Submitted:
14 March 2025
Posted:
18 March 2025
Read the latest preprint version here
Abstract
Keywords:
Biomolecular Condensates
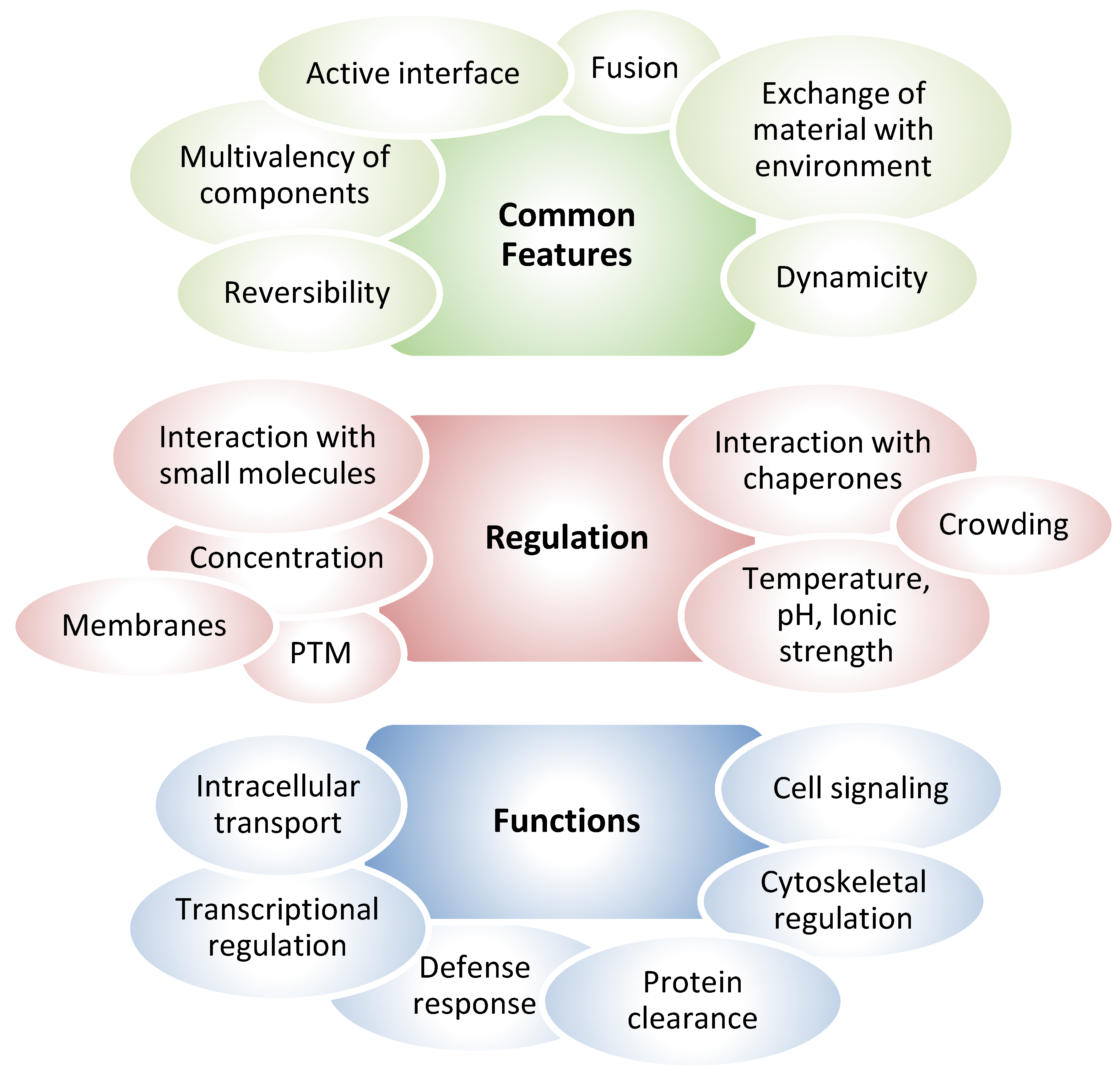
Formation of Protein Condensates. Regulation by Posttranslational Modifications with Focus on Redox Processes
Vimentin Filament Structure and Assembly
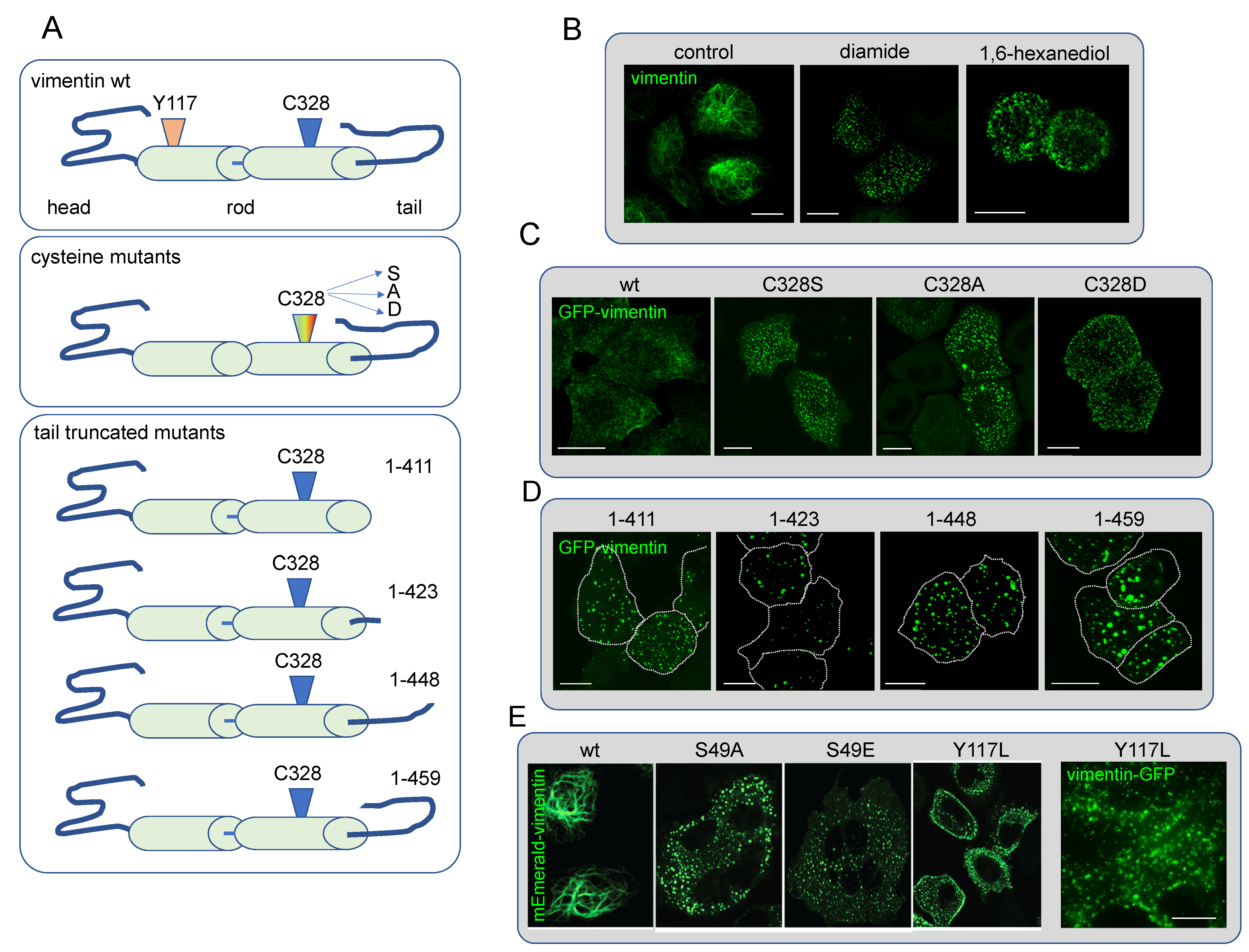
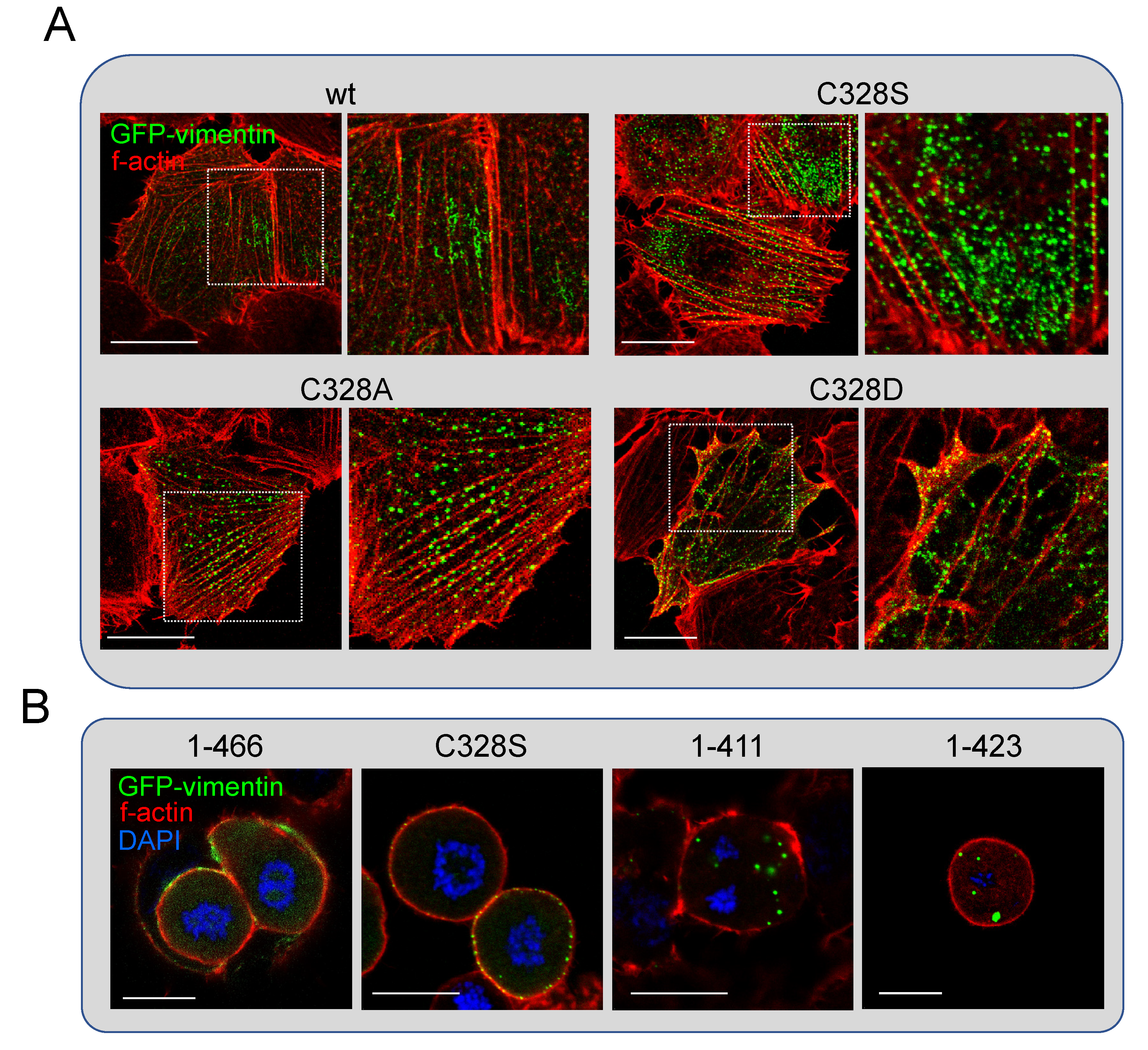
Posttranslational Modifications of Vimentin. Critical Residues
Vimentin Has Been Detected in a Variety of Assemblies Reminiscent of Biomolecular Condensates
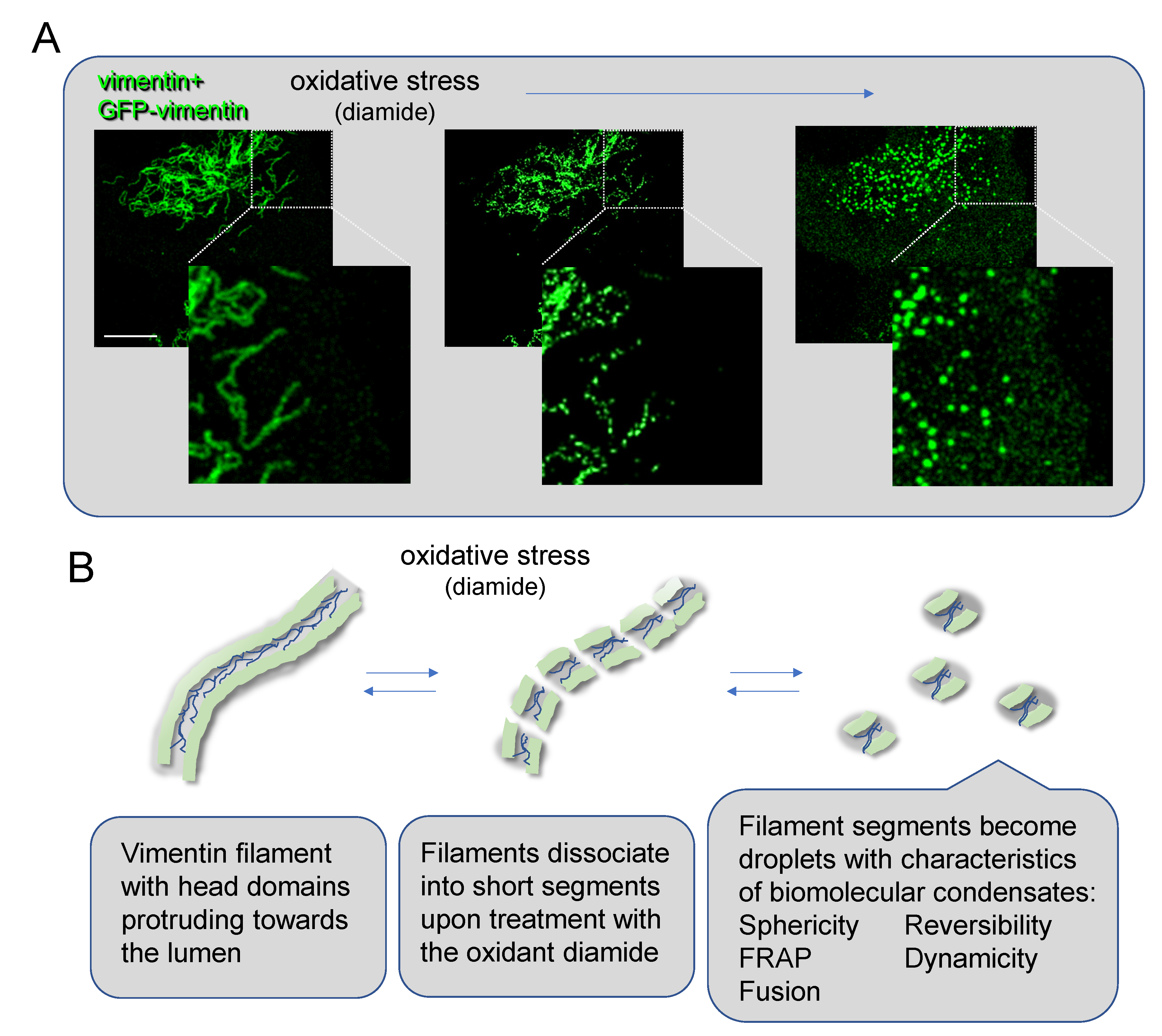
Oxidative Stress Induces Vimentin Condensates
Concluding Remarks and Perspectives
Funding
Acknowledgements
References
