Submitted:
25 March 2025
Posted:
26 March 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Regulatory Framework and Their Challenges
3. Microbiological Testing in Pharmaceuticals
4. Europe’s State of Microbial Contaminations
5. Future Directions
6. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AI | Artificial intelligence |
| AMR | Antimicrobial resistance |
| ATCC | American Type Culture Collection |
| BET | Bacterial endotoxins test |
| CFU | Colony-forming units |
| CPPs | Critical process parameters |
| CQAs | Critical quality attributes |
| EC | European Commission |
| EDQM | European Directorate for the Quality of Medicines & HealthCare |
| EMA | European medicine agency |
| EU | European Union |
| FDA | Food and Drug Administration |
| FTIR | Fourier Transform Infrared |
| GMP | Good Manufacturing Practices |
| ICH | International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use |
| LAL | Limulus Amoebocyte Lysate |
| MHRA | United Kingdom's Medicines and Healthcare products Regulatory Agency |
| NGS | Next-Generation Sequencing |
| Ph. Eur. | European Pharmacopoeia |
| QbD | Quality by Design |
| rBET | recombinant BET |
| rFC | Recombinant Factor C |
| TAMC | Total aerobic microbial count |
| TYMC | Total yeast and mold count |
| USP | United States Pharmacopeia |
| VBNC | Viable but non-culturable cells |
References
- Rodrigues M, Rodrigues I, Conceição J. Importância dos Medicamentos Manipulados na Terapêutica: Revisão histórica e estado atual. Acta Farm Port 2023;12:51–70.
- Blanco Barrantes J, Quesada MS, Rojas G, Loría A. A Journey through the History of Drug Quality Control, from Greece to Costa Rica. Int J Drug Regul Aff 2021;9:33–47. [CrossRef]
- Tabajara de Oliveira Martins D, Rodrigues E, Casu L, Benítez G, Leonti M. The historical development of pharmacopoeias and the inclusion of exotic herbal drugs with a focus on Europe and Brazil. J Ethnopharmacol 2019;240:111891. [CrossRef]
- Hashim Z, Celiksoy V. Pharmaceutical products microbial contamination: approaches of detection and avoidance. Microbes Infect Dis 2024;0:0–0. [CrossRef]
- Ekeleme UG, Ikwuagwu VO, Chukwuocha UM, Nwakanma JC, Adiruo SA, Ogini IO, et al. Detection and characterization of micro-organisms linked to unsealed drugs sold in Ihiagwa community, Owerri, Imo State, Nigeria. Access Microbiol 2024;6:1–7. [CrossRef]
- Kauffman CA, Malani AN. Fungal infections associated with contaminated steroid injections. Emerg Infect 10 2016:359–74. [CrossRef]
- Brooks RB, Mitchell PK, Miller JR, Vasquez AM, Havlicek J, Lee H, et al. Multistate Outbreak of Burkholderia cepacia Complex Bloodstream Infections after Exposure to Contaminated Saline Flush Syringes: United States, 2016-2017. Clin Infect Dis 2019;69:445–9. [CrossRef]
- Chen YY, Huang WT, Chen CP, Sun SM, Kuo FM, Chan YJ, et al. An Outbreak of Ralstonia pickettii Bloodstream Infection Associated with an Intrinsically Contaminated Normal Saline Solution. Infect Control Hosp Epidemiol 2017;38:444–8. [CrossRef]
- Eissa ME. Distribution of bacterial contamination in non-sterile pharmaceutical materials and assessment of its risk to the health of the final consumers quantitatively. Beni-Suef Univ J Basic Appl Sci 2016;5:217–30. [CrossRef]
- Casiraghi A, Centin G, Selmin F, Picozzi C, Minghetti P, Zanon D. Critical aspects in the preparation of extemporaneous flecainide acetate oral solution for paediatrics. Pharmaceutics 2021;13:1–12. [CrossRef]
- European Association of Hospital Pharmacists. EAHP 2023 Shortage Survey Report. Eur Assoc Hosp Pharm 2023:1–67.
