Submitted:
14 February 2025
Posted:
17 February 2025
You are already at the latest version
Abstract
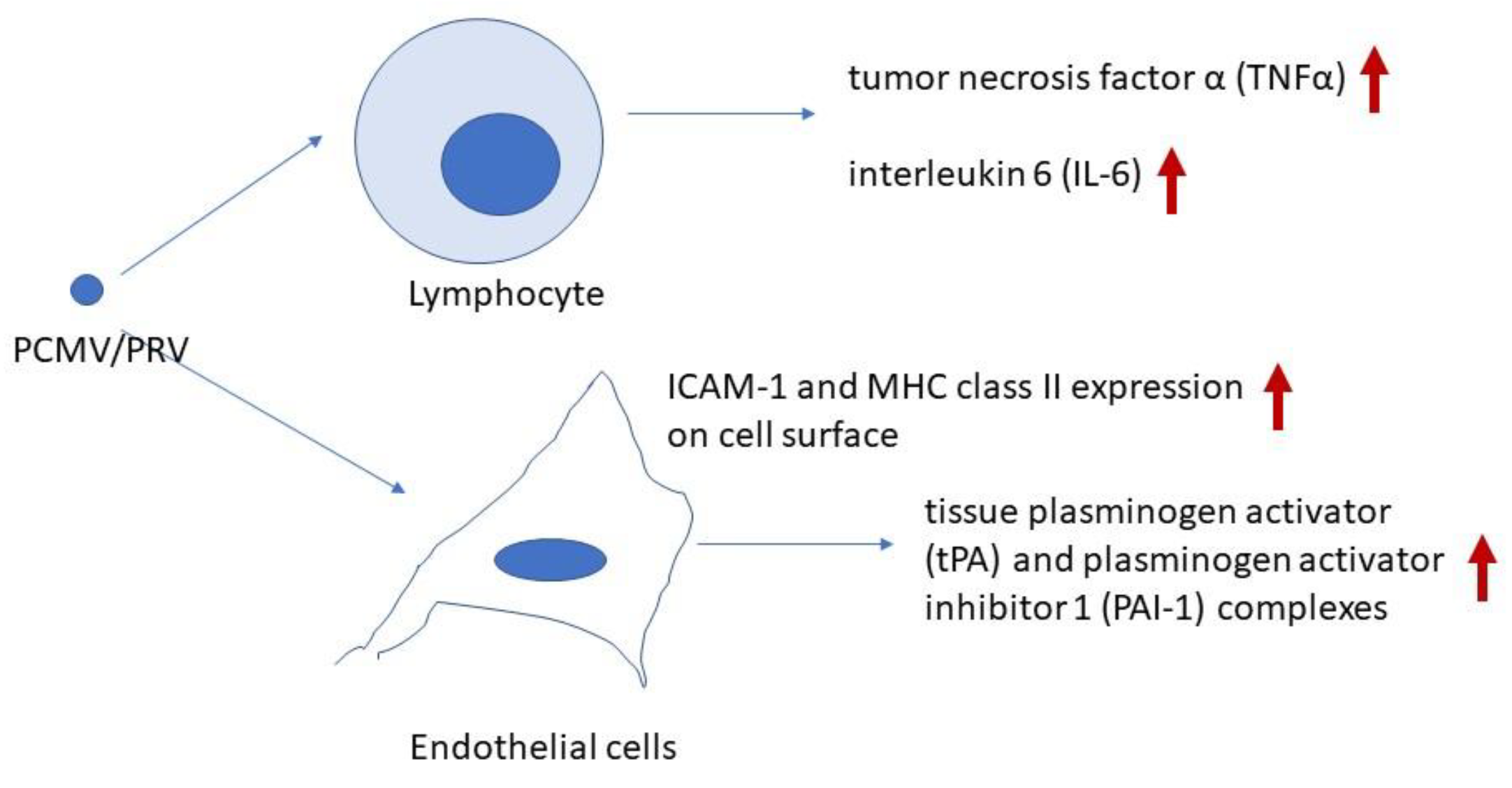
Porcine cytomegalovirus/porcine roseolovirus (PCMV/PRV), a porcine herpesvirus, has been shown to significantly reduce the survival time of porcine xenotransplants in non-human primates. The virus was detected in all examined organs of baboons transplanted with PCMV/PRV-positive organs and it was also transmitted to the first human recipient of a pig heart, contributing to the patient’s death. PCMV/PRV induces consumptive coagulopathy and thrombocytopenia in xenotransplant recipients. Initial studies in baboons revealed that the virus triggers increased release of tumor necrosis factor α (TNFα) and interleukin 6 (IL-6), along with elevated levels of tissue plasminogen activator (tPA) and plasminogen activator inhibitor 1 (PAI-1) complexes. This suggests a complete loss of endothelial cells' pro-fibrinolytic function. Although there is no evidence that PCMV/PRV infects primate cells, including human cells, the virus appears to directly interact with immune and endothelial cells, disrupting cytokine signaling and coagulation pathways. Since it affects only xenotransplant recipients and not healthy humans, this condition should be classified as a xenozoonosis. Interestingly, antibodies against human herpesvirus 6 (HHV-6) cross-react with PCMV/PRV and may contribute to protection against infection in humans. Further research is needed to uncover the molecular mechanisms underlying this xenozoonotic disease.
Keywords:
1. PCMV/PRV—molecular biology
2. Pathogenesis in the pig
3. Pathogenesis in non-human primate xenotransplant recipients
4. Transmission to a human pig heart recipient
5. Molecular insights into PCMV/PRV-induced xenozoonosis