- H. Herrmann, S.V. Strelkov, P. Burkhard, U. Aebi, Intermediate filaments: primary determinants of cell architecture and plasticity, J Clin Invest 119(7) (2009) 1772-83.
- D.M. Toivola, P. Strnad, A. Habtezion, M.B. Omary, Intermediate filaments take the heat as stress proteins, Trends Cell Biol 20(2) (2010) 79-91.
- D. Pérez-Sala, R. Quinlan, The redox-responsive roles of intermediate filaments in cellular stress detection, integration and mitigation, Curr Opin Cell Biol 86 (2024) 102283.
- S. Etienne-Manneville, Cytoplasmic Intermediate Filaments in Cell Biology, Annu Rev Cell Dev Biol 34 (2018) 1-28.
- C.J. Hutchison, M. Alvarez-Reyes, O.A. Vaughan, Lamins in disease: why do ubiquitously expressed nuclear envelope proteins give rise to tissue-specific disease phenotypes?, J Cell Sci 114(Pt 1) (2001) 9-19.
- H.J. Worman, G. Bonne, "Laminopathies": a wide spectrum of human diseases, Exp Cell Res 313(10) (2007) 2121-33.
- J.E. Eriksson, T. Dechat, B. Grin, B. Helfand, M. Mendez, H.M. Pallari, R.D. Goldman, Introducing intermediate filaments: from discovery to disease, J Clin Invest 119(7) (2009) 1763-71.
- E.M. Hol, Y. Capetanaki, Type III Intermediate Filaments Desmin, Glial Fibrillary Acidic Protein (GFAP), Vimentin, and Peripherin, Cold Spring Harbor perspectives in biology 9(12) (2017) a021642.
- A. Messing, M. Brenner, GFAP at 50, ASN neuro 12 (2020) 1759091420949680.
- H. Herrmann, U. Aebi, Intermediate Filaments: Structure and Assembly, Cold Spring Harbor perspectives in biology 8(11) (2016) a018242.
- F. Nunes Vicente, M. Lelek, J.Y. Tinevez, Q.D. Tran, G. Pehau-Arnaudet, C. Zimmer, S. Etienne-Manneville, G. Giannone, C. Leduc, Molecular organization and mechanics of single vimentin filaments revealed by super-resolution imaging, Sci Adv 8(8) (2022) eabm2696.
- M. Eibauer, M.S. Weber, R. Kronenberg-Tenga, C.T. Beales, R. Boujemaa-Paterski, Y. Turgay, S. Sivagurunathan, J. Kraxner, S. Koster, R.D. Goldman, O. Medalia, Vimentin filaments integrate low-complexity domains in a complex helical structure, Nat Struct Mol Biol 31 (2024) 939–949.
- Y.H. Chou, F.W. Flitney, L. Chang, M. Mendez, B. Grin, R.D. Goldman, The motility and dynamic properties of intermediate filaments and their constituent proteins, Exp Cell Res 313(10) (2007) 2236-43.
