1. Introduction
Hypoplasia of tongue and mandible is an uncommon combination of congenital malformations, despite their shared origin. Tongue and mandible come from the same embryonic structure. During the 4th week of embryonic development, the first branchial arch divides into maxillary and mandibular processes and tuberculum impar. The latter two form mandible and anterior two-thirds of tongue. Posterior third of the tongue develops from second and third branchial arch. Hypoglossia and micrognathia most likely develop together, caused by the same unidentified factor, which probably affects the first branchial arch during organogenesis [
1].
Situs inversus totalis, however, is more prevalent in the general population. It is estimated that it occurs in 1 in 8000 to 25000 people. It can be inherited as an autosomal recessive or X-chromosome-linked trait. However, in a certain number of cases, the existence of this anomaly cannot be found in the family at all. Situs inversus is sometimes associated with heart and/or spleen anomalies, and mucociliary disfunction [
2].
It is unclear whether hypoglossia, micrognathia and situs inversus can stem from a same cause, but we know that this fetus was exposed to SARS-CoV-2 infection during the first 6 weeks of intrauterine, embryonic development.
It is also unclear whether the combination of these three malformations can be classified as its own syndrome or not. Cases like these are sporadic and information about them is scarce. That is exactly why it is important to show this case. Here we present one such a case.
2. Case Presentation
The patient is a female term newborn that was hospitalized at the Neonatology department of the Pediatric Clinic in Novi Sad, 1 day after birth. The parents are non-consanguineous, healthy 18 years old mother and 19 years old father, with no history of congenital illnesses or malformations in their respective families. The mother denies using alcohol, nicotine, or drugs during pregnancy, and also denies being exposed to toxic substances during that time. She didnt use any medication other than acetaminophen, which she took while being sick during 4th week of pregnancy. She recalls having diarrhea, vomiting and headache with an elevated temperature of around 37°C. Pregnant women was tested with a PCR test that was positive for the SARS-CoV-19 virus but no further diagnostic testing, genetic or other, was conducted until birth. In the later weeks of pregnancy the woman felt healthy, without symptoms. Polyhydramnios was found on an ultrasound scan during 26th week of pregnancy.
She was delivered at term by Caesarean section, which was indicated due to dystocia. Her birth weight was 3800 g, birth length was 51 cm, and head circumference was 35 cm (all three were 75th centile).
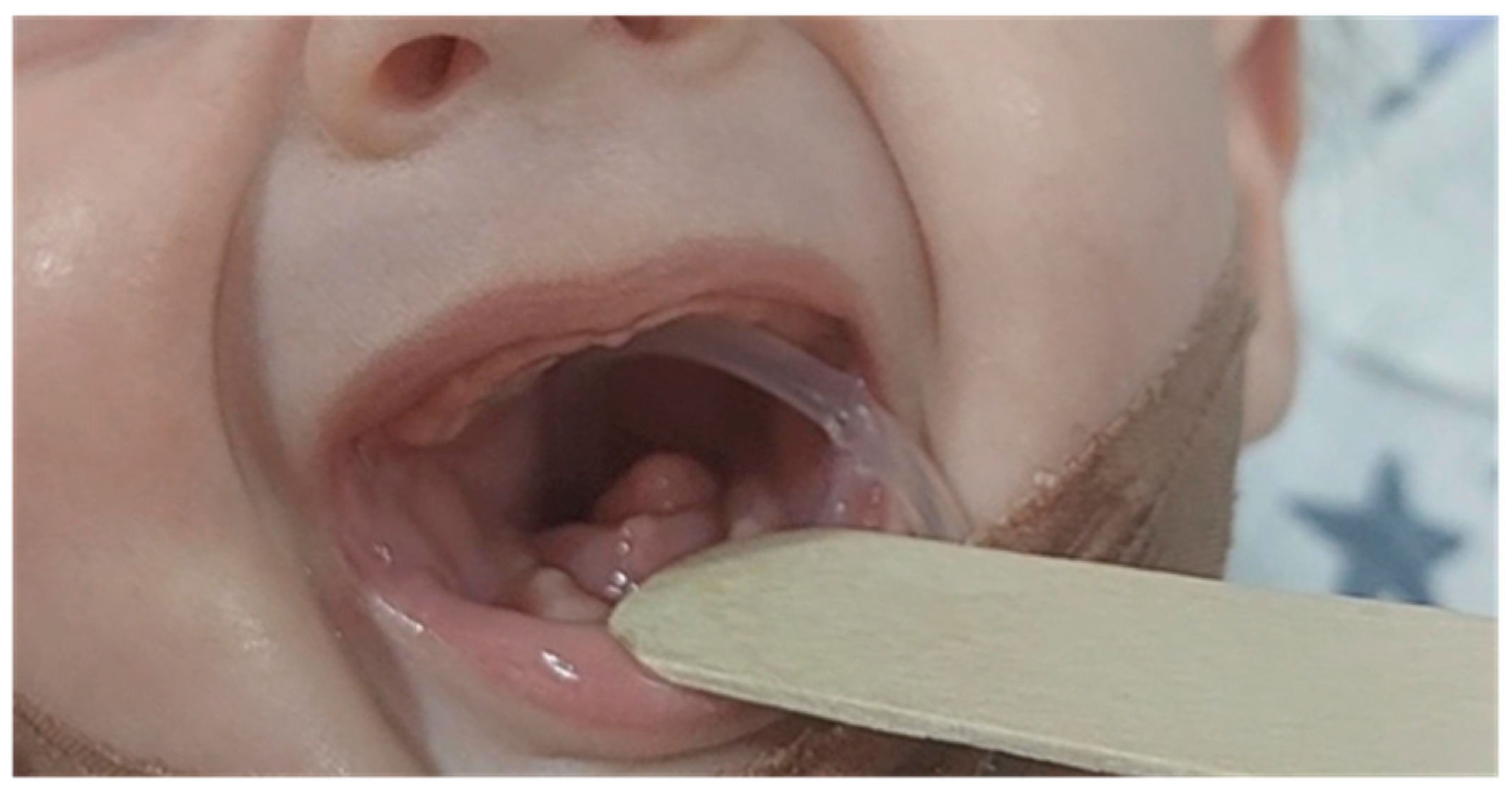
