1. Introduction
Fistulas are connections formed between two epithelial surfaces. They can occur for example between the intestinal loops (enteroenteric), the intestine and the skin (enterocutaneous), or between the rectum and the skin of the buttocks (perianal) [
1,
2].
Patients with perianal fistulas develop symptoms significantly reducing the quality of life, such as anal pain, unpleasant leakage, and sometimes fecal incontinence [
1,
3,
4,
5]. It is reported that it has a negative impact on many aspects of life, including relationships, social and work life and it may even lead to the inability to take part in physical activities [
3].
The classic form of perianal fistula is a late, chronic stage of rectal abscess. Such abscess, through overgrowth, has formed a connection with the skin of the gluteal region, so that a narrow channel connecting the rectum to the skin surface has been formed. These lesions are usually initiated in the crypt glands (cryptoglandular) at the level of the dentate line [
4,
5].
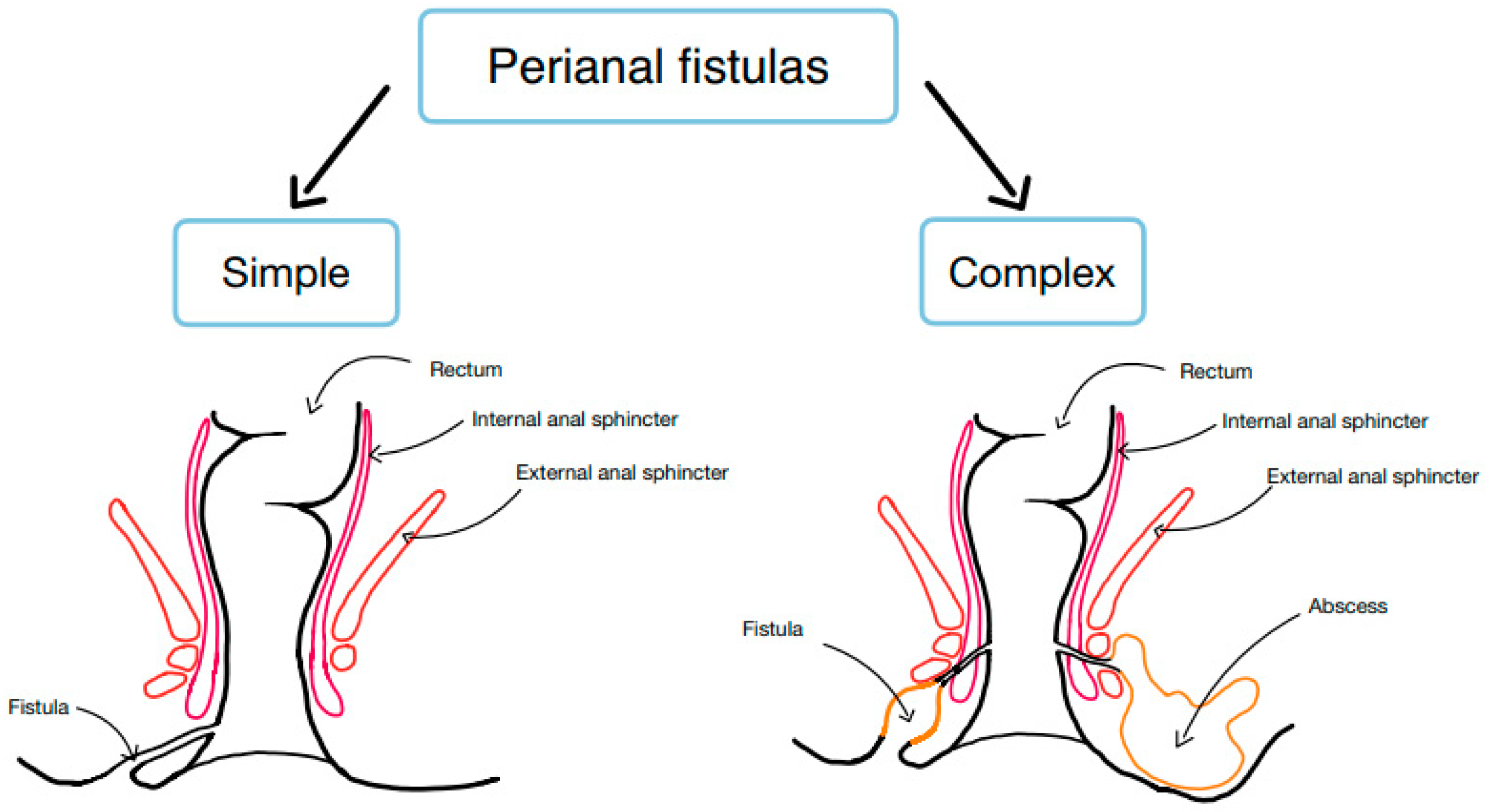
One of the most significant fistula classification systems is proposed by the American Gastroenterology Association [
6], which divides perianal fistulas into simple and complex. A simple fistula in this system involves a connection between the rectum and a single opening in the skin, without being associated with an abscess. A complex fistula, on the other hand, passes through or over the anal sphincter muscles and is associated with multiple end openings in the skin, as well as abscesses or anal strictures. The term complex fistula is also applied to fistulas that are a connection to other internal organs (e.g. bladder, vagina, bowel) [
1].
Based on current knowledge, EMT and matrix remodeling enzymes appear to play a special role in the pathophysiology of fistulas associated with CD. The EMT process produces transitional cells (TCs). In this case, epithelial cells transform into mesenchymal cells (like myofibroblasts) while gaining the ability to migrate and penetrate adjacent tissues. A regulatory factor crucial to the development of EMT is TGF-β. Its concentration increases in the zone between transitional and epithelial cells. The other important factor is TNF, which stimulates both the EMT process and fibroblasts to produce TGF-β. Other molecules upregulated in the TCs of fistula ducts in CD include IL-13, IL-13 receptor, ETS1, and DKK1 [
2,
7].
According to data from a survey conducted in 2023 by A. Spinelli et al. Crohn’s perianal fistula has a great negative impact on patients’ everyday life [
3]. The goal of this paper is to identify the scientific areas that require further research to better understand the development of fistulas, ultimately leading to more effective treatments.
2. Methodology
This literature review aims to explore the current understanding and management of perianal fistulas, focusing on biochemical, genetic, and epigenetic influences in their formation.
We used the keyword “perianal fistula” and checked the first 100 papers on PubMed, Embase, Web of Science sites, and Google Scholar. When selecting research papers, we adhered to two primary principles: the selected papers must address the etiology, pathogenesis, management, or impact of perianal fistulas, with a specific focus on CD-related fistulae and preference was given to papers published from 2010 to 2024, to ensure the incorporation of up-to-date information and latest advancements in the field. In this way, we chose 7 articles to base our paper on [
1,
2,
4,
5,
7,
8,
9]. These selected articles collectively offer a comprehensive perspective on perianal fistulas, molecular insights, and pathogenesis.
