Submitted:
04 February 2025
Posted:
06 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Paraneoplastic Syndromes That Pose Diagnostic Challenges
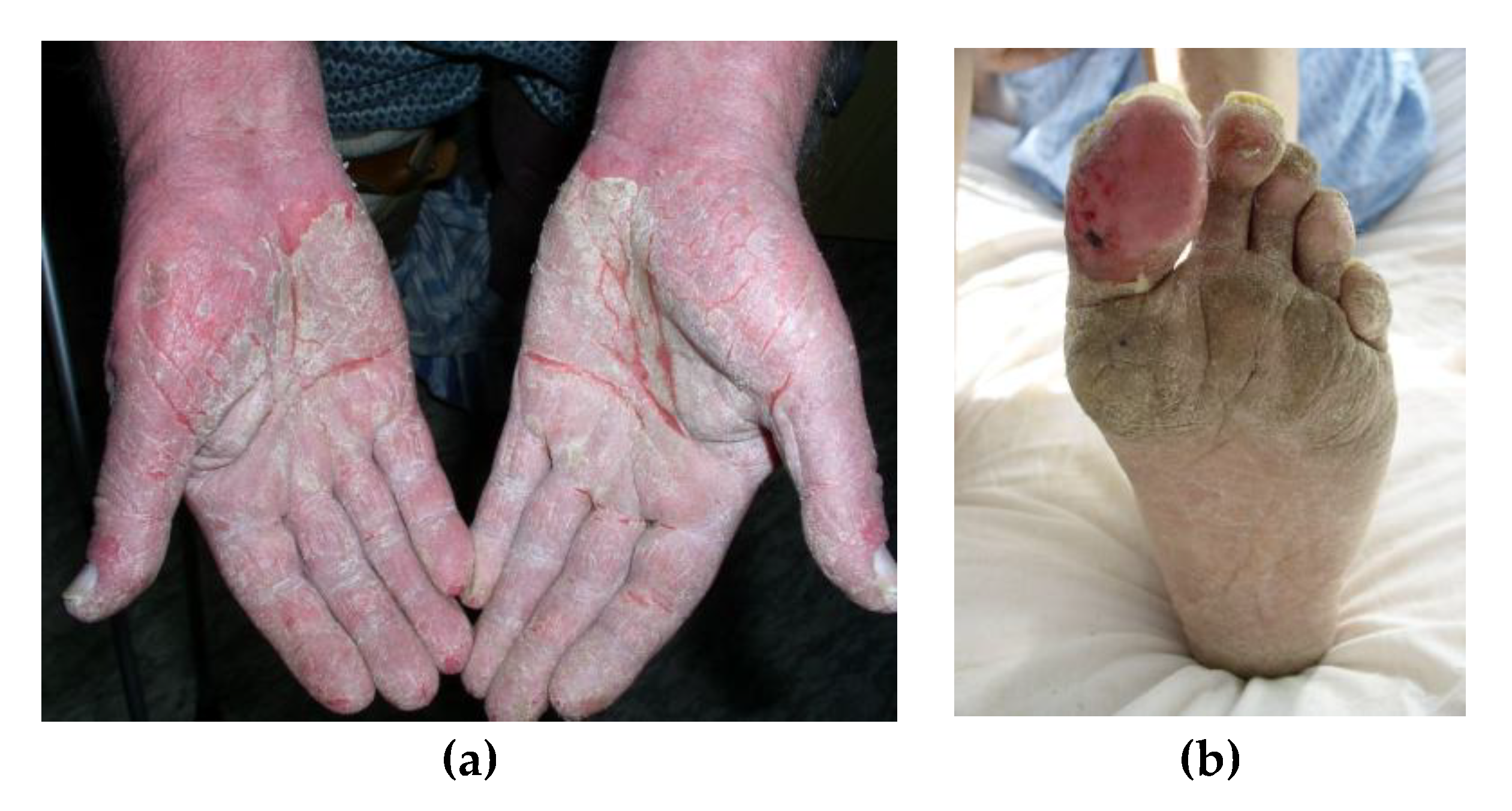
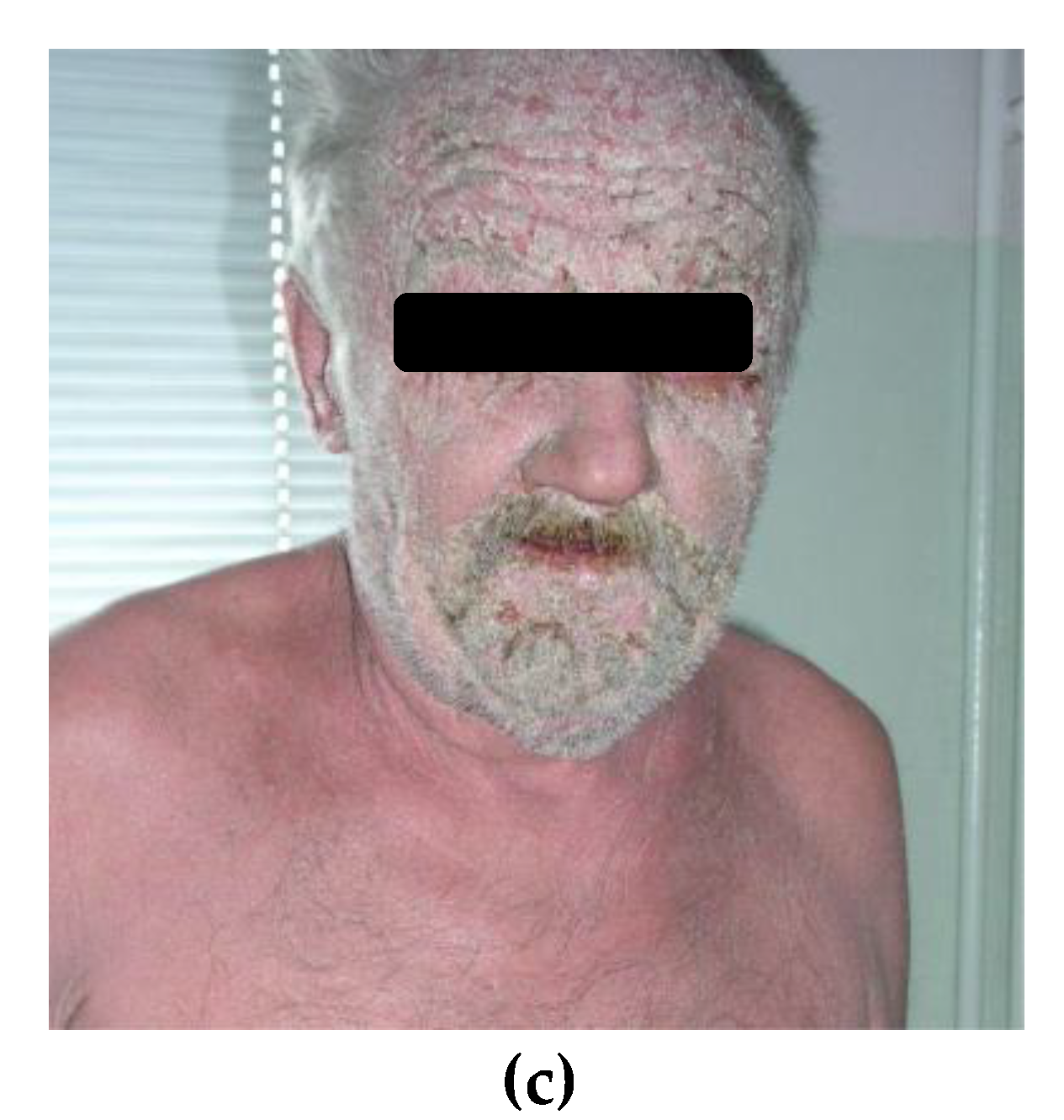
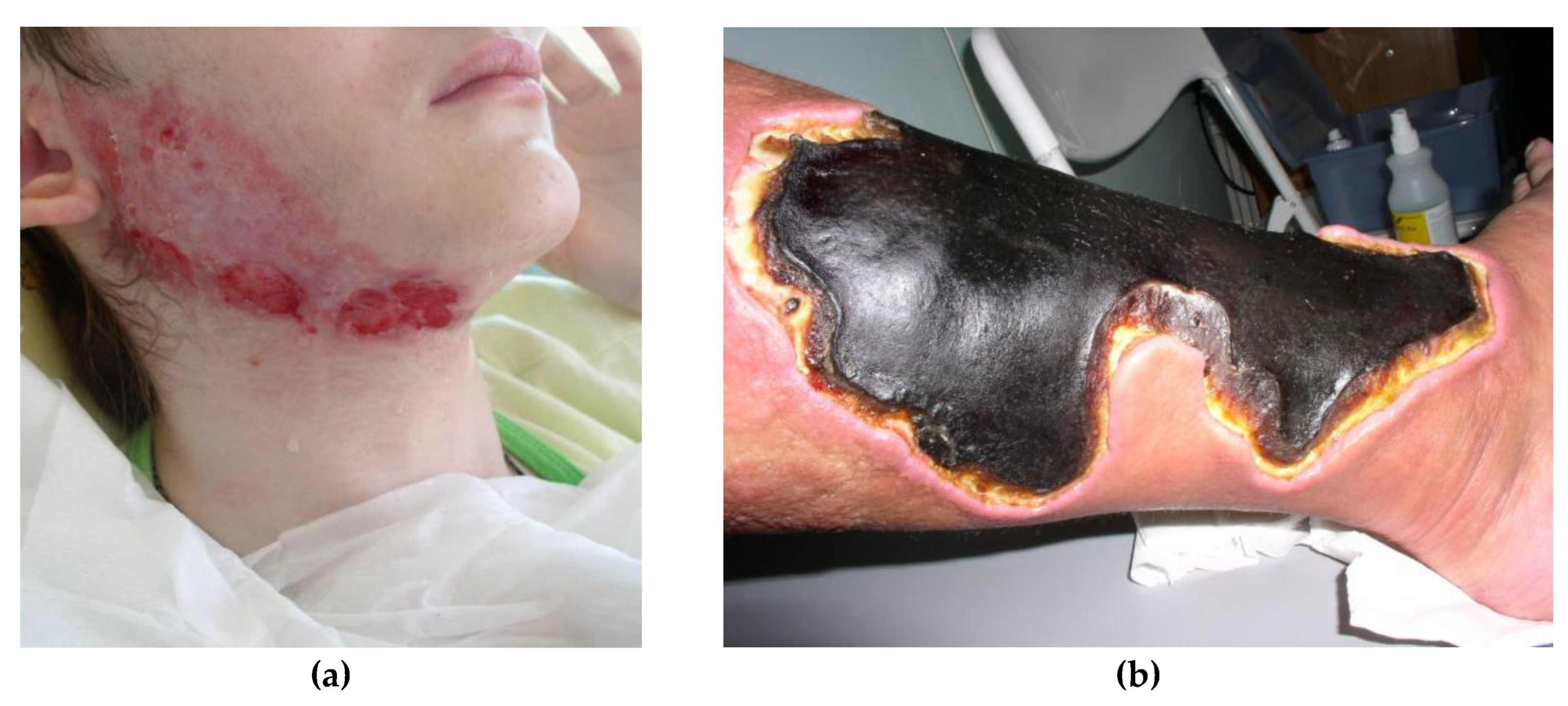
3. Paraneoplastic Syndromes That Are Easier to Recognize
