Submitted:
26 March 2025
Posted:
27 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
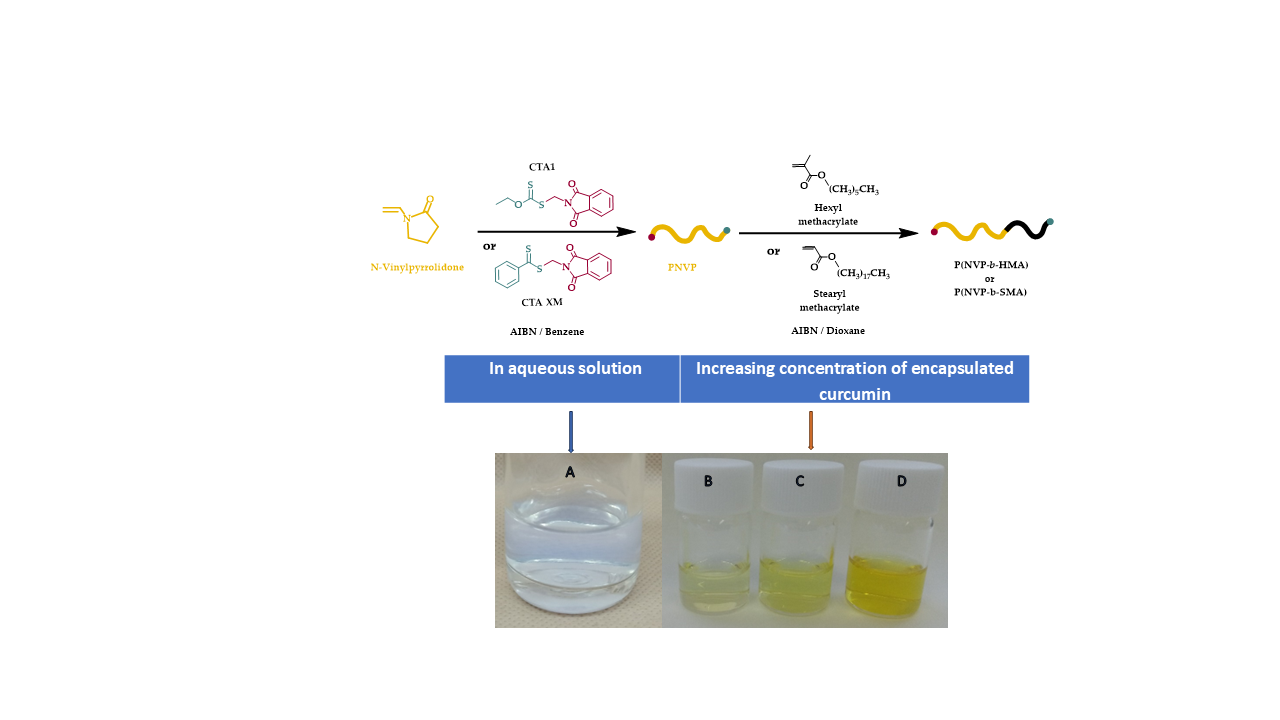
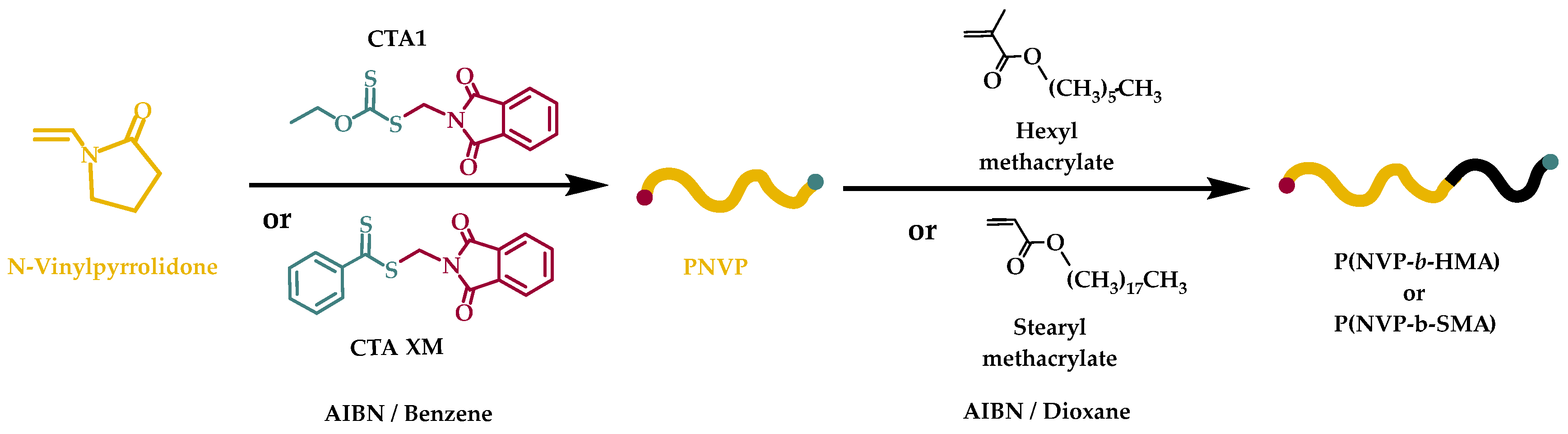
2.2. Synthesis of PNVP-b-PHMA and PNVP-b-PSMA Block Copolymers via RAFT Polymerization
2.3. Encapsulation Process
2.4. Characterization Techniques
3. Results and Discussion
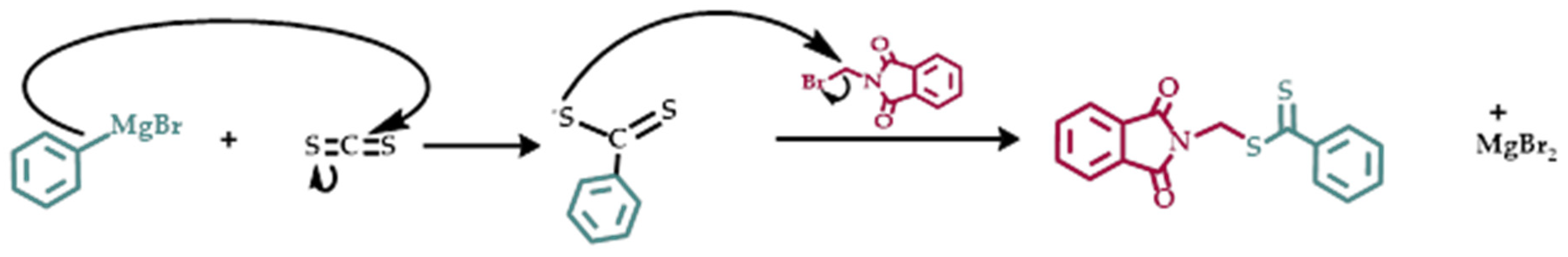
3.1. Synthesis of Phthalimidylmethyl Dithiobenzoate, CTA XM
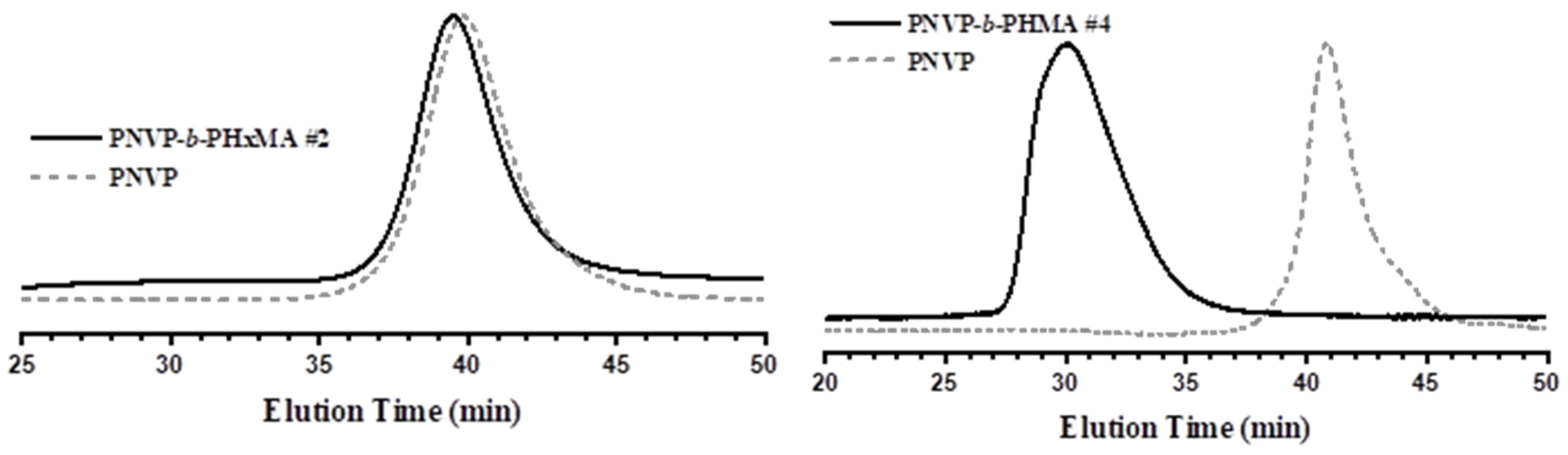
3.2. Synthesis of the Block Copolymers PNVP-b-PHMA and PNVP-b-PSMA
| macro CTA (PNVP) * |
block copolymers * |
NVP |
HMA |
|||
|---|---|---|---|---|---|---|
| Sample | Mn 103 (Daltons) |
Ð | Mn 103 (Daltons) | Ð | % mol** | % mol** |
| PNVP-b-PHMA #1 | 29.8 | 1.29 | 36 | 1.26 | 87 | 13 |
| PNVP-b-PHMA #2 | 10.6 | 1.26 | 13 | 1.27 | 84 | 16 |
| PNVP-b-PHMA #3 | 27.0 | 1.60 | 32 | 1.68 | 74 | 26 |
