Submitted:
28 January 2025
Posted:
29 January 2025
You are already at the latest version
Abstract
Background/Objectives: This study aimed to determine whether interim PET/CT (iP-ET) scans could identify follicular lymphoma (FL) patients at high risk of relapse fol-lowing first-line therapy. Additionally, the potential of cell-free DNA (cfDNA) analysis to complement iPET in predicting outcomes was explored. Methods: A total of 121 FL patients who underwent iPET scans were included, with responses interpreted using the Deauville score (DS). Progression-free survival (PFS) was evaluated over a median follow-up of 34 months. Interim cfDNA data were analyzed for 14 patients to assess its potential for detecting false-positive PET results. Results: Overall, 34% of patients were classified as iPET(+), with significantly worse estimated 5-year PFS compared to iPET(-) patients (29% vs. 72%, hazard ratio 4.31, p < 0.001). Multivariate analysis confirmed iPET(+) as an independent predictor of PFS. Rituximab maintenance was predictive of reduced progression within the iPET(+) group but not among iPET(-) patients. cfDNA analysis identified two false-positive iPET cases and showed potential to identify pa-tients with complete response at risk of early progression. Conclusions: Interim PET results are significant predictors of PFS in FL first-line therapy and could inform re-sponse-adapted treatment strategies. cfDNA analysis has the potential to complement PET/CT by improving specificity and identifying patients at risk of early progression, offering a more precise approach to managing FL.
Keywords:
1. Introduction
2. Materials and Methods
Patients
Analysis of PET/CT Imaging
Baseline Genotyping and iMRD
Statistics
3. Results
3.1. Patient Characteristics
3.2. Risk Factors for Progression-Free Survival Analysis
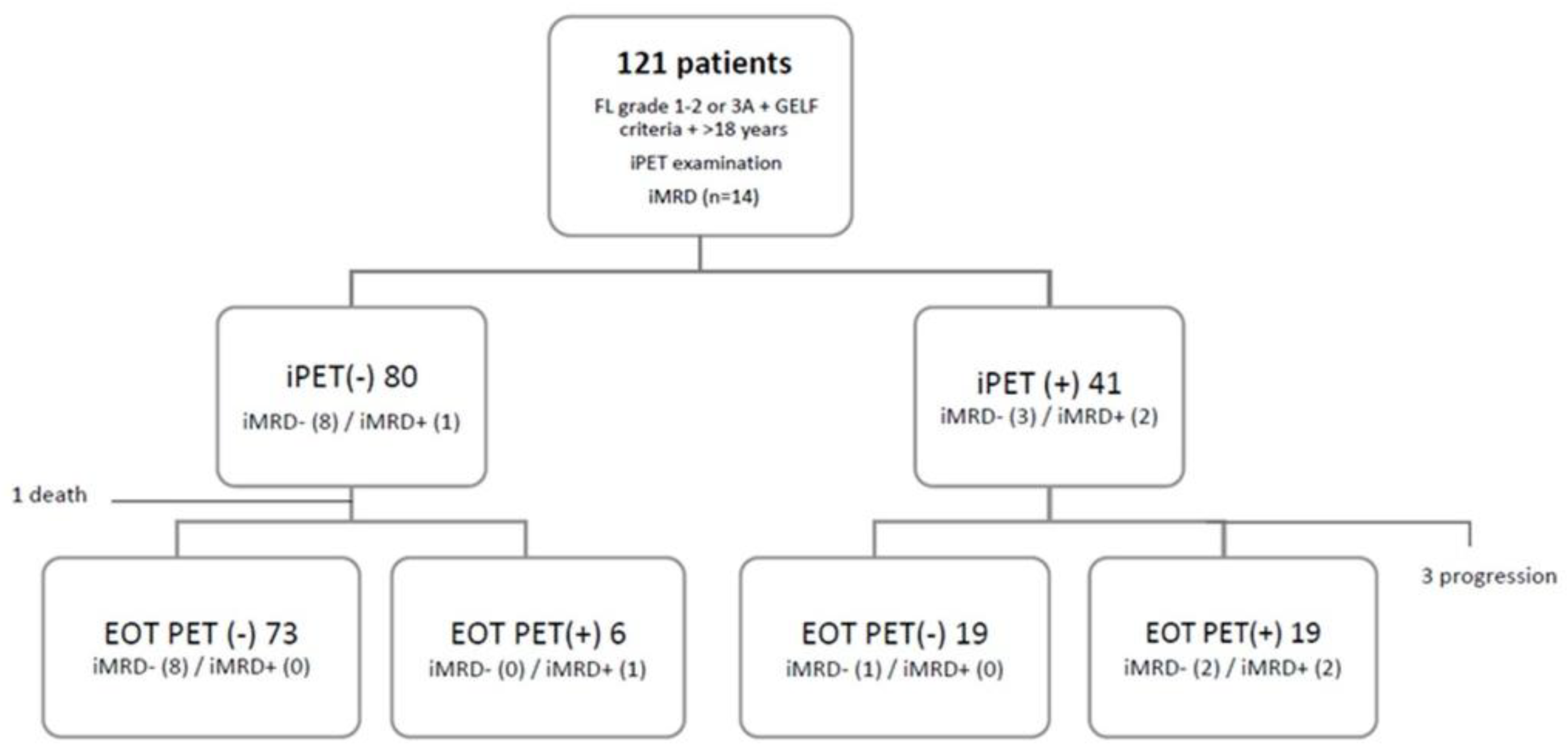
3.3. PET Analysis