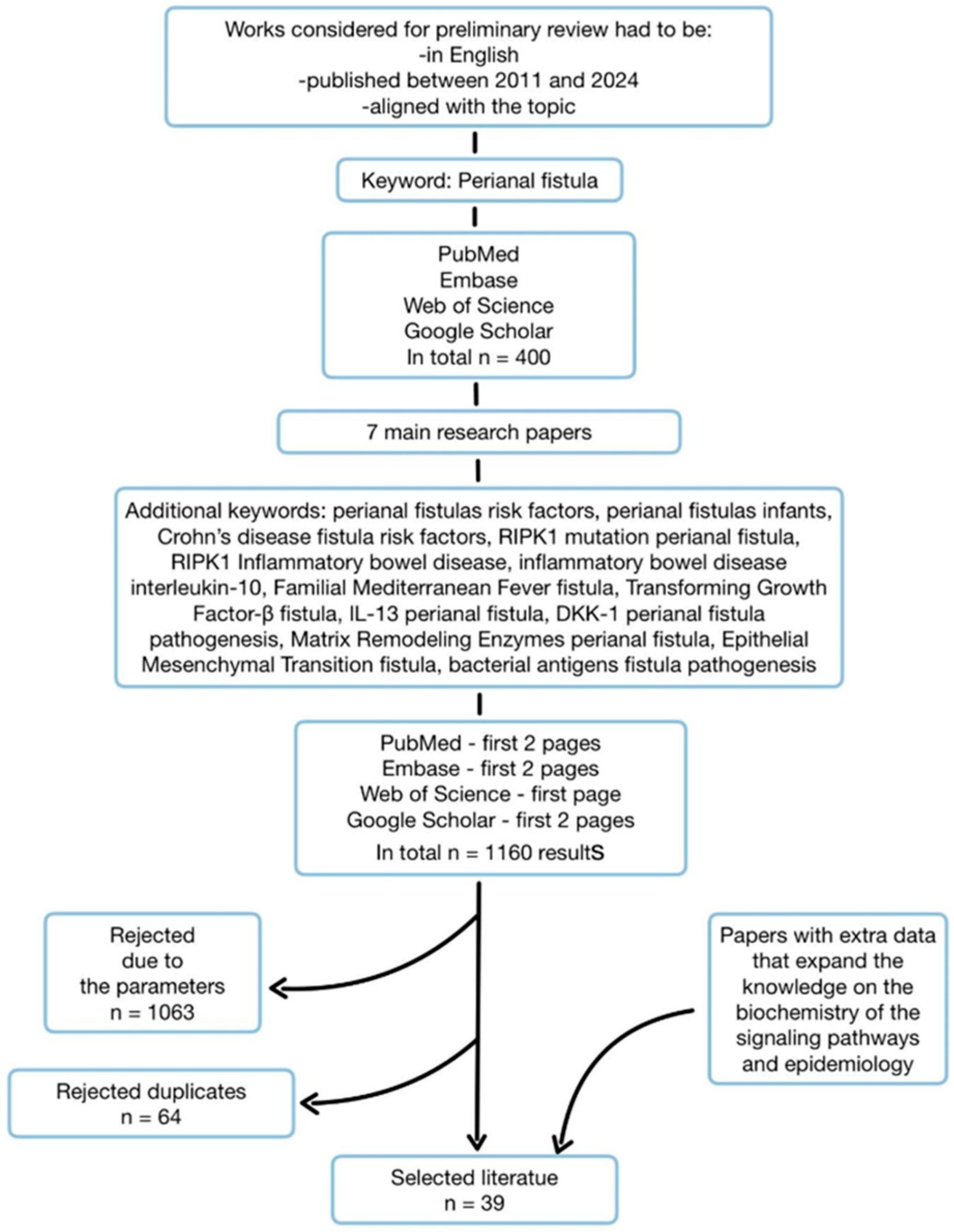
With this foundation, we began expanding the scope by seeking additional studies to delve deeper into the biochemical, genetic, and epigenetic aspects of fistula etiopathogenesis. We used additional keywords to expand our search field (
Figure 1).
That gave us 1160 results. We included papers that: were published for broad, free access and written in English. We were interested only in research conducted on human models, thus excluding those based on animals and cell line cultures. We didn’t have a preference for the type of article, including case studies, original works and review papers. As stated previously, papers had to cover the etiology, pathogenesis, management, or impact of perianal fistulas, with a specific focus on CD-related fistulae.
Next, we scanned papers on the basis of their abstracts and full texts using the same standards. Those directly addressing the role of sought factors in other diseases and didn’t contribute to the focus of this study, e.g. when the topic of fistulas was only briefly mentioned in the text, were not included.
All these criteria let us shorten our list of results to 97 papers. From these papers we excluded duplicates and we were left with 26 publications.
Because some topics weren’t sufficiently explored in our search results, we created new search keywords: “fistula impact on life quality”, “RIPK1 role in inflammation”, “TGF- β signaling pathway”, “β-caterin signaling pathway”, “SATB1 regulation β-caterin”, “β6 integrin activation”. For each of them, we found additional scientific papers that respectively expand the knowledge on each of the mentioned topics [
3,
10,
11,
12,
13,
14]. This allowed us to build a better foundation for important aspects: epidemiology of the perianal fistulas in CD and biochemistry of signaling pathways in the pathogenesis of fistulas.
At that point, we gathered studies on which we based our study, the papers that expand the topic of fistulas, and additional papers that expand and clarify data collected in this study, which makes 39 papers, to which we refer in this study.
We gathered data from these papers and organized them into sections covering similar topics, which we also used to divide our paper. First, we focused on the risk factors and pathogenesis of the fistulas, which gave us insight into the factors that may provoke fistula formation. Based on that, we created separate sections in this work, into which we placed information extracted from the papers, referring consecutively to the EMT, RIPK1, IL-10R, MEFV, TGF-β, TNF-α, IL-13, DKK-1, matrix remodeling enzymes, and bacterial remnants. Using these sections as a basis we wrote this paper by merging knowledge from the articles of similar topics. If any biochemical or epidemiological aspects were not sufficiently explained, we used additional papers to clarify and expand our understanding of the subject (
Figure 2).
3. Results
3.1. Risk Factors
The most important factors associated with fistulation are the male sex and the presence of ischiorectal or intersphincteric abscesses. The presence of inflammatory bowel disease (IBD) is also considered one of the main factors that increase the risk of fistula occurrence [
15].
According to data from 2015, the incidence of IBD is 0-39,4 per 100,000 in North America and 0,9-37.0 in Europe. The susceptibility to IBD is favoured by the family history of IBD, stress, unhealthy diet, for example: low on fiber, high omega-6 and low omega-3 PUFA intake, low on vitamin D; infections of the gastrointestinal tract and intake of NSAIDs, aspirin, oral contraceptives and undergoing postmenopausal hormone therapy [
16]. Other sources also indicate obesity (BMI>25 kg/m2), high daily salt intake, diabetes, hyperlipidemia, dermatosis, anorectal surgery, smoking, alcohol intake, sedentary lifestyle, excessive intake of spicy and high-fat food, infrequent participation in sports and prolonged sitting on the toilet for defecation [
17]. Preventing the development of IBD may be one of the strategies to help prevent the formation of perianal fistulas.
It is shown that 0.5% to 4.3% of all perianal abscesses (PA) or fistulas occur during infancy, especially among males [
18,
19]. The progression of these conditions among infants tends to resolve spontaneously in most instances, while older children exhibit patterns like PA and perianal fistulas seen in adults.
