Submitted:
18 February 2025
Posted:
19 February 2025
You are already at the latest version
Abstract
There are many RNA polymerase inhibitors, such as rifampicin, which acts as an antituberculosis agent, and relatively new antiviral drugs, such as sofosbuvir. These drugs have a safe profile and strong efficacy in treating tuberculosis and hepatitis C infections. Studies have shown that high doses of these compounds promote cell longevity (prolonging life span) in humans. It is hypothesized that these drugs also affect human RNA polymerases at high doses. We predicted that such inhibition inhibits the RNA transcription machinery, which is considered a pacemaker of the biological clock. It is important to shift research to human RNA polymerase modulators to develop new versions of drugs treating diseases such as neurodegenerative disorders. Although there is no data about the crystal structure of the human RNA polymerase, the mammalian and even the bacterial structures look conserved. It is important to raise concerns about cancer risk from potential new drugs targeting RNA polymerase. However, adding adjuvant therapy, such as vascular endothelial growth factor inhibitors, to RNA polymerase inhibitors can be a wise strategy at the end of the tunnel. The speed of RNA polymerase elongation modulates longevity. Aging affects the transcription machinery, and transcription activity affects longevity, creating a loop.
Keywords:
Introduction
Eukaryotic RNA Polymerases
Double Effects of Sofosbuvir on the Liver
Effects of Rifampicin on Longevity in a Worm Model
Allicin Longevity Effects
Ribavirin and Longevity
Favipiravir and Longevity
Other RNA Polymerase Inhibitors and Longevity
| The drug | The target | Effect | Mechanism |