Mandibular hypoplasia, hypoglossia, and high-arched palate were evident upon delivery. There was a significant retrognathia and the anterior two-thirds of the tongue were missing. The posterior third of the tongue was present and it was bud-shaped and barely motile. This case is depicted in
Figure 1. Due to severity of these malformations, breastfeeding and bottle feeding were not possible. Nasogastric tube was inserted. The newborn was otherwise in good physical condition and she had no breathing problems. Apgar score was 9 at 1st, 5th and 10th minute respectively, and all vital signs were normal. Physical examination findings also included low-set ears, hypertelorism, broad nasal bridge, and prolonged and smooth philtrum. Ear canal and tympanic membrane appeared normal on both sides. Hands and feet were normal. There were no other musculoskeletal deformities. During auscultation, heartbeats were mainly heard on the right hemithorax. Chest X-ray and echocardiogram confirmed that there is mirror-image dextrocardia.
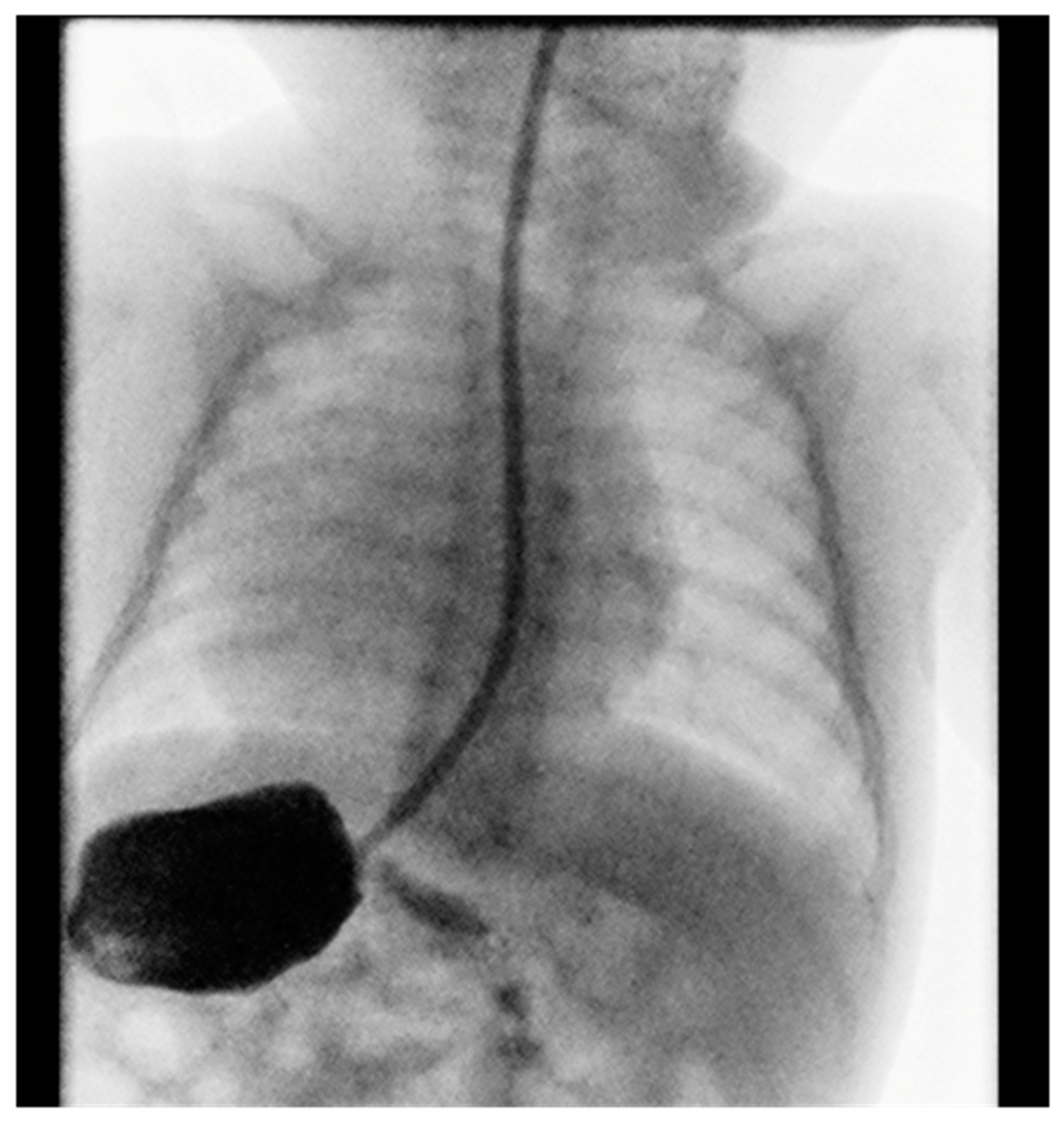
Contrast imaging of the esophagus and stomach shows a normal presentation of the esophagus with the stomach positioned on the right (
Figure 2).
Electrocardiogram showed sinus rhythm with right axis deviation. The pulse was 120 beats per minute. Abdominal ultrasound found liver in the left hemiabdomen, and spleen in the right, with all observable visceral organs showing normal morphology and dimensions.
