Introduction
Radiotherapy (RT) is a cornerstone of modern cancer treatment, serving as a curative intervention in the majority of cases. RT is frequently employed, either alone or in combination with surgery and chemotherapy, to eradicate malignancies. It is also used as a palliative measure to improve quality of life in patients with advanced or incurable cancers.[
1]
Radiotherapy uses ionizing radiation (IR) to destroy cancer cells and reduce tumor size. IR is highly effective in treating a wide range of solid tumors, including skin, brain, breast, prostate, and cervical cancers, as well as hematological malignancies such as leukemia, lymphoma, and glioma (Baskar et al., 2012). However, the application of IR is limited by the development of radioresistance in neoplastic cells and radiation-induced toxicity in surrounding healthy tissues.[
2]
One of the critical adverse effects of IR is its influence on biological membranes which are composed of lipids. The degradation of phospholipids, the principal constituents of the plasma membrane, leads to structural and functional damage. The pathological spectrum of radiation-induced heart disease encompasses both acute and chronic conditions, with extensive toxic effects on almost all cardiac structures, including the pericardial sac, coronary arteries, myocardium, and heart valves.[
3] The mechanisms underlying radiation-induced cardiac damage are multifaceted and include oxidative stress, inflammation, apoptosis, fibrosis, and endothelial-mesenchymal transition (EndMT). Chronic inflammation resulting from cellular damage induced by radiotherapy triggers the accumulation of inflammatory cells and the sustained production of nitric oxide (NO) and reactive oxygen species (ROS).[
4]
To mitigate the risk of cardiac toxicity associated with IR, researchers have explored various adjuvants and antioxidant agents. Among these, natural products with antioxidant properties have demonstrated efficacy in mitigating cellular damage caused by exposure to IR. Propolis is a natural resinous substance produced by honey bees from plant resins, flowers and buds, and mixed with beeswax. Propolis has been used in folk medicine for the treatment of many health conditions due to its antioxidant, antitumor, anti-inflammatory, and antiulcer properties. The components of propolis are responsible for many of its biological and pharmacological activities and have been shown to exhibit detoxification activity against the harmful effects of free radicals.
To the best of our knowledge, the potential protective effects of propolis against radiation-induced oxidative stress have not been previously studied in the cardiac tissue. This study aimed to demonstrate the effect of propolis on cardiac toxicity induced by cranial irradiation.
Materials and Methods
Study Protocol and Experimental Animals
The experimental protocol was approved by the Institutional Review Board of Gaziantep University (Approval number: 2017/2).
Sprague-Dawley rats, weighing 250 - 400 grams were obtained from the Gaziantep University Laboratory Animal Unit. The rats were housed under standard conditions (22°C ± 2°C,12-hour light-dark cycle), with unrestricted access to water and food. All animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals, as published by the US National Institutes of Health (NIH Publications No. 8023, revised 1978).
Experimental Procedur
Thirty-two rats were randomly allocated into four groups, each containing eight rats.
Control Group: Received 1 ml of saline orally for 10 days.
Propolis Control (PC) Group: Received dimethyl sulfoxide (DMSO), in which propolis was dissolved, via oral gavage daily for 10 days.
IR Group: Exposed to 5 Gy of gamma ionizing radiation (IR) to the entire cranium and received 1 ml of saline orally for 10 days post-irradiation.
IR+ Propolis Group: Received propolis (80 mg/kg/day) via oral gavage prior to IR exposure and for 10 days post-procedure. Propolis was dissolved in (DMSO immediately before administration to the rats.
Application of Ionizing Radiation
All rats were anesthetized with 80 mg/kg of ketamine hydrochloride (Pfizer Ilac, Istanbul, Turkey) before undergoing total cranial irradiation. Gamma ionizing radiation was delivered to a 5 x 5 cm anterior field using a Cobalt-60 teletherapy unit (Picker, C9, Maryland, NY) at a source-to-surface distance of 80 cm. A single dose of 5 Gy was applied to the total cranium at a dose rate of 0.49 Gy/min, with the central axis dose calculated at a depth of 0.5 cm. The rats in the control and PC groups received sham irradiation.
