Submitted:
20 February 2025
Posted:
20 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
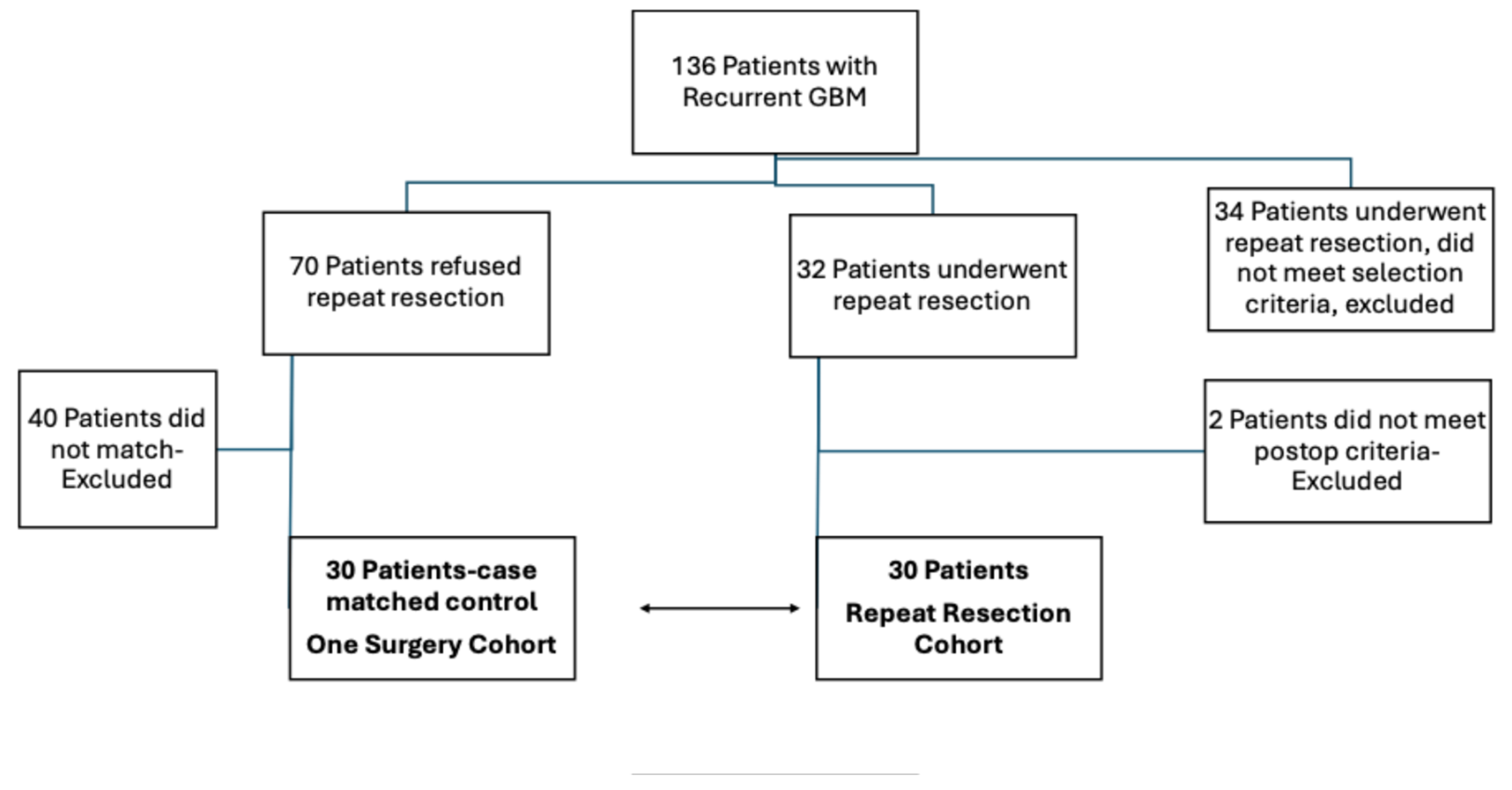
2.1. Study Design
2.2. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. EOR and Molecular Markers
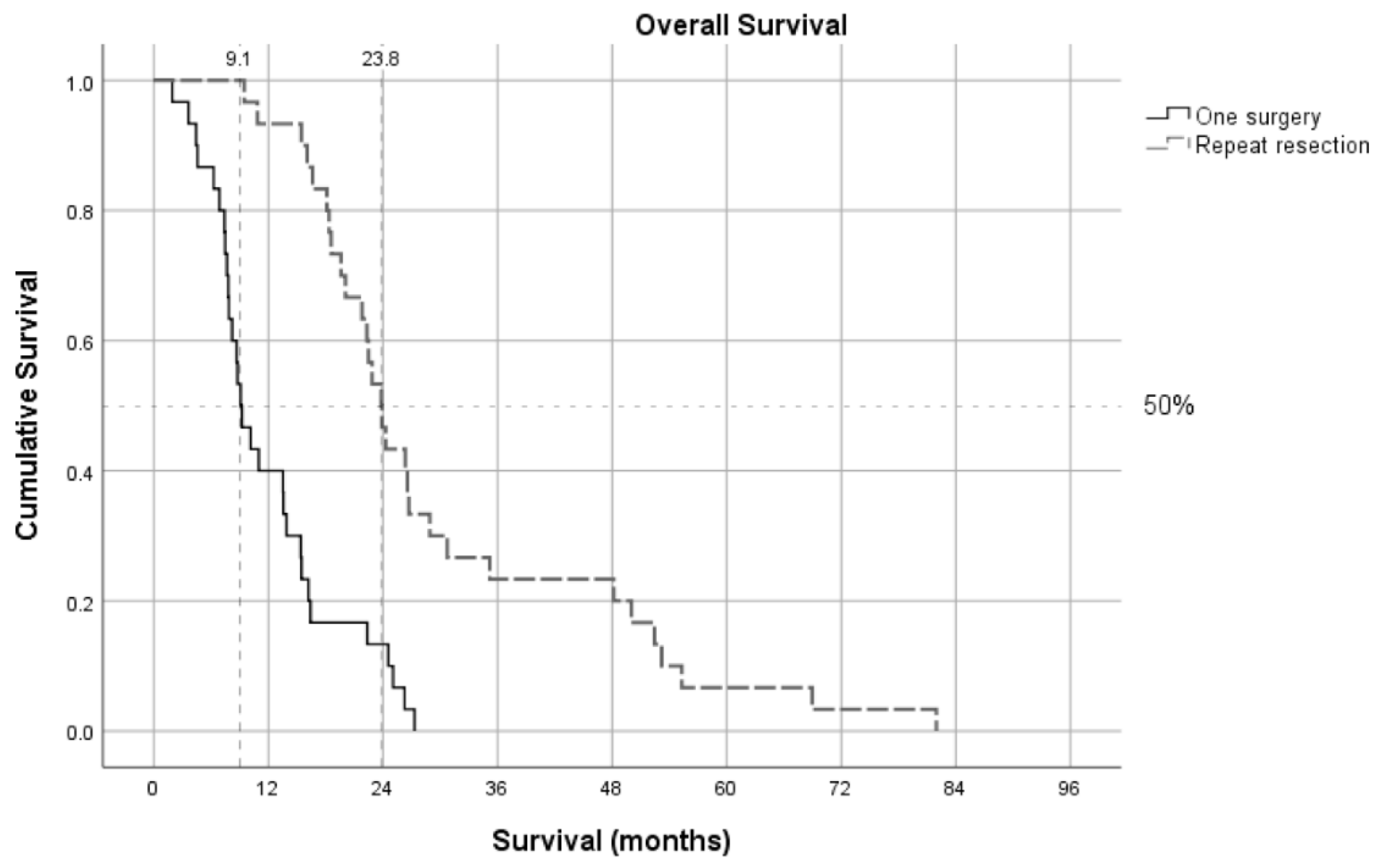
3.3. Overall Survival
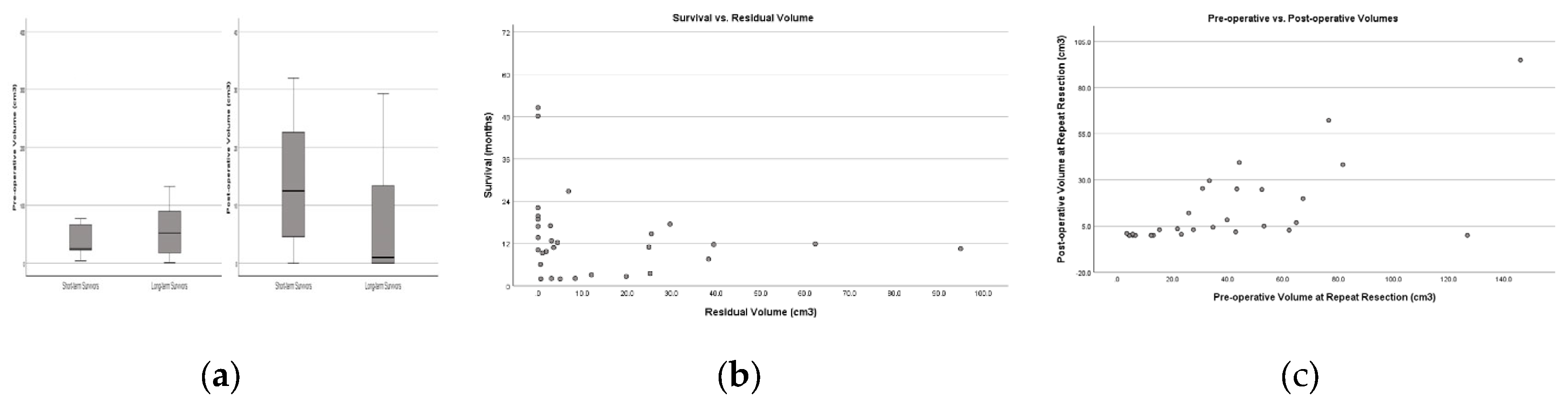
3.4. Factors Related to Long Survival After Repeat Resection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EOR | Extent of resection |
| GBM | Glioblastoma |
| rGBM | Recurrent GBM |
| MGMT | Methylguanine-DNA methyltransferase |
| ASA | The American Society of Anesthesiologists score |
| CE | Contrast-enhancing |
| n-CE | non-contrast-enhancing |
References
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [CrossRef]
- Wen PY, W.M., Lee EQ, . and et al. "Glioblastoma in adults: A society for neuro-oncology (sno) and european society of neuro-oncology (eano) consensus review on current management and future directions." Neuro Oncol (2020): 1073-113.
