Submitted:
06 March 2025
Posted:
07 March 2025
You are already at the latest version
Abstract
Background: Functional gastrointestinal disorders, including irritable bowel syndrome (IBS) and functional abdominal bloating, are prevalent in gastroenterology. The unclear pathogenesis complicates the development of effective therapies, which may be pharmacological or non-pharmacological in nature. Objective: This pilot study aimed to evaluate the efficacy of a food supplement containing probiotics, digestive enzymes, and Melissa officinalis extract in patients suffering from IBS and functional abdominal bloating. Methods: We conducted a randomized, double-blind, placebo-controlled, monocentric trial involving adult patients diagnosed with IBS and functional abdominal bloating. Participants were randomly assigned to receive either the active product or a placebo for 20 days. The primary outcome measure was the change in the global Visual Analogue Scale (VAS) for satisfaction. Secondary measures included symptom scales related to bloating and abdominal pain. Results: A total of 31 participants were enrolled, with 29 completing the study. Patients receiving the active supplement reported a positive effect in the global VAS for satisfaction and demonstrated improvement in most secondary symptom scales compared to the placebo group. However, no statistically significant differences were found between the active and placebo groups, the treatment resulted in a higher tendency to improvement in patient-reported outcomes compared to the placebo group. Conclusion: This pilot study provides valuable insights into non-pharmacological therapies for IBS and functional abdominal bloating. Although the results were promising, larger studies with longer treatment durations and based on more complex approach also including diet intervention and psychological support are needed to fully evaluate the long-term effects of probiotics and the active ingredients in the supplement.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Product in Use
