Submitted:
11 February 2025
Posted:
12 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
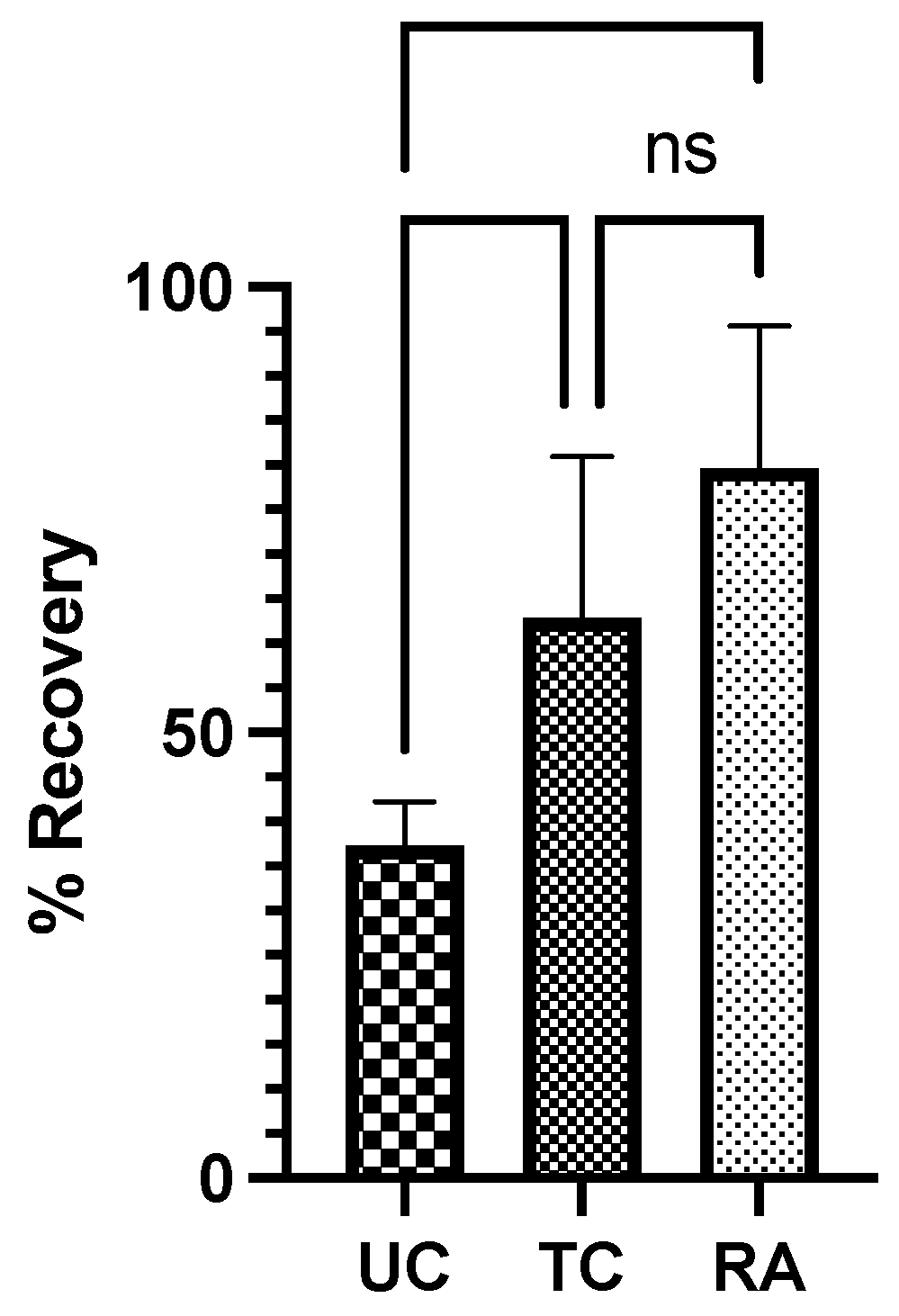
2.1. Functional Outcome Measurements
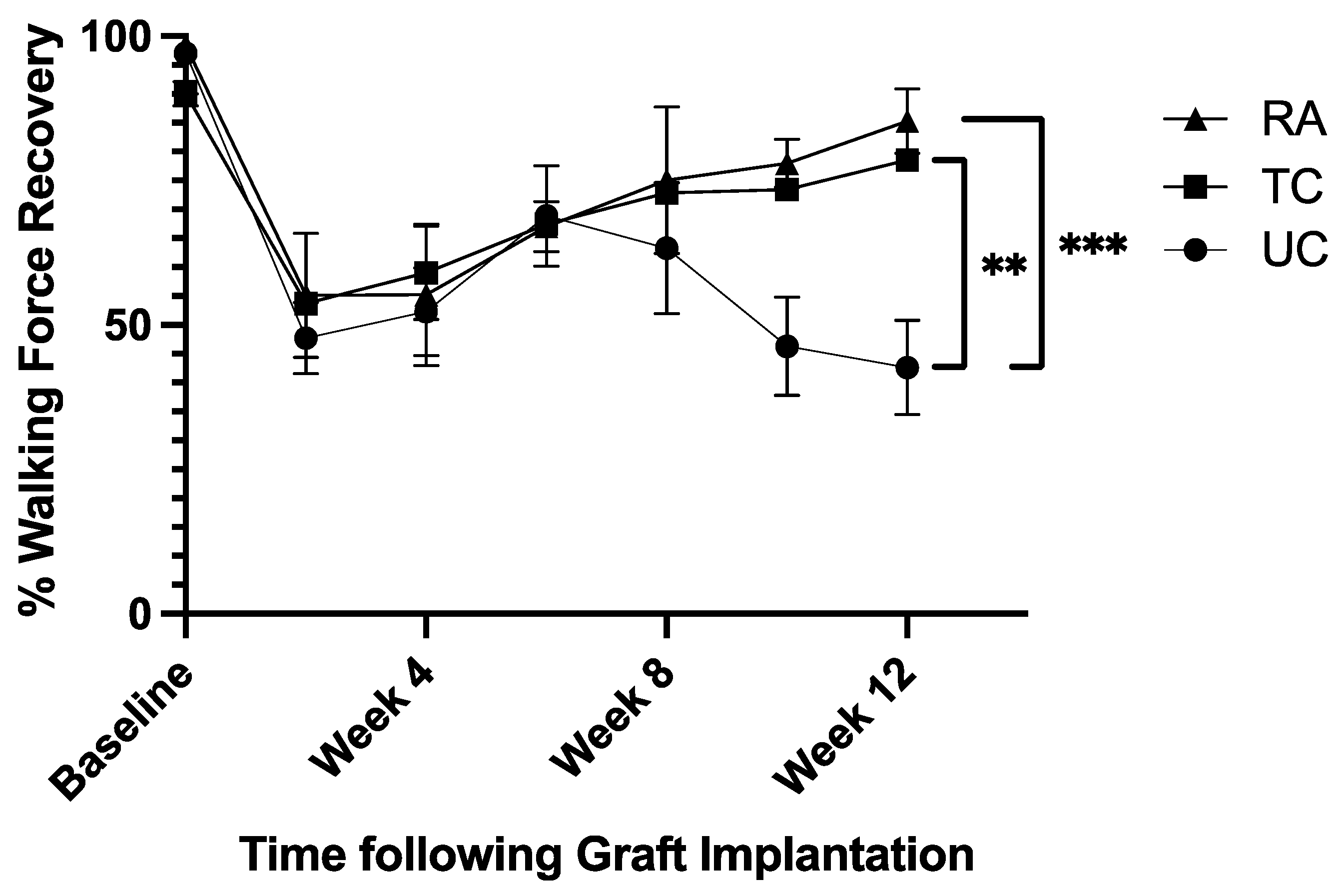
2.2. Walking Track Force Analysis
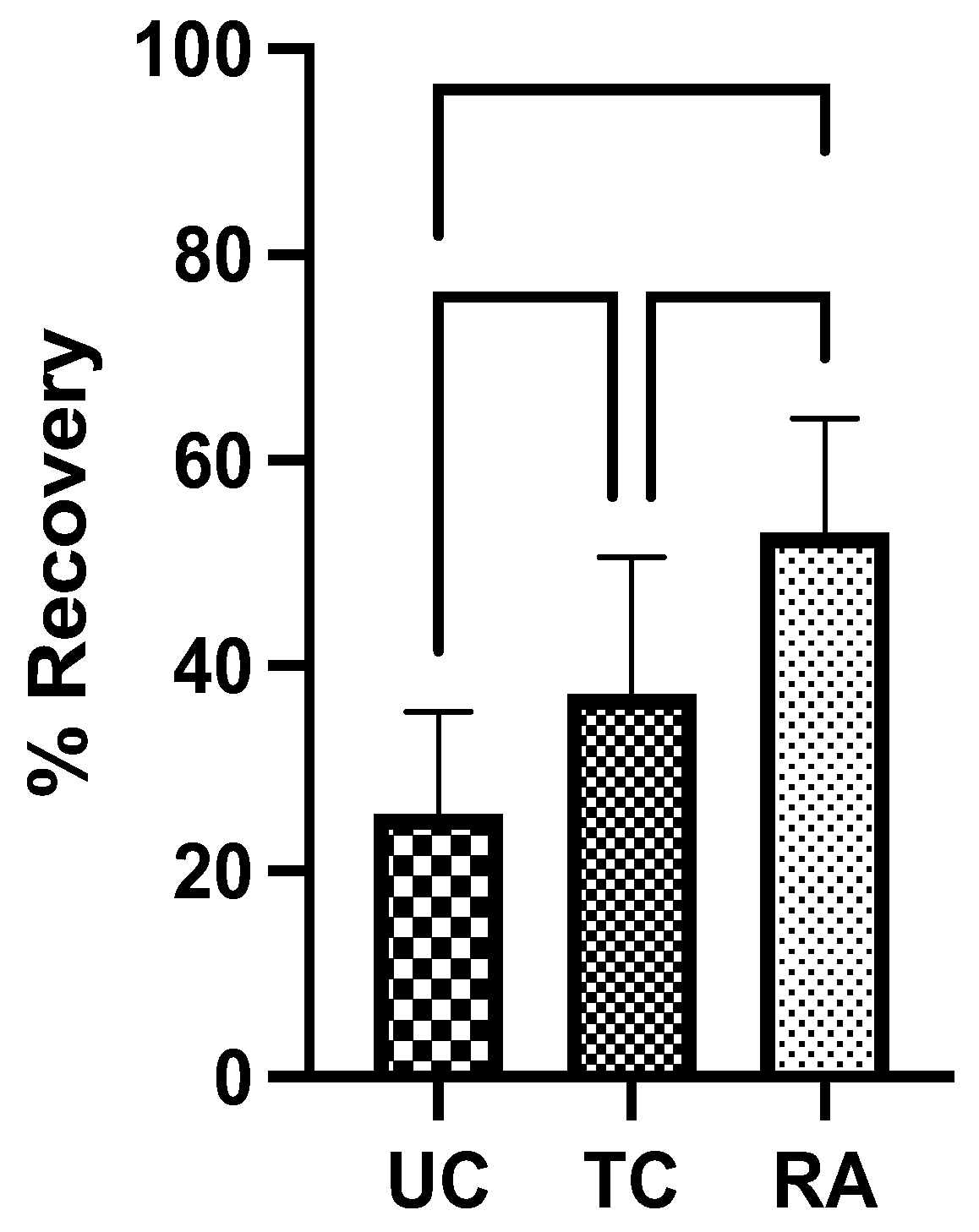
