Submitted:
20 March 2025
Posted:
21 March 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. The Electronic Domain of a Cell
2.1. The Mitochondrion Can Be Imagined as a Tesla-Type Battery Supply
2.2. The Cellular Response Times Are Shorter Than Imagined
3. Electron Current Is Crucial for the Nucleus's Physiology
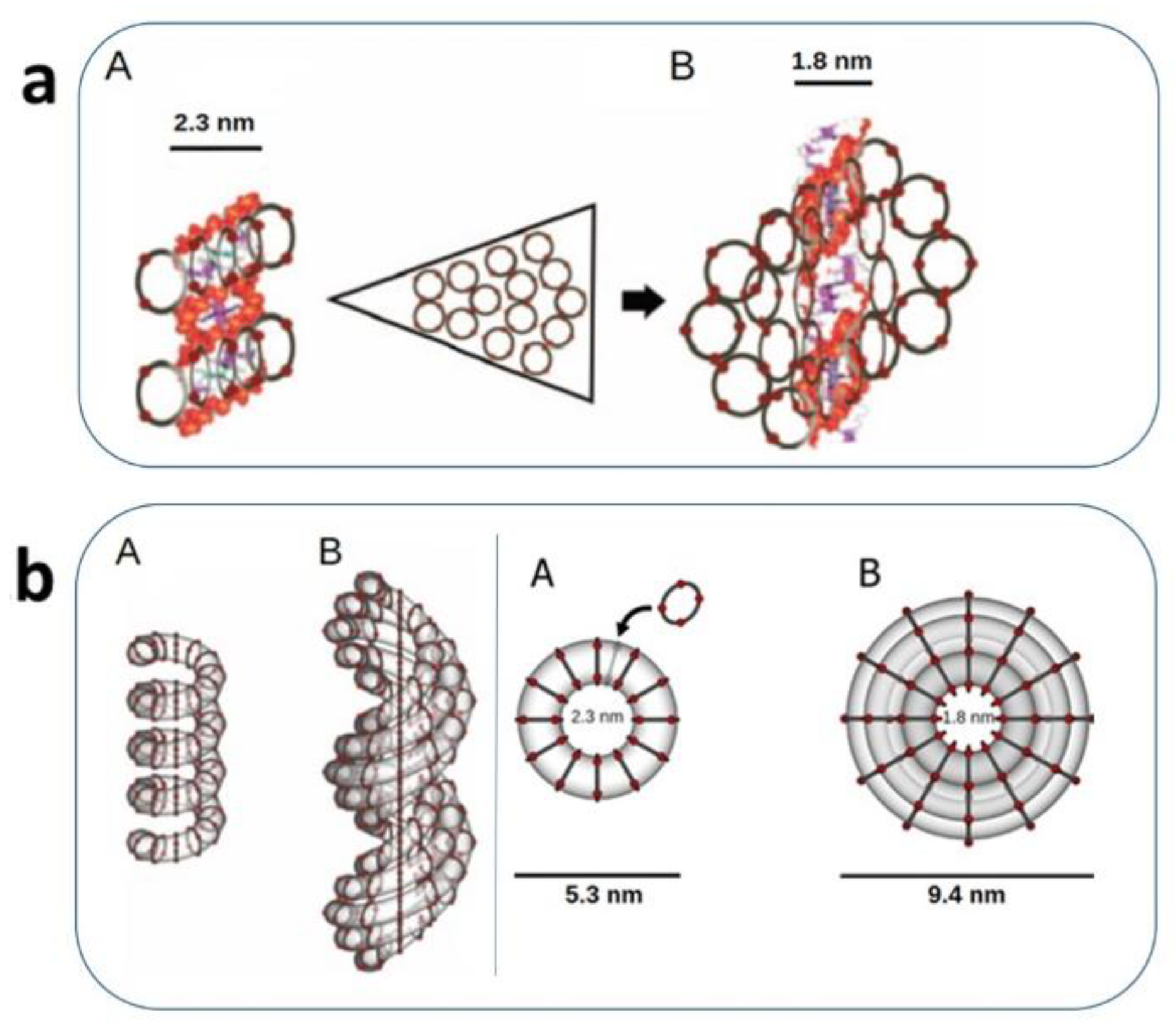
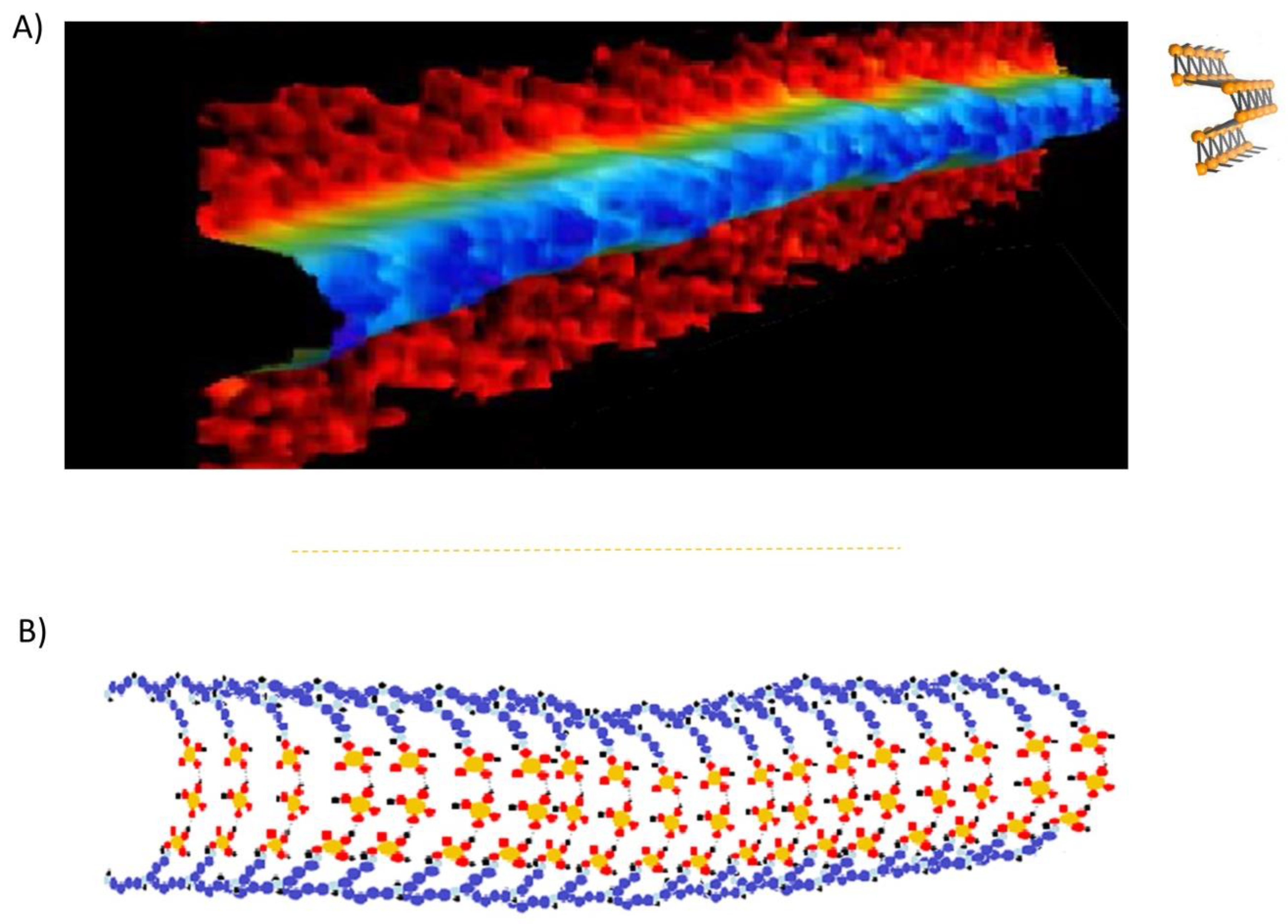
4. The Nuclear Aggregates of Polyamines
4.1. Nuclear Aggregates of Polyamines as Possible Semiconductors
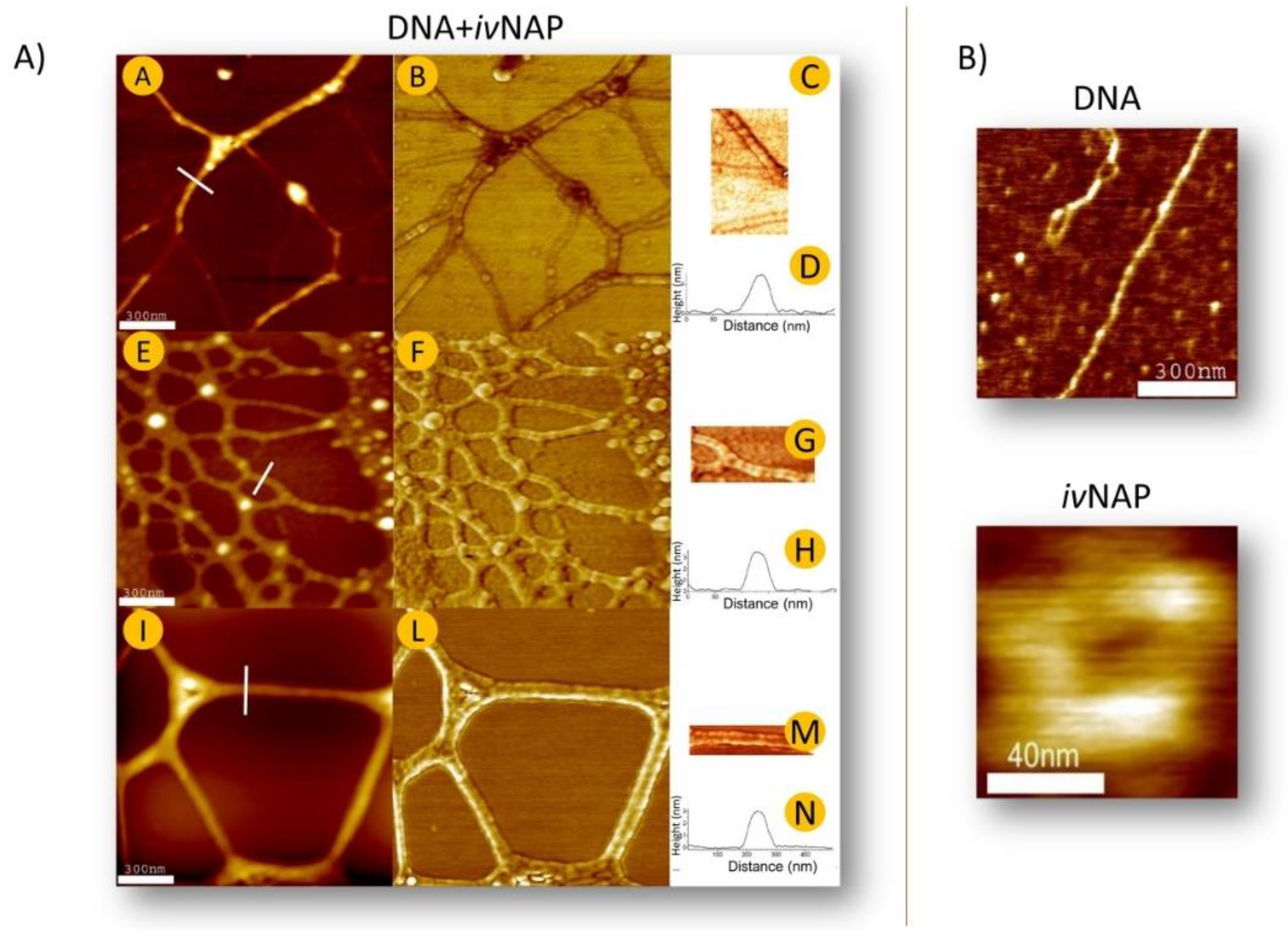
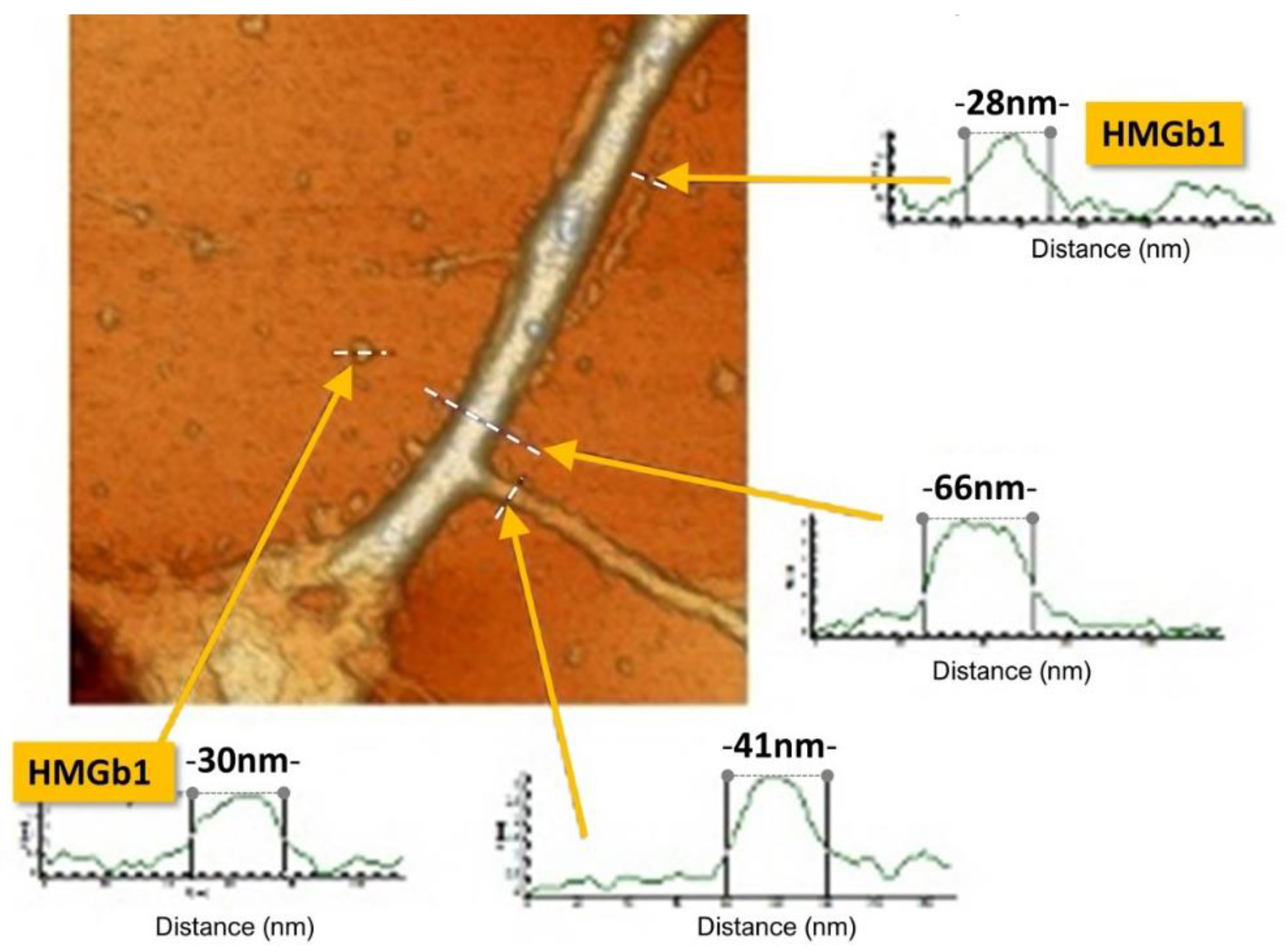
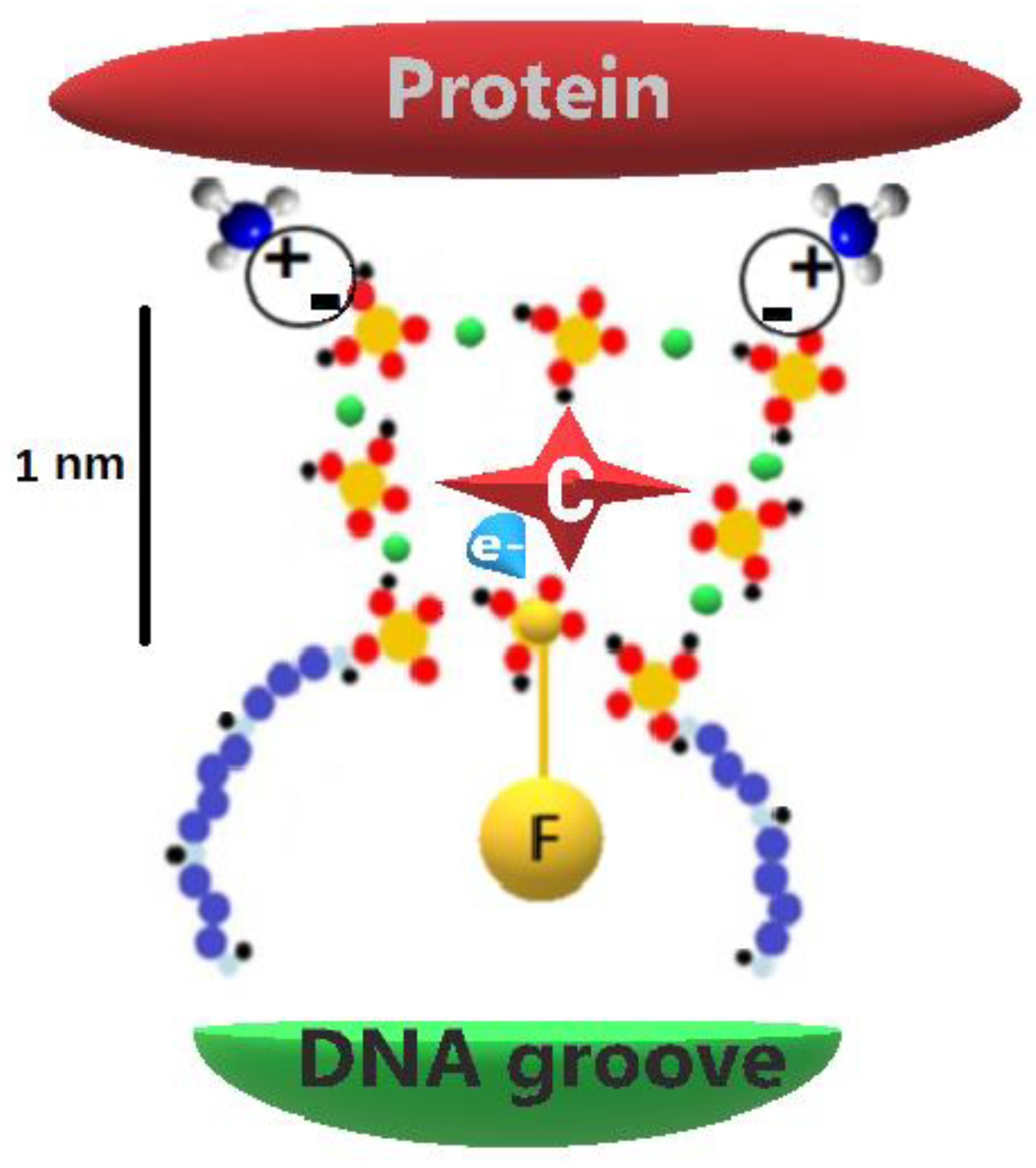
4.2. Nuclear Aggregates of Polyamines and nuclear proteins
4.3. Role of Phosphates in electronic DNA conductivity
5. Conclusions
Abbreviations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Teo, J.J.Y.; Sarpeshkar, R. The Merging of Biological and Electronic Circuits. iScience 2020, 23, 101688. [Google Scholar] [CrossRef] [PubMed]
- Selberg, J.; Gomez, M.; Rolandi, M. The Potential for Convergence between Synthetic Biology and Bioelectronics. Cell Syst 2018, 7, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Schofield, Z.; Meloni, G.N.; Tran, P.; Zerfass, C.; Sena, G.; Hayashi, Y.; Grant, M.; Contera, S.A.; Minteer, S.D.; Kim, M.; et al. Bioelectrical understanding and engineering of cell biology. J R Soc Interface 2020, 17, 20200013. [Google Scholar] [CrossRef]
- Zeng, J.; Teo, J.; Banerjee, A.; Chapman, T.W.; Kim, J.; Sarpeshkar, R. A Synthetic Microbial Operational Amplifier. ACS Synth Biol 2018, 7, 2007–2013. [Google Scholar] [CrossRef]
- Marais, A.; Adams, B.; Ringsmuth, A.K.; Ferretti, M.; Gruber, J.M.; Hendrikx, R.; Schuld, M.; Smith, S.L.; Sinayskiy, I.; Kruger, T.P.J.; et al. The future of quantum biology. J R Soc Interface 2018, 15. [Google Scholar] [CrossRef]
- Al-Khalili, J.; Lilliu, S. Quantum Biology. Scientific Video Protocols 2020, 1, 1. [Google Scholar] [CrossRef]
- Bendandi, A.; Dante, S.; Zia, S.R.; Diaspro, A.; Rocchia, W. Chromatin Compaction Multiscale Modeling: A Complex Synergy Between Theory, Simulation, and Experiment. Frontiers in Molecular Biosciences 2020, 7. [Google Scholar] [CrossRef]
- Zimatore, G.; Tsuchiya, M.; Hashimoto, M.; Kasperski, A.; Giuliani, A. Self-organization of whole-gene expression through coordinated chromatin structural transition. Biophysics Reviews 2021, 2, 031303. [Google Scholar] [CrossRef] [PubMed]
- D'Agostino, L. Native DNA electronics: is it a matter of nanoscale assembly? Nanoscale 2018, 10, 12268–12275. [Google Scholar] [CrossRef]
- D'Agostino, L.; Di Luccia, A. Polyamines interact with DNA as molecular aggregates. European journal of biochemistry / FEBS 2002, 269, 4317–4325. [Google Scholar] [CrossRef]
- Di Luccia, A.; Picariello, G.; Iacomino, G.; Formisano, A.; Paduano, L.; D'Agostino, L. The in vitro nuclear aggregates of polyamines. The FEBS journal 2009, 276, 2324–2335. [Google Scholar] [CrossRef]
- D'Agostino, L.; di Pietro, M.; Di Luccia, A. Nuclear aggregates of polyamines are supramolecular structures that play a crucial role in genomic DNA protection and conformation. The FEBS journal 2005, 272, 3777–3787. [Google Scholar] [CrossRef] [PubMed]
- D'Agostino, L.; di Pietro, M.; Di Luccia, A. Nuclear aggregates of polyamines. IUBMB Life 2006, 58, 75–82. [Google Scholar] [CrossRef]
- Iacomino, G.; Picariello, G.; D'Agostino, L. DNA and nuclear aggregates of polyamines. Biochimica et biophysica acta 2012, 1823, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Iacomino, G.; Picariello, G.; Sbrana, F.; Di Luccia, A.; Raiteri, R.; D'Agostino, L. DNA is wrapped by the nuclear aggregates of polyamines: the imaging evidence. Biomacromolecules 2011, 12, 1178–1186. [Google Scholar] [CrossRef] [PubMed]
