Submitted:
05 February 2025
Posted:
06 February 2025
You are already at the latest version
Abstract
Background: This study examines the challenges associated with pharmacological management in fibromyalgia (FM), a multifaceted syndrome characterized by widespread pain, cognitive impairment and fatigue. A comparative analysis of drug usage, potential drug interactions, and adherence to clinical guidelines highlights the critical need to evaluate how medications may contribute to symptoms such as fibro-fog and affect overall quality of life, with the aim of optimizing patient` well-being. Objectives: The primary aim of this study was to assess medication usage, identify potential drug interactions, and evaluate treatment suitability in FM patients, with a particular focus on recent pharmacological advancements and their impact on symptom management. Methods: This cross-sectional study analyzed medication usage and potential drug interactions in women with FM. Tools such as CheckTheMeds® and the CRIDECO Anticholinergic Load Scale were employed to assess the pharmacological appropriateness of treatments in alignment with clinical guidelines, as well as to evaluate the anticholinergic burden and its potential cognitive impacts. Results: The study included 108 women with FM, with a mean age of 54.06 ± 8.43 years. Pain management medications accounted for 72.94% of total medication prescribed. The analysis revealed a significant anticholinergic burden in 50.93% of the patients, and a total of (881 alerts were recorded for potential drug interactions). Conclusions: Pharmacological reviews are essential in the management of FM, as they enhance treatment safety, minimize risks such as CNS depression and high anticholinergic burden, and help to alleviate symptoms like fibro-fog or cognitive impairment. Personalized medication approaches can improve patient outcomes and quality of life, particularly given the complexities and lack of consensus regarding FM treatment strategies.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Medication Analysis
2.2. Fibromyalgia Guidelines
2.3. Anticholinergic Load Scale
3. Results
3.1. Descriptive Patients
3.2. Interactions
3.3. Study of Fibromyalgia Clinical Guidelines
3.4. Anticholinergic Burden
| Without Anticholinergic Load | Low Potency (Score 1) | Medium Potency (Score 2) | High Potency (Score 3) | ||
|---|---|---|---|---|---|
|
Almotriptan (N02CC05) 0.93% |
Levosulpiride (N05AL07) 0.93% |
Alprazolam (N05BA12) 15.74% |
Lorazepam (N05BA06) 12.04% |
Baclofen (M03BX01) 2.78% |
Amitriptyline* (N06AA09)18.52% |
| Bromazepam (N05BA08)9.26% | Lormetazepam (N05CD06)4.63% | Celecoxib (M01AH01) 1.85% |
Methocarbamol (M03BA03) 3.70% |
Carbamazepine (N03AF01) 0.93% |
Biperiden (N04AA02) 0.93% |
| Chondroitin Sulfate (M01AX25) 2.78% |
Medazepam (N05BA03) 0.93% |
Citalopram (N06AB04) 1.85% |
Mirtazapine (N06AX11) 1.85% |
Maprotiline (N06AA21) 17.59% |
Chlorpromazine (N05AA01) 0.93% |
| Denosumab (M05BX04)2.78% | Melatonin (N05CH01)0.93% | Clonazepam (N03AE01) 9.26% |
Oxycodone (N02AA05) 3.70% |
Olanzapine (N05AH03) 2.78% |
Hydroxyzine (N05BB01) 1.85% |
| Dexketoprofen (M01AE17) 8.33% |
Metamizole Sodium (N02BB02) 13.89% |
Desvenlafaxine (N06AX23) 2.78% |
Potassium clorazepate (N05BA05) 1.85% |
Paroxetine (N06AB05) 2.78% |
Tizanidine (M03BX02) 1.85% |
