Submitted:
05 March 2025
Posted:
06 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
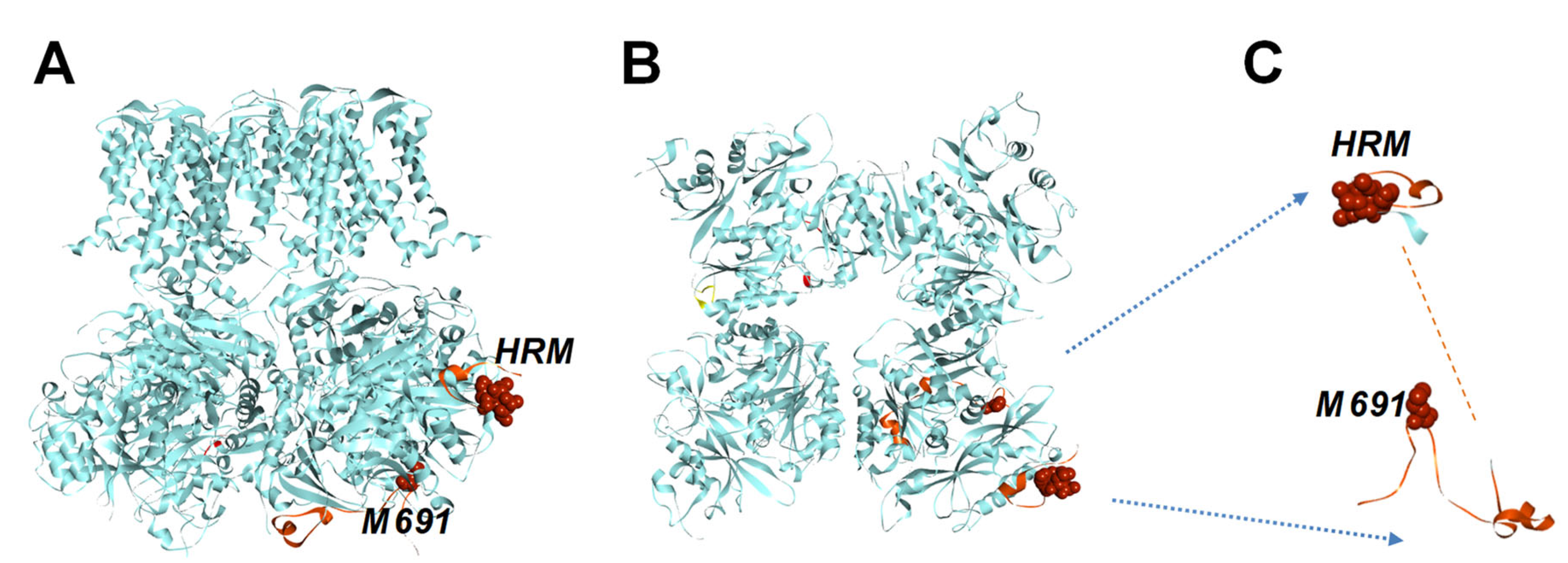
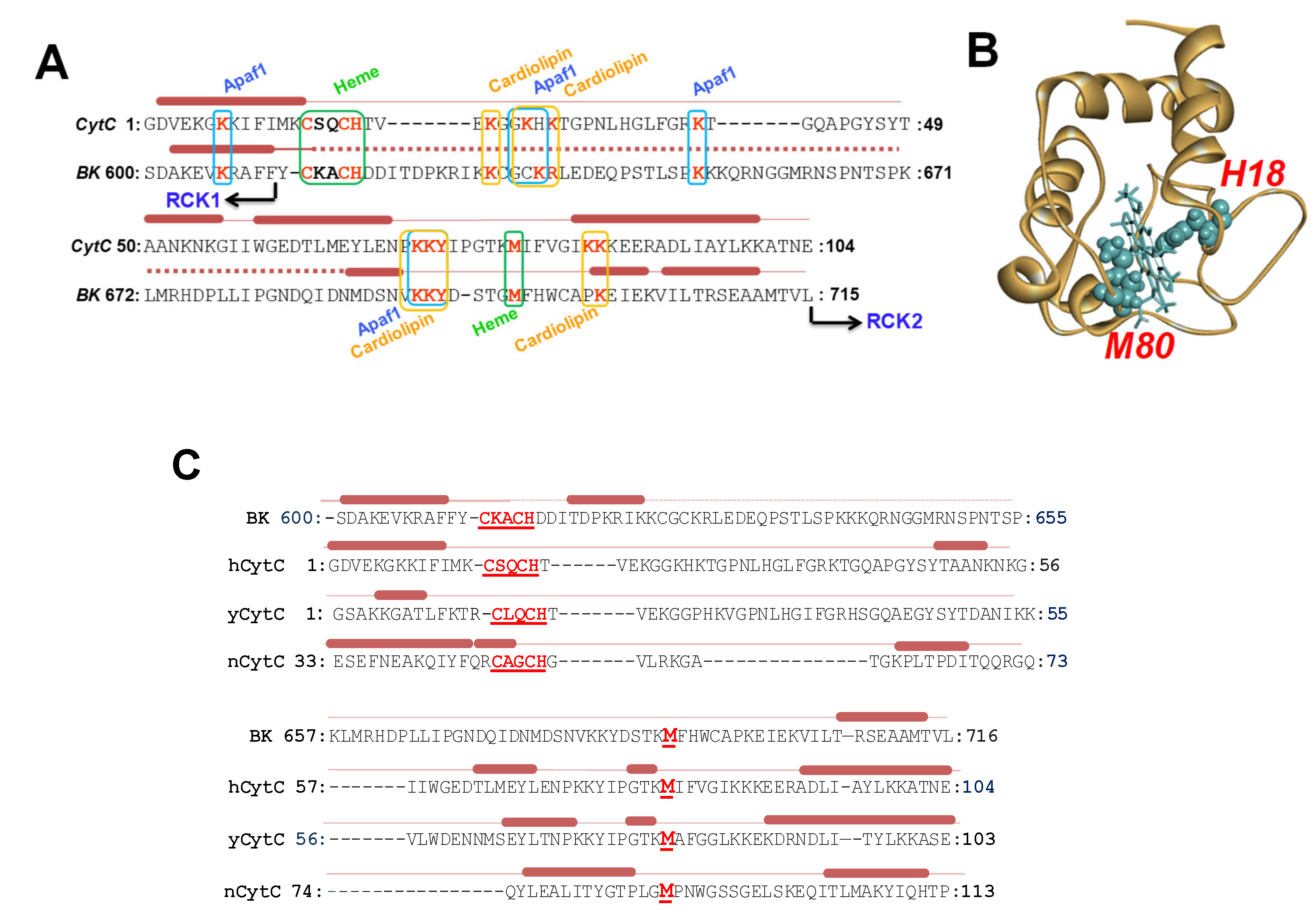
2.1. The Human BK Channel Structure Comprises a Cytochrome c-like Feature Within the Gating Ring
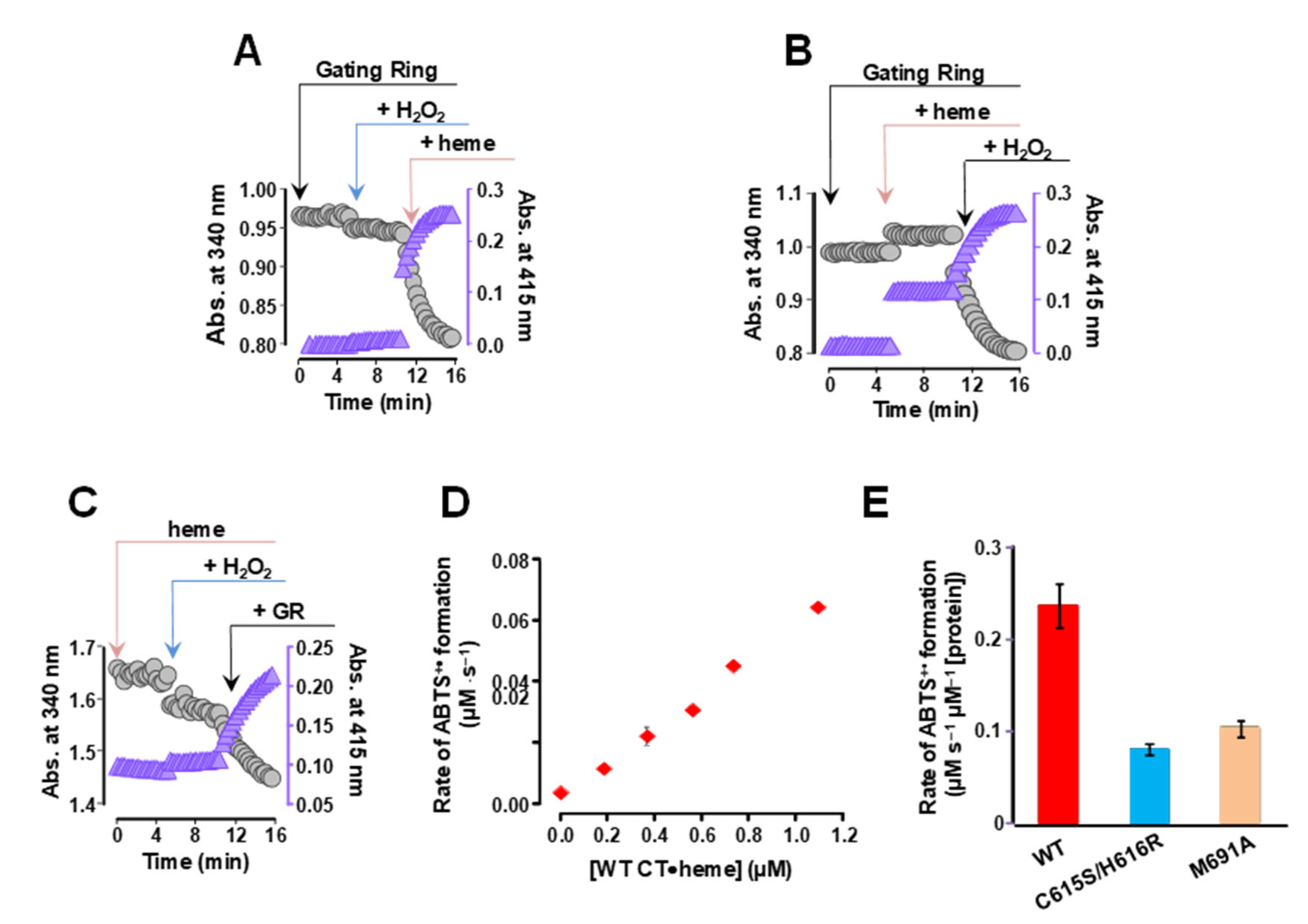
2.2. The Human BK Channel Confers Peroxidase Activity
2.3. Peroxidase Activity of the Human BK Channel C-Terminal Domain
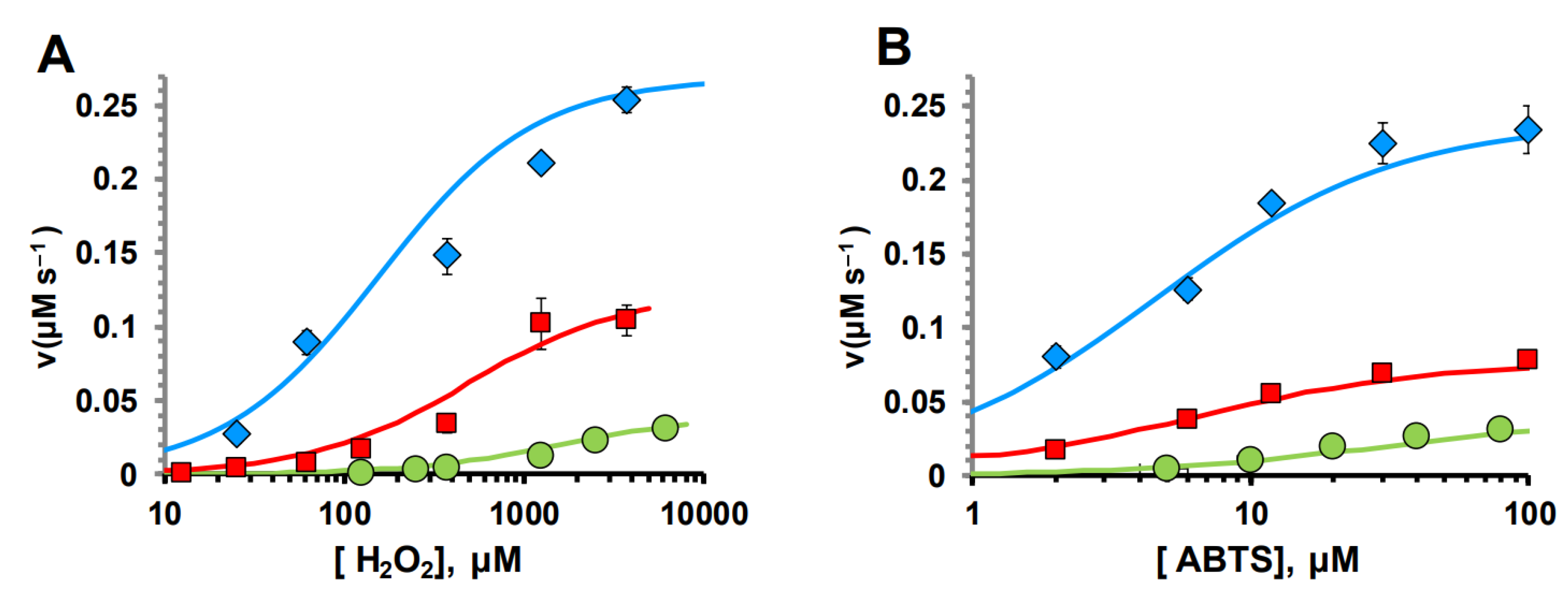
2.4. STREX Significantly Augmented CTD Peroxidase Activity
2.5. The Full Human BK Channel Exhibits Peroxidase Activity
3. Discussion
3.1. Cytochrome-c-like Structure Within BK Channel Gating Ring