Biochemical Assessments
Tissue Preparation for Analysis
All animals were euthanized via decapitation, and their cardiac tissues were excised ten days post-IR exposure. The cardiac tissues were homogenized in a physiological saline solution using an IKA-NERKE homogenizer (Staufen, Germany). The homogenate was centrifuged at 10,000 g for 1 hour, and the clear supernatant was collected for subsequent analysis. All procedures were conducted at 4 °C.
Analysis of Oxidative Stress Parameters
Lipid Hydroperoxide (LOOH)
The modified FOX2 assay method was employed to measure lipid hydroxide levels. Results are presented as μmol/g protein.[
5]
Total Oxidant Status (TOS)
TOS was quantified using a colorimetric technique that measures the cumulative oxidation of ferrous ions to ferric ions by oxidants available in the sample. TOS results are reported as μmol H2O2 equivalent per gram of protein.
Oxidative Stress Index (OSI)
The OSI was calculated using the formula: OSI (arbitrary unit, AU) = [(TOS μmol/L)/(TAS μmol/L)] x 100, after determining TOS and TAS units.
Analysis of Antioxidant Parameters
Total Antioxidant Status (TAS)
TAS was assessed using the methodology described by Erel.[
6] TAS results are presented as mmol Trolox Equivalent per liter.
Arylesterase (ARE) Activity
Arylesterase activity was determined by conducting the enzymatic reaction of phenylacetate and quantifying the resulting phenol colorimetrically. ARE results are expressed as U per gram of protein. [
7]
Ceruloplasmin (CER)
CER levels were measured using the automated colorimetric method described by Erel et al. CER results are reported as U/g protein.[
8]
Paraoxonase (PON) Activity
PON activity was assessed by quantifying the production of 4-nitrophenol through the enzymatic hydrolysis of paraoxon at 412 nm in kinetic mode.[
9] Results are presented as U per gram of protein.
Total-SH
The total sulfhydryl (thiol) groups were assessed using Ellman's method.[
10] Results are expressed as mmol per gram of protein.
Statistical Analysis
