosphate by Dialysis
Dietary Management of Hyperphosphatemia
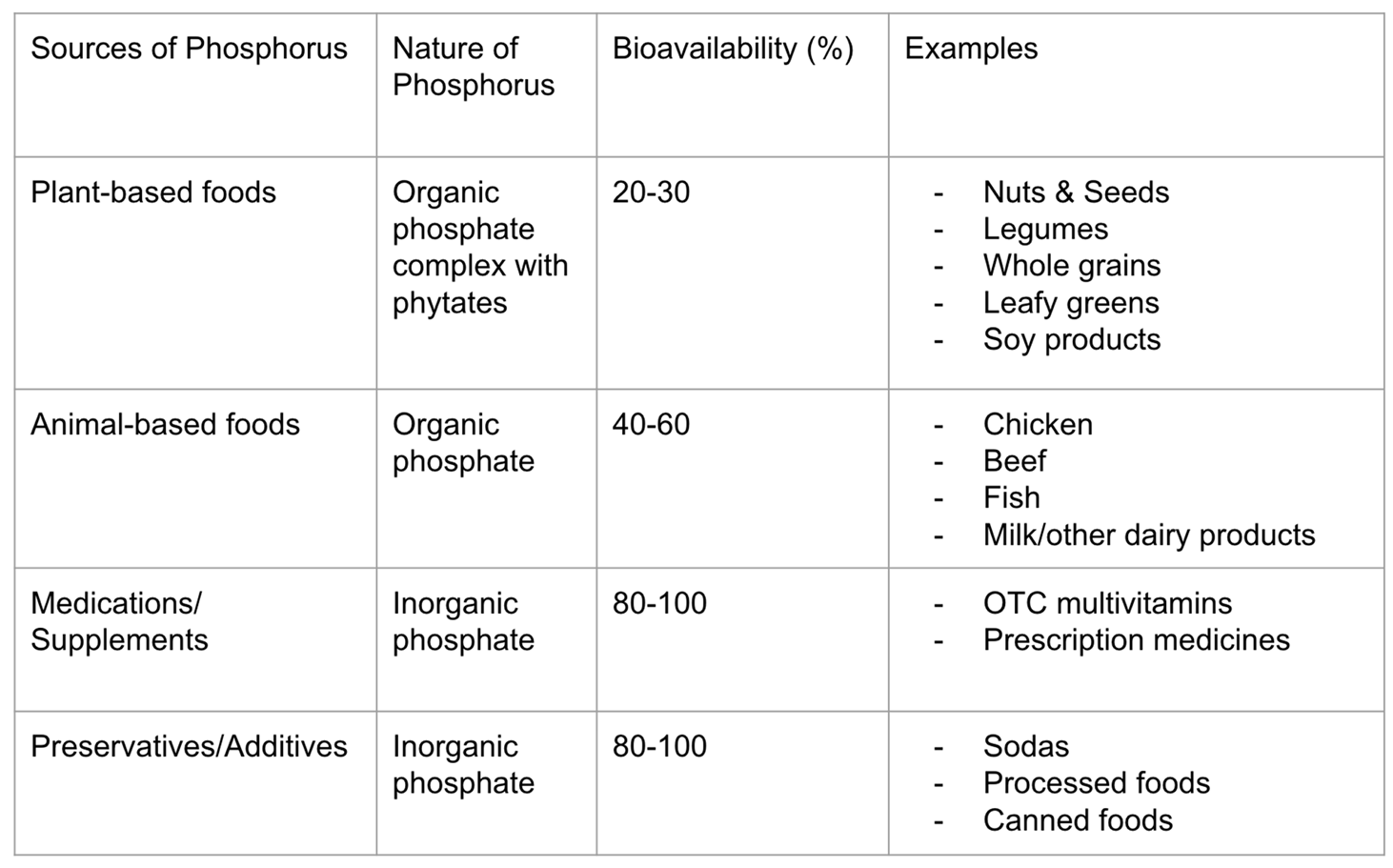
Forms of Phosphorus in the Diet: Organic and Inorganic
Intervention
Reducing Intestinal Phosphate Absorption
Metal-Based Phosphate Binders
Iron-Based Binders: Ferric Citrate and Sucroferric Oxyhydroxide
Drugs Inhibiting Intestinal Phosphate Transport
Controversies and Challenges in the Management of Hyperphosphatemia
Disclosures
References
- Heaney, R.P. Phosphorus. In Present Knowledge in Nutrition, 10th ed.; Erdman, J.W., Macdonald, I.A., Zeisel, S.H., Eds.; Wiley-Blackwell: Washington, DC, 2012; pp. 447–458. [Google Scholar]
- Hu, M.C.; Moe, O.W. Phosphate and Cellular Senescence. Adv. Exp. Med. Biol. 2022, 1362, 55–72. [Google Scholar] [PubMed]
- Phosphorus. Facts Sheet For Health Professionals. https://ods.od.nih.gov/factsheets/Phosphorus-HealthProfessional/#en1, updated May 4, 2023. (accessed on 24 March 2025).
- The United States Renal Data System Annual Data Report 2024. www.usrds.org. (accessed on 24 March 2025).
- Shroff, R.; Long, D.A.; Shanahan, C. Mechanistic Insights into Vascular Calcification in CKD. J. Am. Soc. Nephrol. 2013, 24, 179–189. [Google Scholar] [CrossRef]
- Jono, S.; McKee, M.D.; Murry, C.E.; Shioi, A.; Nishizawa, Y.; Mori, K.; Morii, H.; Giachelli, C.M. Phosphate Regulation of Vascular Smooth Muscle Cell Calcification. Circ. Res. 2000, 87, E10–E17. [Google Scholar] [CrossRef] [PubMed]
- Paloian, N.J.; Giachelli, C.M. A current understanding of vascular calcification in CKD. Am. J. Physiol. Physiol. 2014, 307, F891–F900. [Google Scholar] [CrossRef]
- Isakova, T.; on behalf of the Chronic Renal Insufficiency Cohort (CRIC) Study Group; Wahl, P.; Vargas, G.S.; Gutiérrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Simic, P.; Zhou, I.Y.; Caravan, P.; Parada, X.V.; Wen, D.; Washington, O.L.; Shvedova, M.; Pierce, K.A.; Clish, C.B.; et al. Kidney glycolysis serves as a mammalian phosphate sensor that maintains phosphate homeostasis. J. Clin. Investig. 2023, 133, e164610. [Google Scholar] [CrossRef]
- Sabbagh, Y.; Giral, H.; Caldas, Y.; Levi, M.; Schiavi, S.C. Intestinal Phosphate Transport. Adv. Chronic Kidney Dis. 2011, 18, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.; Hasegawa, H.; Yamazaki, Y.; Muto, T.; Hino, R.; Takeuchi, Y.; Fujita, T.; Nakahara, K.; Fukumoto, S.; Yamashita, T. FGF-23 Is a Potent Regulator of Vitamin D Metabolism and Phosphate Homeostasis. J. Bone Miner. Res. 2004, 19, 429–435. [Google Scholar] [CrossRef]
- Berndt, T.J. Kumar R: Clinical Disturbances of Phosphate Homeostasis. In Seldin and Giebisch’s The Kidney, Fifth Edition ed. [CrossRef]
- Walton, J.; Gray, T.K. Absorption of Inorganic Phosphate in the Human Small Intestine. Clin. Sci. 1979, 56, 407–412. [Google Scholar] [CrossRef]
- Danisi, G.; Straub, R.W. Unidirectional influx of phosphate across the mucosal membrane of rabbit small intestine. Pfl?gers Arch. Eur. J. Physiol. 1980, 385, 117–122. [Google Scholar] [CrossRef]
- Davis, G.R.; Zerwekh, J.E.; Parker, T.F.; Krejs, G.J.; Pak, C.; Fordtran, J.S. Absorption of phosphate in the jejunum of patients with chronic renal failure before and after correction of vitamin D deficiency. Gastroenterology 1983, 85, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Marks, J.; Debnam, E.S.; Unwin, R.J. The role of the gastrointestinal tract in phosphate homeostasis in health and chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2013, 22, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Sabbagh, Y.; O'Brien, S.P.; Song, W.; Boulanger, J.H.; Stockmann, A.; Arbeeny, C.; Schiavi, S.C. Intestinal Npt2b Plays a Major Role in Phosphate Absorption and Homeostasis. J. Am. Soc. Nephrol. 2009, 20, 2348–2358. [Google Scholar] [CrossRef]
- Marks, J.; Debnam, E.S.; Unwin, R.J. Phosphate homeostasis and the renal-gastrointestinal axis. Am. J. Physiol. Physiol. 2010, 299, F285–F296. [Google Scholar] [CrossRef]
- Larsson, T.E.; Kameoka, C.; Nakajo, I.; Taniuchi, Y.; Yoshida, S.; Akizawa, T.; Smulders, R.A. NPT-IIb Inhibition Does Not Improve Hyperphosphatemia in CKD. Kidney Int. Rep. 2018, 3, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Saurette, M.; Alexander, R.T. Intestinal phosphate absorption: the paracellular pathway predominates? Exp Biol Med. 2019, 244, 646–654. [Google Scholar] [CrossRef]
- Knöpfel, T.; Himmerkus, N.; Günzel, D.; Bleich, M.; Hernando, N.; Wagner, C.A. Paracellular transport of phosphate along the intestine. Am. J. Physiol. Liver Physiol. 2019, 317, G233–G241. [Google Scholar] [CrossRef]
- Lee, D.B.; Walling, M.W.; Corry, D.B.; Candeal, E.; Caldas, Y.A.; Guillén, N.; Levi, M.; Sorribas, V.; Marks, J.; Debnam, E.S.; et al. Phosphate transport across rat jejunum: influence of sodium, pH, and 1,25-dihydroxyvitamin D3. Am. J. Physiol. Liver Physiol. 1986, 251, G90–G95. [Google Scholar] [CrossRef]
- King, A.J.; Siegel, M.; He, Y.; Nie, B.; Wang, J.; Koo-McCoy, S.; Minassian, N.A.; Jafri, Q.; Pan, D.; Kohler, J.; et al. Inhibition of sodium/hydrogen exchanger 3 in the gastrointestinal tract by tenapanor reduces paracellular phosphate permeability. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Dudeja, P.K.; Rao, D.D.; Syed, I.; Joshi, V.; Dahdal, R.Y.; Gardner, C.; Risk, M.C.; Schmidt, L.; Bavishi, D.; Kim, K.E.; et al. Intestinal distribution of human Na+/H+ exchanger isoforms NHE-1, NHE-2, and NHE-3 mRNA. Am. J. Physiol. Liver Physiol. 1996, 271, G483–G493. [Google Scholar] [CrossRef]