3.2. The Novel Multifunctional Module Within BK Channels
4. Materials and Methods
4.1. Expression and Purification of BK-Terminal Proteins
4.2. ABTS Assay
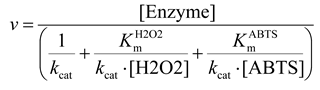
4.3. Peroxidase Activity Assay in Cell Lysates
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoshi, T.; Pantazis, A.; Olcese, R. Transduction of Voltage and Ca2+Signals by Slo1 BK Channels. Physiology 2013, 28, 172–189. [Google Scholar] [CrossRef]
- Cui, J. BK Channel Gating Mechanisms: Progresses Toward a Better Understanding of Variants Linked Neurological Diseases. Front. Physiol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, F.; Gonzalez-Sanabria, N.; Alvarado-Sanchez, R.; Fernández, M.; Castillo, K.; Latorre, R. Large conductance voltage-and calcium-activated K+ (BK) channel in health and disease. Front. Pharmacol. 2024, 15, 1373507. [Google Scholar] [CrossRef] [PubMed]
- Sancho, M.; Kyle, B.D. The Large-Conductance, Calcium-Activated Potassium Channel: A Big Key Regulator of Cell Physiology. Front. Physiol. 2021, 12. [Google Scholar] [CrossRef]
- Yusifov, T.; Savalli, N.; Pantazis, A.; Heinemann, S.H.; Hoshi, T.; Olcese, R. Carbon Monoxide May Regulate BK slo1 Channel Activity by Partially Disrupting Heme Coordination. Biophys. J. 2017, 112, 112a. [Google Scholar] [CrossRef]
- Yusifov, T.; Javaherian, A.; Gandhi, C.; Olcese, R. Calcium-Dependent Operation of the Human BK Channel Gating Ring Apparatus. Biophys. J. 2011, 100, 581a. [Google Scholar] [CrossRef]
- Yusifov, T. , Javaherian, AD., Heinemann, SH., Hoshi T., Olcese R. The human BK Channel Gating Ring is a PIP2 Sensor. The human BK Channel Gating Ring is a PIP2 Sensor. Am. Soc. Anesthesiol. Annu. Meet. Pap. ( 2012. [CrossRef]
- Javaherian, A.D.; Yusifov, T.; Pantazis, A.; Franklin, S.; Gandhi, C.S.; Olcese, R. Metal-driven Operation of the Human Large-conductance Voltage- and Ca2+-dependent Potassium Channel (BK) Gating Ring Apparatus. J. Biol. Chem. 2011, 286, 20701–20709. [Google Scholar] [CrossRef]
- Pantazis, A.; Olcese, R. . Biophysics of BK Channel Gating. Int Rev Neurobiol.