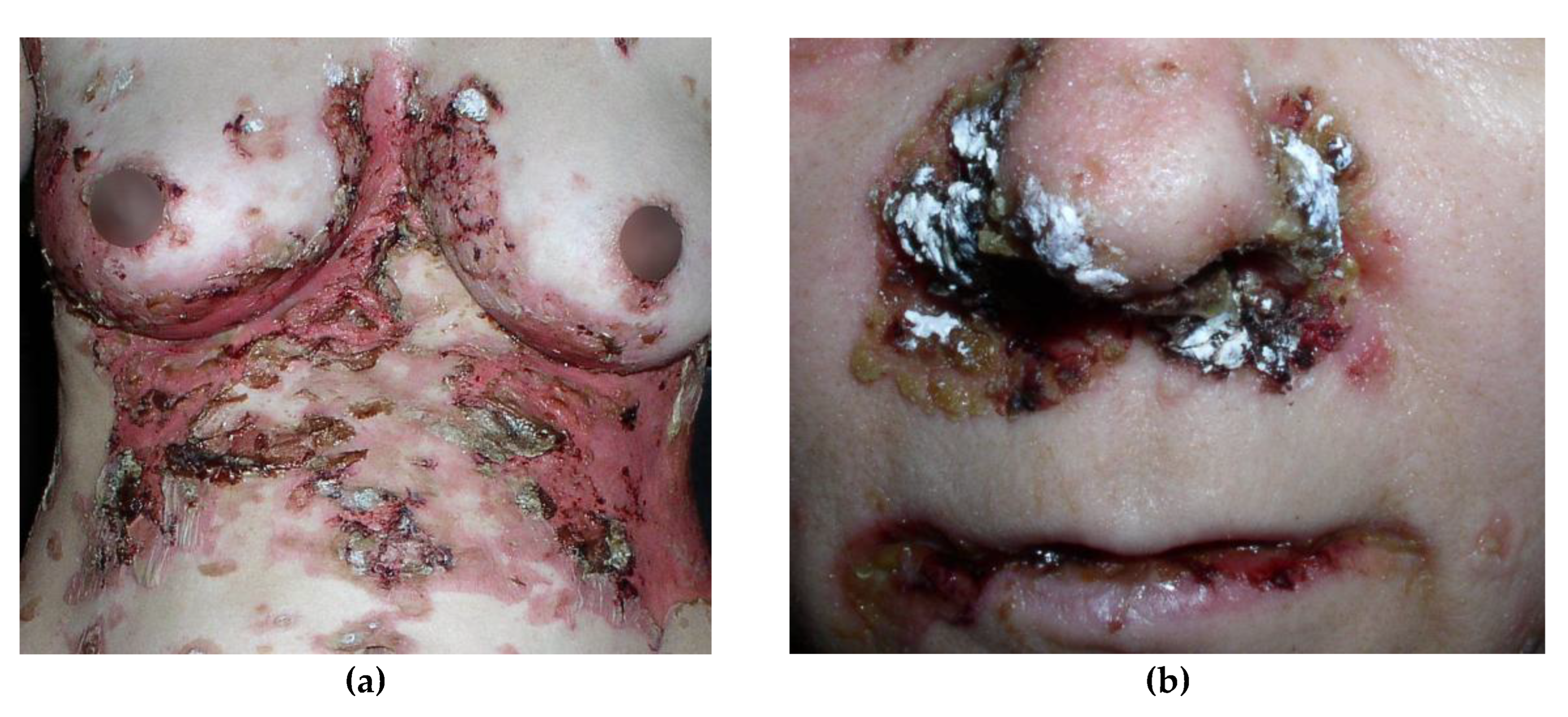
- Perivascular and perimysial inflammatory infiltrate: The infiltrate in dermatomyositis is concentrated around the perivascular and interfascicular regions and consists of B cells, CD4+ T helper cells, macrophages, and plasmacytoid dendritic cells. In contrast to polymyositis, CD8+ T cells and NK cells are rarely present.