| PNVP-b-PHMA #4 | 6.3 | 1.34 | 130 | 1.53 | 5 | 95 |
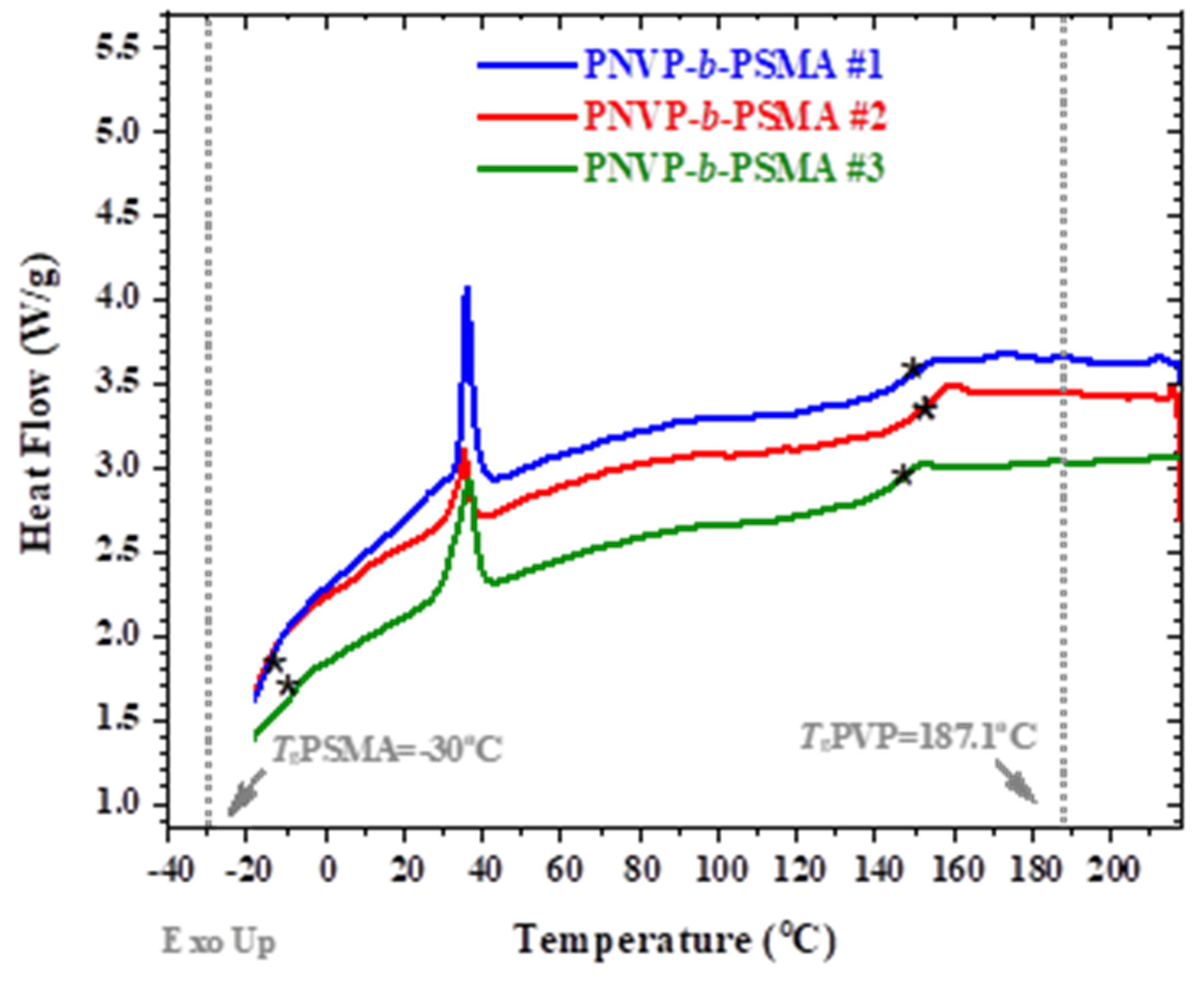
3.2. Thermal Properties
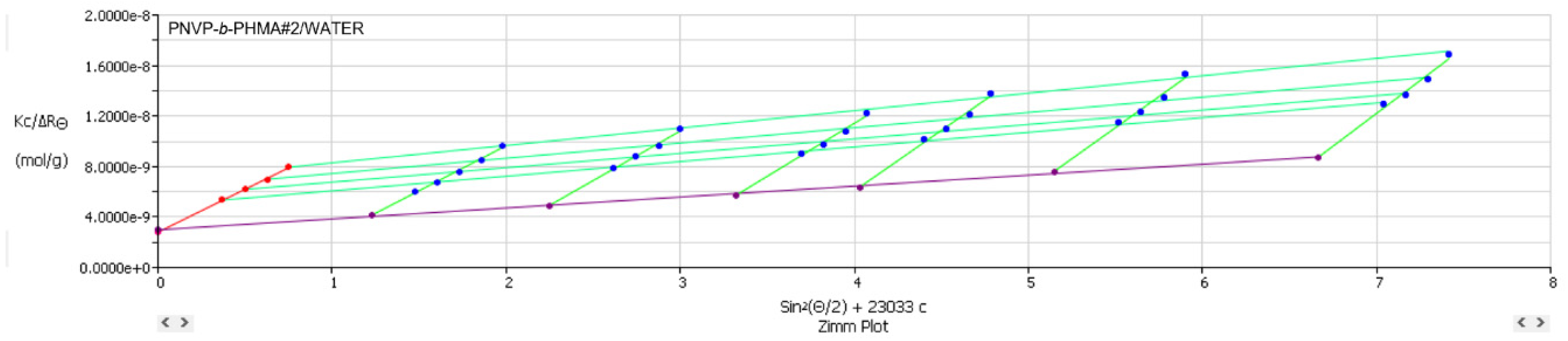
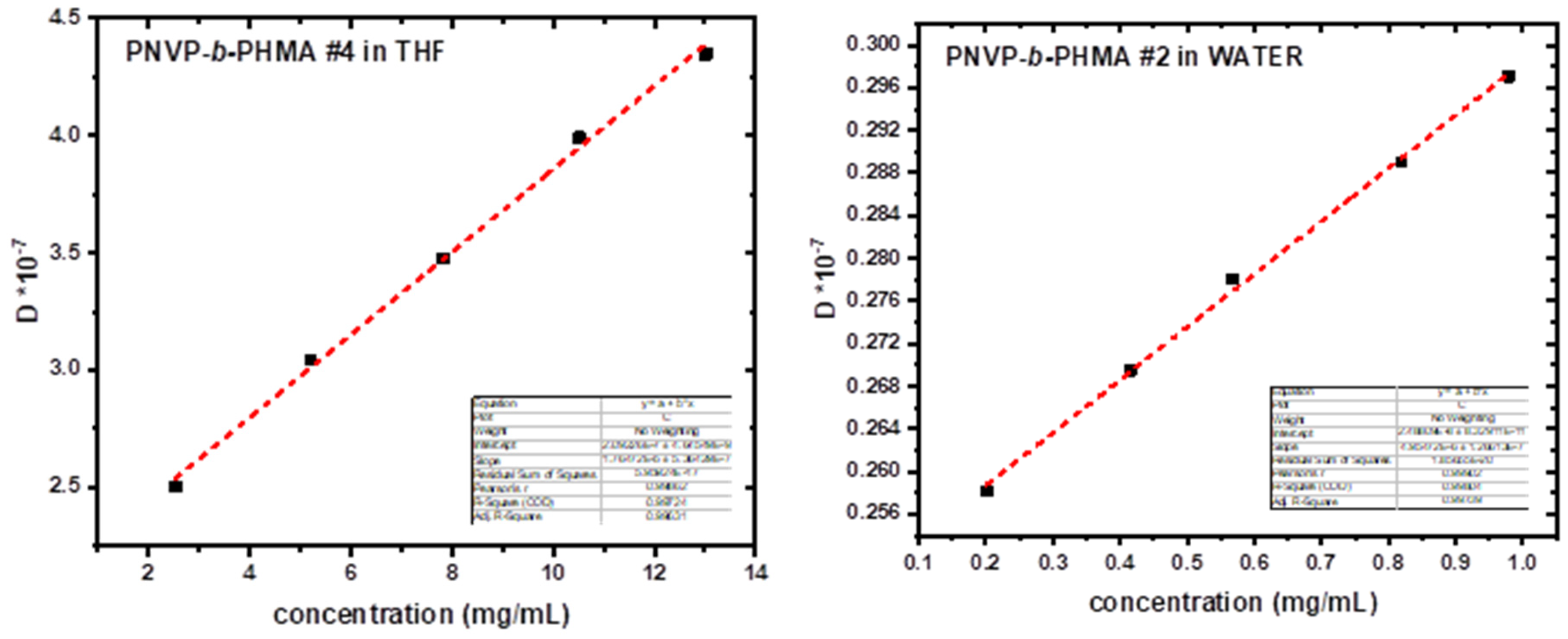
3.3. Self-Assembly Behavior of the PNVP-b-PHMA Block Copolymers in THF and Aqueous Solutions
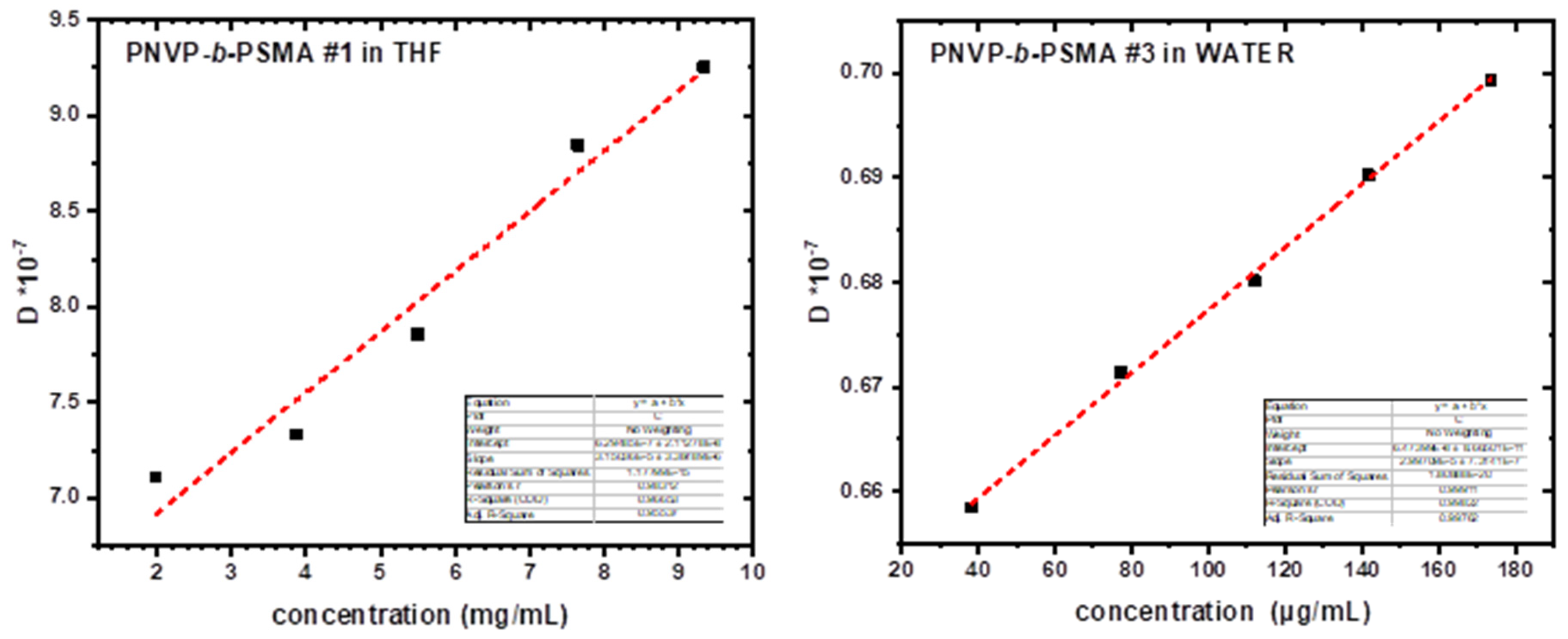
3.4. Self-Assembly Behavior of the PNVP-b-PSMA Block Copolymers in THF and Aqueous Solutions
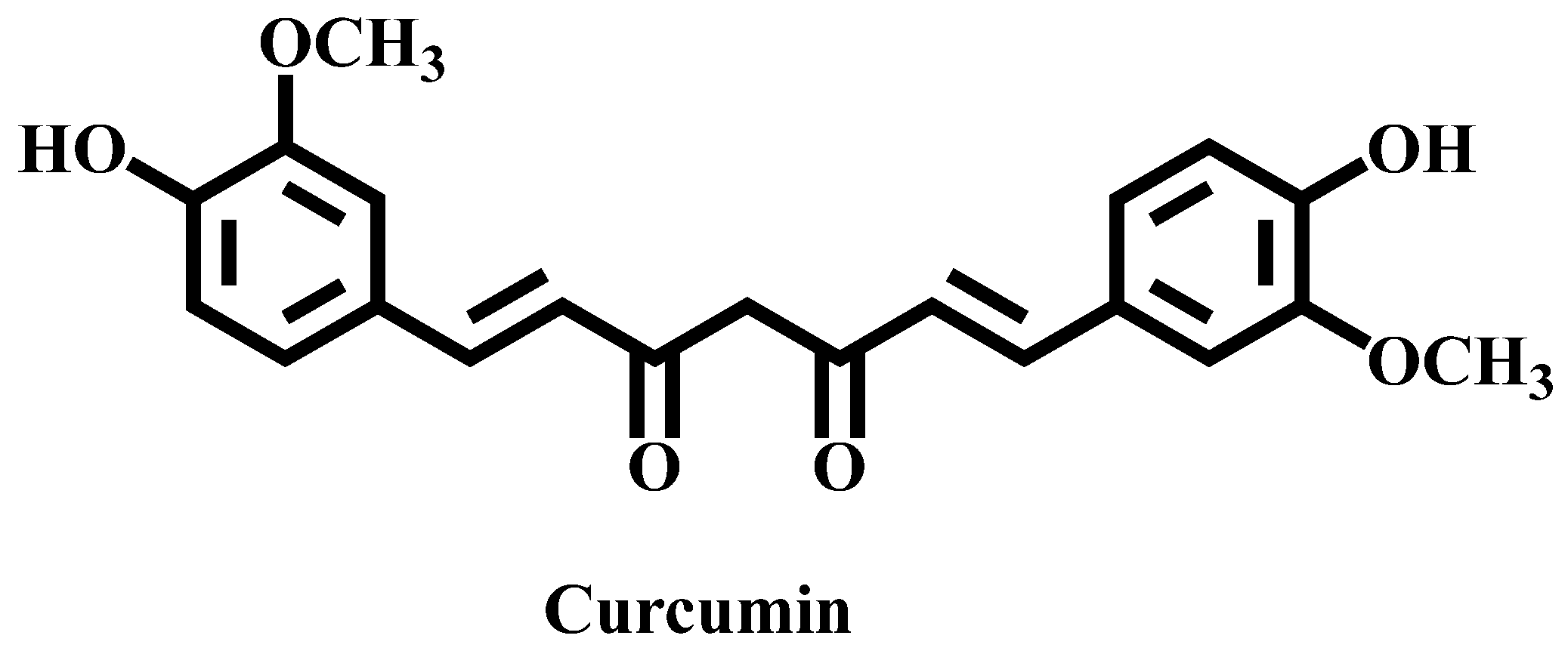
3.5. Encapsulation of Curcumin into the Micellar Solutions
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Teodorescu, M.; Bercea, M. Poly(vinylpyrrolidone)—A Versatile Polymer for Biomedical and Beyond Medical Applications. Polymer-Plastics Technology and Engineering 2015, 54:9, 923-943. [CrossRef]
- Franco, F.; De Marco, I. The Use of Poly(N-vinyl pyrrolidone) in the Delivery of Drugs: A Review. Polymers 2020, 12, 1114.