- Tsien CI, P.S., Dicker AP, . and et al. "Nrg oncology/rtog1205: A randomized phase ii trial of concurrent bevacizumab and reirradiation versus bevacizumab alone as treatment for recurrent glioblastoma." Journal of clinical oncology : official journal of the American Society of Clinical Oncology (2022): 36260832.
- Ringel F, P.H., Sabel M, . and et al. "Clinical benefit from resection of recurrent glioblastomas: Results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection." Neuro Oncol (2016): 96-104.
- Karschnia, P.; Karschnia, P.; Young, J.S.; Young, J.S.; Dono, A.; Dono, A.; Häni, L.; Häni, L.; Sciortino, T.; Sciortino, T.; et al. Prognostic validation of a new classification system for extent of resection in glioblastoma: A report of the RANO resect group. Neuro-Oncology 2022, 25, 940–954. [CrossRef]
- Dono A, Z.P., Holmes E, . and et al. "Impacts of genotypic variants on survival following reoperation for recurrent glioblastoma." J Neurooncol (2022): 353-63.
- Karschnia, P.; Dono, A.; Young, J.S.; Juenger, S.T.; Teske, N.; Häni, L.; Sciortino, T.; Mau, C.Y.; Bruno, F.; Nunez, L.; et al. Prognostic evaluation of re-resection for recurrent glioblastoma using the novel RANO classification for extent of resection: A report of the RANO resect group. Neuro-Oncology 2023, 25, 1672–1685. [CrossRef]
- Karschnia P, Y.J., Dono A, . and et al. "Prognostic validation of a new classification system for extent of resection in glioblastoma: A report of the rano resect group." Neuro Oncol (2022): 35961053.
- Wen, P.Y.; Bent, M.v.D.; Youssef, G.; Cloughesy, T.F.; Ellingson, B.M.; Weller, M.; Galanis, E.; Barboriak, D.P.; de Groot, J.; Gilbert, M.R.; et al. RANO 2.0: Update to the Response Assessment in Neuro-Oncology Criteria for High- and Low-Grade Gliomas in Adults. J. Clin. Oncol. 2023, 41, 5187–5199. [CrossRef]
- Gorlia T, S.R., Brandes AA, . and et al. "New prognostic factors and calculators for outcome prediction in patients with recurrent glioblastoma: A pooled analysis of eortc brain tumour group phase i and ii clinical trials." European journal of cancer (2012): 1176-84.