- Rosenbaum, D.P.; Yan, A.; Jacobs, J.W. Pharmacodynamics, Safety, and Tolerability of the NHE3 Inhibitor Tenapanor: Two Trials in Healthy Volunteers. Clin. Drug Investig. 2018, 38, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Block, G.A.; Rosenbaum, D.P.; Yan, A.; Chertow, G.M. Efficacy and Safety of Tenapanor in Patients with Hyperphosphatemia Receiving Maintenance Hemodialysis: A Randomized Phase 3 Trial. J. Am. Soc. Nephrol. 2019, 30, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, S.; Miyagawa, A.; Kaneko, I.; Shiozaki, Y.; Segawa, H.; Miyamoto, K.-I. Regulation of renal phosphate handling: inter-organ communication in health and disease. J. Bone Miner. Metab. 2015, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lederer, E. Renal phosphate transporters. Curr. Opin. Nephrol. Hypertens. 2014, 23, 502–506. [Google Scholar] [CrossRef]
- Erben, R.G. Physiological Actions of Fibroblast Growth Factor-23. Front. Endocrinol. 2018, 9, 267. [Google Scholar] [CrossRef]
- Martin, A.; David, V.; Quarles, L.D. Regulation and Function of the FGF23/Klotho Endocrine Pathways. Physiol. Rev. 2012, 92, 131–155. [Google Scholar] [CrossRef]
- Lee, M.; Partridge, N.C. Parathyroid hormone signaling in bone and kidney. Curr. Opin. Nephrol. Hypertens. 2009, 18, 298–302. [Google Scholar] [CrossRef]
- Bargagli, M.; Arena, M.; Naticchia, A.; Gambaro, G.; Mazzaferro, S.; Fuster, D.; Ferraro, P.M. The Role of Diet in Bone and Mineral Metabolism and Secondary Hyperparathyroidism. Nutrients 2021, 13, 2328. [Google Scholar] [CrossRef]
- Zhou, W.; Simic, P.; Zhou, I.Y.; Caravan, P.; Parada, X.V.; Wen, D.; Washington, O.L.; Shvedova, M.; Pierce, K.A.; Clish, C.B.; et al. Kidney glycolysis serves as a mammalian phosphate sensor that maintains phosphate homeostasis. J. Clin. Investig. 2023, 133, e164610. [Google Scholar] [CrossRef]
- Ritz, E.; Gross, M.-L. Hyperphosphatemia in renal failure. Blood Purif 2005, 23, 6–9. [Google Scholar] [CrossRef]
- Chen, W.; Bushinsky, D. Chronic kidney disease–mineral and bone disorder. In Handbook of Dialysis Therapy, 5th edn.; Nissenson, A.R., Fine, R.N.B.T., Eds.; Elsevier, 2017; pp. 685–697. [Google Scholar]
- Block, G.A.; Hulbert-Shearon, T.E.; Levin, N.W.; Port, F.K. Association of serum phosphorus and calcium x phosphate product with mortality risk in chronic hemodialysis patients: A national study. Am. J. Kidney Dis. 1998, 31, 607–617. [Google Scholar] [CrossRef]
- Dhingra, R.; Sullivan, L.M.; Fox, C.S.; Wang, T.J.; D’agostino, R.B.; Gaziano, J.M.; Vasan, R.S. Relations of Serum Phosphorus and Calcium Levels to the Incidence of Cardiovascular Disease in the Community. Arch. Intern. Med. 2007, 167, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Hayen, A.; Macaskill, P.; Pellegrini, F.; Craig, J.C.; Elder, G.J.; Strippoli, G.F. Serum levels of phosphorus, parathyroid hormone, and calcium and risks of death and cardiovascular disease in individuals with chronic kidney disease: a systematic review and meta-analysis. JAMA 2011, 305, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.; Trivedi, B.K.; Kalantar-Zadeh, K.; Kovesdy, C.P. Association of Disorders in Mineral Metabolism with Progression of Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2006, 1, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Liu, M.; Li, H.; Luo, Z.; Zhong, X.; Huang, J.; Liu, R.; He, F.; Fu, J. Mortality and Associated Risk Factors in Dialysis Patients with Cardiovascular Disease. Kidney Blood Press. Res. 2016, 41, 479–487. [Google Scholar] [CrossRef]
- Cozzolino, M.; Mangano, M.; Stucchi, A.; Ciceri, P.; Conte, F.; Galassi, A. Cardiovascular disease in dialysis patients. Nephrol. Dial. Transplant. 2018, 33, iii28–iii34. [Google Scholar] [CrossRef]
- Ganesh, S.K.; Stack, A.G.; Levin, N.W.; Hulbert-Shearon, T.; Port, F.K. Association of Elevated Serum PO4, Ca × PO4 Product, and Parathyroid Hormone with Cardiac Mortality Risk in Chronic Hemodialysis Patients. J. Am. Soc. Nephrol. 2001, 12, 2131–2138. [Google Scholar] [CrossRef]
- Cozzolino, M.; Dusso, A.S.; Slatopolsky, E. Role of Calcium-Phosphate Product and Bone-Associated Proteins on Vascular Calcification in Renal Failure. J. Am. Soc. Nephrol. 2001, 12, 2511–2516. [Google Scholar] [CrossRef]
- Zhang, D.; Bi, X.; Liu, Y.; Huang, Y.; Xiong, J.; Xu, X.; Xiao, T.; Yu, Y.; Jiang, W.; Huang, Y.; et al. High Phosphate-Induced Calcification of Vascular Smooth Muscle Cells is Associated with the TLR4/NF-κb Signaling Pathway. Kidney Blood Press. Res. 2017, 42, 1205–1215. [Google Scholar] [CrossRef]
- Amann, K. Media Calcification and Intima Calcification Are Distinct Entities in Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1599–1605. [Google Scholar] [CrossRef]
- El-Abbadi, M.M.; Pai, A.S.; Leaf, E.M.; et al. Phosphate feeding induces arterial medial calcification in uremic mice: role of serum phosphorus, fibroblast growth factor-23, and osteopontin. Kidney Int 2009, 75, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Pai, A.S.; Giachelli, C.M. Matrix remodeling in vascular calcification associated with chronic kidney disease. J Am Soc Nephrol 2010, 21, 1637–1640. [Google Scholar] [CrossRef]
- Luong, T.T.D.; Schelski, N.; Boehme, B.; Makridakis, M.; Vlahou, A.; Lang, F.; Pieske, B.; Alesutan, I.; Voelkl, J. Fibulin-3 Attenuates Phosphate-Induced Vascular Smooth Muscle Cell Calcification by Inhibition of Oxidative Stress. Cell. Physiol. Biochem. 2018, 46, 1305–1316. [Google Scholar] [CrossRef] [PubMed]
- Voelkl, J.; Lang, F.; Eckardt, K.; et al. Signaling pathways involved in vascular smooth muscle cell calcification during hyperphosphatemia. Cell Mol Life Sci 2019, 76, 2077–2091. [Google Scholar] [CrossRef]
- Faul, C.; Amaral, A.P.; Oskouei, B.; Hu, M.-C.; Sloan, A.; Isakova, T.; Gutiérrez, O.M.; Aguillon-Prada, R.; Lincoln, J.; Hare, J.M.; et al. FGF23 induces left ventricular hypertrophy. J. Clin. Investig. 2011, 121, 4393–4408. [Google Scholar] [CrossRef]
