Submitted:
21 March 2025
Posted:
21 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Peptides and Reagents
2.3. Cell Viability and Experimental Designs
2.4. Detection of Apoptosis by Flow Cytometry
2.5. Independence Assessment
2.6. Plate-Seq Data Analysis
2.7. Statistical Analyses
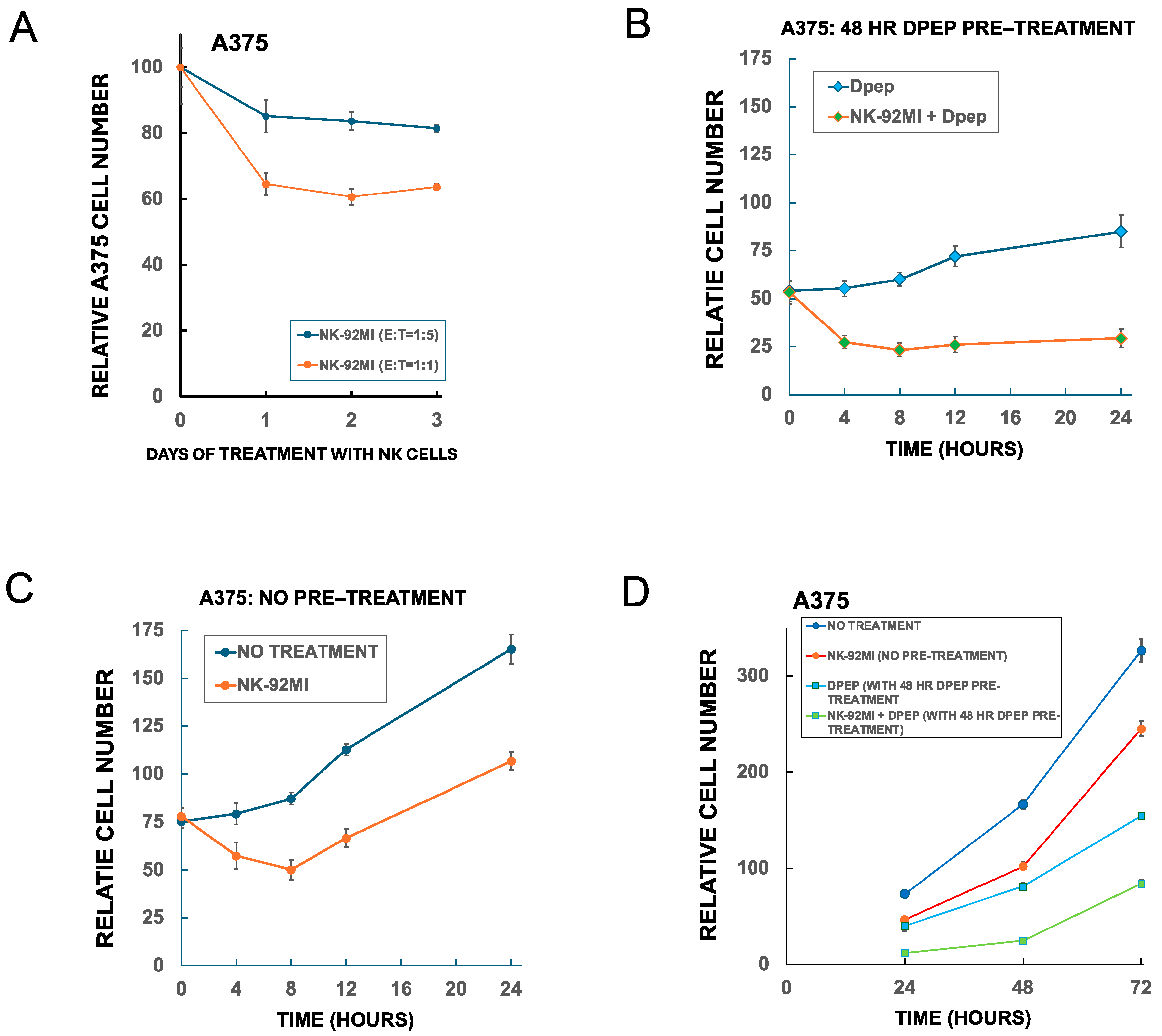
3. Results
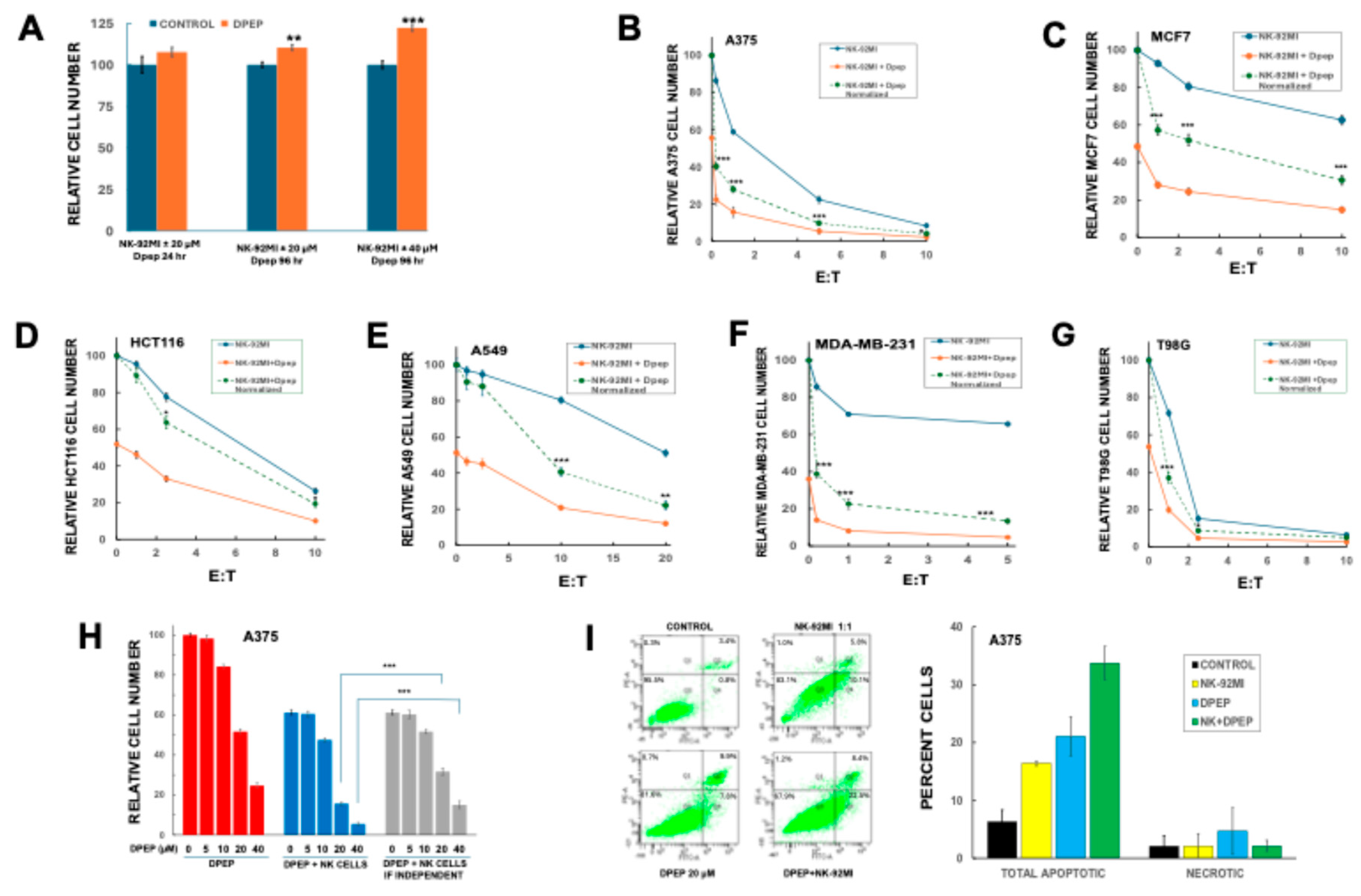
3.1. Dpep Does Not Adversely Affect NK-92MI Cell Growth/Survival
3.2. Dpep Sensitizes Multiple Tumor Cell Lines to NK-92MI Cell Killing
3.3. Dpep Sensitizes Tumor Cells to NK-92MI Cells in a Dose-Dependent Manner
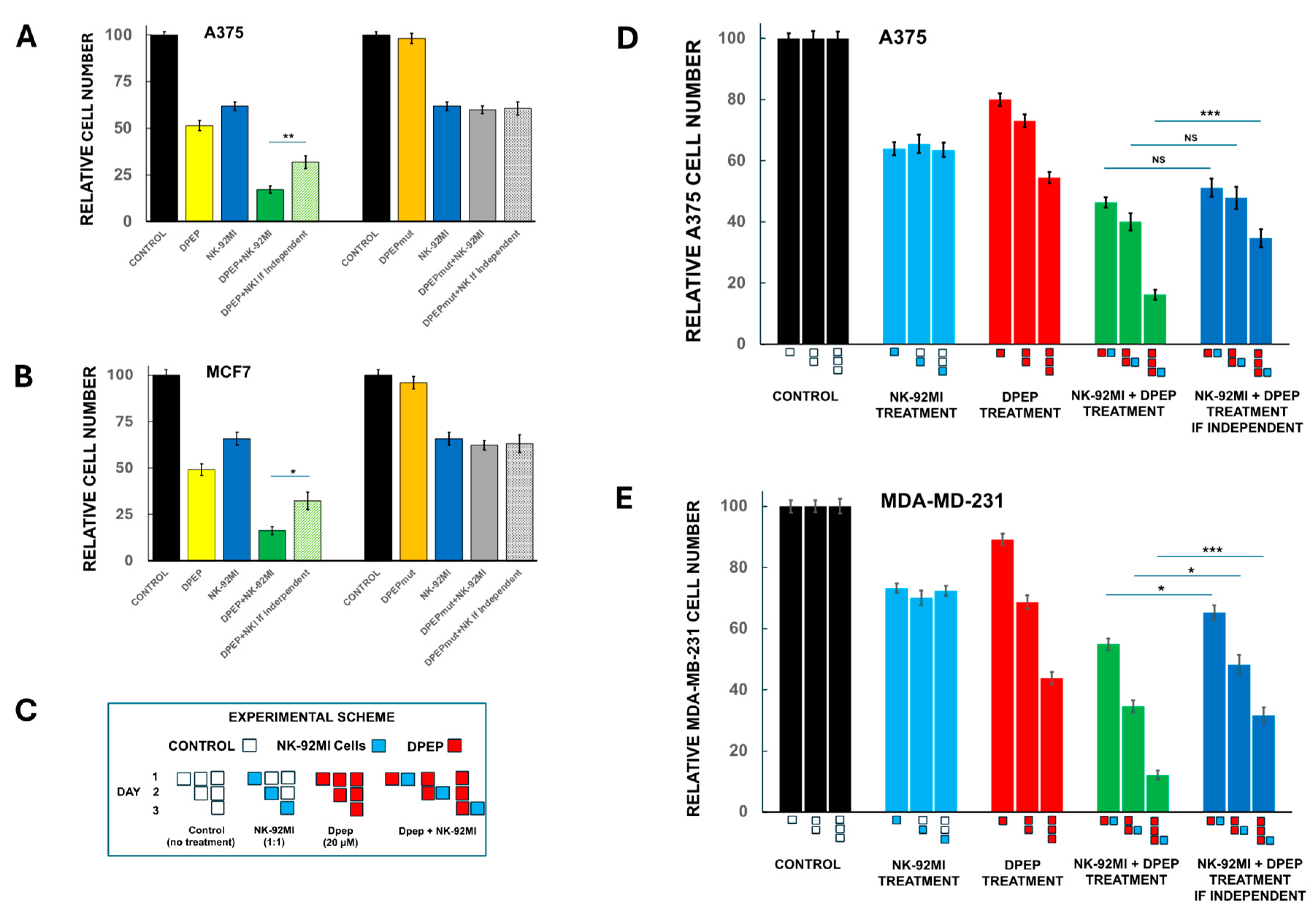
3.4. Sensitization of Tumor Cells to NK-92MI Cells Requires Active Peptide
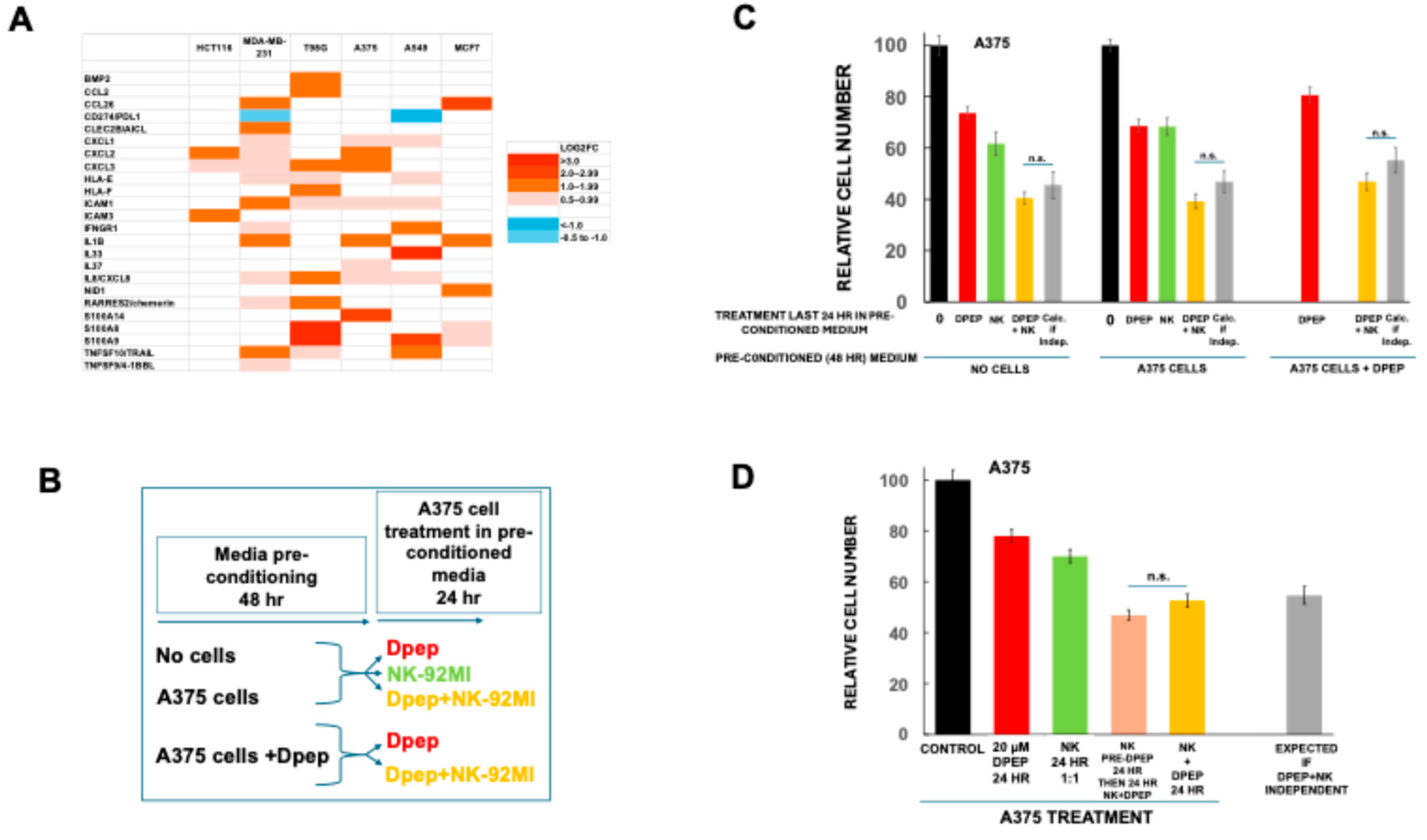
3.5. Sensitization of Tumor Cells to NK-92MI Cells Increases as a Function of Dpep Pre-Treatment Time
3.6. Medium Conditioned by Dpep-Treated Tumor Cells Does Not Affect NK-92MI Cell Cytotoxicity
3.7. Direct Treatment of NK-92MI Cells with Dpep Does Not Substantially Affect Their Activity
3.8. Tumor Cell Sensitization to NK-92MI Cells Requires Susceptibility to Dpep Killing
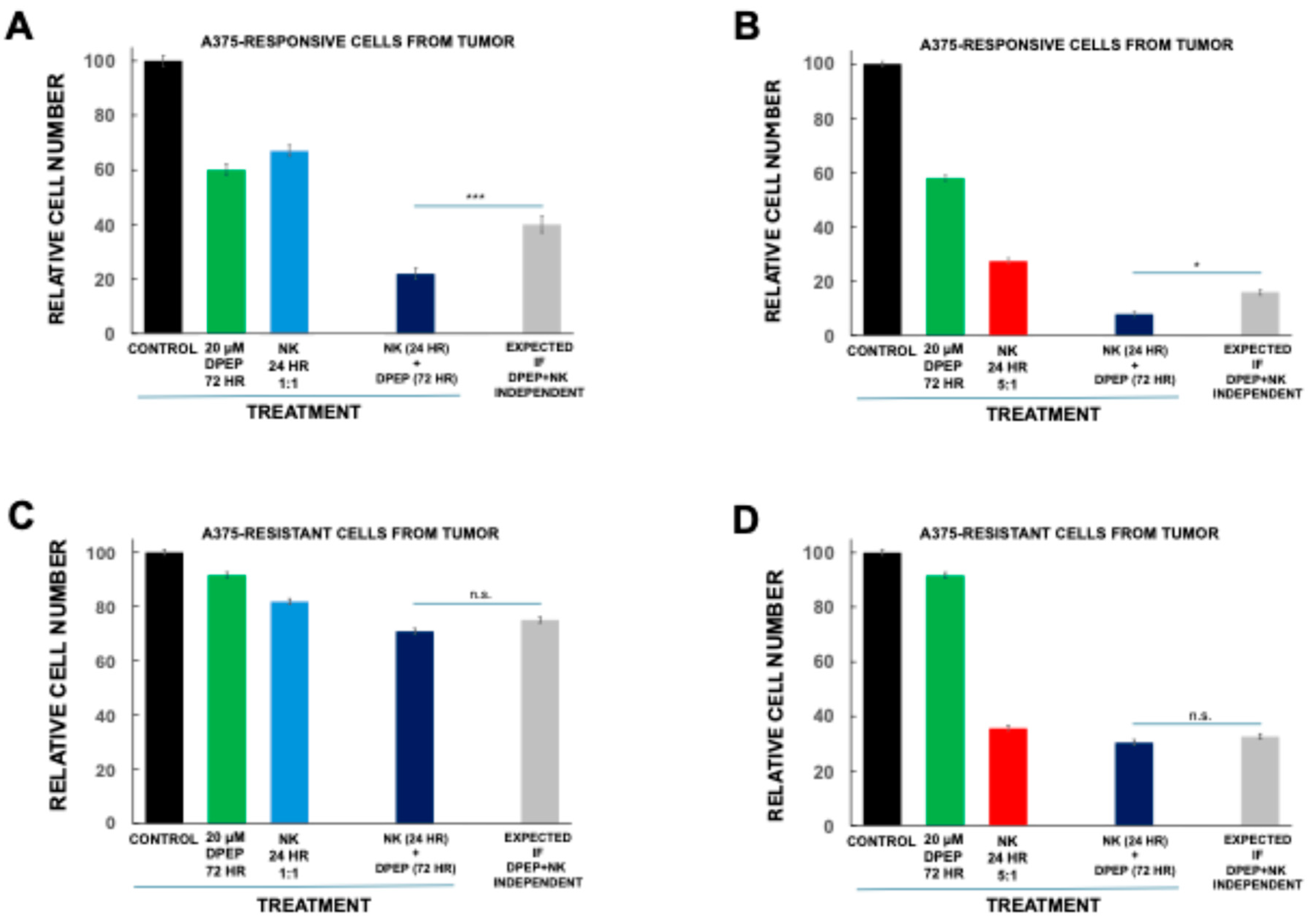
