Submitted:
18 March 2025
Posted:
18 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
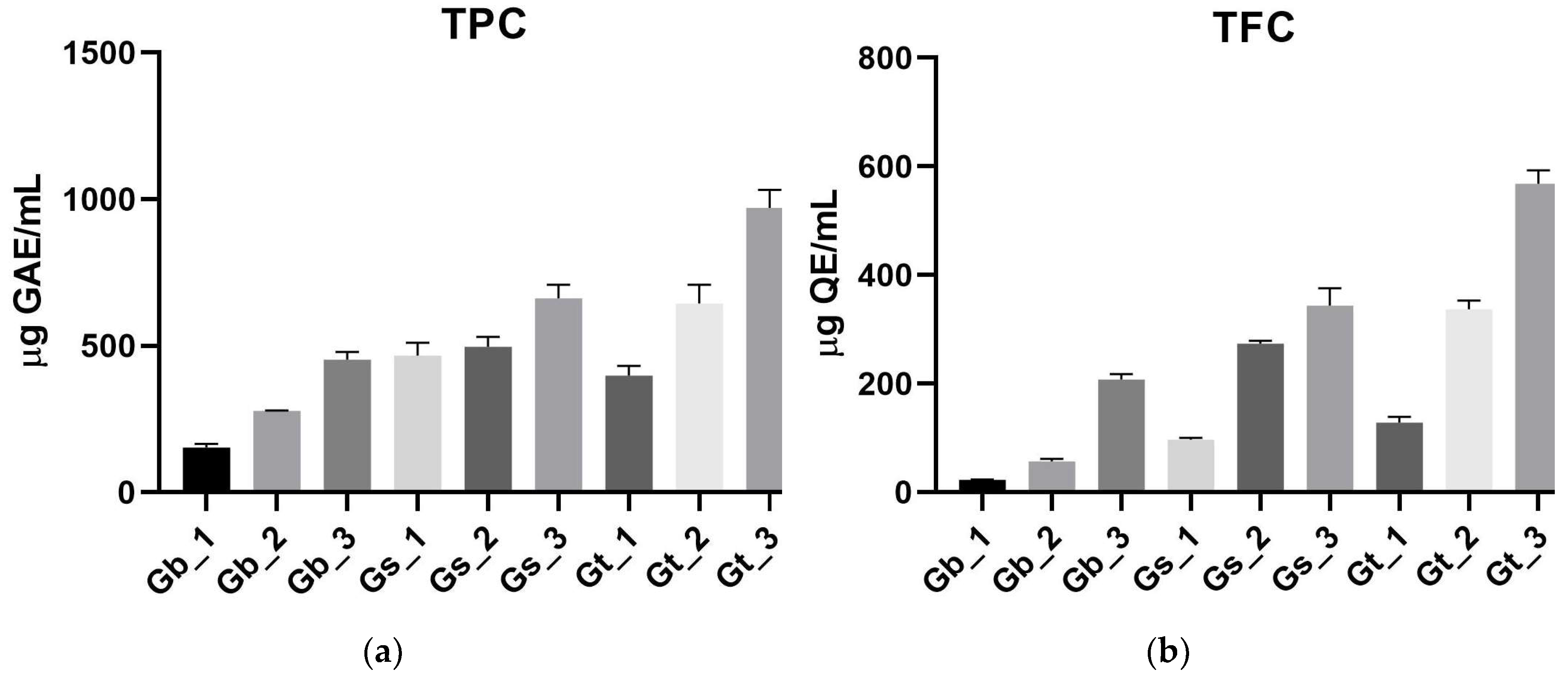
2.1. Total Polyphenols and Flavonoids
2.1.1. Effect of Species and Plant Parts on TPC
2.1.2. Effect of Species and Plant Parts on TFC
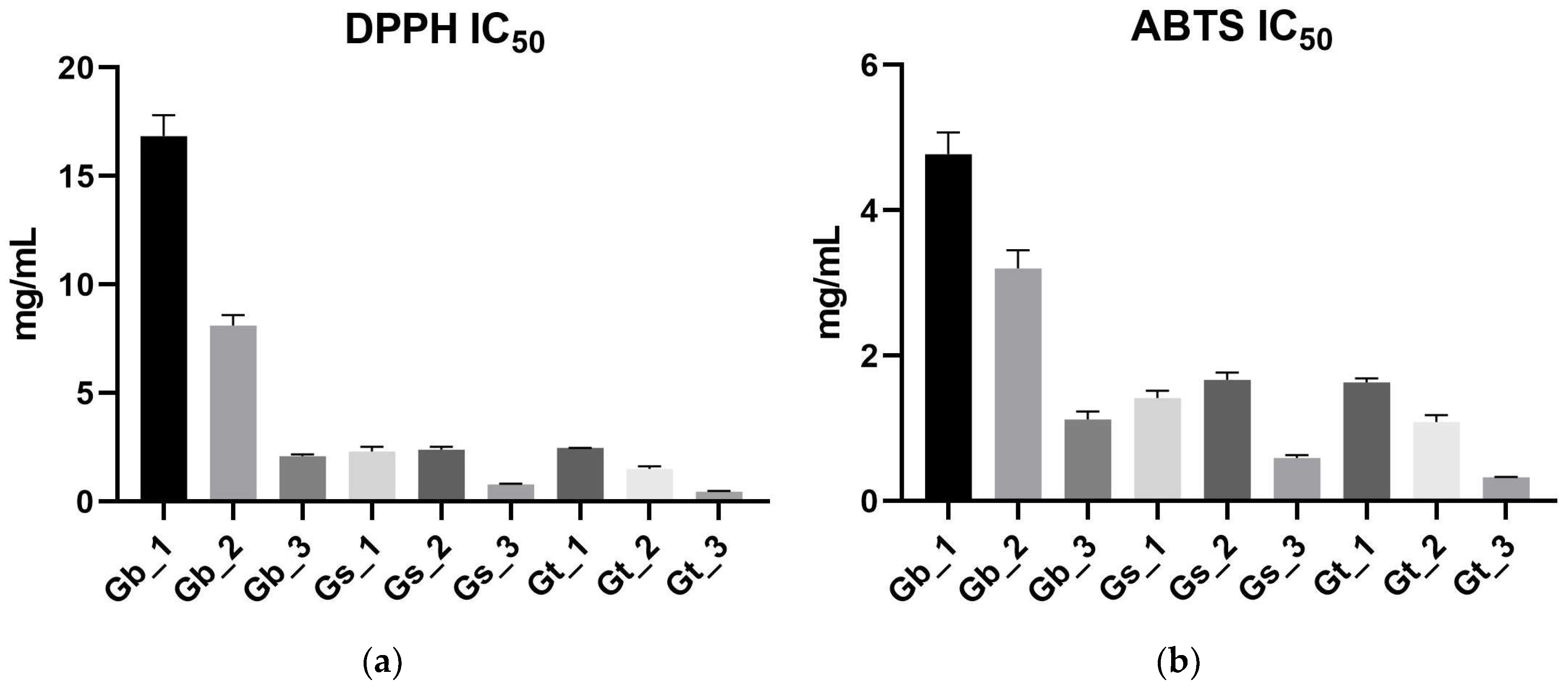
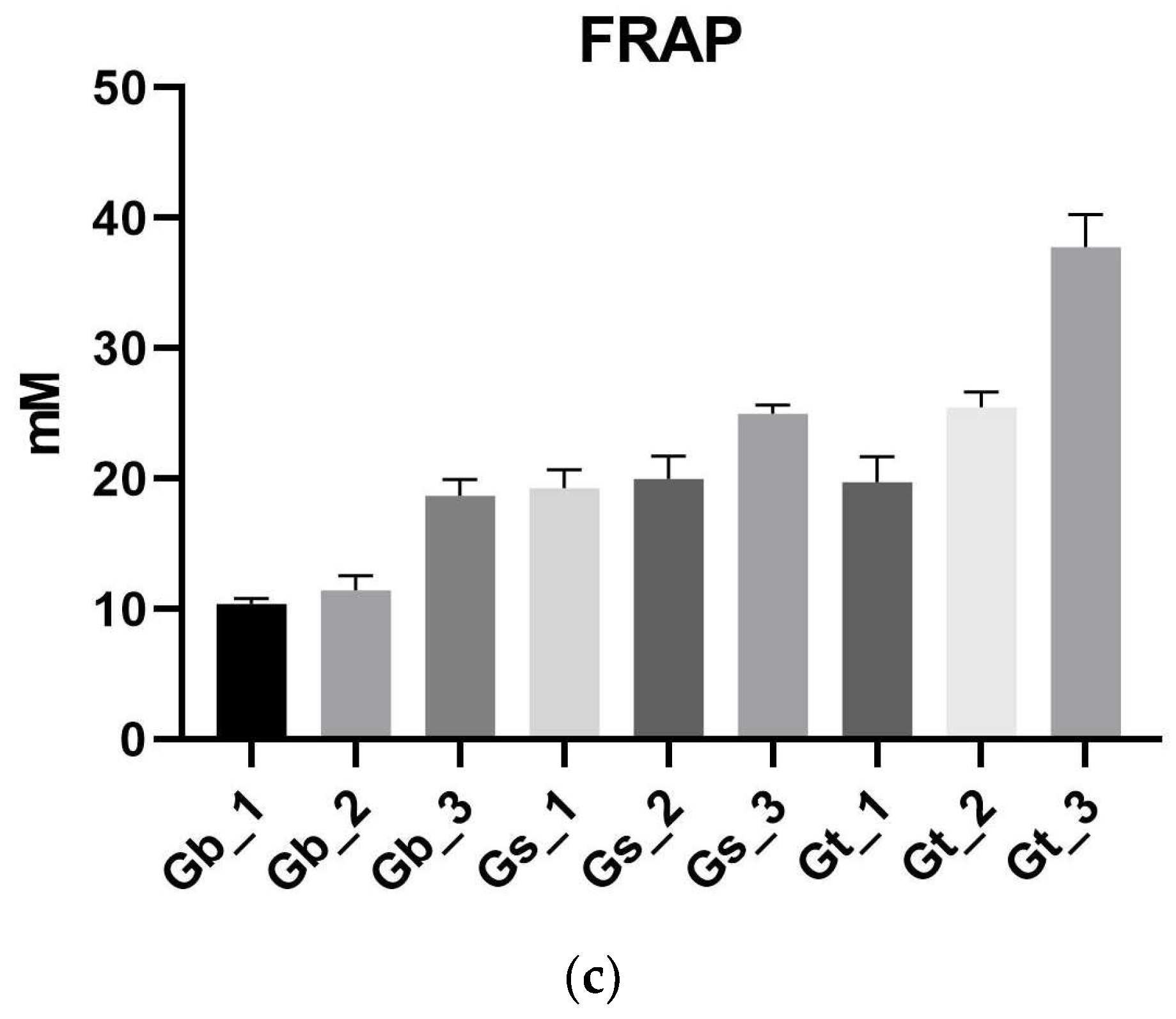
2.2. Antioxidant Activity (DPPH, ABTS, FRAP)
2.2.1. DPPH Radical Scavenging Activity
2.2.2. ABTS Radical Scavenging Activity
2.2.3. FRAP Assay
