Submitted:
10 March 2025
Posted:
11 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
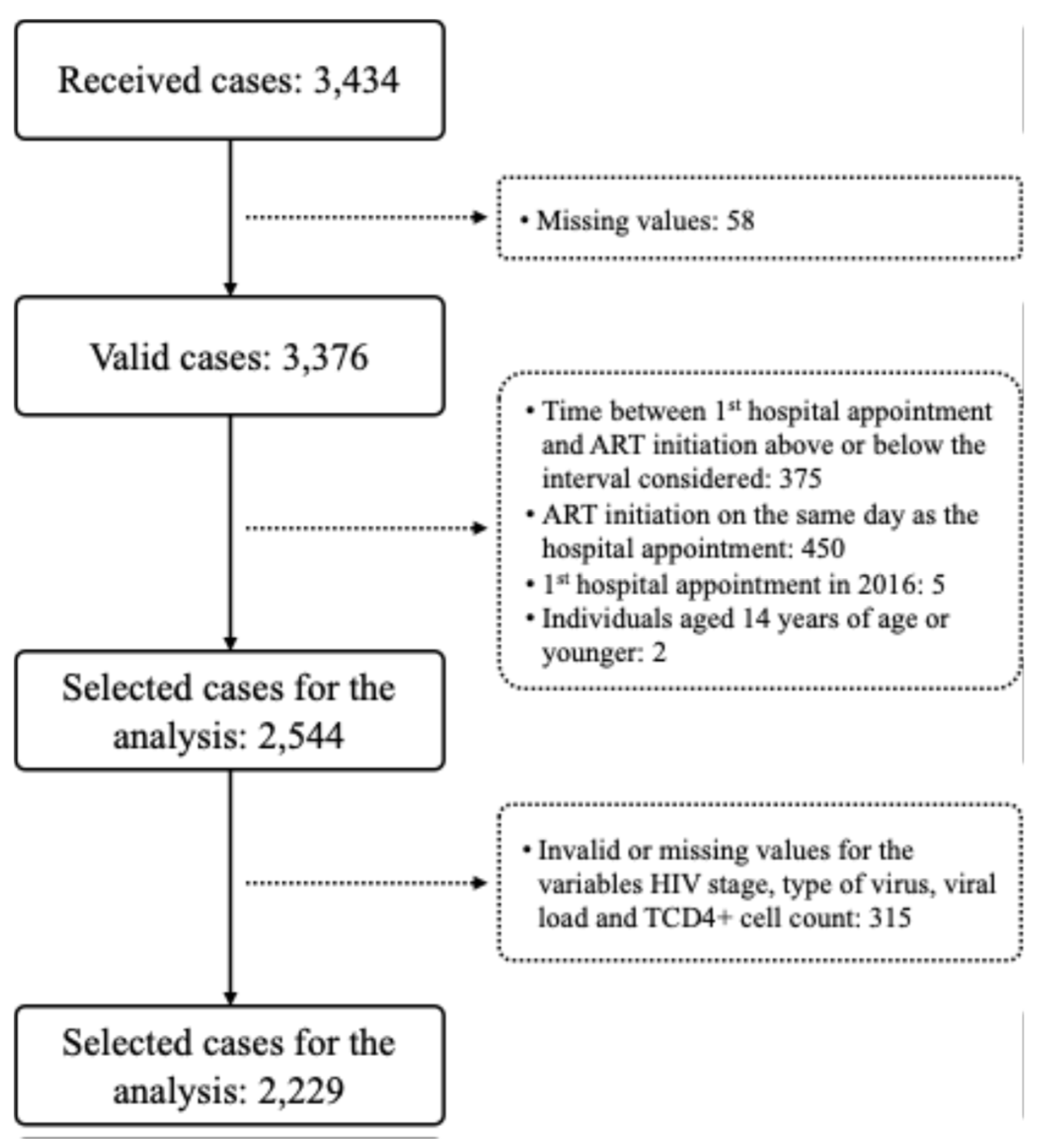
2.1. Data Collection
2.2. Variables
2.3. Data Analysis
2.4. Ethical Considerations
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hammer, S.M.; Squires, K.E.; Hughes, M.D.; Grimes, J.M.; Demeter, L.M.; Currier, J.S.; Eron, J.J.J.; Feinberg, J.E.; Balfour, H.H.J.; Deyton, L.R.; et al. A Controlled Trial of Two Nucleoside Analogues plus Indinavir in Persons with Human Immunodeficiency Virus Infection and CD4 Cell Counts of 200 per Cubic Millimeter or Less. New Engl. J. Med. 1997, 337, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Palella, F.J., Jr.; Delaney, K.M.; Moorman, A.C.; Loveless, M.O.; Fuhrer, J.; Satten, G.A.; Aschman, D.J.; Holmberg, S.D.; the HIV Outpatient Study Investigators. Declining Morbidity and Mortality among Patients with Advanced Human Immunodeficiency Virus Infection. N. Engl. J. Med. 1998, 338, 853–860. [Google Scholar] [CrossRef]
- Mateo-Urdiales A, Johnson S, Smith R, Nachega JB, Eshun-Wilson I. Rapid initiation of antiretroviral therapy for people living with HIV. Cochrane Database Syst Rev [Internet]. 2019 [cited 2024 Oct 12];2019. Available from: /pmc/articles/PMC6575156/.