- Moulay, S. Molecular iodine/polymer complexes. J. Polym. Eng. 2013, 33(5), 389–443.
- Husain, M.S.B.; Gupta, A.; Alashwal, B.Y.; Sharma S. Synthesis of PVA/PVP based hydrogel for biomedical applications: a review. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects 2018, 40:20, 2388-2393. [CrossRef]
- Reis, C.P.; Silva, C.; Martinho, N.; Rosado, C. Drug carriers for oral delivery of peptides and proteins: accomplishments and future Perspectives. Therap. Deliv. 2013, 4(2), 251–265. [CrossRef]
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1638–1649.
- Dolman, M.E.M.; Harmsen, S.; Storm, G.; Hennink, W.E.; Kok, R.J. Drug targeting to the kidney: Advances in the active targeting of therapeutics to proximal tubular cells. Adv. Drug Deliv. Rev. 2010, 62, 1344-1357. [CrossRef]
- Kurakula, M.; Koteswara Rao, G.S.N. Pharmaceutical assessment of polyvinylpyrrolidone (PVP): As excipient from conventional to controlled delivery systems with a spotlight on COVID-19 inhibition. J. Drug Deliv. Sci. Technol. 2020, 60, 102046. [CrossRef]
- Liang, D.; Hsiao, B.S.; Chu, B. Functional electrospun nanofibrous scaffolds for biomedical applications. Adv. Drug Deliv. Rev. 2007, 59, 1392-1412. [CrossRef]
- Culbertson, B.M. Glass-ionomer dental restoratives. Prog. Polym. Sci. 2001, 26, 577-604. [CrossRef]
- Godlewska, K.; Stepnowski, P.; Paszkiewicz, M. Pollutant analysis using passive samplers: principles, sorbents, calibration and applications: A review. Environm. Chem. Lett. 2020, 19, 465–520. [CrossRef]
- Lowe, S.; O’Brien-Simpson, N.M.; Connal, L.A. Antibiofouling polymer interfaces: poly(ethylene glycol) and other promising candidates. Polym. Chem. 2015, 6, 198. [CrossRef]
- Halake, K.; Birajdar, M.; Soo Kim, B.; Bae, H.; Lee, C-C.; Jin Kim, Y.; Kim, S.; Jin Kim, H.; Ahn, S.; Yeoung An, S.; Lee, J. Recent application developments of water-soluble synthetic polymers. J. Industr. Engin. Chem. 2014, 20, 3913–3918. [CrossRef]
- Liu, X.; Xu, Y.; Wu, Z.; Chen, H.; Poly(N-vinylpyrrolidone)-Modified Surfaces for Biomedical Applications. Macromol. Biosci. 2013, 13, 147–154.
- Haaf, F.; Sanner, A.; Straub, F. Polymers of N-Vinylpyrrolidone: Synthesis, Characterization and Uses. Polymer J. 1985, 17, 143-152. [CrossRef]
- Afzal, A.; Kausar, A.; Siddiq, M. A Review on Polymer/Cement Composite with Carbon Nanofiller and Inorganic Filler. Polymer-Plastics Technol. Engin. 2016, 55:12, 1299-1323. [CrossRef]
- Ye, Y-S.; Rick, J. Hwang, B-J. Water Soluble Polymers as Proton Exchange Membranes for Fuel Cells. Polymers 2012, 4, 913-963. [CrossRef]
- Yang, J.; Zhang, H.; Zhou, Q.; Qu, H.; Dong, T.; Zhang, M.; Tang, B.; Zhang, J.; Cui, G. Safety-Enhanced Polymer Electrolytes for Sodium Batteries: Recent Progress and Perspectives. ACS Appl. Mater. Interfaces 2019, 11, 17109−17127. [CrossRef]
- Koczkur, K.M.; Mourdikoudis, S.; Polavarapu, L.; Skrabalak, S.E. Polyvinylpyrrolidone (PVP) in nanoparticle synthesis. Dalton Trans. 2015, 44, 17883. [CrossRef]
- Wiley, B.; Sun, Y.; Mayers, B.; Xia, Y. Shape-Controlled Synthesis of Metal Nanostructures: The Case of Silver. Chem. Eur. J. 2005, 11, 454–463. [CrossRef]
- Qiu, L.Y.; Bae, Y.H. Polymer Architecture and Drug Delivery. Pharm. Res. 2006, 23, 1-30.
- Karanikolopoulos, N.; Pitsikalis, M.; Hadjichristidis, N.; Georgikopoulou, K.; Calogeropoulou, T.; Dunlap, J. R. “pH-Responsive aggregates from double hydrophilic block copolymers carrying zwitterionic groups. Encapsulation of antiparasitic compounds for the treatment of leishmaniasis” Langmuir. 2007, 23, 4214-4224.
- Kwon, G.S.; Okano, T. Polymeric micelles as new drug carriers. Adv. Drug Deliv. Rev. 1996, 21, 107–116. [CrossRef]
- Karanikolopoulos, N.; Zamurovic, M.; Pitsikalis, M.; Hadjichristidis, N. Poly(DL-lactide)-b-Poly(N,N-dimethylamino-2-ethyl methacrylate): Synthesis, characterization, micellization behaviour in aqueous solutions and encapsulation of the hydrophobic drug dipyridamole. Biomacromolecules. 2010, 11, 430-438.
- Trubetskoy, V.S. Polymeric micelles as carriers of diagnostic agents. Adv. Drug Deliv. Rev. 1999, 37, 81–88. [CrossRef]
- Gerst, M.; Schuch, H.; Urban, D. Amphiphilic Block Copolymers as Surfactants in Emulsion Polymerization. ACS Symposium Series; Glass, J.E., Ed.; ACS Publications: Washington, DC, USA, 2000, 765, 37–51.
- Riess, G.; Hurtrez, G.; Bahadur, P. Block Copolymers, Encyclopedia Polymer Science and Engineering; Wiley: New York, NY, USA, 1985; Volume 2, pp. 324–434.
- Hong, S.C.; Rief, U.; Kristen, M.O. Ethylene (co) Polymerization with metallocene catalysts encapsulated in gel-type poly(styrene-co-divinylbenzene) beads. Macromol. Rapid Commun. 2001, 22, 1447–1454. [CrossRef]
- Kataoka, K.; Harada, A.; Nagasaki, Y. Block copolymer micelles for drug delivery: Design, characterization and biological significance. Adv. Drug Deliv. Rev. 2001, 47, 113–131. [CrossRef]
- Kakizawa, Y.; Kataoka, K. Block copolymer micelles for delivery of gene and related compounds. Adv. Drug Deliv. Rev. 2002, 54, 203–222. [CrossRef]
- Sidorov, S.N.; Bronstein, L.M.; Valetsky, P.M.; Hartmann, J.; Coelfen, H.; Schnablegger, H.; Antonietti, M. Stabilization of metal nanoparticles in aqueous medium by poly(ethylene oxide)-poly(ethylene imine) block copolymers. J. Colloid Interface Sci. 1999, 212, 197–211.
- Cao, T.; Yin, W.; Armstrong, J.L.; Webber, S.E. Adsorption of photoactive amphiphilic polymers onto hydrophobic polymer films polystyrene-b-poly(2-vinylnaphthalene)-b-poly(methacrylic acid). Langmuir 1994, 10, 1841–1847. [CrossRef]
- Spatz, J.P.; Sheiko, S.; Möller, M. Ion stabilized block copolymer micelles film formation and inter micellar interaction. Macromolecules 1996, 29, 3220–3226.