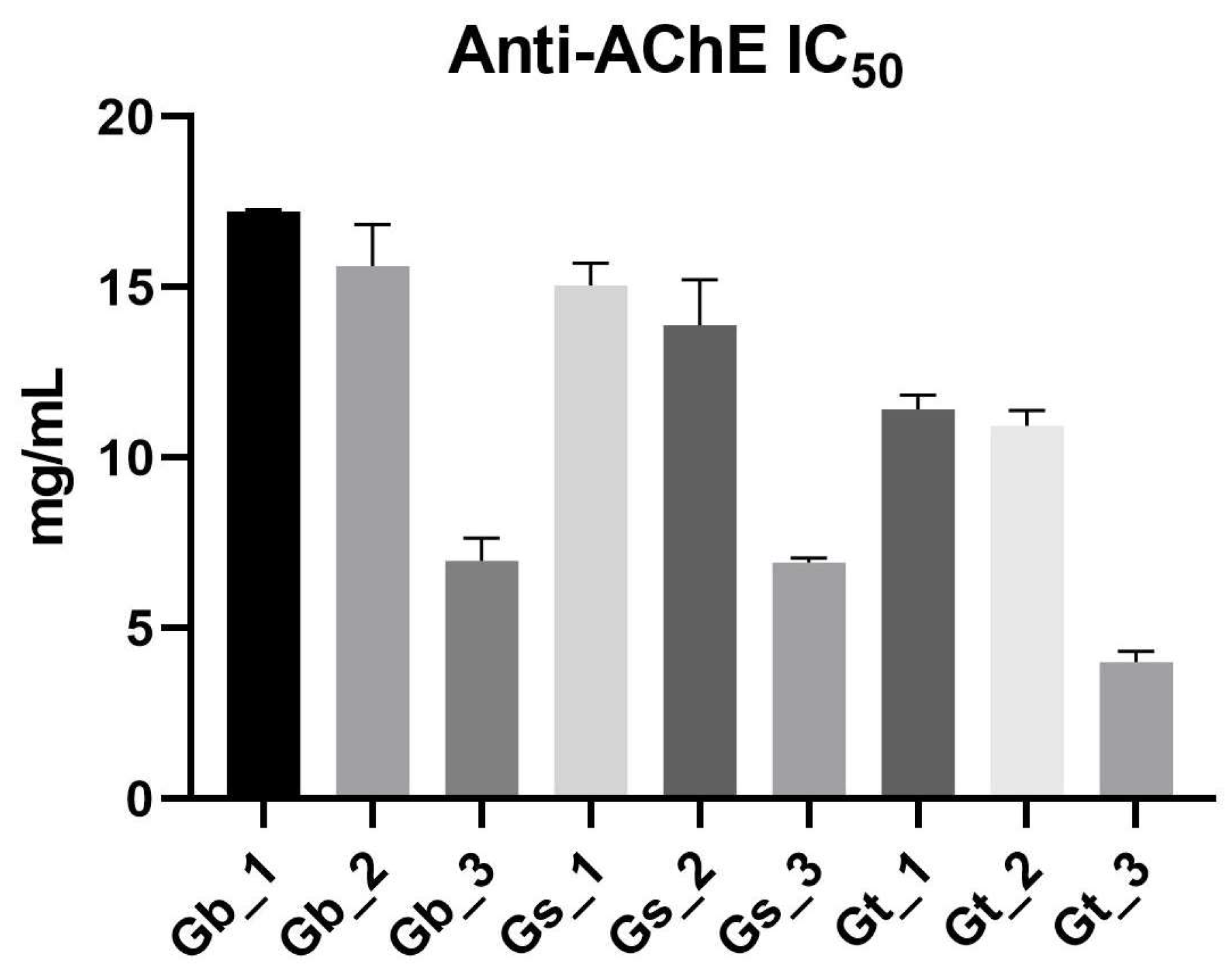
2.3. Neuroprotective (AChE Inhibition) Activity
2.3.1. Influence of Species and Plant Part on AChE Inhibition (IC50 Values)
2.4. HPTLC Fingerprinting for Antioxidant and Neuroprotective Activity
2.5. Phenolic Acids Profile (UHPLC Analysis)
3. Discussion
3.1. Total Polyphenols and Flavonoids
3.2. Antioxidant Activity
- DPPH IC50 vs. ABTS IC50: A strong positive correlation (r=0.983, p<0.05) was observed, indicating that extracts with higher radical scavenging efficiency in the DPPH assay also exhibited strong activity in the ABTS assay;
- DPPH IC50 vs. FRAP (mM Fe2+): A negative correlation (r=-0.833, p<0.05) was found, suggesting that extracts requiring higher concentrations to inhibit 50% of DPPH radicals tended to exhibit higher reducing power in the FRAP assay;
- ABTS IC50 vs. FRAP (mM Fe2+): A negative correlation (r=-0.817, p<0.05) was also observed, indicating an inverse relationship between radical scavenging capacity and ferric-reducing ability.