- Perifascicular atrophy: Atrophy of muscle fibers, especially around the periphery of fascicles, is a hallmark histopathological feature of dermatomyositis. Degenerating and regenerating muscle fibers may be observed in the perifascicular region.
- Microangiopathy: Injury to intramuscular blood vessels takes the form of immunoglobulin and complement (C5b-C9 membrane attack complex) deposits on endomysial capillaries. A reduced capillary density and endothelial hyperplasia may be observed.
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Alter, M.; Mengoni, M.; Gaffal, E. Cutaneous manifestations of internal malignancy. J Dtsch Dermatol Ges. 2020, 18, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.A.; Mesquita Kde, C.; Igreja, A.C.; Lucas, I.C.; Freitas, A.F.; Oliveira, S.M.; Costa, I.M.; Campbell, I.T. Paraneoplastic cutaneous manifestations: Concepts and updates. An Bras Dermatol. 2013, 88, 9–22. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Eubanks, L.E.; McBurney, E.; Reed, R. Erythema gyratum repens. Am J Med Sci. 2001, 321, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Rongioletti, F.; Fausti, V.; Parodi, A. Erythema gyratum repens is not an obligate paraneoplastic disease: A systematic review of the literature and personal experience. J Eur Acad Dermatol Venereol. 2014, 28, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.S.; Neldner, K.H.; Menter, A. Erythema gyratum repens: A paraneoplastic eruption. J Am Acad Dermatol. 1992, 26 Pt 1, 757–762. [Google Scholar] [CrossRef] [PubMed]
- Holt, P.J.; Davies, M.G. Erythema gyratum repens--an immunologically mediated dermatosis? Br J Dermatol. 1977, 96, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Caux, F.; Lebbe, C.; Thomine, E.; Benyahia, B.; Flageul, B.; Joly, P.; Rybojad, M.; Morel, P. Erythema gyratum repens. A case studied with immunofluorescence, immunoelectron microscopy and immunohistochemistry. Br J Dermatol. 1994, 131, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Stone, S.P.; Buescher, L.S. Life-threatening paraneoplastic cutaneous syndromes. Clin Dermatol. 2005, 23, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Kleyn, C.E.; Lai-Cheong, J.E.; Bell, H.K. Cutaneous manifestations of internal malignancy. Am J Clin Dermatol. 2006, 7, 71–84. [Google Scholar] [CrossRef]
- Loucas, E.; Russo, G.; Millikan, L.E. Genetic and acquired cutaneous disorders associated with internal malignancy. Int J Dermatol. 1995, 34, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.S.; Neldner, K.H. Erythema gyratum repens without underlying disease. J Am Acad Dermatol. 1993, 28, 132. [Google Scholar] [CrossRef] [PubMed]
- Jablonska S, Blaszczyk M, Kozlowska A. Erythema gyratum repens-like psoriasis. Int J Dermatol. 2000 Sep;39(9):695-7. [CrossRef] [PubMed]
- Dermatol Rev/Przegl Dermatol 2017, 104, 439–445. [CrossRef]
- Skolnick, M.; Mainman, E.R. Erythema gyratum repens with metastatic adenocarcinoma. Arch Dermatol. 1975, 111, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.H.; Li, C.Y. Necrolytic migratory erythema-like eruption associated with erlotinib in a patient with lung adenocarcinoma. Lung Cancer. 2020, 148, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Trojan, A.; Jacky, E.; Follath, F.; Dummer, R. Necrolytic migratory erythema (glucagenoma)-like skin lesions induced by EGF-receptor inhibition. Swiss Med Wkly. 2003, 133, 22–23. [Google Scholar] [CrossRef] [PubMed]
- Seong, J.Y.; Kong, S.H.; Choi, Y.S.; Suh, H.S. Necrolytic Migratory Erythema–like Skin Lesion During Gefitinib Treatment: A Rare Cutaneous Adverse Reaction. JAMA Dermatol. 2016, 152, 947–948. [Google Scholar] [CrossRef]
- Tremblay, C.; Marcil, I. Necrolytic Migratory Erythema: A Forgotten Paraneoplastic Condition. J Cutan Med Surg. 2017, 21, 559–561. [Google Scholar] [CrossRef] [PubMed]
- Vinik, A.; Feliberti, E.; Perry, R. Glucagonoma syndrome. Endotext. South Dartmouth, MA: MDText.com; 2014. https://www.ncbi.nlm.nih.gov/books/NBK279041/.