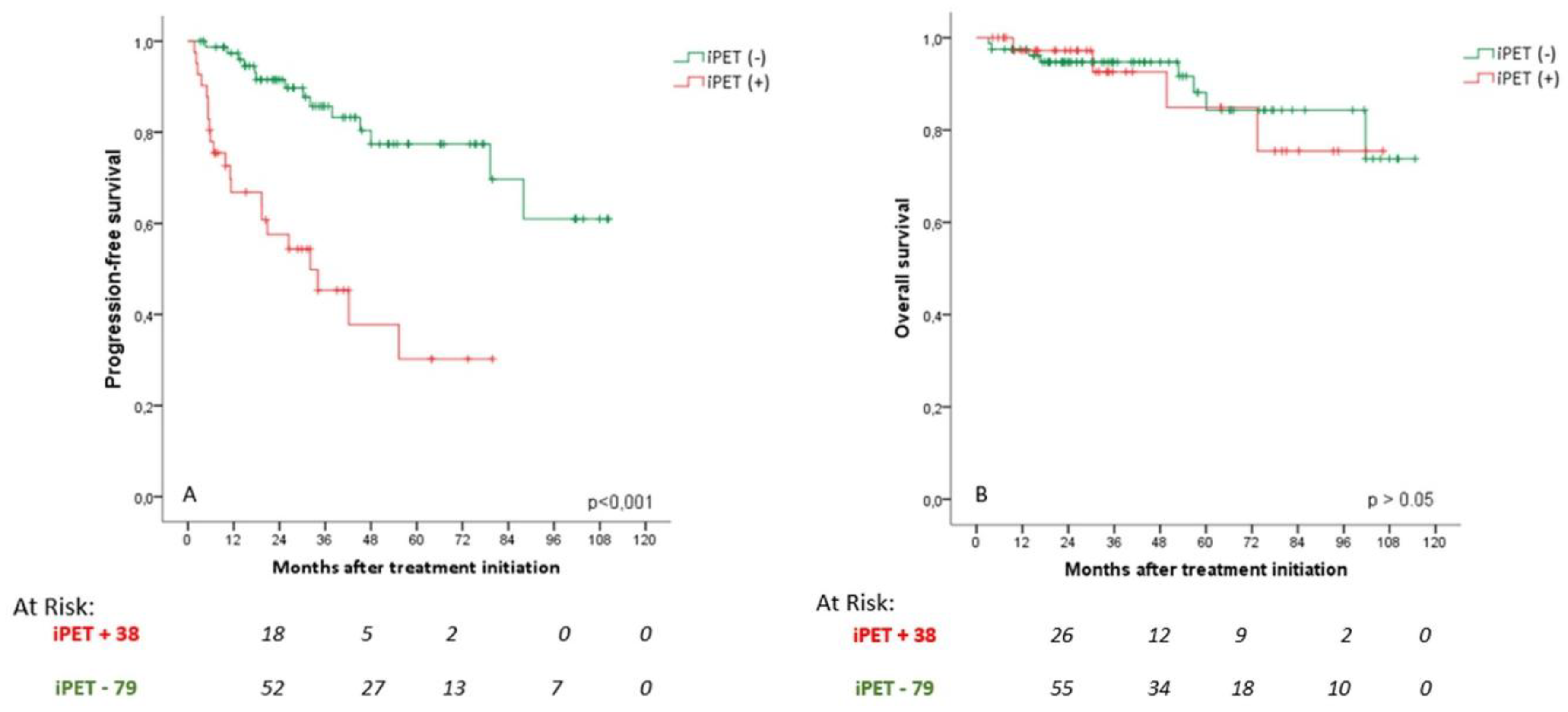
3.4. Prognostic Value of iPET
3.5. Prognostic Value of EOT PET
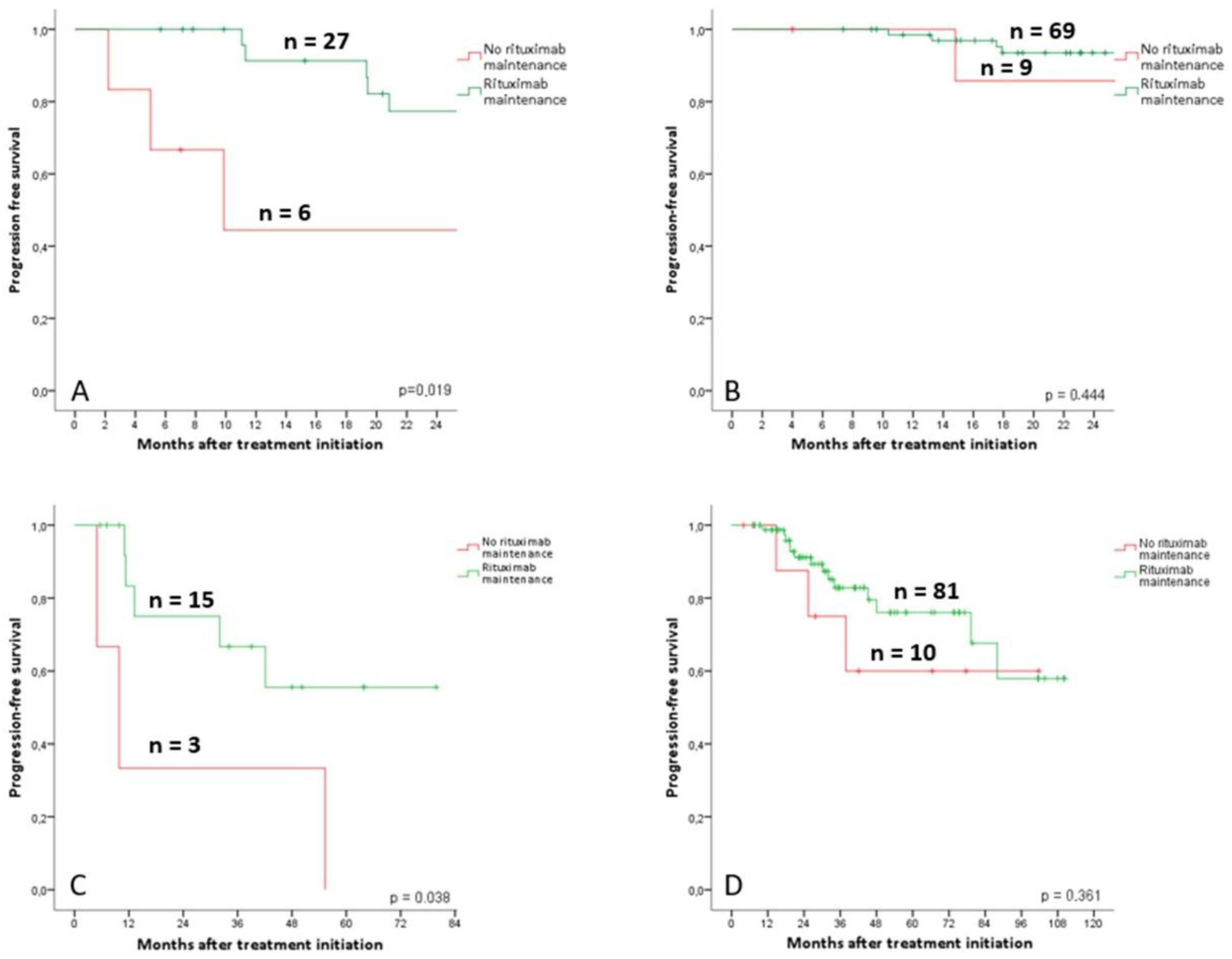
3.6. Rituximab Maintenance Value According to PET Response
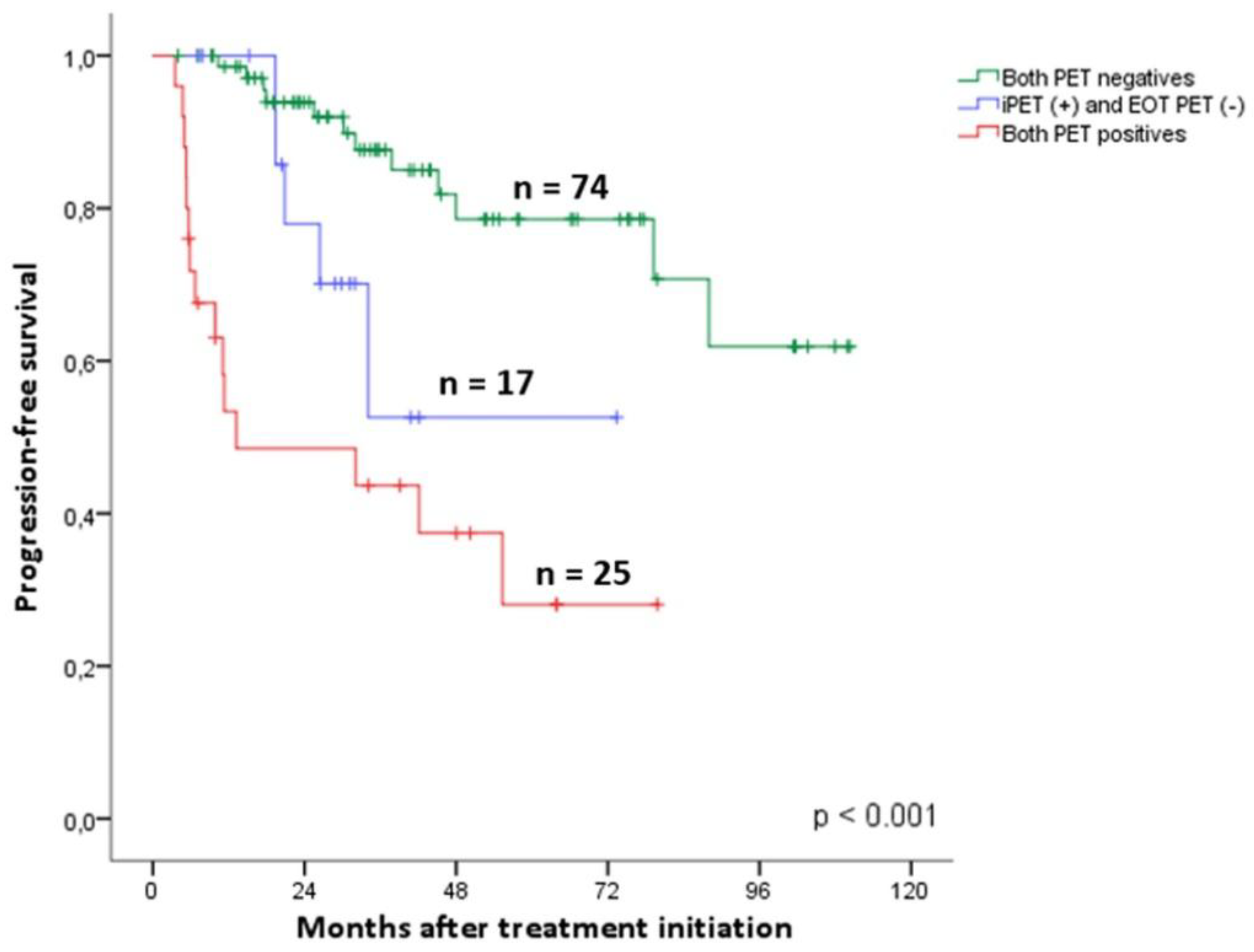
3.7. Dynamics of PET Analysis
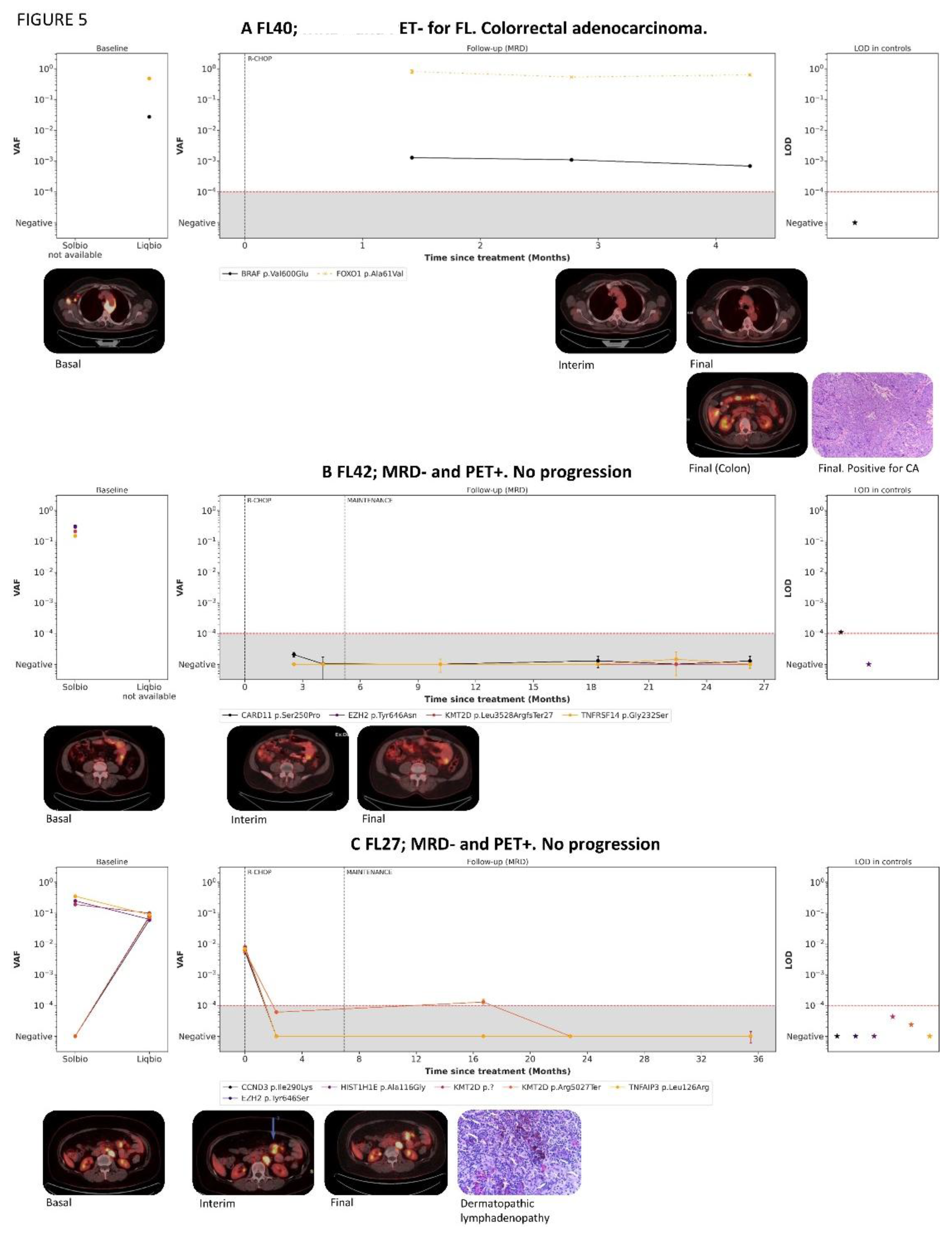
3.8. cfDNA Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cheah CY, Chihara D, Ahmed M, Davis RE, Nastoupil LJ, Phansalkar K, et al. Factors influencing outcome in advanced stage, low-grade follicular lymphoma treated at MD Anderson Cancer Center in the rituximab era. Ann Oncol 2016;27:895–901. [CrossRef]
- Jiménez-Ubieto A, Grande C, Caballero D, Yáñez L, Novelli S, Hernández-Garcia MT, et al. Autologous stem cell transplantation for follicular lymphoma: favorable long-term survival irrespective of pretransplantation rituximab exposure. Biol Blood Marrow Transplant 2017;23:1631–40. [CrossRef]