- Iacomino, G.; Picariello, G.; Sbrana, F.; Raiteri, R.; D'Agostino, L. DNA-HMGB1 interaction: The nuclear aggregates of polyamine mediation. Biochimica et biophysica acta 2016, 1864, 1402–1410. [Google Scholar] [CrossRef]
- Iacomino, G.; Picariello, G.; Stillitano, I.; D'Agostino, L. Nuclear aggregates of polyamines in a radiation-induced DNA damage model. The international journal of biochemistry & cell biology 2014, 47, 11–19. [Google Scholar] [CrossRef]
- Picariello, G.; Iacomino, G.; D'Agostino, L. Phosphate-Induced Polyamine Self-Assembly. In Encyclopedia of Biomedical Polymers and Polymeric Biomaterials, Mishra, M., Ed.; Taylor & Francis: New York, 2015; Volume 8, pp. 5951–5964. [Google Scholar]
- Picariello, G.; Iacomino, G.; Di Luccia, A.; D'Agostino, L. Mass spectrometric analysis of in vitro nuclear aggregates of polyamines. Rapid communications in mass spectrometry : RCM 2014, 28, 499–504. [Google Scholar] [CrossRef]
- Li, J.; Beuerman, R.; Verma, C.S. Mechanism of polyamine induced colistin resistance through electrostatic networks on bacterial outer membranes. Biochim Biophys Acta Biomembr 2020, 1862, 183297. [Google Scholar] [CrossRef]
- Lutz, K.; Groger, C.; Sumper, M.; Brunner, E. Biomimetic silica formation: analysis of the phosphate-induced self-assembly of polyamines. Phys Chem Chem Phys 2005, 7, 2812–2815. [Google Scholar] [CrossRef]
- Lomozik, L.; Gasowska, A.; Bregier-Jarzebowska, R.; Jastrzab, R. Coordination chemistry of polyamines and their interactions in ternary systems including metal ions, nucleosides and nucleotides. Coordination Chemistry Reviews 2005, 249, 2335–2350. [Google Scholar] [CrossRef]
- Marmisollé, W.A.; Irigoyen, J.; Gregurec, D.; Moya, S.; Azzaroni, O. Supramolecular Surface Chemistry: Substrate-Independent, Phosphate-Driven Growth of Polyamine-Based Multifunctional Thin Films. Advanced Functional Materials 2015, 25, 4144–4152. [Google Scholar] [CrossRef]
- Cuenca, V.E.; Martinelli, H.; Ramirez, M.d.l.A.; Ritacco, H.A.; Andreozzi, P.; Moya, S.E. Polyphosphate Poly(amine) Nanoparticles: Self-Assembly, Thermodynamics, and Stability Studies. Langmuir : the ACS journal of surfaces and colloids 2019, 35, 14300–14309. [Google Scholar] [CrossRef]
- Eckert, H.; Montagna, M.; Dianat, A.; Gutierrez, R.; Bobeth, M.; Cuniberti, G. Exploring the organic–inorganic interface in biosilica: atomistic modeling of polyamine and silica precursors aggregation behavior. BMC Materials 2020, 2, 6. [Google Scholar] [CrossRef]
- Laucirica, G.; Marmisollé, W.A.; Azzaroni, O. Dangerous liaisons: anion-induced protonation in phosphate–polyamine interactions and their implications for the charge states of biologically relevant surfaces. Physical Chemistry Chemical Physics 2017, 19, 8612–8620. [Google Scholar] [CrossRef] [PubMed]
- Andreozzi, P.; Simó, C.; Moretti, P.; Porcel, J.M.; Lüdtke, T.U.; Ramirez, M.d.l.A.; Tamberi, L.; Marradi, M.; Amenitsch, H.; Llop, J.; et al. Novel Core–Shell Polyamine Phosphate Nanoparticles Self-Assembled from PEGylated Poly(allylamine hydrochloride) with Low Toxicity and Increased In Vivo Circulation Time. Small 2021, 17, 2102211. [Google Scholar] [CrossRef]
- Berlin, Y.A.; Voityuk, A.A.; Ratner, M.A. DNA base pair stacks with high electric conductance: a systematic structural search. ACS nano 2012, 6, 8216–8225. [Google Scholar] [CrossRef]
- Liu, C.; Xiang, L.; Zhang, Y.; Zhang, P.; Beratan, D.N.; Li, Y.; Tao, N. Engineering nanometre-scale coherence in soft matter. Nat Chem 2016, 8, 941–945. [Google Scholar] [CrossRef]
