Submitted:
26 February 2025
Posted:
28 February 2025
You are already at the latest version
Abstract
Vitamin D-dependent rickets type 1A (VDDR1A) is a rare autosomal recessive disorder caused by mutations in the CYP27B1 gene, leading to a deficiency in active vitamin D (1,25-dihydroxyvitamin D). This study examines the genotypic and phenotypic characteristics of VDDR1A in Vietnamese children. A retrospective analysis was conducted on 19 Vietnamese children diagnosed with VDDR1A. Clinical, radiological, biochemical, and molecular data were collected. Rickets severity score (RSS), biochemical parameters, and height standard deviation score (HtSDS) were used to assess the severity of the condition. The study included 19 children from 17 families (10 males and nine females). The median age of rickets diagnosis was 19.2 months and with VDDR1A, while the median age of VDDR1A diagnosis was 7.5 months. Common symptoms among the children included thickened wrists and ankles (19/19), genu varum or genu valgum (18/19), failure to thrive (18/19), rachitic rosary (12/19), and delayed walking (11/19). Radiographic features showed that all children had cupping, splaying, and fraying, 12 children had rachitic rosary, and six exhibited pseudofracture. Biochemical findings showed severe hypocalcemia, normal or mildly low serum phosphate, elevated alkaline phosphatase and parathyroid hormone levels, and normal serum 25-hydroxyvitamin D level. Genetic analysis identified biallelic CYP27B1 variants, including one known pathogenic frameshift mutation c.1319_1325dup p.(Phe443Profs*24), one novel likely pathogenic missense variant c.616C>T p.(Arg206Cys), and one novel pathogenic frameshift mutation c.96_97del p.(Ala33Thrfs*299). The c.1319_1325dup p.(Phe443Profs*24) variant was the most common, present in 18 out of 19 children. Children with VDDR1A in this study presented with growth failure and skeletal deformities. Key findings included severe hypocalcemia, elevated alkaline phosphatase and parathyroid hormone levels, normal or elevated 25(OH)D, and high RSS. Predominant frameshift mutations in CYP27B1, especially c.1319_1325dup, highlight the importance of early genetic diagnosis for optimal management. Additionally, two novel CYP27B1 variants were identified, expanding the known mutation spectrum of VDDR1A.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Clinical Characteristics