|
Duloxetine* (N06AX21) 44.44% |
Naproxen (M01AE02)3.70% | Diazepam (N05BA01) 23.15% |
Prednisone (H02AB07) 0.93% |
Quetiapine (N05AH04) 2.78% |
|
| Etoricoxib (M01AH05) 20.37% |
Paracetamol (N02BE01) 55.56% |
Escitalopram (N06AB10) 7.41% |
Tapentadol (N02AX06) 5.56% |
Tramadol* (N02AX02) 6.44% |
|
| Gabapentin (N02BF01) 7.41% |
Pregabalin* (N02BF02) 20.37% |
Fentanyl (N02AB03) 0.93% |
Trazodone (N06AX05) 18.52% |
||
| Glucosamine (M01AX05) 0.93% |
Risedronic Acid (M05BA07) 3.70% |
Fluoxetine (N06AB03) 7.41% |
Venlafaxine (N06AX16) 6.48% |
||
| Ibuprofen (M01AE01) 4.63% |
Topiramate (N03AX11) 4.63% |
Flurazepam (N05CD01) 1.85% |
Zolmitriptan (N02CC03) 0.93% |
||
| Ketazolam (N05BA10) 1.85% |
Vortioxetine (N06AX26) 3.70% |
||||
| Lacosamide (N03AX18) 2.78% |
Zolpidem (N05CF02) 2.78% |
||||
| Lamotrigine (N03AX09) 0.93% |
|||||
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- O’Malley, P.G.; Balden, E.; Tomkins, G.; Santoro, J.; Kroenke, K.; Jackson, J.L. Treatment of fibromyalgia with antidepressants: A meta-analysis. J Gen Intern Med. 2000, 15, 659–666. [Google Scholar] [CrossRef]
- Varrassi, G.; Rekatsina, M.; Perrot, S.; Bouajina, E.; Paladini, A.; Coaccioli, S.; et al. Is fibromyalgia a fashionable diagnosis or a medical mystery? Cureus 2023. [Google Scholar] [CrossRef] [PubMed]
- Gil-Ugidos, A.; Rodríguez-Salgado, D.; Pidal-Miranda, M.; Samartin-Veiga, N.; Fernández-Prieto, M.; Carrillo-de-la-Peña, M.T. Working memory performance, pain and associated clinical variables in women with fibromyalgia. Front Psychol. 2021, 12. [Google Scholar] [CrossRef]
- Gmuca, S.; Sonagra, M.; Xiao, R.; Mendoza, E.; Miller, K.S.; Thomas, N.H.; et al. Characterizing neurocognitive impairment in juvenile fibromyalgia syndrome: Subjective and objective measures of dyscognition. Front Pediatr. 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, K.R.; Gracely, R.H.; Glass, J.M. Fibromyalgia dyscognition: Concepts and issues. Reumatismo. 2012, 64. [Google Scholar] [CrossRef] [PubMed]
- Dass, R.; Kalia, M.; Harris, J.; Packham, T. Understanding the experience and impacts of brain fog in chronic pain: A scoping review. Can J Pain. 2023, 7. [Google Scholar] [CrossRef]
- Kratz, A.L.; Whibley, D.; Kim, S.; Williams, D.A.; Clauw, D.J.; Sliwinski, M. The role of environmental distractions in the experience of fibrofog in real-world settings. ACR Open Rheumatol. 2020, 2, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Kratz, A.L.; Whibley, D.; Kim, S.; Sliwinski, M.; Clauw, D.; Williams, D.A. Fibrofog in daily life: An examination of ambulatory subjective and objective cognitive function in fibromyalgia. Arthritis Care Res (Hoboken) 2020, 72, 1669–1677. [Google Scholar] [CrossRef] [PubMed]
- Metyas, C.; Aung, T.T.; Cheung, J.; Joseph, M.; Ballester, A.M.; Metyas, S. Diet and lifestyle modifications for Fibromyalgia. Curr Rheumatol Rev. 2024, 20, 405–413. [Google Scholar] [CrossRef]
- Pettersen, P.S.; Haugmark, T.; Berg, I.J.; Hammer, H.B.; Neogi, T.; Zangi, H.; et al. Pain sensitization in fibromyalgia. Cross-sectional associations between quantitative sensory testing of pain sensitization and fibromyalgia disease burden. Eur J Pain. 2025, 29. [Google Scholar] [CrossRef]
- Who.int. Available online: https://iris.who.int/bitstream/handle/10665/39524/WHO_TRS_804.pdf.