2.3. Compound Muscle Action Potential (CMAP)
2.4. Tibialis Anterior and Gastrocnemius Muscle Weights
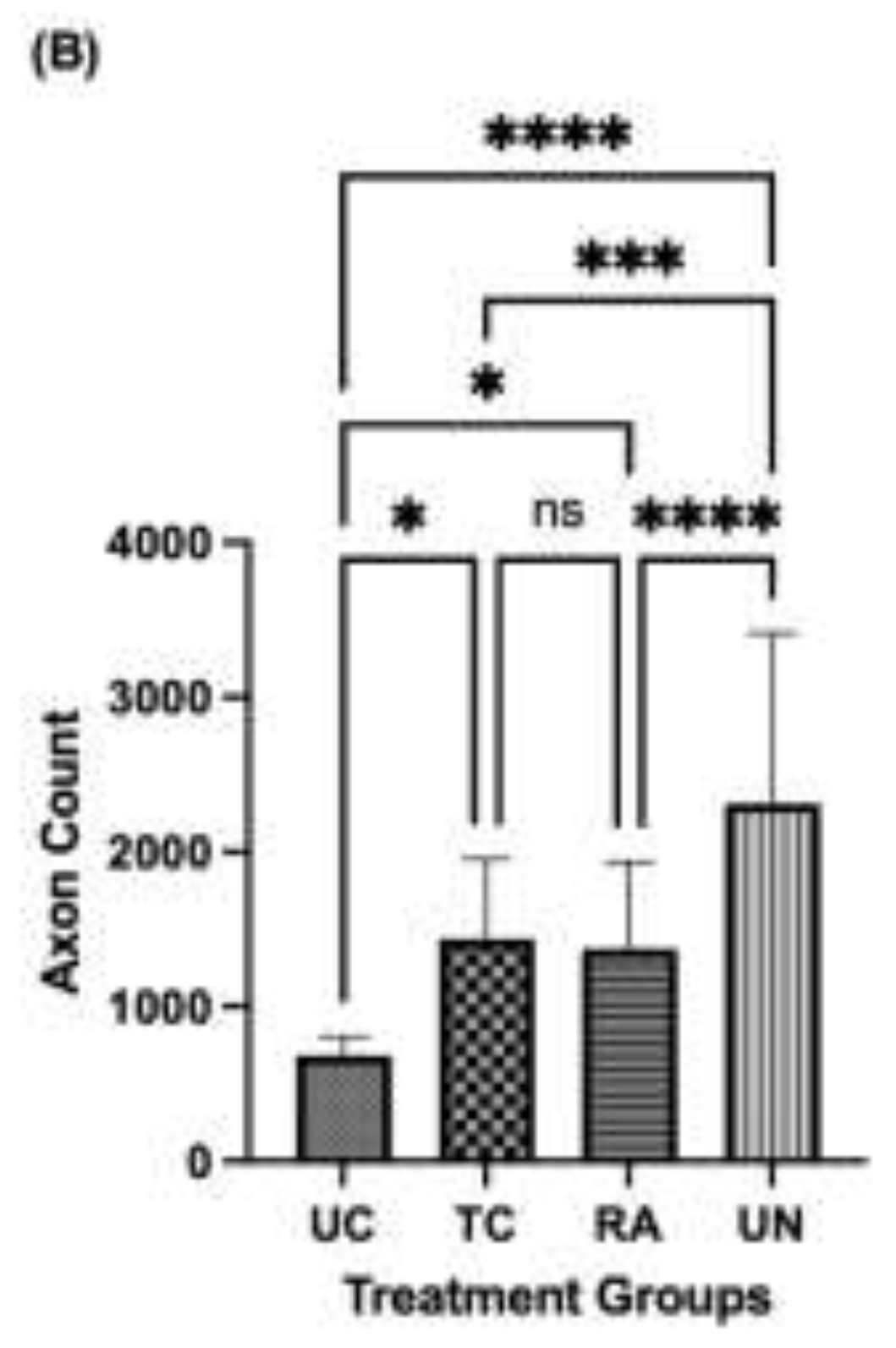
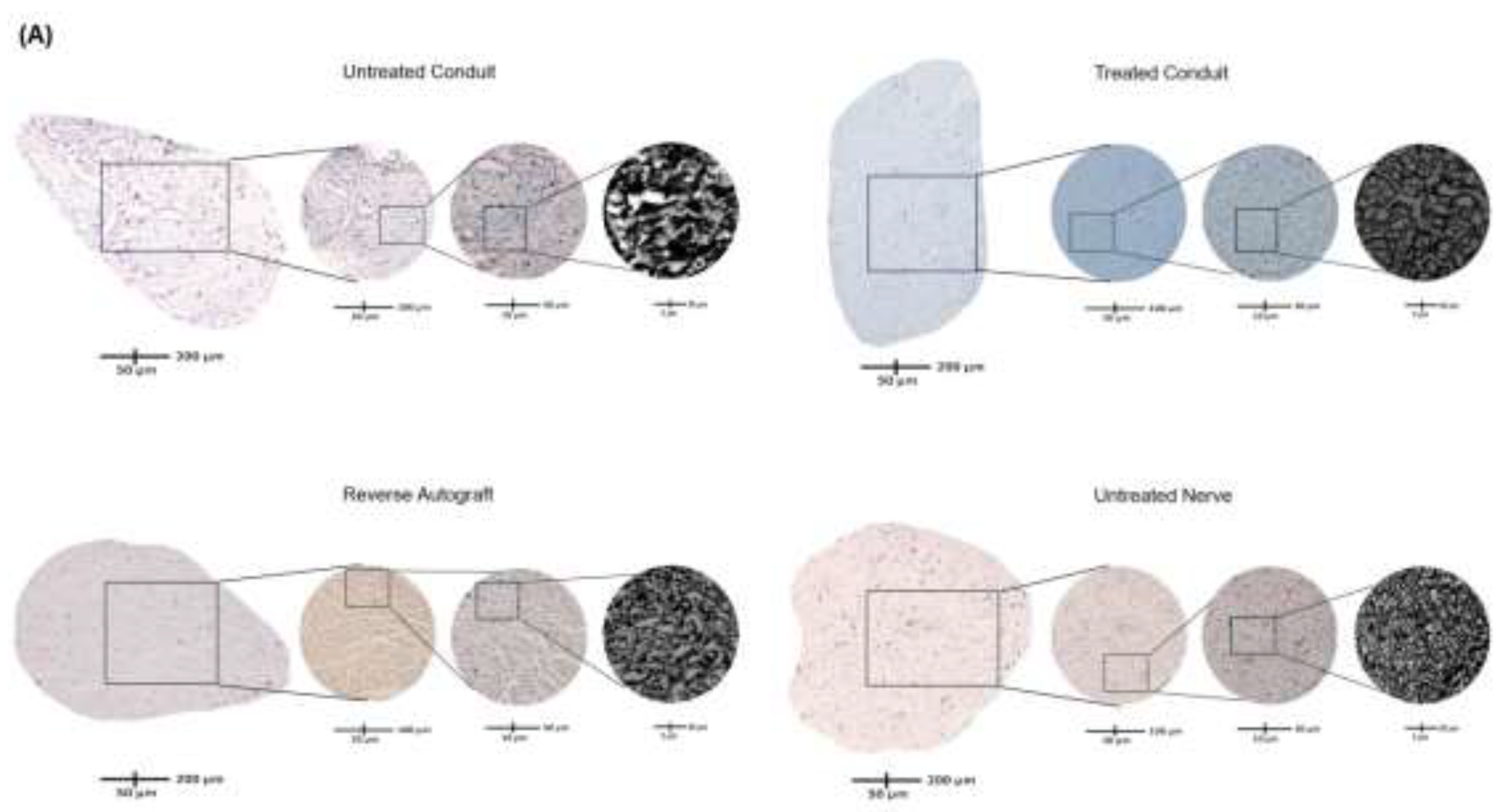
2.5. Axon Analysis
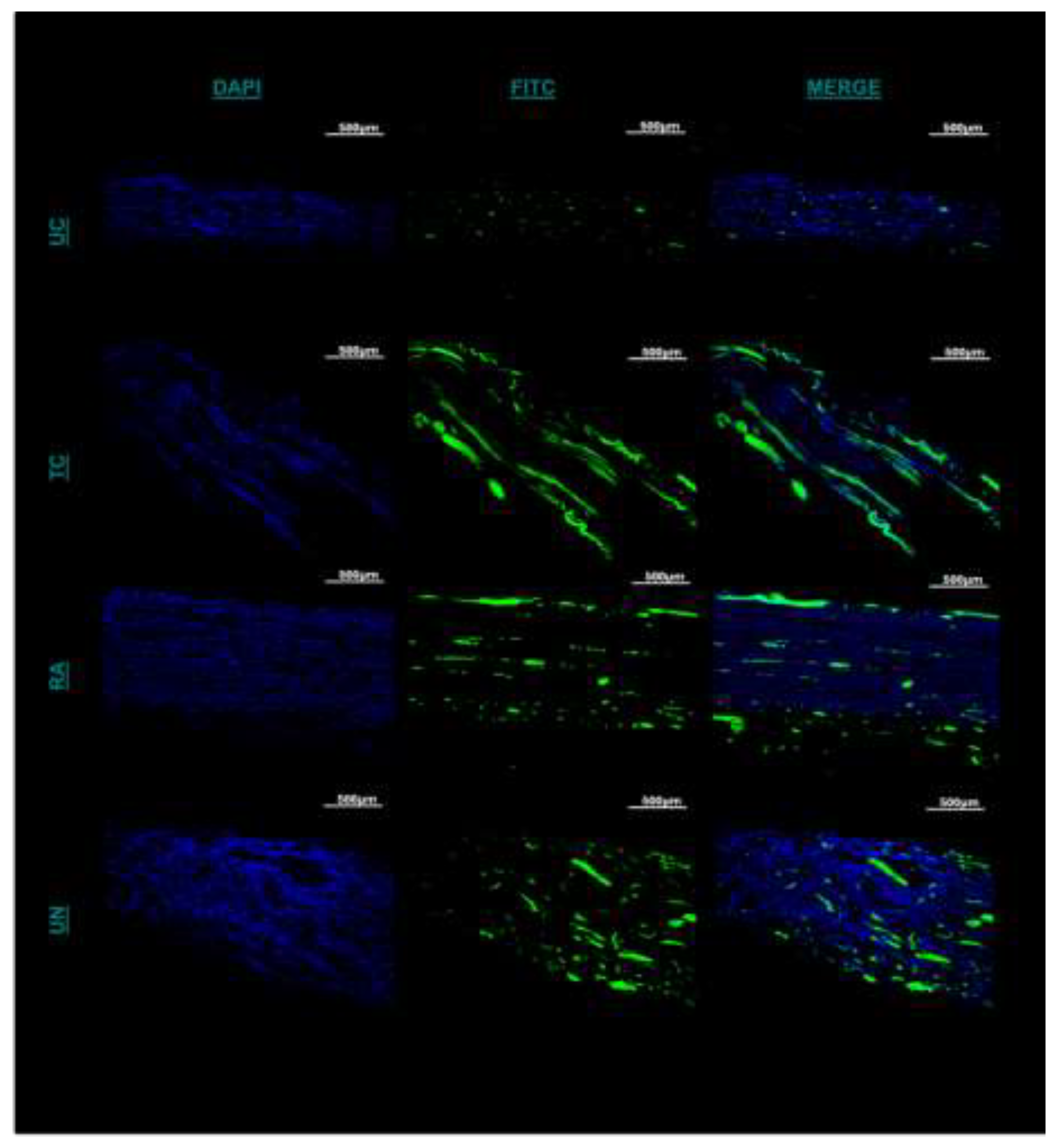
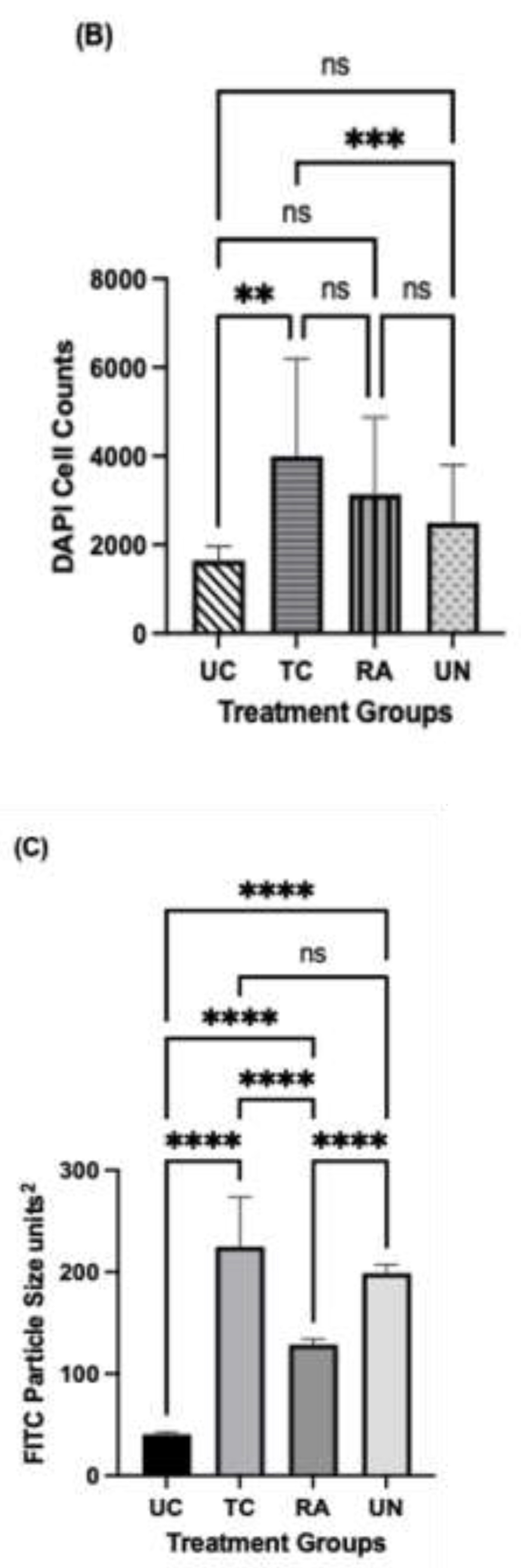
2.6. Schwann Cell Immunofluorescence