There are two theories regarding the formation of perianal fistulas in infants. According to the first theory, congenital abnormalities result in deeper crypts of Morgagni, which leads to blockage in their narrowest parts. In this way, a PA forms, which, if it drains through the skin, creates a perianal fistula. The second theory suggests that congenital perianal fistulas progress to a PA and then develop into an epithelial lining of the fistula. Due to these discrepancies, the pathogenesis of PA/perianal fistulas remains elusive [
19].
3.2. Pathogenesis
More than 90% of perianal fistulas are cryptoglandular fistulas [
4]. They can arise as a complication after hidradenitis suppurativa, trauma, cancer, or tuberculosis [
5]. The second most common type is CD-associated fistulas. It is important to note that due to differences in pathogenesis, the treatment of cryptoglandular and CD-related fistulas differ significantly. CD-related fistulas require a combination of drug therapy and surgical interventions, while cryptoglandular fistulas are mainly treated surgically [
4,
5,
8].
The CD is a chronic immune-mediated inflammatory condition that can affect any segment along the length of the entire gastrointestinal tract. It is often complicated by intestinal strictures and fistulas [
1]. It represents, along with ulcerative colitis (UC), the main form of IBD [
2]. CD can have one of 3 types of course: inflammatory, constrictive, and penetrating; with the penetrating form often leading to fistulas, although any type of CD can progress and change over time. [
1,
9,
20]. Single Nucleotide Polymorphism (SNP) in gene loci of
ZMIZ1,
LOC105373831, and
KSR1 can increase the probability of development of constrictive or penetrating disease phenotype. The SNPs within gene loci
TNFSF15 and
CEBPB-PTPN1 tend to protect from the progression to those phenotypes of the disease [
21].
Understanding of the pathophysiology of CD-associated fistulas is not complete yet. Two mechanisms seem to have a major role: EMT and matrix remodeling enzymes.
Table 1.
Mutations that predict disease progression in CD patients[
22].
Table 1.
Mutations that predict disease progression in CD patients[
22].
| Mutation |
Effect |
| ZMIZ1 (Zinc Finger MIZ-Type Containing 1) |
ZMIZ1 is associated with transcription regulation and interaction with other transcription factors. SNPs (single nucleotide polymorphisms) in this gene may affect its function, which could have implications for inflammatory and immune processes related to CD. |
| LOC105373831 |
LOC105373831 is a gene with a less well-known function that is considered a region susceptible to genetic changes associated with IBD. SNPs in this locus may influence the expression of nearby genes or the functioning of signaling pathways. |
| KSR1 (Kinase Suppressor of Ras 1) |
KSR1 is involved in the Ras/MAPK signaling pathways, which regulate cell growth and inflammatory responses. SNPs in KSR1 may alter its function, potentially affecting inflammatory mechanisms related to CD. |
| TNFSF15 (Tumour Necrosis Factor Superfamily Member 15) |
TNFSF15, also known as TL1A, is an inflammatory factor that plays a role in regulating immune and inflammatory responses. SNPs in this gene can affect its level or function, potentially leading to increased inflammation and progression of CD. |
| CEBPB-PTPN1 (CCAAT/Enhancer Binding Protein Beta–Protein Tyrosine Phosphatase Non-Receptor Type 1) |
CEBPB is a transcription factor that regulates inflammatory response, and PTPN1 is a phosphatase protein that can modulate inflammatory signaling. SNPs in this region may affect the expression or function of these genes, which could impact the development and progression of CD. |
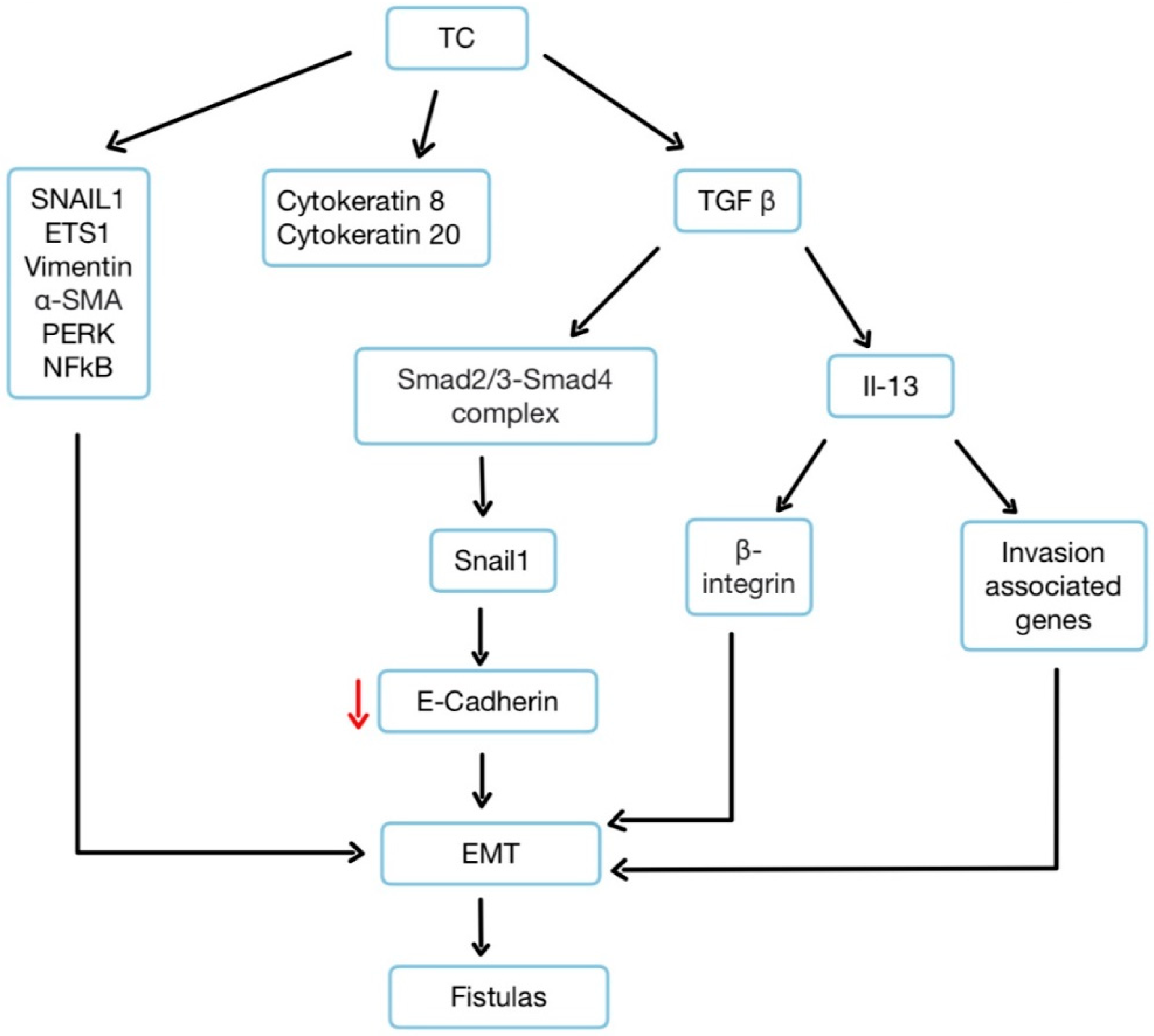
3.3. Epithelial-Mesenchymal Transition
Approximately 27% of CD fistulas exhibit epithelial cells lining the inner surface (epithelialized fistulas), while the remaining are classified as non-epithelialized fistulas covered by myofibroblast-like TCs. A distinct region was identified within most of the patients’ latter fistulas where epithelial cells appeared to transition into TCs, suggesting EMT involvement in CD-associated fistula development [
2].