- Spatz, J.P.; Mössmer, S.; Hartmann, C.; Möller, M. Ordered deposition of inorganic clusters from micellar block copolymer films. Langmuir 2000, 16, 407–415. [CrossRef]
- Lazzari, M.; Scalarone, D.; Hoppe, C.E.; Vazquez-Vazquez, C.; Lòpez-Quintela, M.A. Tunable polyacrylonitrile based micellar aggregates as a potential tool for the fabrication of carbon nanofibers. Chem. Mater. 2007, 19, 5818–5820. [CrossRef]
- Dau, H.; Jones, G.R.; Tsogtgerel, E.; Nguyen, D.; Keyes, A.; Liu, Y.-S.; Rauf, H.; Ordonez, E.; Puchelle, V.; Alhan, H.B.; Zhao, C.; Harth, E. Linear block copolymer synthesis Chem. Rev. 2022, 122, 14471-14553. [CrossRef]
- Hadjichristidis, N.; Iatrou, H.; Pitsikalis, M.; Mays, J.W. Macromolecular architectures by living and controlled/living polymerizations. Progr. Polym. Sci. 2006, 31, 1068–1132. [CrossRef]
- Hadjichristidis, N.; Pitsikalis, M.; Iatrou, H.; Driva, P.; Sakellariou, G.; Chatzichristidi, M. Polymers with star-related structures: Synthesis, properties and applications. Polym. Sci. Compr. Ref. 2012, 6, 29–111.
- Hadjichristidis, N.; Pispas, S.; Iatrou, H.; Pitsikalis, M. Linking chemistry and anionic polymerization. Curr. Org. Chem. 2002, 6, 155–176. [CrossRef]
- Dey, A.; Hldar, U.; De, P. Block copolymer synthesis by the combination of living cationic polymerization and other polymerization methods Frontiers in Chemistry 2021, 9, 644547.
- Diaz, C.; Mehrkhodavandi, P. Strategies for the synthesis of block copolymers with biodegradable polyester segments Polym. Chem. 2021, 12, 783-806. [CrossRef]
- Theodosopoulos, G.; Pitsikalis, M. “Block Copolymers: Recent Synthetic Routes and Developments” Chapter in “Anionic Polymerization: Principles, Practice, Strength, Consequences and Applications” Springer, 2014.
- .
- Kennedy J.P.; Iván B. in Designed polymers by carbocationic macromolecular engineering. Theory and practice Hanser Publishers 1992.
- Macromolecular design of polymeric materials Hatada K.; Kitayama; T.; Vogl, O. Eds Marcel Dekker Inc Chapters 3,4, 1997.
- Matyjaszewski, K.; Xia, Atom transfer radical polymerization. J Chem Rev. 2001, 101, 2921-2990.
- Coessens, V.; Pintauer, T.; Matyjaszewski, K. Functional polymers by atom transfer radical polymerization. Progr Polym Sci. 2001, 26, 337-377. [CrossRef]
- Goto, A.; Fukuda, T. Kinetics of living radical polymerization. Progr Polym Sci. 2004, 29, 329-385. [CrossRef]
- Hawker, C.J.; Bosman, A,W.; Harth, E. New polymer synthesis by nitroxide mediated living radical polymerizations. Chem Rev 2001, 101, 3661-3688. [CrossRef]
- McCormick, C,J.; Lowe, A.B. Aqueous RAFT polymerization: recent developments in synthesis of functional water-soluble (co) polymers with controlled structures. Acc Chem Res 2004, 37, 312-325.
- Webster, O.W.; Anderson, B.C. in New methods for polymer synthesis, Mijs W.J. Ed. 1992, Plenum Press.
- Buchmeiser, M.R. Homogeneous metathesis polymerization by well-defined group VI and group VIII transition-metal alkylidenes: Fundamentals and applications in the preparation of advanced materials. Chem Rev 2000, 100, 1565-1604. [CrossRef]
- Ofstead, E.A.; Wagener, K.B. in New methods for polymer synthesis, Mijs W.J. Ed. 1992 Plenum Press.
- Trnka, T.M. Grubbs, R.H. The development of L2X2Ru CHR olefin metathesis catalysts: an organometallic success story. Acc Chem Res 2001, 34, 18-29.
- Kabanov, A.V.; Batrakova, E.V.; Alakhov, V.Y. Pluronic (R) block copolymers as novel polymer therapeutics for drug and gene delivery. J. Control. Release 2002, 82, 189–212. [CrossRef]
- Kwon, G.S.; Okano, T. Polymeric micelles as new drug carriers.Adv. Drug Deliv. Rev. 1996, 21, 107-116. [CrossRef]
- Munch, M.R.; Gast, A.P. Kinetics of block copolymer adsorption on dielectric surfaces from a selective solvent. Macromolecules 1990, 23, 2313–2320. [CrossRef]
- Breulmann, M.; Förster, S.; Antonietti, M. Mesoscopic surface patterns formed by block copolymer micelles. Macromol. Chem. Phys. 2000, 201, 204–211. [CrossRef]
- Yu-Su, S.Y.; Thomas, D.R.; Alford, J.E.; LaRue, I.; Pitsikalis, M.; Hadjichristidis, N.; DeSimone, J.M.; Dobrynin, A.V.; Sheiko, S.S. Molding copolymer micelles: A framework for molding of discrete objects on surfaces. Langmuir 2008, 24, 12671–12679. [CrossRef]
- Torchilin, V.P. Structure and design of polymeric surfactant-based drug delivery systems. J. Control. Release 2001, 73, 137–172. [CrossRef]
- Bronstein, L.M.; Sidorov, S.N.; Valetsky, P.M.; Hartmann, J.; Coelfen, H.; Antonietti, M. Induced micellization by interaction of poly(2-vinylpyridine)-block-poly(ethylene oxide) with metal compounds. Micelle characteristics and metal nanoparticle formation. Langmuir 1999, 15, 6256–6262.
- Mortensen, K. PEO-related block copolymer surfactants. Colloids Surf. A 2001, 183, 277-292. [CrossRef]
- Adams, M.L.; Lavasanifar, A.; Kwon, G.S. Amphiphilic block copolymers for drug delivery. J. Pharm. Sci. 2003, 92, 1343-1355. [CrossRef]
- Ulrich, K.E.; Cannizzaro, S.M.; Langer, R.S.; Shakesheff, K.M. Polymeric systems for controlled drug release.Chem. Rev. 1999, 99, 3181-3198. [CrossRef]
- Xiong, X.Y.; Tam, K.C.; Gan, L.H. Release kinetics of hydrophobic and hydrophilic model drugs from pluronic F127/poly (lactic acid) nanoparticles. J. Contr. Release 2005, 103, 73-82.
- Kim, S.Y.; Ha, J.C.; Lee, Y.M. Poly (ethylene oxide)-poly (propylene oxide)-poly (ethylene oxide)/poly(ϵ-caprolactone)(PCL) amphiphilic block copolymeric nanospheres: II. Thermo-responsive drug release behaviors. J. Contr. Release 2000, 65, 345-358. [CrossRef]
- Son, K.; Ueda, M.; Taguchi, K.; Maruyama, T.; Takeoka, S.;Ito, Y. Evasion of the accelerated blood clearance phenomenon by polysarcosine coating of liposomes. J. Control. Release 2020, 322, 209-216. [CrossRef]
- Hatori, Y.; Tamaki, K.; Sakasai, S.; Ozaki, K.J.; Onishi, H. Effects of PEG anchors in PEGylated siRNA lipoplexes on in vitro gene-silencing effects and siRNA biodistribution in mice. Mol. Med. Rep. 2020, 22, 4183-4196. [CrossRef]
- Kozma, G.T.; Shimizu, T.; Ishida, T.; Szebeni, J. Anti-PEG antibodies: properties, formation, testing and role in adverse immune reactions to PEGylated nano-biopharmaceuticals. Adv. Drug Deliv. Rev. 2020, 154-155, 163-175. [CrossRef]
- De Vrieze, J. Suspicions grow that nanoparticles in Pfizer’s COVID-19 vaccine trigger rare allergic reactions. Science 2020, 80.
- Berger, M.; Toussaint, F.; Ben Djemaa, S.; Laloy, J.; Pendeville, H.; Evrard, B.; Jerôme, C.; Lechanteur, A.; Mottet, D.; Debuigne, A. Piel, G. Poly(vinyl pyrrolidone) derivatives as PEG alternatives for stealth, non-toxic and less immunogenic siRNA-containing lipoplex delivery. J. Control. Release 2023, 361, 87-101. [CrossRef]
- Torchilin, V.; Levchenko, T.; Whiteman, K.; Yaroslav, A.; Tsatsakis, A.; Rizos, A.; Michailova, E.; Shtilman, M. Amphiphilic poly-N-vinylpyrrolidone synthesis, properties and liposome surface modification. Biomaterials 2001, 22, 3035-3044.
- Yamskov, L.A.; Kuskov, A.N.; Babievsky, K.K.; Berezin, B.B.; Krayukhina, N.; Samoylova, A.; Tikhonov, V.E.; Shtilman Novel liposomal forms of antifungal antibiotics modified by amphiphilic polymers. Appl. Biochem. Microbiol. 2008, 44, 624-628.
- Liu, Y.; Luo, X.; Xu, X.; Gao, N.; Liu, X. Preparation, characterization and in vivo pharmacokinetic study of PVP-modified oleanolic acid liposomes. Int. J. Pharm. 2017, 517, 1-7. [CrossRef]
- Roka, N.; Kokkorogianni, O.; Kontoes-Georgoudakis, P.; Choinopoulos, I.; Pitsikalis, M. Recent Advances in the Synthesis of Complex Macromolecular Architectures Based on Poly(N-vinyl pyrrolidone) and the RAFT Polymerization Technique Polymers 2022, 14, 701. [CrossRef]
- Perrier, S. 50th Anniversary Perspective: RAFT Polymerization—A User Guide. Macromolecules 2017, 50, 7433-7447.
- Pagidi, S.; Park, H.S., DaYeon Lee, D.Y.; Kim, M.S.; Hee Lee S.H. Nanosize-confined nematic liquid crystals at slippery interfaces of polymer composites consisting of poly (hexyl methacrylate). J. Molec. Liquids 2022, 350, 118540. [CrossRef]
- Nielsen, B.V.; Nevell, T.G.; Barbu, E.; Smith, J.R.; Rees, G.D.; Tsibouklis, J. Multifunctional poly(alkyl methacrylate) films for dental care. Biomed. Mater. 2011, 6, 015003. [CrossRef]
- Li, J.; Barrow, D.; Howell, H.; Kalachandra, S. In vitro drug release study of methacrylate polymer blend system: effect of polymer blend composition, drug loading and solubilizing surfactants on drug release. J. Mater. Sci.: Materials in Medicine 2010, 21, 583–588. [CrossRef]
- Heinzmann, C.; Salz, U.; Moszner, N.; Fiore, G.L.; Weder, C. Supramolecular Cross-Links in Poly(alkyl methacrylate) Copolymers and Their Impact on the Mechanical and Reversible Adhesive Properties. ACS Appl. Mater. & Interf. 2015, 7, 13395−13404. [CrossRef]
- Khai, N.; Nguyen, H.; Dang, H.H.; Nguyen, L.T.; Nguyen, L.M.T.; Truong, T.T.; Nguyen, H.T.; Nguyen, T.Q.; Tran, C.D.; Nguyen, L.-T.T. Self-healing elastomers from supramolecular random copolymers of 4-vinyl pyridine. Europ. Polym. J. 2023, 199, 112474. [CrossRef]
- Oktay, B.; Baştürk, E.; Kahraman, M.V.; Apohan, N.K. Designing Coconut Oil Encapsulated Poly(stearyl methacrylate-co-hydroxylethyl metacrylate) Based Microcapsule for Phase Change Materials. Chem. Select 2019, 4, 5110 –51. [CrossRef]
- Zhang, X.; Zhang, X.; Yang, B.; Wang, S.; Liu, M.; Zhang, Y.; Tao, L.; Wei, Y. Aggregation-induced emission material based fluorescent organic nanoparticles: facile PEGylation and cell imaging applications RSC Adv., 2013, 3, 9633-9636. [CrossRef]
- Jennings, J.; Butler, M.F.; McLeod, M.; Csányi, E.; Ryan, A.J.; Mykhaylyk, O.O. Stearyl Methacrylate-Based Polymers as Crystal Habit Modifiers for Triacylglycerols Cryst. Growth Des. 2018, 18, 7094–7105. [CrossRef]
- Pingpin, Z.;Yuanli L.; Haiyang Y.; Xiaoming C. Effect of non-ideal mixed solvents on dimensions of poly(N-vinylpyrrolidone) and poly(methyl methacrylate) coils J. Macromol. Sci. Part B: Polym. Phys. 2006, 45, 1125-1134. [CrossRef]
- Roka, N.; Pitsikalis, M. Synthesis and micellization behavior of amphiphilic block copolymers of poly(N-vinyl pyrrolidone) and poly(benzyl methacrylate): Block versus statistical copolymers. Polymers 2023, 15, 2215. [CrossRef]
- Kontoes-Georgoudakis, P.; Plachouras, N.V.; Kokkorogianni, O.; Pitsikalis, M. Amphiphilic block copolymers of poly(N-vinyl pyrrolidone) and poly (isobornyl methacrylate). Synthesis, characterization and micellization behaviour in selective solvents. Europ. Polym. J. 2024, 208, 112873.