| Sofosbuvir | HCV | May Increase the risk of hepatocellular carcinoma In vitro (Tsai, Cheng et al. 2022). | PHOSPHO2, KLHL23, TRIM39, TSNAX-DISC1, and RPP21 gene expression (Tsai, Cheng et al. 2022). |
| Ribavirin | HCV | Anemia if used in combination (Krishnan and Dixit 2011). | Decrease liver inflammation and induce RBCs hemolysis (Soota and Maliakkal 2014). |
| Remdesivir | COVID-19 | Decrease inflammation (Kandhaya-Pillai, Yang et al. 2022) | JAC/STAT1 pathway (Kandhaya-Pillai, Yang et al. 2022) |
| zidovudine | HIV | Prolog life span and antiaging (McIntyre, Molenaars et al. 2023) | Inhibit mTOR and activate the AMPK pathway. other mechanisms (Thanapairoje, Junsiritrakhoon et al. 2023) |
| Favipiravir | Anti -influenzas | No experiments on longevity | Not confirmed |
| Rifampicin | Mycobacteria (antibiotic). | Promote longevity and inhibit oxidative stress (Lee, Baek et al. 2020). | Increase IGF-1 and SERB lipid signaling (Admasu, Chaithanya Batchu et al. 2018). |
Transcription Machinery and Aging
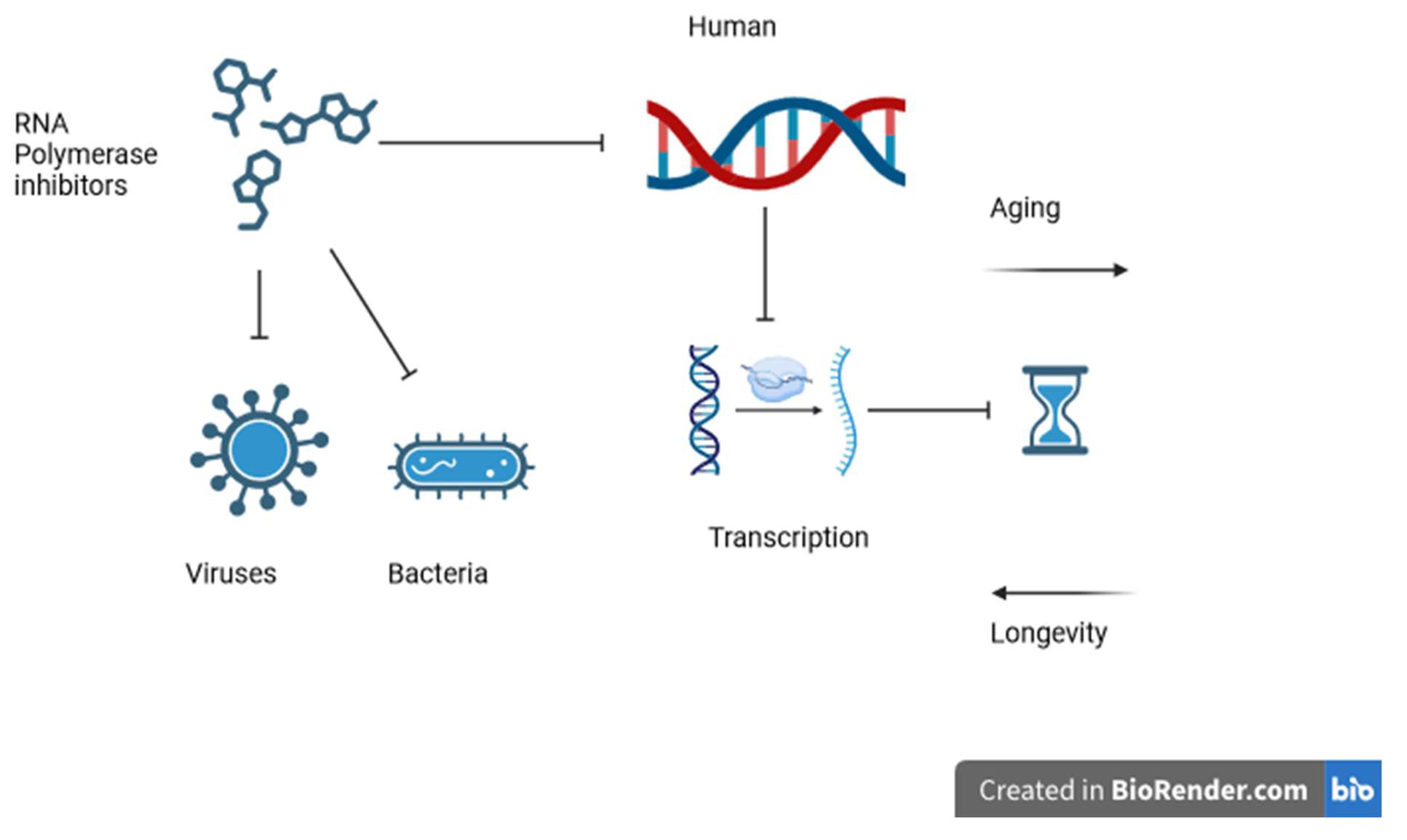
Partial Inhibition of RNA Polymerase and Aging
NRF2 and RNA Polymerase II Inhibition
Aging and Transcriptome Changes
Interaction Between Environmental Stress and Polymerase Inhibition
MAF1 Inhibition of Pol III as a Target for Treating Aging Disorders
MAF1 Regulation of Cancer
Other Functions of MAF1
PTEN Control of RNA Polymerase
Polymerases and Cancer
Pol III and Longevity
The Danger of Carcinogenic Effects of Human RNA Polymerase Inhibitors
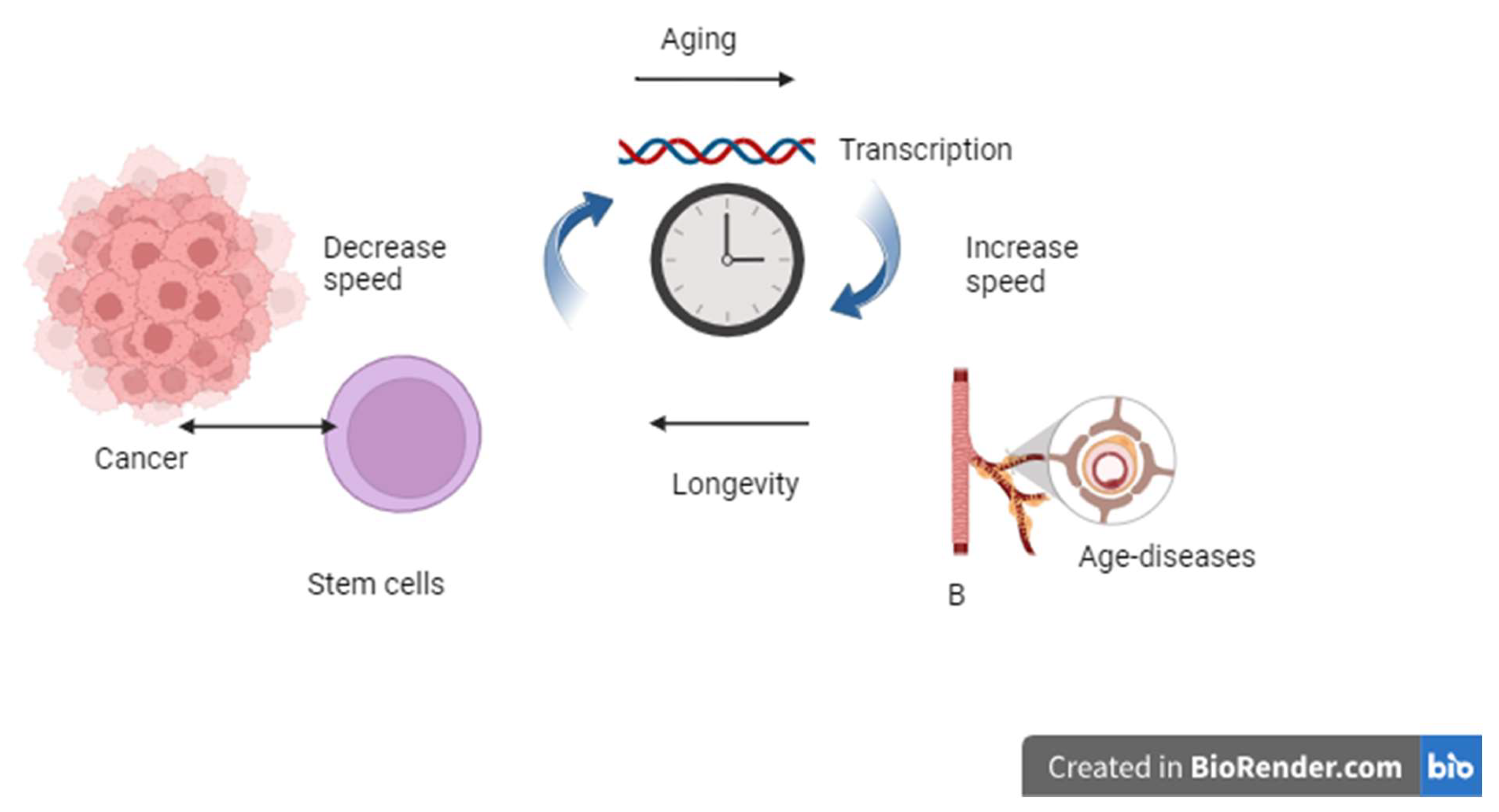
Antiangiogenic Properties and Carcinogenesis
Future Prospects
Conclusion
Author Contributions
Funding
Acknowledgments
Conflict of Interest
Consent to Participate
Human Ethics
References
- Sofosbuvir. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda (MD).
