Submitted:
14 March 2025
Posted:
14 March 2025
You are already at the latest version
Abstract
This comprehensive review explores the critical role of microbial adaptation in enhancing food safety by responding to diverse environmental stressors—an essential aspect of microbial ecology with profound implications for biotechnology, environmental management, and public health. We examine the intricate mechanisms underlying microbial adaptation, including genetic modifications such as mutation and horizontal gene transfer, as well as phenotypic plasticity and epigenetic regulation, which enable microorganisms to thrive under adverse conditions. Case studies illustrate microbial resilience in extreme environments, shedding light on their sophisticated adaptive strategies. Additionally, we discuss the practical applications of microbial adaptation in biotechnological domains, including bioremediation, industrial processes, and its emerging contributions to drug development. By addressing future research directions and challenges, this review underscores the necessity of advancing our understanding of microbial-environment interactions to inform innovative strategies for food safety and broader scientific applications.
Keywords:
Highlights:
- A.
- Introduction to Microbial Adaptation:
- A.
- Microorganisms dynamically adjust to environmental changes.
- A.
- Adaptability seen across bacteria, fungi, viruses, and protists.
- B.
- Factors Driving Microbial Adaptation:
- B.
- Various stressors including temperature, osmotic stress, pH changes, toxins, and pollutants.
- B.
- Dependency on nutrient availability.
- B.
- Host-associated adaptation and genetic variation.
- C.
- Implications for Biotechnology and Environmental Management:
- C.
- Applications in bioremediation, industrial fermentation, and bioprospecting.
- C.
- Potential for drug development and precision medicine.
- D.
- Incidence Studies of Microbial Adaptation:
- D.
- Case studies on bacterial adaptation to extreme environments, anthropogenic disturbances, and engineered systems.
- D.
- Fungal and archaeal adaptation to chemical stress and osmotic stress, respectively.
- E.
- Future Directions and Challenges:
- E.
- Emerging environmental stressors like climate change and urbanization.
- E.
- Unexplored mechanisms of microbial adaptation including microbial dark matter and non-genetic determinants.
- E.
- Technological advancements for studying adaptation such as omics approaches, single-cell analysis, and CRISPR-based tools.
1. Introduction to Microbial Adaptation:
1.1. Significance of Understanding Microbial Adaptation to Environmental Stress
1.1.1. Biotechnological Applications
1.1.2. Human Health
1.1.3. Environmental Sustainability
1.1.4. Climate Change
2. Factors Driving Microbial Adaptation
2.1. Natural and Synthetic Microbial Stressors
2.1.1. Temperature Variations
2.1.2. Osmotic Stress
2.1.4. Toxins and Pollutants
2.2. Dependancy on Nutrient Availability
2.2.1. Chemical Stressors
2.3. Host-Associated Adaptation:
2.4. Genetic Variation and Evolution
2.4.1. Mutation and Genetic Diversity
2.4.2. Horizontal Gene Transfer (HGT)
2.4.3. Selection Pressures and Evolutionary Dynamics
2.5. Ecological Interactions
2.5.1. Microbial Interactions and Community Dynamics
2.5.2. Host-Microbe Interactions
2.6. Anthropogenic Influences
2.6.1. Environmental Perturbations and Anthropogenic Stressors
2.6.2. Biotechnological Applications and Engineered Microbes
2.6.3. Mechanisms of Microbial Adaptation
3. Implications for Biotechnology and Environmental Management:
3.1. Bioremediation Strategies
3.1.1. Adaptive Microbial Syndicates
3.1.2. Engineered Biodegradation Pathways
3.1.3. In Situ Bioremediation Technologies
3.2. Industrial Applications of Stress-Resistant Microorganisms
3.2.1. Industrial Fermentation
3.2.2. Bioprocessing Technologies
3.2.3. Biopolymer Production
3.3. Potential for Drug Development
3.3.1. Antibiotic Discovery
3.3.2. Targeting Virulence Factors
3.3.3. Precision Antimicrobial Therapy
4. Future Directions and Challenges
4.1. Emerging Environmental Stressors
4.1.1. Climate Change-Induced Stressors
4.1.2. Pollutant and Contaminant Dynamics
4.1.3. Urbanization and Habitat Fragmentation
4.2. Unexplored Mechanisms of Microbial Adaptation
4.2.1. Microbial Dark Matter
4.2.2. Microbial Interactions and Community Dynamics
4.2.3. Non-Genetic Determinants of Adaptation
4.3. Technological Advancements for Studying Adaptation
4.3.1. Omics Approaches
4.3.2. Single-Cell Analysis
4.3.3. Machine Learning and Predictive Modeling
4.3.4. CRISPR-Based Tools for Genetic Manipulation
4.3.5. Microfluidics and High-Throughput Cultivation
5. Conclusion
5.1. Recap of Key Findings
5.1.1. Adaptability of Microorganisms
5.1.2. Mechanisms of Microbial Adaptation
5.1.3. Implications for Biotechnology and Environmental Management
5.1.4. Applications in Bioremediation and Industry
5.2. Summary of Implications
5.2.1. Biotechnological Advancements
5.2.2. Human Health and Disease Management
5.2.3. Environmental Sustainability
5.3. Suggestions for Further Research
5.3.1. Emerging Environmental Stressors
5.3.2. Host-Microbe Interactions
5.3.3. Molecular Mechanisms of Adaptation
5.3.4. Integrative Approaches
Conflict of Interests
Acknowledgement
References