- Wick, M.R.; Patterson, J.W. Cutaneous Paraneoplastic Syndromes. Semin. Diagn. Pathol. 2019. [Google Scholar] [CrossRef]
- Foss, M.G.; Hashmi, M.F.; Ferrer-Bruker, S.J. Necrolytic Migratory Erythema. 2023 Jul 25. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan–. [PubMed]
- Kovács, R.K.; Korom, I.; Dobozy, A.; Farkas, G.; Ormos, J.; Kemény, L. Necrolytic migratory erythema. J Cutan Pathol. 2006, 33, 242–245. [Google Scholar] [CrossRef] [PubMed]
- El Rassi, Z.; Partensky, C.; Valette, P.J.; Berger, F.; Chayvialle, J.A. Necrolytic migratory erythema, first symptom of a malignant glucagonoma: Treatment by long-acting somatostatin and surgical resection. Report of three cases. Eur J Surg Oncol. 1998, 24, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Al-Faouri, A.; Ajarma, K.; Alghazawi, S.; Al-Rawabdeh, S.; Zayadeen, A. Glucagonoma and Glucagonoma Syndrome: A Case Report with Review of Recent Advances in Management. Case Rep Surg. 2016, 2016, 1484089. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Caplin, M.E.; Pavel, M.; Ćwikła, J.B.; Phan, A.T.; Raderer, M.; Sedláčková, E.; Cadiot, G.; Wolin, E.M.; Capdevila, J.; Wall, L.; Rindi, G.; Langley, A.; Martinez, S.; Blumberg, J.; Ruszniewski, P.; CLARINET Investigators. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014, 371, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Shah, M.H.; Ito, T.; Bohas, C.L.; Wolin, E.M.; Van Cutsem, E.; Hobday, T.J.; Okusaka, T.; Capdevila, J.; de Vries, E.G.; Tomassetti, P.; Pavel, M.E.; Hoosen, S.; Haas, T.; Lincy, J.; Lebwohl, D.; Öberg, K.; RAD001 in Advanced Neuroendocrine Tumors, Third Trial (RADIANT-3) Study Group. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011, 364, 514–523. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Poole, S.; Fenske, N.A. Cutaneous markers of internal malignancy, I.I. Paraneoplastic dermatoses and environmental carcinogens. Journal of the American Academy of Dermatology 1993, 28, 147–164. [Google Scholar] [CrossRef]
- Sarkar, B.; Knecht, R.; Sarkar, C.; Weidauer, H. Bazex syndrome (acrokeratosis paraneoplastica). Eur Arch Otorhinolaryngol. 1998, 255, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Roy, B.; Lipner, S.R. A Review of Nail Changes in Acrokeratosis Paraneoplastica (Bazex Syndrome). Skin Appendage Disord. 2021, 7, 163–172. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Räßler, F.; Goetze, S.; Elsner, P. Acrokeratosis paraneoplastica (Bazex syndrome) - a systematic review on risk factors, diagnosis, prognosis and management. J Eur Acad Dermatol Venereol. 2017, 31, 1119–1136. [Google Scholar] [CrossRef] [PubMed]
- Amano, M.; Hanafusa, T.; Chikazawa, S.; Ueno, M.; Namiki, T.; Igawa, K.; Miura, K.; Yokozeki, H. Bazex Syndrome in Lung Squamous Cell Carcinoma: High Expression of Epidermal Growth Factor Receptor in Lesional Keratinocytes with Th2 Immune Shift. Case Rep Dermatol. 2016, 8, 358–362. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Valdivielso, M.; Longo, I.; Suárez, R.; Huerta, M.; Lázaro, P. Acrokeratosis paraneoplastica: Bazex syndrome. J Eur Acad Dermatol Venereol. 2005, 19, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.H.; Ferrazzano, C.; Karthikeyan, A.; Hejazi, H.; Bhattacharya, A.; Andrew Awuah, W.; Isik, A. Bazex Syndrome (Acrokeratosis Paraneoplastica): A Narrative Review of Pathogenesis, Clinical Manifestations, and Therapeutic Approaches. Cureus. 2023, 15, e45368. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Huilaja, L.; Soronen, M.; Karjalainen, A.; Tasanen, K. Acrokeratosis Paraneoplastica-like Findings as a Manifestation of Systemic Lupus Erythematosus. Acta Derm Venereol. 2019, 99, 333–334. [Google Scholar] [CrossRef] [PubMed]
- Bolognia, J.L. Bazex’ syndrome. Clin Dermatol. 1993, 11, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Vaynshtok, P.M.; Tian, F.; Kaffenberger, B.H. Acitretin amelioration of Acrokeratosis Paraneoplastica (Bazex Syndrome) in cases of incurable squamous cell carcinoma of the hypopharynx. Dermatol Online J. 2016, 22, 13030–qt8n96v45x. [Google Scholar] [CrossRef] [PubMed]
- Sweet, R.D. An acute febrile neutrophilic dermatosis. Br J Dermatol 1964, 76, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Fazili, T.; Duncan, D.; Wani, L. Sweet’s syndrome. Am J Med. 2010, 123, 694–696. [Google Scholar] [CrossRef] [PubMed]
- Burrall, B. Sweet’s syndrome (acute febrile neutrophilic dermatosis). Dermatol Online J. 1999, 5, 8. [Google Scholar] [PubMed]
- Su, W.P.; Liu, H.N. Diagnostic criteria for Sweet’s syndrome. Cutis. 1986, 37, 167–174. [Google Scholar] [PubMed]
- von den Driesch, P. Sweet’s syndrome (acute febrile neutrophilic dermatosis). J Am Acad Dermatol. 1994, 31, 535–556, quiz 557-60. [Google Scholar] [CrossRef] [PubMed]
- Villarreal-Villarreal, C.D.; Ocampo-Candiani, J.; Villarreal-Martínez, A. Sweet Syndrome: A Review and Update. Actas Dermosifiliogr. 2016, 107, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, L.; Baraf, C.S.; Richheimer, L.L. Sweet’s syndrome (acute febrile neutrophilic dermatosis). Arch Dermatol 1971, 103, 81–84. [Google Scholar] [CrossRef]
- Pirard, C.; Delannoy, A. Sweet syndrome and acute myeloid leukemia. Ann Dermatol Venereol 1977, 104, 160–161. [Google Scholar]
- Cohen, P.R.; Kurzrock, R. Chronic myelogenous leukemia and Sweet syndrome. Am J Hematol 1989, 32, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Hussein, K.; Nanda, A.; Al-Sabah, H.; Alsaleh, Q.A. Sweet’s syndrome (acute febrile neutrophilic dermatosis) associated with adenocarcinoma of prostate and transitional cell carcinoma of urinary bladder. J Eur Acad Dermatol Venereol 2005, 19, 597–599. [Google Scholar] [CrossRef] [PubMed]
- Adil, A.; Goyal, A.; Quint, J.M. Behcet Disease. 2023 Feb 22. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. [PubMed]
- Arun Kumar, A.U.; Elsayed, M.E.; Alghali, A.; Ali, A.A.; Mohamed, H.; Hussein, W.; Hackett, C.; Leonard, N.; Stack, A.G. Sweet syndrome: A rare feature of ANCA-associated vasculitis or unusual consequence of azathioprine-induced treatment. Allergy Asthma Clin Immunol. 2018, 14, 46. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Corbeddu, M.; Pilloni, L.; Pau, M.; Pinna, A.L.; Rongioletti, F.; Atzori, L. Treatment of Sweet’s syndrome in pregnancy. Dermatol Ther. 2018, 31, e12619. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.R.; Kurzrock, R. Sweet’s syndrome: A review of current treatment options. Am J Clin Dermatol. 2002, 3, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Kleyn, C.E.; Lai-Cheong, J.E.; Bell, H.K. Cutaneous Manifestations of Internal Malignancy. American Journal of Clinical Dermatology 2006, 7, 71–84. [Google Scholar] [CrossRef]
- Shah, M.; Sachdeva, M.; Gefri, A.; Jfri, A. Paraneoplastic pyoderma gangrenosum in solid organ malignancy: A literature review. Int J Dermatol. 2020, 59, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Duguid, C.M.; Powell, F.C. Pyoderma gangrenosum. Clin Dermatol. 1993, 11, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Marzano, A.V.; Le, S.T.; Callen, J.P.; Brüggen, M.C.; Guenova, E.; Dissemond, J.; Shinkai, K.; Langan, S.M. Pyoderma gangrenosum. Nat Rev Dis Primers. 2020, 6, 81. [Google Scholar] [CrossRef] [PubMed]
- Alavi, A.; French, L.E.; Davis, M.D.; Brassard, A.; Kirsner, R.S. Pyoderma Gangrenosum: An Update on Pathophysiology, Diagnosis and Treatment. Am J Clin Dermatol. 2017, 18, 355–372. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Ma, C.; Shinkai, K.; Fiorentino, D.; Callen, J.P.; Wollina, U.; Marzano, A.V.; Wallach, D.; Kim, K.; Schadt, C.; Ormerod, A.; Fung, M.A.; Steel, A.; Patel, F.; Qin, R.; Craig, F.; Williams, H.C.; Powell, F.; Merleev, A.; Cheng, M.Y. Diagnostic Criteria of Ulcerative Pyoderma Gangrenosum: A Delphi Consensus of International Experts. JAMA Dermatol. 2018, 154, 461–466. [Google Scholar] [CrossRef] [PubMed]
- George, C.; Deroide, F.; Rustin, M. Pyoderma gangrenosum - a guide to diagnosis and management . Clin Med 2019, 19, 224–228. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- You, H.R.; Ju, J.K.; Yun, S.J.; Lee, J.B.; Kim, S.J.; Won, Y.H.; Lee, S.C. Paraneoplastic Pyoderma Gangrenosum Associated with Rectal Adenocarcinoma. Ann Dermatol. 2018, 30, 79–82. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Karakayli, G.; Beckham, G.; Orengo, I.; Rosen, T. Exfoliative dermatitis. Am Fam Physician. 1999, 59, 625–630. [Google Scholar] [PubMed]
- Zervakis, S.G.; Spernovasilis, N.; Boutakoglou, E.; Panagiotakis, S.; Thomopoulou, K.; Samonis, G. Erythroderma as a paraneoplastic manifestation of small cell lung cancer. Curr Probl Cancer. 2020, 44, 100499. [Google Scholar] [CrossRef] [PubMed]
- Kerasia Maria Plachouri Paraneoplastic erythroderma: An insight on the existing data.