3.9. Dpep and NK Cell Exhaustion
3.10. Dpep-Treated Tumor Cells Respond to Serial Treatments with NK-92MI Cells
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| NK cells | Natural killer cells |
| CAR-NK | Chimeric Antigen Receptor-engineered NK cells |
| FBS | Fetal bovine serum |
| E:T | Effector-to-target |
| FITC | Fluorescein isothiocyanate |
| PI | Propidium iodide |
References
- Laskowski, T.J.; Biederstadt, A.; Rezvani, K. Natural killer cells in antitumour adoptive cell immunotherapy. Nat Rev Cancer 2022, 22(10), 557–575. [Google Scholar] [CrossRef] [PubMed]
- Wolf, N.K.; Kissiov, D.U.; Raulet, D.H. Roles of natural killer cells in immunity to cancer, and applications to immunotherapy. Nat Rev Immunol 2023, 23(2), 90–105. [Google Scholar] [CrossRef]
- Chen, S.; Zhu, H.; Jounaidi, Y. Comprehensive snapshots of natural killer cells functions, signaling, molecular mechanisms and clinical utilization. Signal Transduct Target Ther 2024, 9(1), 302. [Google Scholar] [CrossRef]
- Franks, S.E.; Wolfson, B.; Hodge, J.W. Natural Born Killers: NK Cells in Cancer Therapy. Cancers (Basel) 2020, 12(8). [Google Scholar] [CrossRef]