- Abdullah-Al-Mahin, S.S.; Higashi, C.; Matsumoto, S.; Sonomoto, K. Improvement of multiple-stress tolerance and lactic acid production in lactococcus lactis NZ9000 under conditions of thermal stress by heterologous expression of escherichia coli dnaK. Applied and Environmental Microbiology 2010, 76, 4277–4285. [Google Scholar] [PubMed]
- Abee, T.; Wouters, J.A. Microbial stress response in minimal processing. International Journal of Food Microbiology, 1999; 50, 65–91. [Google Scholar]
- Addo-Bankas, O.; Zhao, Y.; Wei, T.; Stefanakis, A. From past to present: Tracing the evolution of treatment wetlands and prospects ahead. Journal of Water Process Engineering 2024, 60. [Google Scholar] [CrossRef]
- Aggarwal, A.; Frey, H.; McDowell, G.; Drenkhan, F.; Nusser, M.; Racoviteanu, A.; Hoelzle, M. Adaptation to climate change induced water stress in major glacierized mountain regions. Climate and Development 2022, 14, 665–677. [Google Scholar]
- Akhtar, A.A.; Turner, D.P. The role of bacterial ATP-binding cassette (ABC) transporters in pathogenesis and virulence: Therapeutic and vaccine potential. Microbial Pathogenesis 2022, 171. [Google Scholar]
- Alper, H.; Moxley, J.; Nevoigt, E.; Fink, G.R.; Stephanopoulos, G. Engineering yeast transcription machinery for improved ethanol tolerance and production. Science 2006, 314, 1565–1568. [Google Scholar] [CrossRef]
- An, S.Q.; Potnis, N.; Dow, M.; Vorholter, F.J.; He, Y.Q.; Becker, A.; Tang, J.L. Mechanistic insights into host adaptation, virulence and epidemiology of the phytopathogen xanthomonas. FEMS Microbiology Reviews 2020, 44, 1–32. [Google Scholar] [CrossRef]
- Andersson, D.I.; Balaban, N.Q.; Baquero, F.; Courvalin, P.; Glaser, P.; Gophna, U.; T?njum, T. Antibiotic resistance: Turning evolutionary principles into clinical reality. FEMS Microbiology Reviews 2020, 44, 171–188. [Google Scholar]
- Arora, S.; Rani, R.; Ghosh, S. Bioreactors in solid state fermentation technology: Design, applications and engineering aspects. Journal of Biotechnology 2018, 269, 16–34. [Google Scholar] [CrossRef]
- Ballesteros, I.; Oliva, J.M.; Ballesteros, M.; Carrasco, J. Optimization of the simultaneous saccharification and fermentation process using thermotolerant yeasts. Applied Biochemistry and Biotechnology 1993, 39, 201–211. [Google Scholar]
- Benedetti, M.; Giuliani, M.E.; Mezzelani, M.; Nardi, A.; Pittura, L.; Gorbi, S.; Regoli, F. Emerging environmental stressors and oxidative pathways in marine organisms: Current knowledge on regulation mechanisms and functional effects. Biocell 2022, 46, 37. [Google Scholar] [CrossRef]
- Bhatt, P.; Gangola, S.; Bhandari, G.; Zhang, W.; Maithani, D.; Mishra, S.; Chen, S. New insights into the degradation of synthetic pollutants in contaminated environments. Chemosphere 2021, 268. [Google Scholar]
- Bilal, M.; Ji, L.; Xu, Y.; Xu, S.; Lin, Y.; Iqbal, H.M.; Cheng, H. Bioprospecting kluyveromyces marxianus as a robust host for industrial biotechnology. Frontiers in Bioengineering and Biotechnology 2022, 10. [Google Scholar]
- Bohnert, H.J.; Nelson, D.E.; Jensen, R.G. Adaptations to environmental stresses. The Plant Cell 1995, 7, 1099. [Google Scholar] [PubMed]
- Bollenbach, T. Antimicrobial interactions: Mechanisms and implications for drug discovery and resistance evolution. Current Opinion in Microbiology 2015, 27, 1–9. [Google Scholar] [CrossRef]
- Boyce, K.J. Mutators enhance adaptive micro-evolution in pathogenic microbes. Microorganisms 2022, 10, 442. [Google Scholar] [CrossRef]
- Bremer, E.; Kramer, R. Responses of microorganisms to osmotic stress. Annual Review of Microbiology 2019, 73, 313–334. [Google Scholar]
- Cakar, Z.P.; Seker, U.O.; Tamerler, C.; Sonderegger, M.; Sauer, U. Evolutionary engineering of multiple-stress resistant saccharomyces cerevisiae. FEMS Yeast Research.
- Cho, J.S.; Kim, G.B.; Eun, H.; Moon, C.W.; Lee, S.Y. Designing microbial cell factories for the production of chemicals. Jacs Au 2022, 2, 1781–1799. [Google Scholar]
- Choi, S.H.; Baumler, D.J.; Kaspar, C.W. Contribution of dps to acid stress tolerance and oxidative stress tolerance in escherichia coli O157: H7. Applied and Environmental Microbiology 2000, 66, 3911–3916. [Google Scholar]
- Coleman, S.T.; Fang, T.K.; Rovinsky, S.A.; Turano, F.J.; Moye-Rowley, W.S. Expression of a glutamate decarboxylase homologue is required for normal oxidative stress tolerance in saccharomyces cerevisiae. Journal of Biological Chemistry 2001, 276, 244–250. [Google Scholar] [CrossRef]
- Compan, I.; Touati, D. Interaction of six global transcription regulators in expression of manganese superoxide dismutase in escherichia coli K-12. Journal of Bacteriology 1993, 175, 1687–1696. [Google Scholar]
- Cook, G.M.; Greening, C.; Hards, K.; Berney, M. Energetics of pathogenic bacteria and opportunities for drug development. Advances in Microbial Physiology 2014, 65, 1–62. [Google Scholar] [PubMed]
- Council, N. R. (1993). In situ bioremediation: When does it work? National Academies Press.
- Csonka, L.N. Physiological and genetic responses of bacteria to osmotic stress. Microbiological Reviews 1989, 53, 121–147. [Google Scholar] [PubMed]
- Donohoue, P. D. , Barrangou, R., & May, A. P. (2018a). Advances in industrial biotechnology using CRISPR-cas systems. Trends in Biotechnology, 36.
- Donohoue, P. D. , Barrangou, R., & May, A. P. (2018b). Advances in industrial biotechnology using CRISPR-cas systems. Trends in Biotechnology, 36.