Specialized epithelial cells undergo a change where they transition into mesenchymal-like cells, gaining the capability to migrate and infiltrate neighboring tissues. In these TCs, genes of epithelial cell markers such as cytokeratin 8 and cytokeratin 20 are expressed [
2,
23]. Other biosynthesized factors include mesenchymal markers such as vimentin and smooth muscle actin (α-SMA). They also downregulate the expression of adhesion molecules, such as E-cadherin [
2,
4]. TCs are upregulating transcription factors such as SNAIL1, SLUG (also known as SNAIL2), and β6-integrin [
2,
4,
24]. TGF-β is a pivotal regulator of EMT, inducing IL-13 expression and an EMT-like phenotype in intestinal epithelial cells. IL-13, in turn, promotes the expression of invasion-associated genes, suggesting a synergistic role of TGF-β and IL-13 in fistula pathogenesis [
25]. Other molecules that are upregulated in TCs of fistula tracts in CD include IL-13 receptor, ETS1 (also known as protein C-ets-1) and DKK1 (dickkopf-related protein 1), protein kinase (RNA), PKR-like endoplasmic reticulum kinase (PERK) and nuclear factor B (NFkB) [
4,
25]. Translocation of the β-catenin to the nucleus for gene regulation and increased β6-integrin expression are also considered to be EMT markers [
25]. We will look further into those markers in the following sections of this paper.
SNAIL1 expression is notably elevated in the nuclei of TCs lining fistula tracts and crypts adjacent to the tracts, suggesting the involvement of SNAIL1 in fistula pathogenesis. Although SLUG expression is lower than SNAIL1 in TCs, it is present in fibrotic areas around CD fistulas. This suggests its potential role in fibrosis development but rather minor – if any – in EMT onset leading to CD-associated fistulas [
2].
Cytokine and phosphoprotein profiles of idiopathic and CD-related perianal fistulas are broadly similar. Only two measurements show a significant difference between the groups: IL-12 and the IL-1RA/IL-1β ratio, both at the internal opening. This caused the formation of the theory that cryptoglandular and CD-related perianal fistulas may not be as immunologically distinct as previously supposed, which introduces the possibility that biological agents used in CD-related perianal disease may also have a role in cryptoglandular perianal fistulas [
4].
3.4. RIPK1
It was observed that mutations in RIPK1 may lead to the development of perianal fistulas. These patients exhibited severe intestinal inflammation, as well as skin lesions and perianal inflammation, already in the first months of life. Studies on samples taken from patients with PA allowed to identify two mutations causing RIPK1 protein dysfunction:
c.1934C>
T in exon 11 and
c.580G>A in exon 4. [
26].
RIPK1 mutations lead to deficits in inflammatory regulation, which contribute to the development of fistulas through chronic inflammation and tissue damage in the perianal region. One of the pathways found to be upregulated in RIPK1-deficient macrophages is inflammasome activation [
26]. Inflammasome induces the secretion of pro-inflammatory cytokines IL-1β and IL-18 in response to various stimuli, including lipopolysaccharides (LPS) [
27]. Impaired inflammasome activation has been reported in other monogenic disorders associated with IBD [
26]. Till 2022, 14 patients with deleterious RIPK1 mutations have been reported in the literature and 12 of them were presented with PA or fistulas [
28,
29,
30,
31].
RIPK1 also plays a role in mediating apoptosis, inflammation, and necroptosis by participating in the formation of protein complexes involved in the transduction of inflammation upon stimulation by TNFα [
10]. Consequently, individuals deficient in RIPK1-dependent signaling may experience inflammatory and degenerative diseases such as severe colitis and perianal fistulas, especially in young patients [
10,
26]. Additional studies are still required to better define the role of RIPK1 in regulation of intestinal immune responses [
26].
3.5. IL-10R
Monogenic defects play a crucial role in the development of IBD in infants, and thus, these mutations may be factors that increase the risk of fistula formation [
15,
32]. The first monogenic defects identified in children with infantile IBD were mutations in IL-10R-inducing IL-10 signal pathways. Such mutations lead to abnormal suppression of IL-6 secretion in response to LPS/IL-10 co-stimulated peripheral blood mononuclear cell [
32,
33]. These mutations can disrupt the IL-10-induced signaling pathway, resulting in improper phosphorylation of Transcription Factor 3 and increased levels of TNF-α and other pro-inflammatory cytokines, such as IL-6, IL-1, etc. [
32,
33].
It is hypothesized that without an IL–10–mediated anti-inflammatory reaction, the existence of intestinal commensal bacteria triggers a vigorous immune response, leading to excessive inflammation and subsequent tissue injury. This could promote higher levels of intestinal bacteria migration and lead to persistent intestinal lymphadenopathy or, potentially, organ-specific abscesses [
33]. Abscesses like this, as previously stated, can evolve into fistulas [
5].
Individuals harboring IL-10R mutations, during infancy, typically exhibit treatment-resistant enterocolitis and severe perianal conditions, including PA, fissures, and fistulas [
34]. There is no difference in the course of the disease between the IL-10RA, IL-10RB, and IL-10 mutations [
35].
3.6. Mediterranean Fever Gene (MEFV)
The MEFV gene is responsible for encoding the pyrin protein, which is a mediator regulating apoptosis-associated speck-like protein, containing a caspase recruitment domain and affecting caspase-1 activity in the inflammasome. Mutations in the MEFV gene cause systemic increase in IL-1β [
36]. Symptoms of familial Mediterranean fever (FMF), caused by mutations in the MEFV gene, can mimic those seen in IBD, including CD, UC, and intestinal Behçet's disease (BD) [
36]. The MEFV gene was reported to interact with and modulate the activity of several inflammasome components, including cryopyrin, caspase-1 and its substrate, pro-IL-1β [
36,
37]. Although FMF itself may cause symptoms similar to IBD (in up to 70% of FMF cases, there is a nonspecific chronic inflammation of the bowel [
38]) and thus lead to the formation of fistulas, the probability of FMF-CD is 28.6% in all FMF-IBD cases [
39]). The presence of FMF is not associated with CD susceptibility, but CD patients who are carriers of the MEFV mutation are more likely to experience a structuring disease pattern and extraintestinal manifestations of the disease [
40].
Table 2.
The mutations that elevate the risk of fistula development.
Table 2.