- Abbas, A.; Padmanabhan, R.; Romigh, T.; Eng, C. PTEN modulates gene transcription by redistributing genome-wide RNA polymerase II occupancy. Hum. Mol. Genet. 2019, 28, 2826–2834. [CrossRef]
- Abbas, A.; Romigh, T.; Eng, C. PTEN interacts with RNA polymerase II to dephosphorylate polymerase II C-terminal domain. Oncotarget 2019, 10, 4951–4959. [CrossRef]
- Abdelhamed, W.; El-Kassas, M. Hepatocellular carcinoma and hepatitis C virus treatments: The bold and the beautiful. J. Viral Hepat. 2022, 30, 148–159. [CrossRef]
- Admasu, T.D.; Batchu, K.C.; Barardo, D.; Ng, L.F.; Lam, V.Y.M.; Xiao, L.; Cazenave-Gassiot, A.; Wenk, M.R.; Tolwinski, N.S.; Gruber, J. Drug Synergy Slows Aging and Improves Healthspan through IGF and SREBP Lipid Signaling. Dev. Cell 2018, 47, 67–79.e5. [CrossRef]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. Hormesis: Highly Generalizable and Beyond Laboratory. Trends Plant Sci. 2020, 25, 1076–1086. [CrossRef]
- Augusti, K.T.; Jose, R.; Sajitha, G.R.; Augustine, P. A Rethinking on the Benefits and Drawbacks of Common Antioxidants and a Proposal to Look for the Antioxidants in Allium Products as Ideal Agents: A Review. Indian J. Clin. Biochem. 2011, 27, 6–20. [CrossRef]
- Bader, C.D.; Panter, F.; Garcia, R.; Tchesnokov, E.P.; Haid, S.; Walt, C.; Spröer, C.; Kiefer, A.F.; Götte, M.; Overmann, J.; et al. Sandacrabins – Structurally Unique Antiviral RNA Polymerase Inhibitors from a Rare Myxobacterium**. Chem. – A Eur. J. 2022, 28, e202104484. [CrossRef]
- Biswas, B. B., A. Ganguly and A. Das (1975). “Eukaryotic RNA polymerases and the factors that control them.” Prog Nucleic Acid Res Mol Biol 15(0): 145-184. [CrossRef]
- Blagosklonny, M.V. Aging and Immortality: Quasi-Programmed Senescence and Its Pharmacologic Inhibition. Cell Cycle 2006, 5, 2087–2102. [CrossRef]
- Blagosklonny, M.V. An anti-aging drug today: from senescence-promoting genes to anti-aging pill. Drug Discov. Today 2007, 12, 218–224. [CrossRef]
- Blair, D. G. (1988). “Eukaryotic RNA polymerases.” Comp Biochem Physiol B 89(4): 647-670.
- Bozukova, M.; Nikopoulou, C.; Kleinenkuhnen, N.; Grbavac, D.; Goetsch, K.; Tessarz, P. Aging is associated with increased chromatin accessibility and reduced polymerase pausing in liver. Mol. Syst. Biol. 2022, 18, e11002. [CrossRef]
- Bywater, M.J.; Pearson, R.B.; McArthur, G.A.; Hannan, R.D. Dysregulation of the basal RNA polymerase transcription apparatus in cancer. Nat. Rev. Cancer 2013, 13, 299–314. [CrossRef]
- Bywater, M.J.; Poortinga, G.; Sanij, E.; Hein, N.; Peck, A.; Cullinane, C.; Wall, M.; Cluse, L.; Drygin, D.; Anderes, K.; et al. Inhibition of RNA Polymerase I as a Therapeutic Strategy to Promote Cancer-Specific Activation of p53. Cancer Cell 2012, 22, 51–65. [CrossRef]
- Cabarcas-Petroski, S.; Olshefsky, G.; Schramm, L. MAF1 is a predictive biomarker in HER2 positive breast cancer. PLOS ONE 2023, 18, e0291549. [CrossRef]
- Calabrese, E. J. and L. A. Baldwin (1998). “Can the concept of hormesis Be generalized to carcinogenesis?” Regul Toxicol Pharmacol 28(3): 230-241.