- Calandre, E.P.; Rico-Villademoros, F.; Slim, M. An update on pharmacotherapy for the treatment of fibromyalgia. Expert Opin Pharmacother. 2015, 16, 1347–1368. [Google Scholar] [CrossRef]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet. 2021, 397, 2082–2097. [Google Scholar] [CrossRef]
- Ramos, H.; Moreno, L.; Pérez-Tur, J.; Cháfer-Pericás, C.; García-Lluch, G.; Pardo, J. CRIDECO Anticholinergic Load Scale: An Updated Anticholinergic Burden Scale. Comparison with the ACB Scale in Spanish Individuals with Subjective Memory Complaints. J Pers Med. 2022, 12, 207. [Google Scholar] [CrossRef] [PubMed]
- Moran, K.M.; Calip, G.S.; Lee, T.A.; Koronkowski, M.J.; Lau, D.T.; Schumock, G.T. Risk of fall-related injury and all-cause hospitalization of select concomitant central nervous system medication prescribing in older adult persistent opioid users: A case-time-control analysis. Pharmacotherapy 2021, 41, 733–742. [Google Scholar] [CrossRef]
- Center for Drug Evaluation, Research. La FDA exige un Recuadro de advertencia actualizado para mejorar el uso seguro de los medicamentos de la clase de las benzodiazepinas. U.S. Food and Drug Administration. FDA. 2020. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/la-fda-exige-un-recuadro-de-advertencia-actualizado-para-mejorar-el-uso-seguro-de-los-medicamentos.
- Jackson, N.; Doherty, J.; Coulter, S. Neuropsychiatric complications of commonly used palliative care drugs. Postgrad Med J. 2008, 84, 121–126. [Google Scholar] [CrossRef]
- Pilgrim, J.L.; Gerostamoulos, D.; Drummer, O.H. Deaths involving contraindicated and inappropriate combinations of serotonergic drugs. Int J Legal Med. 2011, 125, 803Y815. [Google Scholar] [CrossRef]
- Bartlett, D. Drug-induced serotonin syndrome. Crit Care Nurse. 2017, 37, 49–54. [Google Scholar] [CrossRef] [PubMed]
- WHO Collaborating Centre for Drug Statistics Methodology. ATC Classification Index with DDDs, 2022. Oslo, Norway 2021. Available online: https://www.whocc.no/atc_ddd_index/.
- CheckTheMeds Technology, S.L. CheckTheMeds. Inicio. Checkthemeds.com. Available online: https://www.checkthemeds.com.
- Ser.es. Available online: https://www.ser.es/wp-content/uploads/2020/11/Recomendaciones_SER_FM_DEF.pd.
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Häuser, W.; Fluß, E.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef]
- Bair, M.J.; Krebs, E.E. Fibromyalgia. Ann Intern Med. 2020, 172, ITC33. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Biton, L.; Buskila, D.; Nissanholtz-Gannot, R. Review of fibromyalgia (FM) syndrome treatments. Int J Environ Res Public Health. 2022, 19, 12106. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, V.; Bazzichi, L.; Batticciotto, A.; Pellegrino, G.; Di Franco, M.; Sirotti, S.; et al. Fibromyalgia: One year in review 2023. Clin Exp Rheumatol. 2023, 41, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Ignacio Expósito, J.M.; Carrillo Peñas, N.; Rosety Rodríguez, M.; Lagares Franco, C. El consumo de medicamentos como factor asociado al estado de fragilidad en personas mayores de 65 años en España. Semergen. 2024, 50, 102177. [Google Scholar] [CrossRef] [PubMed]
- Coles, M.L.; Uziel, Y. Juvenile primary fibromyalgia syndrome: A Review- Treatment and Prognosis. Pediatr Rheumatol Online J. 2021, 19. [Google Scholar] [CrossRef]
- Agencia Española de Medicamentos y Productos Sanitarios. Agencia Española de Medicamentos y Productos Sanitarios. 2022. Available online: https://www.aemps.gob.es.
- Mabuchi, T.; Hosomi, K.; Yokoyama, S.; Takada, M. Polypharmacy in three different spontaneous adverse drug event databases. Int J Clin Pharmacol Ther. 2020, 58, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Lee, G.-H.; Kim, S.; Kim, S.; Kim, Y.; Choi, S.-A. Risk factors for respiratory depression associated with tramadol based on the global pharmacovigilance database (VigiBase). Pharmaceuticals (Basel) 2024, 17, 205. [Google Scholar] [CrossRef] [PubMed]
- Rawle, M.J.; Lau, W.C.Y.; Gonzalez-Izquierdo, A.; Patalay, P.; Richards, M.; Davis, D. Associations Between Midlife Anticholinergic Medication Use and Subsequent Cognitive Decline: A British Birth Cohort Study. Drugs Aging 2024. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.T.; McPherson, M.L.; Noble, B.N.; Furuno, J.P. Prevalence and characteristics of patients prescribed opioids and central nervous system depression agents on discharge to hospice care. Ann Palliat Med. 2024, 13, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Robson, M.J.; Quinlan, M.A.; Blakely, R.D. Immune system activation and depression: Roles of serotonin in the central nervous system and periphery. ACS Chem Neurosci. 2017, 8, 932–942. [Google Scholar] [CrossRef] [PubMed]
- Aemps.es. Available online: https://cima.aemps.es/cima/pdfs/es/ft/63734/63734_ft.pdf.