- Pal, S.; Haroon, T.S. Erythroderma: A clinico-etiologic study of 90 cases. Int J Dermatol 1998, 37104, 10.1046/j.1365-4362.1998.00228.x. [Google Scholar]
- Rothe, M.J.; Bialy, T.L.; Grant-Kels, J.M. Erythroderma. Dermatol Clin. 2000, 18, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, V.N. Srivastava G Exfoliative dermatitis: A prospective study of 80 patients. Dermatologica 1986, 173, 278. [Google Scholar] [CrossRef]
- Duangurai, K.; Piamphongsant, T.; Himmungnan, T. Sézary cell count in exfoliative dermatitis. Int J Dermatol. 1988, 27, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.C.; Jester, J.D.; King, L.E., Jr. Erythroderma and exfoliative dermatitis. Clin Dermatol. 1993, 11, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Muller CS, Lorenz MT, Tilgen W, € et al. Primary manifestation of erythema gyratum repens as a transient erythroderma in a patient with bronchial carcinoma. Int J Dermatol 2010, 49, 676–678. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Lee, K.M.; Ucmak, D.; Brodsky, M.; Atanelov, Z.; Farahnik, B.; Abrouk, M.; Nakamura, M.; Zhu, T.H.; Liao, W. Erythrodermic psoriasis: Pathophysiology and current treatment perspectives. Psoriasis 2016, 6, 93–104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Roenneberg, S.; Biedermann, T. Pityriasis rubra pilaris: Algorithms for diagnosis and treatment. J Eur Acad Dermatol Venereol. 2018, 32, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Jawed, S.I.; Myskowski, P.L.; Horwitz, S.; Moskowitz, A.; Querfeld, C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): Part I. Diagnosis: Clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014, 70, 205.e1-16, quiz 221–222. [Google Scholar] [CrossRef] [PubMed]
- Roccuzzo, G.; Giordano, S.; Avallone, G.; Rubatto, M.; Canonico, S.; Funaro, A.; Ortolan, E.; Senetta, R.; Fava, P.; Fierro, M.T.; Ribero, S.; Quaglino, P. Sézary Syndrome: Different Erythroderma Morphological Features with Proposal for a Clinical Score System. Cells. 2022, 11, 333. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sharma, G.; Govil, D.C. Allopurinol induced erythroderma. Indian J Pharmacol. 2013, 45, 627–628. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Reynolds, N.J.; Jones, S.K.; Crossley, J.; Harman, R.R. Exfoliative dermatitis due to nifedipine. Br J Dermatol. 1989, 121, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Tietze, K.J.; Gaska, J.A. Cefoxitin-associated exfoliative dermatitis. Clin Pharm. 1983, 2, 582–584. [Google Scholar] [PubMed]
- Mistry, Nisha MD, FRCPC.; Gupta, Ambika; Alavi, Afsaneh MD, FRCPC.; Sibbald, R. Gary BSc, MD, MEd, FRCPC(Med)(Derm), FACP, FAAD, MAPWCA. A Review of the Diagnosis and Management of Erythroderma (Generalized Red Skin). Advances in Skin & Wound Care 28, p 228-236, May 2015. |. [CrossRef]
- Joly P, Richard C, Gilbert D, Courville P, Chosidow O, Roujeau JC; et al. Sensitivity and specificity of clinical, histologic, and immunologic features in the diagnosis of paraneoplastic pemphigus. J Am Acad Dermatol 2000, 43: 619-26. [CrossRef]
- Martel, P.; Loiseau, P.; Joly, P.; Busson, M.; Lepage, V.; Mouquet, H.; Courville, P.; Flageul, B.; Charron, D.; Musette, P.; Gilbert, D.; Tron, F. Paraneoplastic pemphigus is associated with the DRB1*03 allele. J Autoimmun. 2003, 20, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Bu, D.F.; Li, D.; Zhu, X.J. Genotyping of HLA-I and HLA-II alleles in Chinese patients with paraneoplastic pemphigus. Br J Dermatol. 2008, 158, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Sklavounou, A.; Laskaris, G. Paraneoplastic pemphigus: A review. Oral Oncol 1998, 34, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Kimyai-Asadi, A.; Jih, M.H. Paraneoplastic pemphigus. Int J Dermatol. 2001, 40, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Edgin, W.A.; Pratt, T.C.; Grimwood, R.E. Pemphigus Vulgaris and Paraneoplastic Pemphigus. Oral Maxillofac Surg Clin North Am. 2008, 20, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Paolino, G.; Didona, D.; Magliulo, G.; Iannella, G.; Didona, B.; Mercuri, S.R.; Moliterni, E.; Donati, M.; Ciofalo, A.; Granata, G.; et al. Paraneoplastic Pemphigus: Insight into the Autoimmune Pathogenesis, Clinical Features and Therapy. Int. J. Mol. Sci. 2017, 18, 2532. [Google Scholar] [CrossRef]
- Meyers, S.J.; Varley, G.A.; Meisler, D.M.; Camisa, C.; Wander, A.H. Conjunctival involvement in paraneoplastic pemphigus. Am J Ophthalmol. 1992, 114, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Nousari, H.C.; Deterding, R.; Wojtczack, H.; Aho, S.; Uitto, J.; Hashimoto, T.; Anhalt, G.J. The mechanism of respiratory failure in paraneoplastic pemphigus. N Engl J Med. 1999, 340, 1406–1410. [Google Scholar] [CrossRef] [PubMed]
- Nguyen VT, Ndoye A, Bassler KD et al. Classification, clinical manifestations, and immunopathological mechanisms of the epithelial variant of paraneoplastic autoimmune multiorgan syndrome: A reappraisal of paraneoplastic pemphigus. Arch. Dermatol. 2001; 137: 193–206.