- Wan, D., Satoh, K., Kamigaito, M., Okamoto, Y. Xanthate-mediated radical polymerization of N-vinylpyrrolidone in fluoroalcohols for simultaneous control of molecular weight and tacticity. Macromolecules 2005, 38, 10397-10405. [CrossRef]
- Postma, A., Davis, T. P., Li, G., Moad, G., O’Shea, M. S. RAFT polymerization with phthalimidomethyl trithiocarbonates or xanthates. On the origin of bimodal molecular weight distributions in living radical polymerization. Macromolecules 2006, 39, 5307-5318. [CrossRef]
- Hadjichristidis, N.; Iatrou, H.; Pispas, S.; Pitsikalis, M. Anionic polymerization: High vacuum techniques. J. Polym. Sci. Part A Polym. Chem. 2000, 38, 3211–3234.
- Uhrig, D.; Mays, J.W. Experimental techniques in high-vacuum anionic polymerization. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 6179–6222. [CrossRef]
- Provencher, S.W. CONTIN: A general purpose constrained regularization program for inverting noisy linear algebraic and integral equations. Comput. Phys. Commun. 1982, 27, 229–242. [CrossRef]
- Roka, N.; Pitsikalis, M. Statistical copolymers of N-vinylpyrrolidone and benzyl methacrylate via RAFT: Monomer reactivity ratios, thermal properties and kinetics of thermal decomposition J. Macromol. Sci., Part A: Pure Appl. Chem. 2018, 55, 222–23. [CrossRef]
- Hwang, Y.; Patterson, G. D.; Stevens J. R. Photon correlation spectroscopy of bulk poly(n-hexyl methacrylate) near the glass transition J. Polym. Sci. Part B Polym. Phys. Ed. 1996, 34, 2291-2305.
- Hempel, E.; Beiner, M.; Huth, H.; Donth, E. Temperature modulated DSC for the multiple glass transition in poly(n-alkyl methacrylates) Thermochimica Acta 2002, 391, 219-225.
- Gedde, U. W. Polymer Physics, 3rd ed.; Kluwer Academic Publishers: Dordrecht, 1999.
- Alig, I.; Jarek, M.; Hellmann, G.P. Restricted segmental mobility in side-chain crystalline comblike polymers, studied by dielectric relaxation measurements. Macromolecules 1998, 31, 2245–2251. [CrossRef]
- Pitsikalis, M.; Siakali-Kioulafa, E.; Hadjichristidis, N. Block Copolymers of Styrene and Stearyl Methacrylate. Synthesis and Micellization Properties in Selective Solvents Macromolecules 2000, 33, 5460-5469.
- Ruzette, A.-V. G.; Banerjee, P.; Mayes, A. M.; Pollard, M.; Russell, T. P.; Jerome, R.; Slawecki, T.; Hjelm, R.; Thiyagarajan, P. Phase behavior of diblock copolymers between styrene and n-alkyl methacrylates Macromolecules 1998, 31, 8509-8516.