3. Discussion
3.1. Importance of Microarchitecture
3.2. Benefits of Matrilin-2
3.3. Benefits of Chitosan
3.4. Functional Outcomes
3.5. Axonal Regeneration and Schwann Cell Migration
4. Materials and Methods
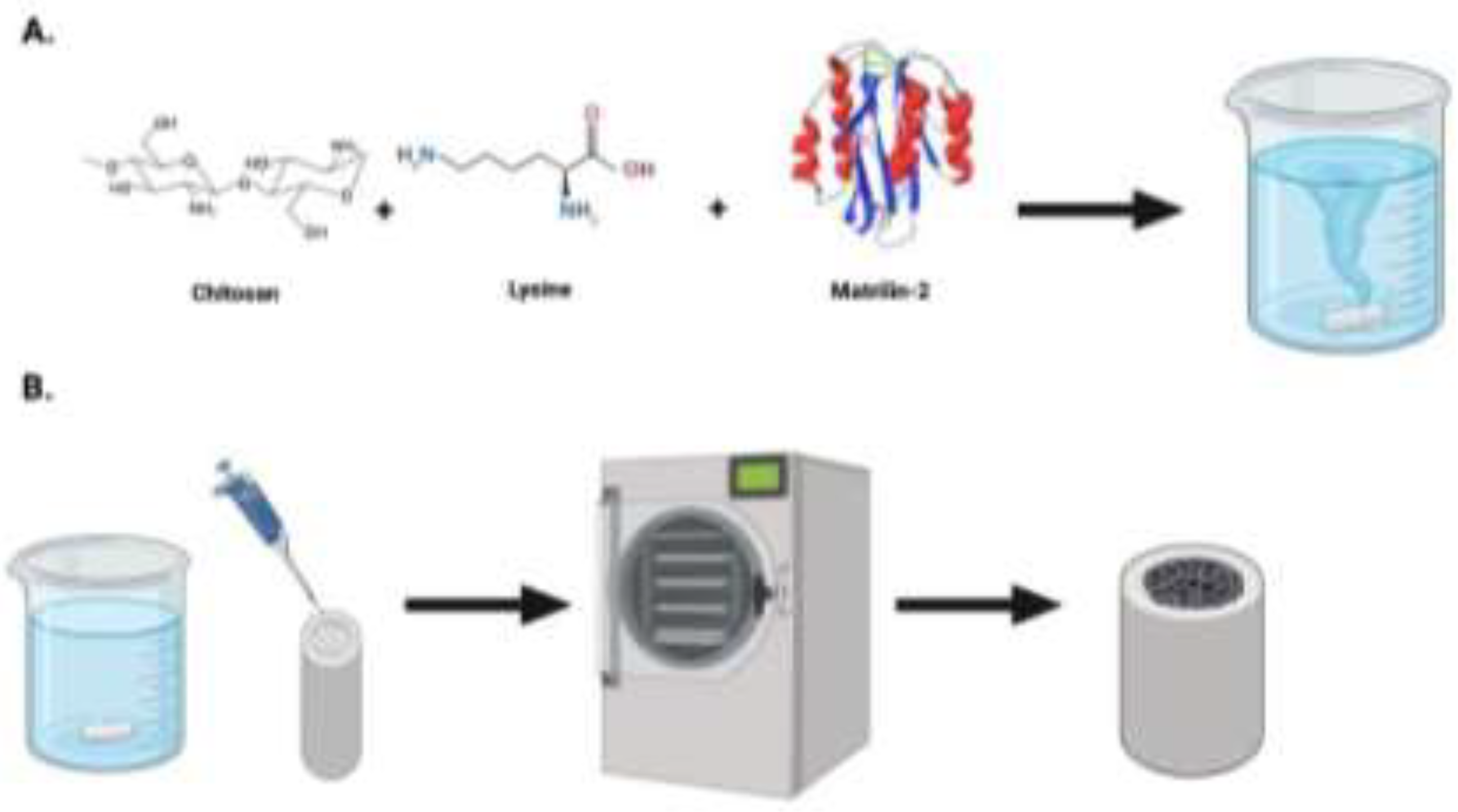
4.1. Preparation of Matrillin-2
4.2. Animals