2.3. Questionnaires
2.4. Statistical Analysis
3. Results
3.1. Subjects’ Demographics
3.2. Global VAS
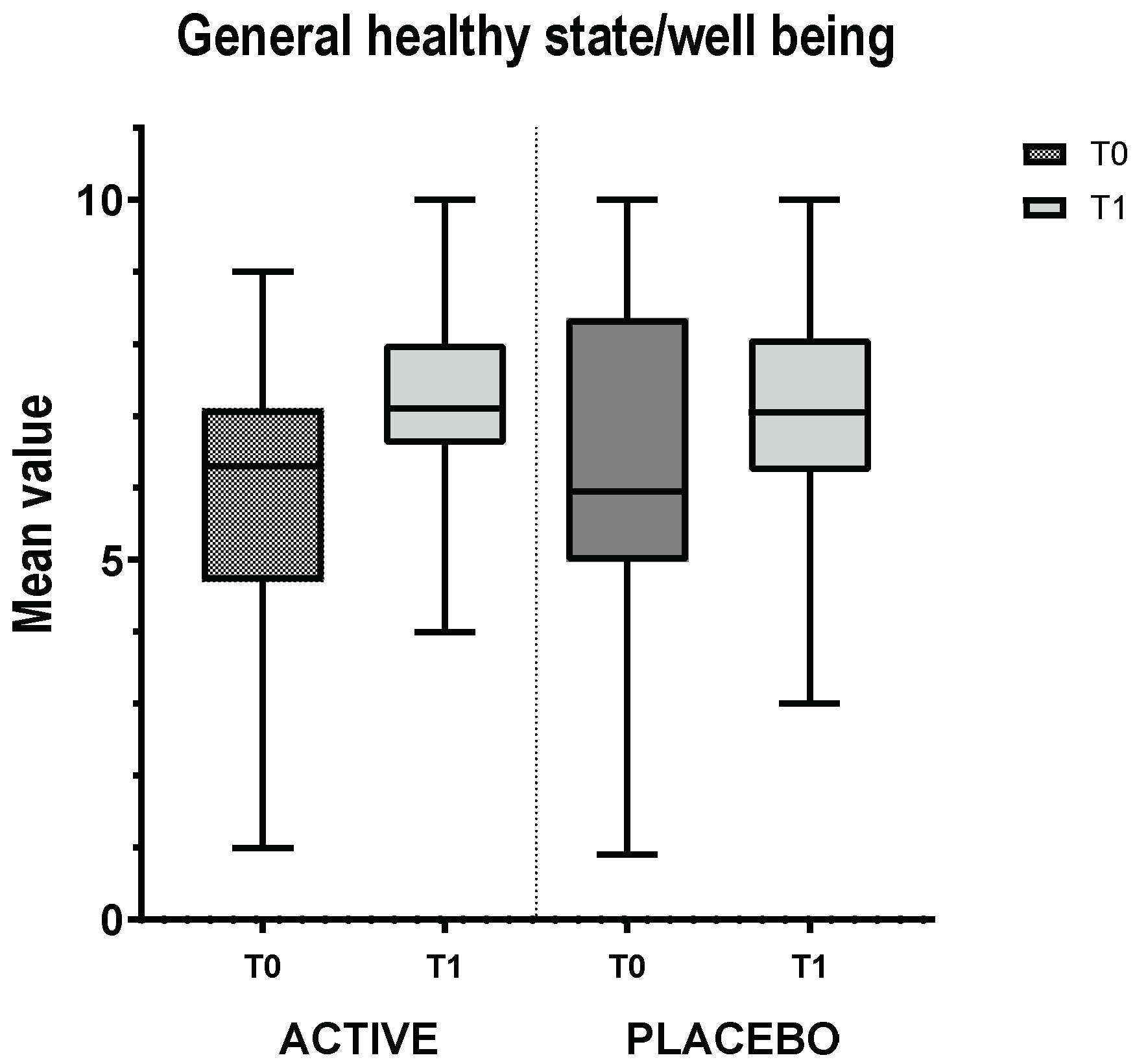
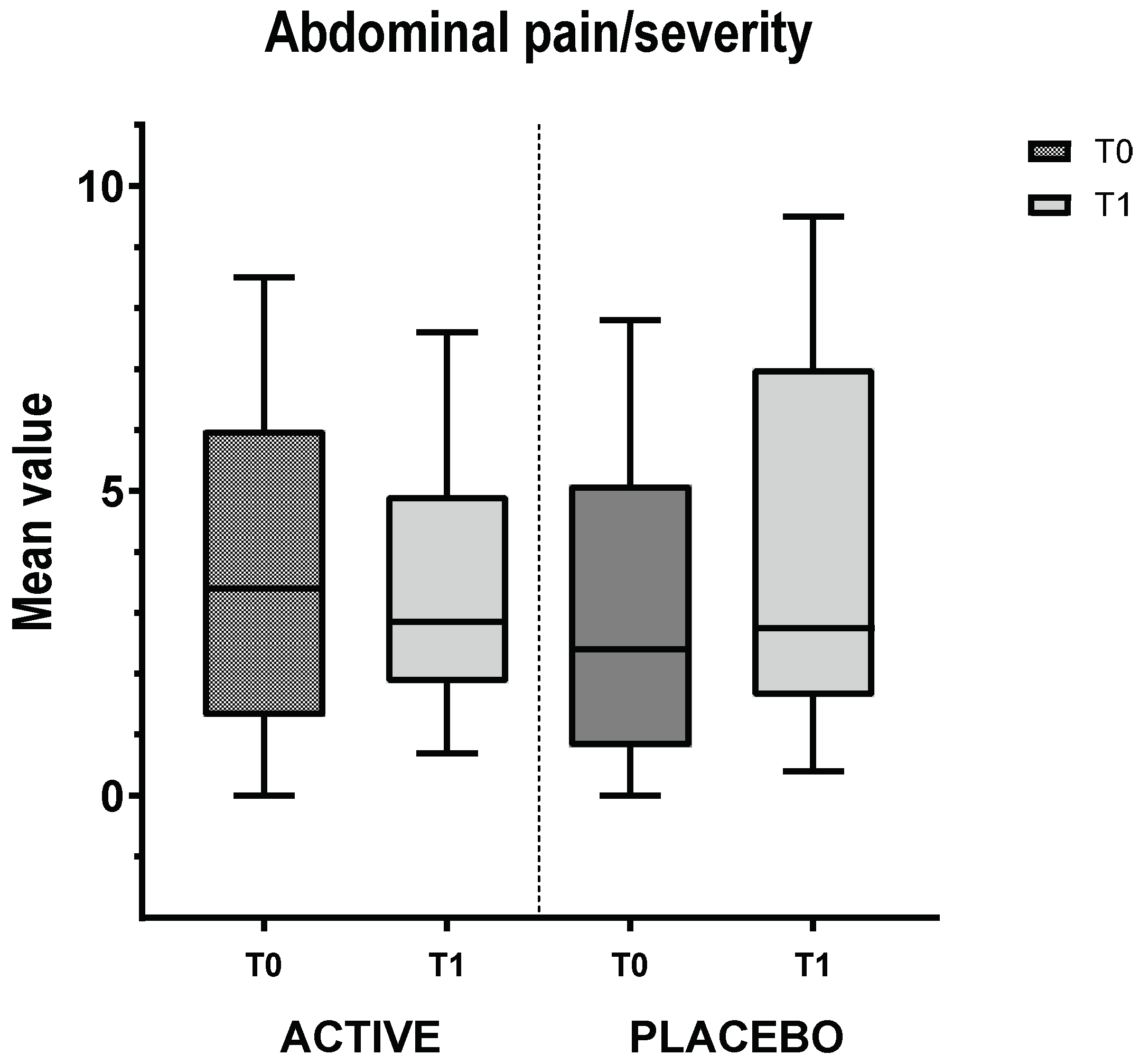
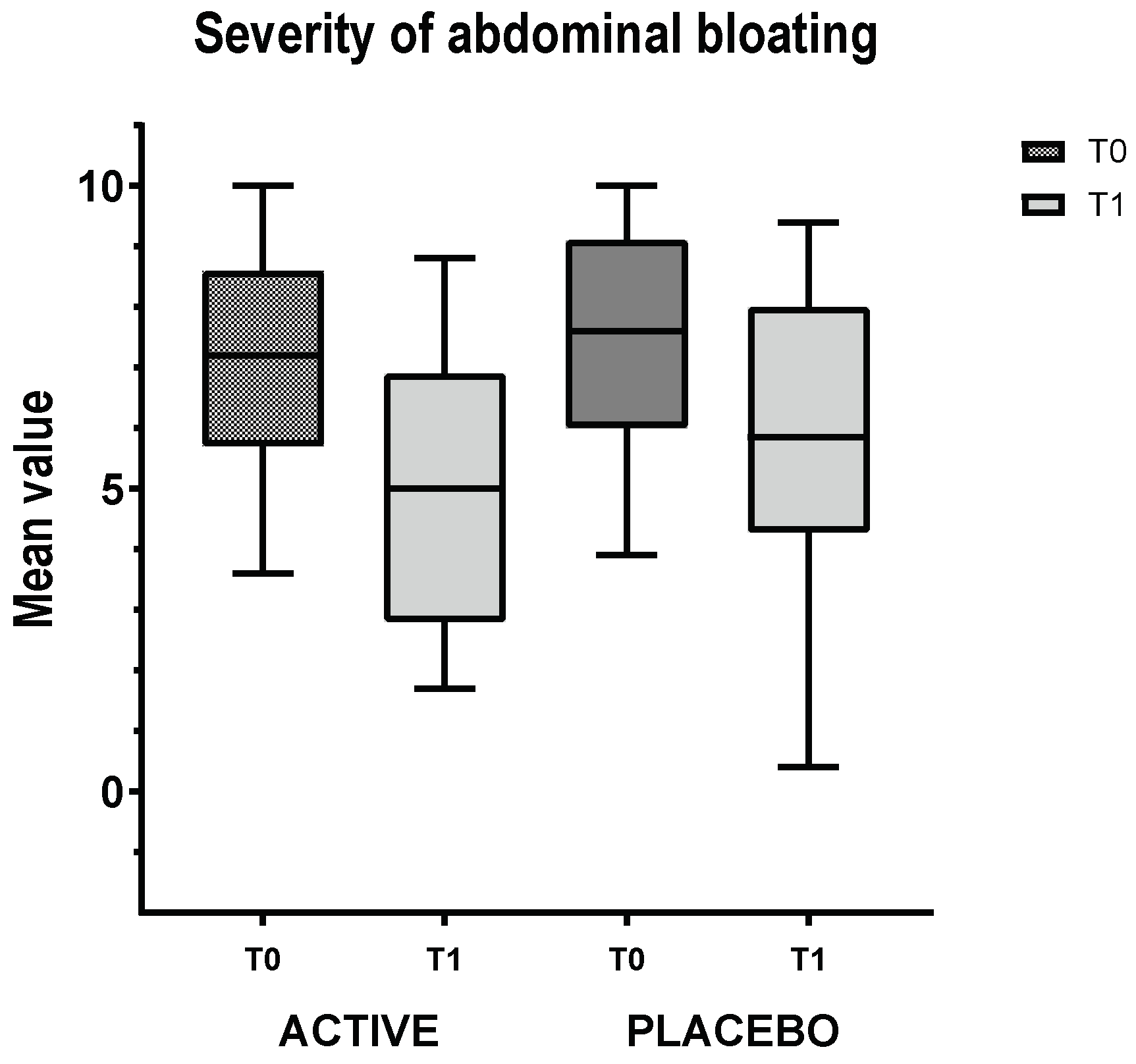
3.3. IBS Group VAS
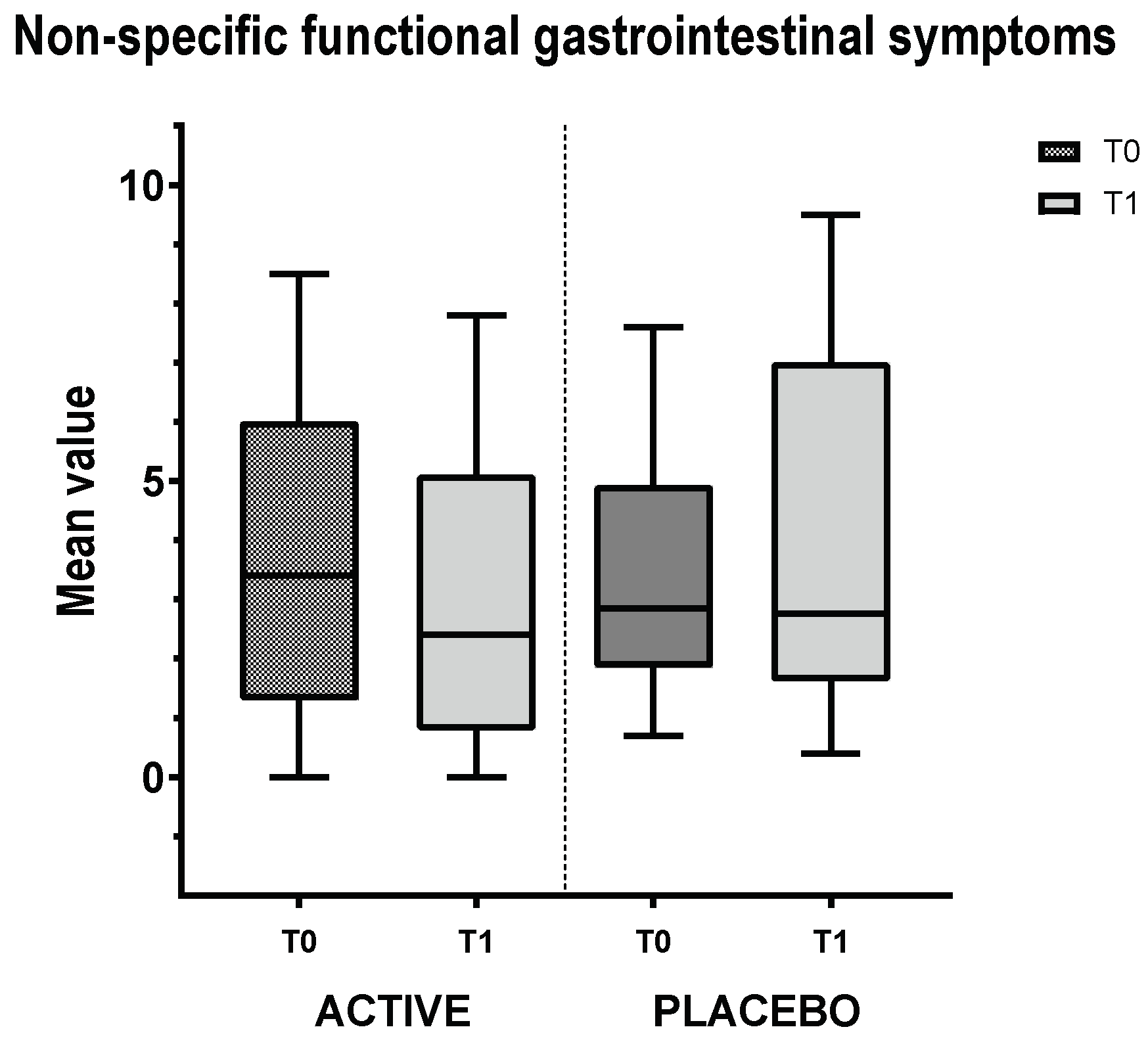
3.5. Non-Specific Functional Gastrointestinal Symptoms Group VAS
3.6. SF-36 Questionnaire
3.7. SCL-90 Questionnaire
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CFR | Case Report Form |
| FGID | Functional Gastrointestinal disorder |
| IBS | Irritable Bowel Syndrome |
| IBS-C | IBS with constipation |
| IBS-D | IBS with diarrhea |
| IBS-M | IBS with mixed symptoms |
| IBS-U | IBS unsubtyped |
| SCL-90 | Symptom Checklist-90 |
| SD | Standard Deviation |
| SF-36 | Short Form Healthy Survey |
| VAS | Visual Analogue Scale |
References
- Alonso-Bermejo C, Barrio J, Fernández B, García-Ochoa E, Santos A, Herreros M, Pérez C. Functional gastrointestinal disorders frequency by Rome IV criteria. Anales de Pediatría (English Edition). 2022;96(5):441–7.
