Submitted:
23 February 2025
Posted:
24 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Sources and Incidence of Exposure
2. Pathophysiology of Carbon Monoxide Poisoning
2.1. Mechanism of Toxicity: Interaction with Hemoglobin and Cellular Effects
2.2. Mechanisms of Cardiac Injury in Carbon Monoxide Poisoning
2.3. Functional and Structural Cardiac Consequences
2.4. Pathophysiology of Brain Injuries in Acute Carbon Monoxide Poisoning
3. Clinical Manifestations of Carbon Monoxide Poisoning
3.1. Neurological Effects
3.2. Cardiovascular and Other Systemic Effects
4. Diagnosis of Carbon Monoxide Poisoning
4.1. Clinical Assessment and History
4.1.1. Identifying Risk Factors and Exposure Sources
4.2. Diagnostic Tools for Carbon Monoxide Poisoning
4.2.1. Carboxyhemoglobin Levels
4.2.2. Pulse CO-Oximetry
4.2.3. Arterial Blood Gas Analysis
4.3. Comparison and Modern Perspective
4.4. Challenges in Early Detection of Carbon Monoxide (CO) Poisoning
5. Prognosis: Factors Affecting Outcomes in Carbon Monoxide (CO) Poisoning
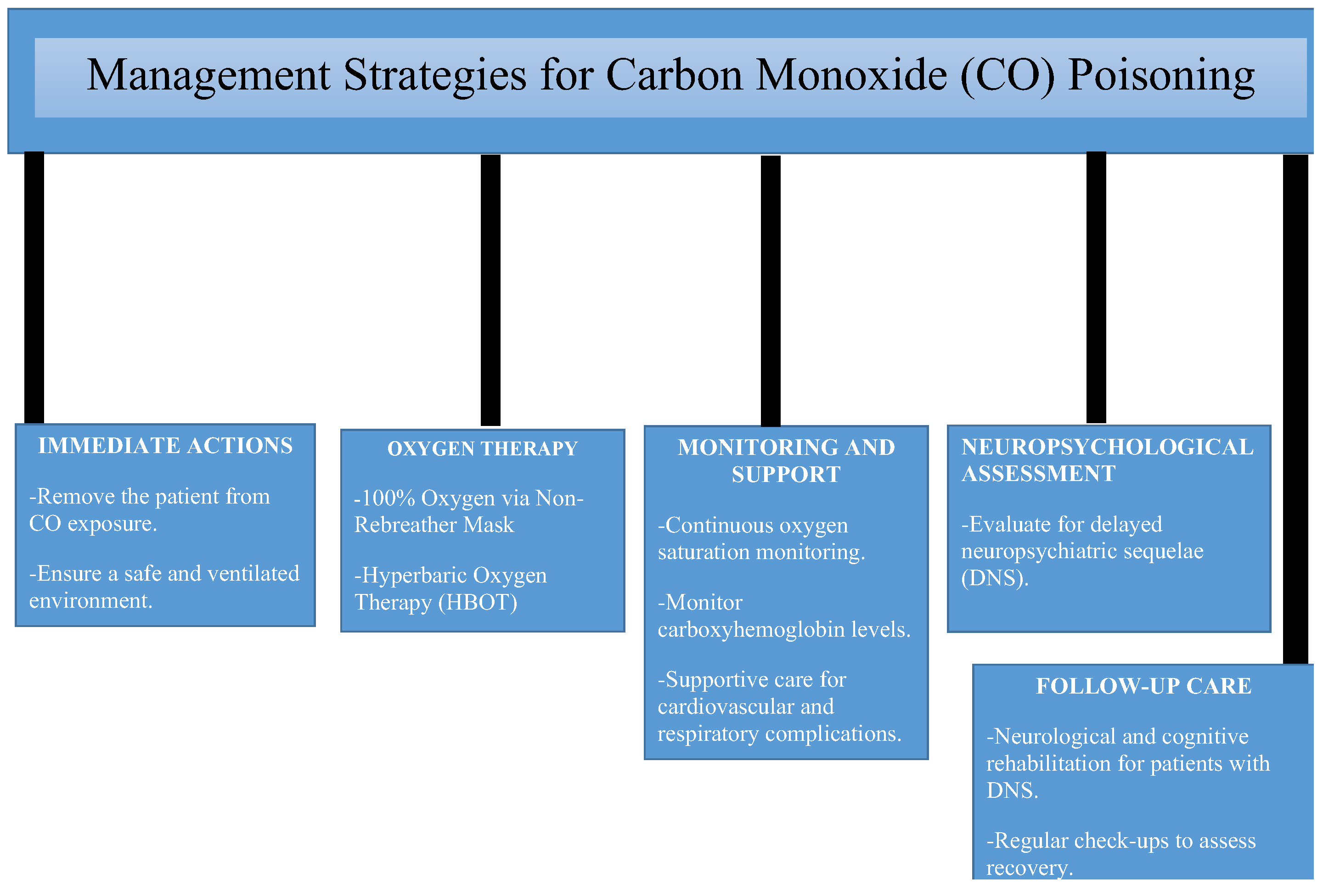
6. Management Strategies for Carbon Monoxide (CO) Poisoning
6.1. Immediate Interventions
6.2. Removal From Source of Exposure
6.3. Oxygen Therapy (100% Oxygen)
6.4. Role of Hyperbaric Oxygen Therapy (HBOT)
6.5. Others Comprehensive Management Approach for CO Poisoning
6.6. Managing Neurological and Cardiovascular Complications
7. Laboratory Diagnostic Challenges
7.1. Limitations of Current Techniques
7.2. Advances in COHb Testing and Diagnosis
7.3. Challenges in CO Poisoning Diagnosis in Resource-Limited Settings
8. Prevention and Public Health Measures
8.1. Maintenance of Combustion Appliances
8.2. Public Awareness Campaigns
8.2.1. Strategies for Public Awareness Campaigns
9. Future Perspectives
9.1. Advances in Diagnostics and Therapeutics
9.2. Development of Portable Detection Devices
9.3. Strategies for Enhanced Healthcare Delivery
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Mattiuzzi, C.; Lippi, G. Worldwide epidemiology of carbon monoxide poisoning. Hum Exp Toxicol. 2020, 39, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Olas, B. Carbon monoxide is not always a poison gas for human organism: Physiological and pharmacological features of CO. Chem Biol Interact. 2014, 222, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Siebenmann, C.; Keiser, S.; Robach, P.; Lundby, C. CORP: The assessment of total hemoglobin mass by carbon monoxide rebreathing. J Appl Physiol (1985). 2017, 123, 645–654. [Google Scholar] [CrossRef]
- Dong, Y.; Peng, R.; Kang, H.; Song, K.; Guo, Q.; Zhao, H.; Zhu, M.; Zhang, Y.; Guan, H.; Li, F. Global incidence, prevalence, and disability of vertebral fractures: a systematic analysis of the global burden of disease study 2019. Spine, J. 2022, 22, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.J.; Wang, L.; Xu, Q.; McTiernan, C.F.; Shiva, S.; Tejero, J.; Gladwin, M.T. Carbon Monoxide Poisoning: Pathogenesis, Management, and Future Directions of Therapy. Am J Respir Crit Care Med. 2017, 195, 596–606. [Google Scholar] [CrossRef]
- Zonta, G.; Rispoli, G.; Malagù, C.; Astolfi, M. Overview of gas sensors focusing on chemoresistive ones for cancer detection. Chemosensors. 2023, 11, 519. [Google Scholar] [CrossRef]