- Wick W, G.T., Bendszus M, . and et al. "Lomustine and bevacizumab in progressive glioblastoma." The New England journal of medicine (2017): 1954-63.
- Park JK, H.T., Arko L, . and et al. "Scale to predict survival after surgery for recurrent glioblastoma multiforme." Journal of clinical oncology : official journal of the American Society of Clinical Oncology (2010): 3838-43.
- Suchorska, B.; Weller, M.; Tabatabai, G.; Senft, C.; Hau, P.; Sabel, M.C.; Herrlinger, U.; Ketter, R.; Schlegel, U.; Marosi, C.; et al. Complete resection of contrast-enhancing tumor volume is associated with improved survival in recurrent glioblastoma—results from the DIRECTOR trial. Neuro-Oncology 2016, 18, 549–556. [CrossRef]
- Behling F, R.J., Dangel E, . and et al. "Complete and incomplete resection for progressive glioblastoma prolongs post-progression survival." Front Oncol (2022): 755430.
- González, V.; Brell, M.; Fuster, J.; Moratinos, L.; Alegre, D.; López, S.; Ibáñez, J. Analyzing the role of reoperation in recurrent glioblastoma: a 15-year retrospective study in a single institution. World J. Surg. Oncol. 2022, 20, 1–10. [CrossRef]
- Tully, P.A.; Gogos, A.J.; Love, C.; Liew, D.; Drummond, K.J.; Morokoff, A.P. Reoperation for Recurrent Glioblastoma and Its Association With Survival Benefit. Neurosurgery 2016, 79, 678–689. [CrossRef]
- Ringel, F.; Pape, H.; Sabel, M.; Krex, D.; Bock, H.C.; Misch, M.; Weyerbrock, A.; Westermaier, T.; Senft, C.; Schucht, P.; et al. Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro-Oncology 2015, 18, 96–104. [CrossRef]
- Yong, R.L.; Wu, T.; Mihatov, N.; Shen, M.J.; Brown, M.A.; Zaghloul, K.A.; Park, G.E.; Park, J.K. Residual tumor volume and patient survival following reoperation for recurrent glioblastoma. J. Neurosurg. 2014, 121, 802–809. [CrossRef]
- Molinaro AM, H.-J. S., Morshed RA, . and et al. "Association of maximal extent of resection of contrast-enhanced and non-contrast-enhanced tumor with survival within molecular subgroups of patients with newly diagnosed glioblastoma." JAMA Oncol (2020): 495-503.
- Bjorland, L.S.; Mahesparan, R.; Fluge, Ø.; Gilje, B.; Kurz, K.D.; Farbu, E. Impact of extent of resection on outcome from glioblastoma using the RANO resect group classification system: a retrospective, population-based cohort study. Neuro-Oncology Adv. 2023, 5, vdad126. [CrossRef]
- Louis DN, P.A., Wesseling P, . and et al. "The 2021 who classification of tumors of the central nervous system: A summary." Neuro Oncol (2021): 34185076.
- Chen, M.W.; Morsy, A.A.; Liang, S.; Ng, W.H. Re-do Craniotomy for Recurrent Grade IV Glioblastomas: Impact and Outcomes from the National Neuroscience Institute Singapore. World Neurosurg. 2015, 87, 439–445. [CrossRef]
- Park, Y.W.; Choi, K.S.; Foltyn-Dumitru, M.; Brugnara, G.; Banan, R.; Kim, S.; Han, K.; Park, J.E.; Kessler, T.; Bendszus, M.; et al. Incorporating Supramaximal Resection into Survival Stratification of IDH-wildtype Glioblastoma: A Refined Multi-institutional Recursive Partitioning Analysis. Clin. Cancer Res. 2024, 30, 4866–4875. [CrossRef]
- Askun, M.M.; Zengin, Y.; Azizova, A.; Karli-Oguz, K.; Saydam, O.; Strobel, T.; Soylemezoglu, F. Repeat Resection for Recurrent Glioblastoma in the WHO 2021 Era: A Prospective Matched Case-Control Study. Neurosurgery 2024, 70, 200–200. [CrossRef]