- Santos AMC, Doria MS, Meirinhos-Soares L, Almeida AJ, Menezes JC. A QRM discussion of microbial contamination of non-sterile drug products, using FDA and EMA warning letters recorded between 2008 and 2016. PDA J Pharm Sci Technol 2018;72:62–72. [CrossRef]
- The European Medicines Agency - National competent authorities (human). EMA 2025. https://www.ema.europa.eu/en/partners-networks/eu-partners/eu-member-states/national-competent-authorities-human (accessed February 12, 2025).
- GOV.UK Alerts, recalls and safety information: medicines and medical devices. GOVUK 2025. https://www.gov.uk/drug-device-alerts (accessed January 29, 2025).
- Swissmedic - Batch recalls human medicines. SWISSMEDIC 2025. https://www.swissmedic.ch/swissmedic/en/home/humanarzneimittel/market-surveillance/qualitaetsmaengel-und-chargenrueckrufe/batch-recalls.html (accessed February 12, 2025).
- European Medicines Agency (EMA) 2025. https://www.ema.europa.eu/en/homepage (accessed February 24, 2025).
- European Directorate for the Quality of Medicines & HealthCare 2025. https://www.edqm.eu/en/ (accessed February 24, 2025).
- Commission E. Good Manufacturing Practice (GMP) guidelines In: The rules governing medicinal products in the European Union. Eur Comm 2011;4. https://health.ec.europa.eu/medicinal-products/eudralex/eudralex-volume-4_en (accessed February 18, 2025).
- Commission E. Regulation (EC) No 852/2004 of the European Parliament on the hygiene of foodstuffs. Eur Comm 2004. https://eur-lex.europa.eu/eli/reg/2004/852/oj/eng (accessed February 18, 2025).
- Commission E. Regulation (EC) No 1223/2009 of the European Parliament on cosmetic products. Eur Comm 2009. https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009R1223 (accessed February 18, 2025).
- Commission E. Regulation (EC) No 83/2001 on the Community code relating to medicinal products for human use. Eur Comm 2001. https://eur-lex.europa.eu/eli/dir/2001/83/oj/eng (accessed February 18, 2025).
- Commission E. Regulation (EC) No 2073/2005 on microbiological criteria for foodstuffs. Eur Comm 2005. https://eur-lex.europa.eu/eli/reg/2005/2073/oj/eng.
- HealthCare ED for the Q of M&. European Pharmacopoeia. 11th Editi. Strasbourg, France: Council of Europe; 2025.
- Hałasa R, Turecka K, Smoktunowicz M, Mizerska U, Orlewska C. Application of tris-(4,7-Diphenyl-1,10 phenanthroline)ruthenium(II) Dichloride to Detection of Microorganisms in Pharmaceutical Products †. Pharmaceuticals 2023;16:1–15. [CrossRef]
- Austin PD, Hand KS, Elia M. Systematic review and meta-analysis of the risk of microbial contamination of parenteral doses prepared under aseptic techniques in clinical and pharmaceutical environments: An update. J Hosp Infect 2015;91:306–18. [CrossRef]
- Lourenço FR, Bettencourt da Silva RJN. Simplified and Detailed Evaluations of the Uncertainty of the Measurement of Microbiological Contamination of Pharmaceutical Products. J AOAC Int 2024;107:856–66. [CrossRef]
- Ansari FA, Perazzolli M, Husain FM, Khan AS, Ahmed NZ, Meena RP. Novel decontamination approaches for stability and shelf-life improvement of herbal drugs: A concise review. The Microbe 2024;3:100070. [CrossRef]
- Dao H, Lakhani P, Police A, Kallakunta V, Ajjarapu SS, Wu KW, et al. Microbial Stability of Pharmaceutical and Cosmetic Products. AAPS PharmSciTech 2018;19:60–78. [CrossRef]
- Center for Drug Evaluation and Research. Microbiological Quality Considerations in Non-sterile Drug Manufacturing Guidance for Industry. Food Drug Adm 2021:1–24.