- Ng, P.C.; Long, B.; Koyfman, A. Clinical chameleons: an emergency medicine focused review of carbon monoxide poisoning. Intern Emerg Med. 2018, 13, 223–229. [Google Scholar] [CrossRef]
- Dey, S.; Dhal, G.C. Materials progress in the control of CO and CO2 emission at ambient conditions: An overview. Materials Science for Energy Technologies. 2019, 2, 607–23. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Hirsch, A.; Etienne, S.; Melnikova, N.; Wu, J.; Sircar, K.; Orr, M. Surveillance of carbon monoxide-related incidents - Implications for prevention of related illnesses and injuries, 2005-2014. Am J Emerg Med. 2018, 36, 1837–1844. [Google Scholar] [CrossRef]
- Votsmeier, M.; Kreuzer, T.; Gieshoff, J.; Lepperhoff, G. Automobile exhaust control. Ullmann's Encyclopedia of Industrial Chemistry. 2009, 4, 407–24. [Google Scholar]
- Hampson, N.B.; Dunn, S.L. Carbon Monoxide Poisoning from Portable Electrical Generators. J Emerg Med. 2015, 49, 125–9. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.W.; Bevan, R.J.; Harrison, P.T.; Youngs, L.C.; Crump, D. Public health impacts of exposure to carbon monoxide from gas appliances in UK homes–are we missing something? . Indoor and Built Environment. 2012, 21, 229–40. [Google Scholar] [CrossRef]
- Alinejad, S.; Zamani, N.; Abdollahi, M.; Mehrpour, O. A Narrative Review of Acute Adult Poisoning in Iran. Iran J Med Sci. 2017, 42, 327–346. [Google Scholar]
- Eggert, E.; Huss, F. Medical and biological factors affecting mortality in elderly residential fire victims: a narrative review of the literature. Scars Burn Heal. 2017, 3, 2059513117707686. [Google Scholar] [CrossRef]
- Roderique, J.D.; Josef, C.S.; Feldman, M.J.; Spiess, B.D. A modern literature review of carbon monoxide poisoning theories, therapies, and potential targets for therapy advancement. Toxicology. 2015, 334, 45–58. [Google Scholar] [CrossRef]
- Motterlini, R.; Foresti, R. Biological signaling by carbon monoxide and carbon monoxide-releasing molecules. Am J Physiol Cell Physiol. 2017, 312, C302–C313. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhang, Y. Mechanisms and therapeutic targets of carbon monoxide poisoning: A focus on reactive oxygen species. Chem Biol Interact. 2024, 403, 111223. [Google Scholar] [CrossRef]
- Guzman, J.A. Carbon monoxide poisoning. Crit Care Clin. 2012, 28, 537–48. [Google Scholar] [CrossRef]
- Jain, K.K.; Jain, K.K. Carbon monoxide and other tissue poisons. Textbook of hyperbaric medicine. 2017, 131–54. [Google Scholar]
- Lippi, G.; Rastelli, G.; Meschi, T.; Borghi, L.; Cervellin, G. Pathophysiology, clinics, diagnosis and treatment of heart involvement in carbon monoxide poisoning. Clinical biochemistry. 2012, 45, 1278–85. [Google Scholar] [CrossRef]
- Fracasso, T.; Pfeiffer, H.; Michaud, K.; Köhler, H.; Sauerland, C.; Schmeling, A. Immunohistochemical expression of fibronectin and C5b-9 in the myocardium in cases of carbon monoxide poisoning. Int J Legal Med. 2011, 125, 377–84. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yu, W.; Cao, J.; Gao, H. Harnessing carbon monoxide-releasing platforms for cancer therapy. Biomaterials. 2020, 255, 120193. [Google Scholar] [CrossRef] [PubMed]
- Cardoso-Pires, C.; Vieira, H.L. Carbon monoxide and mitochondria: Cell energy and fate control. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease. 2024, 167446.