| One-surgery (n=30) |
Repeat Resection (n=30) | p | |
|---|---|---|---|
| Age (years) | 54.1±11.05 | 49.3±10.74 | 0.093* |
| Preoperative Volume at first surgery (cm3) | 46.1 (14.1-191.3) | 45.5 (1-320.4) | 0.871# |
| Female/Male | 15/15 | 21/9 | 0.114$ |
| Hypertension (patients) | 11 | 7 | 0.260$ |
| DM (patients) | 4 | 8 | 0.219$ |
| ASA score at first surgery-median (SD) | 2 (0.573) | 2 (0.765) | 0.788§ |
| GCS ≤13 (patients) | 2 | 2 | 1π |
| KPS ≤70 (patients) | 7 | 6 | 0.754 $ |
| EOR at 1st surgery (%) | 86.9 (27.4-100) | 85.8 (23.6-100) | 0.917# |
| Postoperative volume (cm3) | 5,61 (0-59,1) | 2,38 (0-32) | 0,347# |
| RANO class 1 (patients) | 7 | 10 | 0.39$ |
| Left Hemisphere (patients) | 19 | 17 | 0.598$ |
| Eloquent (motor-sensory-speech) (patients) | 18 | 18 | 1.000$ |
| MGMT methylated (patients) | 13 | 11 | 0.764$ |
| TERT mutated (patients) | 7 | 10 | 0.280$ |
| Completed Stupp regimen (patients) | 30 | 27 | 0.333$ |
| Time to first progression since 1st surgery (RANO 2.0) (mo.s) | 5.6 (3-9) | 5.5(3-9.8) | 0.48π |
| 2nd-line chemotherapy prior to offering surgery (patients) | 10 | 11 | 1.000π |
| One-surgery | Repeat Resection | p | |
|---|---|---|---|
| Survival-median (mo.s) | 9.17 (1.9-27.3) | 23.87 (9.4-81.9) | <0.001$ |
| Time to 2nd surgery after first surgery-median (mo.s) | - | 11.63 (3.8-57.1) | |
| Survival after 2nd surgery-median (mo.s) | - | 11.35 (1.97-50.6) | |
| EOR at 1st surgery (%) | 86.7 (0.27-100) | 85.8 (0.24-100) | 0.917# |
| Residual volume at 1st surgery (cm3) | 5,61(0-59,09) | 2,38(0-31,99) | 0,347# |
| RANO class 1 at 1st surgery (patients) | 7 | 10 | 0.56# |
| EOR at 2nd surgery-median (%) | - | 88.2 (0.11-100) | |
| Residual volume at 2nd surgery-median (cm3) | - | 3.3 (0-94.8) | |
| RANO class 1 at 2nd surgery (patients) | 8 |
| Repeat Resection Cohort | Short-term Survivors After 2nd Surgery (n=7) | Long-term Survivors After 2nd Surgery (n=23) | p |
|---|---|---|---|
| Survival after repeat resection (mo.s)-median | 2.1 (2-3.5) | 12.77 (6.1-50.6) | |
| Clinical Markers | |||
| Age (years) | 50.6±10.34 | 49±40 | 0 .734* |
| Gender (Female) | 3 | 18 | 0.153π |
| GCS ≤13 (patients) | 5 | 0 | <0.001π |
| Park Score | 2 (1-3) | 1 (0-2) | 0.086# |
| Volume > 50 cm3 | 2 | 7 | 1.000π |
| MSM (patients) | 3 | 11 | 1.000 π |
| KPS≤70 (patients) | 6 | 0 | 0.007π |
| Hypertension (patients) | 3 | 4 | 0.25 π |
| DM (patients) | 1 | 7 | 0.638 π |
| Stupp protocol completed (patients) | 7 | 20 | 1.000 π |
| Surgical Markers | |||
| Eloquent location (patients) | 3 | 15 | 0.392π |
| Left hemisphere (patients) | 4 | 13 | 1.000π |
| Touch to ventricle (patients) | 6 | 16 | 0.638π |
| Ependymal enhancement (patients) | 2 | 11 | 0.427π |
| Preoperative Volume-median (cm3) | 24.4 (3.6-320.4) | 52 (1-132.6) | 0.774# |
| Postoperative Volume-median (cm3) | 8.35 (0.6-25.1) | 2.79 (0-94.8) | 0.322# |
| EOR-median (%) | 79 (41.9-90.6) | 89.5 (10.7-100) | 0.287# |
| RANO Class 1 (patients) | 0 | 8 | 0.143 π |
| Pathological Markers | |||
| MGMT methylated (patients) | 3 | 8 | 1.000π |
| TERT mutation (patients) | 1 | 9 | 0.179 π |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