- TPC vs. DPPH IC50: A strong negative correlation (r=-0.9333, p=0.0007), indicating that extracts with higher polyphenol content required lower concentrations to inhibit 50% of DPPH radicals, thus demonstrating stronger radical scavenging activity;
- TPC vs. ABTS IC50: A moderate negative correlation (r=-0.8833, p=0.0031), suggesting that higher polyphenol levels were associated with greater ABTS radical scavenging efficiency;
- TPC vs. FRAP (mM Fe2+ equivalents): A strong positive correlation (r=0.9333, p=0.0007), indicating that extracts with higher polyphenol content exhibited greater ferric-reducing power.
- TFC vs. DPPH IC50: A strong negative correlation (r=-0.9167, p=0.0013), indicating that extracts with higher flavonoid content required lower concentrations to inhibit 50% of DPPH radicals, confirming their potent radical scavenging capacity;
- TFC vs. ABTS IC50: A moderate negative correlation (r=-0.8833, p=0.0031), suggesting that an increase in flavonoid content was associated with improved ABTS radical scavenging efficiency;
- TFC vs. FRAP (mM Fe2+ equivalents): A strong positive correlation (r=0.9333, p=0.0007), indicating that extracts with higher flavonoid content exhibited greater ferric-reducing power.
3.3. Neuroprotective Activity
- AChE inhibition vs. TPC: A moderate negative correlation (r=-0.8266, p=0.0060), suggesting that extracts with higher total polyphenol content exhibited greater AChE inhibition. The 95% confidence interval (CI) ranged from -0.9624 to -0.3603, supporting the statistical robustness of this relationship;
- AChE inhibition vs. TFC: A moderate negative correlation (r=-0.8335, p=0.0053), indicating that an increase in flavonoid content was associated with stronger AChE inhibition. The 95% CI ranged from -0.9640 to -0.3793, reinforcing the reliability of the association.
- AChE inhibition vs. DPPH IC50: A moderate positive correlation (r=0.6887, p=0.0402), indicating that extracts with higher AChE inhibition also tended to require lower concentrations to scavenge 50% of DPPH radicals. However, the correlation was weaker compared to other parameters, as reflected by the 95% CI ranging from 0.04535 to 0.9283;
- AChE inhibition vs. ABTS IC50: A strong positive correlation (r=0.8085, p=0.0083), suggesting that extracts with greater AChE inhibition demonstrated enhanced ABTS radical scavenging activity. The 95% CI (0.3117 to 0.9581) reinforces the statistical robustness of this relationship;
- AChE inhibition vs. FRAP (mM Fe2+ equivalents): A moderate negative correlation (r=-0.8238, p=0.0063), showing that extracts with higher AChE inhibition exhibited stronger reducing power. The negative correlation suggests that extracts with high AChE inhibition had greater ferric-reducing capacity, a trend supported by the 95% CI of -0.9617 to -0.3526.
3.4. Study Limitations
3.4.1. Variability in Plant Material and Environmental Influence
3.4.2. Extraction Method and Solvent Specificity
3.4.3. HPTLC and UHPLC Identification Constraints
3.4.4. Lack of In Vivo Validation
3.4.5. Potential Interference in Quantification Assays
4. Materials and Methods
4.1. Plant Material
4.2. Chemicals and Reagents
4.3. Extraction Procedure
4.4. Standards Preparation
4.5. Total Polyphenols and Flavonoids
4.5.1. TPC Assay
4.5.2. TFC Assay
4.6. Antioxidant Activity Assays
4.6.1. DPPH Antioxidant Assay