Statistical analysis was performed by using GraphPad Prism 6.0 (GraphPad Software, San Diego, CA, USA). Data are expressed as mean ± standard error of the mean (S.E.M.). One-way analysis of variance (ANOVA) was used to compare the groups and p-values less than 0.05 considered statistically significant
Results
Oxidative Stress Parameters
The oxidative stress parameters assessed are presented in
Table 1.
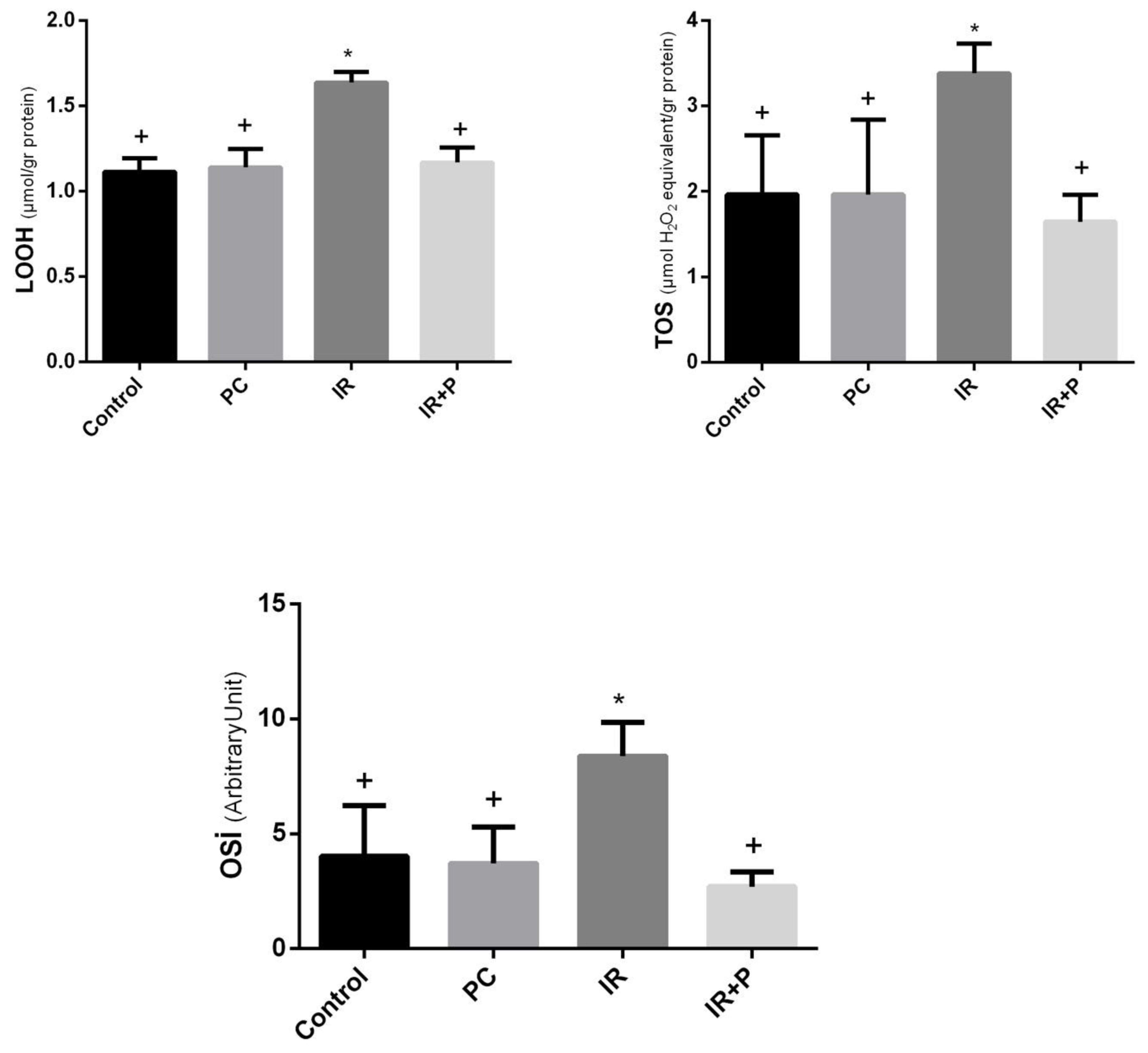
LOOH, TOS, and OSI were markedly elevated in the IR group compared to the control and PC groups (
Table 1). Treatment with propolis significantly reduced these oxidative stress parameters in the IR + propolis group in comparison to the IR group (
Figure 1).
Antioxidant Parameters
The antioxidant parameters evaluated are summarized in
Table 2.
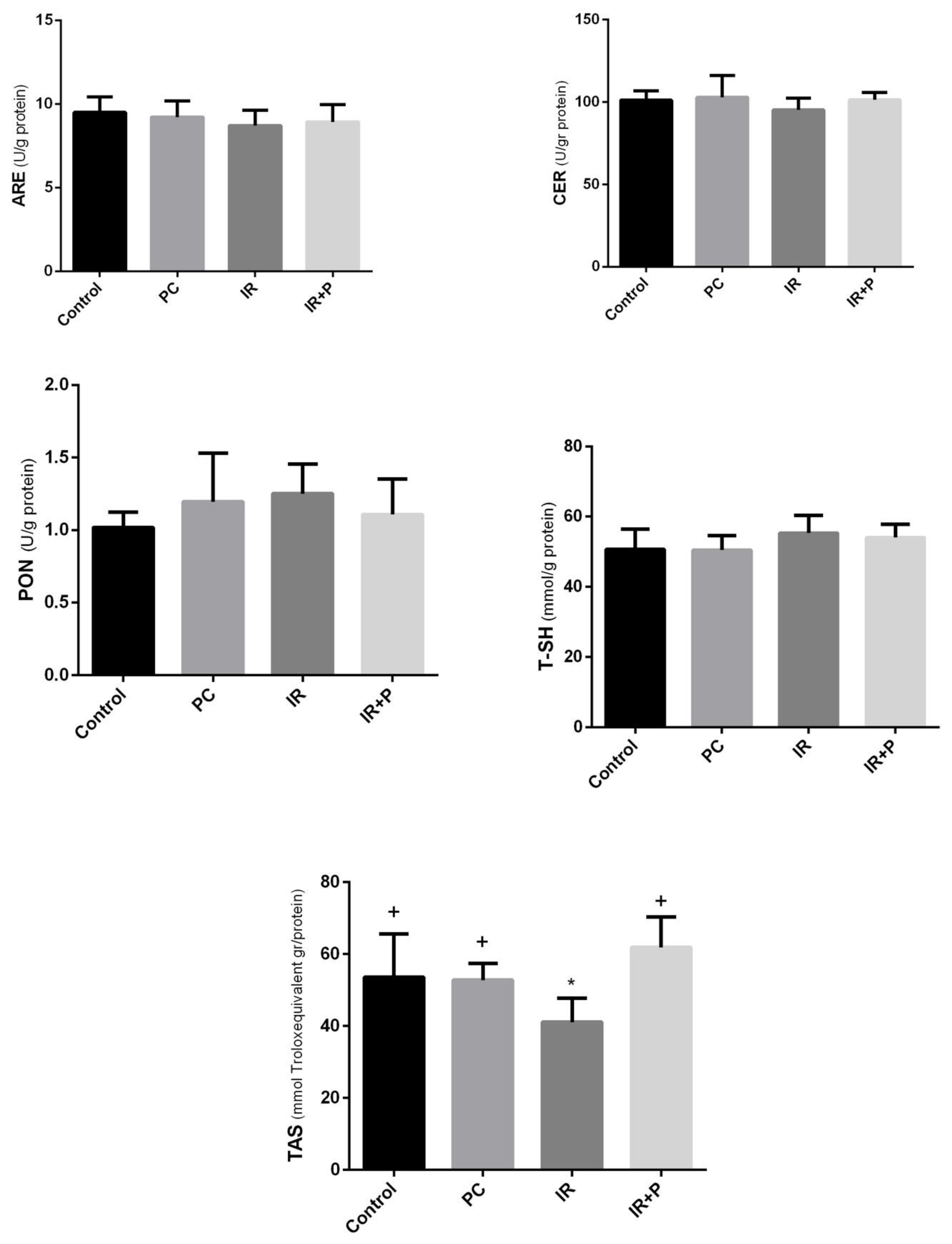
While ARE and CER levels were lower in the IR group compared to the control group, the reduction was not statistically significant. Similarly, PON and Total-SH levels were reduced in the IR group relative to the control, but the difference was also non-significant. However, TAS levels showed a significant decrease in the IR group, while treatment with propolis elevated TAS levels (
Figure 2).
Discussion
The data from the current study showed that systemic administration of propolis effectively mitigates oxidative damage in the heart caused by exposure to total head irradiation in rats.
Radiotherapy (RT) is a cornerstone in the treatment of various cancer types. As the use radiotherapy increases and survival rates improve, the associated risks must be carefully evaluated. Cardiovascular disease is the major cause of non-cancer-related death among cancer survivors.[
11] Cranial radiotherapy presents an additional risk when combined with chemotherapy, especially anthracyclines, causing long-term cardiac and metabolic complications in pediatric cancer patients.[
12]