- Wannamethee, S.G.; Welsh, P.; Papacosta, O.; Lennon, L.; Whincup, P.H.; Sattar, N. Elevated Parathyroid Hormone, But Not Vitamin D Deficiency, Is Associated With Increased Risk of Heart Failure in Older Men With and Without Cardiovascular Disease. Circ. Hear. Fail. 2014, 7, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Vogt, I.; Haffner, D.; Leifheit-Nestler, M. FGF23 and Phosphate–Cardiovascular Toxins in CKD. Toxins 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Ix, J.H.; Katz, R.; Kestenbaum, B.R.; de Boer, I.H.; Chonchol, M.; Mukamal, K.J.; Rifkin, D.; Siscovick, D.S.; Sarnak, M.J.; Shlipak, M.G. Fibroblast Growth Factor-23 and Death, Heart Failure, and Cardiovascular Events in Community-Living Individuals. CHS (Cardiovascular Health Study) J Am Coll Cardiol. 2012, 60, 200–207. [Google Scholar] [CrossRef]
- Edmonston, D.; Grabner, A.; Wolf, M. FGF23 and klotho at the intersection of kidney and cardiovascular disease. Nat. Rev. Cardiol. 2023, 21, 11–24. [Google Scholar] [CrossRef]
- Zhong, Z.; Feng, S.; Fu, D.; Li, B.; Li, Z.; Mao, H. Serum fibroblast growth factor 23 concentration and the risk of mortality in patients undergoing peritoneal dialysis. Perit. Dial. Int. J. Int. Soc. Perit. Dial. 2024, 44, 194–202. [Google Scholar] [CrossRef]
- Cheng, S.-P.; Liu, C.-L.; Liu, T.-P.; Hsu, Y.-C.; Lee, J.-J. Association between Parathyroid Hormone Levels and Inflammatory Markers among US Adults. Mediat. Inflamm. 2014, 2014, 709024. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, D.J.; Covic, A.A.; Venning, M.C.; Ackrill, P. Blood pressure reduction after parathyroidectomy for secondary hyperparathyroidism: Further evidence implicating calcium homeostasis in blood pressure regulation. Am. J. Kidney Dis. 1996, 27, 819–825. [Google Scholar] [CrossRef]
- Smogorzewski, M.; Perna, A.F.; Borum, P.R.; Massry, S.G. Fatty acid oxidation in the myocardium: Effects of parathyroid hormone and CRF. Kidney Int. 1988, 34, 797–803. [Google Scholar] [CrossRef]
- Rodríguez-Ayala, E.; Ávila-Díaz, M.; Foyo-Niembro, E.; Amato, D.; Ramirez-San-Juan, E.; Paniagua, R. Effect of Parathyroidectomy on Cardiac Fibrosis and Apoptosis: Possible Role of Aldosterone. Nephron Physiol. 2006, 103, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Saleh, F.; Schirmer, H.; Sundsfjord, J.; Jorde, R. Parathyroid hormone and left ventricular hypertrophy. Eur Heart J. 2003, 24, 2054–2060. [Google Scholar] [CrossRef]
- Li, Y.; Chen, C.; Liu, H.L.; Qian, G. Vitamin D, Parathyroid Hormone, and Heart Failure in A Chinese Elderly Population. Endocr. Pr. 2015, 21, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Kestenbaum, B.; Sampson, J.N.; Rudser, K.D.; Patterson, D.J.; Seliger, S.L.; Young, B.; Sherrard, D.J.; Andress, D.L. Serum Phosphate Levels and Mortality Risk among People with Chronic Kidney Disease. J. Am. Soc. Nephrol. 2005, 16, 520–528. [Google Scholar] [CrossRef]
- Voormolen, N.; Noordzij, M.; Grootendorst, D.C.; Beetz, I.; Sijpkens, Y.W.; van Manen, J.G.; Boeschoten, E.W.; Huisman, R.M.; Krediet, R.T.; Dekker, F.W.; et al. High plasma phosphate as a risk factor for decline in renal function and mortality in pre-dialysis patients. Nephrol. Dial. Transplant. 2007, 22, 2909–2916. [Google Scholar] [CrossRef]
- Tonelli, M.; Curhan, G.; Pfeffer, M.; Sacks, F.; Thadhani, R.; Melamed, M.L.; Wiebe, N.; Muntner, P. Relation between alkaline phosphatase, serum phosphate, and all-cause or cardiovascular mortality. Circulation 2009, 120, 1784–1792. [Google Scholar] [CrossRef]
- Li, J.-W.; Xu, C.; Fan, Y.; Wang, Y.; Xiao, Y.-B. Can Serum Levels of Alkaline Phosphatase and Phosphate Predict Cardiovascular Diseases and Total Mortality in Individuals with Preserved Renal Function? A Systemic Review and Meta-Analysis. PLOS ONE 2014, 9, e102276. [Google Scholar] [CrossRef]
- Schwarz, S.; Trivedi, B.K.; Kalantar-Zadeh, K.; Kovesdy, C.P. Association of Disorders in Mineral Metabolism with Progression of Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2006, 1, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Maique, J.; Flores, B.; Shi, M.; Shepard, S.; Zhou, Z.; Yan, S.; Moe, O.W.; Hu, M.C. High Phosphate Induces and Klotho Attenuates Kidney Epithelial Senescence and Fibrosis. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.-J.; Hu, J.; Shiizaki, K.; Kuro-O, M.; Malter, J.S. Phosphate–Induced Renal Fibrosis Requires the Prolyl Isomerase Pin1. PLOS ONE 2016, 11, e0150093. [Google Scholar] [CrossRef]
- Sim, J.J.; Bhandari, S.K.; Smith, N.; Chung, J.; Liu, I.L.A.; Jacobsen, S.J.; Kalantar-Zadeh, K. Phosphorus and Risk of Renal Failure in Subjects with Normal Renal Function. Am. J. Med. 2013, 126, 311–318. [Google Scholar] [CrossRef]
- Zoccali, C.; Ruggenenti, P.; Perna, A.; Leonardis, D.; Tripepi, R.; Tripepi, G.; Mallamaci, F.; Remuzzi, G. ; REINStudy Group Phosphate may promote CKDprogression attenuate renoprotective effect of ACEinhibition, J. Am. Soc. Nephrol. 2011, 22, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Da, J.; Xie, X.; Wolf, M.; Disthabanchong, S.; Wang, J.; Zha, Y.; Lv, J.; Zhang, L.; Wang, H. Serum Phosphorus and Progression of CKD and Mortality: A Meta-analysis of Cohort Studies. Am. J. Kidney Dis. 2015, 66, 258–265. [Google Scholar] [CrossRef]
- Isakova, T.; Wahl, P.; Vargas, G.S.; Gutiérrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; et al.; on behalf of the Chronic Renal Insufficiency Cohort (CRIC) Study Group Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef]