- Black CJ, Drossman DA, Talley NJ, Ruddy J, Ford AC. Functional gastrointestinal disorders: advances in understanding and management. The Lancet. 2020;396(10263):1664–74.
- Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional Bowel Disorders. Gastroenterology. 2006;130(5):1480–91.
- Sperber AD, Bangdiwala SI, Drossman DA, Ghoshal UC, Simren M, Tack J, Whitehead WE, Dumitrascu DL, Fang X, Fukudo S, Kellow J, Okeke E, Quigley EMM, Schmulson M, Whorwell P, Archampong T, Adibi P, Andresen V, Benninga MA, Bonaz B, Bor S, Fernandez LB, Choi SC, Corazziari ES, Francisconi C, Hani A, Lazebnik L, Lee YY, Mulak A, Rahman MM, Santos J, Setshedi M, Syam AF, Vanner S, Wong RK, Lopez-Colombo A, Costa V, Dickman R, Kanazawa M, Keshteli AH, Khatun R, Maleki I, Poitras P, Pratap N, Stefanyuk O, Thomson S, Zeevenhooven J, Palsson OS. Worldwide Prevalence and Burden of Functional Gastrointestinal Disorders, Results of Rome Foundation Global Study. Gastroenterology. 2021;160(1):99-114.e3.
- Drossman DA, Hasler WL. Rome IV—Functional GI Disorders: Disorders of Gut-Brain Interaction. Gastroenterology. 2016;150(6):1257–61.
- Kasarello K, Cudnoch-Jedrzejewska A, Czarzasta K. Communication of gut microbiota and brain via immune and neuroendocrine signaling. Front Microbiol. 2023;14:1118529.
- Radovanovic-Dinic B, Tesic-Rajkovic S, Grgov S, Petrovic G, Zivkovic V. Irritable bowel syndrome - from etiopathogenesis to therapy. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2018;162(1):1–9.
- Saha L. Irritable bowel syndrome: pathogenesis, diagnosis, treatment, and evidence-based medicine. WJG. 2014;20(22):6759–73.
- Shang X, E FF, Guo KL, Li YF, Zhao HL, Wang Y, Chen N, Nian T, Yang CQ, Yang KH, Li XX. Effectiveness and Safety of Probiotics for Patients with Constipation-Predominant Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis of 10 Randomized Controlled Trials. Nutrients. 2022;14(12):2482.