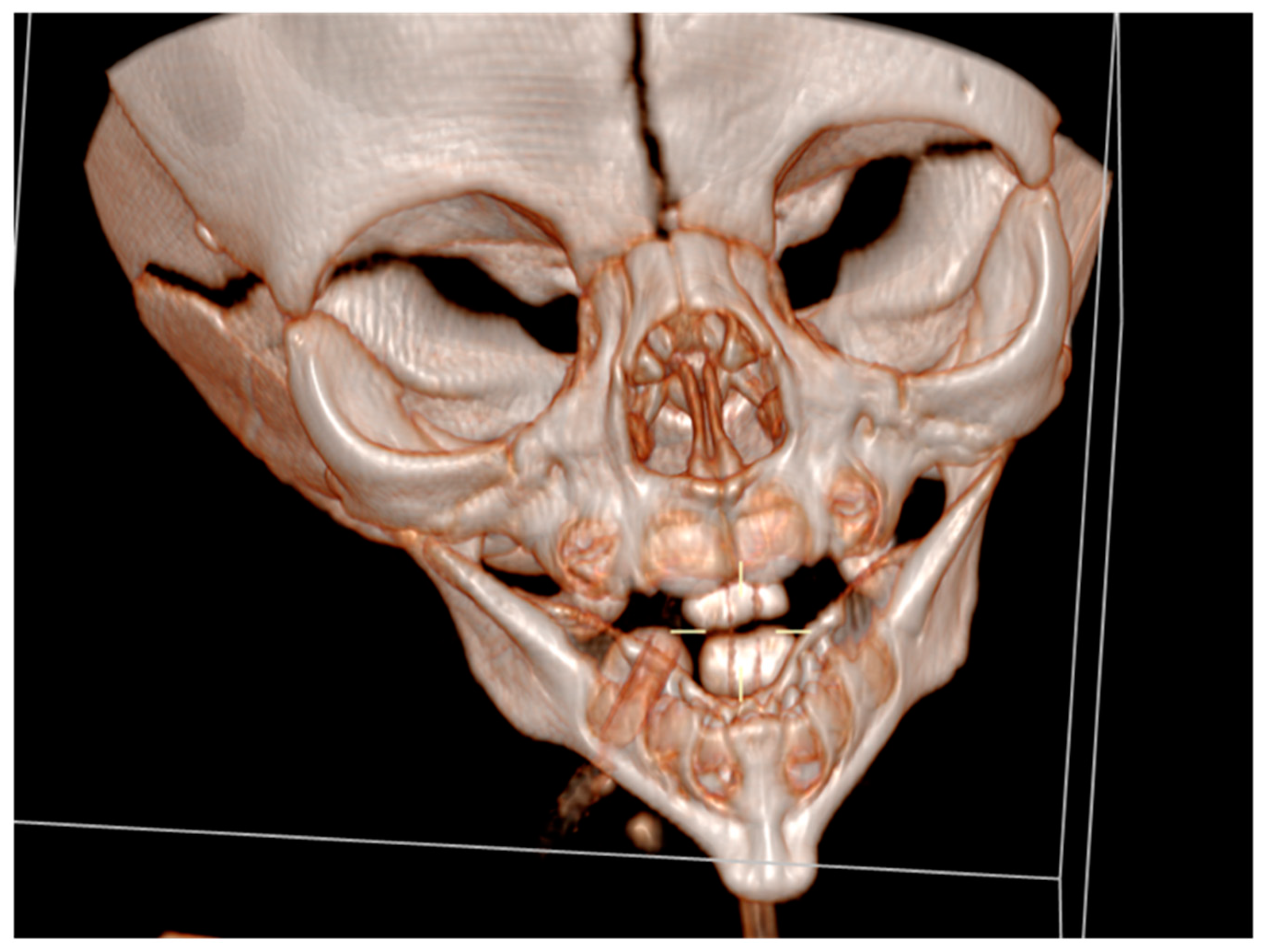
Cranial ultrasound showed no pathological findings, except for discretely hyperechoic periventricular white matter. A computed tomography of facial region described mandibula as hypoplastic and narrow (
Figure 3).
It had underdeveloped body region and pointed chin. Only three tooth germ of the lower frontal teeth was present, that was only one lower medial incisor and two lateral incisors. Temporomandibular joints were normal (
Figure 4).
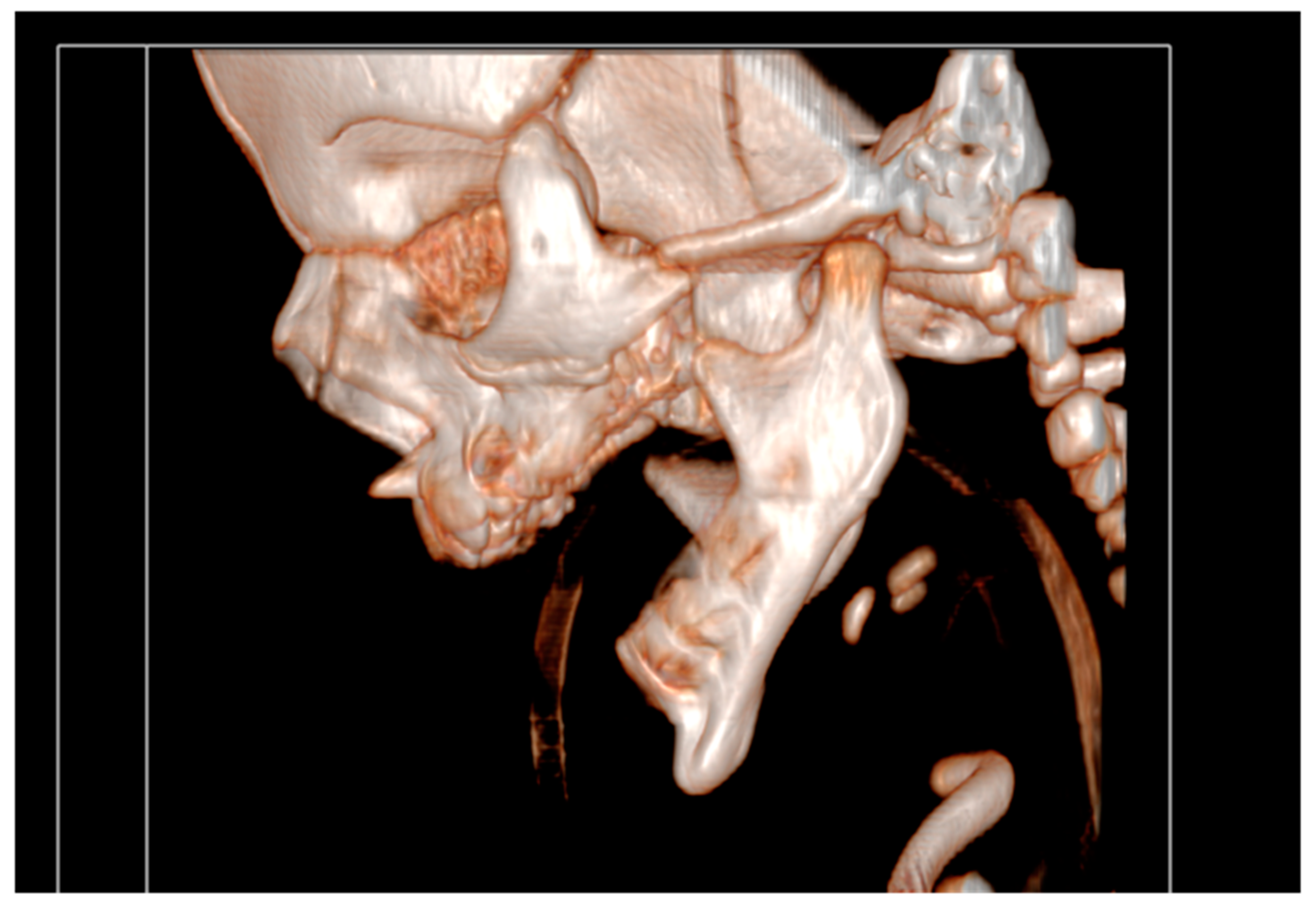
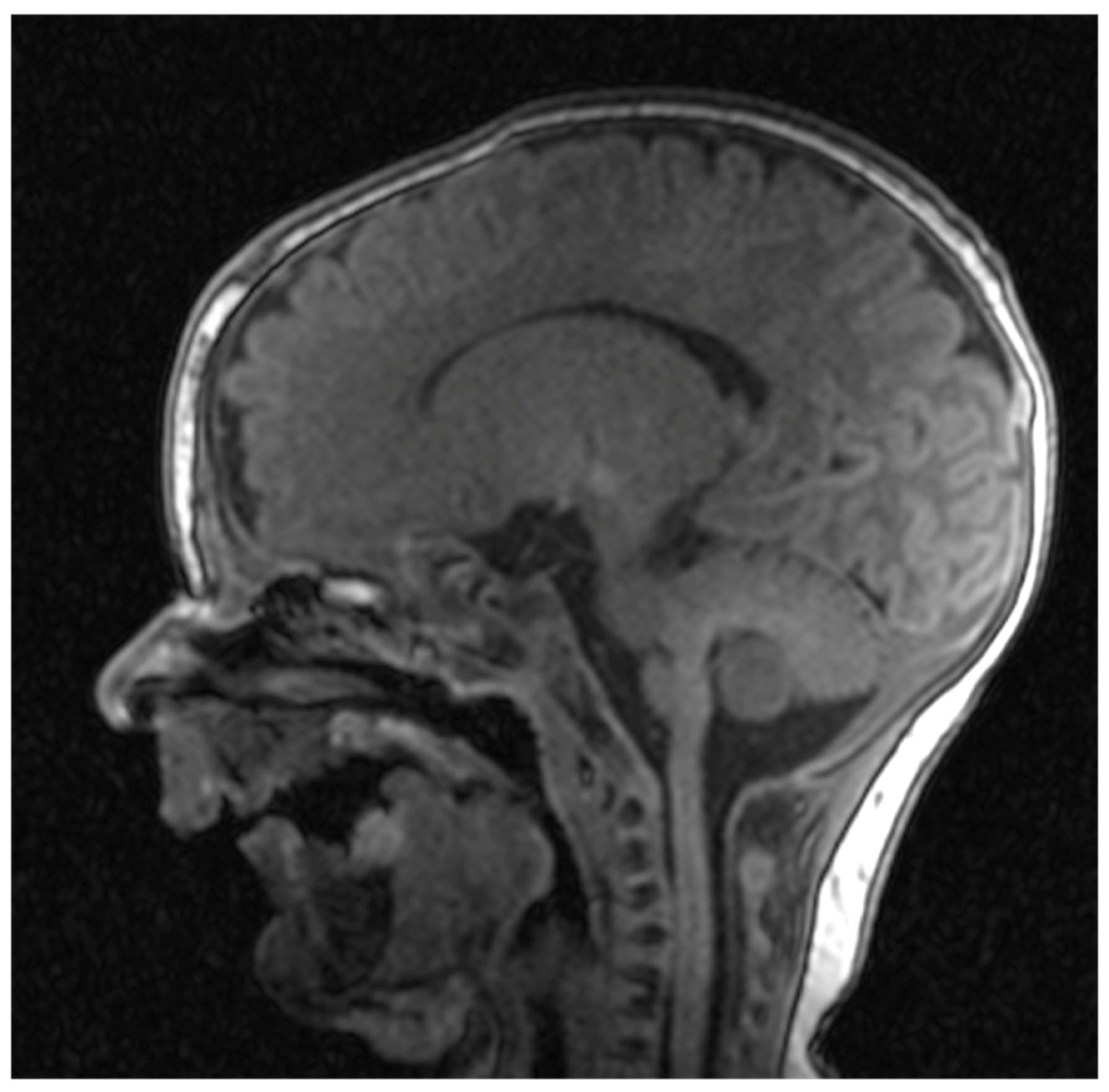
Cranial nuclear magnetic resonance showed no detectable pathological changes in brain structures and tissues. Brain ventricles and extracerebral cerebrospinal fluid spaces had physiological dimensions. Pronounced mycognathia and oral cavity without a developed tongue (
Figure 5).
Pituitary gland and sella turcica were described as normally developed for the age. Ophthalmological examination found no pathological changes in retina and papilla of the optic nerve of both eyes. Antibodies for TORCH infections IgG and all IgM were negative two days after birth.
The otolaryngological findings indicated a rudimentary tongue in the form of a bud with pronounced micrognathia. Examination of the larynx showed normal anatomy without associated anomalies. Hearing screening using transient evoked otoacoustic emission was normal.