- Barrera-Baena, P.; Rodríguez-García, M.; Rodríguez-Rubio, E.; González-Llorente, L.; Ortiz, A.; Zoccali, C.; Locatelli, F.; Floege, J.; Cohen-Solal, M.; Ferreira, M.A.; et al. Serum phosphate is associated with increased risk of bone fragility fractures in haemodialysis patients. Nephrol. Dial. Transplant. 2023, 39, 618–626. [Google Scholar] [CrossRef]
- Tentori, F.; McCullough, K.; Kilpatrick, R.D.; Bradbury, B.D.; Robinson, B.M.; Kerr, P.G.; Pisoni, R.L. High rates of death and hospitalization follow bone fracture among hemodialysis patients. Kidney Int. 2014, 85, 166–173. [Google Scholar] [CrossRef]
- Fusaro, M.; Holden, R.; Lok, C.; Iervasi, G.; Plebani, M.; Aghi, A.; Gallieni, M.; Cozzolino, M. Phosphate and bone fracture risk in chronic kidney disease patients. Nephrol. Dial. Transplant. 2019, 36, 405–412. [Google Scholar] [CrossRef]
- Meleti, Z.; Shapiro, I.; Adams, C. Inorganic phosphate induces apoptosis of osteoblast-like cells in culture. Bone 2000, 27, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Mozar, A.; Haren, N.; Chasseraud, M.; Louvet, L.; Mazière, C.; Wattel, A.; Mentaverri, R.; Morlière, P.; Kamel, S.; Brazier, M.; et al. High extracellular inorganic phosphate concentration inhibits RANK–RANKL signaling in osteoclast-like cells. J. Cell. Physiol. 2007, 215, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Guedes, M.; Bieber, B.; Dasgupta, I.; Vega, A.; Nitta, K.; Brunelli, S.; Hartman, J.; Raimann, J.G.; Robinson, B.M.; Pisoni, R.L. Serum Phosphorus Level Rises in US Hemodialysis Patients Over the Past Decade: A DOPPS Special Report. Kidney Med. 2022, 5, 100584. [Google Scholar] [CrossRef]
- KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 1–59. [CrossRef]
- Kuhlmann, M.K. Phosphate Elimination in Modalities of Hemodialysis and Peritoneal Dialysis. Blood Purif. 2010, 29, 137–144. [Google Scholar] [CrossRef] [PubMed]
- ADeSoi, C.; Umans, J.G. Phosphate kinetics during high-flux hemodialysis. J. Am. Soc. Nephrol. 1993, 4, 1214–1218. [Google Scholar] [CrossRef]
- Sugisaki, H.; Onohara, M.; Kunitomo, T. Dynamic behavior of plasma phosphate in chronic dialysis patients. Trans Am Soc Artif Intern Organs. 1982, 28, 302–307. [Google Scholar]
- Zucchelli, P.; Santoro, A. Inorganic Phosphate Removal during Different Dialytic Procedures. Int. J. Artif. Organs 1987, 10, 173–178. [Google Scholar] [CrossRef]
- Haas, T.; Hillion, D.; Dongradi, G. Phosphate kinetics in dialysis patients. Nephrol Dial Transplant. 1991, 6, 108–113. [Google Scholar]
- Man, N.K.; Chauveau, P.; Kuno, T.; Poignet, J.L.; Yanai, M. Phosphate removal during hemodialysis, hemodiafiltration, and hemofiltration. A reappraisal. ASAIO Trans. 1991, 37, M463–5. [Google Scholar] [PubMed]
- Daugirdas, J.T. Removal of Phosphorus by Hemodialysis. Semin. Dial. 2015, 28, 620–623. [Google Scholar] [CrossRef]
- Kooienga, L. Phosphorus Balance with Daily Dialysis. Semin. Dial. 2007, 20, 342–345. [Google Scholar] [CrossRef] [PubMed]
- Minutolo, R.; Bellizzi, V.; Cioffi, M.; Iodice, C.; Giannattasio, P.; Andreucci, M.; Terracciano, V.; Di Iorio, B.R.; Conte, G.; De Nicola, L. Postdialytic rebound of serum phosphorus: pathogenetic and clinical insights. . 2002, 13, 1046–1054. [Google Scholar] [CrossRef]
- Stremke, E.R.; Trevino, L.; Doshi, S.; Moorthi, R.N.; Gallant, K.M.H.; Moe, S.M. Postdialysis serum phosphate equilibrium in hemodialysis patients on a controlled diet and no binders. Hemodial. Int. 2021, 26, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Perl, J.; Bargman, J.M. Peritoneal dialysis: from bench to bedside and bedside to bench. Am. J. Physiol. Physiol. 2016, 311, F999–F1004. [Google Scholar] [CrossRef]
- Courivaud, C.; Davenport, A. Phosphate Removal by Peritoneal Dialysis: The Effect of Transporter Status and Peritoneal Dialysis Prescription. Perit. Dial. Int. J. Int. Soc. Perit. Dial. 2016, 36, 85–93. [Google Scholar] [CrossRef]
- Bammens, B.; Evenepoel, P.; Verbeke, K.; Vanrenterghem, Y. Removal of middle molecules and protein-bound solutes by peritoneal dialysis and relation with uremic symptoms. Kidney Int. 2003, 64, 2238–2243. [Google Scholar] [CrossRef] [PubMed]
- Debowska, M.; Gomez, R.; Pinto, J.; Waniewski, J.; Lindholm, B. Phosphate clearance in peritoneal dialysis. Sci. Rep. 2020, 10, 17504. [Google Scholar] [CrossRef]
- Peruzzo, D.; Guedes, M.; Larkin, J.W.; Yokoyama, G.; dos Santos, T.L.; Pecoits-Filho, R.; Ribeiro, S.C.; Ramos, A.; Barretti, P.; de Moraes, T.P.; et al. Peritoneal dialysis modality transition and impact on phosphate and potassium serum levels. PLOS ONE 2021, 16, e0257140. [Google Scholar] [CrossRef]
- Calvo, M.S.; Park, Y.K. Changing Phosphorus Content of the U.S. Diet: Potential for Adverse Effects on Bone. J. Nutr. 1996, 126, 1168S–1180S. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Gutekunst, L.; Mehrotra, R.; Kovesdy, C.P.; Bross, R.; Shinaberger, C.S.; Noori, N.; Hirschberg, R.; Benner, D.; Nissenson, A.R.; et al. Understanding Sources of Dietary Phosphorus in the Treatment of Patients with Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.S.; Uribarri, J. Dietary phosphorus intake and the risk for cardiovascular disease in the general population. Advances in Chronic Kidney Disease 2011, 18, 266–272. [Google Scholar]
- Dang, Z.; He, Y.; Xie, R.; Chen, P.; Dong, F. Plant-based Diet and Chronic Kidney Disease: A Systematic Review and Meta-analysis. J. Ren. Nutr. 2025. [Google Scholar] [CrossRef] [PubMed]
- Huml, A.M.; Sullivan, C.M.; Leon, J.B.; Sehgal, A.R. The Adequacy of Phosphorus Binder Prescriptions among American Hemodialysis Patients. Ren. Fail. 2012, 34, 1258–1263. [Google Scholar] [CrossRef]
- Brown-Tortorici, A.R.; Narasaki, Y.; You, A.S.; Norris, K.C.; Streja, E.; Peralta, R.A.; Guerrero, Y.; Daza, A.; Arora, R.; Lo, R.; et al. The Interplay between Dietary Phosphorous, Protein Intake, and Mortality in a Prospective Hemodialysis Cohort. Nutrients 2022, 14, 3070. [Google Scholar] [CrossRef]