- Maruta, C.W.; Miyamoto, D.; Aoki, V.; Carvalho, R.G.R.; Cunha, B.M.; Santi, C.G. Paraneoplastic pemphigus: A clinical, laboratorial, and therapeutic overview. An Bras Dermatol. 2019, 94, 388–398. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Probst C, Schlumberger W, Stöcker W et al. Development of ELISA for the specific determination of autoantibodies against envoplakin and periplakin in paraneoplastic pemphigus. Clin. Chim. Acta 2009; 410: 13–18. [CrossRef]
- Kartan, S.; Shi, V.Y.; Clark, A.K.; Chan, L.S. Paraneoplastic Pemphigus and Autoimmune Blistering Diseases Associated with Neoplasm: Characteristics, Diagnosis, Associated Neoplasms, Proposed Pathogenesis, Treatment. Am J Clin Dermatol. 2017, 18, 105–126. [Google Scholar] [CrossRef] [PubMed]
- Tsuchisaka, A.; Numata, S.; Teye, K.; Natsuaki, Y.; Kawakami, T.; Takeda, Y.; Wang, W.; Ishikawa, K.; Goto, M.; Koga, H.; Sogame, R.; Ishii, N.; Takamori, S.; Hoshino, T.; Brandt, O.; Pas, H.H.; Fujiwara, S.; Hashimoto, T. Epiplakin Is a Paraneoplastic Pemphigus Autoantigen and Related to Bronchiolitis Obliterans in Japanese Patients. J Invest Dermatol. 2016, 136, 399–408. [Google Scholar] [CrossRef]
- Didona, D.; Fania, L.; Didona, B.; Eming, R.; Hertl, M.; Di Zenzo, G. Paraneoplastic Dermatoses: A Brief General Review and an Extensive Analysis of Paraneoplastic Pemphigus and Paraneoplastic Dermatomyositis. Int J Mol Sci. 2020, 21, 2178. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yong, A.A.; Tey, H.L. Paraneoplastic pemphigus. Australas J Dermatol. 2013, 54, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Nam, K.H.; Lee, J.B.; Lee, J.Y.; Ihm, C.W.; Lee, S.E.; Oh, S.H.; Hashimoto, T.; Kim, S.C. Retrospective analysis of 12 Korean patients with paraneoplastic pemphigus. J Dermatol. 2012, 39, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Kershenovich, R.; Hodak, E.; Mimouni, D. Diagnosis and classification of pemphigus and bullous pemphigoid. Autoimmun Rev. 2014, 13, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Bowen, G.M.; Peters, N.T.; Fivenson, D.P.; Su, L.D.; Nousari, H.C.; Anhalt, G.J.; Cooper, K.D.; Stevens, S.R. Lichenoid dermatitis in paraneoplastic pemphigus: A pathogenic trigger of epitope spreading? Arch Dermatol. 2000, 136, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Wade, M.S.; Black, M.M. Paraneoplastic pemphigus: A brief update. Australas J Dermatol. 2005, 46, 1–8, quiz 9-10. [Google Scholar] [CrossRef] [PubMed]
- Murrell, D.F.; Peña, S.; Joly, P.; Marinovic, B.; Hashimoto, T.; Diaz, L.A.; Sinha, A.A.; Payne, A.S.; Daneshpazhooh, M.; Eming, R.; et al. Diagnosis and management of pemphigus: Recommendations of an international panel of experts. J Am Acad Dermatol. 2020, 82, 575–585.e1. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kikuchi, T.; Mori, T.; Shimizu, T.; Koda, Y.; Abe, R.; Kurihara, Y.; Funakoshi, T.; Yamagami, J.; Sato, H.; Tsunoda, K.; Amagai, M.; Okamoto, S. Successful treatment with bendamustine and rituximab for paraneoplastic pemphigus. Ann Hematol. 2017, 96, 1221–1222. [Google Scholar] [CrossRef]
- Bech, R.; Baumgartner-Nielsen, J.; Peterslund, N.A.; Steiniche, T.; Deleuran, M.; d’Amore, F. Alemtuzumab is effective against severe chronic lymphocytic leukaemia-associated paraneoplastic pemphigus. Br J Dermatol. 2013, 169, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Hohwy, T.; Bang, K.; Steiniche, T.; Peterslund, N.A.; d’Amore, F. Alemtuzumab-induced remission of both severe paraneoplastic pemphigus and leukaemic bone marrow infiltration in a case of treatment-resistant B-cell chronic lymphocytic leukaemia. Eur J Haematol. 2004, 73, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Sevim, E.; Kobrin, D.; Casal-Dominguez, M.; Pinal-Fernandez, I. A comprehensive review of dermatomyositis treatments - from rediscovered classics to promising horizons. Expert Rev Clin Immunol. 2024, 20, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Kronzer, V.L.; Kimbrough, B.A.; Crowson, C.S.; Davis JM 3rd Holmqvist, M.; Ernste, F.C. Incidence, Prevalence, and Mortality of Dermatomyositis: A Population-Based Cohort Study. Arthritis Care Res 2023, 75, 348–355. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- O’Hanlon, T.P.; Rider, L.G.; Mamyrova, G.; Targoff, I.N.; Arnett, F.C.; Reveille, J.D.; Carrington, M.; Gao, X.; Oddis, C.V.; Morel, P.A.; Malley, J.D.; Malley, K.; Shamim, E.A.; Chanock, S.J.; Foster, C.B.; Bunch, T.; Reed, A.M.; Love, L.A.; Miller, F.W. HLA polymorphisms in African Americans with idiopathic inflammatory myopathy: Allelic profiles distinguish patients with different clinical phenotypes and myositis autoantibodies. Arthritis Rheum. 2006, 54, 3670–81. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Han, L.; Yuan, L.; Yang, Y.; Gou, G.; Sun, H.; Lu, L.; Bao, L. HLA class II alleles may influence susceptibility to adult dermatomyositis and polymyositis in a Han Chinese population. BMC Dermatol. 2014, 14, 9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Okiyama, N.; Fujimoto, M. Cutaneous manifestations of dermatomyositis characterized by myositis-specific autoantibodies. F1000Res. 2019, 8, F1000. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Strowd, L.C.; Jorizzo, J.L. Review of dermatomyositis: Establishing the diagnosis and treatment algorithm. J Dermatolog Treat. 2013, 24, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Pappu, R.; Seetharaman, M. Polymoysitis. In Medscape Reference. Available from http://emedicine.medscape.com/article/335925..accessed 8 June 2011.