Exposure of cells to ionizing irradiation (IR) is known to induce multiple forms of DNA damage, including double strand breaks, single strand breaks, oxidized bases, and abasic sites resulting from base loss. The harmful effects of IR primarily stem from the generation of electrons that, through interactions with water, produce free radicals. These free radicals can then damage DNA, proteins, and lipids. Direct energy deposition from radiation also contributes to cellular damage. Such interactions with biological materials are collectively referred to as targeted effects, as they exclusively involve the irradiated cells.[
13] Another critical phenomenon association with IR is the radiation-induced bystander effect, where non-irradiated cells are damaged due to their proximity to irradiated cells. The bystander effect can occur through various mechanisms. Ionizing radiation induces extensive DNA damage, triggering DNA damage response and repair mechanisms at levels exceeding those typically associated with endogenous cellular damage. The reactive nitrogen species (RNS) and ROS generated by irradiated cells may be transferred to bystander cells through gap junctional intercellular communication (GJIC) or be released extracellularly in a NO-dependent fashion. Moreover, inflammatory cytokines secreted by irradiated cells can bind to bystander cells through their membrane receptors, activating specific signaling pathways, such as Janus kinase/ signal transducer and activator of transcription (JAK2/STAT3), nuclear factor kappa B (NF-κB), and mitogen-activated protein kinase (MAPK). These pathways ultimately lead to the production of cytokines and the expression of cyclooxygenase-2 (COX-2) and nitric oxide synthase (NOS). Additionally, irradiated cells undergoing apoptosis due to extensive DNA damage may release danger' signals to adjacent cells, which seem to play an integral role in disseminating the stress systemically and to distant sites (systemic response).[
14]
Thus, oxidative stress factors act as key mediators of the direct effects of radiotherapy on cells, as well as the propagation of effects in tissues beyond the targeted region. This oxidative damage contributes to the potential long-term adverse effects observed in cancer survivors.