- Drake, J.W. A constant rate of spontaneous mutation in DNA-based microbes. Proceedings of the National Academy of Science 1991, 88, 7160–7164. [Google Scholar]
- Elena, S.F.; Lenski, R.E. Evolution experiments with microorganisms: The dynamics and genetic bases of adaptation. Nature Reviews Genetics 2003, 4, 457–469. [Google Scholar] [PubMed]
- Feckler, A.; Goedkoop, W.; Konschak, M.; Bundschuh, R.; Kenngott, K.G.; Schulz, R.; Bundschuh, M. History matters: Heterotrophic microbial community structure and function adapt to multiple stressors. Global Change Biology 2018, 24, e402–e415. [Google Scholar]
- Frankel, N.W.; Pontius, W.; Dufour, Y.S.; Long, J.; Hernandez-Nunez, L.; Emonet, T. Adaptability of non-genetic diversity in bacterial chemotaxis. Elife 2014, 3, e03526. [Google Scholar] [CrossRef]
- Gallardo, C.; Monras, J.P.; Plaza, D.O.; Collao, B.; Saona, L.A.; Duran-Toro, V.; Perez-Donoso, J.M. Low-temperature biosynthesis of fluorescent semiconductor nanoparticles (CdS) by oxidative stress resistant antarctic bacteria. Journal of Biotechnology 2014, 187, 108–115. [Google Scholar] [CrossRef]
- Garcia-Pausas, J.; Paterson, E. Microbial community abundance and structure are determinants of soil organic matter mineralisation in the presence of labile carbon. Soil Biology and Biochemistry 2011, 43, 1705–1713. [Google Scholar]
- Gatti, M.; Pea, F. The expert clinical pharmacological advice program for tailoring on real-time antimicrobial therapies with emerging TDM candidates in special populations: How the ugly duckling turned into a swan. Expert Review of Clinical Pharmacology 2023, 16, 1035–1051. [Google Scholar] [CrossRef]
- Goel, R. Metagenomics-A tool for identification and characterization of uncultivable microbial diversity HS Tripathi. HS Tripathi.
- Grifoni 2009, M. , Franchi, E., Fusini, D., Vocciante, M., Barbafieri, M., Pedron, F.,... Petruzzelli, G. Soil remediation: Towards a resilient and adaptive approach to deal with the ever-changing environmental challenges. Environments 2022, 9, 18. [Google Scholar]
- Guan, N.; Li, J.; Shin, H.D.; Du, G.; Chen, J.; Liu, L. Metabolic engineering of acid resistance elements to improve acid resistance and propionic acid production of propionibacterium jensenii. Biotechnology and Bioengineering. 2016, 3, 1294–1304. [Google Scholar]
- Guan, N.; Liu, L.; Zhuge, X.; Xu, Q.; Li, J.; Du, G.; Chen, J. Genome shuffling improves acid tolerance of propionibacterium acidipropionici and propionic acid production. Adv Chem Res 2012, 15, 143–152. [Google Scholar]
- Guillén, S.; Nadal, L.; Álvarez, I.; Mañas, P.; Cebrián, G. Impact of the Resistance Responses to Stress Conditions Encountered in Food and Food Processing Environments on the Virulence and Growth Fitness of Non-Typhoidal Salmonellae. Foods 2021, 10, 617. [Google Scholar] [CrossRef] [PubMed]
- Hegab, H.M.; ElMekawy, A.; Stakenborg, T. Review of microfluidic microbioreactor technology for high-throughput submerged microbiological cultivation. Biomicrofluidics 2013, 7. [Google Scholar]
- Heux, S.; Meynial-Salles, I.; O'Donohue, M.J.; Dumon, C. White biotechnology: State of the art strategies for the development of biocatalysts for biorefining. Biotechnology Advances 2015, 33, 1653–1670. [Google Scholar]
- Hirasawa, T.; Yoshikawa, K.; Nakakura, Y.; Nagahisa, K.; Furusawa, C.; Katakura, Y.; Shioya, S. Identification of target genes conferring ethanol stress tolerance to saccharomyces cerevisiae based on DNA microarray data analysis. Journal of Biotechnology 2007, 131, 34–44. [Google Scholar]
- Hyde, E.D.; Seyfaee, A.; Neville, F.; Moreno-Atanasio, R. Colloidal silica particle synthesis and future industrial manufacturing pathways: A review. Industrial & Engineering Chemistry Research 2016, 55, 8891–8913. [Google Scholar]
- Joshi, S.S.; Jadhav MP, K.; Chourasia, N.K.; Jadhav MP, J.; Pathade, K.N. ; K; K Bioremediation strategies for soil and water pollution harnessing the power of microorganisms. Migration Letters 2024, 21(S8), 1–20. [Google Scholar]
- Keasling, J.; Garcia Martin, H.; Lee, T.S.; Mukhopadhyay, A.; Singer, S.W.; Sundstrom, E. Microbial production of advanced biofuels. Nature Reviews Microbiology 2021, 19, 701–715. [Google Scholar] [CrossRef]
- Li, F.; Chen, L.; Zhang, J.; Yin, J.; Huang, S. Bacterial community structure after long-term organic and inorganic fertilization reveals important associations between soil nutrients and specific taxa involved in nutrient transformations. Frontiers in Microbiology 2017, 8, 187. [Google Scholar] [CrossRef]
- Liang, G.B.; Du, G.C.; Chen, J. A novel strategy of enhanced glutathione production in high cell density cultivation of candida utilis?cysteine addition combined with dissolved oxygen controlling. Enzyme and Microbial Technology 2008, 42, 284–289. [Google Scholar] [CrossRef]
- Liang, G.; Liao, X.; Du, G.; Chen, J. Elevated glutathione production by adding precursor amino acids coupled with ATP in high cell density cultivation of candida utilis. Journal of Applied Microbiology 2008, 105, 1432–1440. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wisniewski, M.; Droby, S.; Vero, S.; Tian, S.; Hershkovitz, V. Glycine betaine improves oxidative stress tolerance and biocontrol efficacy of the antagonistic yeast cystofilobasidium infirmominiatum. International Journal of Food Microbiology 2011, 146, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Liu, S. , Ren, H., Shen, L., Lou, L., Tian, G., Zheng, P., & Hu, B. pH levels drive bacterial community structure in sediments of the qiantang river as determined by 454 pyrosequencing. Frontiers in Microbiology 2015, 6, 285. [Google Scholar]
- Losapio, G.; Genes, L.; Knight, C.J.; McFadden, T.N.; Pavan, L. Monitoring and modelling the effects of ecosystem engineers on ecosystem functioning. Functional Ecology 2024, 38, 8–21. [Google Scholar] [CrossRef]
- Lu, X.; Jiang, Y. Advancements in studying the effects of climate change on forest ecosystems. Advances in Resources Research 2023, 3, 151–177. [Google Scholar]
- Ma, R.; Zhang, Y.; Hong, H.; Lu, W.; Lin, M.; Chen, M.; Zhang, W. Improved osmotic tolerance and ethanol production of ethanologenic escherichia coli by IrrE, a global regulator of radiation-resistance of deinococcus radiodurans. Current Microbiology 2011, 62, 659–664. [Google Scholar] [CrossRef]
- Maitra, S. (2018). In situ bioremediation?an overview, Responsibility of Life Science Informatics Publications.