- Song M, Li Q, Liu C, Wang P, Qin F, Zhang L, et al. A comprehensive technology strategy for microbial identification and contamination investigation in the sterile drug manufacturing facility—a case study. Front Microbiol 2024;15:1–10. [CrossRef]
- Rayan, RA. Pharmaceutical effluent evokes superbugs in the environment: A call to action. Biosaf Heal 2023;5:363–71. [CrossRef]
- Endale H, Mathewos M, Abdeta D. Potential Causes of Spread of Antimicrobial Resistance and Preventive Measures in One Health Perspective-A Review. Infect Drug Resist 2023;16:7515–45. [CrossRef]
- Cen T, Zhang X, Xie S, Li D. Preservatives accelerate the horizontal transfer of plasmid-mediated antimicrobial resistance genes via differential mechanisms. Environ Int 2020;138:105544. [CrossRef]
- Naveed M, Chaudhry Z, Bukhari SA, Meer B, Ashraf H. Antibiotics resistance mechanism. Elsevier Inc.; 2019. [CrossRef]
- The United States Pharmacopeial Convention. United States Pharmacopeia 11th Edition. 11th ed. United States Pharmacopeial Convention; 2024.
- Shukla AK, Boruah JS, Park S, Kim B. Rapid Detection and Counting of Bacteria Using Impact Electrochemical Impedance Spectroscopy. J Phys Chem C 2024;128:13458–63. [CrossRef]
- Jia K, Ionescu RE. Measurement of Bacterial Bioluminescence Intensity and Spectrum: Current Physical Techniques and Principles. Adv. Biochem. Eng. Biotechnol., vol. 123, 2015, p. 19–45. [CrossRef]
- Ferone M, Gowen A, Fanning S, Scannell AGM. Microbial detection and identification methods: Bench top assays to omics approaches. Compr Rev Food Sci Food Saf 2020;19:3106–29. [CrossRef]
- Deng Y, Beahm DR, Ionov S, Sarpeshkar R. Measuring and modeling energy and power consumption in living microbial cells with a synthetic ATP reporter. BMC Biol 2021;19:1–21. [CrossRef]
- Perry, JD. A Decade of Development of Chromogenic Culture Media for Clinical Microbiology in an Era of Molecular Diagnostics. Clin Microbiol Rev 2017;30:449–79. [CrossRef]
- Vanhee LME, D’Haese E, Cools I, Nelis HJ, Coenye T. Detection and quantification of bacteria and fungi using solid-phase cytometry. NATO Sci Peace Secur Ser A Chem Biol 2010:25–41. [CrossRef]
- Müller V, Sousa JM, Ceylan Koydemir H, Veli M, Tseng D, Cerqueira L, et al. Identification of pathogenic bacteria in complex samples using a smartphone based fluorescence microscope. RSC Adv 2018;8:36493–502. [CrossRef]
- Cody RB, McAlpin CR, Cox CR, Jensen KR, Voorhees KJ. Identification of bacteria by fatty acid profiling with direct analysis in real time mass spectrometry. Rapid Commun Mass Spectrom 2015;29:2007–12. [CrossRef]
- Tiquia-Arashiro S, Li X, Pokhrel K, Kassem A, Abbas L, Coutinho O, et al. Applications of Fourier Transform-Infrared spectroscopy in microbial cell biology and environmental microbiology: advances, challenges, and future perspectives. Front Microbiol 2023;14. [CrossRef]
- Arbefeville SS, Timbrook TT, Garner CD. Evolving strategies in microbe identification-a comprehensive review of biochemical, MALDI-TOF MS and molecular testing methods. J Antimicrob Chemother 2024;79:2–8. [CrossRef]
- Takahashi H, Horio K, Kato S, Kobori T, Watanabe K, Aki T, et al. Direct detection of mRNA expression in microbial cells by fluorescence in situ hybridization using RNase H-assisted rolling circle amplification. Sci Rep 2020;10:1–8. [CrossRef]
- Trinh TND, Lee NY. Advances in Nucleic Acid Amplification-Based Microfluidic Devices for Clinical Microbial Detection. Chemosensors 2022;10. [CrossRef]
- Jeon S, Lim N, Park S, Park M, Kim S. Comparison of PFGE, IS6110-RFLP, and 24-Locus MIRU-VNTR for molecular epidemiologic typing of Mycobacterium tuberculosis isolates with known epidemic connections. J Microbiol Biotechnol 2018;28:338–46. [CrossRef]
- Ahmad S, Lohiya S, Taksande A, Meshram RJ, Varma A, Vagha K. A Comprehensive Review of Innovative Paradigms in Microbial Detection and Antimicrobial Resistance: Beyond Traditional Cultural Methods. Cureus 2024;16:1–8. [CrossRef]
- Rychert, J. Benefits and Limitations of MALDI-TOF Mass Spectrometry for the Identification of Microorganisms. J Infect 2019;2:1–5. [CrossRef]
- Singhal N, Kumar M, Kanaujia PK, Virdi JS. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front Microbiol 2015;6:1–16. [CrossRef]
- Wespel M, Geiss M, Nägele M, Combé S, Reich J, Studts J, et al. The impact of endotoxin masking on the removal of endotoxin during manufacturing of a biopharmaceutical drug product. J Chromatogr A 2022;1671:462995. [CrossRef]
- Schneier M, Razdan S, Miller AM, Briceno ME, Barua S. Current technologies to endotoxin detection and removal for biopharmaceutical purification. Biotechnol Bioeng 2020;117:2588–609. [CrossRef]
- Baker E, Ponder J, Oberdorfer J, Spreitzer I, Bolden J, Marius M, et al. Barriers to the Use of Recombinant Bacterial Endotoxins Test Methods in Parenteral Drug, Vaccine and Device Safety Testing. Altern to Lab Anim 2023;51:401–10. [CrossRef]
- Cundell, T. What Microbial Tests Should be Considered Stability Test Parameters? Am Pharm Rev 2024;26:18–22.