- Klingemann, H. The NK-92 cell line-30 years later: Its impact on natural killer cell research and treatment of cancer. Cytotherapy 2023, 25(5), 451–457. [Google Scholar] [CrossRef]
- Zhang, C.; Oberoi, P.; Oelsner, S.; Waldmann, A.; Lindner, A.; Tonn, T.; Wels, W.S. Chimeric Antigen Receptor-Engineered NK-92 Cells: An Off-the-Shelf Cellular Therapeutic for Targeted Elimination of Cancer Cells and Induction of Protective Antitumor Immunity. Front Immunol 2017, 8, 533. [Google Scholar] [CrossRef]
- Tam, Y.K.; Maki, G.; Miyagawa, B.; Hennemann, B.; Tonn, T.; Klingemann, H.G. Characterization of genetically altered, interleukin 2-independent natural killer cell lines suitable for adoptive cellular immunotherapy. Hum Gene Ther 1999, 10(8), 1359–1373. [Google Scholar] [CrossRef] [PubMed]
- Tam, Y.K.; Miyagawa, B.; Ho, V.C.; Klingemann, H.G. Immunotherapy of malignant melanoma in a SCID mouse model using the highly cytotoxic natural killer cell line NK-92. J Hematother 1999, 8(3), 281–290. [Google Scholar] [CrossRef] [PubMed]
- Fabian, K.P.; Hodge, J.W. The emerging role of off-the-shelf engineered natural killer cells in targeted cancer immunotherapy. Mol Ther Oncolytics 2021, 23, 266–276. [Google Scholar] [CrossRef]