Echocardiogram showed a mirror-image dextrocardia. Aorta was shown coming out of anatomical left ventricle, and pulmonary artery was coming out of anatomical right ventricle. Aortic arch was right-positioned, and inferior vena cava could not be clearly visualized. Hepatic veins were found to be flowing directly into the anatomical right atrium. There were no septal defects or shunting. All heart chambers and great vessels had normal dimensions and blood flow.
Gastroduodenal radiography with contrast showed stomach in the right hemiabdomen. Stomach and duodenum had normal structure and motility. There were no signs of hiatus hernia or gastroesophageal reflux, and there were no obstructions to the passage of contrast fluid.
Genetic analysis yielded a normal karyotype, with no microdeletions or microduplications of clinical significance. The pathological finding was not confirmed by genome analysis, exome sequencing.
Newborn primitive reflexes are present and physiological, except for the “rooting” reflex, which is not well coordinated. The child notices faces and makes eye contact. An 8-month-old infant shows proper psychomotor development. For a long time, she was fed through an orogastric tube with stimulation of the sucking reflex all the time. At the age of 8 months, milks was swallowed easily, feeding mushy food became possible but chewing is still difficult. With intensive speech therapy exercises, the child started to vocalize but does not produce syllables. Multidisciplinary following this child we will establish greater or lesser difficulties in talking and speaking.
4. Discussion
Hypoglossia and micrognathia can appear as isolated anomalies, as described by Klaphake et al [
3]. They presented a case of a female patient with hypoglossia, micrognathia, and an 18p deletion on one chromosome. More often, hypoplasia of tongue and mandible appear together with other congenital anomalies. In the case we described, the associated malformation was situs inversus totalis. This combination of anomalies is extremely rare. We found only few cases that had similar clinical presentation. Some of them had additional findings, like atrial bradycardia, patent ductus arteriosus, asplenia, polysplenia and sick sinus rhythm [
4,
5,
6,
7].
One of the cases even described that anterior part of the tongue was present, but not fused with the posterior part. These two parts were split by a smooth groove, and the posterior part created breathing problems when the child was in supine position [
8].
In the case presented by Chabrolle et al. pituitary gland was absent [
9]. Some of these cases described normal or mildly delayed mental and physical development. Others only confirmed that children were able to live with anomalies at the time of writing [
3,
8,
10,
11,
12,
13]. Only two cases ended fatally, due to respiratory distress and asphyxiation, respectfully [
9,
14].