- Genereux, J.C.; Barton, J.K. Mechanisms for DNA charge transport. Chemical reviews 2010, 110, 1642–1662. [Google Scholar] [CrossRef]
- Jimenez-Monroy, K.L.; Renaud, N.; Drijkoningen, J.; Cortens, D.; Schouteden, K.; van Haesendonck, C.; Guedens, W.J.; Manca, J.V.; Siebbeles, L.D.; Grozema, F.C.; et al. High Electronic Conductance through Double-Helix DNA Molecules with Fullerene Anchoring Groups. The journal of physical chemistry. A 2017, 121, 1182–1188. [Google Scholar] [CrossRef]
- Xu; Zhang; Li; Tao. Direct Conductance Measurement of Single DNA Molecules in Aqueous Solution. Nano Letters 2004, 4, 1105–1108. [Google Scholar] [CrossRef]
- Zhuravel, R.; Huang, H.; Polycarpou, G.; Polydorides, S.; Motamarri, P.; Katrivas, L.; Rotem, D.; Sperling, J.; Zotti, L.A.; Kotlyar, A.B.; et al. Backbone charge transport in double-stranded DNA. Nature nanotechnology 2020, 15, 836–840. [Google Scholar] [CrossRef]
- Bhaskaran, R.; Sarma, M. Low-Energy Electron Interaction with the Phosphate Group in DNA Molecule and the Characteristics of Single-Strand Break Pathways. The journal of physical chemistry. A 2015, 119, 10130–10136. [Google Scholar] [CrossRef] [PubMed]
- Sayer, M.; Mansingh, A. Transport Properties of Semiconducting Phosphate Glasses. Physical Review B 1972, 6, 4629–4643. [Google Scholar] [CrossRef]
- Gomes, P.J.; Coelho, M.; Dionísio, M.; Ribeiro, P.A.; Raposo, M. Probing radiation damage by alternated current conductivity as a method to characterize electron hopping conduction in DNA molecules. Applied Physics Letters 2012, 101, 123702. [Google Scholar] [CrossRef]
- Stevens, C.F. Neurophysiology: a Primer; John Wiley & Sons, Inc.: New York, 1966. [Google Scholar]
- Sarpeshkar, R. Ultra low power bioelectronics : fundamentals, biomedical applications, and bio-inspired systems; Cambridge University Press: Cambridge, UK ; New York, 2010; p. xviii, 889 p. [Google Scholar]
- Voet, D.; Voet, J.G.; Pratt, C.W. Fundamentals of biochemistry : life at the molecular level, 2nd ed.; Wiley: Hoboken, N.J, 2006. [Google Scholar]
- Boyer, P.D. The ATP synthase--a splendid molecular machine. Annual review of biochemistry 1997, 66, 717–749. [Google Scholar] [CrossRef]
- Lodish, H.F. Molecular cell biology, 4th ed.; W.H. Freeman: New York, 2000. [Google Scholar]
- Pokorný, J. Physical aspects of biological activity and cancer. AIP Advances 2012, 2, 011207. [Google Scholar] [CrossRef]
- Tyner, K.M.; Kopelman, R.; Philbert, M.A. "Nanosized voltmeter" enables cellular-wide electric field mapping. Biophysical journal 2007, 93, 1163–1174. [Google Scholar] [CrossRef]
- Cunningham, J.; Estrella, V.; Lloyd, M.; Gillies, R.; Frieden, B.R.; Gatenby, R. Intracellular electric field and pH optimize protein localization and movement. PloS one 2012, 7, e36894. [Google Scholar] [CrossRef]
- Giorgio, V.; von Stockum, S.; Antoniel, M.; Fabbro, A.; Fogolari, F.; Forte, M.; Glick, G.D.; Petronilli, V.; Zoratti, M.; Szabo, I.; et al. Dimers of mitochondrial ATP synthase form the permeability transition pore. Proceedings of the National Academy of Sciences of the United States of America 2013, 110, 5887–5892. [Google Scholar] [CrossRef]
- Halestrap, A.P.; Richardson, A.P. The mitochondrial permeability transition: a current perspective on its identity and role in ischaemia/reperfusion injury. Journal of molecular and cellular cardiology 2015, 78, 129–141. [Google Scholar] [CrossRef] [PubMed]
- O'Reilly, C.M.; Fogarty, K.E.; Drummond, R.M.; Tuft, R.A.; Walsh, J.V., Jr. Quantitative analysis of spontaneous mitochondrial depolarizations. Biophysical journal 2003, 85, 3350–3357. [Google Scholar] [CrossRef]