- Suck, G.; Odendahl, M.; Nowakowska, P.; Seidl, C.; Wels, W.S.; Klingemann, H.G.; Tonn, T. NK-92: An 'off-the-shelf therapeutic' for adoptive natural killer cell-based cancer immunotherapy. Cancer Immunol Immunother 2016, 65(4), 485–492. [Google Scholar] [CrossRef]
- Lamers-Kok, N.; Panella, D.; Georgoudaki, A.M.; Liu, H.; Ozkazanc, D.; Kucerova, L.; Duru, A.D.; Spanholtz, J.; Raimo, M. Natural killer cells in clinical development as non-engineered, engineered, and combination therapies. J Hematol Oncol 2022, 15(1), 164. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Wang, M.; Yan, Y.; Jin, X.; Ning, L.; Xu, B.; Wang, Y.; Hao, Y.; Luo, Z.; Guo, C.; et al. Challenges in the Development of NK-92 Cells as an Effective Universal Off-the-Shelf Cellular Therapeutic. J Immunol 2024, 213(9), 1318–1328. [Google Scholar] [CrossRef] [PubMed]
- Maki, G.; Krystal, G.; Dougherty, G.; Takei, F.; Klingemann, H.G. Induction of sensitivity to NK-mediated cytotoxicity by TNF-alpha treatment: Possible role of ICAM-3 and CD44. Leukemia 1998, 12(10), 1565–1572. [Google Scholar] [CrossRef]