The mutations that elevate the risk of fistula development.
| Reference and number of patients |
Mutated protein |
Affected mechanism |
Effect |
| Sultan et al., 2022 [26] – 14 people with the mutation, 12 of them with PA or fistula |
RIPK1 |
inflammasome activation - increased secretion of IL-1β and IL-18, decreased production of IL-6 |
development of fistulas through severe intestinal inflammation and tissue damage in the perianal region |
Glocker et al., 2009; [33] - 4
Hung et al., 2021; [32] - 1
Kotlarz et al., 2012b; [34] - 10
Pigneur et al., 2013b; [35] – 16
n=31 |
IL-10R |
suppression of IL-6 secretion in response to LPS/IL-10, increased levels of TNF-α and other pro-inflammatory cytokines, such as IL-6, IL-1 |
excessive inflammation of the intestinal tract |
Asakura et al., 2018; [36] - 1
Baran et al., 2018; [37] - 1
Fidder et al., 2005; [40] – 18
n=20 |
MEFV |
reduces amounts of pyrin produced or produces malformed pyrin protein |
inappropriate or prolonged inflammatory response |
3.7. TGF-β, TNF-α, IL-13
TGF- β stands out as the primary driver of EMT [
11,
24,
25]. It prompts epithelial cells to lose their typical arrangement. The activated TGF-β receptors phosphorylate Smad2 and Smad3 heterodimer, which interacts with Smad4. The Smad2/3-Smad4 complexes translocate from the cytosol to the nucleus and bind to the additional DNA-binding cofactors to regulate specific TGF-β target genes [
41]. As a result, it causes a reduction in E-Cadherin levels. The inhibition of E-Cadherin activity is mediated by SNAIL1 [
24]. Furthermore, TGF-β prompts the relocation of β-catenin to the cell nucleus, where it regulates the expression of specific target genes [
24,
25,
41]. It has been revealed that IL-13 secretion can be induced by TGF-β in fibroblasts derived from patients with fistulizing CD. IL-13 then causes an increased expression of β6-integrin and the transcription factor SLUG (
Figure 3). It's noteworthy that TNF-α can stimulate the production of TGF-β and both of them have the capability to induce EMT in colonic organoids [
24]. TNF-α, TGF-β, and IL-13 are involved in regulatory feedback loops, likely controlling cell migration and invasion in the pathogenesis of CD-associated perianal fistulas [
24].
3.8. DKK-1
DKK-1 is known to play a key role during EMT [
24]. At the mRNA level, the amount of DKK-1 mRNA was increased in the respective intestinal tissue samples from patients with active CD and UC compared to samples from non-IBD control patients or patients with CD in remission [
24]. Additionally, DKK-1 is a potent antagonist of the canonical Wnt/β-catenin signaling pathway[
12,
24] and has been implicated to act as a mediator of inflammation [
24].
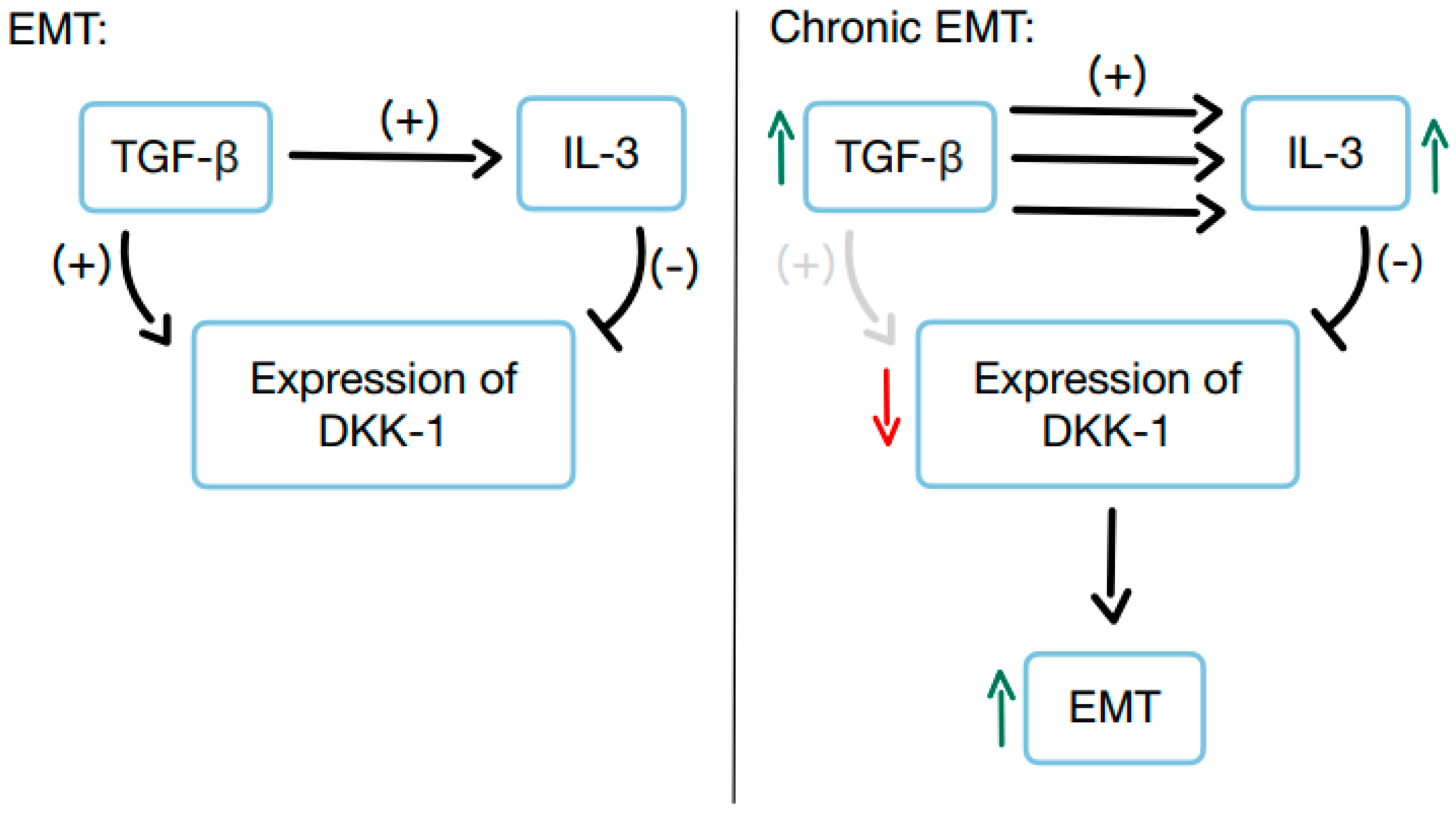
It was found that TGF-β increases the expression of DKK-1 in epithelial cells, but at the same time decreases it in fibroblasts derived from fistulas in CD patients and in control fibroblasts from patients without IBD [
24]. When exposed to TGF-β, epithelial cells undergo EMT and produce more DKK-1 [
24]. Higher DKK-1 levels in these cells can limit cell activity by blocking Wnt signaling, which slows down cell growth, movement, and invasion. It has been found that DKK-1 is also needed for TGF-β to induce IL-13 expression [
13,
24]. This suggests that DKK-1 plays a dual role: it can inhibit TGF-β/Wnt signaling, but it also helps activate IL-13, promoting cell invasion. Moreover, during EMT, TGF-β boosts IL-13 production, which in turn suppresses DKK-1 expression. This creates a complex feedback loop in which TGF-β induces DKK-1, while IL-13 inhibits it (
Figure 4). In inflammatory conditions like chronic intestinal inflammation, excessive TGF-β secretion can lead to overproduction of IL-13, which suppresses DKK-1 and promotes EMT. As shown previously, IL-13 then triggers the expression of genes involved in cell invasion, such as β6-integrin, leading to the formation of fistulas (
Figure 3) [
24,
25]. This effect can be reversed in all cell types studied using anti-IL-13 antibodies. These findings support the hypothesis that inhibiting IL-13 can prevent the formation of fistulas [
24,
25].