- Wolf, D.M.; Segawa, M.; Kondadi, A.K.; Anand, R.; Bailey, S.T.; Reichert, A.S.; van der Bliek, A.M.; Shackelford, D.B.; Liesa, M.; Shirihai, O.S. Individual cristae within the same mitochondrion display different membrane potentials and are functionally independent. The EMBO journal 2019, 38, e101056. [Google Scholar] [CrossRef]
- Harner, M.; Korner, C.; Walther, D.; Mokranjac, D.; Kaesmacher, J.; Welsch, U.; Griffith, J.; Mann, M.; Reggiori, F.; Neupert, W. The mitochondrial contact site complex, a determinant of mitochondrial architecture. The EMBO journal 2011, 30, 4356–4370. [Google Scholar] [CrossRef]
- Rabl, R.; Soubannier, V.; Scholz, R.; Vogel, F.; Mendl, N.; Vasiljev-Neumeyer, A.; Korner, C.; Jagasia, R.; Keil, T.; Baumeister, W.; et al. Formation of cristae and crista junctions in mitochondria depends on antagonism between Fcj1 and Su e/g. The Journal of cell biology 2009, 185, 1047–1063. [Google Scholar] [CrossRef] [PubMed]
- Hoppins, S.; Collins, S.R.; Cassidy-Stone, A.; Hummel, E.; Devay, R.M.; Lackner, L.L.; Westermann, B.; Schuldiner, M.; Weissman, J.S.; Nunnari, J. A mitochondrial-focused genetic interaction map reveals a scaffold-like complex required for inner membrane organization in mitochondria. The Journal of cell biology 2011, 195, 323–340. [Google Scholar] [CrossRef] [PubMed]
- von der Malsburg, K.; Muller, J.M.; Bohnert, M.; Oeljeklaus, S.; Kwiatkowska, P.; Becker, T.; Loniewska-Lwowska, A.; Wiese, S.; Rao, S.; Milenkovic, D.; et al. Dual role of mitofilin in mitochondrial membrane organization and protein biogenesis. Developmental cell 2011, 21, 694–707. [Google Scholar] [CrossRef]
- Barbot, M.; Jans, D.C.; Schulz, C.; Denkert, N.; Kroppen, B.; Hoppert, M.; Jakobs, S.; Meinecke, M. Mic10 oligomerizes to bend mitochondrial inner membranes at cristae junctions. Cell metabolism 2015, 21, 756–763. [Google Scholar] [CrossRef]
- Friedman, J.R.; Mourier, A.; Yamada, J.; McCaffery, J.M.; Nunnari, J. MICOS coordinates with respiratory complexes and lipids to establish mitochondrial inner membrane architecture. eLife 2015, 4. [Google Scholar] [CrossRef]
- Guarani, V.; McNeill, E.M.; Paulo, J.A.; Huttlin, E.L.; Frohlich, F.; Gygi, S.P.; Van Vactor, D.; Harper, J.W. QIL1 is a novel mitochondrial protein required for MICOS complex stability and cristae morphology. eLife 2015, 4. [Google Scholar] [CrossRef]
- Hessenberger, M.; Zerbes, R.M.; Rampelt, H.; Kunz, S.; Xavier, A.H.; Purfurst, B.; Lilie, H.; Pfanner, N.; van der Laan, M.; Daumke, O. Regulated membrane remodeling by Mic60 controls formation of mitochondrial crista junctions. Nature communications 2017, 8, 15258. [Google Scholar] [CrossRef] [PubMed]
- Rampelt, H.; van der Laan, M. The Yin & Yang of Mitochondrial Architecture - Interplay of MICOS and F1Fo-ATP synthase in cristae formation. Microbial cell 2017, 4, 236–239. [Google Scholar] [CrossRef] [PubMed]
- Wollweber, F.; von der Malsburg, K.; van der Laan, M. Mitochondrial contact site and cristae organizing system: A central player in membrane shaping and crosstalk. Biochimica et biophysica acta. Molecular cell research 2017, 1864, 1481–1489. [Google Scholar] [CrossRef] [PubMed]
- Frezza, C.; Cipolat, S.; Martins de Brito, O.; Micaroni, M.; Beznoussenko, G.V.; Rudka, T.; Bartoli, D.; Polishuck, R.S.; Danial, N.N.; De Strooper, B.; et al. OPA1 controls apoptotic cristae remodeling independently from mitochondrial fusion. Cell 2006, 126, 177–189. [Google Scholar] [CrossRef]
- Cogliati, S.; Enriquez, J.A.; Scorrano, L. Mitochondrial Cristae: Where Beauty Meets Functionality. Trends in biochemical sciences 2016, 41, 261–273. [Google Scholar] [CrossRef]