4.6.2. ABTS Antioxidant Assay
4.6.3. FRAP Antioxidant Assay
4.7. Neuroprotective Activity Assay
4.8. HPTLC Fingerprinting for Antioxidant and Neuroprotective Activity
4.9. UHPLC Analysis of Phenolic Acids
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABTS | 2,2’-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| AChE | Acetylcholinesterase |
| AlCl3 | Aluminum chloride |
| ANOVA | Analysis of variance |
| CI | Confidence interval |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| FeCl3 | Ferric chloride |
| FeSO4·7H2O | Ferrous sulfate heptahydrate |
| FRAP | Ferric-reducing antioxidant power |
| GAE | Gallic acid equivalents |
| Gb | Galeopsis bifida |
| Gs | Galeopsis speciosa |
| Gt | Galeopsis tetrahit |
| HCl | Hydrochloric acid |
| HPTLC | High-performance thin-layer chromatography |
| IC50 | Half-maximal inhibitory concentration |
| LC | Liquid chromatography |
| m/z | Mass-to-charge ratio |
| MS | Mass spectrometry |
| NMR | Nuclear magnetic resonance |
| NP–PEG | Natural products–polyethylene glycol |
| PDA | Photodiode array |
| QE | Quercetin equivalents |
| Rf | Retention factor |
| RT | Room temperature |
| SD | Standard deviation |
| TFC | Total flavonoid content |
| TPC | Total phenolic content |
| TPTZ | 2,4,6-Tris(2-pyridyl)-1,3,5-triazine |
| UAE | Ultrasound-assisted extraction |
| UHPLC | Ultra-high-performance liquid chromatography |
| UV | Ultraviolet |
| WWPTFE | Water wettable polytetrafluoroethylene |
References
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. (Eds). Flora Europaea. Vol. 3: Diapensiaceae to Myoporaceae, 1st ed.; Cambridge University Press: Cambridge, UK, 1972; pp. 145–147. [Google Scholar]
- Săvulescu, T. (Ed). Flora R.P.R., 1st ed.; Romanian Academy Publishing House: Bucharest, Romania, 1961; Volume VIII. (in Romanian) [Google Scholar]
- Olennikov, D.N. Synanthropic Plants as an Underestimated Source of Bioactive Phytochemicals: A Case of Galeopsis bifida (Lamiaceae). Plants 2020, 9, 1555. [Google Scholar] [CrossRef] [PubMed]
- Ciocârlan, V. Flora ilustrată a României. Pteridophyta et Spermatophyta, 3rd ed.; Ceres Publishing House: Bucharest, Romania, 2009. (in Romanian) [Google Scholar]
- Bendiksby, M.; Thorbek, L.; Scheen, A.-C.; Lindqvist, C.; Ryding, O. An updated phylogeny and classification of Lamiaceae subfamily Lamioideae. Taxon 2011, 60, 471–484. [Google Scholar] [CrossRef]
- Sârbu, I.; Ştefan, N.; Oprea, A. Plante vasculare din România. Determinator ilustrat de teren, 1st ed.; Victor B Victor Publishing House: Bucharest, Romania, 2013. (in Romanian) [Google Scholar]
- Frezza, C.; Venditti, A.; Serafini, I.; Carassiti, A.; Foddai, S.; Bianco, A.; Serafini, M. Phytochemical characteristics of Galeopsis ladanum subsp. angustifolia (Ehrh. ex Hoffm.) Gaudin collected in Abruzzo region (Central Italy) with chemotaxonomic and ethnopharmacological implications. Trends Phytochem. Res. 2017, 1, 61–68. [Google Scholar]
- Trotin, F.; Pinkas, M. Sur les polyphenols du Galeopsis ochroleuca Lam. (Labiées) [The polyphenols of Galeopsis ochroleuca Lam. (Labiatae)]. Plant. Med. Phytother. 1979, 13, 94–98. [Google Scholar]
- Gritsenko, E.N.; Litvinenko, V.I. New flavonoid compounds from Galeopsis ladanum. Chem. Nat. Compd. 1969, 5, 48–49. [Google Scholar] [CrossRef]