2.3. Genetic Testing
2.4. Statistical Analysis
3. Results
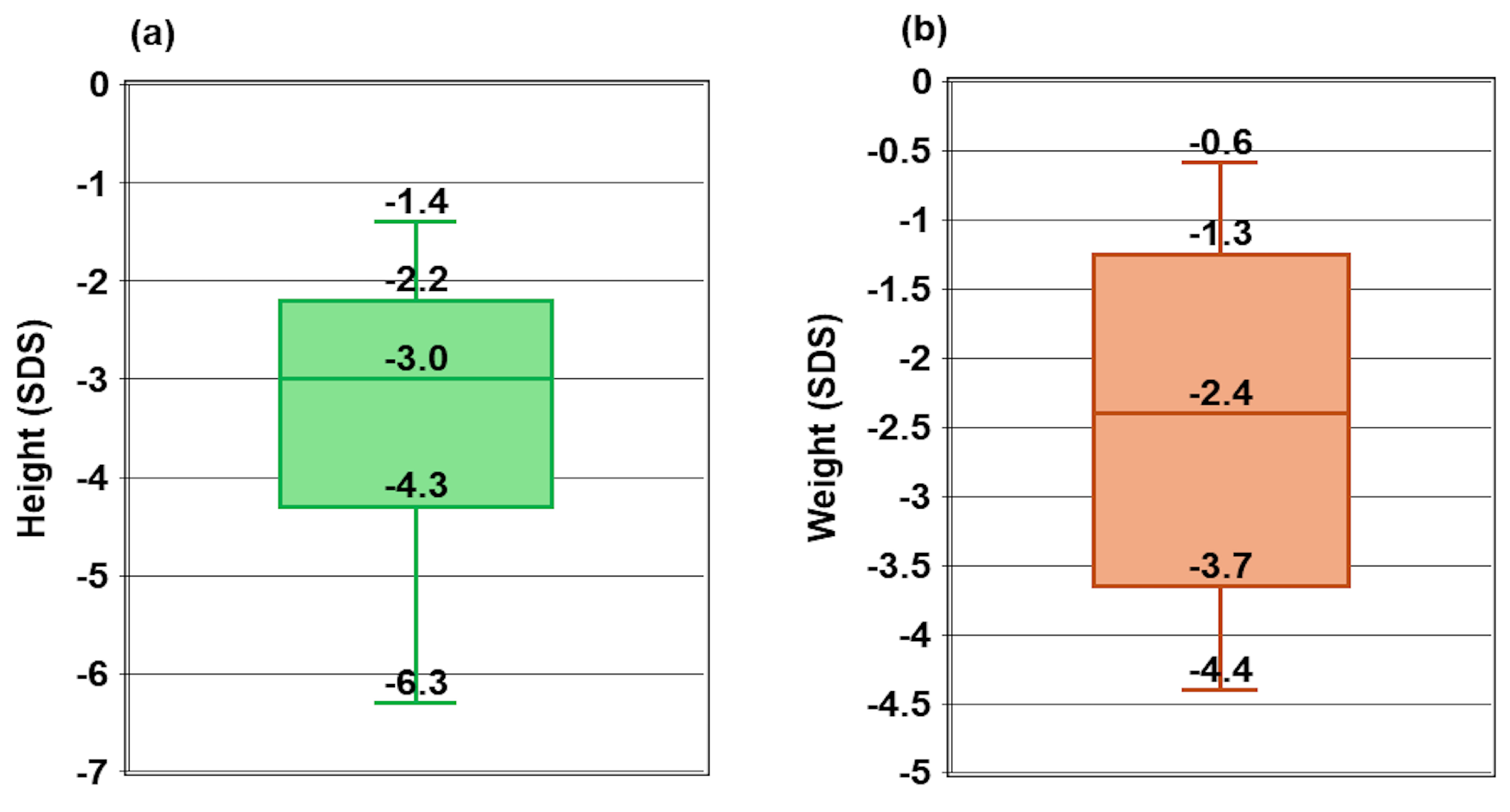
3.1. Demographics and Clinical Presentation
| Subclinical Testings | Normal Range | n | Results | Note |
|---|---|---|---|---|
| Serum total calcium (mmol/L) | 2.2–2.6 | 19 | 1.5±0.3 | 18 hypocalcemia |
| Serum phosphate (mmol/L) | 1.05–1.95 | 19 | 0.8 ± 0.4 | 11 hypophosphatemia |
| Alkaline phosphatase (UI/L) | 156-369 | 18 | 1644.2 ± 917.1 | 18 elevated |
| Parathyroid hormone (ng/mL) | 11-69 | 16 | 457.7 ± 260.7 | 16 elevated |
| 25-hydroxyvitamin D (nmol/L) | 50-250 | 17 | 140.5±109.0 |
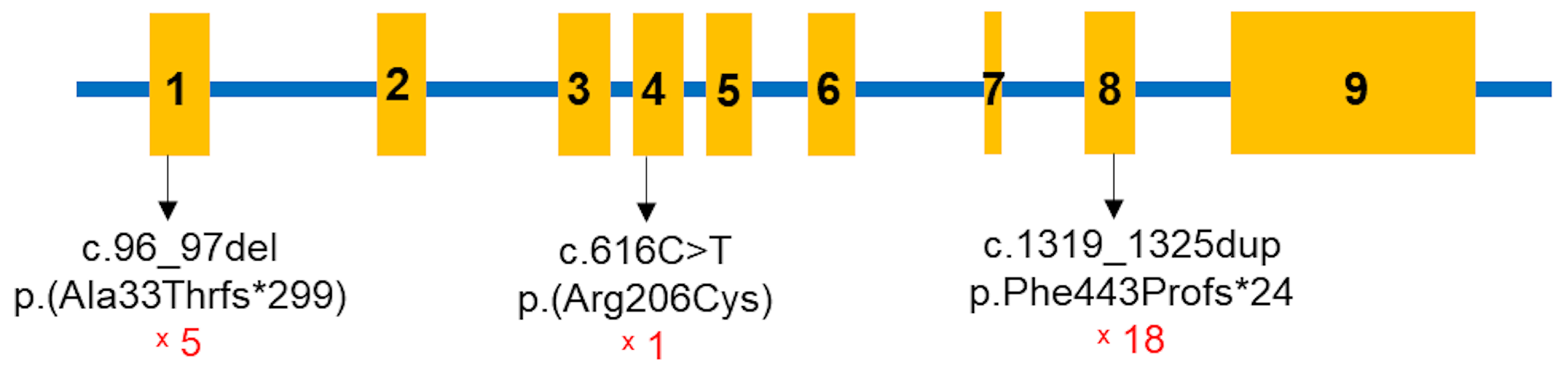
3.2. Genetic Findings
| Patient | Sex | Age of Onset (Months) |
Exon | State in the Children | c.DNA Change | Protein Change | Inheritance |
|---|---|---|---|---|---|---|---|
| 1 | M | 12.1 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 2a | M | 24.9 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 2b | F | 28.5 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 3 | M | 22.9 | 1/4 | Compound heterozygous | c.96_97del/ c.616C>T |
p.Ala33Thrfs*299/ p.Arg206Cys |
n/a |
| 4a | F | 11.1 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 4b | M | 17.5 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Paternal /Maternal |
| 5 | F | 19.3 | 8/1 | Compound heterozygous | c.1319_1325dup/ c.96_97del |
p.Phe443Profs*24/ p.Ala33Thrfs*299 | Paternal /Maternal |
| 6 | F | 31.4 | 8/1 | Compound heterozygous | c.1319_1325dup/ c.96_97del |
p.Phe443Profs*24/ p.Ala33Thrfs*299 | n/a |
| 7 | F | 17.1 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 8 | F | 25.0 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 9 | F | 20.4 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 10 | M | 14.4 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 11 | M | 34.4 | 8/1 | Compound heterozygous | c.1319_1325dup/ c.96_97del |