- Chen, H.; He, Y.; Chen, S.; Qi, S.; Shen, J. Therapeutic targets of oxidative/nitrosative stress and neuroinflammation in ischemic stroke: Applications for natural product efficacy with omics and systemic biology. Pharmacol Res. 2020, 158, 104877. [Google Scholar] [CrossRef]
- Öztürk, S.; Ayhan, S.; Dönmez, İ.; Erdem, F.; Erdem, A. Very late stent thrombosis in a patient presenting with acute carbon monoxide poisoning. Journal of Emergency Medicine Case Reports. 2017, 8, 37–9. [Google Scholar] [CrossRef]
- Liu, C.; Ni, C.; Liu, W.; Yang, X.; Zhang, R.; Zhang, J.; Luo, M.; Xu, J.; Yu, J. Effects of long-term nonylphenol exposure on myocardial fibrosis and cardiac function in rats. Environmental Sciences Europe. 2021, 33, 1–3. [Google Scholar] [CrossRef]
- Kronenbitter, A.; Funk, F.; Hackert, K.; Gorreßen, S.; Glaser, D.; Boknik, P.; Poschmann, G.; Stühler, K.; Isić, M.; Krüger, M.; Schmitt, J.P. Impaired Ca2+ cycling of nonischemic myocytes contributes to sarcomere dysfunction early after myocardial infarction. J Mol Cell Cardiol. 2018, 119, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.J. Adrenergic signaling in heart failure: a balance of toxic and protective effects. Pflugers Arch. 2014, 466, 1139–50. [Google Scholar] [CrossRef]
- Armandeh, M.; Bameri, B.; Baeeri, M.; Haghi-Aminjan, H.; Rahimifard, M.; Hassani, S.; Hooshangi Shayesteh, M.R.; Khalid, M.; Samadi, M.; Hosseini, R.; Masoudi Fard, M.; Abdollahi, M. The role of levosimendan in phosphine-induced cardiotoxicity: evaluation of electrocardiographic, echocardiographic, and biochemical parameters. Toxicol Mech Methods. 2021, 31, 631–643. [Google Scholar] [CrossRef]
- Park, E.J.; Min, Y.G.; Kim, G.W.; Cho, J.P.; Maeng, W.J.; Choi, S.C. Pathophysiology of brain injuries in acute carbon monoxide poisoning: a novel hypothesis. Med Hypotheses. 2014, 83, 186–9. [Google Scholar] [CrossRef]
- Kassa, B.D.; Yigzaw, A.A.; Kassie, Y.G.; Kedimu, M.W.; Mekuanint, Y.F.; Moges, N. Delayed neuropsychiatric sequelae due to long-term effects of carbon monoxide poisoning in Ethiopia: A case report. Toxicology Reports. 2023, 11, 36–9. [Google Scholar] [CrossRef]
- Beppu, T. The role of MR imaging in assessment of brain damage from carbon monoxide poisoning: a review of the literature. AJNR Am J Neuroradiol. 2014, 35, 625–31. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Choi, S.C. Acute carbon monoxide poisoning and delayed neurological sequelae: a potential neuroprotection bundle therapy. Neural Regen Res. 2015, 10, 36–8. [Google Scholar]
- Bauer, N.; Liu, D.; Nguyen, T.; Wang, B. Unraveling the Interplay of Dopamine, Carbon Monoxide, and Heme Oxygenase in Neuromodulation and Cognition. ACS Chem Neurosci. 2024, 15, 400–407. [Google Scholar] [CrossRef]
- Sireesha, M.; Jagadeesh Babu, V.; Kranthi Kiran, A.S.; Ramakrishna, S. A review on carbon nanotubes in biosensor devices and their applications in medicine. Nanocomposites. 2018, 4, 36–57. [Google Scholar] [CrossRef]
- Bielecka-Papierz, G.; Poleszak, E.; Serafko, A. Mephedrone–a synthetic derivative of cathinone. Current Issues in Pharmacy and Medical Sciences. 2023, 36, 54–64. [Google Scholar] [CrossRef]
- Bandeira, G.A.; Lucato, L.T. Toxic leukoencephalopathies. Handb Clin Neurol. 2024, 204, 455–486. [Google Scholar]
- Macnow, T.E.; Waltzman, M.L. Carbon Monoxide Poisoning In Children: Diagnosis And Management In The Emergency Department. Pediatr Emerg Med Pract. 2016, 13, 1–24. [Google Scholar]
- Hampson, N.B.; Piantadosi, C.A.; Thom, S.R.; Weaver, L.K. Practice recommendations in the diagnosis, management, and prevention of carbon monoxide poisoning. Am J Respir Crit Care Med. 2012, 186, 1095–101. [Google Scholar] [CrossRef]
- Ran, T.; Nurmagambetov, T.; Sircar, K. Economic implications of unintentional carbon monoxide poisoning in the United States and the cost and benefit of CO detectors. Am J Emerg Med. 2018, 36, 414–419. [Google Scholar] [CrossRef]
- Bleecker, M.L. Carbon monoxide intoxication. Handb Clin Neurol. 2015, 131, 191–203. [Google Scholar]
- Taylor, F.R. Tobacco, Nicotine, and Headache. Headache. 2015, 55, 1028–44. [Google Scholar] [CrossRef] [PubMed]
- Hedges, D.; Farrer, T.J.; Bigler, E.D.; Hopkins, R.O.; Hedges, D.; Farrer, T.J.; Bigler, E.D.; Hopkins, R.O. Carbon monoxide poisoning and cognitive impairment. The Brain at Risk: Associations between Disease and Cognition. 2019, 225-44.
- Vural, A.; Dolanbay, T. Early and late adverse clinical outcomes of severe carbon monoxide intoxication: A cross-sectional retrospective study. PLoS One. 2024, 19, e0301399. [Google Scholar] [CrossRef] [PubMed]
- Coşkun, A. A new method in foreseeing late-period neuro-psychosis in carbon monoxide poisoning: COHb/Troponin, COHb/Neutrophil, and COHb/Lymphocyte. Cumhuriyet Medical Journal. 2019, 41, 163–73. [Google Scholar] [CrossRef]