- Fenton, T.R.; et al. Phosphorus additives in food and their impact on human health. Current Opinion in Clinical Nutrition & Metabolic Care 2017, 20, 192–196. [Google Scholar]
- Younes, M., Aquilina; et al. Re-evaluation of phosphoric acid–phosphates – di-, tri- and polyphosphates (E 338–341, E 343, E 450–452) as food additives and the safety of proposed extension of use. EFSA J. 2019, 17, e05674. [Google Scholar] [CrossRef]
- Capra, B.T.; Hudson, S.; Helder, M.; Laskaridou, E.; Johnson, A.L.; Gilmore, C.; Marinik, E.; Hedrick, V.E.; Savla, J.; David, L.A.; et al. Ultra-processed food intake, gut microbiome, and glucose homeostasis in mid-life adults: Background, design, and methods of a controlled feeding trial. Contemp. Clin. Trials 2024, 137, 107427–107427. [Google Scholar] [CrossRef]
- FoodNavigator-USA (2014, July 17). Until phosphorus gets on the USDA’s radar, labeling policy won’t change – NKF. FoodNavigator-USA. https://www.foodnavigator-usa.com/Article/2014/07/17/Until-phosphorus-gets-on-the-USDA-s-radar-labeling-policy-won-t-change-NKF/. (accessed on 23 March 2025).
- Nelson, S.M.; Sarabia, S.R.; Christilaw, E.; Ward, E.C.; Lynch, S.K.; Adams, M.A.; Holden, R.M. Phosphate-Containing Prescription Medications Contribute to the Daily Phosphate Intake in a Third of Hemodialysis Patients. J. Ren. Nutr. 2017, 27, 91–96. [Google Scholar] [CrossRef]
- Sherman, R.A.; Ravella, S.; Kapoian, T. A dearth of data: the problem of phosphorus in prescription medications. Kidney Int. 2015, 87, 1097–1099. [Google Scholar] [CrossRef]
- Moe, S.M.; Chen, N.X.; Seifert, M.F.; Sinders, R.M.; Duan, D.; Chen, X.; Liang, Y.; Radcliff, J.S.; White, K.E.; Gattone, V.H. , 2nd. A rat model of chronic kidney disease-mineral bone disorder. Kidney Int. 2009, 75, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Zidehsarai, M.P.; Chambers, M.A.; Jackman, L.A.; Radcliffe, J.S.; Trevino, L.L.; Donahue, S.E.; Asplin, J.R. Vegetarian Compared with Meat Dietary Protein Source and Phosphorus Homeostasis in Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Esmaillzadeh, A. Soy-Protein Consumption and Kidney-Related Biomarkers Among Type 2 Diabetics: A Crossover, Randomized Clinical Trial. J. Ren. Nutr. 2009, 19, 479–486. [Google Scholar] [CrossRef]
- Wu, T.; Chang, C.; Hsu, W.; Wang, I.; Hsu, C.; Cheng, S.; Liang, C.; Chang, C.; Huang, C. Nutritional status of vegetarians on maintenance haemodialysis. Nephrology 2011, 16, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Torres, R.; Young, L.; Murray, D.P.; Kheda, M.; Nahman, N.S. Dietary Protein Source and Phosphate Levels in Patients on Hemodialysis. J. Ren. Nutr. 2020, 30, 423–429. [Google Scholar] [CrossRef]
- Kistler, B.M.; Moore, L.W.; Benner, D.; Biruete, A.; Boaz, M.; Brunori, G.; Chen, J.; Drechsler, C.; Guebre-Egziabher, F.; Hensley, M.K.; et al. The International Society of Renal Nutrition and Metabolism Commentary on the National Kidney Foundation and Academy of Nutrition and Dietetics KDOQI Clinical Practice Guideline for Nutrition in Chronic Kidney Disease. J. Ren. Nutr. 2020, 31, 116–120.e1. [Google Scholar] [CrossRef]
- Stevens, P.E.; Ahmed, S.B.; Carrero, J.J.; Foster, B.; Francis, A.; Hall, R.K.; Herrington, W.G.; Hill, G.; Inker, L.A.; Kazancıoğlu, R.; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Cavanaugh, K.L.; İkizler, T.A. Preventing and correcting metabolic acidosis in chronic kidney disease: Implications for bone and mineral disease. Am. J. Kidney Dis. 2013, 61, 508–516. [Google Scholar] [CrossRef]
- Goraya, N.; Wesson, D.E. Whole-Food Low-Protein Plant-Based Nutrition to Prevent or Slow Progression of Chronic Kidney Disease. Journal of Renal Nutrition 2020, 30, 480–488. [Google Scholar]
- KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. American Journal of Kidney Diseases 2020, 76, S1–S107. [CrossRef]
- Zarantonello, D.; Brunori, G. The Role of Plant-Based Diets in Preventing and Mitigating Chronic Kidney Disease: More Light than Shadows. J. Clin. Med. 2023, 12, 6137. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Shah, S.; Kalantar-Zadeh, K. Adequacy of Plant-Based Proteins in Chronic Kidney Disease. J. Ren. Nutr. 2019, 29, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Consultant360. (n.d.). Dietary modification in renal disease. Retrieved from https://www.consultant360.com/blog/consultant360/nephrology/dietary-modification-renal-disease. (accessed on 23 March 2025).
- Escoffier. (n.d.). The true costs of processed foods on health and the planet. Retrieved from https://www.escoffier.edu/blog/world-food-drink/true-costs-processed-foods-health-planet/#:~:text=So%20although%20processed%20food%20is,And%20agricultural%20byproducts%20are%20destroyed. (accessed on 23 March 2025).
- Springmann, M.; AClark, M.; Rayner, M.; Scarborough, P.; Webb, P. The global and regional costs of healthy and sustainable dietary patterns: a modelling study. Lancet Planet. Heal. 2021, 5, e797–e807. [Google Scholar] [CrossRef] [PubMed]
- Pais, D.F.; Marques, A.C.; Fuinhas, J.A. The cost of healthier and more sustainable food choices: Do plant-based consumers spend more on food? Agric. Food Econ. 2022, 10, 1–21. [Google Scholar] [CrossRef]
- Kahleova, H.; Sutton, M.; Maracine, C.; Nichols, D.; Monsivais, P.; Holubkov, R.; Barnard, N.D. Vegan Diet and Food Costs Among Adults With Overweight. JAMA Netw. Open 2023, 6, e2332106. [Google Scholar] [CrossRef]
- GFI (2022). Reducing the price of alternative proteins. Retrieved from https://gfi.org/reducing-the-price-of-alternative-proteins/. (accessed on 23 March 2025).