- Camilleri M. Diagnosis and Treatment of Irritable Bowel Syndrome: A Review. JAMA. 2021;325(9):865–77.
- Serra J. Management of bloating. Neurogastroenterology & Motility. 2022;34(3):e14333.
- Lacy BE, Cangemi D, Vazquez-Roque M. Management of Chronic Abdominal Distension and Bloating. Clinical Gastroenterology and Hepatology. 2021;19(2):219-231.e1.
- Jiang X, G R Locke III, Choung RS, Zinsmeister AR, Schleck CD, Talley NJ. Prevalence and risk factors for abdominal bloating and visible distention: A population-based study. Gut. 2008;57(6):756.
- Chang L, Lee OY, Naliboff B, Schmulson M, Mayer EA. Sensation of bloating and visible abdominal distension in patients with irritable bowel syndrome. Am J Gastroenterol. 2001;96(12):3341–7.
- Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical review. JAMA. 2015;313(9):949–58.
- Lacy BE, Mearin F, Chang L, Chey WD, Lembo AJ, Simren M, Spiller R. Bowel Disorders. Gastroenterology. 2016;150(6):1393-1407.e5.
- Chang L, Lembo A, Sultan S. American Gastroenterological Association Institute Technical Review on the pharmacological management of irritable bowel syndrome. Gastroenterology. 2014;147(5):1149-1172.e2.
- Eriksson EM, Andrén KI, Kurlberg GK, Eriksson HT. Aspects of the non-pharmacological treatment of irritable bowel syndrome. WJG. 2015;21(40):11439–49.
- Halland M, Saito YA. Irritable bowel syndrome: new and emerging treatments. BMJ. 2015;350:h1622.
- Jent S, Bez NS, Haddad J, Catalano L, Egger KS, Raia M, Tedde GS, Rogler G. The efficacy and real-world effectiveness of a diet low in fermentable oligo-, di-, monosaccharides and polyols in irritable bowel syndrome: A systematic review and meta-analysis. Clinical Nutrition. 2024;43(6):1551–62.
- Galica AN, Galica R, Dumitrașcu DL. Diet, fibers, and probiotics for irritable bowel syndrome. JMedLife. 2022;15(2):174–9.
- Napolitano M, Fasulo E, Ungaro F, Massimino L, Sinagra E, Danese S, Mandarino FV. Gut Dysbiosis in Irritable Bowel Syndrome: A Narrative Review on Correlation with Disease Subtypes and Novel Therapeutic Implications. Microorganisms. 2023;11(10):2369.
- Zhang T, Zhang C, Zhang J, Sun F, Duan L. Efficacy of Probiotics for Irritable Bowel Syndrome: A Systematic Review and Network Meta-Analysis. Front Cell Infect Microbiol. 2022.
- Catanzaro R, Sciuto M, Marotta F. Lactose intolerance: An update on its pathogenesis, diagnosis, and treatment. Nutrition Research. 2021;89:23–34.
- Böhn L, Törnblom H, Van Oudenhove L, Simrén M, Störsrud S. A randomized double-blind placebo-controlled crossover pilot study: Acute effects of the enzyme α-galactosidase on gastrointestinal symptoms in irritable bowel syndrome patients. Neurogastroenterol Motil. 2021;33(7):e14094.
- de Vrese M, Laue C, Offick B, Soeth E, Repenning F, Thoß A, Schrezenmeir J. A combination of acid lactase from Aspergillus oryzae and yogurt bacteria improves lactose digestion in lactose maldigesters synergistically: A randomized, controlled, double-blind cross-over trial. Clin Nutr. 2015;34(3):394–9.
- Zam W, Quispe C, Sharifi-Rad J, López MD, Schoebitz M, Martorell M, Sharopov F, Fokou PVT, Mishra AP, Chandran D, Kumar M, Chen JT, Pezzani R. An Updated Review on The Properties of Melissa officinalis L.: Not Exclusively Anti-anxiety. Front Biosci (Schol Ed). 2022;14(2):16.
- Miraj S, Rafieian-Kopaei, Kiani S. Melissa officinalis L: A Review Study With an Antioxidant Prospective. J Evid Based Complementary Altern Med. 2017;22(3):385–94.