- Quinn, D.K.; Abbott, C.C. Catatonia after cerebral hypoxia: do the usual treatments apply? Psychosomatics. 2014, 55, 525–35. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.; Salazar, L.; Duarte-Costa, S.; Fraga, C.; Monteiro, S.; Camacho, Ó. Hyperbaric Medicine in Pediatrics - reality of a Portuguese reference center. Rev Paul Pediatr. 2024, 43, e2023230. [Google Scholar] [CrossRef]
- Cho, D.H.; Thom, S.R.; Son, J.W.; Ko, S.M.; Cha, Y.S. Practical Recommendations for the Evaluation and Management of Cardiac Injury Due to Carbon Monoxide Poisoning. JACC Heart Fail. 2024, 12, 1343–1352. [Google Scholar] [CrossRef]
- Haliga, R.E.; Morărașu, B.C.; Șorodoc, V.; Lionte, C.; Sîrbu, O.; Stoica, A.; Ceasovschih, A.; Constantin, M.; Șorodoc, L. Rare Causes of Acute Coronary Syndrome: Carbon Monoxide Poisoning. Life (Basel). 2022, 12, 1158. [Google Scholar] [CrossRef]
- Elfakharany, Y.M. Acute poisoning during pregnancy poses a particular challenge to health care providers. Egyptian Society of Clinical Toxicology Journal. 2022, 10, 104–20. [Google Scholar] [CrossRef]
- Dutta, S.; Gorain, B.; Choudhury, H.; Roychoudhury, S.; Sengupta, P. Environmental and occupational exposure of metals and female reproductive health. Environ Sci Pollut Res Int. 2022, 29, 62067–62092. [Google Scholar] [CrossRef]
- Savioli, G.; Gri, N.; Ceresa, I.F.; Piccioni, A.; Zanza, C.; Longhitano, Y.; Ricevuti, G.; Daccò, M.; Esposito, C.; Candura, S.M. Carbon Monoxide Poisoning: From Occupational Health to Emergency Medicine. J Clin Med. 2024, 13, 2466. [Google Scholar] [CrossRef]
- Stephenson, L.; Tiemensma, M.; Van Den Heuvel, C.; Byard, R.W. The spectrum of presentations of unintentional carbon monoxide poisoning. Med Sci Law. 2024, 64, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Reumuth, G.; Alharbi, Z.; Houschyar, K.S.; Kim, B.S.; Siemers, F.; Fuchs, P.C.; Grieb, G. Carbon monoxide intoxication: What we know. Burns. 2019, 45, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Turgut, K.; Yavuz, E. Comparison of non-invasive CPAP with mask use in carbon monoxide poisoning. Am J Emerg Med. 2020, 38, 1454–1457. [Google Scholar] [CrossRef]
- Jeoung, J.H.; Fesseler, J.; Goetzl, S.; Dobbek, H. Carbon monoxide. Toxic gas and fuel for anaerobes and aerobes: carbon monoxide dehydrogenases. Met Ions Life Sci. 2014, 14, 37–69. [Google Scholar] [PubMed]
- Mureşan, C.O.; Zăvoi, R.E.; Dumache, R.O.; Precup, C.V.; Ciocan, V.; Bulzan, O.Ş.; Florou, C.; Enache, A. Co-morbidities in the multiple victims of the silent killer in carbon monoxide poisoning. Rom J Morphol Embryol. 2019, 60, 125–131. [Google Scholar]
- Araya, S.S.; Zhou, F.; Liso, V.; Sahlin, S.L.; Vang, J.R.; Thomas, S.; Gao, X.; Jeppesen, C.; Kær, S.K. A comprehensive review of PBI-based high temperature PEM fuel cells. International Journal of Hydrogen Energy. 2016, 41, 21310–44. [Google Scholar] [CrossRef]
- Buchelli Ramirez, H.; Fernández Alvarez, R.; Rubinos Cuadrado, G.; Martinez Gonzalez, C.; Rodriguez Jerez, F.; Casan Clara, P. Elevated carboxyhemoglobin: sources of carbon monoxide exposure. Arch Bronconeumol. 2014, 50, 465–8. [Google Scholar] [CrossRef]
- Oliverio, S.; Varlet, V. What are the limitations of methods to measure carbon monoxide in biological samples? . Forensic Toxicology. 2020, 38, 1–4. [Google Scholar] [CrossRef]
- Andropoulos, D.B. Monitoring and vascular access. Gregory's Pediatric Anesthesia. 2020, 406-43.
- Pretto, J.J.; Roebuck, T.; Beckert, L.; Hamilton, G. Clinical use of pulse oximetry: official guidelines from the Thoracic Society of Australia and New Zealand. Respirology. 2014, 19, 38–46. [Google Scholar] [CrossRef]
- Kumar V J, Reddy, K.A. Pulse oximetry for the measurement of oxygen saturation in arterial blood. Studies in Skin Perfusion Dynamics: Photoplethysmography and its Applications in Medical Diagnostics. 2021, 51-78.
- Ascha, M.; Bhattacharyya, A.; Ramos, J.A.; Tonelli, A.R. Pulse Oximetry and Arterial Oxygen Saturation during Cardiopulmonary Exercise Testing. Med Sci Sports Exerc. 2018, 50, 1992–1997. [Google Scholar] [CrossRef]
- Tan, R.N.; Mulder, E.E.; Lopriore, E.; Te Pas, A.B. Monitoring Oxygenation and Gas Exchange in Neonatal Intensive Care Units: Current Practice in the Netherlands. Front Pediatr. 2015, 3, 94. [Google Scholar] [CrossRef] [PubMed]
- Rajsic, S.; Breitkopf, R.; Bachler, M.; Treml, B. Diagnostic Modalities in Critical Care: Point-of-Care Approach. Diagnostics (Basel). 2021, 11, 2202. [Google Scholar] [CrossRef]
- Schoen, J.C.; Campbell, R.L.; Sadosty, A.T. Headache in pregnancy: an approach to emergency department evaluation and management. West J Emerg Med. 2015, 16, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Tekin, K.; Karadogan, M.; Gunaydin, S.; Kismet, K. Everything About Pulse Oximetry-Part 1: History, Principles, Advantages, Limitations, Inaccuracies, Cost Analysis, the Level of Knowledge About Pulse Oximeter Among Clinicians, and Pulse Oximetry Versus Tissue Oximetry. J Intensive Care Med. 2023, 38, 775–784. [Google Scholar] [CrossRef]
- Bradberry, S.M.; Vale, P.A.; Jefferson, R.D.; Buckley, N.; Bateman, D.N.; Thanacoody, H.R.; Wood, D. Common chemical poisonings. Oxford Desk Reference: Toxicology. 2014, 205.