- Hou, S.; Heinemann, S.H.; Hoshi, T. Modulation of BKCaChannel Gating by Endogenous Signaling Molecules. Physiology 2009, 24, 26–35. [Google Scholar] [CrossRef]
- Lewandowska, J.; Kalenik, B.; Wrzosek, A.; Szewczyk, A. Redox Regulation of Mitochondrial Potassium Channels Activity. Antioxidants 2024, 13, 434. [Google Scholar] [CrossRef]
- Hoshi, T. & S.H. Heinemann. Modulation of BK Channels by Small Endogenous Molecules and Pharmaceutical Channel Openers. (2016). International Review of Neurobiology. [CrossRef]
- Tao, X.; MacKinnon, R.; States, U. Molecular structures of the human Slo1 K+ channel in complex with β4. eLife 2019, 8. [Google Scholar] [CrossRef]
- Hite, R.K.; Tao, X.; MacKinnon, R. Structural basis for gating the high-conductance Ca2+-activated K+ channel. Nature 2016, 541, 52–57. [Google Scholar] [CrossRef]
- Yuan, P.; Leonetti, M.D.; Hsiung, Y.; MacKinnon, R. Open structure of the Ca2+ gating ring in the high-conductance Ca2+-activated K+ channel. Nature 2011, 481, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Hite, R.K.; MacKinnon, R. Cryo-EM structure of the open high-conductance Ca2+-activated K+ channel. Nature 2016, 541, 46–51. [Google Scholar] [CrossRef]
- Peng Yuan, Manuel D Leonetti, Alexander R Pico, Yichun Hsiung, R. M. Structure of the Human BK Channel Ca2+-Activation Apparatus at 3.0 Å Resolution. Science (80-. ). 182–186 (2010).
- Walewska, A.; Szewczyk, A.; Koprowski, P. External Hemin as an Inhibitor of Mitochondrial Large-Conductance Calcium-Activated Potassium Channel Activity. Int. J. Mol. Sci. 2022, 23, 13391. [Google Scholar] [CrossRef] [PubMed]
- Horrigan, F.T.; Heinemann, S.H.; Hoshi, T. Heme Regulates Allosteric Activation of the Slo1 BK Channel. J. Gen. Physiol. 2005, 126, 7–21. [Google Scholar] [CrossRef]
- Tang, X.D.; Xu, R.; Reynolds, M.F.; Garcia, M.L.; Heinemann, S.H.; Hoshi, T. Haem can bind to and inhibit mammalian calcium-dependent Slo1 BK channels. Nature 2003, 425, 531–535. [Google Scholar] [CrossRef]
- Sahoo, N.; Yang, K.; Coburger, I.; Bernert, A.; Swain, S.M.; Gessner, G.; Kappl, R.; Kühl, T.; Imhof, D.; Hoshi, T.; et al. Intracellular hemin is a potent inhibitor of the voltage-gated potassium channel Kv10.1. Sci. Rep. 2022, 12, 1–12. [Google Scholar] [CrossRef]
- Burton, M.J.; Kapetanaki, S.M.; Chernova, T.; Jamieson, A.G.; Dorlet, P.; Santolini, J.; Moody, P.C.E.; Mitcheson, J.S.; Davies, N.W.; Schmid, R.; et al. A heme-binding domain controls regulation of ATP-dependent potassium channels. Proc. Natl. Acad. Sci. 2016, 113, 3785–3790. [Google Scholar] [CrossRef]
- Wang, S.; Publicover, S.; Gu, Y. An oxygen-sensitive mechanism in regulation of epithelial sodium channel. Proc. Natl. Acad. Sci. 2009, 106, 2957–2962. [Google Scholar] [CrossRef]
- Bertini, I. &, Gabriele Cavallaro, A. R. Cytochrome c: occurrence and functions. J Biol Chem. 15194–15200 (2006).