p.Phe443Profs*24/ p.Ala33Thrfs*299 | Maternal/Paternal |
| 12 | F | 15.1 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | n/a |
| 13 | M | 25.2 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Paternal /Maternal |
| 14 | M | 13.5 | 8 | Compound heterozygous | c.1319_1325dup/ c.96_97del |
p.Phe443Profs*24/ p.Ala33Thrfs*299 | Maternal/Paternal |
| 15 | M | 20.2 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 16 | M | 12.3 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | Maternal/Paternal |
| 17 | F | 8.3 | 8 | Homozygous | c.1319_1325dup | p.Phe443Profs*24 | n/a |
| c.DNA Change | Aa Change | Effect | Mutation Taster | dbSNP155 | ClinVar | ACMG Classification | Literature |
|---|---|---|---|---|---|---|---|
| c.96_97delAG | p.Ala33Thrfs*299 | Frameshift | Disease causing | rs1955367513 | Pathogenic | Pathogenic (PVS1, PM2, PM3, PP1, PP3, PP4, and PP5) | Novel |
| c.616C>T | p.Arg206Cys | Missense | Disease causing | Likely pathogenic (PM2, PM3, PP3, and PP4) | Novel | ||
| c.1319_1325dup | p.Phe443Profs*24 | Frameshift | Disease causing | rs780950819 | Pathogenic | Pathogenic (PVS1, PM2, PM3, PP3, PP4, and PP5) | [10,15,19,20,21,22] |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miller, W.L.; Imel, E.A. Rickets, Vitamin D, and Ca/P Metabolism. Horm Res Paediatr 2022, 95, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, B.J.; Snoddy, A.M.E.; Munns, C.; Simm, P.; Siafarikas, A.; Jefferies, C. A Brief History of Nutritional Rickets. Front Endocrinol (Lausanne) 2019, 10, 795. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.A. Diagnosis and Management of Vitamin D Dependent Rickets. Front Pediatr 2020, 8, 315. [Google Scholar] [CrossRef] [PubMed]
- Acar, S.; Demir, K.; Shi, Y. Genetic Causes of Rickets. J Clin Res Pediatr Endocrinol 2017, 9, 88–105. [Google Scholar] [CrossRef]
- Beck-Nielsen, S.S.; Brock-Jacobsen, B.; Gram, J.; Brixen, K.; Jensen, T.K. Incidence and Prevalence of Nutritional and Hereditary Rickets in Southern Denmark. Eur J Endocrinol 2009, 160, 491–497. [Google Scholar] [CrossRef]
- De Braekeleer, M.; Larochelle, J. Population Genetics of Vitamin D-Dependent Rickets in Northeastern Quebec. Ann Hum Genet 1991, 55, 283–290. [Google Scholar] [CrossRef]