- Food & Wine. (2024, April 5). Why going vegetarian is a recipe for a cheaper grocery bill. Retrieved from https://www.foodandwine.com/vegetarian-cheapest-groceries-report-11685113. (accessed on 23 March 2025).
- The Guardian. (2024, August 28). "Don't be scared of beans": how readers are handling US grocery inflation. Retrieved from https://www.theguardian.com/environment/article/2024/aug/28/inflation-groceries-tips. (accessed on 23 March 2025).
- Kimura, M.; Itokawa, Y. Cooking losses of minerals in foods and its nutritional significance. J Nutr Sci Vitaminol (Tokyo) 1990, 36 (Suppl 1), S25–S32. discussion S33. [Google Scholar] [CrossRef]
- Jones, W.L. Demineralization of a wide variety of foods for the renal patient. J Ren Nutr. 2001, 11, 90–96. [Google Scholar] [CrossRef]
- Naber, T.; Purohit, S. Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease. Nutrients 2021, 13, 3277. [Google Scholar] [CrossRef]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.-J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. Am. J. Kidney Dis. 2020, 76 (Suppl 1), S1–S107. [Google Scholar] [CrossRef]
- Noce, A.; Marrone, G.; Jones, G.W.; Di Lauro, M.; Zaitseva, A.P.; Ramadori, L.; Celotto, R.; Mitterhofer, A.P.; Di Daniele, N. Nutritional Approaches for the Management of Metabolic Acidosis in Chronic Kidney Disease. Nutrients 2021, 13, 2534. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Bellizzi, V.; Piccoli, G.B.; Shi, Y.; Lim, S.K.; Riaz, S.; Urbina Arronte, R.; Lau, W.P.; Fouque, D. Caring for Patients With Advanced Chronic Kidney Disease: Dietary Considerations. Nutrition in Clinical Practice 2023, 38, 22–34. [Google Scholar]
- US-DOPPS Practice Monitor, May 2021; http://www.dopps.org/DPM.
- Fresenius Medical Care North America. PhosLo® gelcaps (calcium acetate): 667 mg [prescribing information]. Waltham, MA: Fresenius Medical Care North America; 2011.
- Fresenius Medical Care North America. VELPHORO ® (sucroferric oxyhydroxide) [prescribing information]. Waltham, MA: Fresenius Medical Care North America; 2013.
- Shire US Inc. FOSRENAL® (lanthanum carbonate) [prescribing information]. Lexington, MA: Shire US Inc.; 2016.
- Keryx Biopharmaceuticals Inc. AURYXIA® (ferric citrate) tablets [prescribing information]. Cambridge, MA: Keryx Biopharmaceuticals Inc.; 2017.
- Genzyme Corp. RENVELA® (sevelamer carbonate) [prescribing information]. Cambridge, MA: Genzyme Corp.; 2020.
- Salusky, I.; Foley, J.; Nelson, P.; Goodman, W. Aluminum Accumulation during Treatment with Aluminum Hydroxide and Dialysis in Children and Young Adults with Chronic Renal Disease. New Engl. J. Med. 1991, 324, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Elliott, H.L.; Dryburgh, F.; Fell, G.S.; Sabet, S.; IMacdougall, A. Aluminium toxicity during regular haemodialysis. BMJ 1978, 1, 1101–1103. [Google Scholar] [CrossRef]
- Parkinson, I.; Feest, T.; Kerr, D.; Ward, M.; Fawcett, R. FRACTURING DIALYSIS OSTEODYSTROPHY AND DIALYSIS ENCEPHALOPATHY: An Epidemiological Survey. Lancet 1979, 313, 406–409. [Google Scholar] [CrossRef]
- Young, E.W.; Albert, J.M.; Satayathum, S.; Goodkin, D.A.; Pisoni, R.L.; Akiba, T.; Akizawa, T.; Kurokawa, K.; Bommer, J.; Piera, L.; et al. Predictors and consequences of altered mineral metabolism: The Dialysis Outcomes and Practice Patterns Study. Kidney Int. 2005, 67, 1179–1187. [Google Scholar] [CrossRef] [PubMed]
- K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 2003, 42 (Suppl 3), S1–S201. [CrossRef]
- Janssen, M.J.; Van Der Kuy, A.; Ter Wee, P.M.; Van Boven, W.P. Aluminum hydroxide, calcium carbonate and calcium acetate in chronic intermittent hemodialysis patients. Clin Nephrol. 1996, 45, 111–119. [Google Scholar]
- Navaneethan, S.D.; Palmer, S.C.; Craig, J.C.; Elder, G.J.; Strippoli, G.F. Benefits and Harms of Phosphate Binders in CKD: A Systematic Review of Randomized Controlled Trials. Am. J. Kidney Dis. 2009, 54, 619–637. [Google Scholar] [CrossRef]
- Suki, W.; Zabaneh, R.; Cangiano, J.; Reed, J.; Fischer, D.; Garrett, L.; Ling, B.; Chasan-Taber, S.; Dillon, M.; Blair, A.; et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 2007, 72, 1130–1137. [Google Scholar] [CrossRef]
- Tonelli, M.; Wiebe, N.; Culleton, B.; Lee, H.; Klarenbach, S.; Shrive, F.; Manns, B.; Network, F.T.A.K.D. Systematic review of the clinical efficacy and safety of sevelamer in dialysis patients. Nephrol. Dial. Transplant. 2007, 22, 2856–2866. [Google Scholar] [CrossRef]
- Block, G.A.; Wheeler, D.C.; Persky, M.S.; Kestenbaum, B.; Ketteler, M.; Spiegel, D.M.; Allison, M.A.; Asplin, J.; Smits, G.; Hoofnagle, A.N.; et al. Effects of Phosphate Binders in Moderate CKD. J. Am. Soc. Nephrol. 2012, 23, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.M.; Martin, B.R.; Wastney, M.E.; McCabe, G.P.; Moe, S.M.; Weaver, C.M.; Peacock, M. Oral calcium carbonate affects calcium but not phosphorus balance in stage 3–4 chronic kidney disease. Kidney Int. 2013, 83, 959–966. [Google Scholar] [CrossRef]
- Spiegel, D.M.; Brady, K. Calcium balance in normal individuals and in patients with chronic kidney disease on low- and high-calcium diets. Kidney Int. 2012, 81, 1116–1122. [Google Scholar] [CrossRef]
- Di Iorio, B.; Bellasi, A.; Russo, D. Mortality in kidney disease patients treated with phosphate binders: a randomized study. Clin J Am Soc Nephrol. 2012, 7, 87–493. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.; Molony, D.; Bell, C.; Cucciniello, E.; Bellizzi, V.; Russo, D.; Bellasi, A. Sevelamer Versus Calcium Carbonate in Incident Hemodialysis Patients: Results of an Open-Label 24-Month Randomized Clinical Trial. Am. J. Kidney Dis. 2013, 62, 771–778. [Google Scholar] [CrossRef]
- Zheng, C.; Liu, J.; Wang, T.; Hu, H.; Chen, Y. A network meta-analysis of therapies for hyperphosphatemia in CKD based on randomized trials. Sci. Rep. 2025, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.M.; Plosker, G.L. Sevelamer Carbonate: A Review in Hyperphosphataemia in Adults with Chronic Kidney Disease. Drugs 2014, 74, 771–792. [Google Scholar] [CrossRef]
- Raggi, P.; Vukicevic, S.; Moysés, R.M.; Wesseling, K.; Spiegel, D.M. Ten-Year Experience with Sevelamer and Calcium Salts as Phosphate Binders. Clin. J. Am. Soc. Nephrol. 2010, 5, S31–S40. [Google Scholar] [CrossRef]