4.3. Gait Analysis
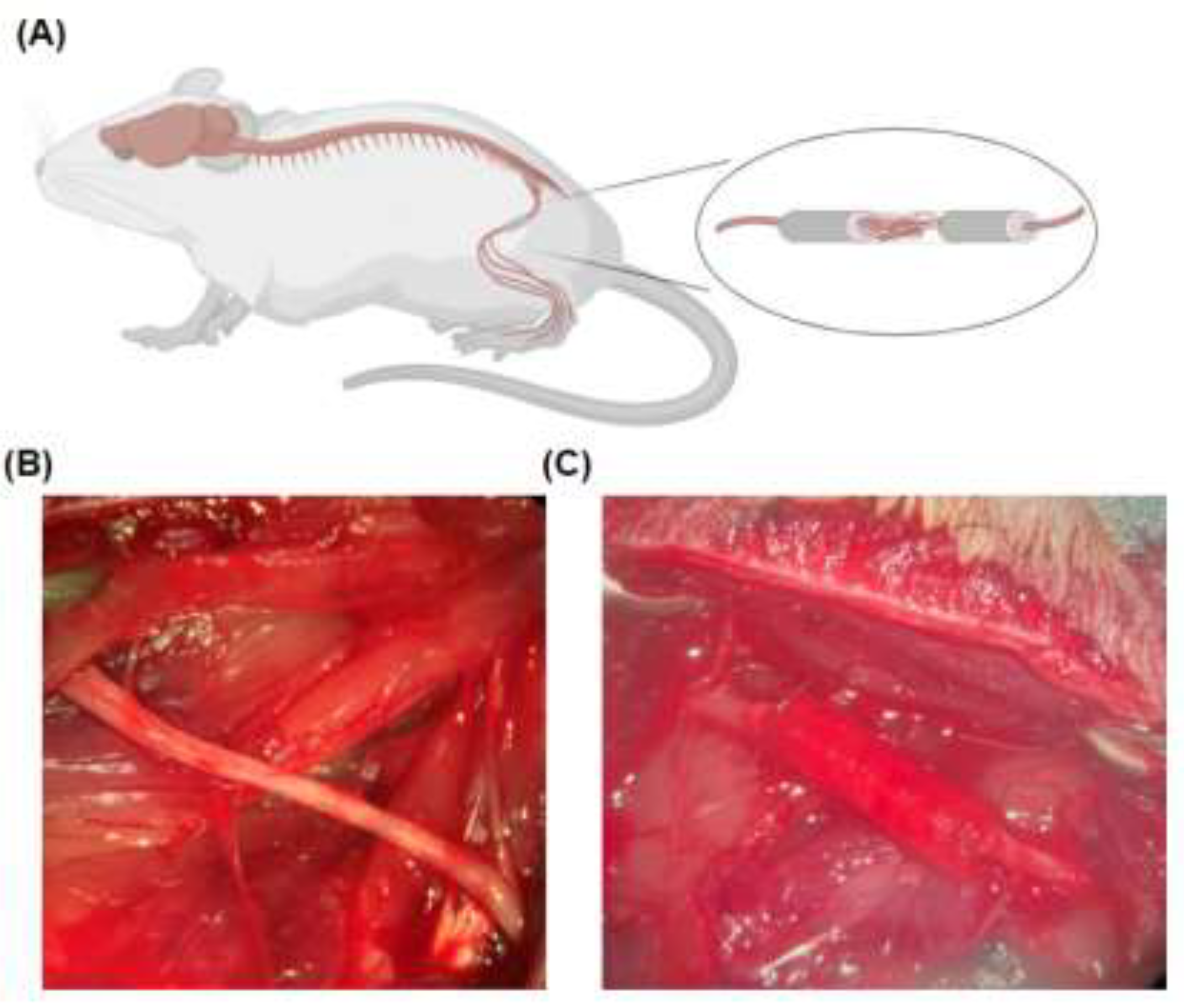
4.4. Sciatic Nerve Defect and Reconstruction Model
4.5. Compound Muscle Action Potential Testing
4.6. Nerve and Muscle Extraction
4.7. Axon Analysis
4.8. Schwann Cell Immunofluorescence
5. Conclusions
Funding
Author contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of interest
Abbreviations
| UC | Untreated Conduit |
| TC | Treated Conduit |
| RA | Reverse Autograft |
| UN | Uninjured Nerve |
| SCs | Schwann cells |
| CMAP | Compound Muscle Action Potential |
| MATN2 | Matrilin-2 |
References
- Li, N.Y.; Onor, G.I.; Lemme, N.J.; Gil, J.A. Epidemiology of Peripheral Nerve Injuries in Sports, Exercise, and Recreation in the United States, 2009 – 2018. Physician Sportsmed. 2021, 49, 355–362. [Google Scholar] [CrossRef]
- NINDS. Peripheral Neuropathy Fact Sheet. National Institute of Neurological Disorders and Stroke.