- Jimenez L. Analysis of FDA Enforcement Reports (2012-2019) to Determine the Microbial Diversity in Contaminated Non-Sterile and Sterile Drugs. Am Pharm Rev 2019. https://www.americanpharmaceuticalreview.com/Featured-Articles/518912-Analysis-of-FDA-Enforcement-Reports-2012-2019-to-Determine-the-Microbial-Diversity-in-Contaminated-Non-Sterile-and-Sterile-Drugs/ (accessed February 17, 2025).
- Tavares M, Kozak M, Balola A, Sá-Correia I. Burkholderia cepacia complex bacteria: A feared contamination risk in water-based pharmaceutical products. Clin Microbiol Rev 2020;33. [CrossRef]
- Sutton S, Jimenez L. A Review of Reported Recalls Involving Microbiological Control (2004-2011) with Emphasis on FDA Considerations of Objectionable Organisms. Am Pharm Rev 2012. https://www.americanpharmaceuticalreview.com/Featured-Articles/38382-A-Review-of-Reported-Recalls-Involving-Microbiological-Control-2004-2011-with-Emphasis-on-FDA-Considerations-of-Objectionable-Organisms/ (accessed February 17, 2025).
- European product recalls break records for fifth consecutive year in 2023. Sedwick 2024. https://www.sedgwick.com/press-release/european-product-recalls-break-records-for-fifth-consecutive-year-in-2023/?loc=eu.
- European product recalls on track to reach a 10-year high in 2024. Sedwick 2024. https://www.sedgwick.com/en-gb/press-release/european-product-recalls-on-track-to-reach-a-10-year-high-in-2024/?loc=eu (accessed February 17, 2025).
- Sandle, T. Review of FDA warning letters for microbial bioburden issues (2001-2011). Pharma Times 2012;44:29–30.
- Jain SK, Jain RK. Review of fda warning letters to pharmaceuticals: Cause and effect analysis. Res J Pharm Technol 2018;11:3219–26. [CrossRef]
- Hall K, Stewart T, Chang J, Kelly Freeman M. Characteristics of FDA drug recalls: A 30-month analysis. Am J Heal Pharm 2016;73:235–40. [CrossRef]
- Alsulimani A, Akhter N, Jameela F, Ashgar RI, Jawed A, Hassani MA, et al. The Impact of Artificial Intelligence on Microbial Diagnosis. Microorganisms 2024;12:1–20. [CrossRef]
- Elshafei, AM. Exploring the Role of Artificial Intelligence in Revolutionizing Microbial Diagnostics. Asian J Res Infect Dis 2024;15:79–90. [CrossRef]
- Tsitou VM, Rallis D, Tsekova M, Yanev N. Microbiology in the era of artificial intelligence: transforming medical and pharmaceutical microbiology. Biotechnol Biotechnol Equip 2024;38. [CrossRef]
- Cheruvari A, Kammara R. Bacteriocins future perspectives: Substitutes to antibiotics. Food Control 2025;168:110834. [CrossRef]
- Darbandi A, Asadi A, Mahdizade Ari M, Ohadi E, Talebi M, Halaj Zadeh M, et al. Bacteriocins: Properties and potential use as antimicrobials. J Clin Lab Anal 2022;36:1–40. [CrossRef]
- Kaleem AM, Koilpillai J, Narayanasamy D. Mastering Quality: Uniting Risk Assessment With Quality by Design (QbD) Principles for Pharmaceutical Excellence. Cureus 2024;16. [CrossRef]
- Sangshetti JN, Deshpande M, Zaheer Z, Shinde DB, Arote R. Quality by design approach: Regulatory need. Arab J Chem 2017;10:S3412–25. [CrossRef]
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). ICH Qual Guidel 2025. https://www.ich.org/ (accessed February 25, 2025).