- Zerbes, R.M.; van der Klei, I.J.; Veenhuis, M.; Pfanner, N.; van der Laan, M.; Bohnert, M. Mitofilin complexes: conserved organizers of mitochondrial membrane architecture. Biological chemistry 2012, 393, 1247–1261. [Google Scholar] [CrossRef]
- Barrera, M.; Koob, S.; Dikov, D.; Vogel, F.; Reichert, A.S. OPA1 functionally interacts with MIC60 but is dispensable for crista junction formation. FEBS letters 2016, 590, 3309–3322. [Google Scholar] [CrossRef]
- Amchenkova, A.A.; Bakeeva, L.E.; Chentsov, Y.S.; Skulachev, V.P.; Zorov, D.B. Coupling membranes as energy-transmitting cables. I. Filamentous mitochondria in fibroblasts and mitochondrial clusters in cardiomyocytes. The Journal of cell biology 1988, 107, 481–495. [Google Scholar] [CrossRef]
- Skulachev, V.P. Mitochondrial filaments and clusters as intracellular power-transmitting cables. Trends in biochemical sciences 2001, 26, 23–29. [Google Scholar] [CrossRef]
- Glancy, B.; Hartnell, L.M.; Malide, D.; Yu, Z.X.; Combs, C.A.; Connelly, P.S.; Subramaniam, S.; Balaban, R.S. Mitochondrial reticulum for cellular energy distribution in muscle. Nature 2015, 523, 617–620. [Google Scholar] [CrossRef]
- Williams, S. Mitochondria work much like Tesla battery packs, study finds. Available online: https://phys.org/news/2019-10-mitochondria-tesla-battery.html.
- Sun, J.; Chen, J.; Mohagheghian, E.; Wang, N. Force-induced gene up-regulation does not follow the weak power law but depends on H3K9 demethylation. Science advances 2020, 6, eaay9095. [Google Scholar] [CrossRef] [PubMed]
- Ting, A.Y.; Kain, K.H.; Klemke, R.L.; Tsien, R.Y. Genetically encoded fluorescent reporters of protein tyrosine kinase activities in living cells. Proceedings of the National Academy of Sciences of the United States of America 2001, 98, 15003–15008. [Google Scholar] [CrossRef] [PubMed]
- Tajik, A.; Zhang, Y.; Wei, F.; Sun, J.; Jia, Q.; Zhou, W.; Singh, R.; Khanna, N.; Belmont, A.S.; Wang, N. Transcription upregulation via force-induced direct stretching of chromatin. Nature materials 2016, 15, 1287–1296. [Google Scholar] [CrossRef]
- Xie, L.H.; Ling, Q.D.; Hou, X.Y.; Huang, W. An effective Friedel-Crafts postfunctionization of poly(N-vinylcarbazole) to tune carrier transportation of supramolecular organic semiconductors based on pi-stacked polymers for nonvolatile flash memory cell. Journal of the American Chemical Society 2008, 130, 2120–2121. [Google Scholar] [CrossRef]
- Gomez-Gualdron, D.A.; Burgos, J.C.; Yu, J.; Balbuena, P.B. Carbon nanotubes: engineering biomedical applications. Prog Mol Biol Transl Sci 2011, 104, 175–245. [Google Scholar] [CrossRef]
- Bellucci, L.; Tozzini, V. Engineering 3D Graphene-Based Materials: State of the Art and Perspectives. Molecules 2020, 25. [Google Scholar] [CrossRef]
- Watts, M.C.; Picco, L.; Russell-Pavier, F.S.; Cullen, P.L.; Miller, T.S.; Bartuś, S.P.; Payton, O.D.; Skipper, N.T.; Tileli, V.; Howard, C.A. Production of phosphorene nanoribbons. Nature 2019, 568, 216–220. [Google Scholar] [CrossRef]
- Bhalla, V.; Bajpai, R.P.; Bharadwaj, L.M. DNA electronics. EMBO reports 2003, 4, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Inokuchi, H. The discovery of organic semiconductors. Its light and shadow. Organic Electronics 2006, 7, 62–76. [Google Scholar] [CrossRef]
- Rios, C.; Salcedo, R. Computational study of electron delocalization in hexaarylbenzenes. Molecules (Basel, Switzerland) 2014, 19, 3274–3296. [Google Scholar] [CrossRef]
- Jeppesen, J.O.; Nielsen, M.B.; Becher, J. Tetrathiafulvalene Cyclophanes and Cage Molecules. Chemical reviews 2004, 104, 5115–5132. [Google Scholar] [CrossRef]