- Anderson JR, Armitage JO, Weisenburger DD. Epidemiology of the non-Hodgkin’s lymphomas: Distributions of the major subtypes differ by geographic locations. Ann Oncol 1998;9(7):717-20. [CrossRef]
- Bastos-Oreiro M, Muntañola A, Panizo C, González-Barca E, Villambrosia S, Córdoba R, et al. RELINF: prospective epidemiological registry of lymphoid neoplasms in Spain. A project from the GELTAMO group. Ann Hematol 2020;99(4):799-808. [CrossRef]
- Flinn IW, van der Jagt R, Kahl BS, Wood P, Hawkins TE, Macdonald D, et al. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood 2014; 123: 2944–2952. [CrossRef]
- Hiddemann W, Kneba M, Dreyling M, Schmitz N, Lengfelder E, Schmits R, et al. Frontline therapy with rituximab added to the combination of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) significantly improves the outcome for patients with advanced-stage follicular lymphoma compared with therapy with CHOP alone: results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood 2005;106(12):3725-32. [CrossRef]
- Morschhauser F, Fowler NH, Feugier P, Bouabdallah R, Tilly H, Palomba ML, et al. Rituximab plus lenalidomide in advanced untreated follicular lymphoma. N Engl J Med 2018;379: 934–947. [CrossRef]
- Marcus R, Davies A, Ando K, Klapper A, Opat S, Owen C, et al. Obinutuzumab for the first-line treatment of follicular lymphoma. N Engl J Med 2017; 377:1331–1344. [CrossRef]
- Bachy E, Seymour JF, Feugier P, Offner F, López-Guillermo A, Belada D, et al. Sustained Progression-Free Survival Benefit of Rituximab Maintenance in Patients With Follicular Lymphoma: Long-Term Results of the PRIMA Study. J Clin Oncol 2019;37(31):2815-2824. [CrossRef]
- Salles G, Seymour JF, Offner F, López-Guillermo A, Velada D, Xerri L, et al. Rituximab maintenance for 2 years in patients with high tumour burden follicular lymphoma responding to rituximab pluschemotherapy (PRIMA): A phase 3, randomised controlled trial. Lancet 2011;377:42-51.