5. PCMV/PRV and HHV-6
6. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BaCMV | baboon cytomegalovirus |
| CXCL-9 | chemokine (C-X-C motif) ligand 9 |
| HCMV | human cytomegalovirus |
| HHV6, 7 | human herpesvirus 6, 7 |
| HUVEC | human umbilical vein endothelial cells |
| ICAM-1 | intercellular adhesion molecule-1 |
| IFNγ | interferon γ |
| IL-1α, -1β, -4, -5, -6, -7, -8, -12B, -15 | interleukin-1α, -1β, -6, -7, - 8, -12B, 15 |
| ITCV | International Committee on Taxonomy of Viruses |
| IVIG | intravenous immunoglobulin |
| MCP-1, -2 | monocyte chemoattractant protein-1, -2 |
| MDMs | monocyte derived macrophages |
| MIP-1α, -3 | macrophage inflammatory protein-1α, -3 |
| MHC | major histocompatibility complex |
| miRNA | micro RNA |
| MIG | monokine induced by gamma interferon |
| PAEC | porcine aortic endothelial cells |
| PAI-1 | plasminogen activator inhibitor 1 |
| PCMV/PRV | porcine cytomegalovirus/porcine roseolovirus |
| PTEC | proximal tubular epithelial cells |
| SuBHV2 | suid betaherpesvirus 2 |
| TCR | T cell receptor |
| TGF-β1 | transforming growth factor β1 |
| TF | porcine tissue factor |
| TNFα | tumor necrosis factor α |
| tPA | tissue plasminogen activator |
References
- Denner, J.; Bigley, T.M.; Phan, T.L.; Zimmermann, C.; Zhou, X.; Kaufer, B.B. Comparative Analysis of Roseoloviruses in Humans, Pigs, Mice, and Other Species. Viruses. 2019, 11, 1108. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Zeng, N.; Zhou, L.; Ge, X.; Guo, X.; Yang, H. Genomic organization and molecular characterization of porcine cytomegalovirus. Virology. 2014, 460-461, 165–172. [Google Scholar] [CrossRef] [PubMed]
- https://ictv.global/report/chapter/orthoherpesviridae/orthoherpesviridae/roseolovirus (accessed on 6 February 2025).