- Piozzi, F.; Savona, G.; Rodriguez, B.; Servettaz, O. Galangustin, a New Flavone from Galeopsis angustifolia. Heterocycles 1982, 19, 1581–1584. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Gil, M.I.; Ferreres, F.; Tomás-Lorente, F. Correlations between flavonoid composition and infrageneric taxonomy of some European Galeopsis species. Phytochemistry 1991, 30, 3311–3314. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Gil, M.I.; Ferreres, F.; Tomás-Lorente, F. Flavonoid p-coumaroylglucosides and 8-hydroxyflavone allosylglucosides in some Labiatae. Phytochemistry 1992, 31, 3097–3102. [Google Scholar] [CrossRef]
- Uriarte-Pueyo, I.; Calvo, M.I. Structure–activity relationships of acetylated flavone glycosides from Galeopsis ladanum L. (Lamiaceae). Food Chem. 2010, 120, 679–683. [Google Scholar] [CrossRef]
- Venditti, A.; Serrilli, A.M.; Bianco, A. A new flavonoid and other polar compounds from Galeopsis angustifolia Ehrh. ex Hoffm. Nat. Prod. Res. 2013, 27, 412–416. [Google Scholar] [CrossRef]
- Calis, I.; Lahloub, M.F.; Rogenmoser, E.; Sticher, O. Isomartynoside, a phenylpropanoid glycoside from Galeopsis pubescens. Phytochemistry 1984, 23, 2313–2315. [Google Scholar] [CrossRef]
- Sticher, O.; Rogenmoser, E.; Weisflog, A. Neue iridoidglucoside aus Galeopsis tetrahit L. und Galeopsis pubescens Bess. (Labiatae). Tetrahedron Lett. 1975, 16, 291–294. [Google Scholar] [CrossRef]
- Wieffering, J.H. Chromosome numbers, scutellarin and iridoid patterns in the genus Galeopsis (Labiatae). Bot. Helv. 1983, 93, 239–253. [Google Scholar]
- Frezza, C.; Venditti, A.; Giuliani, C.; Foddai, S.; Maggi, F.; Fico, G.; Bianco, A.; Serafini, M. Preliminary study on the phytochemical evolution of different Lamiaceae species based on iridoids. Biochem. Syst. Ecol. 2019, 82, 44–51. [Google Scholar] [CrossRef]
- Rodríguez, B.; Savona, G. Diterpenoids from Galeopsis angustifolia. Phytochemistry 1980, 19, 1805–1807. [Google Scholar] [CrossRef]
- Pérez-Sirvent, L.; Rodríguez, B.; Savona, G.; Servetta, O. Rearranged labdane diterpenoids from Galeopsis angustifolia. Phytochemistry 1983, 22, 527–530. [Google Scholar] [CrossRef]
- Savona, G.; Bruno, M.; Servettaz, O.; Rodríguez, B. Galeuterone and pregaleuterone, labdane diterpenoids from Galeopsis reuteri. Phytochemistry 1984, 23, 2958–2959. [Google Scholar] [CrossRef]
- Flamini, G.; Cioni, P.L.; Morelli, I. Essential oils of Galeopsis pubescens and G. tetrahit from Tuscany (Italy). Flavour Fragr. J. 2004, 19, 327–329. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Dudareva, L.V.; Tankhaeva, L.M. Chemical composition of essential oils from Galeopsis bifida and Phlomoides tuberosa. Chem. Nat. Compd. 2010, 46, 316–318. [Google Scholar] [CrossRef]
- Gusakova, S.D.; Vinokurov, I.I.; Umarov, A.U. Epoxy and hydroxy acids of the seed oil of Galeopsis bifida. Chem. Nat. Compd. 1981, 17, 217–223. [Google Scholar] [CrossRef]
- Khomova, T.V.; Gusakova, S.D.; Umarov, A.U. Structure of the triacyl- and epoxyacyldiacylglycerols of the seeds of Galeopsis bifida. Chem. Nat. Compd. 1983, 19, 225–226. [Google Scholar] [CrossRef]
- Gusakova, S.D.; Khomova, T.V. New oxo acids of the seed oil of Galeopsis bifida. Chem. Nat. Compd. 1984, 20, 266–270. [Google Scholar] [CrossRef]
- Asilbekova, D.T.; Gusakova, S.D.; Moiseeva, G.P.; Glushenkova, A.I. New epoxy acids of Galeopsis bifida. Chem. Nat. Compd. 1987, 23, 186–192. [Google Scholar] [CrossRef]