We observed that oxidative stress markers, LOOH, TOS, and OSI, were markedly increased in cardiac tissue following cranial radiotherapy in rats. This suggests that the bystander effect is also present in cardiac tissue. Propolis is a naturally occurring resinous substance produced by honey bees from a variety of botanical materials, floral elements, and exudates.[
15] One of the key properties of propolis is its antioxidant activity. The antioxidant properties of propolis may be associated with specific biological functions, including chemoprevention. The flavonoids in propolis are potent free radical scavengers, safeguarding against lipid peroxidation and serving as efficient antioxidants for the cell membrane.[
16]
Propolis samples from Europe, South America and Asia have different chemical compositions as a result of the geographical differences.[
17,
18] Flavonoids and phenolic compounds are the main components of propolis. The studies stated that the strong antioxidant activity of propolis is due to compounds such as caffeic acid, ferulic acid and caffeic acid phenethyl ester.[
19,
20]
In a recent study, it was showed that propolis samples, collected from four different regions of Turkey contained high amounts of flavonoids, phenolic acids and caffeoylquinic acids showing a wide range of biological activities and most of these samples exhibited similar phenolic profiles.[
21] These compounds in the content of propolis samples obtained from Turkey have been shown to show antioxidant activity in various studies.[
22,
23]
Chrysin (5,7-dihydroxyflavone), a flavonoid found in propolis, honey, and various plants, has been shown to possess antioxidant activity and reduce oxidative stress. [
24,
25]
The findings of the current study showed that administering propolis after radiotherapy reduced all oxidative stress parameters to levels similar to those observed before radiotherapy, demonstrating the antioxidant effectiveness of propolis. Research on the bystander effect in cardiac tissue is limited.
In parallel with our findings, Barary reported that propolis treatment significantly improved cardiotoxicity induced by 5-fluorouracil in rats by reducing oxidative stress.[
26] In another study, the protective effects of Chinese propolis and its active compounds against hydrogen peroxide-induced oxidative stress in cardiac H9c2 cells were demonstrated.[
27]
In the present study, the TAS, which reflects the body's overall antioxidant capacity, decreased following radiotherapy in cardiac tissue but returned to baseline levels with propolis treatment. Supporting our data, Barary et al. also showed an increase in TAS in the heart tissues of the rats with 5-fluorouracil-induced cardiac toxicity. In addition to TAS, the antioxidant enzymes, ARE, CER, PON, and Total-SH were also reduced after cranial radiotherapy, though these changes were not statistically significant. Consequently, although the levels of these enzymes remained relatively unchanged in the cardiac tissue after radiotherapy, the alterations in TAS, which may serve as a marker for total antioxidant balance, suggests the potential involvement of other antioxidant enzymes and molecules in this context.
The OSI (TOS/TAS) represents a more precise measure of oxidative stress in the body by evaluating both TAS and TOS comprehensively.
The major strength of this study is that, as an animal experiment, it is free from confounding variables, such as comorbidities, commonly affecting humans. Furthermore, the direct evaluation of cardiac tissue for oxidative damage adds to the robustness of the findings. However, the absence of histological assessment is a notable limitation. The study, designed as an animal experiment, was limited by a small sample size. Therefore, conducting similar studies with larger animal cohorts would improve the accuracy and generalizability of the findings.
Conclusion
This animal study showed that, as a distant organ, the heart is also affected by oxidative stress caused by cranial irradiation, and that propolis mitigates oxidative damage by reducing oxidant levels and enhancing antioxidant activity. Propolis may serve as a potential cardioprotective agent for patients undergoing radiotherapy. However, further research involving human subjects is warranted to corroborate these findings and evaluate the clinical utility of propolis as a radioprotective agent.