- Kitahata, M.M.; Gange, S.J.; Abraham, A.G.; Merriman, B.; Justice, A.C.; Hogg, R.S.; Deeks, S.G.; Eron, J.J.; Brooks, J.T.; Rourke, S.B.; et al. Effect of Early versus Deferred Antiretroviral Therapy for HIV on Survival. New Engl. J. Med. 2009, 360, 1815–1826. [Google Scholar] [CrossRef]
- Andrade, H.B.; Shinotsuka, C.R.; da Silva, I.R.F.; Donini, C.S.; Li, H.Y.; de Carvalho, F.B.; Brasil, P.E.A.A.D.; Bozza, F.A.; Japiassu, A.M. Highly active antiretroviral therapy for critically ill HIV patients: A systematic review and meta-analysis. PLOS ONE 2017, 12, e0186968. [Google Scholar] [CrossRef]
- Lundgren JD, Babiker AG, Gordin F, Emery S, Grund B, Sharma S, et al. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med. 2015;373:795–807.
- Temprano ANRS 12136 Study Group. A trial of early antiretrovirals and isoniazid preventive therapy in Africa. N Engl J Med. 2015;373:808–22.
- Samji, H.; Cescon, A.; Hogg, R.S.; Modur, S.P.; Althoff, K.N.; Buchacz, K.; Burchell, A.N.; Cohen, M.; Gebo, K.A.; Gill, M.J.; et al. Closing the Gap: Increases in Life Expectancy among Treated HIV-Positive Individuals in the United States and Canada. PLOS ONE 2013, 8, e81355. [Google Scholar] [CrossRef]
- Lohse, N.; Obel, N. Update of Survival for Persons With HIV Infection in Denmark. Ann. Intern. Med. 2016, 165, 749–750. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Verma, A.; Kashyap, M.; Gautam, P. ART in Prevention of Mother-to-Child Transmission of HIV. J. Obstet. Gynecol. India 2020, 70, 18–22. [Google Scholar] [CrossRef]
- Cohen, M.S.; Chen, Y.Q.; McCauley, M.; Gamble, T.; Hosseinipour, M.C.; Kumarasamy, N.; Hakim, J.G.; Kumwenda, J.; Grinsztejn, B.; Pilotto, J.H.; et al. Prevention of HIV-1 Infection with Early Antiretroviral Therapy. N. Engl. J. Med. 2011, 365, 493–505. [Google Scholar] [CrossRef]
- Zolopa, A.R.; Andersen, J.; Komarow, L.; Sanne, I.; Sanchez, A.; Hogg, E.; Suckow, C.; Powderly, W. Early Antiretroviral Therapy Reduces AIDS Progression/Death in Individuals with Acute Opportunistic Infections: A Multicenter Randomized Strategy Trial. PLOS ONE 2009, 4, e5575. [Google Scholar] [CrossRef]
- Koenig, S.P.; Dorvil, N.; Dévieux, J.G.; Hedt-Gauthier, B.L.; Riviere, C.; Faustin, M.; Lavoile, K.; Perodin, C.; Apollon, A.; Duverger, L.; et al. Same-day HIV testing with initiation of antiretroviral therapy versus standard care for persons living with HIV: A randomized unblinded trial. PLOS Med. 2017, 14, e1002357–e1002357. [Google Scholar] [CrossRef]
- Pilcher, C.D.; Ospina-Norvell, C.F.-P.; Dasgupta, A.B.; Jones, D.; Hartogensis, W.; Torres, S.M.; Calderon, F.M.; Demicco, E.; Geng, E.; Gandhi, M.; et al. The Effect of Same-Day Observed Initiation of Antiretroviral Therapy on HIV Viral Load and Treatment Outcomes in a US Public Health Setting. Am. J. Ther. 2017, 74, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Mendez-Lopez, A.; McKee, M.; Stuckler, D.; Granich, R.; Gupta, S.; Noori, T.; Semenza, J. Population uptake and effectiveness of test-and-treat antiretroviral therapy guidelines for preventing the global spread of HIV: an ecological cross-national analysis. HIV Med. 2019, 20, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Ford, N.; Migone, C.; Calmy, A.; Kerschberger, B.; Kanters, S.; Nsanzimana, S.; Mills, E.J.; Meintjes, G.; Vitoria, M.; Doherty, M.; et al. Benefits and risks of rapid initiation of antiretroviral therapy. AIDS 2018, 32, 17–23. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for managing advanced HIV disease and rapid initiation of antiretroviral therapy. Geneva; 2017.