It has been almost 100 years since the first case of hypoglossia, micrognathia and situs inversus was described in literature (Watkin, 1925) [
12]. There is still no proven cause of these anomalies. Some authors noted that mothers were exposed to fiberglass, nicotine, X-rays and antibiotics [
4,
5,
6,
9,
10]. All exposures happened during critical period of organogenesis. Still, none of them were proven to have an effect on development of these anomalies.This is exactly why it is important to present this case of ours when the mother had fever, nausea, vomiting and diarrhea during the first month of pregnancy. She recalls that it was the 4th week of pregnancy, which is a critical period of organogenesis. By testing a nasopharyngeal swab, the PCR test was positive for SARS-CoV-19. We have not found a case of these anomalies in the literature after the mother's Covid infection. She only used acetaminophen to treat the fever. This infection is the only plausible cause of deformations that’s left. Only one other case reported that mother had a fever during early pregnancy [
9]. Some authors suggested that de novo genetic mutations play a role in these malformations, since all parents were healthy with no previous family history of congenital illnesses [
15]. Some of the affected children had siblings, which were all unaffected and healthy [
7,
12,
14]. One mother had two previous miscarriages of unknown cause [
10]. However, children’s karyotypes were all normal, like in our case, except in one case where deletion of one 18p was detected [
3].
There have been attempts to classify congenital syndromes that share hypoglossia as a common trait. Such an attempt was made by Hall in 1971 [
16]. He proposed a five type classification of congenital syndromes that have hypoglossia in common. This set of syndromes is known as oromandibular limb hypogenesis syndrome. The spectrum of anomalies that these syndromes include is broad. Most notable are cleft lip and/or palate, micrognathia, hypodactylia, and peromelia. However, they do not include situs inversus.
The malformations present in our patient have only some clinical features close to otocephaly. Otocephaly is classified into four groups: 1. agnathia alone; 2. agnathia with holoprosencephaly; 3. agnathia with situs inversus and visceral anomalies; and 4. agnathia with holoprosencephaly, situs inversus, and visceral anomalies. However, hypoglossia was pronounced in our patient and holoprosencephaly was not seen.
Otocephaly can also vary in presence and intensity in individual patients. Even if the brain is not affected, death occurs due to pulmonary hypoplasia and severe breathing problems [
17]. There has been at least one case where the patient with a mild form of this syndrome lived to the age of 3. Unfortunately, the patient was not observed thereafter [
18]. At least four otocephaly cases are known to be caused by a mutation in the gene PRRX1 on the long arm of chromosome 1 [
17]. In our patient, genetic analyses did not confirm any pathological mutation.
Given that these three malformations, hypoglossia, micrognathia and situs inversus, with their phenotypic characteristics and clinical manifestations do not fully fit into the syndromes described so far. It is suspected that the cases of hypoglossia, micrognathia and situs inversus in this particular case described could be caused by Covid infection during pregnancy, because certainly the course of embryonic development was interrupted at an early stage.
The described rare cases of occurrence of these three anomalies together for an as yet unclear reason raises the question of whether they represent one entity or not. Given the orderly findings of the examined genome, the question remains open in which period and at what level should the error occur that leads to the emergence of these three anomalies at the same time.
5. Conclusions
Hypoglossia and micrognathia are rare congenital malformations with wide clinical spectrum. They can be associated with oral and facial malformations as well as anomalies of other organ systems. They most likely occur after the interruption of the normal embryonic development of the tongue and mandible. The causes of its occurrence are numerous, from genetic factors, congenital anomalies, but also harmful factors from the environment during embryonic development. Here we present an infant born with hypoglossia, micrognathia, and situs inversus as a very rare triadic combination without additional ear, musculoskeletal or brain deformities with normal genome findings. The only thing that has been proven is the exposure of the fetus to the SARS-CoV-19 virus in the first weeks of embryonic development.
This clinical presentation (micrognathia, hypoglossia, situs inversus) does not fit the description of existing syndromes that include similar anomalies such as otocephaly or oromandibular limb hypogenesis syndrome.
The possible influence of the SARS-CoV-19 virus on the occurrence of these anomalies and the exact mechanism of action should be confirmed in subsequent research.
Author Contributions
Conceptualization, G.V.-F. (Gordana Velisavljev-Filipovic); investigation, G.V.-F. (Gordana Velisavljev-Filipovic), O.J. (Ognjen Jovanov); resources, (Gordana Velisavljev-Filipovic), O.J.; writing—original draft preparation, G.V.-F. (Gordana Velisavljev-Filipovic), O.J.; writing—review and editing, G.V.-F. (Gordana Velisavljev-Filipovic), O.J.; supervision, G.V.-F. (Gordana Velisavljev-Filipovic). Both authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of INSTITUTE FOR CHILD AND YOUTH HEALTH CARE OF VOJVODINA (protocol code 263, date of approval: 17. January 2025).