- Kagan, V. E.; et al. Cytochrome c/cardiolipin relations in mitochondria: a kiss of death. Cytochrome c/cardiolipin relations mitochondria a kiss death 1439–1453 (2010). [CrossRef]
- Yi, L.; Morgan, J.T.; Ragsdale, S.W. Identification of a Thiol/Disulfide Redox Switch in the Human BK Channel That Controls Its Affinity for Heme and CO. J. Biol. Chem. 2010, 285, 20117–20127. [Google Scholar] [CrossRef] [PubMed]
- Lawrence Salkoff, Alice Butler, Gonzalo Ferreira, C. S. & A. W. High-conductance potassium channels of the SLO family. Nat. Rev. Neurosci. 2006. [CrossRef]
- Cunha, C. A.; et al. Cytochrome c nitrite reductase from Desulfovibrio desulfuricans ATCC 27774. The relevance of the two calcium sites in the structure of the catalytic subunit (NrfA). J. Biol. Chem. 1746. [Google Scholar]
- Oubrie, A. , Rozeboom, H. J., Kalk, K. H., Huizinga, E. G. & Dijkstra, B. W. Crystal structure of quinohemoprotein alcohol dehydrogenase from Comamonas testosteroni: Structural basis for substrate oxidation and electron transfer. J. Biol. Chem. 277, 3727–3732 (2002).
- Ow, Y.-L.P.; Green, D.R.; Hao, Z.; Mak, T.W. Cytochrome c: functions beyond respiration. Nat. Rev. Mol. Cell Biol. 2008, 9, 532–542. [Google Scholar] [CrossRef]
- Steele, H.B.B.; Elmer-Dixon, M.M.; Rogan, J.T.; Ross, J.B.A.; Bowler, B.E. The Human Cytochrome c Domain-Swapped Dimer Facilitates Tight Regulation of Intrinsic Apoptosis. Biochemistry 2020, 59, 2055–2068. [Google Scholar] [CrossRef] [PubMed]
- Santucci, R.; Sinibaldi, F.; Cozza, P.; Polticelli, F.; Fiorucci, L. Cytochrome c: An extreme multifunctional protein with a key role in cell fate. Int. J. Biol. Macromol. 2019, 136, 1237–1246. [Google Scholar] [CrossRef]
- Ascenzi, P.; Polticelli, F.; Marino, M.; Santucci, R.; Coletta, M. Cardiolipin drives cytochrome c proapoptotic and antiapoptotic actions. IUBMB Life 2011, 63, 160–165. [Google Scholar] [CrossRef]
- Belikova, N.A.; Jiang, J.; Tyurina, Y.Y.; Zhao, Q.; Epperly, M.W.; Greenberger, J.; Kagan, V.E. Cardiolipin-Specific Peroxidase Reactions of Cytochrome c in Mitochondria During Irradiation-Induced Apoptosis. Int. J. Radiat. Oncol. 2007, 69, 176–186. [Google Scholar] [CrossRef]
- Latorre, R.; Castillo, K.; Carrasquel-Ursulaez, W.; Sepulveda, R.V.; Gonzalez-Nilo, F.; Gonzalez, C.; Alvarez, O. Molecular Determinants of BK Channel Functional Diversity and Functioning. Physiol. Rev. 2017, 97, 39–87. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Coghill, L.S.; McClafferty, H.; MacDonald, S.H.-F.; Antoni, F.A.; Ruth, P.; Knaus, H.-G.; Shipston, M.J. Distinct stoichiometry of BK Ca channel tetramer phosphorylation specifies channel activation and inhibition by cAMP-dependent protein kinase. Proc. Natl. Acad. Sci. 2004, 101, 11897–11902. [Google Scholar] [CrossRef]
- Shipston, M.J. Ion Channel Regulation by Protein Palmitoylation. J. Biol. Chem. 2011, 286, 8709–8716. [Google Scholar] [CrossRef]
- Yeudall, S.; Upchurch, C.M.; Leitinger, N. The clinical relevance of heme detoxification by the macrophage heme oxygenase system. Front. Immunol. 2024, 15, 1379967. [Google Scholar] [CrossRef]
- Kubo, Y. A new world of heme function. Pfl?gers Arch. Eur. J. Physiol. 2020, 472, 547–548. [Google Scholar] [CrossRef]
- Coburger, I.; Yang, K.; Bernert, A.; Wiesel, E.; Sahoo, N.; Swain, S.M.; Hoshi, T.; Schönherr, R.; Heinemann, S.H. Impact of intracellular hemin on N-type inactivation of voltage-gated K+ channels. Pfl?gers Arch. Eur. J. Physiol. 2020, 472, 551–560. [Google Scholar] [CrossRef]
- Kang, S.A.; Crane, B.R. Effects of interface mutations on association modes and electron-transfer rates between proteins. Proc. Natl. Acad. Sci. 2005, 102, 15465–15470. [Google Scholar] [CrossRef]
- Nugraheni, A.D.; Ren, C.; Matsumoto, Y.; Nagao, S.; Yamanaka, M.; Hirota, S. Oxidative modification of methionine80 in cytochrome c by reaction with peroxides. J. Inorg. Biochem. 2018, 182, 200–207. [Google Scholar] [CrossRef]
- Patel, N. H.; et al. Inhibition of BKCa negatively alters cardiovascular function. Physiol. Rep. 6, 1–11 (2018).