- Newby, P. Newby P. The Significance and Detection of VBNC Microorganisms. Am Pharm Rev 2007. https://www.americanpharmaceuticalreview.com/Featured-Articles/113051-The-Significance-and-Detection-of-VBNC-Microorganisms/ (accessed February 27, 2025).
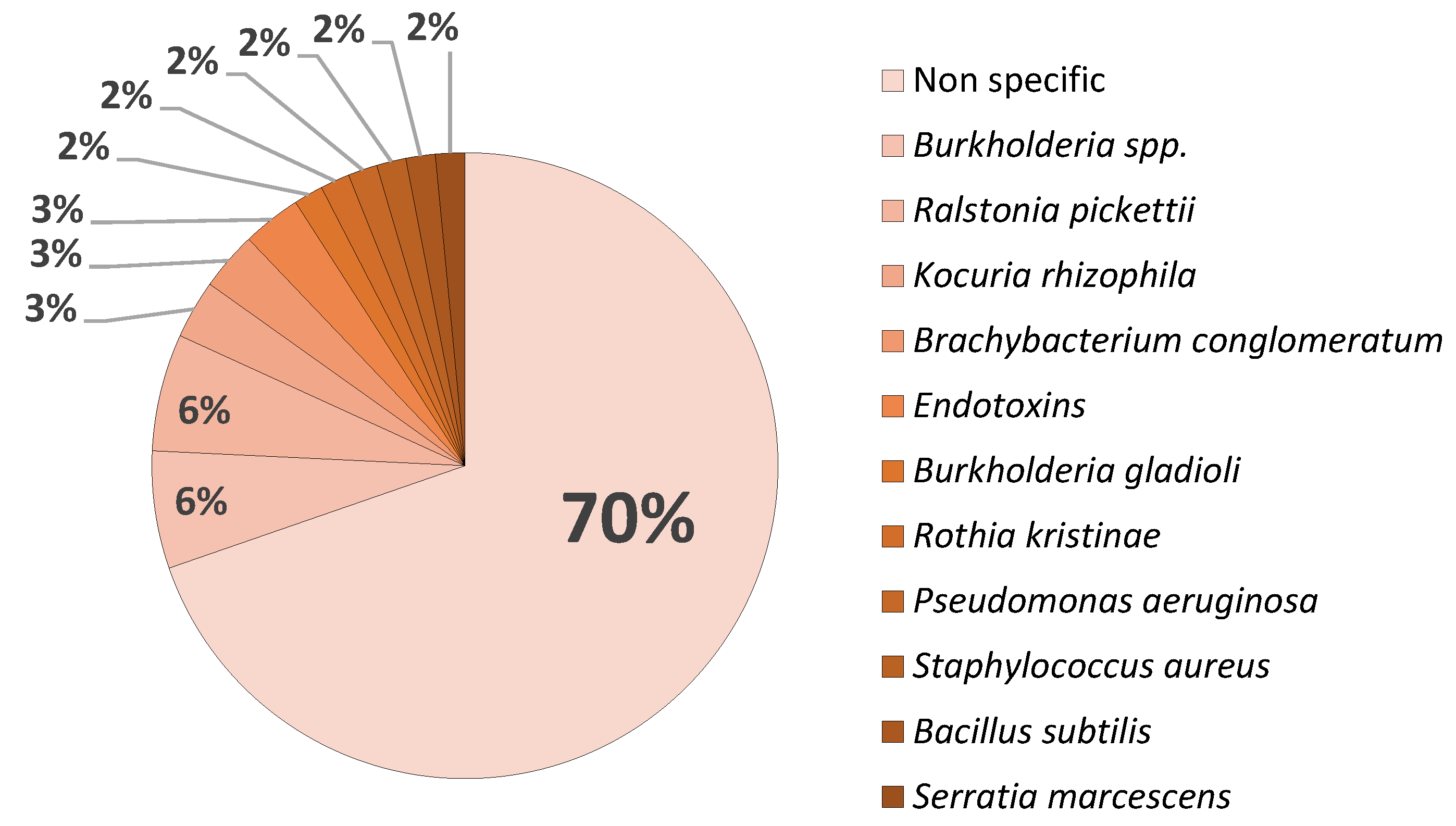
