Submitted:
22 March 2025
Posted:
25 March 2025
You are already at the latest version
Abstract
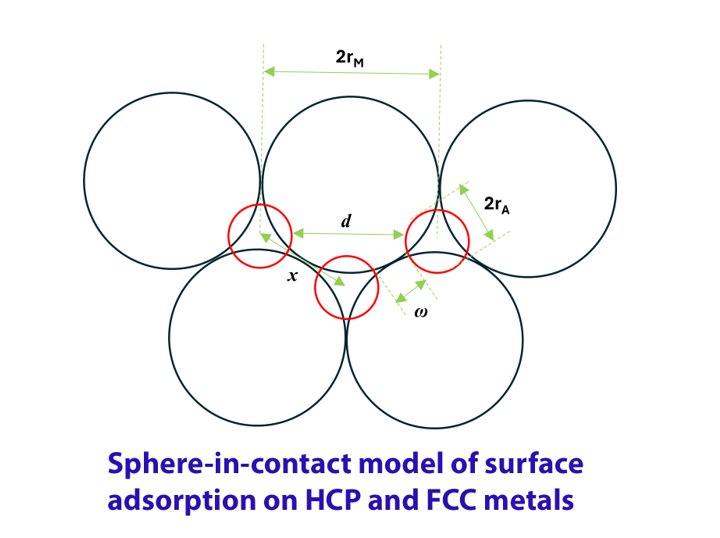
Keywords:
1. Introduction
2. Materials and Methods
3. Results and Discussion
Analytical Equations of the Repulsive Interaction Energy of Monoatomic Adsorbates in the Sphere-in-Contact Model
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ML | Monolayer |
| HCP | Hexagonal close packed |
| FCC | Face centered cubic |
| BCC | Body centered cubic |
| DFT | Density functional theory |
| APC | Article processing charge |
| STM | Scanning tunneling microscopy |
| AFM | Atomic force microscopy |
| LEED | Low energy electron diffraction |
| NP | Nanoparticle |
| CPK | Corey-Pauling-Koltun |
| VdW | Van der Waals |
References
- Wang, Z.; Hu, P. Rational catalyst design for CO oxidation: a gradient-based optimization strategy. Catalysis Science & Technology 2021, 11, 2604-2615. [CrossRef]
- Xu, H.; Cheng, D.; Cao, D.; Zeng, X.C. Revisiting the universal principle for the rational design of single-atom electrocatalysts. Nature Catalysis 2024, 7, 207-218. [CrossRef]
- Saini, S.; Halldin Stenlid, J.; Abild-Pedersen, F. Electronic structure factors and the importance of adsorbate effects in chemisorption on surface alloys. Computational Materials 2022, 8, 163. [CrossRef]
- Sprunger, P.T.; Plummer, E.W. The Interaction of Hydrogen with Simple Metal Surfaces. Surface Science 1994, 307-309, 118-123.
- Comer, B.M.; Li, J.; F., A.-P.; Bajdich, M.; Winther, K.T. Unraveling electronic trends in O* and OH* surface adsorption in the MO2 transition-metaloxide series. J. Phys. Chem. C 2022, 126, 7903.
- Raman, A.S.; Vojvodic, A. Energy Trends in Adsorption at Surfaces. In Handbook of Materials Modeling: Applications: Current and Emerging Materials, Andreoni, W., Yip, S., Eds.; Springer International Publishing: Cham, 2020; pp. 1321-1341.
- Zeinalipour-Yazdi, C.D.L., E. Z. Study of the cap structure of (3,3), (4,4) and (5,5)-SWCNTs: Application of the sphere-in-contact model. Carbon 2017, 115, 819-827.