- Szteyn, K. & Singh, H. Bkca channels as targets for cardioprotection. Antioxidants 9, 1–16 (2020).
- Soltysinska, E.; Bentzen, B.H.; Barthmes, M.; Hattel, H.; Thrush, A.B.; Harper, M.-E.; Qvortrup, K.; Larsen, F.J.; Schiffer, T.A.; Losa-Reyna, J.; et al. KCNMA1 Encoded Cardiac BK Channels Afford Protection against Ischemia-Reperfusion Injury. PLOS ONE 2014, 9, e103402. [Google Scholar] [CrossRef]
- Yusifov, T.; Savalli, N.; Gandhi, C.S.; Ottolia, M.; Olcese, R. The RCK2 domain of the human BK Ca channel is a calcium sensor. Proc. Natl. Acad. Sci. 2008, 105, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Yusifov, T.; Javaherian, A.D.; Pantazis, A.; Gandhi, C.S.; Olcese, R. The RCK1 domain of the human BKCa channel transduces Ca2+ binding into structural rearrangements. J. Gen. Physiol. 2010, 136, 189–202. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N. & Woody, R. W. Computation and Analysis of Protein Circular Dichroism Spectra. Methods Enzymol.
- Meredith, A.L. BK Channelopathies andKCNMA1-Linked Disease Models. Annu. Rev. Physiol. 2024, 86, 277–300. [Google Scholar] [CrossRef]
- Yusifov, T.; Qudretova, F.; Aliyeva, A. Role of Bioelectrical Signaling Networks in Tumor Growth. Am. J. Biomed. Life Sci. 2024, 12, 83–92. [Google Scholar] [CrossRef]
- Sahoo, N.; Goradia, N.; Ohlenschläger, O.; Schönherr, R.; Friedrich, M.; Plass, W.; Kappl, R.; Hoshi, T.; Heinemann, S.H. Heme impairs the ball-and-chain inactivation of potassium channels. Proc. Natl. Acad. Sci. 2013, 110, E4036–E4044. [Google Scholar] [CrossRef]
- Williams, S.E.J.; Wootton, P.; Mason, H.S.; Bould, J.; Iles, D.E.; Riccardi, D.; Peers, C.; Kemp, P.J. Hemoxygenase-2 Is an Oxygen Sensor for a Calcium-Sensitive Potassium Channel. Science 2004, 306, 2093–2097. [Google Scholar] [CrossRef] [PubMed]
- Yusifov, T.; Savalli, N.; Pantazis, A.; Heinemann, S.H.; Hoshi, T.; Olcese, R. Carbon Monoxide May Regulate BK slo1 Channel Activity by Partially Disrupting Heme Coordination. Biophys. J. 2017, 112, 112a. [Google Scholar] [CrossRef]
- Jaggar, J.H.; Li, A.; Parfenova, H.; Liu, J.; Umstot, E.S.; Dopico, A.M.; Leffler, C.W. Heme Is a Carbon Monoxide Receptor for Large-Conductance Ca 2+ -Activated K + Channels. Circ. Res. 2005, 97, 805–812. [Google Scholar] [CrossRef]
- Yi, L.; Morgan, J.T.; Ragsdale, S.W. Identification of a Thiol/Disulfide Redox Switch in the Human BK Channel That Controls Its Affinity for Heme and CO. J. Biol. Chem. 2010, 285, 20117–20127. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, J.; Wasson, J.; Salkoff, L.; Permutt, M.A. Cloning of human pancreatic islet large conductance Ca2+-activated K+ channel (hSlo) cDNAs: evidence for high levels of expression in pancreatic islets and identification of a flanking genetic marker. Diabetologia 1996, 39, 891–898. [Google Scholar] [CrossRef]
- Saito, M.; Nelson, C.; Salkoff, L.; Lingle, C.J. A Cysteine-rich Domain Defined by a Novel Exon in aSlo Variant in Rat Adrenal Chromaffin Cells and PC12 Cells. 1997, 272, 11710–11717. [CrossRef]