- Singh, A.K. Physicochemical, Electronic, and Mechanical Properties of Nanoparticles. Engineered Nanoparticles 2016, 77–123. [Google Scholar] [CrossRef]
- Mima, T.; Kinjo, T.; Yamakawa, S.; Asahi, R. Study of the conformation of polyelectrolyte aggregates using coarse-grained molecular dynamics simulations. Soft Matter 2017, 13, 5991–5999. [Google Scholar] [CrossRef]
- Hossain, M.A.; Işıklan, M.; Pramanik, A.; Saeed, M.A.; Fronczek, F.R. Anion Cluster: Assembly of Dihydrogen Phosphates for the Formation of a Cyclic Anion Octamer. Cryst Growth Des 2012, 12, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Mata, I.; Alkorta, I.; Molins, E.; Espinosa, E. Electrostatics at the Origin of the Stability of Phosphate-Phosphate Complexes Locked by Hydrogen Bonds. Chemphyschem 2012, 13, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Rich, A.; Zhang, S. Z-DNA: the long road to biological function. Nature Reviews Genetics 2003, 4, 566–572. [Google Scholar] [CrossRef]
- Youn, J.H.; Shin, J.-S. Nucleocytoplasmic Shuttling of HMGB1 Is Regulated by Phosphorylation That Redirects It toward Secretion. The Journal of Immunology 2006, 177, 7889–7897. [Google Scholar] [CrossRef]
- Oh, Y.J.; Youn, J.H.; Ji, Y.; Lee, S.E.; Lim, K.J.; Choi, J.E.; Shin, J.-S. HMGB1 Is Phosphorylated by Classical Protein Kinase C and Is Secreted by a Calcium-Dependent Mechanism. The Journal of Immunology 2009, 182, 5800–5809. [Google Scholar] [CrossRef]
- Bannister, A.J.; Kouzarides, T. Regulation of chromatin by histone modifications. Cell Res 2011, 21, 381–395. [Google Scholar] [CrossRef]
- Rossetto, D.; Avvakumov, N.; Côté, J. Histone phosphorylation: a chromatin modification involved in diverse nuclear events. Epigenetics 2012, 7, 1098–1108. [Google Scholar] [CrossRef]
- Mandell, D.J.; Chorny, I.; Groban, E.S.; Wong, S.E.; Levine, E.; Rapp, C.S.; Jacobson, M.P. Strengths of Hydrogen Bonds Involving Phosphorylated Amino Acid Side Chains. Journal of the American Chemical Society 2007, 129, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Hunter, T. Why nature chose phosphate to modify proteins. Philos Trans R Soc Lond B Biol Sci 2012, 367, 2513–2516. [Google Scholar] [CrossRef]
- Thapar, R. Contribution of protein phosphorylation to binding-induced folding of the SLBP-histone mRNA complex probed by phosphorus-31 NMR. FEBS Open Bio 2014, 4, 853–857. [Google Scholar] [CrossRef]
- Krajewska, W.M. Regulation of transcription in eukaryotes by DNA-binding proteins. International Journal of Biochemistry 1992, 24, 1885–1898. [Google Scholar] [CrossRef]
- Erhard, M.; Krenn, M.; Zeilinger, A. Advances in high-dimensional quantum entanglement. Nature Reviews Physics 2020, 2, 365–381. [Google Scholar] [CrossRef]
- D'Acunto, M. Quantum biology.pi-pientanglement signatures in protein-DNA interactions. Phys Biol 2022, 19. [Google Scholar] [CrossRef]
- Kotler, S.; Peterson, G.A.; Shojaee, E.; Lecocq, F.; Cicak, K.; Kwiatkowski, A.; Geller, S.; Glancy, S.; Knill, E.; Simmonds, R.W.; et al. Direct observation of deterministic macroscopic entanglement. Science 2021, 372, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Mercier de Lepinay, L.; Ockeloen-Korppi, C.F.; Woolley, M.J.; Sillanpaa, M.A. Quantum mechanics-free subsystem with mechanical oscillators. Science 2021, 372, 625–629. [Google Scholar] [CrossRef]
- Castelvecchi, D. Minuscule drums push the limits of quantum weirdness. Nature 2021. [Google Scholar] [CrossRef]
- Lau, H.K.; Clerk, A.A. Macroscale entanglement and measurement. Science 2021, 372, 570–571. [Google Scholar] [CrossRef]