- Edington, N.; Plowright, W.; Watt, R.G. Generalized porcine cytomegalic inclusion disease: Distribution of cytomegalic cells and virus. J Comp Pathol. 1976, 86, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Jhelum, H.; Kaufer, B.; Denner, J. Application of Methods Detecting Xenotransplantation-Relevant Viruses for Screening German Slaughterhouse Pigs. Viruses. 2024, 16, 1119. [Google Scholar] [CrossRef]
- Denner, J. Reduction of the survival time of pig xenotransplants by porcine cytomegalovirus. Virol J. 2018, 15, 171. [Google Scholar] [CrossRef]
- Michaels, M.G.; Alcendor, D.J.; St George, K.; Rinaldo, C.R., Jr.; Ehrlich, G.D.; Becich, M.J.; Hayward, G.S. Distinguishing baboon cytomegalovirus from human cytomegalovirus: Importance for xenotransplantation. J Infect Dis. 1997, 176, 1476–1483. [Google Scholar] [CrossRef]
- Michaels, M.G.; Jenkins, F.J.; St George, K.; Nalesnik, M.A.; Starzl, T.E.; Rinaldo, C.R., Jr. Detection of infectious baboon cytomegalovirus after baboon-to-human liver xenotransplantation. J Virol. 2001, 75, 2825–2828. [Google Scholar] [CrossRef]
- Degré, M.; Ranneberg-Nilsen, T.; Beck, S.; Rollag, H.; Fiane, A.E. Human cytomegalovirus productively infects porcine endothelial cells in vitro. Transplantation. 2001, 72, 1334–1337. [Google Scholar] [CrossRef]
- Whitteker, J.L.; Dudani, A.K.; Tackaberry, E.S. Human fibroblasts are permissive for porcine cytomegalovirus in vitro. Transplantation. 2008, 86, 155–162. [Google Scholar] [CrossRef]
- Tucker, A.W.; Galbraith, D.; McEwan, P.; Onions, D. Evaluation of porcine cytomegalovirus as a potential zoonotic agent in xenotransplantation. Transplant Proc. 1999, 31, 915. [Google Scholar] [CrossRef]
- Plowright, W.; Edington, N.; Watt, R.G. The behaviour of porcine cytomegalovirus in commercial pig herds. J Hyg (Lond). 1976, 76, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liao, S.; Zhu, L.; Xu, Z.; Zhou, Y. Molecular epidemiology of porcine Cytomegalovirus (PCMV) in Sichuan Province, China: 2010-2012. PLoS ONE. 2013, 8, e64648. [Google Scholar] [CrossRef] [PubMed]
- Edington, N.; Wrathall, A.; Done, J. Porcine cytomegalovirus (PCMV) in early gestation. Vet microbiol 1988, 17, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, Z.; Zhu, L.; Liao, S.; Guo, W. Transcriptome analysis of porcine thymus following porcine cytomegalovirus infection. PLoS ONE. 2014, 9, e113921. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Zhang, Y.; Peng, L.; Ma, J.; et al. The role of the CD95, CD38 and TGF-b 1 during active human cytomegalovirus infection in liver transplantation. Cytokine 2006, 35, 193–199. [Google Scholar] [CrossRef]
- Liu, X.; Liao, S.; Xu, Z.; Zhu, L.; Yang, F.; Guo, W. Identification and Analysis of the Porcine MicroRNA in Porcine Cytomegalovirus-Infected Macrophages Using Deep Sequencing. PLoS ONE. 2016, 11, e0150971. [Google Scholar] [CrossRef]
- Liu, X.; Wei, H.; Liao, S.; Ye, J.; Zhu, L.; Xu, Z. MicroRNA transcriptome analysis of porcine vital organ responses to immunosuppressive porcine cytomegalovirus infection. Virol J. 2018, 15, 16. [Google Scholar] [CrossRef]