- Martinez, J.L.; Baquero, F. Mutation frequencies and antibiotic resistance. Antimicrobial Agents and Chemotherapy 2000, 44, 1771–1777. [Google Scholar] [CrossRef]
- Mitchell, A.; Romano, G.H.; Groisman, B.; Yona, A.; Dekel, E.; Kupiec, M.; Pilpel, Y. Adaptive prediction of environmental changes by microorganisms. Nature 2009, 460, 220–224. [Google Scholar] [CrossRef]
- Mulder, C. , Boit, A., Bonkowski, M., De Ruiter, P. C., Mancinelli, G., der Heijden, M. G., & Rutgers, M. (2011). A belowground perspective on dutch agroecosystems: How soil organisms interact to support ecosystem services. Advances in ecological research, (pp. 277-357) Academic Press.
- Nguyen, L.N.; Kumar, J.; Vu, M.T.; Mohammed, J.A.; Pathak, N.; Commault, A.S.; Nghiem, L.D. Biomethane production from anaerobic co-digestion at wastewater treatment plants: A critical review on development and innovations in biogas upgrading techniques. Science of the Total Environment 2021, 765. [Google Scholar] [CrossRef]
- Oide, S.; Gunji, W.; Moteki, Y.; Yamamoto, S.; Suda, M.; Jojima, T.; Inui, M. Thermal and solvent stress cross-tolerance conferred to corynebacterium glutamicum by adaptive laboratory evolution. Applied and Environmental Microbiology 2015, 81, 2284–2298. [Google Scholar] [PubMed]
- Olive, A.J.; Sassetti, C.M. Metabolic crosstalk between host and pathogen: Sensing, adapting and competing. Nature Reviews Microbiology 2016, 14, 221–234. [Google Scholar] [PubMed]
- Pan, J.; Wang, G.; Nong, J.; Xie, Q. Biodegradation of benzo (a) pyrene by a genetically engineered bacillus licheniformis: Degradation, metabolic pathway and toxicity analysis. Chemical Engineering Journal 2023, 478. [Google Scholar]
- Pepi, M.; Focardi, S. Antibiotic-resistant bacteria in aquaculture and climate change: A challenge for health in the mediterranean area. International Journal of Environmental Research and Public Health 2021, 18, 5723. [Google Scholar]
- Postollec, F.; Falentin, H.; Pavan, S.; Combrisson, J.; Sohier, D. Recent advances in quantitative PCR (qPCR) applications in food microbiology. Food Microbiology 2011, 28, 848–861. [Google Scholar]
- Purvis, J.E.; Yomano, L.P.; Ingram, L.O. Enhanced trehalose production improves growth of escherichia coli under osmotic stress. Applied and Environmental Microbiology 2005, 71, 3761–3769. [Google Scholar] [CrossRef]
- Rabbani, A.; Zainith, S.; Deb, V.K.; Das, P.; Bharti, P.; Rawat, D.S.; Saxena, G. Microbial technologies for environmental remediation: Potential issues, challenges, and future prospects. Microbe Mediated Remediation of Environmental Contaminants 2021, 271–286. [Google Scholar]
- Ramakrishnan, B.; Venkateswarlu, K.; Sethunathan, N.; Megharaj, M. Local applications but global implications: Can pesticides drive microorganisms to develop antimicrobial resistance? Science of the Total Environment, 654.