- Kim, H.; Kim, S.H.; Kim, M.J.; Kim, S.J.; Park, S.J.; Chung, J.S.; Bae, J.H.; Kang, C.D. EGFR inhibitors enhanced the susceptibility to NK cell-mediated lysis of lung cancer cells. J Immunother 2011, 34(4), 372–381. [Google Scholar] [CrossRef]
- Sawasdee, N.; Wattanapanitch, M.; Thongsin, N.; Phanthaphol, N.; Chiawpanit, C.; Thuwajit, C.; Yenchitsomanus, P.T.; Panya, A. Doxorubicin sensitizes breast cancer cells to natural killer cells in connection with increased Fas receptors. Int J Mol Med 2022, 49(3). [Google Scholar] [CrossRef]
- Raja, R.; Wu, C.; Bassoy, E.Y.; Rubino, T.E., Jr.; Utagawa, E.C.; Magtibay, P.M.; Butler, K.A.; Curtis, M. PP4 inhibition sensitizes ovarian cancer to NK cell-mediated cytotoxicity via STAT1 activation and inflammatory signaling. J Immunother Cancer 2022, 10(12). [Google Scholar] [CrossRef] [PubMed]
- Son, C.H.; Keum, J.H.; Yang, K.; Nam, J.; Kim, M.J.; Kim, S.H.; Kang, C.D.; Oh, S.O.; Kim, C.D.; Park, Y.S.; et al. Synergistic enhancement of NK cell-mediated cytotoxicity by combination of histone deacetylase inhibitor and ionizing radiation. Radiat Oncol 2014, 9, 49. [Google Scholar] [CrossRef]