3.9. Matrix Remodeling Enzymes
Matrix metalloproteinases (MMPSs) facilitate fistula progression by degrading the extracellular matrix (ECM) and promoting cell migration [
4]. Increased MMPS activity has been observed in both experimental studies and humans with IBD [
7,
41]. In CD fistula tracts, there has been a significant increase in the production of MMPSs, particularly MMPS3 protein, as well as elevated mRNA levels in mononuclear cells and fibroblasts [
7].
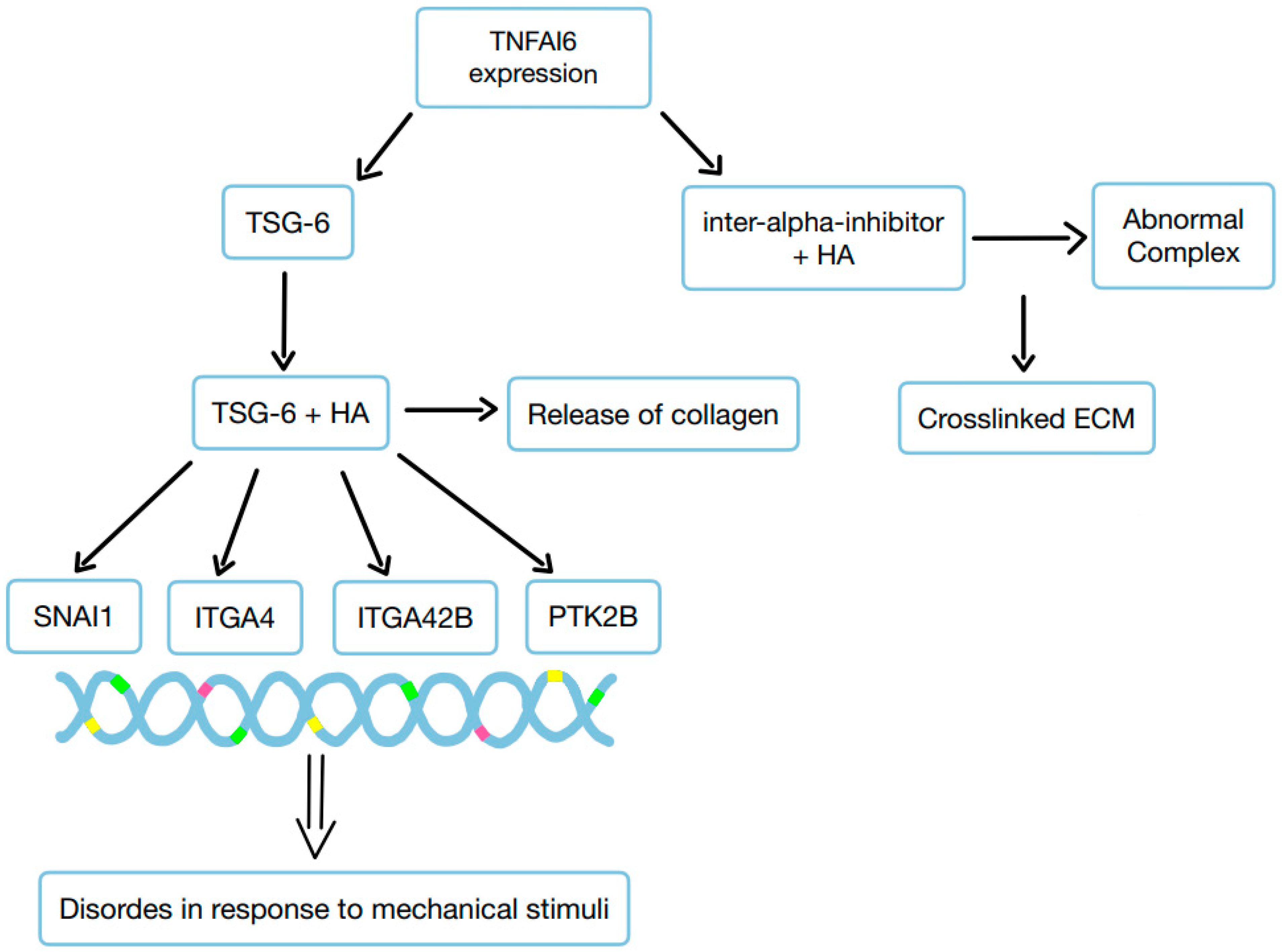
A noticeable difference in the organization of the ECM between the fistula and the surrounding regions was observed [
42]. In the fistula, there was increased expression of integrins and MMPSs, along with hyaluronan (HA) staining, which correlated with heightened production of newly formed collagen fibers and mechanosensitive proteins [
42]. Among the genes associated with ECM regulation, TNFAI6 (which encodes TSG-6) was significantly more expressed in the fistula compared to the surrounding area. In the fistula, TNFAI6 promoted the abnormal formation of complexes between heavy chains of inter-alpha-inhibitor and HA, leading to the creation of a crosslinked ECM. Additionally, enriching an HA scaffold with recombinant human TSG-6 protein promoted the release of collagen. It also increased the expression of genes such as SNAI1, ITGA4, ITGA42B, and PTK2B, which are involved in transmitting responses to mechanical stimuli. TSG-6 triggers the EMT by mediating changes in ECM organization and transcription factor SNAI1 through the activation of mechanosensitive proteins [
42]. EMT triggered in this way may be the first step toward the development of a fistula.
3.10. Bacterial Remnants
LPS and other bacterial remnants play a role in the development of chronic inflammatory lesions by triggering EMT, which leads to abnormal ECM integrity. This disruption causes the release of proinflammatory cytokines and other molecules. Therefore, EMT may promote increased migration of epithelial cells, which could contribute to fistula canal development [
4].
Recent evidence suggests that in CD patients, defects in the epithelial lining – caused by inflammation or injury – lead to the development of pathogen-associated patterns [
4,
43]. These patterns trigger various pathways involving TNF-α, TGF-β, IL-13, MMPSs, and integrin αvβ. These pathways promote EMT, allowing cells to invade and migrate, leading to the formation of a penetrating fistula lined with TCs [
4].