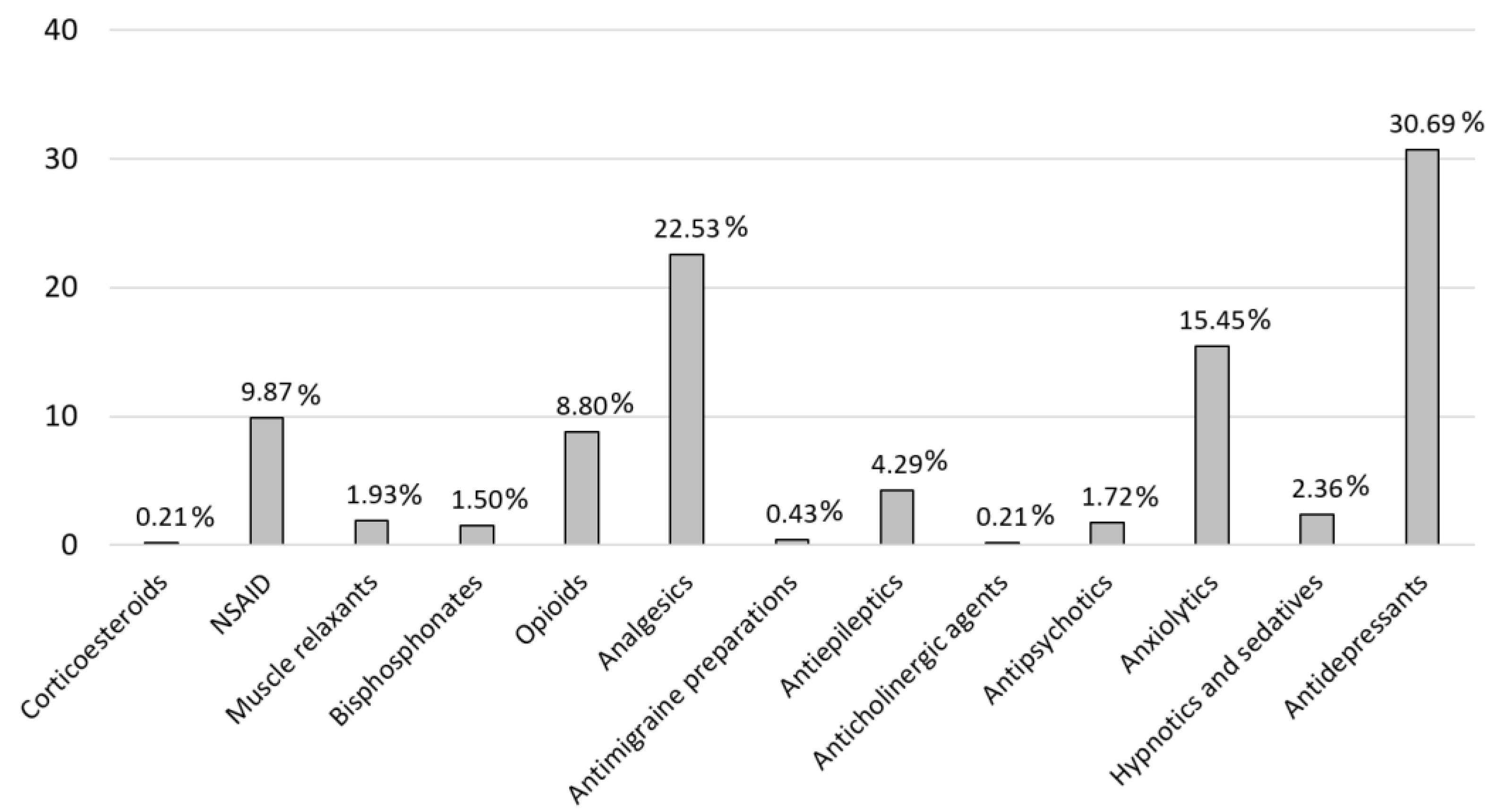
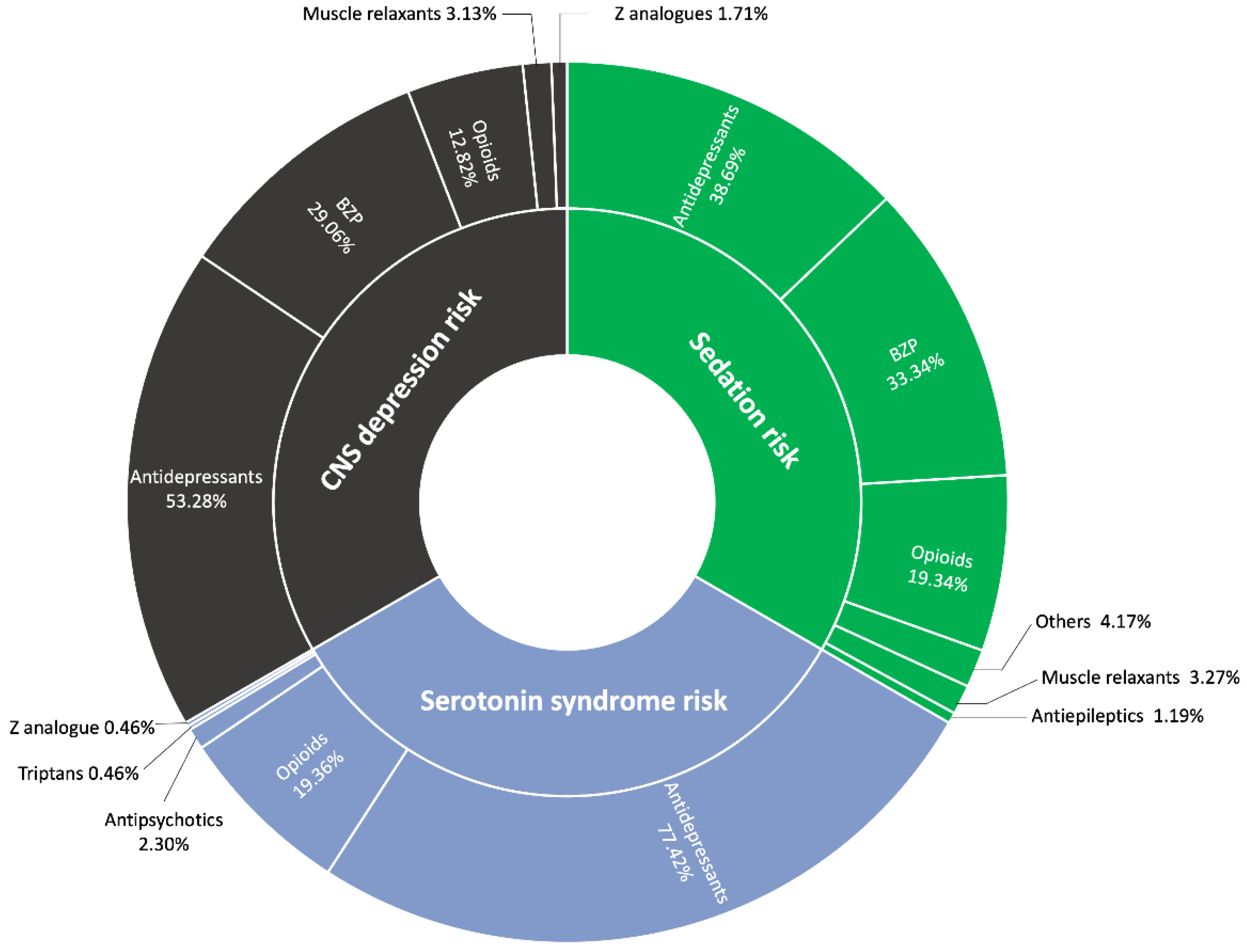
| Pharmacological Treatment | Level of Evidence | Grade | Recommendation | Agreement (%) |
|---|---|---|---|---|
| Amitriptyline (low doses) | Ia | A | Some evidence | 100 |
| Duloxetine o milnacipran | Ia | A | Some evidence | 100 |
| Pregabalin | Ia | A | Some evidence | 94 |
| Cyclobenzaprine | Ia | A | Some evidence | 75 |
| Tramadol | Ib | A | Some evidence | 100 |
| Avoid | Consider Intervention | Know and Assess | Occasional | Total | |
|---|---|---|---|---|---|
| CNS depression | 0.00% | 14.18% | 7.52% | 0.00% | 21.70% |
| Sedation | 0.00% | 14.30% | 4.81% | 1.11% | 20.22% |
| Serotoninergic syndrome | 0.00% | 10.48% | 0.86% | 0.00% | 11.34% |
| Loss of effectiveness | 0.00% | 0.00% | 2.59% | 3.82% | 6.41% |
| Toxicity | 0.00% | 1.73% | 1.97% | 1.23% | 4.93% |
| QT interval prolongation | 0.25% | 1.48% | 0.74% | 1.60% | 4.07% |
| Preventing risk of falls and fractures | 0.00% | 3.82% | 0.00% | 0.00% | 3.82% |
| Duplicity | 0.74% | 2.59% | 0.00% | 0.00% | 3.33% |