- Karpel-Massler, G.; Horst, B.A.; Shu, C.; Chau, L.; Tsujiuchi, T.; Bruce, J.N.; Canoll, P.; Greene, L.A.; Angelastro, J.M.; Siegelin, M.D. A Synthetic Cell-Penetrating Dominant-Negative ATF5 Peptide Exerts Anticancer Activity against a Broad Spectrum of Treatment-Resistant Cancers. Clin Cancer Res 2016, 22(18), 4698–4711. [Google Scholar] [CrossRef]
- Zhou, Q.; Sun, X.; Pasquier, N.; Jefferson, P.; Nguyen, T.T.T.; Siegelin, M.D.; Angelastro, J.M.; Greene, L.A. Cell-Penetrating CEBPB and CEBPD Leucine Zipper Decoys as Broadly Acting Anti-Cancer Agents. Cancers (Basel) 2021, 13(10). [Google Scholar] [CrossRef]
- Greene, L.A.; Zhou, Q.; Siegelin, M.D.; Angelastro, J.M. Targeting Transcription Factors ATF5, CEBPB and CEBPD with Cell-Penetrating Peptides to Treat Brain and Other Cancers. Cells 2023, 12(4). [Google Scholar] [CrossRef]
- Zhou, Q.; Greene, L.A. Dpep Inhibits Cancer Cell Growth and Survival via Shared and Context-Dependent Transcriptome Perturbations. Cancers (Basel) 2023, 15(22). [Google Scholar] [CrossRef]
- Melaiu, O.; Lucarini, V.; Cifaldi, L.; Fruci, D. Influence of the Tumor Microenvironment on NK Cell Function in Solid Tumors. Front Immunol 2019, 10, 3038. [Google Scholar] [CrossRef] [PubMed]