- Kavanová, L.; Moutelíková, R.; Prodělalová, J.; Faldyna, M.; Toman, M.; Salát, J. Monocyte derived macrophages as an appropriate model for porcine cytomegalovirus immunobiology studies. Vet Immunol Immunopathol. 2018, 197, 58–62. [Google Scholar] [CrossRef]
- Gollackner, B.; Mueller, N.J.; Houser, S.; Qawi, I.; Soizic, D.; Knosalla, C.; Buhler, L.; Dor, F.J.; Awwad, M.; Sachs, D.H.; Cooper, D.K.; Robson, S.C.; Fishman, J.A. Porcine cytomegalovirus and coagulopathy in pig-to-primate xenotransplantation. Transplantation. 2003, 75, 1841–1847. [Google Scholar] [CrossRef]
- Mueller, N.J.; Barth, R.N.; Yamamoto, S.; Kitamura, H.; Patience, C.; Yamada, K.; Cooper, D.K.; Sachs, D.H.; Kaur, A.; Fishman, J.A. Activation of cytomegalovirus in pig-to-primate organ xenotransplantation. J Virol. 2002, 76, 4734–440. [Google Scholar] [CrossRef]
- Längin, M.; Mayr, T.; Reichart, B.; Michel, S.; Buchholz, S.; Guethoff, S.; Dashkevich, A.; Baehr, A.; Egerer, S.; Bauer, A.; Mihalj, M.; Panelli, A.; Issl, L.; Ying, J.; Fresch, A.K.; Buttgereit, I.; Mokelke, M.; Radan, J.; Werner, F.; Lutzmann, I.; Steen, S.; Sjöberg, T.; Paskevicius, A.; Qiuming, L.; Sfriso, R.; Rieben, R.; Dahlhoff, M.; Kessler, B.; Kemter, E.; Kurome, M.; Zakhartchenko, V.; Klett, K.; Hinkel, R.; Kupatt, C.; Falkenau, A.; Reu, S.; Ellgass, R.; Herzog, R.; Binder, U.; Wich, G.; Skerra, A.; Ayares, D.; Kind, A.; Schönmann, U.; Kaup, F.J.; Hagl, C.; Wolf, E.; Klymiuk, N.; Brenner, P.; Abicht, J.M. Consistent success in life-supporting porcine cardiac xenotransplantation. Nature. 2018, 564, 430–433. [Google Scholar] [CrossRef] [PubMed]
- Denner, J.; Längin, M.; Reichart, B.; Krüger, L.; Fiebig, U.; Mokelke, M.; Radan, J.; Mayr, T.; Milusev, A.; Luther, F.; Sorvillo, N.; Rieben, R.; Brenner, P.; Walz, C.; Wolf, E.; Roshani, B.; Stahl-Hennig, C.; Abicht, J.M. Impact of porcine cytomegalovirus on long-term orthotopic cardiac xenotransplant survival. Sci Rep. 2020, 10, 17531. [Google Scholar] [CrossRef]
- Morozov, V.A.; Abicht, J.M.; Reichart, B.; Mayr, T.; Guethoff, S.; Denner, J. Active replication of porcine cytomegalovirus (PCMV) following transplantation of a pig heart into a baboon despite undetected virus in the donor pig. Ann Virol Res. 2016, 2, 1018. [Google Scholar]
- Fiebig, U.; Abicht, J.M.; Mayr, T.; Längin, M.; Bähr, A.; Guethoff, S.; Falkenau, A.; Wolf, E.; Reichart, B.; Shibahara, T.; Denner, J. Distribution of Porcine Cytomegalovirus in Infected Donor Pigs and in Baboon Recipients of Pig Heart Transplantation. Viruses. 2018, 10, 66. [Google Scholar] [CrossRef] [PubMed]
- Griffith, B.P.; Goerlich, C.E.; Singh, A.K.; Rothblatt, M.; Lau, C.L.; Shah, A.; Lorber, M.; Grazioli, A.; Saharia, K.K.; Hong, S.N.; Joseph, S.M.; Ayares, D.; Mohiuddin, M.M. Genetically Modified Porcine-to-Human Cardiac Xenotransplantation. N Engl J Med. 2022, 387, 35–44. [Google Scholar] [CrossRef]