Informed Consent Statement
Written informed consent has been obtained from the patient's parent's to publish this paper.
Data Availability Statement
The data presented can be provided on request.
Acknowledgments
There are no acknowledgements.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Meyer, U. (2021). Fundamentals of Craniofacial Malformations.
- Weis, F., & Degrandi. Situs inversus totalis. Anästhesiologie & Intensivmedizin 2021, 62. [Google Scholar]
- Klaphake, S. , van Dooren, M. F., Senden, R. E. M., Hoogeboom, A. J. M., Wolvius, E. B., & Koudstaal, M. J. A patient with chromosome 18p deletion and congenital hypoglossia. Clinical dysmorphology 2018, 27, 46–48. [Google Scholar] [CrossRef] [PubMed]
- McLaren, L. B. , Mactier, H., & Tolmie, J. Situs inversus totalis and congenital hypoglossia associated with atrial ectopic bradycardia and normal neurodevelopmental outcome. Clinical dysmorphology 2013, 22, 36–38. [Google Scholar] [CrossRef]
- Amor, D. J. , & Craig, J. E. Situs inversus totalis and congenital hypoglossia. Clinical dysmorphology 2001, 10, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Oulis, C. J. , & Thornton, J. B. Severe congenital hypoglossia and micrognathia with other multiple birth defects. Journal of oral pathology 1982, 11, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Faqeih, E. , Farra, H., & Al-Hassnan, Z. A further case of micrognathia, aglossia, and situs inversus totalis with additional features. Clinical dysmorphology 2008, 17, 219–220. [Google Scholar] [CrossRef] [PubMed]
- Tan, H. K. , Smith, J. D., & Goh, D. Y. Unfused hypoplastic tongue in a newborn. International journal of pediatric otorhinolaryngology 1999, 49, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Chabrolle, J. P. , Labenne, M., Cailliez, D., Poinsot, J., Bruel, H., & Vercoustre, L. Hypoglossie, situs inversus et absence d'hypophyse chez un nouveau-né: Effet tératogène d'une hyperthermie maternelle? Archives de Pédiatrie 1998, 5, 186–189. [Google Scholar]
- Ren, X. C. , Li, Y. F., Liu, Y., & Zhu, S. S. Mandibular symphyseal midline distraction osteogenesis for micrognathia associated with aglossia and situs inversus totalis. International journal of oral and maxillofacial surgery 2017, 46, 1346–1351. [Google Scholar] [CrossRef] [PubMed]
- Jang, G. Y. , Lee, K. C., Choung, J. T., Son, C. S., & Tockgo, Y. C. Congenital aglossia with situs inversus totalis--a case report. Journal of Korean medical science 1997, 12, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Watkin, H. G. Congenital absence of the tongue. The International Journal of Orthodontia, Oral Surgery, and Radiography 1925, 11, 941–943. [Google Scholar] [CrossRef]
- Hussels I., E. Microglossia, hypodontia, micrognathia, situs inversus. Birth defects original article series 1971, 7, 282. [Google Scholar] [PubMed]
- Dunham, M. E. , & Austin, T. L. Congenital aglossia and situs inversus. International journal of pediatric otorhinolaryngology 1990, 19, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Fabiani M, Libotte F, Margiotti K, Tannous DKI, Sparacino D, D'Aleo MP, Monaco F, Dello Russo C, Mesoraca A, Giorlandino C. Agnathia-Otocephaly Complex Due to a De Novo Deletion in the OTX2 Gene. Genes 2022, 13, 2269. [Google Scholar] [CrossRef] [PubMed]
- Hall B., D. Aglossia-adactylia. Birth defects original article series 1971, 7, 233–236. [Google Scholar]
- Dubucs, C. , Chassaing, N., Sergi, C., Aubert-Mucca, M., Attié-Bitach, T., Lacombe, D., Thauvin-Robinet, C., Arpin, S., Perez, M. J., Cabrol, C., Chen, C. P., Aziza, J., Colin, E., Martinovic, J., Calvas, P., & Plaisancié, J. Re-focusing on Agnathia-Otocephaly complex. Clinical oral investigations 2021, 25, 1353–1362. [Google Scholar] [CrossRef] [PubMed]
- Shermak, M. A. , & Dufresne, C. R. Nonlethal case of otocephaly and its implications for treatment. Journal of Craniofacial Surgery 1996, 7, 372–375. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).