- Rello 2019, J. , Van Engelen, T. S. R., Alp, E., Calandra, T., Cattoir, V., Kern, W. V.,... Wiersinga, W. J. Towards precision medicine in sepsis: A position paper from the european society of clinical microbiology and infectious diseases. Clinical Microbiology and Infection 2018, 24, 1264–1272. [Google Scholar]
- Riley, S.P.; Sauvajot, R.M.; Fuller, T.K.; York, E.C.; Kamradt, D.A.; Bromley, C.; Wayne, R.K. Effects of urbanization and habitat fragmentation on bobcats and coyotes in southern california. Conservation Biology 2003, 17, 566–576. [Google Scholar]
- Rodriguez, R.J.; Redman, R.S.; Henson, J.M. The role of fungal symbioses in the adaptation of plants to high stress environments. Mitigation and Adaptation Strategies for Global Change 2004, 9, 261–272. [Google Scholar]
- Rodriguez-Verdugo, A.; Vulin, C.; Ackermann, M. The rate of environmental fluctuations shapes ecological dynamics in a two?species microbial system. Ecology Letters 2019, 22, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Roebler, M.; Muller, V. Osmoadaptation in bacteria and archaea: Common principles and differences. Environmental Microbiology 2001, 3, 743–754. [Google Scholar]
- Samagaci, L.; Ouattara, H.G.; Goualie, B.G.; Niamke, S.L. Polyphasic analysis of pectinolytic and stress-resistant yeast strains isolated from ivorian cocoa fermentation. Journal of Food Research 2015, 4, 124. [Google Scholar] [CrossRef]
- Schimel, J.; Balser, T.C.; Wallenstein, M. Microbial stress?response physiology and its implications for ecosystem function. Ecology 2007, 88, 1386–1394. [Google Scholar]
- Seufferheld, M.J.; Alvarez, H.M.; Farias, M.E. Role of polyphosphates in microbial adaptation to extreme environments. Applied and Environmental Microbiology 2008, 74, 5867–5874. [Google Scholar] [CrossRef]
- Sharma, P.; Singh, S.P.; Iqbal, H.M.; Tong, Y.W. Omics approaches in bioremediation of environmental contaminants: An integrated approach for environmental safety and sustainability. Environmental Research 2022, 211. [Google Scholar] [CrossRef]
- Shi, D.J.; Wang, C.L.; Wang, K.M. Genome shuffling to improve thermotolerance, ethanol tolerance and ethanol productivity of saccharomyces cerevisiae. Journal of Industrial Microbiology and Biotechnology 2009, 36, 139–147. [Google Scholar]
- Silva, L.N.; Zimmer, K.R.; Macedo, A.J.; Trentin, D.S. Plant natural products targeting bacterial virulence factors. Chemical Reviews 2016, 116, 9162–9236. [Google Scholar]
- Skoneczny, M.; Skoneczna, A. Response mechanisms to chemical and physical stresses in yeast and filamentous fungi. Stress Response Mechanisms in Fungi: Theoretical and Practical Aspects.
- Solden, L.; Lloyd, K.; Wrighton, K. The bright side of microbial dark matter: Lessons learned from the uncultivated majority. Current Opinion in Microbiology 2016, 31, 217–226. [Google Scholar] [CrossRef]
- Subedi, B.; Aguilar, L.; Robinson, E.M.; Hageman, K.J.; Bjorklund, E.; Sheesley, R.J.; Usenko, S. Selective pressurized liquid extraction as a sample-preparation technique for persistent organic pollutants and contaminants of emerging concern. TrAC Trends in Analytical Chemistry 2015, 68, 119–132. [Google Scholar]
- Tan, Y.S.; Zhang, R.K.; Liu, Z.H.; Li, B.Z.; Yuan, Y.J. Microbial adaptation to enhance stress tolerance. Frontiers in Microbiology 2022, 13. [Google Scholar]
- Thorwall, S.; Schwartz, C.; Chartron, J.W.; Wheeldon, I. Stress-tolerant non-conventional microbes enable next-generation chemical biosynthesis. Nature Chemical Biology 2020, 16, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Tiedje, J.M.; Bruns, M.A.; Casadevall, A.; Criddle, C.S.; Eloe-Fadrosh, E.; Karl, D.M.; Zhou, J. Microbes and climate change: A research prospectus for the future. Mbio 2022, 13, 800–822. [Google Scholar] [CrossRef] [PubMed]
- Toft, C.; Andersson, S.G. Evolutionary microbial genomics: Insights into bacterial host adaptation. Nature Reviews Genetics 2010, 11, 465–475. [Google Scholar] [PubMed]
- Trovao, M.; Sch?ler, L.M.; Machado, A.; Bombo, G.; Navalho, S.; Barros, A.; Varela, J. Random mutagenesis as a promising tool for microalgal strain improvement towards industrial production. Marine Drugs 2022, 20, 440. [Google Scholar]
- Uddin, T.M.; Chakraborty, A.J.; Khusro, A.; Zidan BR, M.; Mitra, S.; Emran, T.B.; Koirala, N. Antibiotic resistance in microbes: History, mechanisms, therapeutic strategies and future prospects. Journal of Infection and Public Health 2021, 14, 1750–1766. [Google Scholar]
- Valliere, J.M.; Wong, W.S.; Nevill, P.G.; Zhong, H.; Dixon, K.W. Preparing for the worst: Utilizing stress?tolerant soil microbial communities to aid ecological restoration in the anthropocene. Ecological Solutions and Evidence 2020, 1, e12027. [Google Scholar]
- Verma, M. L. , Kumar, S., Jeslin, J., & Dubey, N. K. (2020). Microbial production of biopolymers with potential biotechnological applications. Biopolymer-based formulations.
- Wang, Y.; Bhattacharya, T.; Jiang, Y.; Qin, X.; Wang, Y.; Liu, Y.; Chen, L. A novel deep learning method for predictive modeling of microbiome data. Briefings in Bioinformatics 2021, 22. [Google Scholar] [CrossRef]
- Wondraczek, L.; Pohnert, G.; Schacher, F.H.; Kohler, A.; Gottschaldt, M.; Schubert, U.S.; Brakhage, A.A. Artificial microbial arenas: Materials for observing and manipulating microbial consortia. Advanced Materials 2019, 31, 190028. [Google Scholar]
- Wu, J.; Tzanakakis, E.S. Deconstructing stem cell population heterogeneity: Single-cell analysis and modeling approaches. Biotechnology Advances 2013, 31, 1047–1062. [Google Scholar]
- Xu, S.; Zhou, J.; Liu, L.; Chen, J. Proline enhances torulopsis glabrata growth during hyperosmotic stress. Biotechnology and Bioprocess Engineering 2010, 15, 285–292. [Google Scholar] [CrossRef]
- Xu, S.; Zhou, J.; Liu, L.; Chen, J. Arginine: A novel compatible solute to protect candida glabrata against hyperosmotic stress. Process Biochemistry 2011, 46, 1230–1235. [Google Scholar] [CrossRef]
- Yang, X.; Li, Y.; Niu, B.; Chen, Q.; Hu, Y.; Yang, Y.; Zhang, G. Temperature and precipitation drive elevational patterns of microbial beta diversity in alpine grasslands. Microbial Ecology 2021, 1–13. [Google Scholar] [CrossRef]
- Yazawa, H.; Iwahashi, H.; Uemura, H. Disruption of URA7 and GAL6 improves the ethanol tolerance and fermentation capacity of saccharomyces cerevisiae. Yeast 2007, 24, 551–560. [Google Scholar] [CrossRef]
- Yong, Y.C.; Zhong, J.J. Recent advances in biodegradation in china: New microorganisms and pathways, biodegradation engineering, and bioenergy from pollutant biodegradation. Process Biochemistry 2010, 45, 1937–1943. [Google Scholar] [CrossRef]
- Zhang, C.; Sun, R.; Xia, T. Adaption/resistance to antimicrobial nanoparticles: Will it be a problem? Nano Today, 34.