- Stamatakis, C.L. Oeste, D. Perez-Sala, Vimentin as a Multifaceted Player and Potential Therapeutic Target in Viral Infections, International journal of molecular sciences 21(13) (2020) 4675.
- P. González-Jiménez, S. Duarte, A. Martínez-Fernández, E. Navarro-Carrasco, V. Lalioti, M.A. Pajares, D. Pérez-Sala, Vimentin single cysteine residue acts as a tunable sensor for network organization and as a key for actin remodeling in response to oxidants and electrophiles, Redox biology 64 (2023) 102756.
- S. Duarte, A. Viedma-Poyatos, E. Navarro-Carrasco, A.E. Martinez, M.A. Pajares, D. Perez-Sala, Vimentin filaments interact with the actin cortex in mitosis allowing normal cell division, Nature communications 10 (2019) 4200.
- Y.H. Chou, S. Khuon, H. Herrmann, R.D. Goldman, Nestin promotes the phosphorylation-dependent disassembly of vimentin intermediate filaments during mitosis, Mol Biol Cell 14(4) (2003) 1468-78.
- B.T. Helfand, M.G. Mendez, S.N. Murthy, D.K. Shumaker, B. Grin, S. Mahammad, U. Aebi, T. Wedig, Y.I. Wu, K.M. Hahn, M. Inagaki, H. Herrmann, R.D. Goldman, Vimentin organization modulates the formation of lamellipodia, Mol Biol Cell 22(8) (2011) 1274-89.
- M.E. Kidd, D.K. Shumaker, K.M. Ridge, The role of vimentin intermediate filaments in the progression of lung cancer, Am J Respir Cell Mol Biol 50(1) (2014) 1-6.
- D. Pérez-Sala, C.L. Oeste, A.E. Martínez, B. Garzón, M.J. Carrasco, F.J. Cañada, Vimentin filament organization and stress sensing depend on its single cysteine residue and zinc binding, Nature communications 6 (2015) 7287.
- A. Mónico, S. Duarte, M.A. Pajares, D. Pérez-Sala, Vimentin disruption by lipoxidation and electrophiles: role of the cysteine residue and filament dynamics, Redox biology 23 (2019) 101098.
- X. Zhou, M. Kato, S.L. McKnight, How do disordered head domains assist in the assembly of intermediate filaments?, Curr Opin Cell Biol 85 (2023) 102262.
- P. Martínez-Cenalmor, A.E. Martínez, D. Moneo-Corcuera, P. González-Jiménez, D. Pérez-Sala, Oxidative stress elicits the remodeling of vimentin filaments into biomolecular condensates, Redox biology 75 (2024) 103282.
- S.F. Banani, H.O. Lee, A.A. Hyman, M.K. Rosen, Biomolecular condensates: organizers of cellular biochemistry, Nat Rev Mol Cell Biol 18(5) (2017) 285-298.
- Y. Shin, C.P. Brangwynne, Liquid phase condensation in cell physiology and disease, Science 357(6357) (2017) eaaf4382.
- S. Alberti, Phase separation in biology, Curr Biol 27(20) (2017) R1097-R1102.
- J.A. Ditlev, L.B. Case, M.K. Rosen, Who's In and Who's Out-Compositional Control of Biomolecular Condensates, J Mol Biol 430(23) (2018) 4666-4684.
- G. Rivas, A.P. Minton, Influence of Nonspecific Interactions on Protein Associations: Implications for Biochemistry In Vivo, Annu Rev Biochem 91 (2022) 321-351.
- B. Monterroso, W. Margolin, A.J. Boersma, G. Rivas, B. Poolman, S. Zorrilla, Macromolecular Crowding, Phase Separation, and Homeostasis in the Orchestration of Bacterial Cellular Functions, Chem Rev 124(4) (2024) 1899-1949.
- W.T. Snead, A.S. Gladfelter, The Control Centers of Biomolecular Phase Separation: How Membrane Surfaces, PTMs, and Active Processes Regulate Condensation, Mol Cell 76(2) (2019) 295-305.
- G. Rivas, A.P. Minton, Surfaces as frameworks for intracellular organization, Trends Biochem Sci 49(11) (2024) 942-954.
- S. Alberti, A.A. Hyman, Biomolecular condensates at the nexus of cellular stress, protein aggregation disease and ageing, Nat Rev Mol Cell Biol 22(3) (2021) 196-213.
- S. Alberti, A. Gladfelter, T. Mittag, Considerations and Challenges in Studying Liquid-Liquid Phase Separation and Biomolecular Condensates, Cell 176(3) (2019) 419-434.