- Adams, E.M.; Chow, C.K.; Premkumar, A.; Plotz, P.H. The idiopathic inflammatory myopathies: Spectrum of MR imaging findings. Radiographics. 1995, 15, 563–574. [Google Scholar] [CrossRef]
- Vattemi, G.; Mirabella, M.; Guglielmi, V.; Lucchini, M.; Tomelleri, G.; Ghirardello, A.; Doria, A. Muscle biopsy features of idiopathic inflammatory myopathies and differential diagnosis. Auto Immun Highlights. 2014, 5, 77–85. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sigurgeirsson, B.; Lindelöf, B.; Edhag, O.; Allander, E. Risk of cancer in patients with dermatomyositis or polymyositis. A population-based study. N Engl J Med. 1992, 326, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Emslie-Smith, A.M.; Engel, A.G. Microvascular changes in early and advanced dermatomyositis: A quantitative study. Ann Neurol. 1990, 27, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Maoz, C.R.; Langevitz, P.; Livneh, A.; Blumstein, Z.; Sadeh, M.; Bank, I.; Gur, H.; Ehrenfeld, M. High incidence of malignancies in patients with dermatomyositis and polymyositis: An 11-year analysis. Semin Arthritis Rheum. 1998, 27, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, D.; Casciola-Rosen, L. Autoantibodies to transcription intermediary factor 1 in dermatomyositis shed insight into the cancer-myositis connection. Arthritis Rheum. 2012, 64, 346–349. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ofori, E.; Ramai, D.; Ona, M.; Reddy, M. Paraneoplastic Dermatomyositis Syndrome Presenting as Dysphagia. Gastroenterology Res. 2017, 10, 251–254. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Callen, J.P.; Hyla, J.F.; Bole, G.G., Jr.; Kay, D.R. The relationship of dermatomyositis and polymyositis to internal malignancy. Arch Dermatol. 1980, 116, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Zhang, L.; Jia, H.; Nie, Z.; Zhang, L. Malignancy in dermatomyositis: A retrospective paired case-control study of 202 patients from Central China. Medicine (Baltimore). 2020, 99, e21733. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ponti, G.; Luppi, G.; Losi, L.; Giannetti, A.; Seidenari, S. Leser-Trélat syndrome in patients affected by six multiple metachronous primitive cancers. J Hematol Oncol. 2010, 3, 2. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Safa, G.; Darrieux, L. Leser-Trélat Sign without Internal Malignancy. Case Rep Oncol. 2011, 4, 175–177. [Hafner C, Vogt T. Seborrheic keratosis. J Dtsch Dermatol Ges. 2013, 6, 664–677. [Google Scholar] [CrossRef]
- Inamadar, A.C.; Palit, A. Eruptive seborrhoeic keratosis in human immunodeficiency virus infection: A coincidence or ’the sign of Leser-Trélat’? Br J Dermatol. 2003, 149, 435–436. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Salafranca, M.; Gómez-Martín, I.; Bañuls, J.; Serrano, P.; Medina, C.; Llambrich, A.; Pizarro, Á.; Ara, M.; Zaballos, P. Dermoscopy of inflamed seborrheic keratosis: A great mimic of malignancy. Australas J Dermatol. 2022, 63, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Myroshnychenko, M.S.; Moiseienko, T.M.; Torianyk, I.I.; Ivannik, V.Y.; Popova, N.G.; Mozhaiev, I.V.; Chastii, T.V.; Minukhin, V.V.; Leontiev, P.A.; Osolodchenko, T.P.; Parkhomenko, K.Y. Seborrheic Keratosis: Current State of the Problem. Wiad Lek. 2022, 75 Pt 2, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Roh, N.K.; Hahn, H.J.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Clinical and Histopathological Investigation of Seborrheic Keratosis. Ann Dermatol. 2016, 28, 152–158. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gaduputi, V.; Chandrala, C.; Tariq, H.; Kanneganti, K. Sign of leser-trélat associated with esophageal squamous cell cancer. Case Rep Oncol Med. 2014, 2014, 825929. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aouali, S.; Bensalem, S.; Saddouk, H.; Aissaoui, A.; Bennani, A.; Zizi, N.; Dikhaye, S. Leser-Trelat sign preceding male breast cancer. Ann Med Surg (Lond). 2021, 72, 103065. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Modi, K.; Chen, R.; Abubshait, L. Leser-Trélat Sign as a Marker for Underlying Pancreatic Cancer. Clin Pract Cases Emerg Med. 2023, 7, 202–204. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mathien, A.; Kuraitis, D. Leser-Trelat sign: Eruptive seborrheic keratoses and primary lung adenocarcinomas with an epidermal growth factor receptor mutation. JAAD Case Rep. 2023, 37, 38–40. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wollina, U. Recent advances in managing and understanding seborrheic keratosis. F1000Res. 2019, 8, F1000. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Valejo Coelho, M.M.; Prokop, J.; Fernandes, C. Acanthosis Nigricans Maligna. JAMA Dermatol. 2019, 155, 1069. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, R.; Liu, Y.; Zhang, S.; Qi, H. Malignant acanthosis nigricans: A case report. BMC Ophthalmol. 2020, 20, 446. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hermanns-Lê, T.; Scheen, A.; Piérard, G.E. Acanthosis nigricans associated with insulin resistance : Pathophysiology and management. Am J Clin Dermatol. 2004, 5, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Chung, V.Q.; Moschella, S.L.; Zembowicz, A.; Liu, V. Clinical and pathologic findings of paraneoplastic dermatoses. J Am Acad Dermatol. 2006, 54, 745–762, quiz 763-6. [Google Scholar] [CrossRef] [PubMed]