- Zhu, Y. 2020, Li, J., Tan, M., Liu, L., Jiang, L., Sun, J., & Chen, J. Optimization and scale-up of propionic acid production by propionic acid-tolerant propionibacterium acidipropionici with glycerol as the carbon source. Bioresource Technology 2010, 101, 8902–8906. [Google Scholar]
- Zingaro, K.A.; Papoutsakis, E.T. GroESL overexpression imparts escherichia coli tolerance to i-, n-, and 2-butanol, 1, 2, 4-butanetriol and ethanol with complex and unpredictable patterns. Metabolic Engineering 2013, 15, 196–205. [Google Scholar] [CrossRef]
- Hosseini Nezhad, M.; Hussain, A.M.; Britz, L.M. Stress responses in probiotic Lactobacillus casei. Critical reviews in food science and nutrition 2015, 55, 740–749. [Google Scholar] [CrossRef]
- Koutsoumanis, K. , Allende, A., Alvarez-Ordonez, A., Bolton, D., Bover-Cid, S., Chemaly, M., Hilbert, F. (2021). Role played by the environment in the emergence and spread of antimicrobial resistance (AMR) through the food. EFSA Panel on Biological Hazards.
- Smidt, H. , Bruijn, F. J., Cocolin, L. S., Sauer, M., Dowling, D. N., & Thomashow, L. (2023). Good microbes in medicine, food production, biotechnology, bioremediation, and agriculture (Vol. 303026).
- van der Meer, J. R. Environmental pollution promotes selection of microbial degradation pathways 4. Frontiers in Ecology and the Environment 2006, 4, 35–42. [Google Scholar] [CrossRef]
- van Vliet, S. , Hauert, C., Fridberg, K., & Ackermann, M. Dal Co A (2022) Global dynamics of microbial communities emerge from local interaction rules. PLoS Comput Biol 2022, 18, e1009877. [Google Scholar] [CrossRef]
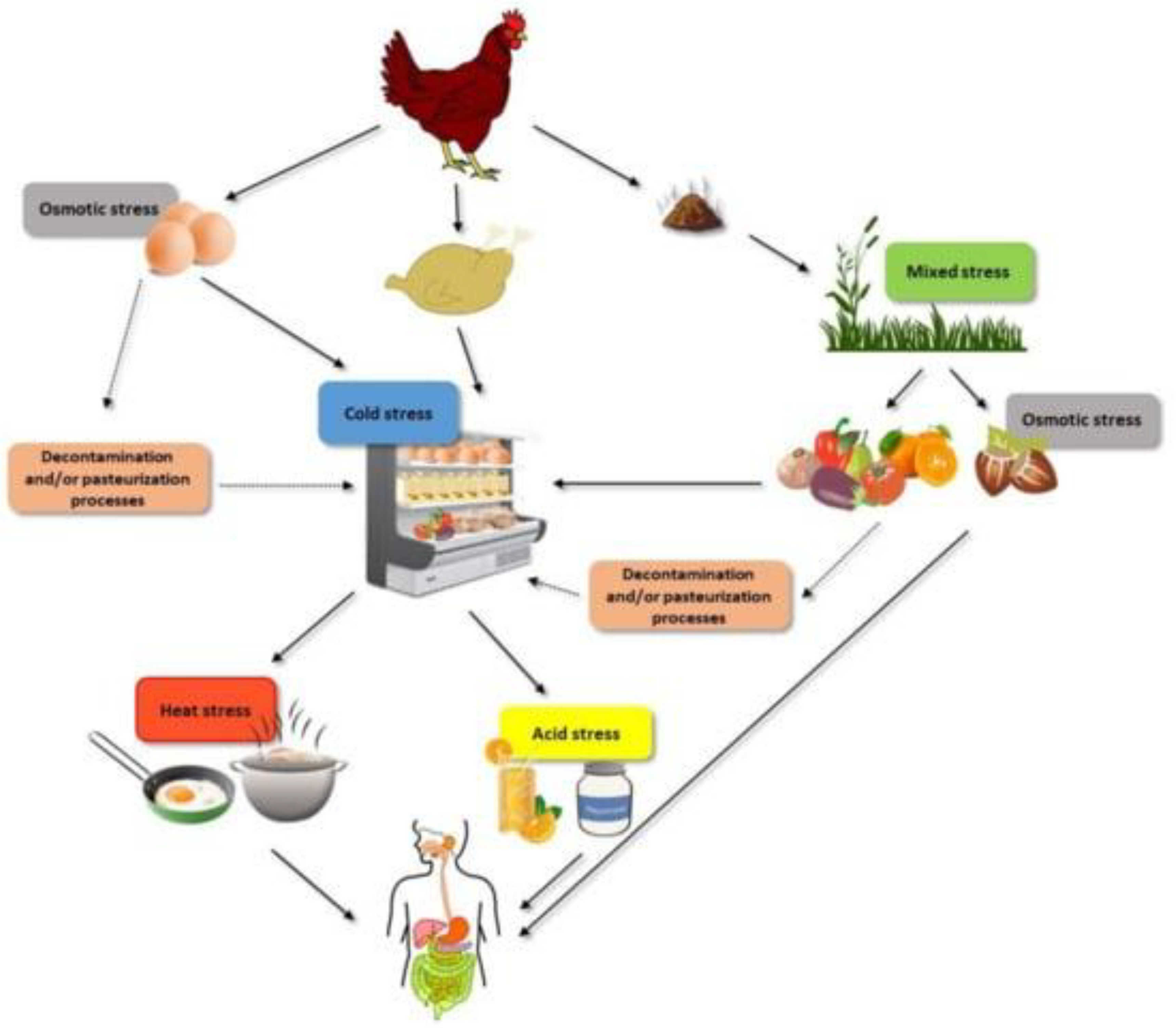
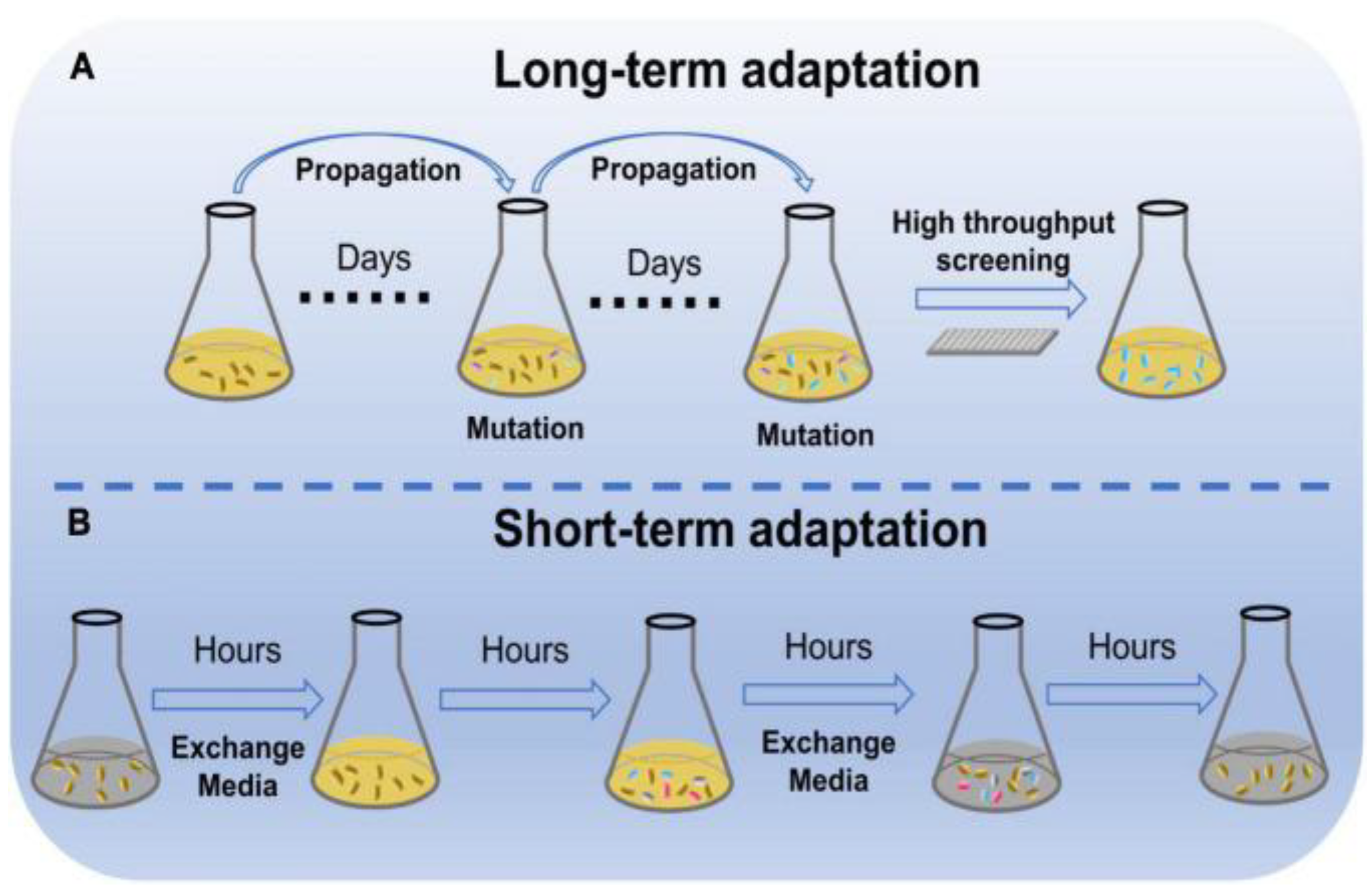
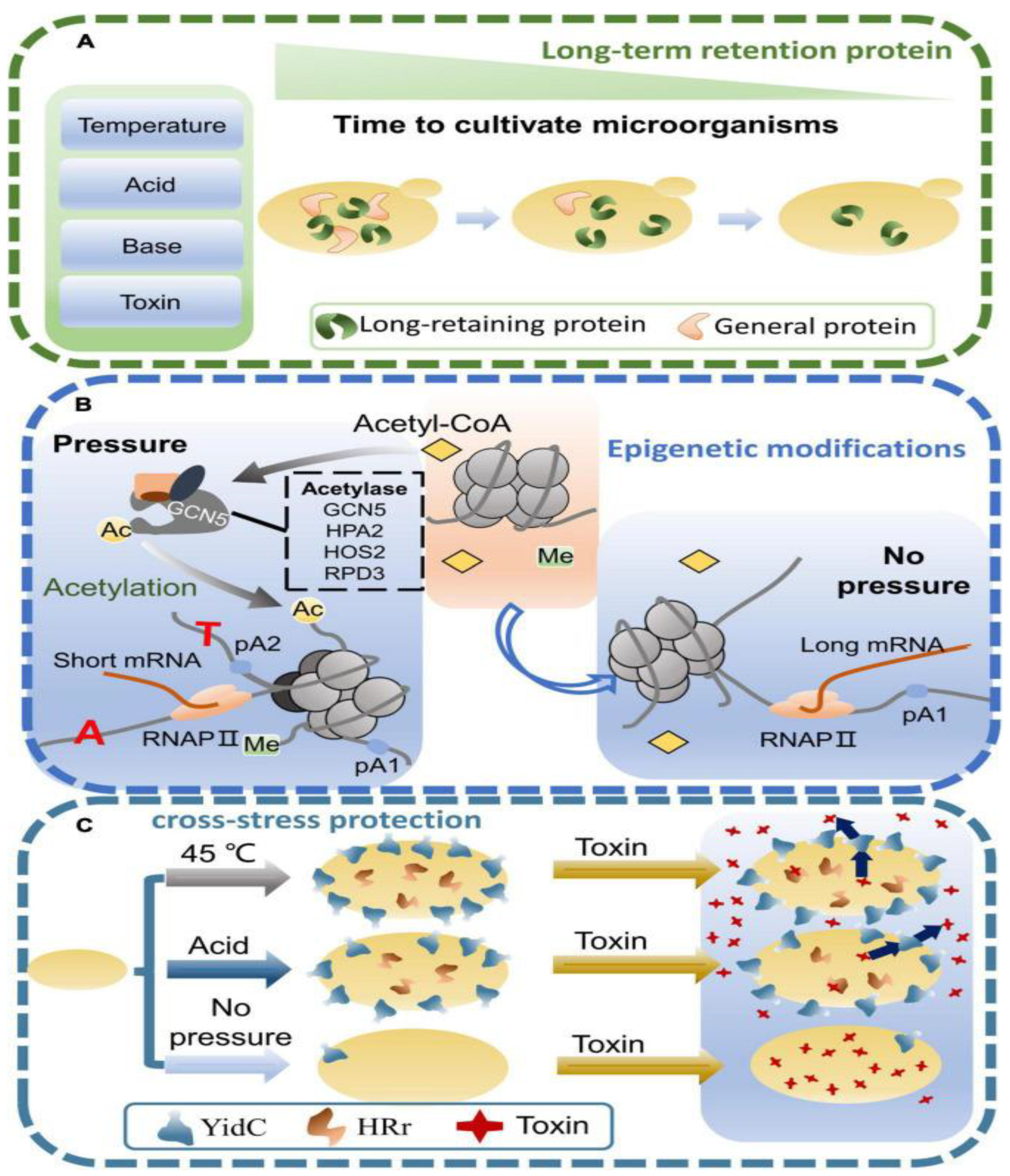
| S.No. | Stress | Organism | Approach | Reference |