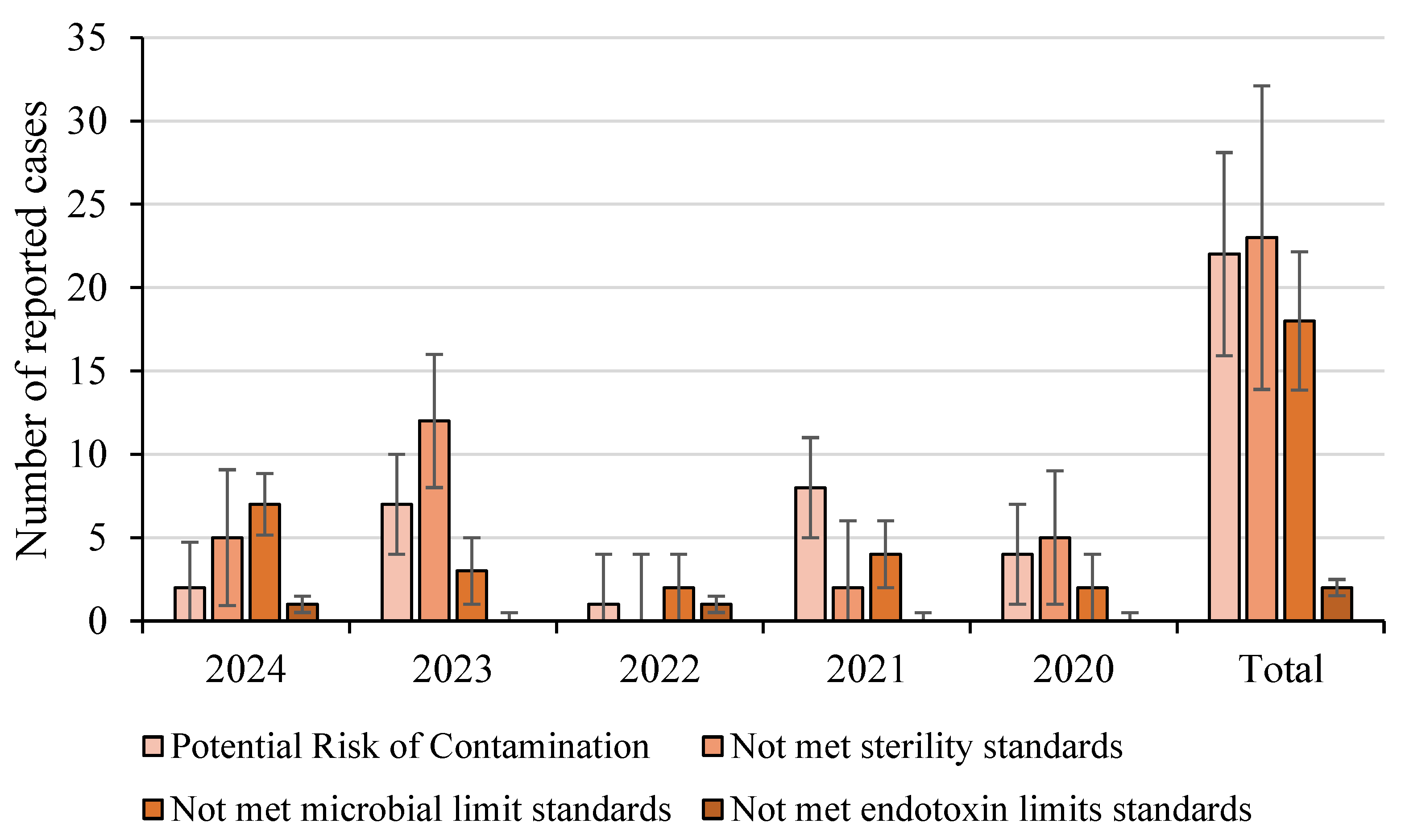
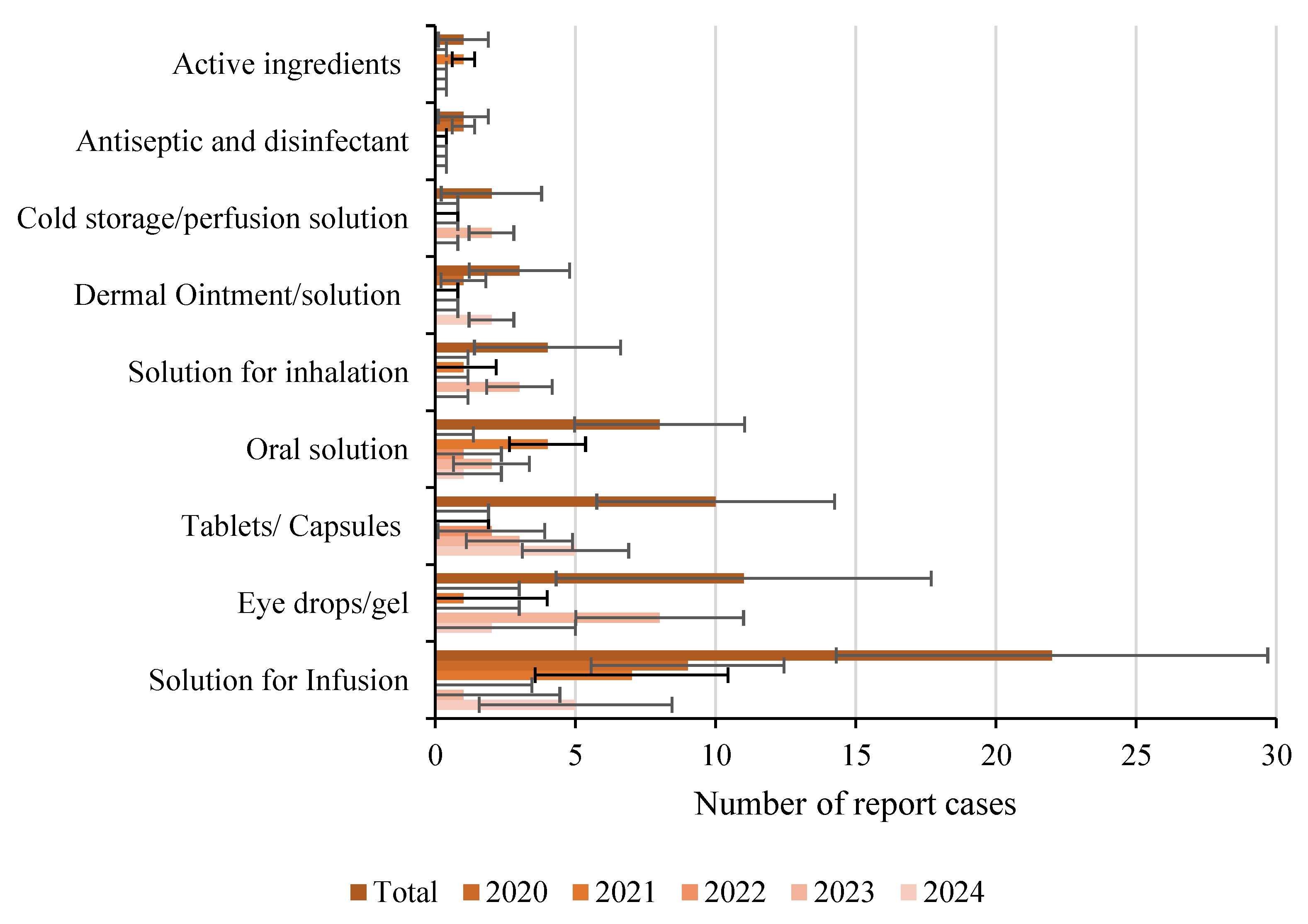
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