- Gikarakis, T.; Pappas, I.; Arvanitaki, P.; Pantazi, E.; Mitsoni, E.; Roka, N.; Pitsikalis, M. Thermal stability and kinetics of thermal decomposition of statistical copolymers of N-vinylpyrrolidone and alkyl methacrylates synthesized via RAFT polymerization J. Chem. 2021, 6633052. [CrossRef]
- Franco, P.; De Marco I. The use of poly(N-vinyl pyrrolidone) in the delivery of drugs: A review. Polymers 2020, 12, 1114. [CrossRef]
- Moghadamtousi, S.Z.; Kadir, H.A.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; zandi, K. A review on antibacterial, antiviral, and antifungal activity of curcumin. BioMed. Res. Int. 2014, 2014, 1-12.
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggrawal, B.B.; Bioavailability of curcumin: Problems and promise. Molecular Pharmaceutics 2007, 4, 807-818. [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A review of its effects on human health. Foods 2017, 6, 92. [CrossRef]
- Tomeh, M.; Hadianamrei, R.; Zhao, X. A review of curcumin and its derivatives as anticancer agents. Int. J. Mol. Sci. 2019, 20, 1033. [CrossRef]
- Scandalis, A.; Selianitis, D.; Pispas, S. PnBA-b-PNIPAM-b-PDMAEMA thermos-responsive triblock terpolymers and their quaternized analogs as gene and drug delivery vectors. Polymers, 2021, 13, 2361. [CrossRef]
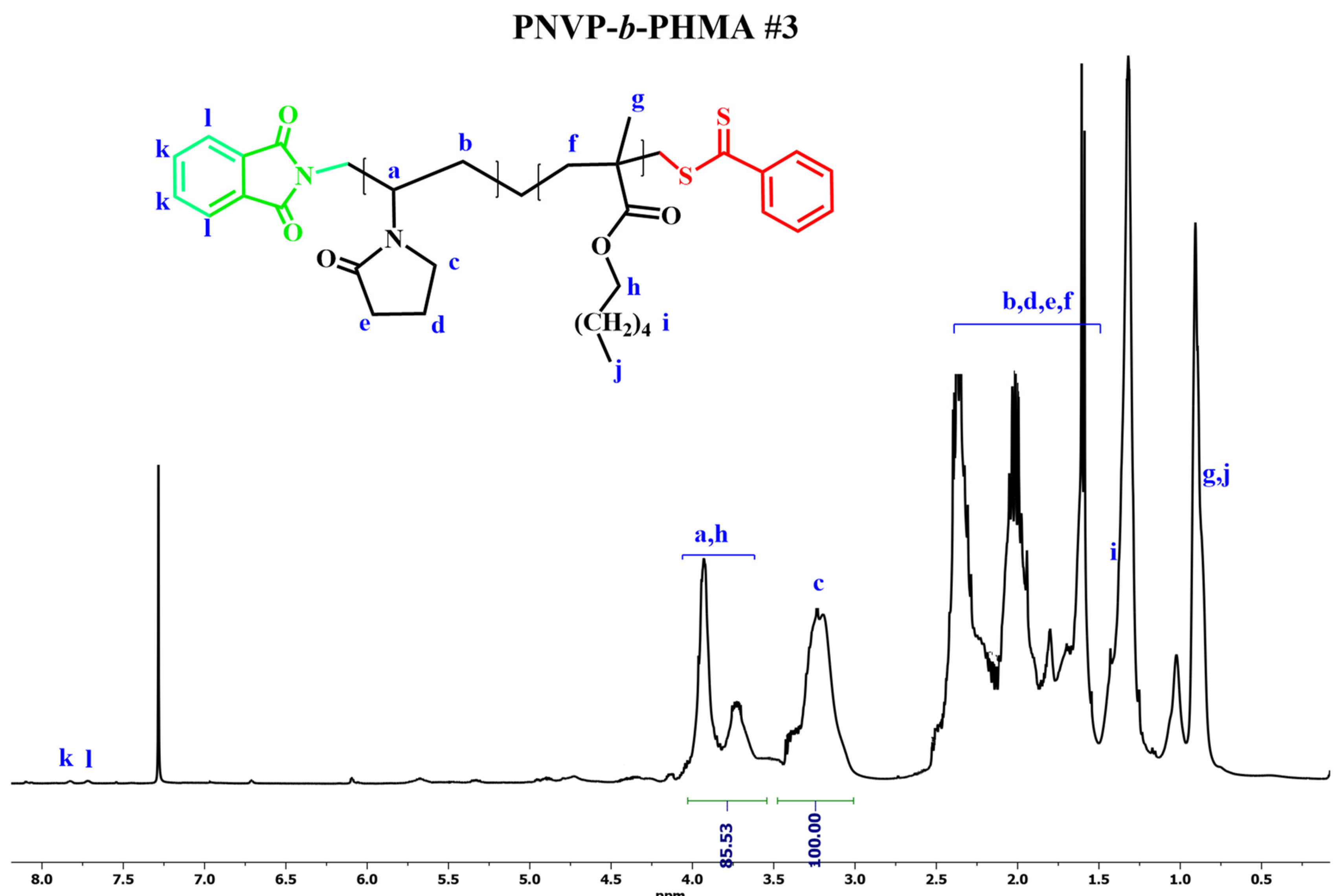
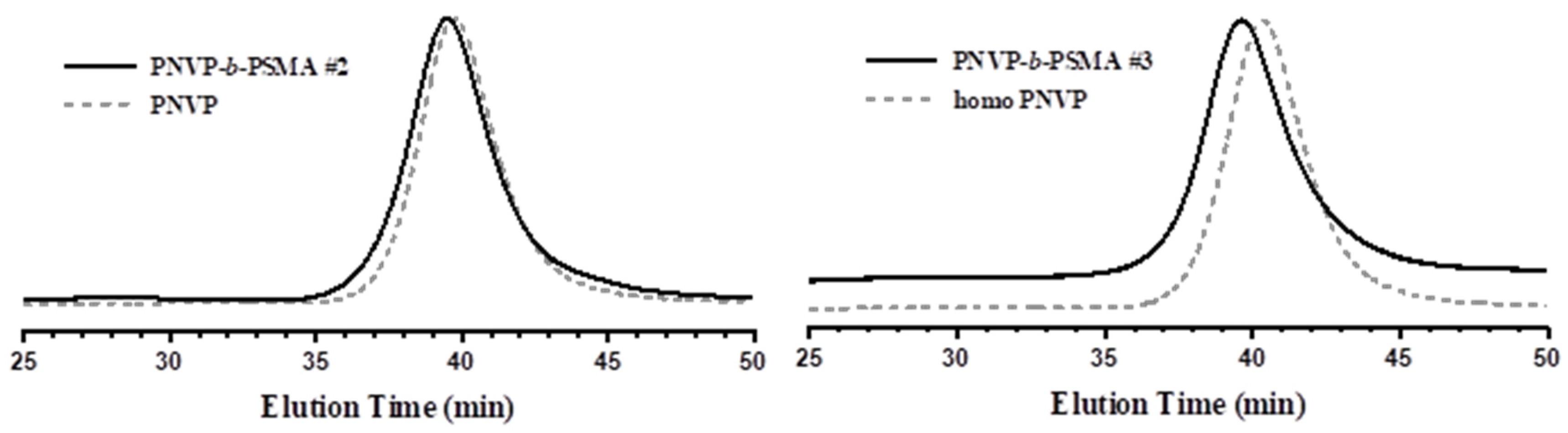
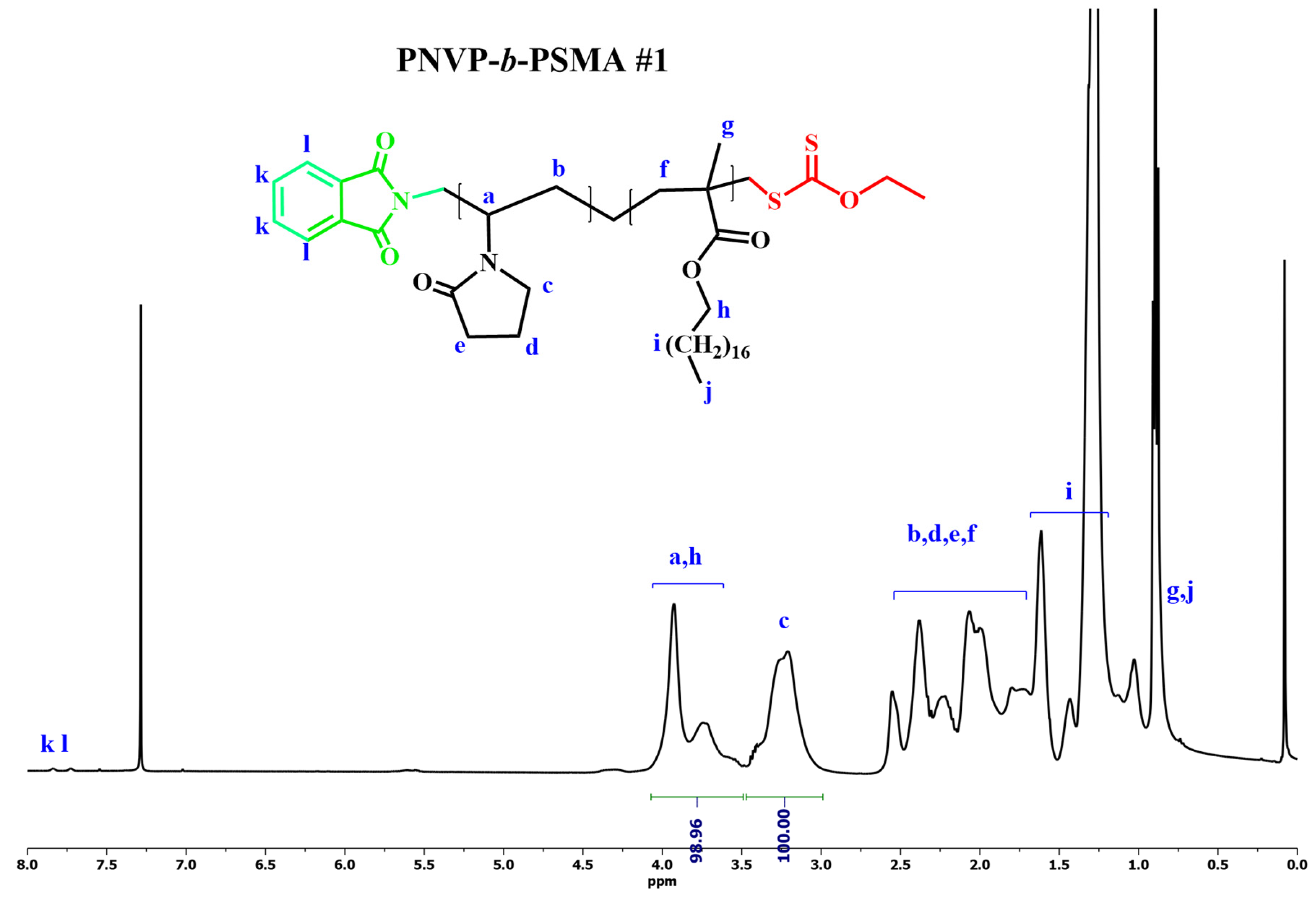
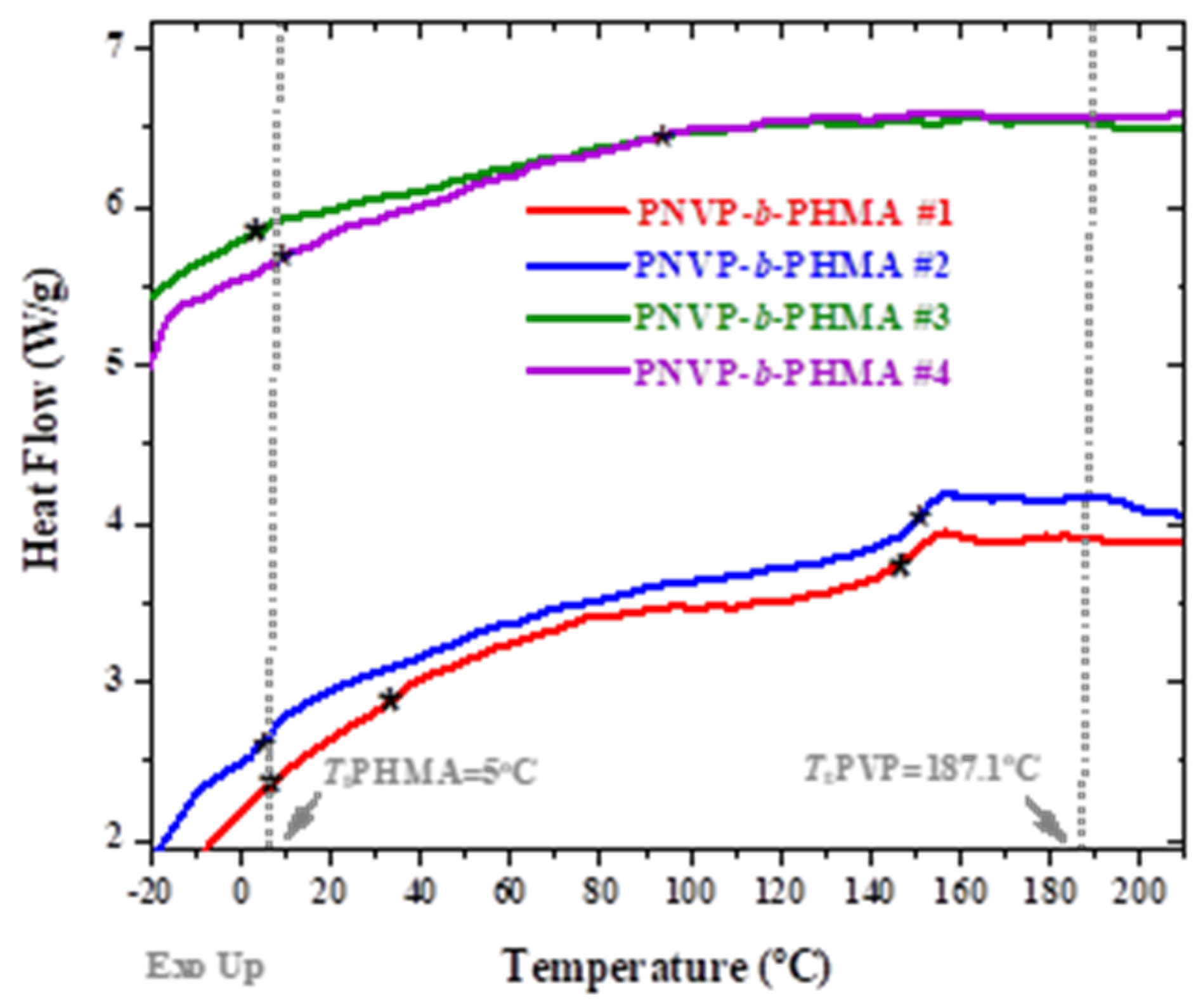
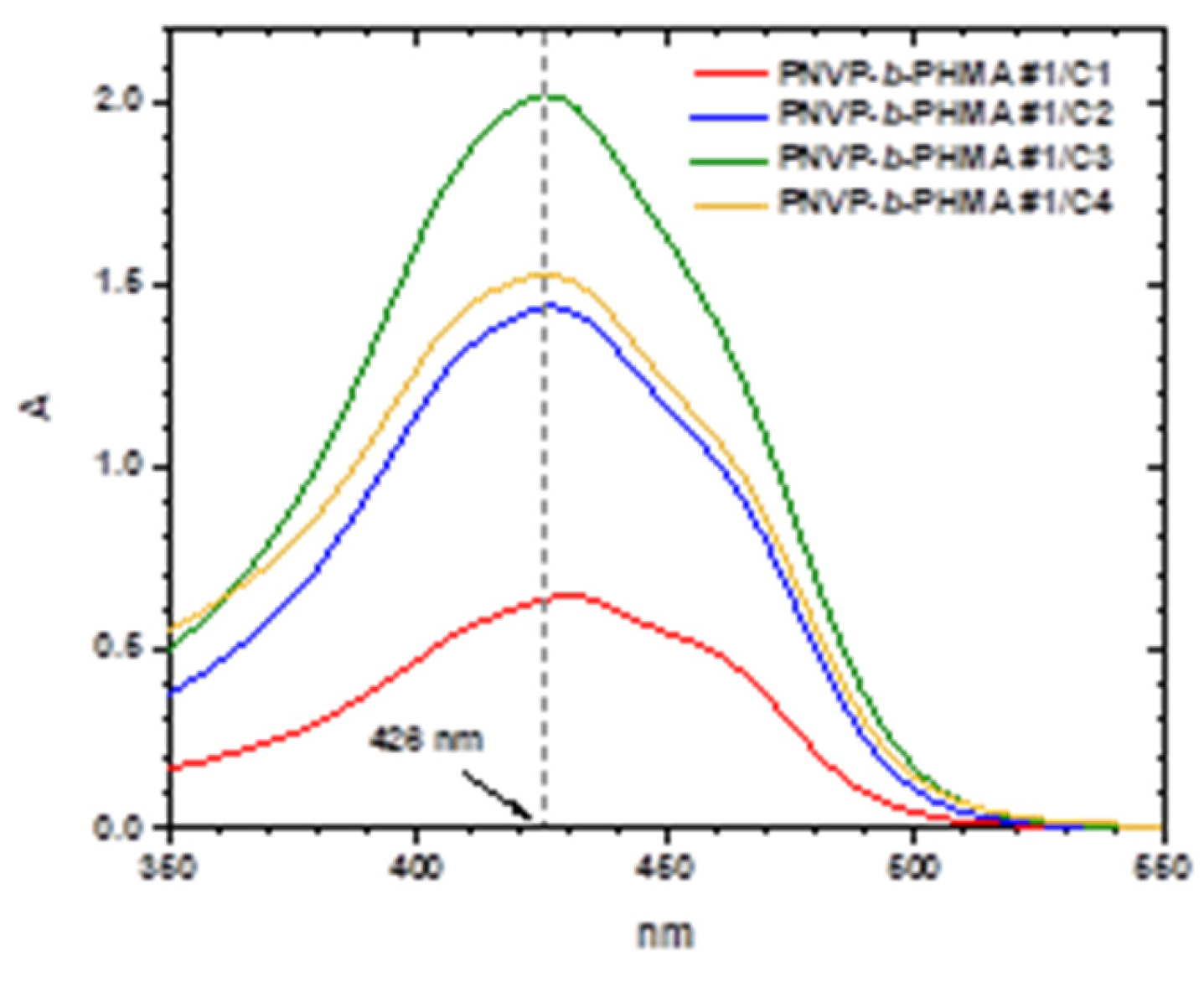
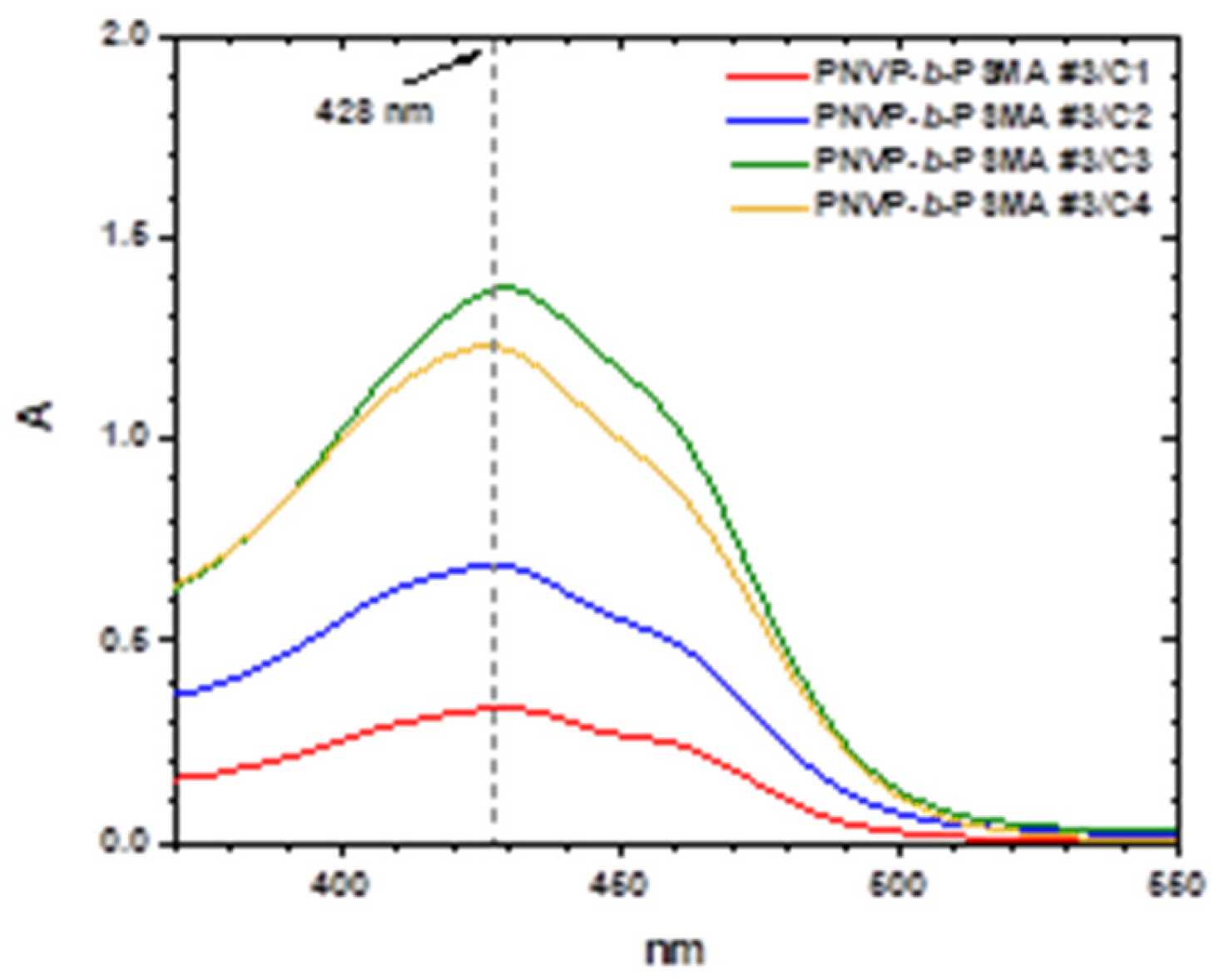
| Sample | PNVP (g) | AIBN (g) | HMA (mL) | Dioxane (mL) |
|---|---|---|---|---|
| PNVP-b-PHMA #1 | 5 | 0.0068 | 1 | 9 |
| PNVP-b-PHMA #2 | 5 | 0.0171 | 1 | 9 |
| PNVP-b-PHMA #3 | 1 | 0.0015 | 3 | 2 |
| PNVP-b-PHMA #4 | 0.5 | 0.0032 | 3 | 2 |
| Sample | PNVP (g) | AIBN (g) | SMA (mL) | Dioxane (mL) |
|---|---|---|---|---|
| PNVP-b-PSMA #1 | 5 | 0.0068 | 1 | 9 |
| PNVP-b-PSMA #2 | 5 | 0.0178 | 1 | 9 |
| PNVP-b-PSMA #3 | 5 | 0.0178 | 3 | 9 |
| macro CTA (PNVP) * |
block copolymers * |
NVP |
SMA |
|||
|---|---|---|---|---|---|---|
| Sample | Mn 103 (Daltons) |
Ð | Mn 103 (Daltons) | Ð | % mol** | % mol** |
| PNVP-b-PSMA #1 | 29.8 | 1.29 | 37 | 1.31 | 67 | 33 |
| PNVP-b-PSMA #2 | 7.9 | 1.28 | 9.3 | 1.35 | 94 | 6 |
| PNVP-b-PSMA #3 | 7.9 | 1.28 | 10.8 | 1.31 | 83 | 17 |
| Sample | Tg Experimental (OC) | ||
|---|---|---|---|
| PNVP-b-PHMA #1 | 11.4 | 36.7 | 151.3 |
| PNVP-b-PHMA #2 | 7.1 | - | 152.8 |
| PNVP-b-PHMA #3 | 5.5 | - | 118.1 |
| PNVP-b-PHMA #4 | 18.5 | - | 114.6 |
| PNVP | - | - | 187.1 |