|---|---|---|---|---|
| 1 | Oxidative | Escherichia coli | Overexpression of Dps, a DNA-binding protein that protects against oxidative stress | (Choi et al. 2000) |
| Saccharomyces cerevisiae | Expression of a glutamate Decarboxylase homolog to enhance stress resistance | (Coleman et al. 2001) | ||
| Lactococcus lactis | Introduction of glutathione Biosynthetic capability through genetic modification | (Fu et al. 2006) | ||
| Candida utilis | Control of dissolved oxygen levels in the environment to mitigate oxidative stress | (Liang, Du, & Chen, 2008) | ||
| Candida utilis | Addition of H2O2, a known oxidative stress inducer, to study stress response mechanisms | (G. Liang et al. 2008) | ||
| Candida infirmominiatum | Addition of glycine betaine, a compatible solute, to enhance stress tolerance | (J. Liu et al. 2011) | ||
| 2 | Hyperosmotic | Escherichia coli | Overproduction of trehalose, a compatible solute, to counteract osmotic stress | (Purvis et al. 2005) |
| Lactococcus lactis | Expression of DnaK, a chaperone protein, to assist in protein folding under stress | (Abdullah-Al-Mahin et al. 2010) | ||
| Torulaspora glabrata | Addition of proline, an osmoprotectant, to alleviate hyperosmotic stress | (Xu et al. 2010) | ||
| Candida glabrata | Accumulation of arginine, an osmoprotectant, to enhance stress resistance | (Xu et al. 2011) | ||
| Escherichia coli | Expression of IrrE, a radiation resistance protein, to confer osmotic stress tolerance | (Ma et al. 2011) | ||
| 3 | Thermal | Kluyveromyces marxianus | Induction of mutation in response to thermal stress | (Ballesteros et al. 1993) |
| Lactococcus lactis, and Lactococcus paracasei | Overproduction of GroESL, a chaperonin complex, to assist in protein folding under heat stress | (Compan and Touati 1993) | ||