- Eswaraiah, V.; Zeng, Q.; Long, Y.; Liu, Z. Black Phosphorus Nanosheets: Synthesis, Characterization and Applications. Small 2016, 12, 3480–3502. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Xie, X.; Duan, G.; Chen, S.H.; Meng, X.-Y.; Zhou, R. Binding patterns and dynamics of double-stranded DNA on the phosphorene surface. Nanoscale 2020, 12, 9430–9439. [Google Scholar] [CrossRef]
- Li, H.-x.; Zhao, K.-c.; Jiang, J.-j.; Zhu, Q.-s. Research progress on black phosphorus hybrids hydrogel platforms for biomedical applications. Journal of Biological Engineering 2023, 17, 8. [Google Scholar] [CrossRef] [PubMed]
- Kumawat, R.L.; Pathak, B. Individual Identification of DNA Nucleobases on Atomically Thin Black Phosphorene Nanoribbons: van der Waals Corrected Density Functional Theory Calculations. The Journal of Physical Chemistry C 2019, 123, 22377–22383. [Google Scholar] [CrossRef]
- Nourbakhsh, Z.; Asgari, R. Charge transport in doped zigzag phosphorene nanoribbons. Physical Review B 2018, 97, 235406. [Google Scholar] [CrossRef]
- Nourbakhsh, Z.; Asgari, R. Phosphorene as a nanoelectromechanical material. Physical Review B 2018, 98, 125427. [Google Scholar] [CrossRef]
- Cheng, T.; Shen, D.X.; Meng, M.; Mallick, S.; Cao, L.; Patmore, N.J.; Zhang, H.L.; Zou, S.F.; Chen, H.W.; Qin, Y.; et al. Efficient electron transfer across hydrogen bond interfaces by proton-coupled and -uncoupled pathways. Nature communications 2019, 10, 1531. [Google Scholar] [CrossRef]
- Benco, L.; Tunega, D.; Hafner, J.; Lischka, H. Upper Limit of the O−H···O Hydrogen Bond. Ab Initio Study of the Kaolinite Structure. The Journal of Physical Chemistry B 2001, 105, 10812–10817. [Google Scholar] [CrossRef]
- Gray, H.B.; Winkler, J.R. Long-range electron transfer. Proceedings of the National Academy of Sciences of the United States of America 2005, 102, 3534–3539. [Google Scholar] [CrossRef]
- Gilbert Gatty, M.; Kahnt, A.; Esdaile, L.J.; Hutin, M.; Anderson, H.L.; Albinsson, B. Hopping versus Tunneling Mechanism for Long-Range Electron Transfer in Porphyrin Oligomer Bridged Donor-Acceptor Systems. The journal of physical chemistry. B 2015, 119, 7598–7611. [Google Scholar] [CrossRef]
- Davis, W.B.; Svec, W.A.; Ratner, M.A.; Wasielewski, M.R. Molecular-wire behaviour in p -phenylenevinylene oligomers. Nature 1998, 396, 60–63. [Google Scholar] [CrossRef]
- Giacalone, F.; Segura, J.L.; Martín, N.; Guldi, D.M. Exceptionally Small Attenuation Factors in Molecular Wires. Journal of the American Chemical Society 2004, 126, 5340–5341. [Google Scholar] [CrossRef]
- Goldsmith, R.H.; Sinks, L.E.; Kelley, R.F.; Betzen, L.J.; Liu, W.; Weiss, E.A.; Ratner, M.A.; Wasielewski, M.R. Wire-like charge transport at near constant bridge energy through fluorene oligomers. Proceedings of the National Academy of Sciences of the United States of America 2005, 102, 3540–3545. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, G.; Giacalone, F.; Segura, J.L.; Martín, N.; Guldi, D.M. Electronic Communication through π-Conjugated Wires in Covalently Linked Porphyrin/C60Ensembles. Chemistry - A European Journal 2005, 11, 1267–1280. [Google Scholar] [CrossRef] [PubMed]
- Sedghi, G.; Esdaile, L.J.; Anderson, H.L.; Martin, S.; Bethell, D.; Higgins, S.J.; Nichols, R.J. Comparison of the Conductance of Three Types of Porphyrin-Based Molecular Wires: beta,meso,beta-Fused Tapes, meso-Butadiyne-Linked and Twisted meso-meso Linked Oligomers. Adv Mater 2012, 24, 653–653. [Google Scholar] [CrossRef] [PubMed]
- Sukegawa, J.; Schubert, C.; Zhu, X.; Tsuji, H.; Guldi, D.M.; Nakamura, E. Electron transfer through rigid organic molecular wires enhanced by electronic and electron–vibration coupling. Nat Chem 2014, 6, 899–905. [Google Scholar] [CrossRef]