- Calabrese, E.J.; Nascarella, M.; Pressman, P.; Hayes, A.W.; Dhawan, G.; Kapoor, R.; Calabrese, V.; Agathokleous, E. Hormesis determines lifespan. Ageing Res. Rev. 2024, 94, 102181. [CrossRef]
- Campbell, E.A.; Korzheva, N.; Mustaev, A.; Murakami, K.; Nair, S.; Goldfarb, A.; Darst, S.A. Structural Mechanism for Rifampicin Inhibition of Bacterial RNA Polymerase. 2001, 104, 901–912. [CrossRef]
- Celik, I., M. Erol and Z. Duzgun (2022). “In silico evaluation of potential inhibitory activity of remdesivir, favipiravir, ribavirin and galidesivir active forms on SARS-CoV-2 RNA polymerase.” Mol Divers 26(1): 279-292. [CrossRef]
- Chaudhary, K.; Deb, S.; Moniaux, N.; Ponnusamy, M.P.; Batra, S.K. Human RNA polymerase II-associated factor complex: dysregulation in cancer. Oncogene 2007, 26, 7499–7507. [CrossRef]
- Chen, Z.; Mai, Q.; Wang, Q.; Gou, Q.; Shi, F.; Mo, Z.; Cui, W.; Zhuang, W.; Li, W.; Xu, R.; et al. CircPOLR2A Promotes Proliferation and Impedes Apoptosis of Glioblastoma Multiforme Cells by Up-regulating POU3F2 to Facilitate SOX9 Transcription. Neuroscience 2022, 503, 118–130. [CrossRef]
- Debès, C.; Papadakis, A.; Grönke, S.; Karalay, Ö.; Tain, L.S.; Mizi, A.; Nakamura, S.; Hahn, O.; Weigelt, C.; Josipovic, N.; et al. Ageing-associated changes in transcriptional elongation influence longevity. Nature 2023, 616, 814–821. [CrossRef]
- Martínez, C.D.; Gómez-Rubio, M.; Gómez-Domínguez, C. Is hepatitis C direct-acting antiviral therapy a risk factor for the development and recurrence of hepatocellular carcinoma? Narrative literature review and clinical practice recommendations. Ann. Hepatol. 2020, 21, 100225. [CrossRef]
- Dienemann, C.; Schwalb, B.; Schilbach, S.; Cramer, P. Promoter Distortion and Opening in the RNA Polymerase II Cleft. Mol. Cell 2019, 73, 97–106.e4. [CrossRef]
- Doğan, M.F.; Kaya, K.; Demirel, H.H.; Başeğmez, M.; Şahin, Y.; Çiftçi, O.; Oban, V.; Dikeç, G. The effect of vitamin C supplementation on favipiravir-induced oxidative stress and proinflammatory damage in livers and kidneys of rats. Immunopharmacol. Immunotoxicol. 2023, 45, 521–526. [CrossRef]
- Drygin, D.; Lin, A.; Bliesath, J.; Ho, C.B.; O’Brien, S.E.; Proffitt, C.; Omori, M.; Haddach, M.; Schwaebe, M.K.; Siddiqui-Jain, A.; et al. Targeting RNA Polymerase I with an Oral Small Molecule CX-5461 Inhibits Ribosomal RNA Synthesis and Solid Tumor Growth. Cancer Res. 2011, 71, 1418–1430. [CrossRef]
- Drygin, D.; Rice, W.G.; Grummt, I. The RNA Polymerase I Transcription Machinery: An Emerging Target for the Treatment of Cancer. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 131–156. [CrossRef]
- Feldberg, R.S.; Chang, S.C.; Kotik, A.N.; Nadler, M.; Neuwirth, Z.; Sundstrom, D.C.; Thompson, N.H. In vitro mechanism of inhibition of bacterial cell growth by allicin. Antimicrob. Agents Chemother. 1988, 32, 1763–1768. [CrossRef]
- Filer, D.; Thompson, M.A.; Takhaveev, V.; Dobson, A.J.; Kotronaki, I.; Green, J.W.M.; Heinemann, M.; Tullet, J.M.A.; Alic, N. RNA polymerase III limits longevity downstream of TORC1. Nature 2017, 552, 263–267. [CrossRef]
- Fukushima, S.; Kinoshita, A.; Puatanachokchai, R.; Kushida, M.; Wanibuchi, H.; Morimura, K. Hormesis and dose–response-mediated mechanisms in carcinogenesis: evidence for a threshold in carcinogenicity of non-genotoxic carcinogens. Carcinog. 2005, 26, 1835–1845. [CrossRef]
- Golegaonkar, S.; Tabrez, S.S.; Pandit, A.; Sethurathinam, S.; Jagadeeshaprasad, M.G.; Bansode, S.; Sampathkumar, S.; Kulkarni, M.J.; Mukhopadhyay, A. Rifampicin reduces advanced glycation end products and activates DAF-16 to increase lifespan in Caenorhabditis elegans. Aging Cell 2015, 14, 463–473. [CrossRef]
- Goodfellow, S. J. and J. C. Zomerdijk (2013). “Basic mechanisms in RNA polymerase I transcription of the ribosomal RNA genes.” Subcell Biochem 61: 211-236. [CrossRef]