- Marcus R, Davies A, Ando K, Klapper W, Opat S, Owen C, et al. Obinutuzumab for the First-Line Treatment of Follicular Lymphoma. N Engl J Med 2017;377(14):1331-1344. [CrossRef]
- Jiménez-Ubieto A, Grande C, Caballero D, Yáñez L, Novelli S, Hernández MT, et al. Progression-free survival at 2 years post-autologous transplant: a surrogate endpoint for overall survival in follicular lymphoma. Cancer Med 2017;6:2766–74. [CrossRef]
- Casulo C, Dixon JG, Le-Rademacher J, Hoster E, Hochster H, Hiddemann W, et al. Validation of POD24 as a robust early clinical endpoint of poor survival in FL from 5225 patients on 13 clinical trials. Blood 2022;139:1684–93. [CrossRef]
- Murakami S, Kato H, Higuchi Y, Yamamoto K, Yamamoto H, Saito T, et al. Prediction of high risk for death in patients with follicular lymphoma receiving rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone in first-line chemotherapy. Ann Hematol 2016;95(8):1259-69. [CrossRef]
- Zhang L, Ghielmini M, Cheson BD, Ujjani C. Pros and cons of rituximab maintenance in follicular lymphoma. Cancer Treat Rev 2017;58:34-40. [CrossRef]
- Federico M, Bellei M, Marcheselli L, Luminari S, Lopez-Guillermo A, Vitolo U, et al. Follicular Lymphoma International Prognostic Index 2: A New Prognostic Index for Follicular Lymphoma Developed by the International Follicular Lymphoma Prognostic Factor Project. J Clin Oncol 2009;27(27):4555-62. [CrossRef]
- Solal-Céligny P, Roy P, Colombat P, White J, Armitage JO, Arranz-Saez R, et al. Follicular Lymphoma International Prognostic Index. Blood 2004;104(5):1258-65. [CrossRef]
- Bachy E, Maurer MJ, Habermann TM, Gelas-Dore B, Maucort-Boulch D, Estell JA, et al. A simplified scoring system in de novo follicular lymphoma treated initially with immunochemotherapy. Blood 2018;132(1):49-58. [CrossRef]
- Mondello P, Fama A, Larson MC, Feldman AL, Villasboas JC, Yang ZZ, et al. Lack of intrafollicular memory CD4 + T cells is predictive of early clinical failure in newly diagnosed follicular lymphoma. Blood Cancer 2021;11(7):130. [CrossRef]
- Mir F, Mattiello F, Grigg A, Herold M, Hiddemann W, Marcus R, et al. Follicular Lymphoma Evaluation Index ( FLEX ): A new clinical prognostic model that is superior to existing risk scores for predicting progression-free survival and early treatment failure after frontline immunochemotherapy. Am J Hematol 2020;95(12):1503-10. [CrossRef]
- Pastore A, Jurinovic V, Kridel R, Hoster E, Staiger AM, Szczepanowski M, et al. Integration of gene mutations in risk prognostication for patients receiving first-line immunochemotherapy for follicular lymphoma: a retrospective analysis of a prospective clinical trial and validation in a population-based registry. Lancet Oncol 2015;16(9):1111-22. [CrossRef]
- Jurinovic V, Kridel R, Staiger AM, Szczepanowski M, Horn H, Dreyling MH, et al. Clinicogenetic risk models predict early progression of follicular lymphoma after first-line immunochemotherapy. Blood 2016;128(8):1112-20. [CrossRef]
- Huet S, Tesson B, Jais JP, Feldman AL, Magnano L, Thomas E, et al. A gene expression profiling score for prediction of outcome in patients with follicular lymphoma: a retrospective training and validation analysis in three international cohorts. Lancet Oncol 2018;19:549–61. [CrossRef]
- Meignan M, Cottereau AS, Versari A, Chartier L, Dupuis J, Boussetta S, et al. Baseline Metabolic Tumor Volume Predicts Outcome in High–Tumor-Burden Follicular Lymphoma: A Pooled Analysis of Three Multicenter Studies. J Clin Oncol 2016;34(30):3618-26. [CrossRef]
- Trotman J, Barrington SF, Belada D, meignan M, MacEwan R, Owen C, et al. PET investigators from the GALLIUM study. Prognostic value of end-of-induction PET response after first-line immunochemotherapy for follicular lymphoma (GALLIUM): secondary analysis of a randomised, phase 3 trial. Lancet Oncol 2018;19:1530–1542. [CrossRef]
- Dupuis J, Berriolo-Riedinger A, Julian A, Brice P, Tychyj-Pinel C, Tilly H, et al. Impact of [(18)F]fluorodeoxyglucose positron emission tomography response evaluation in patients with high-tumor burden follicular lymphoma treated with immunochemotherapy: a prospective study from the Groupe d’Etudes des Lymphomes de l’Adulte and GOELAMS. J Clin Oncol 2012;30(35):4317-22. [CrossRef]
- Galimberti S, Luminari S, Ciabatti E, Grassi S, Guerrini F, Dondi A, et al. Minimal residual disease after conventional treatment significantly impacts on progression-free survival of patients with follicular lymphoma: the FIL FOLL05 trial. Clin Cancer Res 2014;20:6398–405. [CrossRef]
- Levavi H, Lancman G, Gabrilove J. Impact of rituximab on COVID-19 outcomes. Ann Hematol. 2021;100(11):2805-2812. [CrossRef]
- Trotman J, Presgrave P, Carradice DP, Lenton DS, Gandhi MK, Cochrane T, et al. Lenalidomide Consolidation Added to Rituximab Maintenance Therapy in Patients Remaining PET Positive After Treatment for Relapsed Follicular Lymphoma: A Phase 2 Australasian Leukaemia & Lymphoma Group NHL26 Study. Hemasphere 2023;7(3):e836. [CrossRef]
- Luminari S, Manni M, Galimberti S, Versari A, Tucci A, Boccomini C, et al.Response-adapted postinduction strategy in patients with advanced-stage follicular lymphoma: the FOLL12 study. J Clin Oncol 2022;40:729–39. [CrossRef]
- Johnson P, Federico M, Kirkwood, Fossa A, Berkahn L, Carella A, et al. Adapted treatment guided by interim PET-CT scan in advanced Hodgkin’s lymphoma. N Engl J Med 2016;374:2419–2429. [CrossRef]
- Borchmann P, Goergen H, Kobe C, Lohri A, Greil R, Eichenauer DA, et al. PET-guided treatment in patients with advanced-stage Hodgkin’s lymphoma (HD18): final results of an open-label, international, randomised phase 3 trial by the German Hodgkin Study Group. Lancet 2017;390:2790–2802.