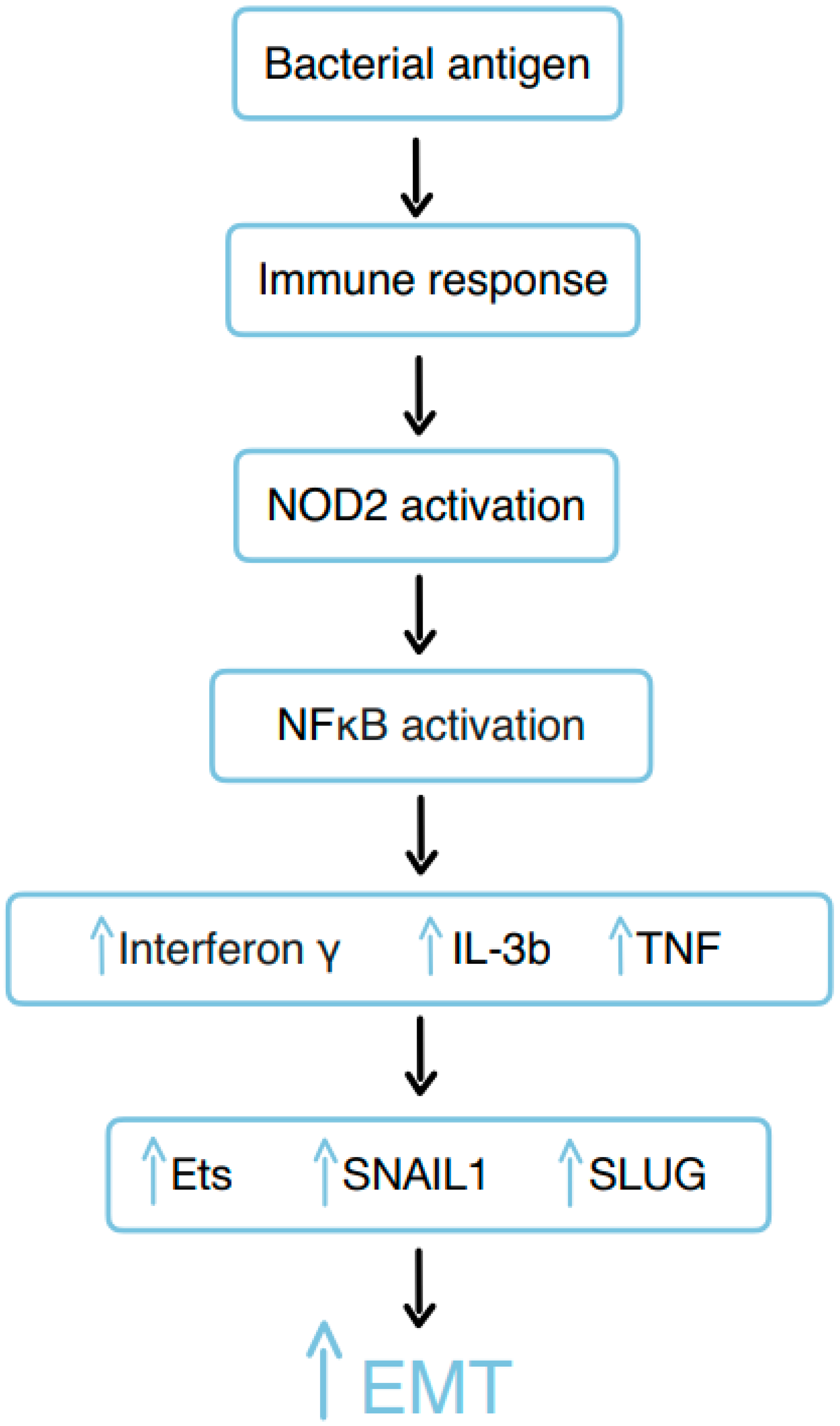
Figure 6.
The role of bacterial remnants in the development of chronic inflammatory lesions through EMT.
Figure 6.
The role of bacterial remnants in the development of chronic inflammatory lesions through EMT.
An incorrect immune response to bacterial antigens (for example, muramyl dipeptide (MDP)) during CD and the failure of intestinal barrier function might result from NOD2 and subsequent activation of NFκB, thus inducing proinflammatory cytokines, including interferon-gamma, IL-3, and TNF [
23]. Subsequently, the cytokines and growth factors induce the expression of transcription factors associated with EMT and cell invasiveness, such as Ets-1, SNAIL1, and SLUG [
2,
23]. As a result, EMT is initiated, likely as part of a physiological response aimed at wound healing, during ongoing inflammation. Simultaneously, the expression of molecules associated with invasive cell growth, such as β6-integrin, increases. Finally, this leads to the manifestation of CD-associated fistulas [
23].
MDP upregulates transcription factor ETS-1, which is a proto-oncogene that mediates activation of the αvβ6-integrin receptor via the subunit β6-integrin [
14,
23]. ETS-1 is induced by TNF in HT29 epithelial cells, control fibroblasts, and fistula fibroblasts from CD patients. It is also known that TNF induces β6-integrin through ETS-1 [
23]. ETS-1 could be a key regulatory element in bacterially induced EMT and fistulas, starting from bacterial promotion of TNF production [
23].
Table 3.
The role of proteins in fistula forming.
Table 3.
The role of proteins in fistula forming.
| Reference |
Protein |
Role of the protein in the fistula forming |
Effect |
| M. Scharl et al., 2013 [25] |
TGF-β |
reduces the E-Cadherin level, induces IL-13 expression |
promotes EMT |
| S. M. Frei et al., 2013 [23] |
TNF-α |
stimulates the production of TGF-β |
promotes EMT |
| M. Scharl et al., 2013 [25] |
IL-13 |
increases expression of β6-integrin and the transcription factor SLUG |
triggers the expression of genes involved in cell invasion, leading to the formation of fistulas |
| S. M. Frei et al., 2013 [23] |
DKK-1 |
antagonist of the Wnt/β-catenin signaling pathway, mediator of inflammation |
inhibits TGF-β/Wnt signaling, but also helps activate IL-13, which promotes cell invasion |
4. Conclusions
The conducted analysis indicates a significant role of genetic, epigenetic factors, and proteins in the pathogenesis of perianal fistulas. Based on numerous studies, the EMT and matrix remodeling enzymes are crucial in the development of fistulas associated with CD. Both EMT and matrix remodeling enzymes facilitate the transformation of epithelial cells into mesenchymal-like TCs, promoting invasion and tissue infiltration that contribute to fistula formation.
The TCs lining the fistulas express factors that upregulate the EMT process, such as TGF-β, ETS1, vimentin, α-SMA, SNAIL1, PERK, NFκB, and DKK1 [
2,
4,
7,
42]. TNF-α stimulates TGF-β production, which then enhances EMT processes and promotes cell migration and invasion in the development of CD-associated fistulas [
2,
7,
11,
24]. TGF-β also induces EMT by activating receptors that phosphorylate Smad2 and Smad3, which then form a complex with Smad4 and translocate to the nucleus to regulate target genes, reducing E-Cadherin levels and promoting the nuclear relocation of β-catenin. The use of anti-TNF-α antibody therapy is widespread and supported by randomized trials; it has been proven to have a 36% chance of permanent closure of the fistula compared to a 19% chance of full recovery while on placebo [
44,
45].
Mutations in RIPK1, IL-10R, and MEFV contribute to fistula development by disrupting immune regulation, which leads to chronic inflammation and tissue damage. RIPK1 mutations result in impaired inflammasome activation and dysregulated immune responses, promoting inflammation and necroptosis, which can cause fistulas. IL-10R mutations prevent the anti-inflammatory signaling pathway, resulting in excessive immune reactions to commensal bacteria, causing tissue injury, abscess formation, and fistula development. MEFV mutations increase IL-1β production through dysregulated inflammasome activity, leading to inflammation that can mimic IBD and contribute to fistula formation [
26,
32,
33,
40]. This suggests that, especially in cases of infantile fistulizing features, genetic differentiation may be necessary to select the appropriate therapy. MEFV mutations treated with colchicine therapy show great results with co-existing fistulas [
37], and fistulas due to the mutation in IL-10R or IL-10 genes may be treated with allogeneic hematopoietic stem cell transplantation. Bacterial remnants, such as LPS and MDP, contribute to fistula formation in CD by triggering EMT and disrupting ECM integrity. These remnants activate immune pathways and induce proinflammatory cytokines and growth factors, leading to increased epithelial cell migration, tissue invasion, and the formation of fistulas lined with TCs due to abnormal immune response and impaired intestinal barrier function [
4,
43]. This indicates that antibiotics could be used for their prophylactic effects against infections and abscesses, or as adjuncts in medical treatments, such as in combination with infliximab; further research is needed to confirm these uses [
44]. The exact mechanisms of their formation remain unknown and require further investigation, particularly regarding the role of genes associated with these conditions.