- Grunewald, M.; Kumar, S.; Sharife, H.; Volinsky, E.; Gileles-Hillel, A.; Licht, T.; Permyakova, A.; Hinden, L.; Azar, S.; Friedmann, Y.; et al. Counteracting age-related VEGF signaling insufficiency promotes healthy aging and extends life span. Science 2021, 373, 533–+. [CrossRef]
- Gunaydin-Akyildiz, A.; Aksoy, N.; Boran, T.; Ilhan, E.N.; Ozhan, G. Favipiravir induces oxidative stress and genotoxicity in cardiac and skin cells. Toxicol. Lett. 2022, 371, 9–16. [CrossRef]
- Gyenis, A.; Chang, J.; Demmers, J.J.P.G.; Bruens, S.T.; Barnhoorn, S.; Brandt, R.M.C.; Baar, M.P.; Raseta, M.; Derks, K.W.J.; Hoeijmakers, J.H.J.; et al. Genome-wide RNA polymerase stalling shapes the transcriptome during aging. Nat. Genet. 2023, 55, 268–279. [CrossRef]
- Harris, C. C. (1976). “The carcinogenicity of anticancer drugs: a hazard in man.” Cancer 37(2 Suppl): 1014-1023. [CrossRef]
- Hokonohara, K.; Nishida, N.; Miyoshi, N.; Takahashi, H.; Haraguchi, N.; Hata, T.; Matsuda, C.; Mizushima, T.; Doki, Y.; Mori, M. Involvement of MAF1 homolog, negative regulator of RNA polymerase III in colorectal cancer progression. Int. J. Oncol. 2019, 54, 1001–1009. [CrossRef]
- Horikawa, I. (2020). “Balancing and Differentiating p53 Activities toward Longevity and No Cancer?” Cancer Res 80(23): 5164-5165. [CrossRef]
- Horvath, S.; Raj, K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat. Rev. Genet. 2018, 19, 371–384. [CrossRef]
- Irschik, H.; Augustiniak, H.; Gerth, K.; Höfle, G.; Reichenbach, H. Antibiotics from gliding bacteria. No. 68. The Ripostatins, Novel Inhibitors of Eubacterial RNA Polymerase Isolated from Myxobacteria. J. Antibiot. 1995, 48, 787–792. [CrossRef]
- Jiang, Q.; Zhang, J.; Li, F.; Ma, X.; Wu, F.; Miao, J.; Li, Q.; Wang, X.; Sun, R.; Yang, Y.; et al. POLR2A Promotes the Proliferation of Gastric Cancer Cells by Advancing the Overall Cell Cycle Progression. Front. Genet. 2021, 12. [CrossRef]
- Johnson, S. A., L. Dubeau, M. Kawalek, A. Dervan, A. H. Schonthal, C. V. Dang and D. L. Johnson (2003). “Increased expression of TATA-binding protein, the central transcription factor, can contribute to oncogenesis.” Mol Cell Biol 23(9): 3043-3051. [CrossRef]
- Kandhaya-Pillai, R., X. Yang, T. Tchkonia, G. M. Martin, J. L. Kirkland and J. Oshima (2022). “TNF-alpha/IFN-gamma synergy amplifies senescence-associated inflammation and SARS-CoV-2 receptor expression via hyper-activated JAK/STAT1.” Aging Cell 21(6): e13646. [CrossRef]
- Kim, J. W., R. K. Ponce and R. A. Okimoto (2021). “Capicua in Human Cancer.” Trends Cancer 7(1): 77-86. [CrossRef]
- Klaunig, J.E. Cancer Biology and Hormesis: Commentary. Crit. Rev. Toxicol. 2005, 35, 593–594. [CrossRef]
- Kobayashi, E.H.; Suzuki, T.; Funayama, R.; Nagashima, T.; Hayashi, M.; Sekine, H.; Tanaka, N.; Moriguchi, T.; Motohashi, H.; Nakayama, K.; et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat. Commun. 2016, 7, 11624. [CrossRef]
- Krishnan, S.M.; Dixit, N.M. Ribavirin-Induced Anemia in Hepatitis C Virus Patients Undergoing Combination Therapy. PLOS Comput. Biol. 2011, 7, e1001072. [CrossRef]
- Kulaberoglu, Y., Y. Malik, G. Borland, C. Selman, N. Alic and J. M. A. Tullet (2021). “RNA Polymerase III, Ageing and Longevity.” Front Genet 12: 705122. [CrossRef]
- Lapierre, J.; Hub, J.S. DNA opening during transcription initiation by RNA polymerase II in atomic detail. Biophys. J. 2022, 121, 4299–4310. [CrossRef]
- Lautré, W.; Richard, E.; Feugeas, J.-P.; Dumay-Odelot, H.; Teichmann, M. The POLR3G Subunit of Human RNA Polymerase III Regulates Tumorigenesis and Metastasis in Triple-Negative Breast Cancer. Cancers 2022, 14, 5732. [CrossRef]
- Lee, E.H.; Baek, S.Y.; Park, J.Y.; Kim, Y.W. Rifampicin activates AMPK and alleviates oxidative stress in the liver as mediated with Nrf2 signaling. Chem. Interactions 2020, 315, 108889. [CrossRef]
- Lee, S.-H.; Lee, J.-H.; Lee, H.-Y.; Min, K.-J. Sirtuin signaling in cellular senescence and aging. BMB Rep. 2019, 52, 24–34. [CrossRef]