- Ford, N.; Vitoria, M.; Doherty, M. Providing antiretroviral therapy to all who are HIV positive: the clinical, public health and programmatic benefits of Treat All. J. Int. AIDS Soc. 2018, 21, e25078. [Google Scholar] [CrossRef] [PubMed]
- Njuguna, I.N.; Cranmer, L.M.; O Otieno, V.; Mugo, C.; Okinyi, H.M.; Benki-Nugent, S.; Richardson, B.; Stern, J.; Maleche-Obimbo, E.; Wamalwa, D.C.; et al. Urgent versus post-stabilisation antiretroviral treatment in hospitalised HIV-infected children in Kenya (PUSH): a randomised controlled trial. Lancet HIV 2018, 5, e12–e22. [Google Scholar] [CrossRef]
- Grant PM, Zolopa AR. When to start ART in the setting of acute AIDS-related opportunistic infections: The time is now! Curr HIV/AIDS Rep. 2012;9:251–8.
- Boyd, M.; Boffito, M.; Castagna, A.; Estrada, V. Rapid initiation of antiretroviral therapy at HIV diagnosis: definition, process, knowledge gaps. HIV Med. 2019, 20, 3–11. [Google Scholar] [CrossRef]
- Bogdanić, N.; Bendig, L.; Lukas, D.; Zekan, Š.; Begovac, J. Timeliness of antiretroviral therapy initiation in the era before universal treatment. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Joint United Nations Programme on HIV/AIDS. Prevailing against pandemics by putting people at the centre: World AIDS Day report 2020 [Internet]. 2020. Available from: https://aidstargets2025.unaids.org/assets/images/prevailing-against-pandemics_en.
- Gardner, E.M.; McLees, M.P.; Steiner, J.F.; del Rio, C.; Burman, W.J. The Spectrum of Engagement in HIV Care and its Relevance to Test-and-Treat Strategies for Prevention of HIV Infection. Clin. Infect. Dis. 2011, 52, 793–800. [Google Scholar] [CrossRef]
- Genberg, B.L.; Lee, Y.; Rogers, W.H.; Wilson, I.B. Four Types of Barriers to Adherence of Antiretroviral Therapy are Associated with Decreased Adherence Over Time. AIDS Behav. 2015, 19, 85–92. [Google Scholar] [CrossRef]
- Croome, N.; Ahluwalia, M.; Hughes, L.D.; Abas, M. Patient-reported barriers and facilitators to antiretroviral adherence in sub-Saharan Africa. AIDS 2017, 31, 995–1007. [Google Scholar] [CrossRef]
- Moges, N.A.; Adesina, O.A.; A Okunlola, M.; Berhane, Y. Barriers and Facilitators of Same-Day Antiretroviral Therapy Initiation Among People Newly Diagnosed with HIV in Ethiopia: Qualitative Study Using the Transtheoretical Model of Behavioral Change. J. Multidiscip. Heal. 2020, 13, 1801–1815. [Google Scholar] [CrossRef]
- Patel, R.C.; Odoyo, J.; Anand, K.; Stanford-Moore, G.; Wakhungu, I.; Bukusi, E.A.; Baeten, J.M.; Brown, J.M. Facilitators and Barriers of Antiretroviral Therapy Initiation among HIV Discordant Couples in Kenya: Qualitative Insights from a Pre-Exposure Prophylaxis Implementation Study. PLOS ONE 2016, 11, e0168057. [Google Scholar] [CrossRef]