| Increased Adverse Drug Reactions | 0.00% | 1.48% | 1.60% | 0.12% | 3.21% |
| Haemorrhages | 0.37% | 1.60% | 1.11% | 0.00% | 3.08% |
| CYP2D6 | 0.00% | 0.37% | 2.47% | 0.00% | 2.84% |
| Consider reducing anticholinergic load | 0.00% | 2.71% | 0.00% | 0.00% | 2.71% |
| Therapeutic cascade | 0.00% | 2.47% | 0.00% | 0.00% | 2.47% |
| Consider alternative to antidepressants | 0.00% | 1.85% | 0.00% | 0.00% | 1.85% |
| Anticholinergic risk | 0.00% | 1.11% | 0.25% | 0.25% | 1.60% |
| Joint use of SSRIs and tricyclic | 0.00% | 1.48% | 0.00% | 0.00% | 1.48% |
| Assess need for 3 analgesics | 0.00% | 1.36% | 0.00% | 0.00% | 1.36% |
| Alternatives to clonazepam | 0.00% | 1.23% | 0.00% | 0.00% | 1.23% |
| Low blood pressure | 0.00% | 0.00% | 0.86% | 0.00% | 0.86% |
| Antagonising the anticonvulsant effect | 0.00% | 0.00% | 0.00% | 0.74% | 0.74% |
| CYP2C19 | 0.00% | 0.00% | 0.37% | 0.00% | 0.37% |
| Hepatotoxicity | 0.00% | 0.00% | 0.00% | 0.25% | 0.25% |
| NSAIDs + bisphosphonates | 0.00% | 0.00% | 0.00% | 0.12% | 0.12% |
| TOTAL | 1.36% | 64.24% | 25.15% | 9.25% | 100% |