- Lee, S.-W.; Chen, L.-S.; Yang, S.-S.; Huang, Y.-H.; Lee, T.-Y. Direct-Acting Antiviral Therapy for Hepatitis C Virus in Patients with BCLC Stage B/C Hepatocellular Carcinoma. Viruses 2022, 14, 2316. [CrossRef]
- Levine, M.E.; Lu, A.T.; Quach, A.; Chen, B.H.; Assimes, T.L.; Bandinelli, S.; Hou, L.; Baccarelli, A.A.; Stewart, J.D.; Li, Y.; et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging 2018, 10, 573–591. [CrossRef]
- Li, X.-L.; Xie, Y.; Chen, Y.-L.; Zhang, Z.-M.; Tao, Y.-F.; Li, G.; Wu, D.; Wang, H.-R.; Zhuo, R.; Pan, J.-J.; et al. The RNA polymerase II subunit B (RPB2) functions as a growth regulator in human glioblastoma. Biochem. Biophys. Res. Commun. 2023, 674, 170–182. [CrossRef]
- Lien, E. J. and X. C. Ou (1985). “Carcinogenicity of some anticancer drugs--a survey.” J Clin Hosp Pharm 10(3): 223-242. [CrossRef]
- Lin, X.-L.; Liu, Y.; Liu, M.; Hu, H.; Pan, Y.; Fan, X.-J.; Hu, X.-M.; Zou, W.-W. Inhibition of Hydrogen Peroxide-Induced Human Umbilical Vein Endothelial Cells Aging by Allicin Depends on Sirtuin1 Activation. Med Sci. Monit. 2017, 23, 563–570. [CrossRef]
- Lopez-Otin, C., M. A. Blasco, L. Partridge, M. Serrano and G. Kroemer (2013). “The hallmarks of aging.” Cell 153(6): 1194-1217. [CrossRef]
- López-Otín, C.; Pietrocola, F.; Roiz-Valle, D.; Galluzzi, L.; Kroemer, G. Meta-hallmarks of aging and cancer. Cell Metab. 2023, 35, 12–35. [CrossRef]
- Loveridge, C.J.; Slater, S.; Campbell, K.J.; Nam, N.A.; Knight, J.; Ahmad, I.; Hedley, A.; Lilla, S.; Repiscak, P.; Patel, R.; et al. BRF1 accelerates prostate tumourigenesis and perturbs immune infiltration. Oncogene 2019, 39, 1797–1806. [CrossRef]
- Lu, L.; Su, S.; Yang, H.; Jiang, S. Antivirals with common targets against highly pathogenic viruses. Cell 2021, 184, 1604–1620. [CrossRef]
- Martinez-Jimenez, C.P.; Eling, N.; Chen, H.-C.; Vallejos, C.A.; Kolodziejczyk, A.A.; Connor, F.; Stojic, L.; Rayner, T.F.; Stubbington, M.J.T.; Teichmann, S.A.; et al. Aging increases cell-to-cell transcriptional variability upon immune stimulation. Science 2017, 355, 1433–1436. [CrossRef]
- Corrales, G.M.; Filer, D.; Wenz, K.C.; Rogan, A.; Phillips, G.; Li, M.; Feseha, Y.; Broughton, S.J.; Alic, N. Partial Inhibition of RNA Polymerase I Promotes Animal Health and Longevity. Cell Rep. 2020, 30, 1661–1669.e4. [CrossRef]
- McIntyre, R. L., M. Molenaars, B. V. Schomakers, A. W. Gao, R. Kamble, A. Jongejan, M. van Weeghel, A. B. P. van Kuilenburg, R. Possemato, R. H. Houtkooper and G. E. Janssens (2023). “Anti-retroviral treatment with zidovudine alters pyrimidine metabolism, reduces translation, and extends healthy longevity via ATF-4.” Cell Rep 42(1): 111928. [CrossRef]
- Moniaux, N., C. Nemos, B. M. Schmied, S. C. Chauhan, S. Deb, K. Morikane, A. Choudhury, M. Vanlith, M. Sutherlin, J. M. Sikela, M. A. Hollingsworth and S. K. Batra (2006). “The human homologue of the RNA polymerase II-associated factor 1 (hPaf1), localized on the 19q13 amplicon, is associated with tumorigenesis.” Oncogene 25(23): 3247-3257. [CrossRef]
- Park, Y.; Lee, S.; Yoon, J.; Kim, E.; Park, S. Allicin protects porcine oocytes against damage during aging in vitro. Mol. Reprod. Dev. 2019, 86, 1116–1125. [CrossRef]
- Pluta, K.; Lefebvre, O.; Martin, N.C.; Smagowicz, W.J.; Stanford, D.R.; Ellis, S.R.; Hopper, A.K.; Sentenac, A.; Boguta, M. Maf1p, a Negative Effector of RNA Polymerase III in Saccharomyces cerevisiae. Mol. Cell. Biol. 2001, 21, 5031–5040. [CrossRef]
- Rangaraju, S., G. M. Solis, R. C. Thompson, R. L. Gomez-Amaro, L. Kurian, S. E. Encalada, A. B. Niculescu, 3rd, D. R. Salomon and M. Petrascheck (2015). “Suppression of transcriptional drift extends C. elegans lifespan by postponing the onset of mortality.” Elife 4: e08833. [CrossRef]
- Rhoads, R. E., T. D. Dinkova and N. L. Korneeva (2006). “Mechanism and regulation of translation in C. elegans.” WormBook: 1-18.