| PHMA | 5 | - | - |
| Sample | WVSt % | Tm(°C) | ΔH(j/g #) | ΔH(j/gVSt) | Xc % | Tg1(°C) | Tg2(°C) |
|---|---|---|---|---|---|---|---|
| PNVP-b-PSMA #1 | 59.9 | 35.9 | 31.6 | 52.7 | 69.4 | -14.8 | 147.0 |
| PNVP-b-PSMA #2 | 16.2 | 35.2 | 9.6 | 59.2 | 77.9 | -15.9 | 154.7 |
| PNVP-b-PSMA #3 | 38.4 | 36.6 | 22.3 | 58.0 | 76.4 | -7.9 | 147.2 |
| PNVP | - | - | - | - | - | - | 187.1 |
| PSMA | - | 34.0 | 75.9 | - | - | -30 | - |
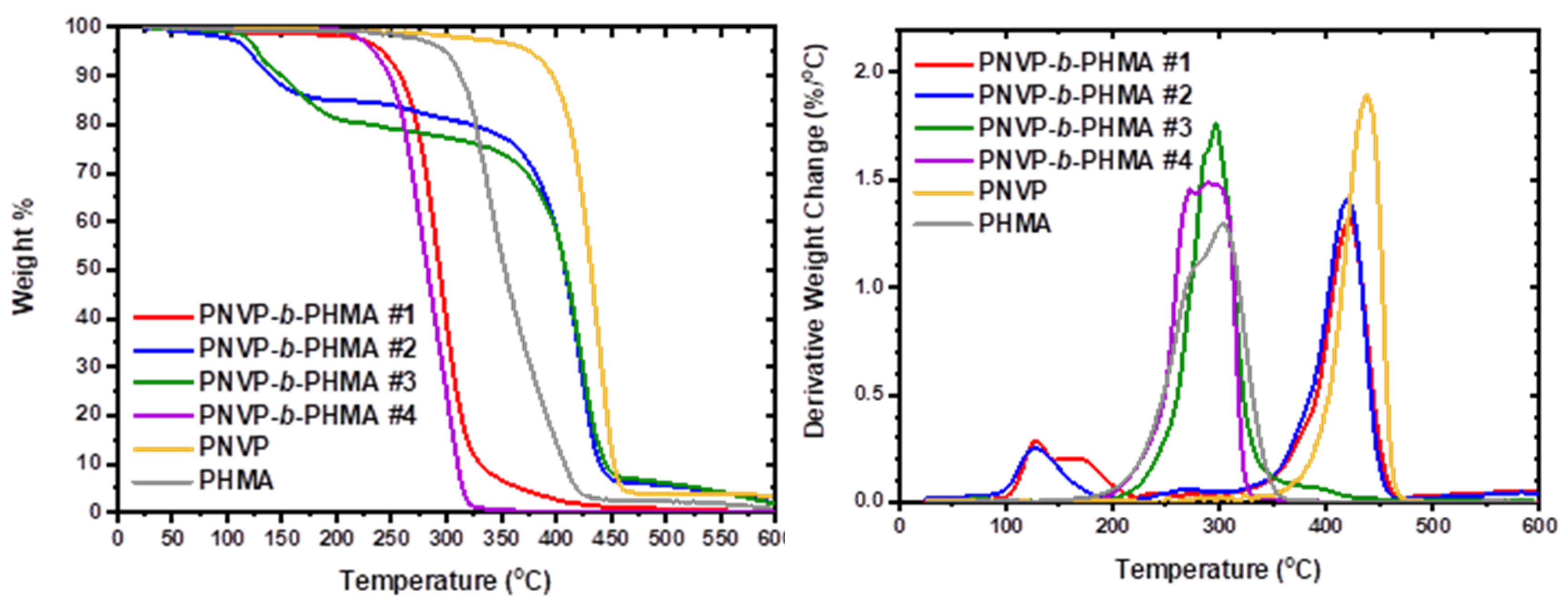
| Sample | Start1 | End1 | Max1 (OC) | Start2 | End2 | Max2 (OC) |
|---|---|---|---|---|---|---|
| PNVP-b-PHMA #1 | 94.9 | 220.4 | 128.7 (broad) | 320.2 | 466.1 | 420.9 |
| PNVP-b-PHMA #2 | 94.9 | 194.5 | 127.7 | 318.9 | 463.8 | 421.0 |
| PNVP-b-PHMA #3 | 204.3 | 413.2 | 296.9 | - | - | - |
| PNVP-b-PHMA #4 | 188.3 | 333.0 | 291.5 (broad) | - | - | - |
| PNVP | - | - | - | 347.63 | 484.06 | 437.53 |
| PHMA | 190.8 | 356.6 | 299.1 (shoulder) | - | - | - |
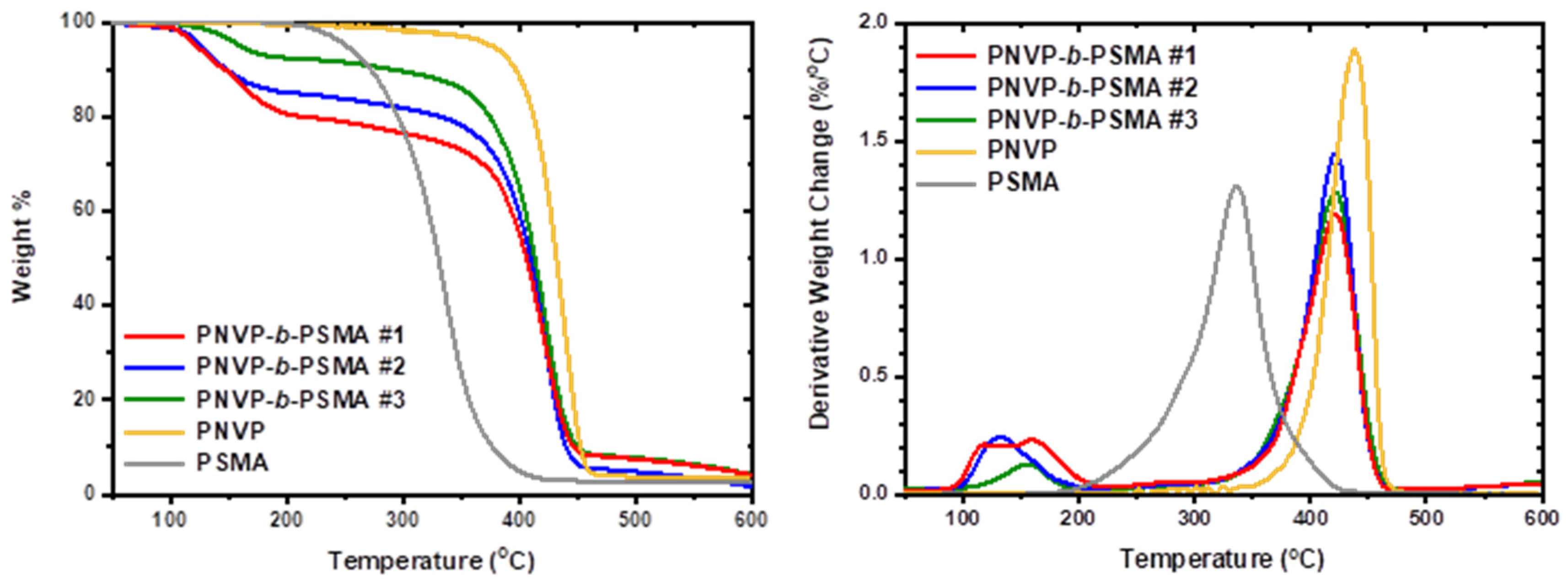
| Sample | Start1 | End1 | Max1 (OC) | Start2 | End2 | Max2 (OC) |
|---|---|---|---|---|---|---|
| PNVP-b-PSMA #1 | 88.8 | 216.6 | 140.8 (broad) | 339.4 | 463.7 | 419.3 |
| PNVP-b-PSMA #2 | 94.3 | 209.2 | 129.1 | 312.6 | 469.2 | 423.5 |
| PNVP-b-PSMA #3 | 113.4 | 198.9 | 154.8 | 323.6 | 470.8 | 421.0 |
| PNVP | - | - | - | 347.6 | 484.0 | 437.5 |
| PSMA | 185.9 | 430.3 | 335.8 | - | - | - |
| Sample | Solvent | MW From SEC |
Mw From SLS |
Nw | Rg (nm) |
A (cm3mol/g2) |
|---|---|---|---|---|---|---|
| PNVP-b-PHMA #1 | CHCl3 | 36x103 | ||||
| PNVP-b-PHMA #1 | THF | 5.59x104 | 1.55 | 2.20x10−4 | ||
| PNVP-b-PHMA #1 | WATER | 5.88x107 | 1633 | 59.9 | 2.96x10−5 | |
| PNVP-b-PHMA #2 | CHCl3 | 13x103 | ||||
| PNVP-b-PHMA #2 | THF | 2.84x104 | 2.18 | 1.75x10−4 | ||
| PNVP-b-PHMA #2 | WATER | 3.58x108 | 27538 | 103.6 | 9.98x10−6 | |
| PNVP-b-PHMA #3 | CHCl3 | 32x103 | ||||
| PNVP-b-PHMA #3 | THF | 1.32x105 | 4.13 | 4.20x10−5 | ||
| PNVP-b-PHMA #3 | WATER | 8.06x108 | 25187 | 124.0 | 1.18x10−5 | |
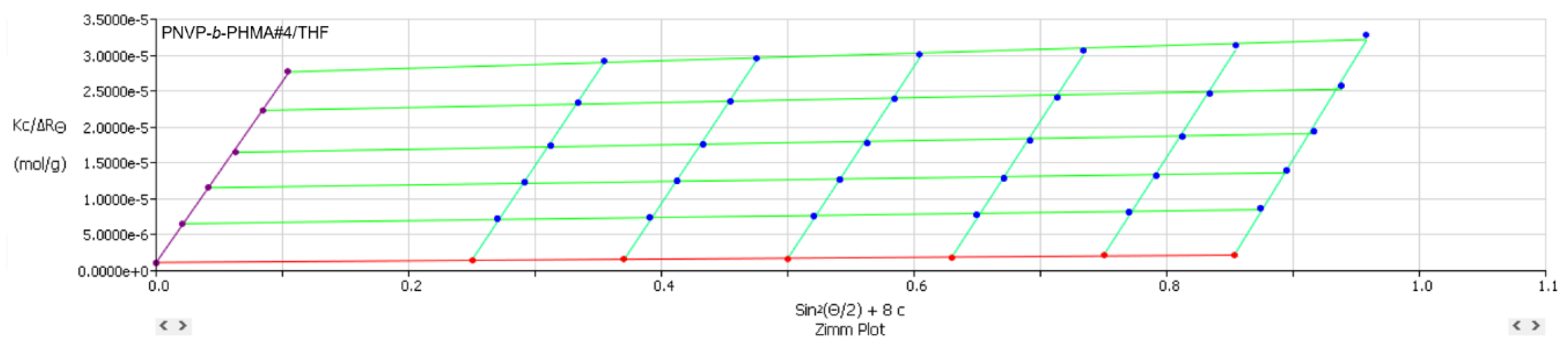
| PNVP-b-PHMA #4 | CHCl3 | 130x103 | ||||
| PNVP-b-PHMA #4 | THF | 8.76x105 | 6.74 | 65.0 | 1.01x10−3 |
| Sample | solvent | Do | Kd | Rg (nm) |
Rho (nm) |
Rg/Rho |
|---|---|---|---|---|---|---|
| PNVP-b-PHMA #1 | CHCl3 | 6.294x10−7 | 23.36 | 6.41 | ||
| PNVP-b-PHMA #1 | THF | 7.635x10−7 | 19.04 | 6.20 | ||
| PNVP-b-PHMA #1 | WATER | 3.469x10−8 | 384 | 59.9 | 70.73 | 0.85 |
| PNVP-b-PHMA #2 | CHCl3 | 3.500x10−7 | 143 | 11.53 | ||