- Pollitzer, S. Acanthosis nigricans: A symptom of a disorder of the abdominal sympathetic. JAMA 1909, 53, 1369–1373. [Google Scholar] [CrossRef]
- Zhang, N.; Qian, Y.; Feng, A.P. Acanthosis nigricans, tripe palms, and sign of Leser-Trélat in a patient with gastric adenocarcinoma: Case report and literature review in China. Int J Dermatol. 2015, 54, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Yeh, J.S.; Munn, S.E.; Plunkett, T.A.; Harper, P.G.; Hopster, D.J.; du Vivier, A.W. Coexistence of acanthosis nigricans and the sign of Leser-Trélat in a patient with gastric adenocarcinoma: A case report and literature review. J Am Acad Dermatol. 2000, 42 Pt 2, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, W.J.; Goldin, H.M.; Bronson, D.M.; Brody, P.E. Acanthosis nigricans associated with mycosis fungoides. J Am Acad Dermatol. 1988, 19 Pt 2, 951–953. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Amador, V.; Esquivel-Pedraza, L.; Caballero-Mendoza, E.; Berumen-Campos, J.; Orozco-Topete, R.; Angeles-Angeles, A. Oral manifestations as a hallmark of malignant acanthosis nigricans. J Oral Pathol Med. 1999, 28, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Rigel, D.S.; Jacobs, M.I. Malignant acanthosis nigricans: A review. J Dermatol Surg Oncol. 1980, 6, 923–927. [Google Scholar] [CrossRef] [PubMed]
- Darmstadt, G.L.; Yokel, B.K.; Horn, T.D. Treatment of acanthosis nigricans with tretinoin. Arch Dermatol. 1991, 127, 1139–1140. [Google Scholar] [CrossRef] [PubMed]
- Blobstein, S.H. Topical therapy with tretinoin and ammonium lactate for acanthosis nigricans associated with obesity. Cutis. 2003, 71, 33–34. [Google Scholar] [PubMed]
- Gregoriou, S.; Anyfandakis, V.; Kontoleon, P.; Christofidou, E.; Rigopoulos, D.; Kontochristopoulos, G. Acanthosis nigricans associated with primary hypogonadism: Successful treatment with topical calcipotriol. J Dermatolog Treat. 2008, 19, 373–375. [Google Scholar] [CrossRef] [PubMed]
| Category | Condition |
| Metabolic and Endocrine Disorders | Acanthosis nigricans |
| Granulomatous Diseases | Acute complications of sarcoidosis |
| Malignancies | Cutaneous T-cell lymphoma |
| Autoimmune Blistering Diseases | Bullous pemphigoid, Pemphigus foliaceus |
| Allergic Diseases | Irritant contact dermatitis, Pediatric atopic dermatitis, Allergic contact dermatitis |
| Inflammatory Skin Diseases | Plaque psoriasis, Lichen planus, Pityriasis rubra pilaris, Seborrheic dermatitis, Stasis dermatitis |
| Reumatologic Diseases | Reactive arthritis |
| Genodermatosis | Familial bening pemhigus (Hailey-Hailey disease) |
| Other diseases | Dermatologic manifestations of graft versus host disease |
| Protein name | Molecular Weight (kD) | Location |
|---|---|---|
| Desmoglein type 1 (Dsg1) Desmoglein type 3 (Dsg3) |
160 130 |
Desmosome, extracellular |
| Desmoplakin 1 Desmoplakin 2 |
250 210 |
Desmosome, intracellular |
| Bullous pemhigoid antigen 1 (BP230) | 230 | Hemidesmosome/lamina lucida |
| Envoplakin | 210 | Desmosome, intracellular |
| Epiplakin | >700 | Desmosome, intracellular |
| Periplakin | 190 | Desmosome, intracellular |
| Alpha-2-macroglobulin-like-1-antigen (A2ML1) | 170 | Protease inhibitor |
| Skin finding | Description | |
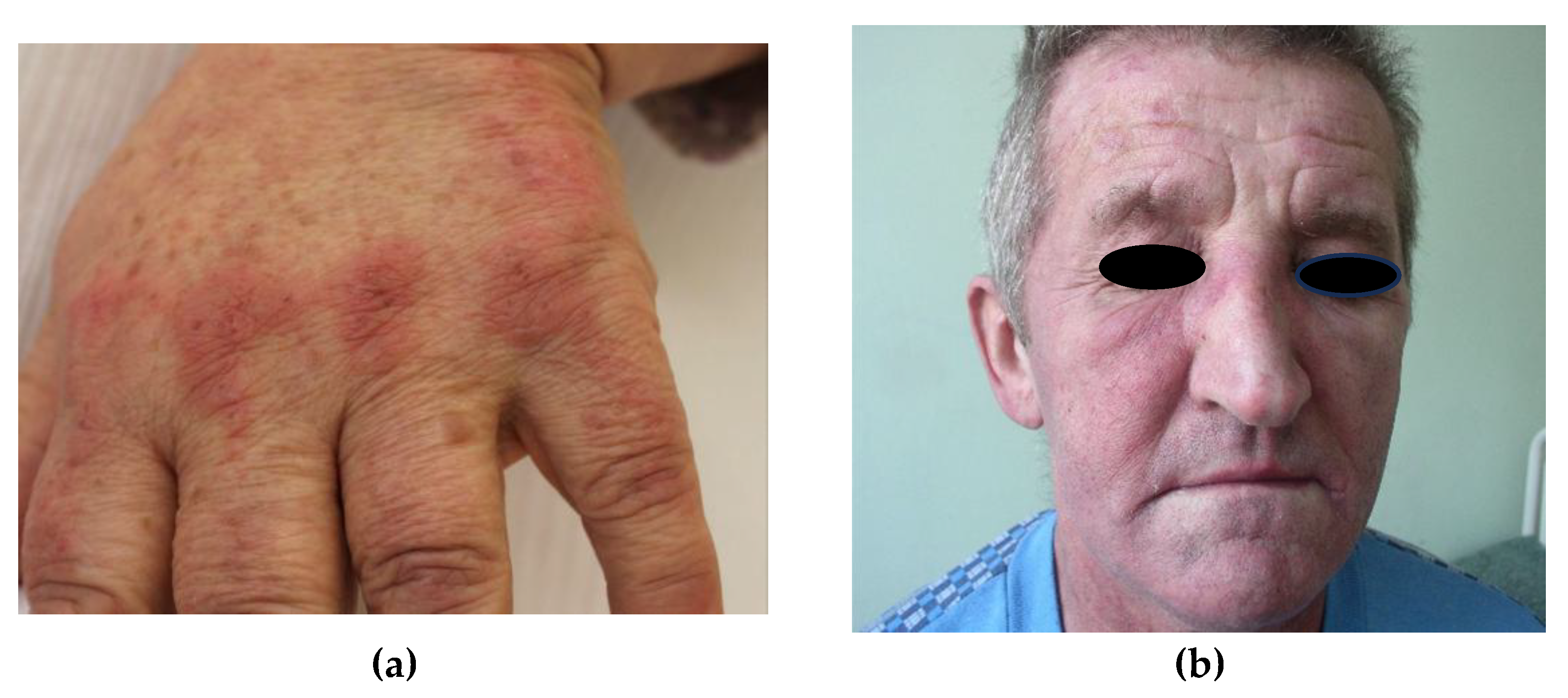
| Pathognomic symptoms | Gottron papules | erythematous or violaceous papules with or without scaling or ulceration located over the interphalangeal or metacarpophalangeal joints |
| Heliotrope rash | violaceous, or an erythematous rash effecting upper eyelids with or without periorbital edema; may not be apparent in patients with dark skin | |
| Other symptoms | Gottron sign | erythematous macules or patches over the elbows or knees |
| Facial erythema | erythema over the cheeks and nasal bridge involving the nasolabial folds, may extend up to the forehead and ears | |
| Shawl sign | erythema over the posterior aspect of the neck, upper back and shoulders at times, extending to the upper arms | |
| V sign | ill-defined erythematous macules involving the anterior aspect of the neck and the upper chest | |
| Poikiloderma | atrophic skin with changes in pigmentation and teleangiectasia in photo-exposed or non-exposed areas | |
| Holster sign | poikilodermia involving the lateral aspects of the thighs | |
| Periungual involvement | teleangiectasis and cuticular overgrowth | |
| Mechanic’s hands | hyperkeratotic, cracked horizontal lines on the palmar and lateral aspects of the fingers | |
| Scalp involvement | diffuse poikilodermia, with scaling and pruritis | |
| Calcinosis cutis | calcium deposits in the skin |
| The most common causes | The rarer causes |
| Adenocarcinomas | Breast cancer |
|
Hematopoietic neoplasms Lung cancer Pancreatic malignancy |
|
Prostate malignancy Kidney malignancy |
| Laryngeal malignancy | |
| Ovarian and uterine malignancy | |
| Bladder malignancy | |
| Nasopharyngeal carcinoma | |
| Melanoma | |
| Mycosis fungoides | |
| Hepatocellular carcinoma | |
| Squamous cell carcinoma |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