- Extremera, J.; Vergara, D.; Rodríguez, S.; Dávila, L.P. Reality-Virtuality Technologies in the Field of Materials Science and Engineering. Applied Sciences 2022, 12. [CrossRef]
- Zeinalipour-Yazdi, C.D.; Pullman, D.P.; Catlow, C.R.A. The sphere-in-contact model of carbon materials. Journal of Molecular Modeling 2016, 22, 40. [CrossRef]
- Corey, R.B.; Pauling, L. Molecular models of amino acids, peptides, and proteins. Rev. Sci. Instr. 1953, 24, 621.
- Papadopoulou, A.; Laucks, J.; Tibbits, S. From Self-Assembly to Evolutionary Structures. Architectural Design 2017, 87, 28-37. [CrossRef]
- Pham, T.A.; Song, F.; Nguyen, M.-T.; Stöhr, M. Self-assembly of pyrene derivatives on Au(111): substituent effects on intermolecular interactions. Chemical Communications 2014, 50, 14089-14092. [CrossRef]
- Bezryadin, A.; Westervelt, R.M.; Tinkham, M. Self-assembled chains of graphitized carbon nanoparticles. Applied Physics Letters 1999, 74, 2699-2701. [CrossRef]
- Hubmann, A.H.; Dietz, D.; Brötz, J.; Klein, A. Interface Behaviour and Work Function Modification of Self-Assembled Monolayers on Sn-Doped In2O3. Surfaces 2019, 2, 241-256. [CrossRef]
- Kosmala, T.; Blanco, M.; Granozzi, G.; Wandelt, K. Potential Driven Non-Reactive Phase Transitions of Ordered Porphyrin Molecules on Iodine-Modified Au(100): An Electrochemical Scanning Tunneling Microscopy (EC-STM) Study. Surfaces 2018, 1, 12-28. [CrossRef]
- Zeinalipour-Yazdi, C.D.; Loizidou, E.Z. Corrigendum to “Study of the cap structure of (3,3), (4,4) and (5,5)-SWCNTs: Application of the sphere-in-contact model” [Carbon 115 (2017) 819–827]. Carbon 2019, 146, 369-370.
- Zeinalipour-Yazdi, C.D.; Pullman, D.P. Study of a rhombohedral graphite X-ray filter using the sphere-in-contact model. Chemical Physics Letters 2019, 734, 136717. [CrossRef]
- Zeinalipour-Yazdi, C.D.; Pullman, D.P. Miniature physical sphere-in-contact models of heterogeneous catalysts and metal nanoparticles. Journal of Molecular Modeling 2023, 29, 312. [CrossRef]
- Zeinalipour-Yazdi, C.D. A study using physical sphere-in-contact models to investigate the structure of close-packed nanoparticles supported on flat hexagonal, square and trigonal lattices. Chemical Physics 2024, 588, 112464. [CrossRef]
- Zeinalipour-Yazdi, C.D.; Willock, D.J.; Thomas, L.; Wilson, K.; Lee, A.F. CO adsorption over Pd nanoparticles: A general framework for IR simulations on nanoparticles. Surface Science 2016, 646, 210-220.