- Ruggero, D. Revisiting the Nucleolus: From Marker to Dynamic Integrator of Cancer Signaling. Sci. Signal. 2012, 5, pe38–pe38. [CrossRef]
- Sancar, A. and R. N. Van Gelder (2021). “Clocks, cancer, and chronochemotherapy.” Science 371(6524). [CrossRef]
- Santana-Salgado, I.; Bautista-Santos, A.; Moreno-Alcántar, R. Risk factors for developing hepatocellular carcinoma in patients treated with direct-acting antivirals. Rev. De Gastroenterol. De Mex. 2022, 87, 455–461. [CrossRef]
- Sapena, V., M. Enea, F. Torres, C. Celsa, J. Rios, G. E. M. Rizzo, P. Nahon, Z. Marino, R. Tateishi, T. Minami, A. Sangiovanni, X. Forns, H. Toyoda, S. Brillanti, F. Conti, E. Degasperi, M. L. Yu, P. C. Tsai, K. Jean, M. El Kassas, H. I. Shousha, A. Omar, C. Zavaglia, H. Nagata, M. Nakagawa, Y. Asahina, A. G. Singal, C. Murphy, M. Kohla, C. Masetti, J. F. Dufour, N. Merchante, L. Cavalletto, L. L. Chemello, S. Pol, J. Crespo, J. L. Calleja, R. Villani, G. Serviddio, A. Zanetto, S. Shalaby, F. P. Russo, R. Bielen, F. Trevisani, C. Camma, J. Bruix, G. Cabibbo and M. Reig (2022). “Hepatocellular carcinoma recurrence after direct-acting antiviral therapy: an individual patient data meta-analysis.” Gut 71(3): 593-604. [CrossRef]
- Schmähl, D. Carcinogenicity of anticancer drugs and especially alkylating agents. 1986, 29–35.
- Scott, P. H., C. A. Cairns, J. E. Sutcliffe, H. M. Alzuherri, A. McLees, A. G. Winter and R. J. White (2001). “Regulation of RNA polymerase III transcription during cell cycle entry.” J Biol Chem 276(2): 1005-1014. [CrossRef]
- Shen, C.; Cheng, X.; Li, D.; Meng, Q. Investigation of rifampicin-induced hepatotoxicity in rat hepatocytes maintained in gel entrapment culture. Cell Biol. Toxicol. 2008, 25, 265–274. [CrossRef]
- Shichiri, M.; Tanaka, Y. Inhibition of cancer progression by rifampicin: Involvement of antiangiogenic and anti-tumor effects. Cell Cycle 2010, 9, 64–68. [CrossRef]
- Soota, K.; Maliakkal, B. Ribavirin induced hemolysis: A novel mechanism of action against chronic hepatitis C virus infection. World J. Gastroenterol. 2014, 20, 16184–90. [CrossRef]
- Sun, Y., C. Chen, R. Xue, Y. Wang, B. Dong, J. Li, C. Chen, J. Jiang, W. Fan, Z. Liang, H. Huang, R. Fang, G. Dai, Y. Yan, T. Yang, X. Li, Z. P. Huang, Y. Dong and C. Liu (2019). “Maf1 ameliorates cardiac hypertrophy by inhibiting RNA polymerase III through ERK1/2.” Theranostics 9(24): 7268-7281.