- Taylor, C.A.; Braza, D.; Rice, J.B.; Dillingham, T. The Incidence of Peripheral Nerve Injury in Extremity Trauma. Am. J. Phys. Med. Rehabilitation 2008, 87, 381–385. [Google Scholar] [CrossRef]
- Grinsell, D.; Keating, C.P. Peripheral Nerve Reconstruction after Injury: A Review of Clinical and Experimental Therapies. BioMed Res. Int. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Ruijs, A.C.J.; Jaquet, J.-B.; Kalmijn, S.; Giele, H.; Hovius, S.E.R. Median and Ulnar Nerve Injuries: A Meta-Analysis of Predictors of Motor and Sensory Recovery after Modern Microsurgical Nerve Repair. Plast. Reconstr. Surg. 2005, 116, 484–494. [Google Scholar] [CrossRef]
- Moore, A.M.; Kasukurthi, R.; Magill, C.K.; Farhadi, H.F.; Borschel, G.H.; Mackinnon, S.E. Limitations of Conduits in Peripheral Nerve Repairs. HAND 2009, 4, 180–186. [Google Scholar] [CrossRef]
- Whitlock, E.L.; Tuffaha, S.H.; Luciano, J.P.; Yan, Y.; Hunter, D.A.; Magill, C.K.; Moore, A.M.; Tong, A.Y.; Mackinnon, S.E.; Borschel, G.H. Processed allografts and type I collagen conduits for repair of peripheral nerve gaps. Muscle Nerve 2009, 39, 787–799. [Google Scholar] [CrossRef]
- Tang, P.; Whiteman, D.R.; Voigt, C.; Miller, M.C.; Kim, H. No Difference in Outcomes Detected Between Decellular Nerve Allograft and Cable Autograft in Rat Sciatic Nerve Defects. J. Bone Jt. Surg. 2019, 101, e42. [Google Scholar] [CrossRef]
- Piecha, D.; Wiberg, C.; Mörgelin, M.; Reinhardt, D.P.; Deák, F.; Maurer, P.; Paulsson, M. Matrilin-2 interacts with itself and with other extracellular matrix proteins. Biochem. J. 2002, 367, 715–721. [Google Scholar] [CrossRef]
- Kiss, I.; Évakorpos; Deák, F. Matrilin-2, an extracellular adaptor protein, is needed for the regeneration of muscle, nerve and other tissues. Neural Regen. Res. 2015, 10, 866–9. [Google Scholar] [CrossRef]
- Malin, D.; Sonnenberg-Riethmacher, E.; Guseva, D.; Wagener, R.; Aszódi, A.; Irintchev, A.; Riethmacher, D. The extracellular-matrix protein matrilin 2 participates in peripheral nerve regeneration. J. Cell Sci. 2009, 122, 995–1004. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Gong, J.; Yang, L.; Niu, C.; Ni, X.; Wang, Y.; Peng, S.; Gu, X.; Sun, C.; et al. Chitosan degradation products facilitate peripheral nerve regeneration by improving macrophage-constructed microenvironments. Biomaterials 2017, 134, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Qulub, F.; Widiyanti, P.; Maulida, H.N.; Indrio, L.W.; Wijayanti, T.R. EFFECT OF DEACETYLATION DEGREES VARIATION ON CHITOSAN NERVE CONDUIT FOR PERIPHERAL NERVE REGENERATION. Folia Medica Indones. 2017, 53, 101–107. [Google Scholar] [CrossRef]
- Neubrech, F.; Sauerbier, M.; Moll, W.; Seegmüller, J.; Heider, S.; Harhaus, L.; Bickert, B.; Kneser, U.; Kremer, T. Enhancing the Outcome of Traumatic Sensory Nerve Lesions of the Hand by Additional Use of a Chitosan Nerve Tube in Primary Nerve Repair: A Randomized Controlled Bicentric Trial. Plast. Reconstr. Surg. 2018, 142, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Li, N.Y.; Vorrius, B.; Ge, J.; Qiao, Z.; Zhu, S.; Katarincic, J.; Chen, Q. Matrilin-2 within a three-dimensional lysine-modified chitosan porous scaffold enhances Schwann cell migration and axonal outgrowth for peripheral nerve regeneration. Front. Bioeng. Biotechnol. 2023, 11. [Google Scholar] [CrossRef]
- Jessen, K.R.; Mirsky, R.; Lloyd, A.C. Schwann Cells: Development and Role in Nerve Repair. Cold Spring Harb. Perspect. Biol. 2015, 7, a020487. [Google Scholar] [CrossRef]
- Führmann, T.; Hillen, L.M.; Montzka, K.; Wöltje, M.; Brook, G.A. Cell–Cell interactions of human neural progenitor-derived astrocytes within a microstructured 3D-scaffold. Biomaterials 2010, 31, 7705–7715. [Google Scholar] [CrossRef]
- Gao, X.; Wang, Y.; Chen, J.; Peng, J. The role of peripheral nerve ECM components in the tissue engineering nerve construction. Prog. Neurobiol. 2013, 24, 443–453. [Google Scholar] [CrossRef]
- Charras, G.; Sahai, E. Physical influences of the extracellular environment on cell migration. Nat. Rev. Mol. Cell Biol. 2014, 15, 813–824. [Google Scholar] [CrossRef]
- Smith, D.H.; Burrell, J.C.; Browne, K.D.; Katiyar, K.S.; Ezra, M.I.; Dutton, J.L.; Morand, J.P.; Struzyna, L.A.; Laimo, F.A.; Chen, H.I.; et al. Tissue-engineered grafts exploit axon-facilitated axon regeneration and pathway protection to enable recovery after 5-cm nerve defects in pigs. Sci. Adv. 2022, 8, eabm3291. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, X.; Cai, M.; Zhao, W.; Ye, D.; Zhou, Y.; Zhu, C.; Zhang, X.; Lu, X.; Zhang, Z. A novel electrospun nerve conduit enhanced by carbon nanotubes for peripheral nerve regeneration. Nanotechnology 2014, 25, 165102. [Google Scholar] [CrossRef]
- Guenard, V.; Kleitman, N.; Morrissey, T.; Bunge, R.; Aebischer, P. Syngeneic Schwann cells derived from adult nerves seeded in semipermeable guidance channels enhance peripheral nerve regeneration. J. Neurosci. 1992, 12, 3310–3320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Peng, J.; Guo, Y.; Javidiparsijani, S.; Wang, G.; Wang, Y.; Liu, H.; Liu, J.; Luo, J. Matrilin-2 Is a Widely Distributed Extracellular Matrix Protein and a Potential Biomarker in the Early Stage of Osteoarthritis in Articular Cartilage. BioMed Res. Int. 2014, 2014, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.K.; Watson, M.A.; Lyman, M.; Perry, A.; Aldape, K.D.; Deák, F.; Gutmann, D.H. Matrilin-2 expression distinguishes clinically relevant subsets of pilocytic astrocytoma. Neurology 2006, 66, 127–130. [Google Scholar] [CrossRef]