- Zeinalipour-Yazdi, C.D.; Cooksy, A.L.; Efstathiou, A.M. CO adsorption on transition metal clusters: Trends from density functional theory. Surface Science 2008, 602, 1858-1862. [CrossRef]

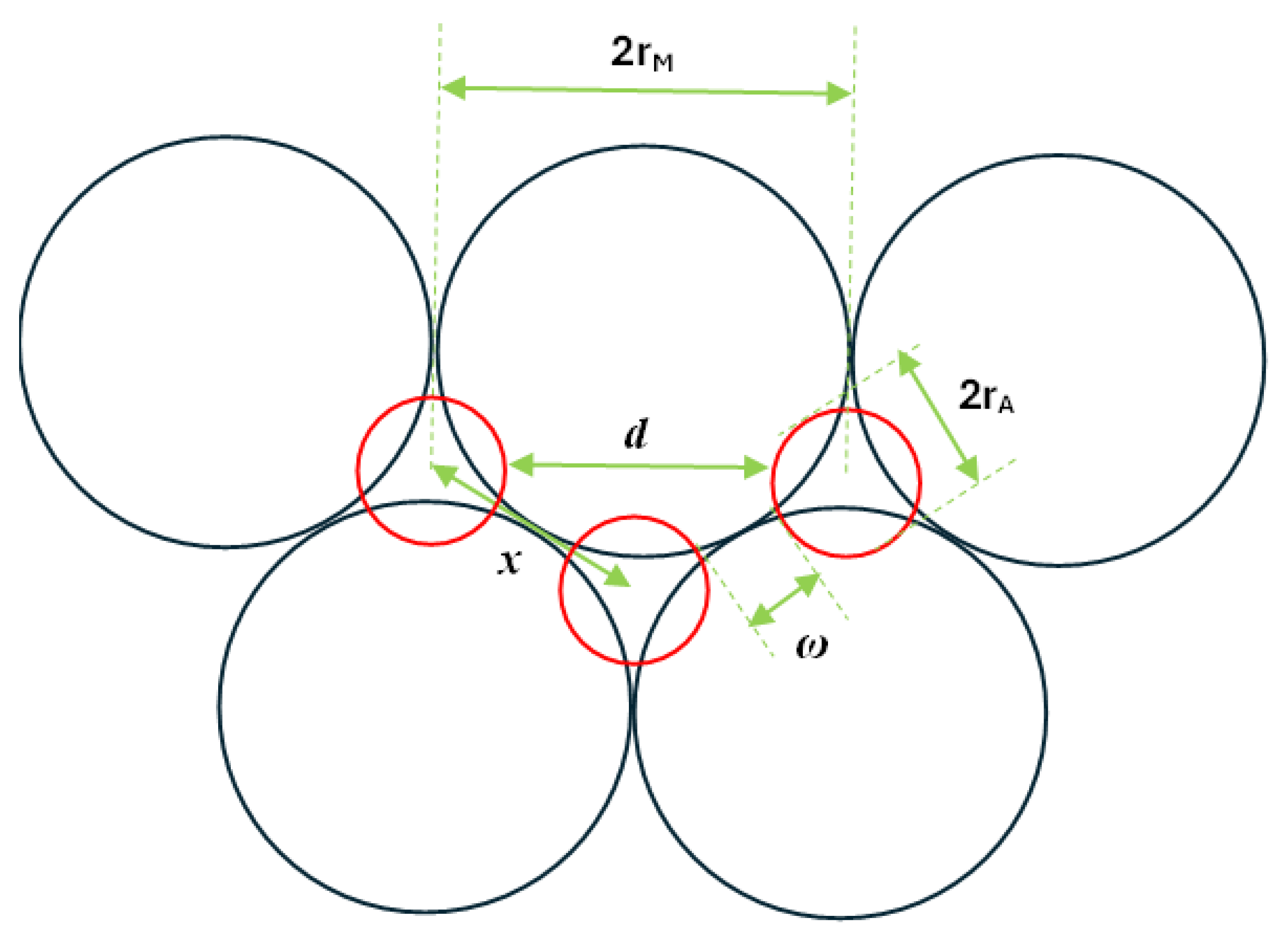
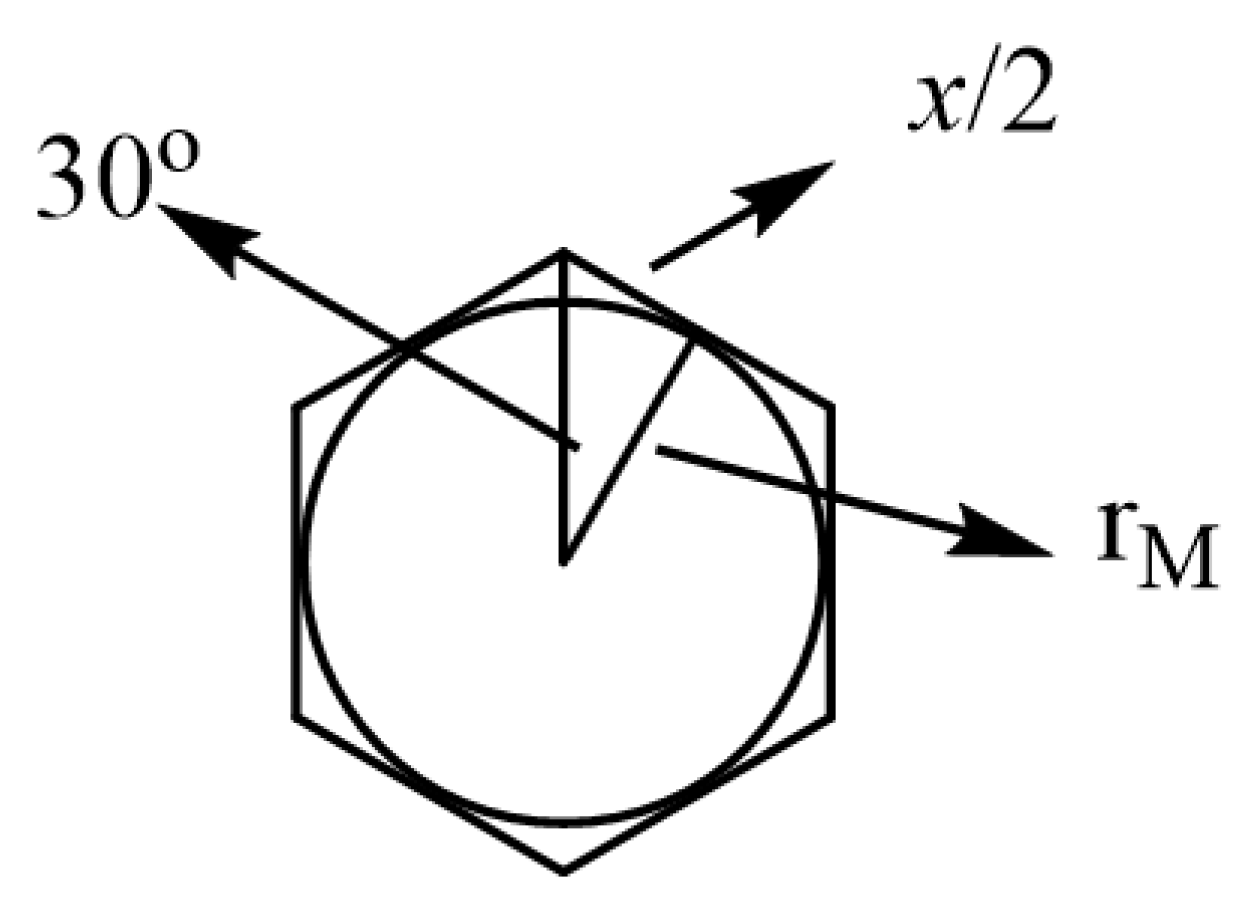
| Surface number | v2 | v3 | Unet (kJ mol-1) δ = ±0.1 e | Unet (kJ mol-1) δ = ±0.2 e |
|---|---|---|---|---|
| 1 | 24 | 4 | 25 | 100 |
| 2 | 26 | 0 | 25 | 100 |
| 3 | 53 | 0 | 51 | 205 |
| 4 | 10 | 17 | 17 | 69 |
| 5 | 16 | 15 | 22 | 89 |
| 6 | 26 | 12 | 30 | 122 |
| 7 | 8 | 25 | 19 | 76 |
| 8 | 26 | 0 | 25 | 100 |
| 9 | 0 | 35 | 16 | 63 |
| 10 | 12 | 22 | 21 | 86 |
| 11 | 26 | 0 | 25 | 100 |
| 12 | 27 | 11 | 31 | 124 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