- Ran, G.H.; Lin, Y.Q.; Tian, L.; Zhang, T.; Yan, D.M.; Yu, J.H.; Deng, Y.C. Natural killer cell homing and trafficking in tissues and tumors: From biology to application. Signal Transduct Target Ther 2022, 7(1), 205. [Google Scholar] [CrossRef]
- Zhi, L.; Wang, X.; Gao, Q.; He, W.; Shang, C.; Guo, C.; Niu, Z.; Zhu, W.; Zhang, X. Intrinsic and extrinsic factors determining natural killer cell fate: Phenotype and function. Biomed Pharmacother 2023, 165, 115136. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.; Tian, Z. NK Cell Exhaustion. Front Immunol 2017, 8, 760. [Google Scholar] [CrossRef]
- Jia, H.; Yang, H.; Xiong, H.; Luo, K.Q. NK cell exhaustion in the tumor microenvironment. Front Immunol 2023, 14, 1303605. [Google Scholar] [CrossRef]
- Cozar, B.; Greppi, M.; Carpentier, S.; Narni-Mancinelli, E.; Chiossone, L.; Vivier, E. Tumor-Infiltrating Natural Killer Cells. Cancer Discov 2021, 11(1), 34–44. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Steinherz, P.; Klingemann, H.G.; Dennig, D.; Childs, B.H.; McGuirk, J.; O'Reilly, R.J. Antileukemia activity of a natural killer cell line against human leukemias. Clin Cancer Res 1998, 4(11), 2859–2868. [Google Scholar]