- Pan, K.T.; Leonardi, G.S.; Croxford, B. Factors Contributing to CO Uptake and Elimination in the Body: A Critical Review. Int J Environ Res Public Health. 2020, 17, 528. [Google Scholar] [CrossRef] [PubMed]
- Nnaji, C.C.; Chibueze, C.; Afangideh, C.B. The menace and mitigation of air pollution in the built environment: A review. Nigerian Journal of Technology. 2023, 42, 12–29. [Google Scholar] [CrossRef]
- Kim, Y.J.; Sohn, C.H.; Seo, D.W.; Oh, B.J.; Lim, K.S.; Kim, W.Y. Clinical Predictors of Acute Brain Injury in Carbon Monoxide Poisoning Patients With Altered Mental Status at Admission to Emergency Department. Acad Emerg Med. 2019, 26, 60–67. [Google Scholar] [CrossRef]
- Ramponi, G.; Gianni, F.; Karlafti, E.; Piazza, I.; Albertoni, F.; Colombo, G.; Casazza, G.; Garegnani, A.; Casella, R.; Costantino, G. The diagnostic accuracy of carbon monoxide pulse oximetry in adults with suspected acute carbon monoxide poisoning: a systematic review and meta-analysis. Front Med (Lausanne). 2023, 10, 1250845. [Google Scholar] [CrossRef]
- Hampson, N.B. U.S. Mortality Due to Carbon Monoxide Poisoning, 1999-2014. Accidental and Intentional Deaths. Ann Am Thorac Soc. 2016, 13, 1768–1774. [Google Scholar]
- Taylor-Williams, M.; Spicer, G.; Bale, G.; Bohndiek, S.E. Noninvasive hemoglobin sensing and imaging: optical tools for disease diagnosis. J Biomed Opt. 2022, 27, 080901. [Google Scholar] [CrossRef]
- Pan, K.T.; Shen, C.H.; Lin, F.G.; Chou, Y.C.; Croxford, B.; Leonardi, G.; Huang, K.L. Prognostic factors of carbon monoxide poisoning in Taiwan: a retrospective observational study. BMJ Open. 2019, 9, e031135. [Google Scholar] [CrossRef]
- Chiew, A.L.; Buckley, N.A. Carbon monoxide poisoning in the 21st century. Critical Care. 2014, 18, 1–8. [Google Scholar] [CrossRef]
- Eriksson, E.; Liu, P.Y.; Schultz, G.S.; Martins-Green, M.M.; Tanaka, R.; Weir, D.; Gould, L.J.; Armstrong, D.G.; Gibbons, G.W.; Wolcott, R.; Olutoye, O.O.; Kirsner, R.S.; Gurtner, G.C. Chronic wounds: Treatment consensus. Wound Repair Regen. 2022, 30, 156–171. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, D.; Marroni, A.; Kot, J. Correction to Mathieu, D.; Marroni, A.; Kot J: Tenth European Consensus Conference on Hyperbaric Medicine: recommendations for accepted and non-accepted clinical indications and practice of hyperbaric oxygen treatment. Diving Hyperb Med. 2017, 47, 24–32. [Google Scholar] [CrossRef]
- Hantson, P. Mechanisms of toxic cardiomyopathy. Clin Toxicol (Phila). 2019, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Luo, X. Extrapulmonary Comorbidities Associated with Chronic Obstructive Pulmonary Disease: A Review. Int J Chron Obstruct Pulmon Dis. 2024, 19, 567–578. [Google Scholar] [CrossRef]
- Manisalidis, I.; Stavropoulou, E.; Stavropoulos, A.; Bezirtzoglou, E. Environmental and Health Impacts of Air Pollution: A Review. Front Public Health. 2020, 8, 14. [Google Scholar] [CrossRef]
- Cosselman, K.E.; Navas-Acien, A.; Kaufman, J.D. Environmental factors in cardiovascular disease. Nature Reviews Cardiology. 2015, 12, 627–42. [Google Scholar] [CrossRef]
- Yeh, Z.T.; Tsai, C.F.; Yip, P.K.; Lo, C.Y.; Peng, S.M.; Chen, S.Y.; Kung, L.Y. Neuropsychological performance in patients with carbon monoxide poisoning. Appl Neuropsychol Adult. 2014, 21, 278–87. [Google Scholar] [CrossRef]
- Mishra, K.; Krishnan, G. Carbon monoxide poisoning. Journal of Marine Medical Society. 2016, 18, 179–86. [Google Scholar] [CrossRef]
- Shrivastav, A.; Swetanshu, S.P. The Impact of Environmental Toxins on Cardiovascular Diseases. Curr Probl Cardiol. 2024, 49, 102120. [Google Scholar] [CrossRef] [PubMed]
- Mladěnka, P.; Applová, L.; Patočka, J.; Costa, V.M.; Remiao, F.; Pourová, J.; Mladěnka, A.; Karlíčková, J.; Jahodář, L.; Vopršalová, M.; Varner, K.J.; Štěrba, M.; TOX-OER and CARDIOTOX Hradec Králové Researchers and Collaborators. Comprehensive review of cardiovascular toxicity of drugs and related agents. Med Res Rev. 2018, 38, 1332–1403. [Google Scholar] [CrossRef]
- Gozubuyuk, A.A.; Dag, H.; Kacar, A.; Karakurt, Y.; Arica, V. Epidemiology, pathophysiology, clinical evaluation, and treatment of carbon monoxide poisoning in child, infant, and fetus. North Clin Istanb. 2017, 4, 100–107. [Google Scholar]
- Whyand, T.; Hurst, J.R.; Beckles, M.; Caplin, M.E. Pollution and respiratory disease: can diet or supplements help? A review. Respir Res. 2018, 19, 79. [Google Scholar] [CrossRef] [PubMed]
- Barber, C.W.; Miller, M.J. Reducing a suicidal person's access to lethal means of suicide: a research agenda. Am J Prev Med. 2014, 47, S264–72. [Google Scholar] [CrossRef]
- Liao, S.C.; Mao, Y.C.; Yang, K.J.; Wang, K.C.; Wu, L.Y.; Yang, C.C. Targeting optimal time for hyperbaric oxygen therapy following carbon monoxide poisoning for prevention of delayed neuropsychiatric sequelae: A retrospective study. J Neurol Sci. 2019, 396, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Lustberg, M.B.; Kuderer, N.M.; Desai, A.; Bergerot, C.; Lyman, G.H. Mitigating long-term and delayed adverse events associated with cancer treatment: implications for survivorship. Nat Rev Clin Oncol. 2023, 20, 527–542. [Google Scholar] [CrossRef]