| Saccharomyces cerevisiae | Application of evolutionary engineering methods to develop thermotolerant strains | (Cakar et al. 2005) | ||
| Saccharomyces cerevisiae | Utilization of genome shuffling techniques to enhance thermal stress resistance | (Shi et al. 2009) | ||
| 4 | Acid | Propionibacterium acidipropionici | Execution of adaptive growth strategies to advance acid stress resistance | (Y. Zhu et al. 2010) |
| Propionibacterium acidipropionici | Implementation of adaptive evolution strategies to improve acid stress resistance | (L. Zhu et al. 2012) | ||
| Propionibacterium acidipropionici | Utilization of genome shuffling techniques to enhance acid stress resistance | (Guan et al. 2012) | ||
| Lactobacillus casei | Application of metabolic engineering approaches to develop acid-tolerant strains | (Y. Zhang et al. 2010) | ||
| Pediococcus jensenii | Implementation of metabolic engineering strategies targeting acid resistance elements | (Guan et al. 2016) | ||
| 5 | Organic solvent | Saccharomyces cerevisiae | Utilization of transcription machinery engineering to enhance solvent tolerance | (Alper et al. 2006) |
| Saccharomyces cerevisiae | Overexpression of TRP1–5 and TAT2 genes to improve solvent tolerance | (Hirasawa et al. 2007) | ||
| Saccharomyces cerevisiae | Deletion of URA7 and GAL6 genes to enhance solvent tolerance | (Yazawa et al. 2007) | ||
| Escherichia coli | Overproduction of GroESL, a chaperonin complex, to enhance solvent tolerance | (Zingaro and Papoutsakis 2013) | ||
| Corynebacterium glutamicum | Implementation of adaptive evolution strategies to enhance solvent tolerance | (Oide et al. 2015) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