- D.M. Mitrea, B. Chandra, M.C. Ferrolino, E.B. Gibbs, M. Tolbert, M.R. White, R.W. Kriwacki, Methods for Physical Characterization of Phase-Separated Bodies and Membrane-less Organelles, J Mol Biol 430(23) (2018) 4773-4805.
- S. Kroschwald, S. Maharana, S. Alberti, Hexanediol: a chemical probe to investigate the material properties of membrane-less compartments, Matters (2017) 1-8.
- R. Duster, I.H. Kaltheuner, M. Schmitz, M. Geyer, 1,6-Hexanediol, commonly used to dissolve liquid-liquid phase separated condensates, directly impairs kinase and phosphatase activities, J Biol Chem 296 (2021) 100260.
- B. Monterroso, M.A. Robles-Ramos, S. Zorrilla, G. Rivas, Reconstituting bacterial cell division assemblies in crowded, phase-separated media, Methods Enzymol 646 (2021) 19-49.
- S. Alberti, D. Dormann, Liquid-Liquid Phase Separation in Disease, Annu Rev Genet 53 (2019) 171-194.
- S. Mohapatra, S. Wegmann, Biomolecular condensation involving the cytoskeleton, Brain Res Bull 194 (2023) 105-117.
- B. Monterroso, S. Zorrilla, M. Sobrinos-Sanguino, M.A. Robles-Ramos, M. Lopez-Alvarez, W. Margolin, C.D. Keating, G. Rivas, Bacterial FtsZ protein forms phase-separated condensates with its nucleoid-associated inhibitor SlmA, EMBO Rep 20(1) (2019) e45946.
- T. Wiegand, A.A. Hyman, Drops and fibers - how biomolecular condensates and cytoskeletal filaments influence each other, Emerg Top Life Sci 4(3) (2020) 247-261.
- J.B. Woodruff, B. Ferreira Gomes, P.O. Widlund, J. Mahamid, A. Honigmann, A.A. Hyman, The Centrosome Is a Selective Condensate that Nucleates Microtubules by Concentrating Tubulin, Cell 169(6) (2017) 1066-1077 e10.
- Y. Lin, E. Mori, M. Kato, S. Xiang, L. Wu, I. Kwon, S.L. McKnight, Toxic PR Poly-Dipeptides Encoded by the C9orf72 Repeat Expansion Target LC Domain Polymers, Cell 167(3) (2016) 789-802 e12.
- M.A. Robles-Ramos, S. Zorrilla, C. Alfonso, W. Margolin, G. Rivas, B. Monterroso, Assembly of bacterial cell division protein FtsZ into dynamic biomolecular condensates, Biochim Biophys Acta Mol Cell Res 1868(5) (2021) 118986.
- B. Guilhas, J.C. Walter, J. Rech, G. David, N.O. Walliser, J. Palmeri, C. Mathieu-Demaziere, A. Parmeggiani, J.Y. Bouet, A. Le Gall, M. Nollmann, ATP-Driven Separation of Liquid Phase Condensates in Bacteria, Mol Cell 79(2) (2020) 293-303 e4.
- S. Keyport Kik, D. Christopher, H. Glauninger, C.W. Hickernell, J.A.M. Bard, K.M. Lin, A.H. Squires, M. Ford, T.R. Sosnick, D.A. Drummond, An adaptive biomolecular condensation response is conserved across environmentally divergent species, Nature communications 15(1) (2024) 3127.
- S. Kroschwald, M.C. Munder, S. Maharana, T.M. Franzmann, D. Richter, M. Ruer, A.A. Hyman, S. Alberti, Different Material States of Pub1 Condensates Define Distinct Modes of Stress Adaptation and Recovery, Cell reports 23(11) (2018) 3327-3339.
- J. Li, M. Zhang, W. Ma, B. Yang, H. Lu, F. Zhou, L. Zhang, Post-translational modifications in liquid-liquid phase separation: a comprehensive review, Mol Biomed 3(1) (2022) 13.
- H.J. Choi, J.Y. Lee, K. Kim, Glutathionylation on RNA-binding proteins: a regulator of liquid‒liquid phase separation in the pathogenesis of amyotrophic lateral sclerosis, Experimental & molecular medicine 55(4) (2023) 735-744.
- H. Sies, D.P. Jones, Reactive oxygen species (ROS) as pleiotropic physiological signalling agents, Nat Rev Mol Cell Biol 21(7) (2020) 363-383.
- H. Sies, R.J. Mailloux, U. Jakob, Fundamentals of redox regulation in biology, Nat Rev Mol Cell Biol 25(9) (2024) 701-719.
- M.J. Davies, Protein oxidation and peroxidation, Biochem J 473(7) (2016) 805-25.
- X. Huang, S. Chen, W. Li, L. Tang, Y. Zhang, N. Yang, Y. Zou, X. Zhai, N. Xiao, W. Liu, P. Li, C. Xu, ROS regulated reversible protein phase separation synchronizes plant flowering, Nat Chem Biol 17(5) (2021) 549-557.