- Sun, X.; Angelastro, J.M.; Merino, D.; Zhou, Q.; Siegelin, M.D.; Greene, L.A. Dominant-negative ATF5 rapidly depletes survivin in tumor cells. Cell Death Dis 2019, 10(10), 709. [Google Scholar] [CrossRef]
- Pan, K.; Farrukh, H.; Chittepu, V.C.S.R.; et al. CAR race to cancer immunotherapy: From CAR T, CAR NK to CAR macrophage therapy. J Exp Clin Cancer Res. 2022, 41, 119. [Google Scholar] [CrossRef]
- Tong, L.; Jimenez-Cortegana, C.; Tay, A.H.M.; Wickstrom, S.; Galluzzi, L.; Lundqvist, A. NK cells and solid tumors: Therapeutic potential and persisting obstacles. Mol Cancer 2022, 21(1), 206. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Yang, H.; Xiong, H.; Luo, K.Q. NK cell exhaustion in the tumor microenvironment. Front Immunol 2023, 14, 1303605. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, J.M.; Rehman, S.; Virumbrales-Munoz, M.; McMinn, P.H.; Geiger, P.; Fitzgerald, C.; Heaster, T.; Skala, M.C.; Beebe, D.J. Microfluidic tumor-on-a-chip model to evaluate the role of tumor environmental stress on NK cell exhaustion. Sci Adv 2021, 7(8). [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