- Esber, A.L.; Coakley, P.; A Ake, J.; Bahemana, E.; Adamu, Y.; Kiweewa, F.; Maswai, J.; Owuoth, J.; Robb, M.L.; Polyak, C.S.; et al. Decreasing time to antiretroviral therapy initiation after HIV diagnosis in a clinic-based observational cohort study in four African countries. J. Int. AIDS Soc. 2020, 23, e25446. [Google Scholar] [CrossRef] [PubMed]
- Medland, N.A.; Chow, E.P.F.; McMahon, J.H.; Elliott, J.H.; Hoy, J.F.; Fairley, C.K. Time from HIV diagnosis to commencement of antiretroviral therapy as an indicator to supplement the HIV cascade: Dramatic fall from 2011 to 2015. PLoS ONE 2017, 12, e0177634. [Google Scholar]
- Lockman, S.; Holme, M.P.; Makhema, J.; Bachanas, P.; Moore, J.; Wirth, K.E.; Lebelonyane, R.; Essex, M. Implementation of Universal HIV Testing and Treatment to Reduce HIV Incidence in Botswana: the Ya Tsie Study. Curr. HIV/AIDS Rep. 2020, 17, 478–486. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, J.; Fu, X.; Yang, F.; Liu, Q.; Li, J.; Tan, Z.; Li, J.; Lin, K.; Yan, Y.; et al. Effect of Late Testing and Antiretroviral Treatment on Mortality Among People Living With HIV in the Era of Treat-All in Guangdong Province, China, 1992–2018: A Cohort Study. Front. Public Heal. 2022, 10, 851117. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Xiao, X.; Zhang, C.; Xie, Y.; Wang, H. Prevalence of delayed antiretroviral therapy initiation among people living with HIV: A systematic review and meta-analysis. PLOS ONE 2023, 18, e0286476. [Google Scholar] [CrossRef]
- Alejos B, Díez C, Galindo MJ, Lopez JC, Hernando V, Ayerdi O, et al. Linkage to care and time to viral suppression in PWH in Spain: 2004-2020. Top Antivir Med. 2022;30:358–358.
- Hill, J.E.; Stephani, A.-M.; Sapple, P.; Clegg, A.J. The effectiveness of continuous quality improvement for developing professional practice and improving health care outcomes: a systematic review. Implement. Sci. 2020, 15, 1–14. [Google Scholar] [CrossRef]
- Powers KA, Miller WC. Building on the HIV cascade: a complementary “HIV States and Transitions” framework for describing HIV diagnosis, care, and treatment at the population level. J Acquir Immune Defic Syndr. 2015;69:341–7.
- Joint United Nations Programme on HIV/AIDS (UNAIDS). 90-90-90: An ambitious treatment target to help end the AIDS epidemic. 2014.
- Ministério da Saúde. Diário da República n.o 228/2015, 1o Suplemento, Série II de 2015-11-20, Despacho n.o 13447-C/2015. Portugal; 2015.
- Ministério da Saúde. Direção-Geral da Saúde. Infeção VIH e SIDA: Desafios e Estratégias. Lisboa; 2018.
- Nicolau, V.; Cortes, R.; Lopes, M.; Virgolino, A.; Santos, O.; Martins, A.; Faria, N.; Reis, A.P.; Santos, C.; Maltez, F.; et al. HIV Infection: Time from Diagnosis to Initiation of Antiretroviral Therapy in Portugal, a Multicentric Study. Healthcare 2021, 9, 797. [Google Scholar] [CrossRef]
- Ministério da Saúde., Direção-Geral da Saúde, Instituto Nacional de Saúde Doutor Ricardo Jorge. Infeção VIH e SIDA em Portugal – 2022. 2022.