- Haffner, D.; Leifheit-Nestler, M.; Grund, A.; Schnabel, D. Rickets Guidance: Part I-Diagnostic Workup. Pediatr Nephrol 2022, 37, 2013–2036. [Google Scholar] [CrossRef]
- Dodamani, M.H.; Sehemby, M.; Memon, S.S.; Sarathi, V.; Lila, A.R.; Chapla, A.; Bhandare, V.V.; Patil, V.A.; Shah, N.S.; Thomas, N.; et al. Genotype and Phenotypic Spectrum of Vitamin D Dependent Rickets Type 1A: Our Experience and Systematic Review. J Pediatr Endocrinol Metab 2021, 34, 1505–1513. [Google Scholar] [CrossRef]
- Edouard, T.; Alos, N.; Chabot, G.; Roughley, P.; Glorieux, F.H.; Rauch, F. Short- and Long-Term Outcome of Patients with Pseudo-Vitamin D Deficiency Rickets Treated with Calcitriol. J Clin Endocrinol Metab 2011, 96, 82–89. [Google Scholar] [CrossRef]
- Dursun, F.; Özgürhan, G.; Kırmızıbekmez, H.; Keskin, E.; Hacıhamdioğlu, B. Genetic and Clinical Characteristics of Patients with Vitamin D Dependent Rickets Type 1A. J Clin Res Pediatr Endocrinol 2019, 11, 34–40. [Google Scholar] [CrossRef]
- Pu, F.; Chen, N.; Xue, S. Calcium Intake, Calcium Homeostasis and Health. Food Sci Hum Wellness 2016, 5, 8–16. [Google Scholar] [CrossRef]
- Bergwitz, C.; Jüppner, H. Regulation of Phosphate Homeostasis by PTH, Vitamin D, and FGF23. Annu Rev Med 2010, 61, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Cannalire, G.; Pilloni, S.; Esposito, S.; Biasucci, G.; Di Franco, A.; Street, M.E. Alkaline Phosphatase in Clinical Practice in Childhood: Focus on Rickets. Front Endocrinol (Lausanne) 2023, 14, 1111445. [Google Scholar] [CrossRef] [PubMed]
- Thacher, T.D.; Fischer, P.R.; Pettifor, J.M.; Lawson, J.O.; Manaster, B.J.; Reading, J.C. Radiographic Scoring Method for the Assessment of the Severity of Nutritional Rickets. J Trop Pediatr 2000, 46, 132–139. [Google Scholar] [CrossRef]
- Kaygusuz, S.B.; Alavanda, C.; Kirkgoz, T.; Eltan, M.; Yavas Abali, Z.; Helvacioglu, D.; Guran, T.; Ata, P.; Bereket, A.; Turan, S. Does Genotype-Phenotype Correlation Exist in Vitamin D-Dependent Rickets Type IA: Report of 13 New Cases and Review of the Literature. Calcif Tissue Int 2021, 108, 576–586. [Google Scholar] [CrossRef]