- Piecha, D.; Muratoglu, S.; Mörgelin, M.; Hauser, N.; Studer, D.; Kiss, I.; Paulsson, M.; Deák, F. Matrilin-2, a Large, Oligomeric Matrix Protein, Is Expressed by a Great Variety of Cells and Forms Fibrillar Networks. J. Biol. Chem. 1999, 274, 13353–13361. [Google Scholar] [CrossRef] [PubMed]
- Pittier, R.; Sauthier, F.; Hubbell, J.A.; Hall, H. Neurite extension andin vitro myelination within three-dimensional modified fibrin matrices. J. Neurobiol. 2005, 63, 1–14. [Google Scholar] [CrossRef]
- Omidinia-Anarkoli, A.; Ephraim, J.W.; Rimal, R.; De Laporte, L. Hierarchical fibrous guiding cues at different scales influence linear neurite extension. Acta Biomater. 2020, 113, 350–359. [Google Scholar] [CrossRef]
- Gholap, A.D.; Rojekar, S.; Kapare, H.S.; Vishwakarma, N.; Raikwar, S.; Garkal, A.; Mehta, T.A.; Jadhav, H.; Prajapati, M.K.; Annapure, U. Chitosan scaffolds: Expanding horizons in biomedical applications. Carbohydr. Polym. 2023, 323, 121394. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Worley, S.D.; Broughton, R. The Chemistry and Applications of Antimicrobial Polymers: A State-of-the-Art Review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar] [CrossRef]
- P. , A.; K., V.; M., S.; T., G.; K., R.; P.N., S.; Sukumaran, A. Removal of toxic heavy metal lead (II) using chitosan oligosaccharide-graft-maleic anhydride/polyvinyl alcohol/silk fibroin composite. Int. J. Biol. Macromol. 2017, 104, 1469–1482. [Google Scholar] [CrossRef]
- Chang, F.; Wang, Y.; Liu, P.; Peng, J.; Han, G.-H.; Ding, X.; Wei, S.; Gao, G.; Huang, K. Role of macrophages in peripheral nerve injury and repair. Neural Regen. Res. 2019, 14, 1335–1342. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Ao, Q.; Gong, K.; Wang, A.; Zheng, L.; Gong, Y.; Zhang, X. The effect of topology of chitosan biomaterials on the differentiation and proliferation of neural stem cells. Acta Biomater. 2010, 6, 3630–3639. [Google Scholar] [CrossRef] [PubMed]
- Dietzmeyer, N.; Förthmann, M.; Leonhard, J.; Helmecke, O.; Brandenberger, C.; Freier, T.; Haastert-Talini, K. Two-Chambered Chitosan Nerve Guides With Increased Bendability Support Recovery of Skilled Forelimb Reaching Similar to Autologous Nerve Grafts in the Rat 10 mm Median Nerve Injury and Repair Model. Front. Cell. Neurosci. 2019, 13, 149. [Google Scholar] [CrossRef]
- Shrestha, S.; Shrestha, B.K.; Kim, J.I.; Ko, S.W.; Park, C.H.; Kim, C.S. Electrodeless coating polypyrrole on chitosan grafted polyurethane with functionalized multiwall carbon nanotubes electrospun scaffold for nerve tissue engineering. Carbon 2018, 136, 430–443. [Google Scholar] [CrossRef]
- Deng, P.; Chen, F.; Zhang, H.; Chen, Y.; Zhou, J. Multifunctional Double-Layer Composite Hydrogel Conduit Based on Chitosan for Peripheral Nerve Repairing. Adv. Heal. Mater. 2022, 11, e2200115. [Google Scholar] [CrossRef] [PubMed]
- Crosio, A.; Fornasari, B.E.; Gambarotta, G.; Geuna, S.; Raimondo, S.; Battiston, B.; Tos, P.; Ronchi, G. Chitosan tubes enriched with fresh skeletal muscle fibers for delayed repair of peripheral nerve defects. Neural Regen. Res. 2019, 14, 1079–1084. [Google Scholar] [CrossRef]
- Ao, Q.; Fung, C.-K.; Tsui, A.Y.-P.; Cai, S.; Zuo, H.-C.; Chan, Y.-S.; Shum, D.K.-Y. The regeneration of transected sciatic nerves of adult rats using chitosan nerve conduits seeded with bone marrow stromal cell-derived Schwann cells. Biomaterials 2011, 32, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Mi, R.; Hoke, A.; Chew, S.Y. Nanofibrous nerve conduit-enhanced peripheral nerve regeneration. J. Tissue Eng. Regen. Med. 2014, 8, 377–385. [Google Scholar] [CrossRef]
- Bian, Y.-Z.; Wang, Y.; Aibaidoula, G.; Chen, G.-Q.; Wu, Q. Evaluation of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) conduits for peripheral nerve regeneration. Biomaterials 2009, 30, 217–225. [Google Scholar] [CrossRef]
- Longo, M.V.L.; de Faria, J.C.M.; Isaac, C.; Nepomuceno, A.C.; Teixeira, N.H.; Gemperli, R. Comparisons of the results of peripheral nerve defect repair with fibrin conduit and autologous nerve graft: An experimental study in rats. Microsurgery 2015, 36, 59–65. [Google Scholar] [CrossRef]
- Kwon, S.W.; Choi, J.; Kim, J.H.; Jang, J.W.; Kim, H.J.; Choi, S.H. Decellularized sciatic nerve matrix as a biodegradable conduit for peripheral nerve regeneration. Neural Regen. Res. 2018, 13, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Langer, H.T.; Senden, J.M.G.; Gijsen, A.P.; Kempa, S.; van Loon, L.J.C.; Spuler, S. Muscle Atrophy Due to Nerve Damage Is Accompanied by Elevated Myofibrillar Protein Synthesis Rates. Front. Physiol. 2018, 9, 1220. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.-S.; Hwang, J.-Y.; Kim, M.S.; Lee, J.-Y.; Kim, J.-W.; Kim, H.-S.; Shin, U.S.; Knowles, J.C.; Kim, H.-W.; Hyun, J.K. Carbon-nanotube-interfaced glass fiber scaffold for regeneration of transected sciatic nerve. Acta Biomater. 2015, 13, 324–334. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