- Eve, D.J.; Steele, M.R.; Sanberg, P.R.; Borlongan, C.V. Hyperbaric oxygen therapy as a potential treatment for post-traumatic stress disorder associated with traumatic brain injury. Neuropsychiatr Dis Treat. 2016, 12, 2689–2705. [Google Scholar] [CrossRef]
- Levy, R.J. Carbon monoxide pollution and neurodevelopment: A public health concern. Neurotoxicol Teratol. 2015, 49, 31–40. [Google Scholar] [CrossRef]
- Ku, C.H.; Hung, H.M.; Leong, W.C.; Chen, H.H.; Lin, J.L.; Huang, W.H.; Yang, H.Y.; Weng, C.H.; Lin, C.M.; Lee, S.H.; Wang, I.K. Outcome of patients with carbon monoxide poisoning at a far-east poison center. PLoS One. 2015, 10, e0118995. [Google Scholar] [CrossRef]
- Thompson, T.M.; Theobald, J.; Lu, J.; Erickson, T.B. The general approach to the poisoned patient. Dis Mon. 2014, 60, 509–24. [Google Scholar] [CrossRef] [PubMed]
- Akyol, S.; Erdogan, S.; Idiz, N.; Celik, S.; Kaya, M.; Ucar, F.; Dane, S.; Akyol, O. The role of reactive oxygen species and oxidative stress in carbon monoxide toxicity: an in-depth analysis. Redox Rep. 2014, 19, 180–9. [Google Scholar] [CrossRef] [PubMed]
- Beuhler, M.C. Treatment and evaluation of specific toxins. Emergency Medical Services: Clinical Practice and Systems Oversight. 2021, 1, 383–90. [Google Scholar]
- Sethi, D.; Subramanian, S. When place and time matter: How to conduct safe inter-hospital transfer of patients. Saudi J Anaesth. 2014, 8, 104–13. [Google Scholar] [CrossRef]
- Fucili, G.; Brauzzi, M. Tips for avoiding common mistakes in out-of-hospital diagnosis of carbon monoxide poisoning. J Anesth Analg Crit Care. 2022, 2, 14. [Google Scholar] [CrossRef]
- Prata, J.C.; da Costa, J.P.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T. Environmental exposure to microplastics: An overview on possible human health effects. Sci Total Environ. 2020, 702, 134455. [Google Scholar] [CrossRef]
- Meitner, C.; Feuerstein, R.A.; Steele, A.M. Nursing strategies for the mechanically ventilated patient. Front Vet Sci. 2023, 10, 1145758. [Google Scholar] [CrossRef]
- Kinoshita, H.; Türkan, H.; Vucinic, S.; Naqvi, S.; Bedair, R.; Rezaee, R.; Tsatsakis, A. Carbon monoxide poisoning. Toxicol Rep. 2020, 7, 169–173. [Google Scholar] [CrossRef]
- Osborne, J.; Sobh, M.; Trudel, G. Carbon monoxide as a clinical marker of hemolysis. Am J Hematol. 2023, 98, 1127–1159. [Google Scholar] [CrossRef]
- Köhler, A.; Zoll, F.M.; Ploner, T.; Hammer, A.; Joannidis, M.; Tilg, H.; Finkenstedt, A.; Hartig, F. Oxygenation Performance of Different Non-Invasive Devices for Treatment of Decompression Illness and Carbon Monoxide Poisoning. Front Physiol. 2022, 13, 885898. [Google Scholar] [CrossRef]
- Heyboer, M., 3rd; Sharma, D.; Santiago, W.; McCulloch, N. Hyperbaric Oxygen Therapy: Side Effects Defined and Quantified. Adv Wound Care (New Rochelle). 2017, 6, 210–224. [Google Scholar] [CrossRef] [PubMed]
- Del Moral-Barbudo, B.; Blancas, R.; Ballesteros-Ortega, D.; Quintana-Díaz, M.; Martínez-González, Ó. Current and research therapies for the prevention and treatment of delayed neurological syndrome associated with carbon monoxide poisoning: A narrative review. Hum Exp Toxicol. 2020, 39, 765–772. [Google Scholar] [CrossRef]
- Marcinkowska, A.B.; Mankowska, N.D.; Kot, J.; Winklewski, P.J. Impact of Hyperbaric Oxygen Therapy on Cognitive Functions: a Systematic Review. Neuropsychol Rev. 2022, 32, 99–126. [Google Scholar] [CrossRef]
- Ahmed, S.; Kumar, S. Carbon monoxide toxicity and its management: A review. International Journal of Advanced Research in Medicinal Chemistry. 2020, 2, 11–9. [Google Scholar]
- Weaver, L.K. Carbon monoxide poisoning. Undersea Hyperb Med. 2020 First Quarter;47, 151-169.
- Valerio, A.; Verzè, M.; Marchiori, F.; Rucci, I.; De Santis, L.; Aprili, I.; Antolini, L.; Sannino, A.; Canello, A.; Checchin, E.; Mazzola, F.; Ferrari, S.; Garon, M.; Schonsberg, A.; Tardivo, S. Managing a Mass CO Poisoning: Critical Issues and Solutions From the Field to the Hyperbaric Chamber. Disaster Med Public Health Prep. 2017, 11, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Barnett, A.; Beasley, R.; Buchan, C.; Chien, J.; Farah, C.S.; King, G.; McDonald, C.F.; Miller, B.; Munsif, M.; Psirides, A.; Reid, L.; Roberts, M.; Smallwood, N.; Smith, S. Thoracic Society of Australia and New Zealand Position Statement on Acute Oxygen Use in Adults: 'Swimming between the flags'. Respirology. 2022, 27, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Cecconi, M.; Hernandez, G.; Dunser, M.; Antonelli, M.; Baker, T.; Bakker, J.; Duranteau, J.; Einav, S.; Groeneveld, A.B.J.; Harris, T.; Jog, S.; Machado, F.R.; Mer, M.; Monge García, M.I.; Myatra, S.N.; Perner, A.; Teboul, J.L.; Vincent, J.L.; De Backer, D. Fluid administration for acute circulatory dysfunction using basic monitoring: narrative review and expert panel recommendations from an ESICM task force. Intensive Care Med. 2019, 45, 21–32. [Google Scholar] [CrossRef]
- Zamani, N.; Hassanian-Moghaddam, H.; Zamani, N. Strategies for the treatment of acute benzodiazepine toxicity in a clinical setting: the role of antidotes. Expert Opin Drug Metab Toxicol. 2022, 18, 367–379. [Google Scholar] [CrossRef]
- Wilkinson, J. Poisoning. RCEM Lecture Notes: Emergency medicine. 2023, 277-309.