- Casasnovas R-O, Bouabdallah R, Brice P, Lazarovici J, Ghesquieres H, Stamatoullas A, et al. PET-adapted treatment for newly diagnosed advanced Hodgkin lymphoma (AHL2011): a randomised, multicentre, non-inferiority, phase 3 study. Lancet Oncol 2019;20:202–215. [CrossRef]
- Dührsen U, Müller S, Hertenstein B, Thomssen H, Kotzerke J, Mesters R, et al. Positron emission tomography-guided therapy of aggressive non-Hodgkin lymphomas (PETAL): a multicenter, randomized phase III trial. J Clin Oncol 2018;36:2024–2034. [CrossRef]
- Boo SH, O JH, Kwon SJ, Yoo IR, Kim SH, Park GS, et al. Predictive value of interim and end-oftherapy 18F-FDG PET/CT in patients with follicular lymphoma. Nucl Med Mol Imaging 2019;53:263–269. [CrossRef]
- Dupuis J, Berriolo-Riedinger A, Julian A, Brice P, Tychyj-Pinel C, Tilly H, et al. Impact of [18 F] fluorodeoxyglucose positron emission tomography response evaluation in patients with high–tumor burden follicular lymphoma treated with immunochemotherapy: a prospective study from the Groupe d’Etudes des Lymphomes de l’Adulte and GOELAMS. J Clin Oncol 2012;30:4317–4322. [CrossRef]
- Merryman RW, Michaud L, Redd R, Mondello P, Park H, Spilberg G, et al. Interim Positron Emission Tomography During Frontline Chemoimmunotherapy for Follicular Lymphoma. Hemasphere 2023;7(2):e826. [CrossRef]
- Bishu S, Quigley JM, Bishu SR, Olsasky SM, Stem RA, Shostrom VK, et al. Predictive value and diagnostic accuracy of F-18-fluoro-deoxy-glucose positron emission tomography treated grade 1 and 2 follicular lymphoma. Leuk Lymphoma 2007;48:1548–1555. [CrossRef]
- Lu Z, Lin M, Downe P, et al. The prognostic value of mid- and post-treatment [(18)F]fluorodeoxyglucose (FDG) positron emission tomography (PET) in indolent follicular lymphoma. Ann Nucl Med. 2014;28:805–811. 22. Zhou Y, Zhao Z, Li J, et al. [CrossRef]
- Jiménez-Ubieto A, Poza M, Martin-Muñoz A, Ruiz-Heredia Y, Dorado S, Figaredo G, et al. Real-life disease monitoring in follicular lymphoma patients using liquid biopsy ultra-deep sequencing and PET/CT. Leukemia. 2023;37(3):659-669. [CrossRef]
- Fernández-Miranda I, Pedrosa L, Llanos M, Franco FF, Gómez S, Martín-Acosta P, et al. Monitoring of Circulating Tumor DNA Predicts Response to Treatment and Early Progression in Follicular Lymphoma: Results of a Prospective Pilot Study. Clin Cancer Res. 2023;29(1):209-220. [CrossRef]
- Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32(27):3059-3068. [CrossRef]
- Kurch L, Hüttmann A, Georgi TW, Rekowski J, Sabri O, Schmitz C, et al. Interim PET in Diffuse Large B-Cell Lymphoma. J Nucl Med. 2021;62(8):1068-1074. [CrossRef]
- Barrington SF, Mir F, El-Galaly TC, Knapp A, Nielsen TG, Sahin D, et al. Follicular Lymphoma Treated with First-Line Immunochemotherapy: A Review of PET/CT in Patients Who Did Not Achieve a Complete Metabolic Response in the GALLIUM Study. J Nucl Med. 2022;63(8):1149-1154. [CrossRef]
- Ansell SM, Armitage JO. Positron Emission Tomographic Scans in Lymphoma: Convention and Controversy. Mayo Clin Proc. 2012;87(6):571-580. [CrossRef]