- Vervloet, M.G.; et al. The role of phosphate in kidney disease. Nat. Rev. Nephrol. 2016, 13, 27–38. [Google Scholar] [CrossRef]
- Lenglet, A.; Fabresse, N.; Taupin, M.; Gomila, C.; Liabeuf, S.; Kamel, S.; Alvarez, J.C.; Drueke, T.B.; Massy, Z.A. Does the Administration of Sevelamer or Nicotinamide Modify Uremic Toxins or Endotoxemia in Chronic Hemodialysis Patients? Drugs 2019, 79, 855–862. [Google Scholar] [CrossRef]
- Wrong, O.; Harland, C. Sevelamer. Nephrol Dial Transplant 2008, 23, 2101–2102. [Google Scholar] [CrossRef]
- Pai, A.B.; Shepler, B.M. Comparison of Sevelamer Hydrochloride and Sevelamer Carbonate: Risk of Metabolic Acidosis and Clinical Implications. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2009, 29, 554–561. [Google Scholar] [CrossRef]
- Delmez, J.; Block, G.; Robertson, J.; Chasan-Taber, S.; Blair, A.; Dillon, M.; Bleyer, A. A randomized, double-blind, crossover design study of sevelamer hydrochloride and sevelamer carbonate in patients on hemodialysis. Clin. Nephrol. 2007, 68, 386–391. [Google Scholar] [CrossRef]
- Fissell, R.B.; Karaboyas, A.; Bieber, B.A.; Sen, A.; Li, Y.; Lopes, A.A.; Akiba, T.; Bommer, J.; Ethier, J.; Jadoul, M.; et al. Phosphate binder pill burden, patient-reported non-adherence, and mineral bone disorder markers: Findings from the DOPPS. Hemodial. Int. 2015, 20, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, D.R.; Scott, L.J.; Cvetković, R.S.; Plosker, G.L. Sevelamer Hydrochloride: A review of its use for hyperphosphataemia in patients with end-stage renal disease on haemodialysis. Drugs 2008, 68, 85–104. [Google Scholar] [CrossRef] [PubMed]
- Pai, A.B.; Shepler, B.M. Comparison of Sevelamer Hydrochloride and Sevelamer Carbonate: Risk of Metabolic Acidosis and Clinical Implications. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2009, 29, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Swanson, B.J.; Limketkai, B.N.; Liu, T.-C.; Montgomery, E.; Nazari, K.; Park, J.Y.; Santangelo, W.C.; Torbenson, M.S.; Voltaggio, L.; Yearsley, M.M.; et al. Sevelamer Crystals in the Gastrointestinal Tract (GIT). Am. J. Surg. Pathol. 2013, 37, 1686–1693. [Google Scholar] [CrossRef]
- Elkalashy, A.; Rainwater, R.R.; Ali, U.; Elbahnasawy, E.; Singh, M.; Karakala, N. Gastrointestinal mucosal cell injury caused by sevelamer crystals- Case series and literature review. J. Ren. Nutr. 2025. [Google Scholar] [CrossRef]
- Braunlin, W.; Zhorov, E.; Guo, A.; et al. Bile acid binding to sevelamer HCl. Kidney Int 2002, 62, 611–619. [Google Scholar] [CrossRef]
- Burke, S.K.; Dillon, M.A.; Hemken, D.E.; et al. Meta-analysis of the effect of sevelamer on phosphorus, calcium, PTH, and serum lipids in dialysis patients. Adv Ren Replace Ther 2003, 10, 133–145. [Google Scholar] [CrossRef]
- Susantitaphong, P.; Jaber, B.L. Potential Interaction Between Sevelamer and Fat-Soluble Vitamins: A Hypothesis. Am. J. Kidney Dis. 2012, 59, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Vemuri, N.; Michelis, M.F.; Matalon, A. Conversion to lanthanum carbonate monotherapy effectively controls serum phosphorus with a reduced tablet burden: a multicenter open-label study. BMC Nephrol. 2011, 12, 49–49. [Google Scholar] [CrossRef]
- Dellanna, F.; Reichel, H.; Seibt, F. Efficacy and safety of lanthanum carbonate in German patients on dialysis. Clin. Nephrol. 2012, 78, 382–390. [Google Scholar] [CrossRef]
- Wilson, R.J.; Keith, M.S.; Preston, P.; Copley, J.B. The Real-World Dose-Relativity of Sevelamer Hydrochloride and Lanthanum Carbonate Monotherapy in Patients with End-Stage Renal Disease. Adv. Ther. 2013, 30, 1100–1110. [Google Scholar] [CrossRef]
- Takahara, Y.; Matsuda, Y.; Takahashi, S.; Shigematsu, T. ; on behalf of the Lanthanum Carbonate Study Group Efficacy and safety of lanthanum carbonate in pre-dialysis CKD patients with hyperphosphatemia: a randomized trial. Clin. Nephrol. 2014, 82, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Toida, T.; Fukudome, K.; Fujimoto, S.; Yamada, K.; Sato, Y.; Chiyotanda, S.; Kitamura, K. Effect of lanthanum carbonate vs. calcium carbonate on serum calcium in hemodialysis patients: a crossover study. Clin. Nephrol. 2012, 78, 216–223. [Google Scholar] [CrossRef]
- Zhang, C.; Wen, J.; Li, Z.; Fan, J. Efficacy and safety of lanthanum carbonate on chronic kidney disease–mineral and bone disorder in dialysis patients: a systematic review. BMC Nephrol. 2013, 14, 226. [Google Scholar] [CrossRef]
- Lacour, B.; Lucas, A.; Auchère, D.; Ruellan, N.; De Serre Patey, N.M.; Drüeke, T.B. Chronic renal failure is associated with increased tissue deposition of lanthanum after 28-day oral administration. Kidney Int. 2005, 67, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, A.J.; E Barnett, M.; Krause, R.; A Siami, G. ; Lanthanum Carbonate Study Group Lanthanum carbonate treatment, for up to 6 years, is not associated with adverse effects on the liver in patients with chronic kidney disease Stage 5 receiving hemodialysis. Clin Nephrol. 2009, 71, 286–295. [Google Scholar]
- Spasovski, G.B.; Sikole, A.; Gelev, S.; Masin-Spasovska, J.; Freemont, T.; Webster, I.; Gill, M.; Jones, C.; De Broe, M.E.; D'Haese, P.C. Evolution of bone and plasma concentration of lanthanum in dialysis patients before, during 1 year of treatment with lanthanum carbonate and after 2 years of follow-up. Nephrol. Dial. Transplant. 2006, 21, 2217–2224. [Google Scholar] [CrossRef]
- Hutchison, A.J.; Wilson, R.J.; Garafola, S.; Copley, J.B. Lanthanum carbonate: safety data after 10 years. Nephrology 2016, 21, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.B.; Sika, M.; Koury, M.J.; Chuang, P.; Schulman, G.; Smith, M.T.; Whittier, F.C.; Linfert, D.R.; Galphin, C.M.; Athreya, B.P.; et al. Ferric Citrate Controls Phosphorus and Delivers Iron in Patients on Dialysis. J. Am. Soc. Nephrol. 2015, 26, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Umanath, K.; Jalal, D.I.; Greco, B.A.; Umeukeje, E.M.; Reisin, E.; Manley, J.; Zeig, S.; Negoi, D.G.; Hiremath, A.N.; Blumenthal, S.S.; et al. Ferric Citrate Reduces Intravenous Iron and Erythropoiesis-Stimulating Agent Use in ESRD. J. Am. Soc. Nephrol. 2015, 26, 2578–2587. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Hirakata, H.; Akiba, T.; Fukagawa, M.; Nakayama, M.; Sawada, K.; Kumagai, Y.; Block, G.A. Ferric Citrate Hydrate for the Treatment of Hyperphosphatemia in Nondialysis-Dependent CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 543–552. [Google Scholar] [CrossRef]