- T. Vignane, M. Hugo, C. Hoffmann, A. Katsouda, J. Petric, H. Wang, M. Miler, F. Comas, D. Petrovic, S. Chen, J.L. Miljkovic, J.L. Morris, S.R. Chowdhury, J. Prudent, N. Polovic, M.P. Murphy, A. Papapetropoulos, D. Milovanovic, M.R. Filipovic, Protein thiol alterations drive aberrant phase separation in aging, bioRxiv (2023) 10.1101/2023.11.07.566021.
- E. Fuentes-Lemus, J.S. Reyes, L.F. Gamon, C. Lopez-Alarcon, M.J. Davies, Effect of macromolecular crowding on protein oxidation: Consequences on the rate, extent and oxidation pathways, Redox biology 48 (2021) 102202.
- E. Fuentes-Lemus, M.J. Davies, Effect of crowding, compartmentalization and nanodomains on protein modification and redox signaling - current state and future challenges, Free Radic Biol Med 196 (2023) 81-92.
- Y. Saito, W. Kimura, Roles of Phase Separation for Cellular Redox Maintenance, Frontiers in genetics 12 (2021) 691946.
- Y. Dai, C.F. Chamberlayne, M.S. Messina, C.J. Chang, R.N. Zare, L. You, A. Chilkoti, Interface of biomolecular condensates modulates redox reactions, Chem 9(6) (2023) 1594-1609.
- B. Macek, K. Forchhammer, J. Hardouin, E. Weber-Ban, C. Grangeasse, I. Mijakovic, Protein post-translational modifications in bacteria, Nat Rev Microbiol 17(11) (2019) 651-664.
- M. Sasazawa, D.T. Tomares, W.S. Childers, S. Saurabh, Biomolecular condensates as stress sensors and modulators of bacterial signaling, PLoS pathogens 20(8) (2024) e1012413.
- F. Heinkel, L. Abraham, M. Ko, J. Chao, H. Bach, L.T. Hui, H. Li, M. Zhu, Y.M. Ling, J.C. Rogalski, J. Scurll, J.M. Bui, T. Mayor, M.R. Gold, K.C. Chou, Y. Av-Gay, L.P. McIntosh, J. Gsponer, Phase separation and clustering of an ABC transporter in Mycobacterium tuberculosis, Proc Natl Acad Sci U S A 116(33) (2019) 16326-16331.
- A. Gupta, A. Joshi, K. Arora, S. Mukhopadhyay, P. Guptasarma, The bacterial nucleoid-associated proteins, HU and Dps, condense DNA into context-dependent biphasic or multiphasic complex coacervates, J Biol Chem 299(5) (2023) 104637.
- N. Grams, M. Charman, E. Halko, R. Lauman, B.A. Garcia, M.D. Weitzman, Phosphorylation regulates viral biomolecular condensates to promote infectious progeny production, EMBO J 43(2) (2024) 277-303.
- S. Gruenheid, B.B. Finlay, Microbial pathogenesis and cytoskeletal function, Nature 422(6933) (2003) 775-81.
- S. Zhao, C. Miao, X. Gao, Z. Li, J.E. Eriksson, Y. Jiu, Vimentin cage - A double-edged sword in host anti-infection defense, Curr Opin Cell Biol 86 (2024) 102317.
- H. Herrmann, M. Haner, M. Brettel, N.O. Ku, U. Aebi, Characterization of distinct early assembly units of different intermediate filament proteins, J Mol Biol 286(5) (1999) 1403-20.
- F. Nunes-Vicente, M. Lelek, J.-Y. Tinevez, Q.D. Tran, G. Pehau-Arnaudet, C. Zimmer, S. Etienne- Manneville, G. Giannone, C. Leduc, Molecular organization and mechanics of single vimentin filaments revealed by super-resolution imaging, bioRxiv (2021). [CrossRef]
- Q.D. Tran, M. Lenz, H. Wioland, A. Jegou, G. Romet-Lemonne, C. Leduc, Continuous self-repair protects vimentin intermediate filaments from fragmentation, bioRxiv (2024) 10.1101/2024.09.02.610785.
- M. Yoon, R.D. Moir, V. Prahlad, R. Goldman, Motile properties of vimentin intermediate filament networks in living cells, J Cell Biol 143 (1998) 147-157.
- G. Colakoglu, A. Brown, Intermediate filaments exchange subunits along their length and elongate by end-to-end annealing, J Cell Biol 185(5) (2009) 769-77.
- E. Griesser, V. Vemula, A. Mónico, D. Pérez-Sala, M. Fedorova, Dynamic posttranslational modifications of cytoskeletal proteins unveil hot spots under nitroxidative stress, Redox biology 44 (2021) 102014.