| PNVP-b-PHMA #2 | THF | 9.611x10−7 | 37.59 | 4.93 | ||
| PNVP-b-PHMA #2 | WATER | 2.488x10−8 | 199 | 103.6 | 98.62 | 1.05 |
| PNVP-b-PHMA #3 | CHCl3 | 2.532x10−7 | 160 | 15.94 | ||
| PNVP-b-PHMA #3 | THF | 5.106x10−7 | 33.23 | 9.28 | ||
| PNVP-b-PHMA #3 | WATER | 2.106x10−8 | 1023 | 124 | 116.51 | 1.06 |
| PNVP-b-PHMA #4 | CHCl3 | 1.621x10−7 | 103 | 24.90 | ||
| PNVP-b-PHMA #4 | THF | 2.092x10−7 | 84.35 | 65 | 22.64 | 2.87 |
| asmple | Solvent | Mw From SEC |
Mw From SLS |
Nw | Rg (nm) |
A2 (cm3mol/g2) |
|---|---|---|---|---|---|---|
| PNVP-b-PSMA #1 | CHCl3 | 37x103 | ||||
| PNVP-b-PSMA #1 | THF | 2.53x105 | 6.84 | 6.00x10−4 | ||
| PNVP-b-PSMA #1 | WATER | 7.53x107 | 2035 | 48.0 | 4.53x10−5 | |
| PNVP-b-PSMA #2 | CHCl3 | 9.3x103 | ||||
| PNVP-b-PSMA #2 | THF | 4.70x104 | 5.05 | 3.50x10−4 | ||
| PNVP-b-PSMA #2 | WATER | 3.68x107 | 3957 | 43.8 | 7.40x10−5 | |
| PNVP-b-PSMA #3 | CHCl3 | 10.8x103 | ||||
| PNVP-b-PSMA #3 | THF | 7.20x104 | 6.67 | 2.50x10−4 | ||
| PNVP-b-PSMA #3 | WATER | 1.192x107 | 1104 | 36.1 | 7.30x10−5 |
| Sample | solvent | Do | Kd | Rg (nm) | Rho (nm) | Rg/Rho |
|---|---|---|---|---|---|---|
| PNVP-b-PSMA #1 | CHCl3 | 3.289x10−7 | 158.8 | 12.27 | ||
| PNVP-b-PSMA #1 | THF | 6.295x10−7 | 50.05 | 7.53 | ||
| PNVP-b-PSMA #1 | WATER | 3.171x10−8 | 1179 | 48 | 77.38 | 0.62 |
| PNVP-b-PSMA #2 | CHCl3 | 3.116x10−7 | 231.2 | 12.96 | ||
| PNVP-b-PSMA #2 | THF | 9.033x10−7 | 28.90 | 5.24 | ||
| PNVP-b-PSMA #2 | WATER | 4.418x10−8 | 952.0 | 43.8 | 55.53 | 0.79 |
| PNVP-b-PSMA #3 | CHCl3 | 2.567x10−7 | 166.0 | 15.72 | ||
| PNVP-b-PSMA #3 | THF | 6.577x10−7 | 156.8 | 7.20 | ||
| PNVP-b-PSMA #3 | WATER | 6.474x10−8 | 462.9 | 36.1 | 37.90 | 0.95 |
| Sample | DLC% | DLE% |
|---|---|---|
| PNVP-b-PHMA #1 | ||
| 1/c1 | 0.59 | 50.54 |
| 1/c2 | 1.35 | 76.50 |
| 1/c3 | 1.92 | 67.96 |
| 1/c4 | 1.46 | 38.58 |
| PNVP-b-PHMA #2 | ||
| 2/c1 | 0.12 | 13.30 |
| 2/c2 | 0.78 | 35.75 |
| 2/c3 | 0.82 | 29.09 |
| 2/c4 | 1.01 | 29.56 |
| PNVP-b-PHMA #3 | ||
| 3/c1 | 0.32 | 34.65 |
| 3/c2 | 0.99 | 48.46 |
| 3/c3 | 1.28 | 47.33 |
| 3/c4 | 1.60 | 48.46 |
| Sample | DLC% | DLE% |
|---|---|---|
| PNVP-b-PSMA #1 | - | - |
| 1/c1 | 0.40 | 39.97 |
| 1/c2 | 0.91 | 40.99 |
| 1/c3 | 1.32 | 46.11 |
| 1/c4 | 1.68 | 47.01 |
| PNVP-b-PSMA #2 | ||
| 2/c1 | 0.14 | 11.88 |
| 2/c2 | 0.92 | 39.21 |
| 2/c3 | 0.81 | 26.74 |
| 2/c4 | 1.43 | 39.57 |
| PNVP-b-PSMA #3 | ||
| 3/c1 | 0.27 | 24.01 |
| 3/c2 | 0.61 | 26.39 |
| 3/c3 | 1.26 | 43.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