- Xie, J.; McCobb, D.P. Control of Alternative Splicing of Potassium Channels by Stress Hormones. Science 1998, 280, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Burton, M.J.; Cresser-Brown, J.; Thomas, M.; Portolano, N.; Basran, J.; Freeman, S.L.; Kwon, H.; Bottrill, A.R.; Llansola-Portoles, M.J.; Pascal, A.A.; et al. Discovery of a heme-binding domain in a neuronal voltage-gated potassium channel. J. Biol. Chem. 2020, 295, 13277–13286. [Google Scholar] [CrossRef]
- Zhou, Y.; Lingle, C.J. Paxilline inhibits BK channels by an almost exclusively closed-channel block mechanism. J. Gen. Physiol. 2014, 144, 415–440. [Google Scholar] [CrossRef]
- Shipston, M. J. , & Tian, L. (2016). Posttranscriptional and Posttranslational Regulation of BKChannels. ( 128, 91–126. [CrossRef]
- McCartney, C.E.; McClafferty, H.; Huibant, J.-M.; Rowan, E.G.; Shipston, M.J.; Rowe, I.C.M. A cysteine-rich motif confers hypoxia sensitivity to mammalian large conductance voltage- and Ca-activated K (BK) channel α-subunits. Proc. Natl. Acad. Sci. 2005, 102, 17870–17876. [Google Scholar] [CrossRef]
- Jeng, W.-Y. , Shiu, J.-H., Tsai, Y.-H., Chuang, W.-J (2003) Solution Structure of Reduced Recombinant Human Cytochrome c. [CrossRef]
- Baistrocchi, P.; Banci, L.; Bertini, I.; Turano, P.; Bren, K.L.; Gray, H.B. Three-Dimensional Solution Structure of Saccharomyces cerevisiae Reduced Iso-1-cytochrome c. Biochemistry 1996, 35, 13788–13796. [Google Scholar] [CrossRef] [PubMed]
- Nurizzo, D.; Silvestrini, M.-C.; Mathieu, M.; Cutruzzolà, F.; Bourgeois, D.; Fülöp, V.; Hajdu, J.; Brunori, M.; Tegoni, M.; Cambillau, C. N-terminal arm exchange is observed in the 2.15 Å crystal structure of oxidized nitrite reductase from Pseudomonas aeruginosa. Structure 1997, 5, 1157–1171. [Google Scholar] [CrossRef] [PubMed]
- Oubrie, A.; Rozeboom, H.J.; Kalk, K.H.; Huizinga, E.G.; Dijkstra, B.W. Crystal Structure of Quinohemoprotein Alcohol Dehydrogenase from Comamonas testosteroni. J. Biol. Chem. 2002, 277, 3727–3732. [Google Scholar] [CrossRef] [PubMed]
- Satoh, A.; Kim, J.-K.; Miyahara, I.; Devreese, B.; Vandenberghe, I.; Hacisalihoglu, A.; Okajima, T.; Kuroda, S.; Adachi, O.; Duine, J.A.; et al. Crystal Structure of Quinohemoprotein Amine Dehydrogenase from Pseudomonas putida. J. Biol. Chem. 2002, 277, 2830–2834. [Google Scholar] [CrossRef]



| WT GR Javaherian, et al., 2010 | (C615R/H616S) GR In this study | M691A GR In this study | |
| α-helix (%) | 28.6 | 27.8 | 28.0 |
| nα-helix | 21.9 | 21.5 | 21.3 |
| β-strand (%) | 21.8 | 22.1 | 21.1 |
| nβ-strand | 31.2 | 30.9 | 30.5 |
| Turn + Unordered (%) | 49.7 | 50.2 | 50.8 |
| NRMSD | 0.018 | 0.015 | 0.017 |
| Protein | Km H2O2 (mM) | KmABTS (mM) | kcat |
|---|---|---|---|
| GR | 0.512±0.1 | 0.012±0.001 | 0.14±0.02 |
| GR/STREX | 0.15±0.01 | 0.0043±0.0009 | 0.29±0.018 |
| CytC | 5.49±0.34 | 0.1±0.002 | 0.13±0.0165 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