- Roncoroni L, Bascuñán KA, Doneda L, et al. Correction: Roncoroni, L. et al. A Low FODMAP Gluten-Free Diet Improves Functional Gastrointestinal Disorders and Overall Mental Health of Celiac Disease Patients: A Randomized Controlled Trial. Nutrients 2018, 10, 1023. Nutrients. 2019;11(3):566.
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977.
- Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473–83.
- Derogatis LR, Lipman RS, Covi L, Rickels K, Uhlenhuth EH. Dimensions of outpatient neurotic pathology: Comparison of a clinical versus an empirical assessment. Journal of Consulting and Clinical Psychology. 1970;34(2):164–71.
- Mayer EA, Nance K, Chen S. The Gut-Brain Axis. Annu Rev Med. 2022;73:439–53.
- Wang Q, Yang Q, Liu X. The microbiota–gut–brain axis and neurodevelopmental disorders. Protein & Cell. 2023;14(10):762–75.
- Satish Kumar L, Pugalenthi LS, Ahmad M, Reddy S, Barkhane Z, Elmadi J. Probiotics in Irritable Bowel Syndrome: A Review of Their Therapeutic Role. Cureus. 2022;14(4):e24240.
- Cozma-Petruţ A, Loghin F, Miere D, Dumitraşcu DL. Diet in irritable bowel syndrome: What to recommend, not what to forbid to patients! WJG. 2017;23(21):3771.
- Zhuang X, Xiong L, Li L, Li M, Chen M. Alterations of gut microbiota in patients with irritable bowel syndrome: A systematic review and meta-analysis. J of Gastro and Hepatol. 2017;32(1):28–38.
- Kerckhoffs AP, Samsom M, Rest MEVD, Vogel JD, Knol J, Ben-Amor K, Akkermans LM. Lower Bifidobacteria counts in both duodenal mucosa-associated and fecal microbiota in irritable bowel syndrome patients. WJG. 2009;15(23):2887.
- Malinen E, Rinttilä T, Kajander K, Mättö J, Kassinen A, Krogius L, Saarela M, Korpela R, Palva A. Analysis of the fecal microbiota of irritable bowel syndrome patients and healthy controls with real-time PCR. Am J Gastroenterol. 2005;100(2):373–82.
- O’Mahony L, McCarthy J, Kelly P, Hurley G, Luo F, Chen K, O’Sullivan GC, Kiely B, Collins JK, Shanahan F, Quigley EMM. Lactobacillus and bifidobacterium in irritable bowel syndrome: Symptom responses and relationship to cytokine profiles. Gastroenterology. 2005;128(3):541–51.
- Ringel-Kulka T, Palsson OS, Maier D, Carroll I, Galanko JA, Leyer G, Ringel Y. Probiotic Bacteria Lactobacillus acidophilus NCFM and Bifidobacterium lactis Bi-07 Versus Placebo for the Symptoms of Bloating in Patients With Functional Bowel Disorders: A Double-blind Study. Journal of Clinical Gastroenterology. 2011;45(6):518–25.
- Quigley EMM. Probiotics in Irritable Bowel Syndrome: The Science and the Evidence. Journal of Clinical Gastroenterology. 2015;49:S60.
- Hyland NP, Quigley EM, Brint E. Microbiota-host interactions in irritable bowel syndrome: Epithelial barrier, immune regulation and brain-gut interactions. WJG. 2014;20(27):8859.
- Latif A, Shehzad A, Niazi S, Zahid A, Ashraf W, Iqbal MW, Rehman A, Riaz T, Aadil RM, Khan IM, Özogul F, Rocha JM, Esatbeyoglu T, Korma SA. Probiotics: mechanism of action, health benefits and their application in food industries. Front Microbiol. 2023;14:1216674.
- Ianiro G, Pecere S, Giorgio V, Gasbarrini A, Cammarota G. Digestive Enzyme Supplementation in Gastrointestinal Diseases. CDM. 14 2016;17(2):187–93.