| N | CALS for EULAR Recommendations | Number of Other Drugs Combinate | Total Cals Treatment | Percentage of Patients with CALS ≥ 3 | |
|---|---|---|---|---|---|
| Amitriptyline | N= 11 (10.19%) | 3 | 4.3 ± 1.16 | 5.4 ± 1.56 | 100% |
| Duloxetine | N= 19 (17.59%) | 0 | 3.16 ± 1.01 | 1.61 ± 0.77 | 35% |
| Tramadol | N= 7 (6.48%) | 2 | 3.57 ± 0.79 | 3.57 ± 1.13 | 85.71% |
| pregabalin | N= 6 (5.56%) | 0 | 3.83 ± 0.72 | 3 ± 0.81 | 50% |
| Amitriptyline + duloxetine | N= 5 (4.63%) | 3 | 3.4 ± 1.14 | 3.6 ± 0.55 | 100% |
| Amitriptyline + tramadol | N= 3 (2.78%) | 5 | 4.33 ± 1.53 | 6.67 ± 1.53 | 100% |
| Duloxetine + pregabalin | N= 7 (6.48%) | 0 | 4.37 ± 2.50 | 2.14 ± 3.64 | 14.29% |
| Duloxetine + tramadol | N= 12 (11.11%) | 2 | 5.08 ± 0.90 | 3.17 ± 1.34 | 75% |
| Pregabalin + tramadol | N= 4 (3.7%) | 2 | 5.5 ± 1.91 | 3.5 ± 1.29 | 75% |
| Amitriptyline + duloxetine + pregabalin | N= 1 (0.93%) | 3 | 8 | 6 | 100% |
| Tramadol + pregabalin + duloxetine | N=3 (2.78%) | 2 | 6.33 ± 2.52 | 3.67 ± 2.89 | 33.33% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