Author Contributions
I.V.D. methodology and conceptualization writing - original draft, writing - review & editing, M.D.S Visualization, A.Y and S.T. evaluation of blood tests. All authors read and approved the final manuscript.
Funding
All contributing authors declared that they received no financial support.
Institutional Review Board Statement
The experimental protocol was approved by the Institutional Review Board of Gaziantep University (Approval number: 2017/2), which is a member of HADYEK, whose directive was approved by HADMEK within the scope of the Regulation on the Working Procedures and Principles of Animal Experiments Ethics Committees dated 15 February 2014 and numbered 28914.
Availability of Data and Materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
Not applicable.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- Alfouzan, A.F. Radiation Therapy in Head and Neck Cancer. Saudi medical journal 2021, 42, 247. [Google Scholar] [CrossRef]
- Nambiar, D.; Rajamani, P.; Singh, R.P. Effects of Phytochemicals on Ionization Radiation-Mediated Carcinogenesis and Cancer Therapy. Mutation Research/Reviews in Mutation Research 2011, 728, 139–157. [Google Scholar] [CrossRef] [PubMed]
- Filopei, J.; Frishman, W. Radiation-Induced Heart Disease. Cardiology in review 2012, 20, 184–188. [Google Scholar] [CrossRef]
- Singh, K.; Alameri, A.; Ali Hamza, A.; E. Al-Gazally, M.; Temurovich Islomov, S.; Fadhel Obaid, R.; Alexis Ramírez-Coronel, A.; Abosaooda, M.; Yahyapour, R.; Najafi, M. Cardiac Injury Following Chemo/Radiation Therapy: An Updated Reviewon Mechanisms and Therapeutic Approaches. CRP 2023, 16, 185–203. [Google Scholar] [CrossRef]
- Nourooz-Zadeh, J. Ferrous Ion Oxidation in Presence of Xylenol Orange for Detection of Lipid Hydroperoxides in Plasma. In Methods in enzymology; Elsevier, 1999; Vol. 300, pp. 58–62.
- Erel, O. A Novel Automated Direct Measurement Method for Total Antioxidant Capacity Using a New Generation, More Stable ABTS Radical Cation. Clinical biochemistry 2004, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Haagen, L.; Brock, A. A New Automated Method for Phenotyping Arylesterase (EC 3.1.1.2) Based Upon Inhibition of Enzymatic Hydrolysis of 4-Nitrophenyl Acetate by Phenyl Acetate. Clinical Chemistry and Laboratory Medicine 1992, 30. [Google Scholar] [CrossRef]
- Erel, O. Automated Measurement of Serum Ferroxidase Activity. Clinical chemistry 1998, 44, 2313–2319. [Google Scholar] [CrossRef]
- Eckerson, H.W.; Wyte, C.M.; La Du, B.N. The Human Serum Paraoxonase/Arylesterase Polymorphism. American journal of human genetics 1983, 35, 1126. [Google Scholar] [PubMed]
- Ellman, G.L. Reprint of: Tissue Sulfhydryl Groups. Archives of Biochemistry and Biophysics 2022, 726, 109245. [Google Scholar] [CrossRef]
- Armanious, M.A.; Mohammadi, H.; Khodor, S.; Oliver, D.E.; Johnstone, P.A.; Fradley, M.G. Cardiovascular Effects of Radiation Therapy. Current problems in cancer 2018, 42, 433–442. [Google Scholar] [CrossRef]
- Landy, D.C.; Miller, T.L.; Lipsitz, S.R.; Lopez-Mitnik, G.; Hinkle, A.S.; Constine, L.S.; Adams, M.J.; Lipshultz, S.E. Cranial Irradiation as an Additional Risk Factor for Anthracycline Cardiotoxicity in Childhood Cancer Survivors: An Analysis from the Cardiac Risk Factors in Childhood Cancer Survivors Study. Pediatr Cardiol 2013, 34, 826–834. [Google Scholar] [CrossRef]