- Pettitt AR, Barrington S, Kalakonda N, Khan UT, Jackson R, Carruthers S, et al. NCRI PETREA TRIAL: A PHASE 3 EVALUATION OF PET-GUIDED, RESPONSE-ADAPTED THERAPY IN PATIENTS WITH PREVIOUSLY UNTREATED, ADVANCED-STAGE, HIGH-TUMOUR-BURDEN FOLLICULAR LYMPHOMA. Hematol Oncol 2019;37: 67-68. [CrossRef]
- Kurtz DM, Soo J, Co Ting Keh L, Alig S, Chabon JJ, Sworder BJ, et al. Enhanced detection of minimal residual disease by targeted sequencing of phased variants in circulating tumor DNA. Nat Biotechnol 2021;39(12):1537–47. [CrossRef]
- Kurtz DM, Scherer F, Jin MC, Soo J, Craig AFM, Esfahani MS, et al. Circulating tumor DNA measurements as early outcome predictors in diffuse Large B-cell lymphoma. J Clin Oncol 2018;36(28):2845-2853. [CrossRef]
- Spina V, Bruscaggin A, Cuccaro A, Martini M, Di Trani M, Forestieri G, et al. Circulating tumor DNA reveals genetics, clonal evolution, and residual disease in classical Hodgkin lymphoma. Blood 2018;131(22):2413-2425. [CrossRef]
- Jiménez-Ubieto A, Martín-Muñoz A, Poza M, Dorado S, García-Ortiz A, Revilla E, et al. Personalized monitoring of circulating tumor DNA with a specific signature of trackable mutations after chimeric antigen receptor T-cell therapy in follicular lymphoma patients. Front Immunol 2023;14:1188818. [CrossRef]
| Parameter | Total n = 121 | |
| Median age, years (range) | 62 (27-87) | |
| Sex male, % | 52 | |
| Grade, % | ||
| 1-2 | 75 | |
| 3A | 25 | |
| Ann-Arbor stage, % | ||
| 2 | 7 | |
| 3 | 21 | |
| 4 | 72 | |
| B symptoms, % | 24 | |
| Bulky disease, % | 38 | |
| Extranodal involvement, %* | 31 | |
| Bone marrow involvement, % | 56 | |
| FLIPI, n=117, % | ||
| Low risk | 8 | |
| Intermediate risk | 34 | |
| High risk | 58 | |
| FLIPI2, n=115, % | ||
| Low risk | 30 | |
| Intermediate risk | 24 | |
| High risk | 46 | |
| PRIMA-PI, n=114, % | ||
| Low risk | 32 | |
| Intermediate risk | 37 | |
| High risk | 31 | |
| Hemoglobin (median g/dL, range) | 13.8 (8.8 - 17.7) | |
| Increased LDH % | 39 | |
| Increased β2-microglobulin % | 58 | |
| Treatment regimen, % | ||
| R-CHOP | 78 | |
| R-Bendamustine | 20 | |
| Other treatments | 2 | |
| Rituximab maintenance, n=105, % | 87 | |
| *Other than bone marrow. |
| Univariate P value |
Univariate HR (95% CI) |
Multivariate P value |
Multivariate HR (95% CI) | |
| Age ≥ 60 | 0.907 | 0.95 (0.43-2.12) | * | |
| Female (vs male) | 0.184 | 0.58 (0.26-1.30) | * | |
| Grade FL 3A (vs 1-2) | 0.102 | 0.41 (0.14-1.14) | * | |
| Advanced Ann-Arbor stage | 0.254 | 3.43 (0.41-28.52) | * | |
| B symptoms | 0.419 | 1.45 (0.59-3.55) | * | |
| > 4 nodal areas | 0.014 | 13.16 (1.70-101.59) | 0.026 | 9.7 (1.3-73.4) |
| BM involvement | 0.491 | 0.88 (0.61-1.27) | * | |
| Bulky disease | 0.510 | 0.75 (0.33-1.75) | * | |
| Extranodal involvement1 | 0.375 | 0.66 (0.27-1.65) | * | |
| Elevated β2 microglobulin | 0.005 | 3.80 (1.49-9.73) | 0.173 | * |
| Hemoglobin <12 | 0.024 | 3.12 (1.16-8.40) | 0.100 | * |
| Elevated LDH | 0.005 | 3.31 (1.45-7.59) | 0.247 | * |
| RB (vs RCHOP) | 0.287 | 1.68 (0.65-4.33) | * | |
| Rituximab maintenance | 0.028 | 028 (0.09-0.87) | 0.016 | 0.3 (0.1-0.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