- Robertson, D.L.; Hahn, B.H.; Sharp, P.M. Recombination in AIDS viruses. J. Mol. Evol. 1995, 40, 249–259. [Google Scholar] [CrossRef]
- Sharp, P.M.; Hahn, B.H. Origins of HIV and the AIDS Pandemic. Cold Spring Harb. Perspect. Med. 2011, 1, a006841–a006841. [Google Scholar] [CrossRef] [PubMed]
- Grinsztejn, B.; Hosseinipour, M.C.; Ribaudo, H.J.; Swindells, S.; Eron, J.; Chen, Y.Q.; Wang, L.; Ou, S.-S.; Anderson, M.; McCauley, M.; et al. Effects of early versus delayed initiation of antiretroviral treatment on clinical outcomes of HIV-1 infection: results from the phase 3 HPTN 052 randomised controlled trial. Lancet Infect. Dis. 2014, 14, 281–290. [Google Scholar] [CrossRef] [PubMed]
- A Levy, J. Caution: should we be treating HIV infection early? Lancet 1998, 352, 982–983. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. HIV/AIDS surveillance in Europe 2022 - 2021 data [Internet]. 2022. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/2022-Annual_HIV_Report_final.pdf.
- European Centre for Disease Prevention and Control. ECDC Special report: Continuum of HIV care - Monitoring implementation of the Dublin Declaration on Partnership to fight HIV/AIDS in Europe and Central Asia: 2021 progress report [Internet]. 2021. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/Dublin-Continuum-of-HIV-care-2021-progress-report-final-with-covers-updated.pdf.
- Ben Farhat, J.; Hessamfar, M.; Farbos, S.; Desclaux, A.; Dumondin, G.; Ferrand, H.; Greib, C.; Castan, B.; Rispal, P.; Duffau, P.; et al. Time to Treatment Initiation and HIV Viral Suppression in People Diagnosed With HIV-1 During COVID-19 Pandemic in Ex-Aquitaine, France (ANRS CO3 AQUIVIH-NA Cohort-QuAliCOV Study). Am. J. Ther. 2024, 95, 1–5. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. World AIDS Day 2022: WHO/Europe and ECDC report reveals increasing numbers living with undiagnosed HIV in the Region [Internet]. 2022. Available from: https://www.ecdc.europa.eu/en/news-events/hiv-increasing-numbers-living-undiagnosed.
| Year of the first appointment | N | Median (IQR) | Mean (standard deviation) | Range (minimum-maximum) |
|---|---|---|---|---|
| 2017 | 493 | 36.00 (33.00) | 48.74 (48.15) | 1-364 |
| 2018 | 497 | 30.00 (23.00) | 39.54 (36.59) | 1-309 |
| 2019 | 428 | 29.00 (30.50) | 39.73 (42.07) | 1-355 |
| 2020 | 348 | 21.00 (28.00) | 36.62 (46.83) | 1-351 |
| 2021 | 353 | 22.00 (23.00) | 31.99 (39.08) | 1-351 |
| 2022 | 110 | 21.00 (22.00) | 26.15 (27.73) | 1-222 |
| Total | 2,229 | 29.00 (30.00) | 39.30 (42.52) | 1-354 |
| Total N |
≤14 days n (%) |
>14 days n (%) |
p-value | |
|---|---|---|---|---|
| HIV stage | ||||
| A | 1512 | 1196 (79.1%) | 316 (20.9%) | |
| B | 458 | 354 (77.3%) | 104 (22.7%) | <.001 |
| C | 259 | 155 (59.8%) | 104 (40.2%) | |
| Type of virus | ||||
| HIV1 | 2190 | 1676 (76.5%) | 514 (23.5%) | |
| HIV2 | 3 | 27 (75.0%) | 1 (33.3%) | .902 |
| HIV1 and 2 | 36 | 2 (66.7%) | 9 (25.0%) | |
| Viral load | ||||
| Mean (SD) | 756,175 (4,409,505) | 637,216 (4232190) | 1,1432,246 (4,927,177) | |
| Median (IQR) | 83,100 (319,702) | 70,718 (245,472) | 138,000 (570,850) | <.001 |
| Minimum-maximum | 0-101,000,000 | 0-101,000,000 | 37-100,000,000 | |
| TCD4+ cell count Mean (SD) Median (IQR) Minimum-maximum |
377.52 (331.06) 338.00 (353) 1-9210 |
392.74 (349.28) 353.23 (345) 2-9210 |
328.01 (257.29) 292.50 (367) 1-1362 |
<.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