- Do, T.T.M.; Vu, C.D.; Dien, T.M.; Can, T.B.N.; Nguyen, T.T.N.; Nguyen, H.H.; Tran, V.K.; Nguyen, N.L.; Tran, H.T.; Mai, T.T.C.; et al. Phenotypes, Genotypes, Treatment, and Outcomes of 14 Children with Sitosterolemia at Vietnam National Children’s Hospital. J Clin Med 2025, 14, 325. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 2015, 17, 405–424. [Google Scholar] [CrossRef]
- Kim, C.J.; Kaplan, L.E.; Perwad, F.; Huang, N.; Sharma, A.; Choi, Y.; Miller, W.L.; Portale, A.A. Vitamin D 1alpha-Hydroxylase Gene Mutations in Patients with 1alpha-Hydroxylase Deficiency. J Clin Endocrinol Metab 2007, 92, 3177–3182. [Google Scholar] [CrossRef]
- Wang, J.T.; Lin, C.J.; Burridge, S.M.; Fu, G.K.; Labuda, M.; Portale, A.A.; Miller, W.L. Genetics of Vitamin D 1alpha-Hydroxylase Deficiency in 17 Families. Am J Hum Genet 1998, 63, 1694–1702. [Google Scholar] [CrossRef]
- Li, Y.; Yuan, X.; Chen, R.; Lin, X.; Shangguan, H.; Yang, X.; Zhang, Y. Clinical and Genetic Analysis of Two Chinese Families with Vitamin D-Dependent Rickets Type IA and Follow-Up. Orphanet J Rare Dis 2020, 15, 273. [Google Scholar] [CrossRef]
- Ito, N.; Peña, A.S.; Perano, S.; Atkins, G.J.; Findlay, D.M.; Couper, J.J. First Australian Report of Vitamin D-Dependent Rickets Type I. Med J Aust 2014, 201, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Durmaz, E.; Zou, M.; Al-Rijjal, R.A.; Bircan, I.; Akçurin, S.; Meyer, B.; Shi, Y. Clinical and Genetic Analysis of Patients with Vitamin D-Dependent Rickets Type 1A. Clin Endocrinol (Oxf) 2012, 77, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Guan, Z.; Mei, H.; Zhang, W.; Zhou, Z.; Su, L.; Cheng, J.; Zheng, R.; Liang, C.; Cai, Y.; et al. Clinical Characteristics and Long-Term Outcomes of 12 Children with Vitamin D-Dependent Rickets Type 1A: A Retrospective Study. Front Pediatr 2022, 10, 1007219. [Google Scholar] [CrossRef] [PubMed]
- Chanchlani, R.; Nemer, P.; Sinha, R.; Nemer, L.; Krishnappa, V.; Sochett, E.; Safadi, F.; Raina, R. An Overview of Rickets in Children. Kidney Int Rep 2020, 5, 980–990. [Google Scholar] [CrossRef]