- Siva, S.; MacManus, M.P.; Martin, R.F.; Martin, O.A. Abscopal Effects of Radiation Therapy: A Clinical Review for the Radiobiologist. Cancer Letters 2015, 356, 82–90. [Google Scholar] [CrossRef]
- Nikitaki, Z.; Mavragani, I.V.; Laskaratou, D.A.; Gika, V.; Moskvin, V.P.; Theofilatos, K.; Vougas, K.; Stewart, R.D.; Georgakilas, A.G. Systemic Mechanisms and Effects of Ionizing Radiation: A New⿿ Old⿿ Paradigm of How the Bystanders and Distant Can Become the Players. In Proceedings of the Seminars in cancer biology; Elsevier, 2016; Vol. 37; pp. 77–95. [Google Scholar]
- Braakhuis, A. Evidence on the Health Benefits of Supplemental Propolis. Nutrients 2019, 11, 2705. [Google Scholar] [CrossRef] [PubMed]
- Chavda, V.P.; Vuppu, S.; Balar, P.C.; Mishra, T.; Bezbaruah, R.; Teli, D.; Sharma, N.; Alom, S. Propolis in the Management of Cardiovascular Disease. International Journal of Biological Macromolecules 2024, 131219. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.-R.; Kumazawa, S.; Hamasaka, T.; Bang, K.-S.; Nakayama, T. Antioxidant Activity and Constituents of Propolis Collected in Various Areas of Korea. J Agric Food Chem 2004, 52, 7286–7292. [Google Scholar] [CrossRef]
- Velikova, M.; Bankova, V.; Sorkun, K.; Houcine, S.; Tsvetkova, I.; Kujumgiev, A. Propolis from the Mediterranean Region: Chemical Composition and Antimicrobial Activity. Z Naturforsch C J Biosci 2000, 55, 790–793. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.K.; Alencar, S.M.; Aguiar, C.L. Botanical Origin and Chemical Composition of Brazilian Propolis. J Agric Food Chem 2002, 50, 2502–2506. [Google Scholar] [CrossRef]
- Medić-Sarić, M.; Rastija, V.; Bojić, M.; Males, Z. From Functional Food to Medicinal Product: Systematic Approach in Analysis of Polyphenolics from Propolis and Wine. Nutr J 2009, 8, 33. [Google Scholar] [CrossRef]
- Bozkuş, T.N.; Değer, O.; Yaşar, A. Chemical Characterization of Water and Ethanolic Extracts of Turkish Propolis by HPLC-DAD and GC-MS. Journal of Liquid Chromatography & Related Technologies 2021, 44, 77–86. [Google Scholar] [CrossRef]
- Kanbur, M.; Eraslan, G.; Silici, S. Antioxidant Effect of Propolis against Exposure to Propetamphos in Rats. Ecotoxicology and Environmental Safety 2009, 72, 909–915. [Google Scholar] [CrossRef]
- Eraslan, G.; Kanbur, M.; Silici, S. Evaluation of Propolis Effects on Some Biochemical Parameters in Rats Treated with Sodium Fluoride. Pesticide Biochemistry and Physiology 2007, 88, 273–283. [Google Scholar] [CrossRef]
- Moghadam, E.R.; Ang, H.L.; Asnaf, S.E.; Zabolian, A.; Saleki, H.; Yavari, M.; Esmaeili, H.; Zarrabi, A.; Ashrafizadeh, M.; Kumar, A.P. Broad-Spectrum Preclinical Antitumor Activity of Chrysin: Current Trends and Future Perspectives. Biomolecules 2020, 10, 1374. [Google Scholar] [CrossRef] [PubMed]
- Mani, R.; Natesan, V. Chrysin: Sources, Beneficial Pharmacological Activities, and Molecular Mechanism of Action. Phytochemistry 2018, 145, 187–196. [Google Scholar] [CrossRef]
- Barary, M.; Hosseinzadeh, R.; Kazemi, S.; Liang, J.J.; Mansoori, R.; Sio, T.T.; Hosseini, M.; Moghadamnia, A.A. The Effect of Propolis on 5-Fluorouracil-Induced Cardiac Toxicity in Rats. Scientific Reports 2022, 12, 8661. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, K.; Xu, X.; Ge, M.; Chen, Y.; Hu, F. Citation for: Potential Protective Effects of Bioactive Constituents from Chinese Propolis against Acute Oxidative Stress Induced by Hydrogen Peroxide in Cardiac H9c2 Cells. Wiley Online Library.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).