- Barletta, J.F.; Muir, J.; Brown, J.; Dzierba, A. A Systematic Approach to Understanding Acid-Base Disorders in the Critically Ill. Ann Pharmacother. 2024, 58, 65–75. [Google Scholar] [CrossRef]
- Krishnamoorthy, A.; Shaikh, I.A.; Mitra, S.; Jasper, A.; Abhilash, K.P. Delayed neurological sequelae of carbon monoxide poisoning. indian journal of medical specialities. 2015, 6, 63–6. [Google Scholar] [CrossRef]
- Wen, T.; Liang, J.; Wei, Y.; Lin, W.; Pan, L. Analysis of prognosis of neurological sequelae in children with carbon monoxide poisoning. Scientific Reports. 2024, 14, 1–1. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jeong, H.; Choo, Y.H.; Kim, M.; Ha, E.J.; Oh, J.; Shim, Y.; Kim, S.B.; Jung, H.G.; Park, S.H.; Kim, J.O.; Kim, J.; Kim, H.S.; Lee, S. Optimizing Mannitol Use in Managing Increased Intracranial Pressure: A Comprehensive Review of Recent Research and Clinical Experiences. Korean J Neurotrauma. 2023, 19, 162–176. [Google Scholar] [CrossRef] [PubMed]
- Mahenthiran, A.K.; Mahenthiran, A.K.; Mahenthiran, J. Cardiovascular system and COVID-19: manifestations and therapeutics. Rev Cardiovasc Med. 2020, 21, 399–409. [Google Scholar] [CrossRef]
- Noel-Morgan, J.; Muir, W.W. Anesthesia-Associated Relative Hypovolemia: Mechanisms, Monitoring, and Treatment Considerations. Front Vet Sci. 2018, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Ruas, F.; Mendonça, M.C.; Real, F.C.; Vieira, D.N.; Teixeira, H.M. Carbon monoxide poisoning as a cause of death and differential diagnosis in the forensic practice: a retrospective study, 2000-2010. J Forensic Leg Med. 2014, 24, 1–6. [Google Scholar] [CrossRef]
- Osborne, J.; Sobh, M.; Trudel, G. Carbon monoxide as a clinical marker of hemolysis. Am J Hematol. 2023, 98, 1127–1159. [Google Scholar] [CrossRef]
- Parmentier-Decrucq, E.; Mathieu, D. Intoxication au monoxyde de carbone [Carbon monoxide poisoning]. Rev Prat. 2019, 69, 75–79. [Google Scholar]
- Reisinger, A.; Rabensteiner, J.; Hackl, G. Diagnosis of acute intoxications in critically ill patients: focus on biomarkers - part 2: markers for specific intoxications. Biomarkers. 2020, 25, 112–125. [Google Scholar] [CrossRef]
- Oliverio, S. Current challenges in carbon monoxide poisoning diagnosis from an analytical perspective. Front Med (Lausanne). 2023, 10, 1304294. [Google Scholar] [CrossRef]
- Dhama, K.; Latheef, S.K.; Dadar, M.; Samad, H.A.; Munjal, A.; Khandia, R.; Karthik, K.; Tiwari, R.; Yatoo, M.I.; Bhatt, P.; Chakraborty, S.; Singh, K.P.; Iqbal, H.M.N.; Chaicumpa, W.; Joshi, S.K. Biomarkers in Stress Related Diseases/Disorders: Diagnostic, Prognostic, and Therapeutic Values. Front Mol Biosci. 2019, 6, 91. [Google Scholar] [CrossRef]
- Beduk, T.; Durmus, C.; Hanoglu, S.B.; Beduk, D.; Salama, K.N.; Goksel, T.; Turhan, K.; Timur, S. Breath as the mirror of our body is the answer really blowing in the wind? Recent technologies in exhaled breath analysis systems as non-invasive sensing platforms. TrAC Trends in Analytical Chemistry. 2021, 143, 116329. [Google Scholar] [CrossRef]
- Al-Beltagi, M.; Saeed, N.K.; Bediwy, A.S.; Elbeltagi, R. Pulse oximetry in pediatric care: Balancing advantages and limitations. World J Clin Pediatr. 2024, 13, 96950. [Google Scholar] [CrossRef] [PubMed]
- Howard, M. A market for diagnostic devices for extreme point-of-care testing: Are we ASSURED of an ethical outcome? Dev World Bioeth. 2024, 24, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Harrington, R.A.; Califf, R.M.; Balamurugan, A.; Brown, N.; Benjamin, R.M.; Braund, W.E.; Hipp, J.; Konig, M.; Sanchez, E.; Joynt Maddox, K.E. Call to Action: Rural Health: A Presidential Advisory From the American Heart Association and American Stroke Association. Circulation. 2020, 141, e615–e644. [Google Scholar] [CrossRef]
- Seow, M.K.; Ali, M.S. Carbon dioxide and carbon monoxide gas detection system for cars. ELEKTRIKA-Journal of Electrical Engineering. 2021, 20, 65–9. [Google Scholar]
- Nandy, T.; Coutu, R.A., Jr.; Ababei, C. Carbon Monoxide Sensing Technologies for Next-Generation Cyber-Physical Systems. Sensors (Basel). 2018, 18, 3443. [Google Scholar] [CrossRef]
- Sircar, K.; Clower, J.; Shin, M.K.; Bailey, C.; King, M.; Yip, F. Carbon monoxide poisoning deaths in the United States, 1999 to 2012. Am J Emerg Med. 2015, 33, 1140–5. [Google Scholar] [CrossRef]
- Kazanskiy, N.L.; Butt, M.A.; Khonina, S.N. Recent Advances in Wearable Optical Sensor Automation Powered by Battery versus Skin-like Battery-Free Devices for Personal Healthcare-A Review. Nanomaterials (Basel). 2022, 12, 334. [Google Scholar] [CrossRef]
- Pullinger, M.; Kilgour, J.; Goddard, N.; Berliner, N.; Webb, L.; Dzikovska, M.; Lovell, H.; Mann, J.; Sutton, C.; Webb, J.; Zhong, M. The IDEAL household energy dataset, electricity, gas, contextual sensor data and survey data for 255 UK homes. Scientific Data. 2021, 8, 146. [Google Scholar] [CrossRef]
- Turner, P.R.; Bales, D.; Thomas, K.; Goldschmidt, M.; Fisher-McLean, K. Program note: Expanding healthy housing and environmental awareness in rural communities through local and national partnerships. Housing and Society. 2017, 44, 190–207. [Google Scholar] [CrossRef]
- Roberto, C.A.; Swinburn, B.; Hawkes, C.; Huang, T.T.; Costa, S.A.; Ashe, M.; Zwicker, L.; Cawley, J.H.; Brownell, K.D. Patchy progress on obesity prevention: emerging examples, entrenched barriers, and new thinking. Lancet. 2015, 385, 2400–9. [Google Scholar] [CrossRef] [PubMed]
- Raub, J.A.; Mathieu-Nolf, M.; Hampson, N.B.; Thom, S.R. Carbon monoxide poisoning--a public health perspective. Toxicology. 2000, 145, 1–14. [Google Scholar] [CrossRef]
- Cahyo, N.; Sitanggang, R.B.; Simareme, A.A.; Paryanto, P. Impact of crude palm oil on engine performance, emission product, deposit formation, and lubricating oil degradation of low-speed diesel engine: An experimental study. Results in Engineering. 2023, 18, 101156. [Google Scholar] [CrossRef]
- Zuazua-Ros, A.; de Brito Andrade, L.; Dorregaray-Oyaregui, S.; Martín-Gómez, C.; Ramos González, J.C.; Manzueta, R.; Sánchez Saiz-Ezquerra, B.; Ariño, A.H. Crosscutting of the pollutants and building ventilation systems: a literature review. Environ Sci Pollut Res Int. 2023, 30, 66538–66558. [Google Scholar] [CrossRef]
- Zhang, L.; Ou, C.; Magana-Arachchi, D.; Vithanage, M.; Vanka, K.S.; Palanisami, T.; Masakorala, K.; Wijesekara, H.; Yan, Y.; Bolan, N.; Kirkham, M.B. Indoor Particulate Matter in Urban Households: Sources, Pathways, Characteristics, Health Effects, and Exposure Mitigation. Int J Environ Res Public Health. 2021, 18, 11055. [Google Scholar] [CrossRef] [PubMed]
- Fonollosa, J.; Solórzano, A.; Marco, S. Chemical Sensor Systems and Associated Algorithms for Fire Detection: A Review. Sensors (Basel). 2018, 18, 553. [Google Scholar] [CrossRef]
- Mathias, J.A.; Juenger, K.M.; Horton, J.J. Advances in the energy efficiency of residential appliances in the US: A review. Energy Efficiency. 2023, 16, 34. [Google Scholar] [CrossRef]
- Zhao, Y.; McDonell, V.; Samuelsen, S. Residential Fuel Transition and Fuel Interchangeability in Current Self-Aspirating Combustion Applications: Historical Development and Future Expectations. Energies. 2022, 15, 3547. [Google Scholar] [CrossRef]
- Wood, M.D.; Thorne, S.; Kovacs, D.; Butte, G.; Linkov, I.; Thorne, S.; Butte, G.; Hailey, S. Saving Lives from a Silent Killer: Using Mental Modeling to Address Homeowners’ Decision Making About Carbon Monoxide Poisoning. Mental Modeling Approach: Risk Management Application Case Studies. 2017, 195-221.