- Gusakova, S.D.; Asilbekova, D.T. Hydroxy acids of the reserve lipids of Galeopsis bifida. Chem. Nat. Compd. 1991, 27, 655–663. [Google Scholar] [CrossRef]
- Czarnecki, R.; Librowski, T.; Zélbala, K.; Kohlmünzer, S. Pharmacological properties of a lyophilizate from Galeopsis ladanum on the central nervous system of rodents. Phytother. Res. 1993, 7, 9–12. [Google Scholar] [CrossRef]
- Uriarte-Pueyo, I.; Calvo, M.I. Phytochemical Study and Evaluation of Antioxidant, Neuroprotective and Acetylcholinesterase Inhibitor Activities of Galeopsis ladanum L. Extracts. Pharmacogn. Mag. 2009, 5, 287–290. [Google Scholar] [CrossRef]
- Matkowski, A.; Piotrowska, M. Antioxidant and free radical scavenging activities of some medicinal plants from the Lamiaceae. Fitoterapia 2006, 77, 346–353. [Google Scholar] [CrossRef]
- Matkowski, A.; Tasarz, P.; Szypuła, E. Antioxidant activity of herb extracts from five medicinal plants from Lamiaceae, subfamily Lamioideae. J. Med. Plants Res. 2008, 2, 321–330. [Google Scholar]
- Pinto, D.; Giuliani, G.; Marzani, B. 209 Galeopsis segetum Necker extracts for the prevention and treatment of hair loss. J. Invest. Dermatol. 2016, 136, S196. [Google Scholar] [CrossRef]
- Uriarte-Pueyo, I.; Goicoechea, M.; Gil, A.G.; López de Cerain, A.; López de Munain, A.; Calvo, M.I. Negative Evidence for Stachydrine or Galeopsis ladanum L. Seeds as the Causal Agents of Coturnism after Quail Meat Ingestion. J. Agric. Food Chem. 2009, 57, 11055–11059. [Google Scholar] [CrossRef]
- Shraim, A.M.; Ahmed, T.A.; Rahman, M.M.; Hijji, Y.M. Determination of total flavonoid content by aluminum chloride assay: A critical evaluation. LWT 2021, 150, 111932. [Google Scholar] [CrossRef]
- Sembiring, E.N.; Elya, B.; Sauriasari, R. Phytochemical Screening, Total Flavonoid and Total Phenolic Content and Antioxidant Activity of Different Parts of Caesalpinia bonduc (L.) Roxb. Phcog. J. 2018, 10, 123–127. [Google Scholar] [CrossRef]
- Mogoşanu, G.D.; Buteică, S.A.; Purcaru, Ş.O.; Croitoru, O.; Georgescu, A.M.; Serban, F.; Tătăranu, L.G.; Alexandru, O.; Dricu, A. Rationale and in vitro efficacy of Ligustrum vulgare hydroalcoholic extract for the treatment of brain tumors. Int. J. Clin. Exp. Pathol. 2016, 9, 8286–8296. [Google Scholar]
- Bejenaru, C.; Segneanu, A.-E.; Bejenaru, L.E.; Biţă, A.; Radu, A.; Mogoşanu, G.D.; Ciocîlteu, M.V.; Manda, C.V. Phenolic Acid Profile and In Vitro Antioxidant and Anticholinesterase Activities of Romanian Wild-Grown Acer spp. (Sapindaceae). Appl. Sci. 2025, 15, 1235. [Google Scholar] [CrossRef]
- Morlock, G.E.; Heil, J.; Bardot, V.; Lenoir, L.; Cotte, C.; Dubourdeaux, M. Effect-Directed Profiling of 17 Different Fortified Plant Extracts by High-Performance Thin-Layer Chromatography Combined with Six Planar Assays and High-Resolution Mass Spectrometry. Molecules 2021, 26, 1468. [Google Scholar] [CrossRef]
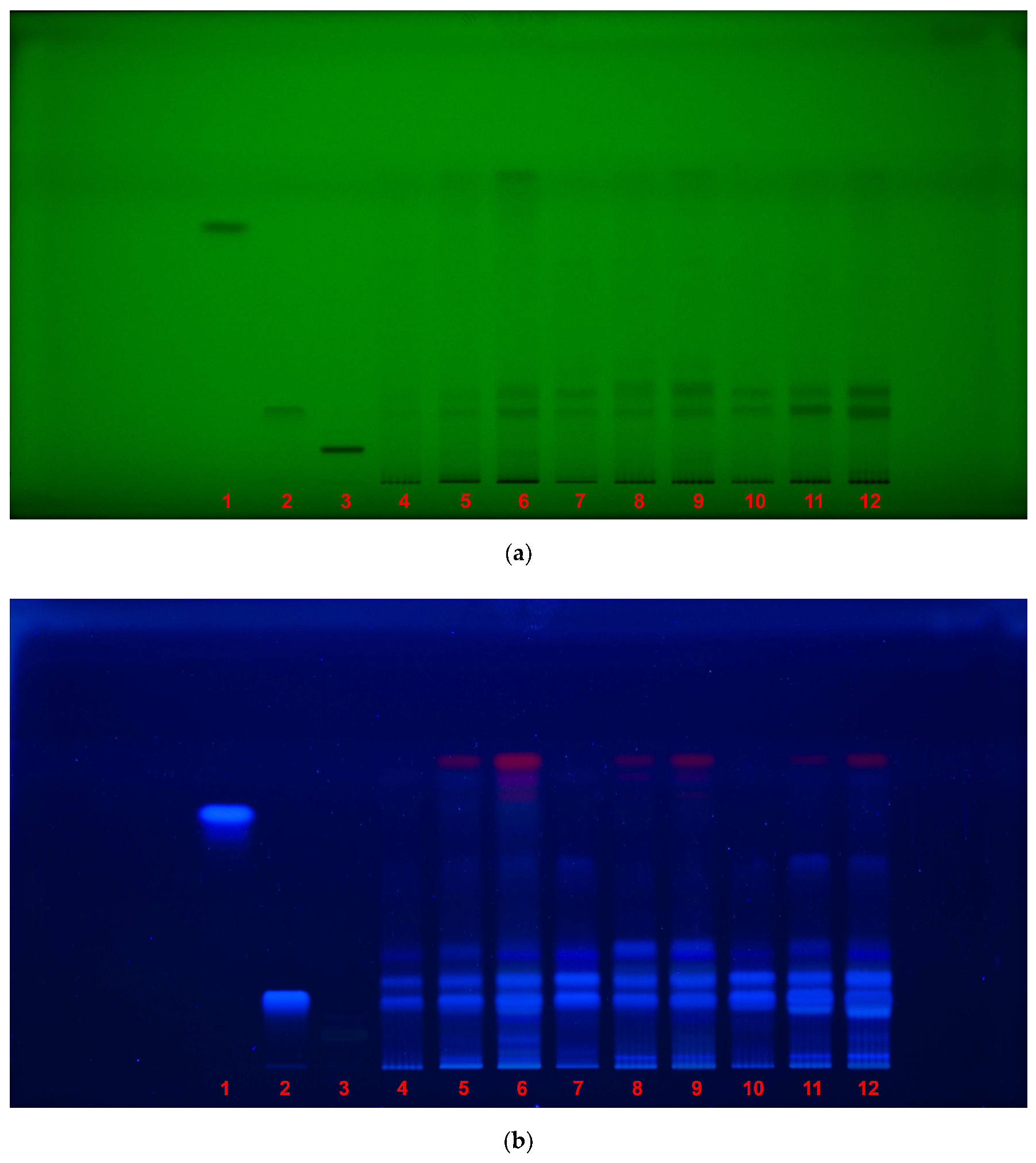
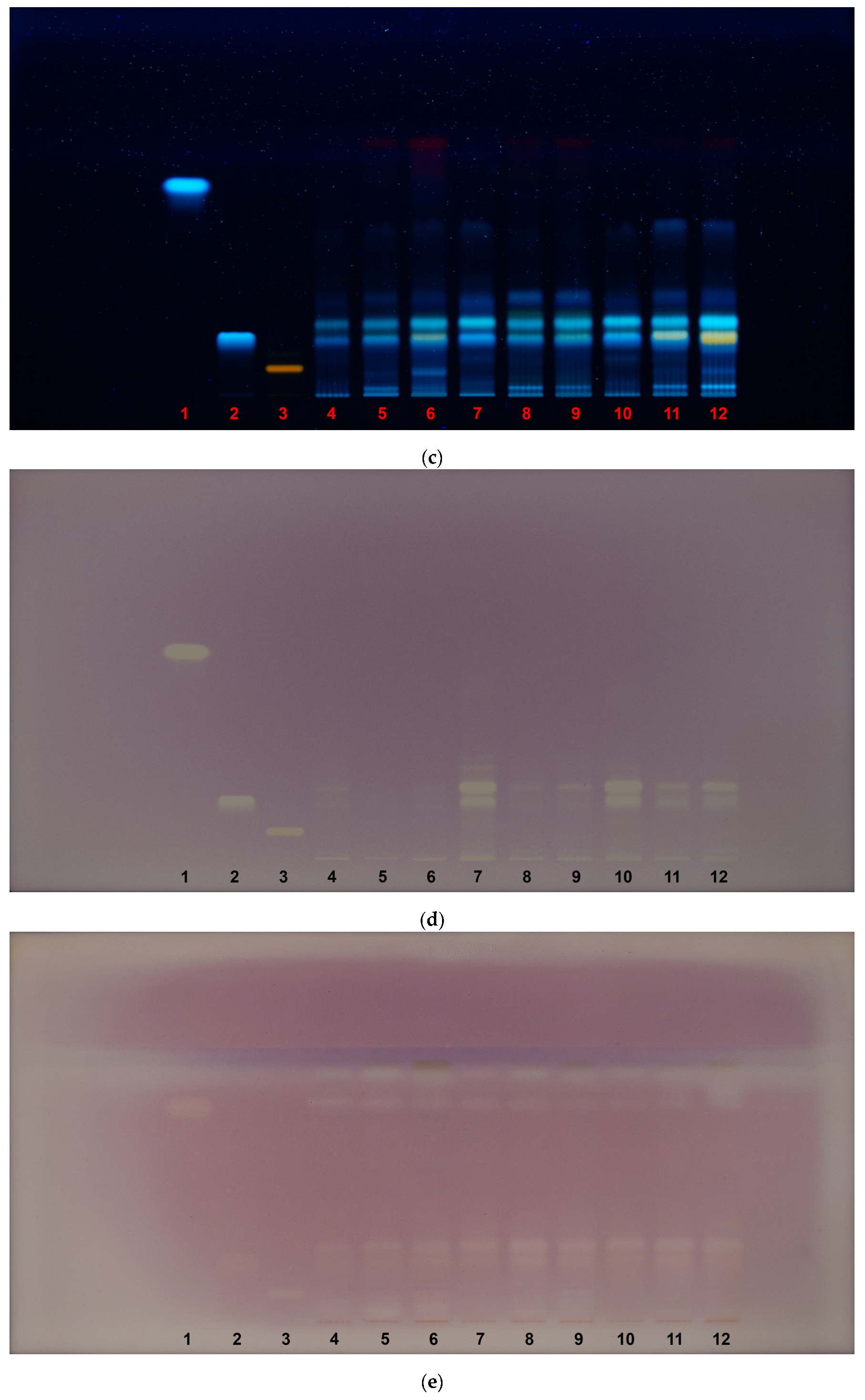
| HPTLC fingerprint | Description |
|---|---|
| 254 nm UV light, without derivatization |
• under shortwave UV light, dark bands indicate the presence of UV-absorbing compounds, such as phenolic acids and flavonoids; • caffeic acid (Rf 0.79) was not detected in any of the samples, confirming its absence or presence at undetectable concentrations (low amount demonstrated by UHPLC assay); • chlorogenic acid (Rf 0.22) was visible in all samples, confirming it as a major component of Galeopsis spp.; • rutin (Rf 0.085) was detected as dark band only as reference. |