- Van Buren, P.N.; Lewis, J.B.; Dwyer, J.P.; Greene, T.; Middleton, J.; Sika, M.; Umanath, K.; Abraham, J.D.; Arfeen, S.S.; Bowline, I.G.; et al. The Phosphate Binder Ferric Citrate and Mineral Metabolism and Inflammatory Markers in Maintenance Dialysis Patients: Results From Prespecified Analyses of a Randomized Clinical Trial. Am. J. Kidney Dis. 2015, 66, 479–488. [Google Scholar] [CrossRef]
- Floege, J. Phosphate binders in chronic kidney disease: a systematic review of recent data. J. Nephrol. 2016, 29, 329–340. [Google Scholar] [CrossRef]
- Cozzolino, M.; Funk, F.; Rakov, V.; Phan, O.; Teitelbaum, I. Preclinical Pharmacokinetics, Pharmacodynamics and Safety of Sucroferric Oxyhydroxide. Curr. Drug Metab. 2015, 15, 953–965. [Google Scholar] [CrossRef]
- Floege, J.; Covic, A.C.; Ketteler, M.; Mann, J.F.; Rastogi, A.; Spinowitz, B.; Chong, E.M.; Gaillard, S.; Lisk, L.J.; Sprague, S.M.; et al. Long-term effects of the iron-based phosphate binder, sucroferric oxyhydroxide, in dialysis patients. Nephrol. Dial. Transplant. 2015, 30, 1037–1046. [Google Scholar] [CrossRef]
- Floege, J.; Covic, A.C.; Ketteler, M.; Rastogi, A.; Chong, E.M.; Gaillard, S.; Lisk, L.J.; Sprague, S.M. A phase III study of the efficacy and safety of a novel iron-based phosphate binder in dialysis patients. Kidney Int. 2014, 86, 638–647. [Google Scholar] [CrossRef]
- Ketteler, M.; Sprague, S.M.; Covic, A.C.; Rastogi, A.; Spinowitz, B.; Rakov, V.; Walpen, S.; Floege, J. Effects of sucroferric oxyhydroxide and sevelamer carbonate on chronic kidney disease–mineral bone disorder parameters in dialysis patients. Nephrol. Dial. Transplant. 2018, 34, 1163–1170. [Google Scholar] [CrossRef]
- Covic, A.C.; Floege, J.; Ketteler, M.; Sprague, S.M.; Lisk, L.; Rakov, V.; Rastogi, A. Iron-related parameters in dialysis patients treated with sucroferric oxyhydroxide. Nephrol. Dial. Transplant. 2016, 32, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Lioulios, G.; Stangou, M.; Sarafidis, P.A.; Tsouchnikas, I.; Minasidis, I.; Vainas, A.; Faitatzidou, D.; Sampani, E.; Papagianni, A. Chronic Therapy with Sucroferric Oxyhydroxide Does Not Affect Iron and Anemia Markers in Dialysis Patients. Blood Purif. 2020, 49, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Schiavi, S.C.; Tang, W.; Bracken, C.; O’brien, S.P.; Song, W.; Boulanger, J.; Ryan, S.; Phillips, L.; Liu, S.; Arbeeny, C.; et al. Npt2b Deletion Attenuates Hyperphosphatemia Associated with CKD. J. Am. Soc. Nephrol. 2012, 23, 1691–1700. [Google Scholar] [CrossRef]
- Diaeresis]Ller, D.M.; Mehling, H.; Otto, B.D.; Bergmann-Lips, R.; Luft, F.; Jordan, J.; Kettritz, R. Niacin Lowers Serum Phosphate and Increases HDL Cholesterol in Dialysis Patients. Clin. J. Am. Soc. Nephrol. 2007, 2, 1249–1254. [Google Scholar] [CrossRef]
- Cheng, S.C.; Young, D.O.; Huang, Y.; Delmez, J.A.; Coyne, D.W. A Randomized, Double-Blind, Placebo-Controlled Trial of Niacinamide for Reduction of Phosphorus in Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2008, 3, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Ix, J.H.; Isakova, T.; Larive, B.; Raphael, K.L.; Raj, D.S.; Cheung, A.K.; Sprague, S.M.; Fried, L.F.; Gassman, J.J.; Middleton, J.P.; et al. Effects of Nicotinamide and Lanthanum Carbonate on Serum Phosphate and Fibroblast Growth Factor-23 in CKD: The COMBINE Trial. J. Am. Soc. Nephrol. 2019, 30, 1096–1108. [Google Scholar] [CrossRef]
- Ketteler, M.; Wiecek, A.; Rosenkranz, A.R.; Ose, C.; Rekowski, J.; Lorenz, H.; Hellmann, B.; Karus, M.; Ruhmann, M.; Ammer, R. Modified-release nicotinamide for the treatment of hyperphosphataemia in haemodialysis patients: 52-week efficacy and safety results of the phase 3 randomized controlled NOPHOS trial. Nephrol. Dial. Transplant. 2022, 38, 982–991. [Google Scholar] [CrossRef]
- Gallant, K.M.H.; Sprague, S.M.; Rosenbaum, D.P.; Spiegel, D.M.; Kozuka, K.; Edelstein, S.; Chertow, G.M. Tenapanor: A Phosphate Absorption Inhibitor for the Management of Hyperphosphatemia in Patients With Kidney Failure. J. Ren. Nutr. 2024, 35, 25–34. [Google Scholar] [CrossRef]
- Pergola, P.E.; Rosenbaum, D.P.; Yang, Y.; Chertow, G.M. A Randomized Trial of Tenapanor and Phosphate Binders as a Dual-Mechanism Treatment for Hyperphosphatemia in Patients on Maintenance Dialysis (AMPLIFY). J. Am. Soc. Nephrol. 2021, 32, 1465–1473. [Google Scholar] [CrossRef]
- https://ir.ardelyx.com/news-releases/news-release-details/fda-approves-xphozahr-tenapanor-first-class-phosphate-absorption#:~:text=FDA%20approval%20of%20XPHOZAH%20is,met%20their%20primary%20and%20key. Accessed March 24, 2025.
- Chiu, Y.-W.; Teitelbaum, I.; Misra, M.; de Leon, E.M.; Adzize, T.; Mehrotra, R. Pill Burden, Adherence, Hyperphosphatemia, and Quality of Life in Maintenance Dialysis Patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1089–1096. [Google Scholar] [CrossRef]
- Nagano, N.; Ito, K.; Ono, T.; Ariyoshi, Y.; Masima, S.; Kobayashi, H.; Ando, T.; Tsutsui, T.; Ogawa, T. Prescription characteristics of phosphate binders in a high pill burden for hemodialysis patients. Ren. Replace. Ther. 2021, 7, 1–11. [Google Scholar] [CrossRef]
- Forfang, D.; Edwards, D.P.; Kalantar-Zadeh, K. The Impact of Phosphorus Management Today on Quality of Life: Patient Perspectives. Kidney Med. 2022, 4, 100437. [Google Scholar] [CrossRef] [PubMed]
- Edmonston, D.L.; Isakova, T.; Dember, L.M.; Brunelli, S.; Young, A.; Brosch, R.; Beddhu, S.; Chakraborty, H.; Wolf, M. Design and Rationale of HiLo: A Pragmatic, Randomized Trial of Phosphate Management for Patients Receiving Maintenance Hemodialysis. Am. J. Kidney Dis. 2021, 77, 920–930.e1. [Google Scholar] [CrossRef] [PubMed]
- Wald, R.; Walsh, M.W. In Search of the Optimal Target for Phosphate Control: Episode 1. J. Am. Soc. Nephrol. 2021, 32, 526–528. [Google Scholar] [CrossRef]
- Gregg, L.P.; Hedayati, S.S. Management of Traditional Cardiovascular Risk Factors in CKD: What Are the Data? Am. J. Kidney Dis. 2018, 72, 728–744. [Google Scholar] [CrossRef]
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