- Muzzio, N.E.; Pasquale, M.A.; Marmisollé, W.A.; von Bilderling, C.; Cortez, M.L.; Pietrasanta, L.I.; Azzaroni, O. Self-assembled phosphate-polyamine networks as biocompatible supramolecular platforms to modulate cell adhesion. Biomaterials Science 2018, 6, 2230–2247. [Google Scholar] [CrossRef]
- Perez-Mitta, G.; Marmisolle, W.A.; Albesa, A.G.; Toimil-Molares, M.E.; Trautmann, C.; Azzaroni, O. Phosphate-Responsive Biomimetic Nanofluidic Diodes Regulated by Polyamine-Phosphate Interactions: Insights into Their Functional Behavior from Theory and Experiment. Small 2018, 14, e1702131. [Google Scholar] [CrossRef]
- Fenoy, G.E.; Piccinini, E.; Knoll, W.; Marmisollé, W.A.; Azzaroni, O. The Effect of Amino–Phosphate Interactions on the Biosensing Performance of Enzymatic Graphene Field-Effect Transistors. Analytical chemistry 2022, 94, 13820–13828. [Google Scholar] [CrossRef]
- Ashoka, A.; Clancy, A.J.; Panjwani, N.A.; Cronin, A.; Picco, L.; Aw, E.S.Y.; Popiel, N.J.M.; Eaton, A.G.; Parton, T.G.; Shutt, R.R.C.; et al. Magnetically and optically active edges in phosphorene nanoribbons. Nature 2025, 639, 348–353. [Google Scholar] [CrossRef]
- Wang, C.-H.; Incorvia, J.A.C.; McClellan, C.J.; Yu, A.C.; Mleczko, M.J.; Pop, E.; Wong, H.S.P. Unipolar n-Type Black Phosphorus Transistors with Low Work Function Contacts. Nano Letters 2018, 18, 2822–2827. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-Y.; Su, S.-K.; Wong, H.S.P.; Li, L.-J. How 2D semiconductors could extend Moore’s law. Nature 2019, 567, 169–170. [Google Scholar] [CrossRef] [PubMed]
- Salomon, A.; Cahen, D.; Lindsay, S.; Tomfohr, J.; Engelkes, V.B.; Frisbie, C.D. Comparison of Electronic Transport Measurements on Organic Molecules. Adv Mater 2003, 15, 1881–1890. [Google Scholar] [CrossRef]
- Coropceanu, V.; Cornil, J.; da Silva Filho, D.A.; Olivier, Y.; Silbey, R.; Brédas, J.-L. Charge Transport in Organic Semiconductors. Chemical reviews 2007, 107, 926–952. [Google Scholar] [CrossRef] [PubMed]
- Facchetti, A. π-Conjugated Polymers for Organic Electronics and Photovoltaic Cell Applications. Chemistry of Materials 2011, 23, 733–758. [Google Scholar] [CrossRef]
- Irimia-Vladu, M.; Kanbur, Y.; Camaioni, F.; Coppola, M.E.; Yumusak, C.; Irimia, C.V.; Vlad, A.; Operamolla, A.; Farinola, G.M.; Suranna, G.P.; et al. Stability of Selected Hydrogen Bonded Semiconductors in Organic Electronic Devices. Chemistry of Materials 2019, 31, 6315–6346. [Google Scholar] [CrossRef]
- Ibusuki, R.; Morishita, T.; Furuta, A.; Nakayama, S.; Yoshio, M.; Kojima, H.; Oiwa, K.; Furuta, K. Programmable molecular transport achieved by engineering protein motors to move on DNA nanotubes. Science 2022, 375, 1159–1164. [Google Scholar] [CrossRef]
- Li, H.X.; Zhao, K.C.; Jiang, J.J.; Zhu, Q.S. Research progress on black phosphorus hybrids hydrogel platforms for biomedical applications. J Biol Eng 2023, 17, 8. [Google Scholar] [CrossRef]
- Li, P.; Kim, S.; Tian, B. Beyond 25 years of biomedical innovation in nano-bioelectronics. Device 2024, 2. [Google Scholar] [CrossRef]
- Alan, Crawford; Jarrell, Dillard; Helene, Fouquet; Reynolds., I. Alan Crawford; Jarrell Dillard; Helene Fouquet; Reynolds., I. The World Is Dangerously Dependent on Taiwan for Semiconductors. Available online: https://www.bloombergquint.com/technology/the-world-is-dangerously-dependent-on-taiwan-for-semiconductors.
- Lee, M.; Weng, M.-H.; Jang, S.-L. The Competitiveness and Future Challenge of the Taiwan Semiconductors Industry. In Technology Rivalry Between the USA and China, C.Y. Chow, P., Ed.; Springer Nature Switzerland: Cham, 2024. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