- Tang, W.; Liu, S.; Degen, D.; Ebright, R.H.; Prusov, E.V. Synthesis and Evaluation of Novel Analogues of Ripostatins. Chem. – A Eur. J. 2014, 20, 12310–12319. [CrossRef]
- Thanapairoje, K.; Junsiritrakhoon, S.; Wichaiyo, S.; Osman, M.A.; Supharattanasitthi, W. Anti-ageing effects of FDA-approved medicines: a focused review. J. Basic Clin. Physiol. Pharmacol. 2023, 34, 277–289. [CrossRef]
- Tsai, W.-L.; Cheng, J.-S.; Liu, P.-F.; Chang, T.-H.; Sun, W.-C.; Chen, W.-C.; Shu, C.-W. Sofosbuvir induces gene expression for promoting cell proliferation and migration of hepatocellular carcinoma cells. Aging 2022, 14, 5710–5726. [CrossRef]
- Turowski, T.W.; Tollervey, D. Transcription by RNA polymerase III: insights into mechanism and regulation. Biochem. Soc. Trans. 2016, 44, 1367–1375. [CrossRef]
- Ugwu, K. O., I. S. Onah, G. C. Mbah and I. M. Ezeonu (2020). “Rifampicin resistance patterns and dynamics of tuberculosis and drug-resistant tuberculosis in Enugu, South Eastern Nigeria.” J Infect Dev Ctries 14(9): 1011-1018. [CrossRef]
- Ulasi, A.; Nwachukwu, N.; Onyeagba, R.; Umeham, S.; Amadi, A. Prevalence of rifampicin resistant tuberculosis among pulmonary tuberculosis patients In Enugu, Nigeria. Afr. Heal. Sci. 2022, 22, 156–161. [CrossRef]
- Vahidi, S.; Agah, S.; Mirzajani, E.; Gharakhyli, E.A.; Norollahi, S.E.; Taramsari, M.R.; Babaei, K.; Samadani, A.A. microRNAs, oxidative stress, and genotoxicity as the main inducers in the pathobiology of cancer development. Horm. Mol. Biol. Clin. Investig. 2024, 45, 55–73. [CrossRef]
- Ramani, M.K.V.; Yang, W.; Irani, S.; Zhang, Y. Simplicity is the Ultimate Sophistication—Crosstalk of Post-translational Modifications on the RNA Polymerase II. J. Mol. Biol. 2021, 433, 166912–166912. [CrossRef]
- Vermulst, M.; Denney, A.S.; Lang, M.J.; Hung, C.-W.; Moore, S.; Moseley, M.A.; Thompson, J.W.; Madden, V.; Gauer, J.; Wolfe, K.J.; et al. Transcription errors induce proteotoxic stress and shorten cellular lifespan. Nat. Commun. 2015, 6, 1–11. [CrossRef]
- Werner, F.; Grohmann, D. Evolution of multisubunit RNA polymerases in the three domains of life. Nat. Rev. Microbiol. 2011, 9, 85–98. [CrossRef]
- White, R.J. RNA polymerase III transcription and cancer. Oncogene 2004, 23, 3208–3216. [CrossRef]
- White, R.J.; Gottlieb, T.M.; Downes, C.S.; Jackson, S.P. Cell Cycle Regulation of RNA Polymerase III Transcription. Mol. Cell. Biol. 1995, 15, 6653–6662. [CrossRef]
- Willis, I. M. (2018). “Maf1 phenotypes and cell physiology.” Biochim Biophys Acta Gene Regul Mech 1861(4): 330-337. [CrossRef]
- Willis, I.M.; Moir, R.D. Signaling to and from the RNA Polymerase III Transcription and Processing Machinery. Annu. Rev. Biochem. 2018, 87, 75–100. [CrossRef]
- Winter, A.G.; Sourvinos, G.; Allison, S.J.; Tosh, K.; Scott, P.H.; Spandidos, D.A.; White, R.J. RNA polymerase III transcription factor TFIIIC2 is overexpressed in ovarian tumors. Proc. Natl. Acad. Sci. 2000, 97, 12619–12624. [CrossRef]
- Xu, J.; Liu, Y.; Li, Y.; Wang, H.; Stewart, S.; Van der Jeught, K.; Agarwal, P.; Zhang, Y.; Liu, S.; Zhao, G.; et al. Precise targeting of POLR2A as a therapeutic strategy for human triple negative breast cancer. Nat. Nanotechnol. 2019, 14, 388–397. [CrossRef]
- Yoshioka, K.-I.; Kusumoto-Matsuo, R.; Matsuno, Y.; Ishiai, M. Genomic Instability and Cancer Risk Associated with Erroneous DNA Repair. Int. J. Mol. Sci. 2021, 22, 12254. [CrossRef]
- Yousefsani, B.S.; Nabavi, N.; Pourahmad, J. Contrasting Role of Dose Increase in Modulating Sofosbuvir-Induced Hepatocyte Toxicity. Drug Res. 2020, 70, 137–144. [CrossRef]
- Zaragoza, D.; Ghavidel, A.; Heitman, J.; Schultz, M.C. Rapamycin Induces the G0 Program of Transcriptional Repression in Yeast by Interfering with the TOR Signaling Pathway. Mol. Cell. Biol. 1998, 18, 4463–4470. [CrossRef]
- Zhang, S., X. Li, H. Y. Wang and X. F. Steven Zheng (2018). “Beyond regulation of pol III: Role of MAF1 in growth, metabolism, aging and cancer.” Biochim Biophys Acta Gene Regul Mech 1861(4): 338-343. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