- Ozden, A.; Doneray, H. The Genetics and Clinical Manifestations of Patients with Vitamin D Dependent Rickets Type 1A. J Pediatr Endocrinol Metab 2021, 34, 781–789. [Google Scholar] [CrossRef]
- Tahir, S.; Demirbilek, H.; Ozbek, M.N.; Baran, R.T.; Tanriverdi, S.; Hussain, K. Genotype and Phenotype Characteristics in 22 Patients with Vitamin D-Dependent Rickets Type I. Horm Res Paediatr 2016, 85, 309–317. [Google Scholar] [CrossRef]
- Tiosano, D.; Hochberg, Z. Hypophosphatemia: The Common Denominator of All Rickets. J Bone Miner Metab 2009, 27, 392–401. [Google Scholar] [CrossRef]
- Calder, A.D. Radiology of Osteogenesis Imperfecta, Rickets and Other Bony Fragility States. Endocr Dev 2015, 28, 56–71. [Google Scholar] [CrossRef]
- Ahmad, F.; Shah, B.; Nabi, G.; Sofi, F. Looser’s Zone. Oman Med J 2010, 25, 141. [Google Scholar] [CrossRef]
| Evaluation Site | Grade | Radiographic Features |
|---|---|---|
| Radius and ulna | 0 | Normal |
| 1 | Widened growth plate, irregularity of metaphyseal margins, no concave cupping | |
| 2 | Metaphyseal concavity with fraying of margins | |
| Femur and tibia | 0 | Normal |
| 1 | Partial lucency, smooth metaphyseal margin visible | |
| 2 | Partial lucency, smooth metaphyseal margin not visible | |
| 3 | Complete lucency, epiphysis appears widely separated from distal metaphysis | |
| Multiplier | Multiplier 0.5 | |
| Multiplier 1 | ||
| Characteristics | Parameter (n=19) | |
|---|---|---|
| Sex | Male | 10 (52.6%) |
| Female | 9 (47.4%) | |
| Age of diagnosis rickets (month) | 19.2 [8.3-34.4] | |
| Time of diagnosis VDDR1A (month) | 7.5 [1.1-148.0] | |
| Delayed walking | 11 (57.9%) | |
| Frontal bossing | 10 (52.6%) | |
| Thickened wrists and ankles | 19 (100%) | |
| Genu varumorgenu valgum | 18 (94.7%) | |
| Rachitic rosary | 12 (63.2%) | |
| Bones frature | 2 (10.5%) | |
| Seizures | 6 (31.5%) | |
| Chest deformity | 10 (52.6%) | |
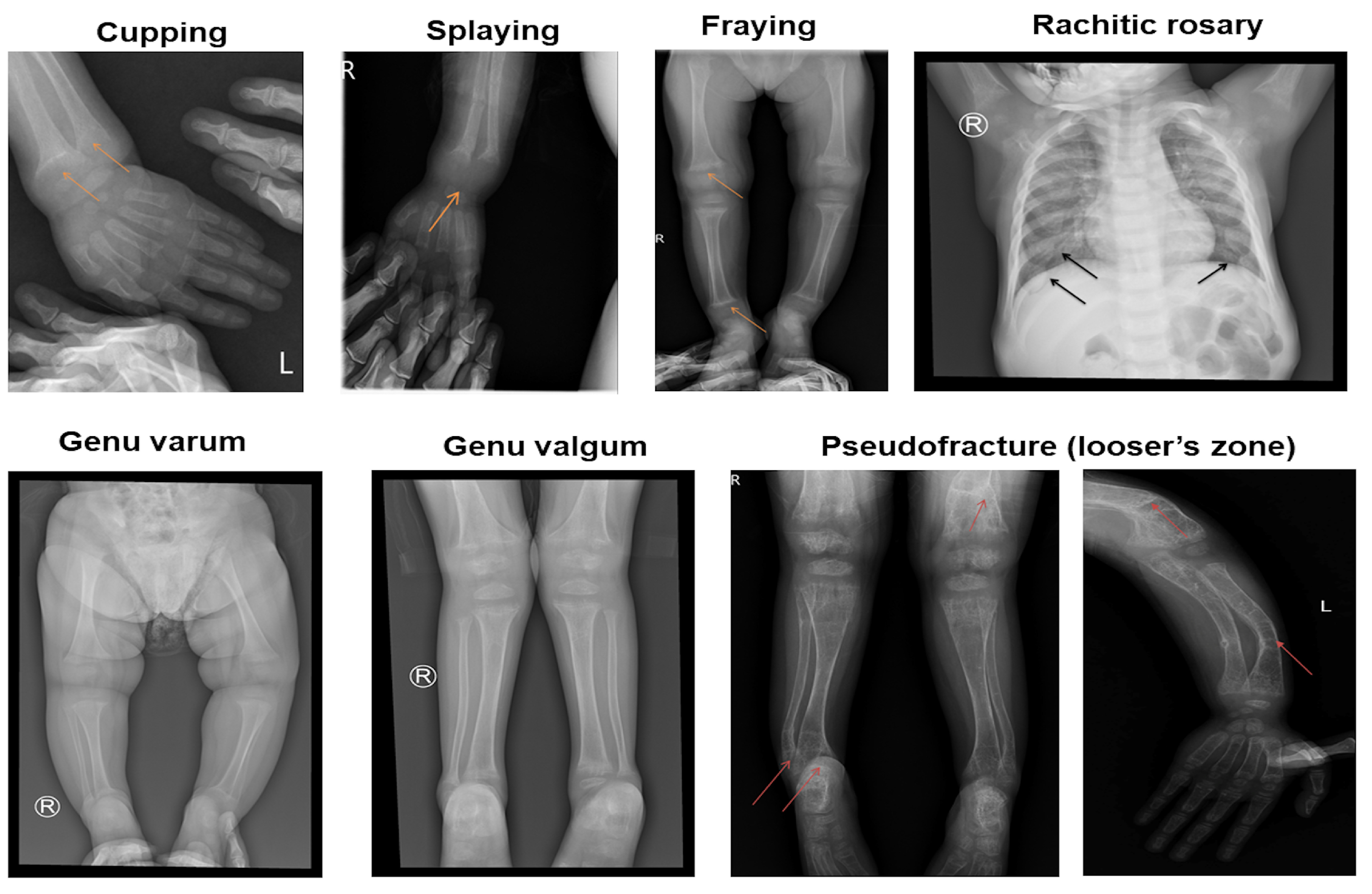
| X-ray features | Cupping and splaying | 19 (100%) |
| Fraying | 19 (100%) | |
| Rachitic rosary | 12 (63.2%) | |
| Pseudofrature (Looser’s zone) |
6 (31.6%) | |
| Rickets severe score = 10 | 19 (100%) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