- Mohiuddin, M.M.; Singh, A.K.; Scobie, L.; Goerlich, C.E.; Grazioli, A.; Saharia, K.; Crossan, C.; Burke, A.; Drachenberg, C.; Oguz, C.; Zhang, T.; Lewis, B.; Hershfeld, A.; Sentz, F.; Tatarov, I.; Mudd, S.; Braileanu, G.; Rice, K.; Paolini, J.F.; Bondensgaard, K.; Vaught, T.; Kuravi, K.; Sorrells, L.; Dandro, A.; Ayares, D.; Lau, C.; Griffith, B.P. Graft dysfunction in compassionate use of genetically engineered pig-to-human cardiac xenotransplantation: A case report. Lancet. 2023, 402, 397–410. [Google Scholar] [CrossRef]
- Denner, J. First transplantation of a pig heart from a multiple gene-modified donor porcine cytomegalovirus/roseolovirus, and antiviral drugs. Xenotransplantation 2023, 30, e12800. [Google Scholar] [CrossRef]
- Yamada, K.; Tasaki, M.; Sekijima, M.; Wilkinson, R.A.; Villani, V.; Moran, S.G.; Cormack, T.A.; Hanekamp, I.M.; Hawley, R.J.; Arn, J.S.; Fishman, J.A.; Shimizu, A.; Sachs, D.H. Porcine cytomegalovirus infection is associated with early rejection of kidney grafts in a pig to baboon xenotransplantation model. Transplantation. 2014, 98, 411–418. [Google Scholar] [CrossRef]
- Sedmak, D.D.; Knight, D.A.; Vook, N.C.; et al. Divergent patterns of ELAM-1, ICAM-1, and VCAM-1 expression on cytomegalovirus-infected endothelial cells. Transplantation 1994, 58, 1379. [Google Scholar]
- van Dorp, W.T.; van Wieringen, P.A.; Marselis-Jonges, E.; et al. Cytomegalovirus directly enhances MHC class I and intercellular adhesion molecule-1 expression on cultured proximal tubular epithelial cells. Transplantation 1993, 55, 1367. [Google Scholar] [CrossRef]
- Pugliese, A.; Vidotto, V.; Beltramo, T.; Petrini, S.; Torre, D. A review of HIV-1 Tat protein biological effects. Cell Biochem Funct. 2005, 23, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Romani, B.; Engelbrecht, S.; Glashoff, R.H. Functions of Tat: The versatile protein of human immunodeficiency virus type 1. J Gen Virol. 2010, 91, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Denner, J. The transmembrane proteins contribute to immunodeficiencies induced by HIV-1 and other retroviruses. AIDS. 2014, 28, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Denner, J.; Eschricht, M.; Lauck, M.; Semaan, M.; Schlaermann, P.; Ryu, H.; Akyüz, L. Modulation of cytokine release and gene expression by the immunosuppressive domain of gp41 of HIV-1. PLoS ONE. 2013, 8, e55199. [Google Scholar] [CrossRef]
- Schlecht-Louf, G.; Renard, M.; Mangeney, M.; Letzelter, C.; Richaud, A.; Ducos, B.; Bouallaga, I.; Heidmann, T. Retroviral infection in vivo requires an immune escape virulence factor encrypted in the envelope protein of oncoretroviruses. Proc Natl Acad Sci U S A. 2010, 107, 3782–3787. [Google Scholar] [CrossRef]
- Denner, J. Zoonosis and xenozoonosis in xenotransplantation: A proposal for a new classification. Zoonoses Public Health. 2023, 70, 578–579. [Google Scholar] [CrossRef]
- Fiebig, U.; Holzer, A.; Ivanusic, D.; Plotzki, E.; Hengel, H.; Neipel, F.; Denner, J. Antibody Cross-Reactivity between Porcine Cytomegalovirus (PCMV) and Human Herpesvirus-6 (HHV-6). Viruses. 2017, 9, 317. [Google Scholar] [CrossRef]
- Looney, R.J.; Huggins, J. Use of intravenous immunoglobulin G (IVIG). Best Pract Res Clin Haematol. 2006, 19, 3–25. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