- Wynn, P.M.; Zou, K.; Young, B.; Majsak-Newman, G.; Hawkins, A.; Kay, B.; Mhizha-Murira, J.; Kendrick, D. Prevention of childhood poisoning in the home: overview of systematic reviews and a systematic review of primary studies. Int J Inj Contr Saf Promot. 2016, 23, 3–28. [Google Scholar] [CrossRef]
- Alexander, D.E. Social media in disaster risk reduction and crisis management. Sci Eng Ethics. 2014, 20, 717–33. [Google Scholar] [CrossRef]
- Tota, P.M. Filling the gaps: the role and impact of international non-governmental organisations in ‘Education for All’. Globalisation, Societies and Education. 2014, 12, 92–109. [Google Scholar] [CrossRef]
- Yared, R.; Abdulrazak, B. Ambient technology to assist elderly people in indoor risks. Computers. 2016, 5, 22. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, X.; Yu, B.; Wang, Z.; Wang, L.; Lei, K.; Zuo, L.; Fan, P.; Liang, T. Exploring the environmental risks and seasonal variations of potentially toxic elements (PTEs) in fine road dust in resource-based cities based on Monte Carlo simulation, geo-detector and random forest model. J Hazard Mater. 2024, 473, 134708. [Google Scholar] [CrossRef]
- Levin, A.; Tonelli, M.; Bonventre, J.; Coresh, J.; Donner, J.A.; Fogo, A.B.; Fox, C.S.; Gansevoort, R.T.; Heerspink, H.J.L.; Jardine, M.; Kasiske, B.; Köttgen, A.; Kretzler, M.; Levey, A.S.; Luyckx, V.A.; Mehta, R.; Moe, O.; Obrador, G.; Pannu, N.; Parikh, C.R.; Perkovic, V.; Pollock, C.; Stenvinkel, P.; Tuttle, K.R.; Wheeler, D.C.; Eckardt, K.U.; ISN Global Kidney Health Summit participants. Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. Lancet. 2017, 390, 1888–1917. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Luan, Y.; Zhang, Q.; Wang, F.; Rao, Y. Effects of Dichloromethane, Nitrate, and Sulfhemoglobin-Induced Substances on Carboxyhemoglobin Detection: A Comprehensive Review. J Anal Toxicol. 2024, bkae096. [Google Scholar] [CrossRef]
- Kumar, P.; Skouloudis, A.N.; Bell, M.; Viana, M.; Carotta, M.C.; Biskos, G.; Morawska, L. Real-time sensors for indoor air monitoring and challenges ahead in deploying them to urban buildings. Sci Total Environ. 2016, 560-561, 150–9. [Google Scholar] [CrossRef] [PubMed]
- Paterson, J.; Berry, P.; Ebi, K.; Varangu, L. Health care facilities resilient to climate change impacts. Int J Environ Res Public Health. 2014, 11, 13097–116. [Google Scholar] [CrossRef]
- Lyon, M.; Fehlmann, C.A.; Augsburger, M.; Schaller, T.; Zimmermann-Ivol, C.; Celi, J.; Gartner, B.A.; Lorenzon, N.; Sarasin, F.; Suppan, L. Evaluation of a Portable Blood Gas Analyzer for Prehospital Triage in Carbon Monoxide Poisoning: Instrument Validation Study. JMIR Form Res. 2023, 7, e48057. [Google Scholar] [CrossRef]
- Farrell, K. Pulse Oximetry and Co-Oximetry. Advanced Monitoring and Procedures for Small Animal Emergency and Critical Care. 2023, 327-38.
- Vasthare, R.; Kumar, S.; Arron, L.Y. Carbon monoxide breath analyzers and its role in tobacco cessation: A narrative review of literature. Journal of International Oral Health. 2018, 10, 71–6. [Google Scholar] [CrossRef]
| Test Name | Principle | Development Time | Advantages | Sensitivity | Limitations | Ref. |
| CO-Oximetry (Spectrophotometry) | Differentiates COHb from oxyhemoglobin using multiple wavelengths of light. | 2–5 minutes | Accurate and direct measurement of COHb levels. | High | Requires specialized equipment; not widely available in all settings. | [60] |
| Blood Gas Analysis with CO-Oximetry | Measures COHb using arterial or venous blood and co-oximetry. | 5–10 minutes | Simultaneous measurement of blood gases and COHb. | High | Time-sensitive; levels decrease quickly after oxygen therapy. | [156] |
| Noninvasive Pulse CO-Oximetry | Uses multi-wavelength light to noninvasively measure COHb. | Real-time | Noninvasive, rapid, and suitable for point-of-care testing. | Moderate to High | Less accurate at very low or very high COHb levels. | [157] |
| Standard Pulse Oximetry | Measures oxygen saturation using two wavelengths of light. | Real-time | Readily available and easy to use. | Low | Cannot distinguish between oxyhemoglobin and carboxyhemoglobin (false normal). | [129] |
| Breath CO Analysis | Measures exhaled CO levels as a proxy for COHb concentration. | 1–2 minutes | Noninvasive, simple, and portable. | Moderate | Limited accuracy; influenced by recent exposure and respiration rate. | [158] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