- H.J. Tarbet, L. Dolat, T.J. Smith, B.M. Condon, E.T. O'Brien, 3rd, R.H. Valdivia, M. Boyce, Site-specific glycosylation regulates the form and function of the intermediate filament cytoskeleton, eLife 7 (2018) e31807.
- González-Jiménez, S. Duarte, M.A. Pajares, D. Pérez-Sala, Vimentin tail segments are differentially exposed at distinct cellular locations and in response to stress, Frontiers in cell and developmental biology 10 (2022) 908263.
- M. Kaus-Drobek, N. Mucke, R.H. Szczepanowski, T. Wedig, M. Czarnocki-Cieciura, M. Polakowska, H. Herrmann, A. Wyslouch-Cieszynska, M. Dadlez, Vimentin S-glutathionylation at Cys328 inhibits filament elongation and induces severing of mature filaments in vitro, FEBS J 287 (2020) 5304-5322.
- Guzman-Caldentey, M.A. Pajares, S. Martin-Santamaria, D. Pérez-Sala, Molecular Insight into the Regulation of Vimentin by Cysteine Modifications and Zinc Binding, Antioxidants (Basel) 10(7) (2021) 1039.
- Zorrilla, G. Rivas, D. Pérez-Sala, Zinc Differentially Modulates the Assembly of Soluble and Polymerized Vimentin, International journal of molecular sciences 21(7) (2020) 2426.
- C.Y. Yang, P.W. Chang, W.H. Hsu, H.C. Chang, C.L. Chen, C.C. Lai, W.T. Chiu, H.C. Chen, Src and SHP2 coordinately regulate the dynamics and organization of vimentin filaments during cell migration, Oncogene 38(21) (2019) 4075-4094.
- M. Meier, G.P. Padilla, H. Herrmann, T. Wedig, M. Hergt, T.R. Patel, J. Stetefeld, U. Aebi, P. Burkhard, Vimentin coil 1A-A molecular switch involved in the initiation of filament elongation, J Mol Biol 390(2) (2009) 245-61.
- Herrmann, M.W. Davidson, V.I. Gelfand, Microtubule-dependent transport of vimentin filament precursors is regulated by actin and by the concerted action of Rho- and p21-activated kinases, FASEB J 28(7) (2014) 2879-90.
- V. Prahlad, M. Yoon, R.D. Moir, R.D. Vale, R.D. Goldman, Rapid movements of vimentin on microtubule tracks: kinesin-dependent assembly of intermediate filament networks, J Cell Biol 143(1) (1998) 159-70.
- W.W. Franke, E. Schmid, C. Grund, B. Geiger, Intermediate filament proteins in nonfilamentous structures: transient disintegration and inclusion of subunit proteins in granular aggregates, Cell 30(1) (1982) 103-13.
- S. Sivagurunathan, A. Vahabikashi, H. Yang, J. Zhang, K. Vazquez, D. Rajasundaram, Y. Politanska, H. Abdala-Valencia, J. Notbohm, M. Guo, S.A. Adam, R.D. Goldman, Expression of vimentin alters cell mechanics, cell-cell adhesion, and gene expression profiles suggesting the induction of a hybrid EMT in human mammary epithelial cells, Frontiers in cell and developmental biology 10 (2022) 929495.
- A. Basu, T. Krug, B.d. Pont, Q. Huang, S. Sun, S.A. Adam, R. Goldman, D.A. Weitz, Phase separated liquid vimentin droplets stabilize actin fibers through wetting, bioRxiv (2024). [CrossRef]
- Rossow, C. Hookway, S.A. Adam, V.I. Gelfand, Vimentin filament precursors exchange subunits in an ATP-dependent manner, Proc Natl Acad Sci U S A 112(27) (2015) E3505-14.
- D. Moneo-Corcuera, A. Viedma-Poyatos, K. Stamatakis, D. Pérez-Sala, Desmin Reorganization by Stimuli Inducing Oxidative Stress and Electrophiles: Role of Its Single Cysteine Residue, Antioxidants (Basel) 12(9) (2023) 1703.
- M. Matsuyama, H. Tanaka, A. Inoko, H. Goto, S. Yonemura, K. Kobori, Y. Hayashi, E. Kondo, S. Itohara, I. Izawa, M. Inagaki, Defect of mitotic vimentin phosphorylation causes microophthalmia and cataract via aneuploidy and senescence in lens epithelial cells, J Biol Chem 288(50) (2013) 35626-35.
- N.H. Lin, Y.S. Huang, P. Opal, R.D. Goldman, A. Messing, M.D. Perng, The role of gigaxonin in the degradation of the glial-specific intermediate filament protein GFAP, Mol Biol Cell 27(25) (2016) 3980-3990.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