| 366 nm UV light, without derivatization |
• under longwave UV light, compounds such as phenolic acids emit fluorescence, revealing their presence; • chlorogenic acid (Rf 0.22) was again observed in all samples, confirming its stability and prevalence across species; • rutin (Rf 0.085) was not visible under this condition, indicating that it does not fluoresce strongly without derivatization. |
| 366 nm UV light, derivatization with NP–PEG reagent |
• NP–PEG derivatization enhances flavonoid fluorescence (orange/yellow), allowing for their clearer visualization; • rutin (Rf 0.085) became visible after derivatization, confirming that its detection requires NP-PEG treatment; • strong flavonoid fluorescence was observed in G. tetrahit leaves, with a unique, orange-colored band that was absent in other species and plant parts, but not at the same Rf as rutin. |
| antioxidant activity (DPPH assay, white light) |
• DPPH assay was used to detect antioxidant activity, where active compounds appear as yellow bands against a purple background, indicating free radical scavenging activity; • chlorogenic acid (Rf 0.22) correlated strongly with antioxidant activity, as yellow bands were observed at this Rf across all samples; • for the DPPH HPTLC assay, extracts from aerial parts and leaves were diluted fivefold to prevent oversaturation of the plate and ensure accurate visualization of antioxidant activity. |
| neuroprotective activity (AChE inhibition assay, white light) |
• AChE inhibition assay was used to detect neuroprotective compounds, where active inhibitors appeared as clear bands against a purple background; • chlorogenic acid (Rf 0.22) demonstrated visible AChE inhibition in all samples, suggesting that it may contribute to the neuroprotective effects observed in Galeopsis spp.; • a distinct inhibition zone appeared at Rf 0.79, the same Rf as caffeic acid; however, since caffeic acid was not detected in the chemical fingerprinting, this suggests the presence of another compound with neuroprotective properties that migrates similarly; • slightly stronger inhibition zones were observed in G. tetrahit leaves, further reinforcing that this plant part contains potent neuroprotective compounds. |
| Sample | Species/Vegetal Product | Date/Site of Collection (Southwest Romania Flora) | Voucher Specimen |
|---|---|---|---|
| Gb_1 | G. bifida/radix | 19 August 2024/Tismana City, Gorj County | GAL-BIF-2024-0819-2 |
| Gb_2 | G. bifida/herba | 19 August 2024/Tismana City, Gorj County | GAL-BIF-2024-0819-2 |
| Gb_3 | G. bifida/folium | 19 August 2024/Tismana City, Gorj County | GAL-BIF-2024-0819-2 |
| Gs_1 | G. speciosa/radix | 19 August 2024/Tismana City, Gorj County | GAL-SPC-2024-0819-2 |
| Gs_2 | G. speciosa/herba | 19 August 2024/Tismana City, Gorj County | GAL-SPC-2024-0819-2 |
| Gs_3 | G. speciosa/folium | 19 August 2024/Tismana City, Gorj County | GAL-SPC-2024-0819-2 |
| Gt_1 | G. tetrahit/radix | 21 July 2024/Lăpuşnicel Village, Caraş Severin County | GAL-TTH-2024-0721-2 |
| Gt_2 | G. tetrahit/herba | 21 July 2024/Lăpuşnicel Village, Caraş Severin County | GAL-TTH-2024-0721-2 |
| Gt_3 | G. tetrahit/folium | 21 July 2024/Lăpuşnicel Village, Caraş Severin County | GAL-TTH-2024-0721-2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