- Vejdani R, Shalmani HRM, Mir-Fattahi M, Sajed-Nia F, Abdollahi M, Zali MR, Alizadeh AHM, Bahari A, Amin G. The Efficacy of an Herbal Medicine, Carmint, on the Relief of Abdominal Pain and Bloating in Patients with Irritable Bowel Syndrome: A Pilot Study. Dig Dis Sci. 2006;51(8):1501–7.
- Konstantis G, Efstathiou S, Pourzitaki C, Kitsikidou E, Germanidis G, Chourdakis M. Efficacy and safety of probiotics in the treatment of irritable bowel syndrome: A systematic review and meta-analysis of randomised clinical trials using ROME IV criteria. Clin Nutr. 2023; 42(5):800–9.
| ACTIVE GROUP (n=15) | PLACEBO GROUP (n=14) | Comparison ACTIVE and PLACEBO | |||
|---|---|---|---|---|---|
|
Women, n (%) Men, n (%) |
11 (73.3) 4 (26.7) |
12 (85.7) 2 (14.3) |
|||
| Mean±SD |
Median (Min-Max) |
Mean±SD |
Median (Min-Max) |
p-value | |
| Age (years) | 44.0 ± 14.3 | 42 (24-64) | 46.1 ± 13.1 | 51 (25-61) | 0.68 |
| Weight (kg) | 62.1 ± 11.2 | 58 (46-87) | 62.7 ± 10.3 | 59.5 (52-82) | 0.87 |
| Height (m) | 1.7 ± 0.1 | 1.7 (1.5-1.9) | 1.7 ± 0.1 | 1.7 (1.6-1.8) | 0.74 |
| BMI (kg/m²) | 22.3 ± 2.7 | 22.3 (18-27) | 22.4 ± 3.0 | 21.6 (19.5-29) | 0.93 |
| ACTIVE GROUP (n=15) | PLACEBO GROUP (n=14) | |||
|---|---|---|---|---|
| Basal visit (T0) | Final visit (T1) |
Basal visit (T0) |
Final visit (T1) |
|
| Mean ± SEM | 5.64 ± 1.74 | 7.15 ± 1.00 | 6.89 ± 1.42 | 6.31 ± 1.79 |
| Median | 6.30 | 7.10 | 7.05 | 5.95 |
| Min-Max | 1.00 – 9.00 | 4.00 -10.00 | 3.00 – 10.00 | 0.90 – 10.00 |
| Variation Final vs Basal visit | Variation Final vs Basal visit | |||
| Mean ± SD | 1.51 ± 2.49 | -0.58 ± 3.14 | ||
| p-value | *0.052 | |||
| ACTIVE GROUP (n=15) | PLACEBO GROUP (n=14) | |||
|---|---|---|---|---|
| Basal visit (T0) | Final visit (T1) |
Basal visit (T0) |
Final visit (T1) |
|
| Mean ± SEM | 101.53 ± 4.58 | 103.20 ± 4.09 | 99.86 ± 5.65 | 99.71 ± 7.70 |
| Median | 102.00 | 105.00 | 101.00 | 101.00 |
| Min-Max | 89.00 – 106.00 | 96.00 – 109.00 | 91.00 – 108.00 | 79.00 – 109.00 |
| Variation Final vs Basal visit | Variation Final vs Basal visit | |||
| Mean ± SD | 1.67 ± 3.66 | -0.14 ± 7.04 | ||
| p-value | 0.388 | |||
| ACTIVE GROUP (n=15) | PLACEBO GROUP (n=14) | |||
| Basal visit (T0) |
Final visit (T1) |
Basal visit (T0) |
Final visit (T1) |
|
| Mean ± SEM | 101.53 ± 4.58 | 103.20 ± 4.09 | 99.86 ± 5.65 | 99.71 ± 7.70 |
| Median | 102.00 | 105.00 | 101.00 | 101.00 |
| Min-Max | 89.00 – 106.00 | 96.00 – 109.00 | 91.00 – 108.00 | 79.00 – 109.00 |
| Variation Final vs Basal visit | Variation Final vs Basal visit | |||
| Mean ± SD | 1.67 ± 3.66 | -0.14 ± 7.04 | ||
| p-value | 0.388 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