This paper primarily aims to highlight the biochemical and genetic aspects of fistula formation. These topics are still not sufficiently researched, and more data are needed to develop effective treatment methods. The aim was to suggest which areas of science should be explored further to better understand the mechanisms of fistula development. We hope that by gathering all factors contributing to fistula formation in one paper, we are creating an anchor point for other researchers to begin their work and avoid missing any crucial aspects influencing new approaches to fistula treatment and prevention.
Conflicts of Interest
All authors declare no conflicts of interest in this paper.
References
- Gold SL, Cohen-Mekelburg S, Schneider Y, et al. (2018) Perianal Fistulas in Patients With Crohn’s Disease, Part 1: Current Medical Management.
- Scharl M, Weber A, Fürst A, et al. (2011) Potential role for SNAIL family transcription factors in the etiology of Crohn’s disease-associated fistulae. Inflamm Bowel Dis 17: 1907–1916. [CrossRef]
- Spinelli A, Yanai H, Girardi P, et al. (2023) The Impact of Crohn’s Perianal Fistula on Quality of Life: Results of an International Patient Survey. Crohns Colitis 360 5. [CrossRef]
- Włodarczyk M, Włodarczyk J, Sobolewska-Włodarczyk A, et al. (2021) Current concepts in the pathogenesis of cryptoglandular perianal fistula. Journal of International Medical Research 49. [CrossRef]
- Nottingham JM, Rentea RM (2023) Anal Fistulotomy.
- American Gastroenterological Association Clinical Practice Committee american-gastroenterological-association-medical-position-statem-2003.
- Panés J, Rimola J (2017) Perianal fistulizing Crohn’s disease: Pathogenesis, diagnosis and therapy. Nat Rev Gastroenterol Hepatol 14: 652–664. [CrossRef]
- Zhou Z, Ouboter LF, Peeters KCMJ, et al. (2023) Crohn’s Disease-Associated and Cryptoglandular Fistulas: Differences and Similarities. J Clin Med 12. [CrossRef]
- Braithwaite GC, Lee MJ, Hind D, et al. (2017) Prognostic factors affecting outcomes in fistulating perianal Crohn’s disease: a systematic review. Tech Coloproctol 21: 501–519. [CrossRef]
- Meng H, Liu Z, Li X, et al. (2018) Death-domain dimerization-mediated activation of RIPK1 controls necroptosis and RIPK1-dependent apoptosis. Proc Natl Acad Sci U S A 115: E2001–E2009. [CrossRef]
- Derynck R, Budi EH (2019) Specificity, versatility, and control of TGF-β family signaling. Sci Signal 12. [CrossRef]
- Liu J, Xiao Q, Xiao J, et al. (2022) Wnt/β-catenin signaling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct Target Ther 7: 3. [CrossRef]
- Notani D, Gottimukkala KP, Jayani RS, et al. (2010) Global regulator SATB1 recruits β-catenin and regulates TH2 differentiation in Wnt-dependent manner. PLoS Biol 8. [CrossRef]
- Bates RC, Bellovin DI, Brown C, et al. (2005) Transcriptional activation of integrin β6 during the epithelial-mesenchymal transition defines a novel prognostic indicator of aggressive colon carcinoma. Journal of Clinical Investigation 115: 339–347. [CrossRef]
- Cooper CR, Keller DS (2020) Perianal Fistulas. Dis Colon Rectum 63: 129–132. [CrossRef]
- Ananthakrishnan AN (2015) Epidemiology and risk factors for IBD. Nat Rev Gastroenterol Hepatol 12: 205–217. [CrossRef]
- Varsamis N, Kosmidis C, Chatzimavroudis G, et al. (2022) Perianal fistulas: A review with emphasis on preoperative imaging. Adv Med Sci 67. [CrossRef]
- Festen C, Van Hat-Ten H Perianal Abscess and Fistula-in-An0 in Infants. [CrossRef]
- Gosemann JH, Lacher M (2020) Perianal Abscesses and Fistulas in Infants and Children. European Journal of Pediatric Surgery 30: 386–390. [CrossRef]
- Papi C, Festa V, Fagnani C, et al. (2005) Evolution of clinical behaviour in Crohn’s disease: Predictive factors of penetrating complications. Digestive and Liver Disease 37: 247–253. [CrossRef]
- Ditrich F, Blümel S, Biedermann L, et al. (2020) Genetic risk factors predict disease progression in Crohn’s disease patients of the Swiss inflammatory bowel disease cohort. Therap Adv Gastroenterol 13. [CrossRef]
- Ditrich F, Blümel S, Biedermann L, et al. (2020) Genetic risk factors predict disease progression in Crohn’s disease patients of the Swiss inflammatory bowel disease cohort. Therap Adv Gastroenterol 13. [CrossRef]
- Frei SM, Pesch T, Lang S, et al. (2013) A Role for Tumor Necrosis Factor and Bacterial Antigens in the Pathogenesis of Crohnʼs Disease–Associated Fistulae. Inflamm Bowel Dis 19: 2878–2887. [CrossRef]
- Frei SM, Hemsley C, Pesch T, et al. (2013) The Role for Dickkopf-Homolog-1 in the Pathogenesis of Crohn’s Disease-Associated Fistulae. PLoS One 8: e78882. [CrossRef]
- Scharl M, Frei S, Pesch T, et al. (2013) Interleukin-13 and transforming growth factor β synergise in the pathogenesis of human intestinal fistulae. Gut 62: 63–72. [CrossRef]
- Sultan M, Adawi M, Kol N, et al. (2022) RIPK1 mutations causing infantile-onset IBD with inflammatory and fistulizing features. Front Immunol 13. [CrossRef]
- He Y, Hara H, Núñez G (2016) Mechanism and Regulation of NLRP3 Inflammasome Activation. Trends Biochem Sci 41: 1012–1021. [CrossRef]
- Li Y, Führer M, Bahrami E, et al. (2019) Human RIPK1 deficiency causes combined immunodeficiency and inflammatory bowel diseases. Proc Natl Acad Sci U S A 116: 970–975. [CrossRef]
- Lin L, Wang Y, Liu L, et al. (2020) Clinical phenotype of a Chinese patient with RIPK1 deficiency due to novel mutation. Genes Dis 7: 122–127. [CrossRef]
- Cuchet-Lourenço D, Eletto D, Wu C, et al. (2018) Biallelic RIPK1 mutations in humans cause severe immunodeficiency, arthritis, and intestinal inflammation . Science (1979) 361: 810–813. [CrossRef]
- Uchiyama Y, Kim CA, Pastorino AC, et al. (2019) Primary immunodeficiency with chronic enteropathy and developmental delay in a boy arising from a novel homozygous RIPK1 variant. J Hum Genet 64: 955–960. [CrossRef]
- Hung HH, Lee HC, Yeung CY, et al. (2021) Importance of early detection of infantile inflammatory bowel disease with defective IL-10 pathway: A case report. Medicine (United States) 100: E25868. [CrossRef]
- Glocker E-O, Kotlarz D, Boztug K, et al. (2009) Inflammatory Bowel Disease and Mutations Affecting the Interleukin-10 Receptor. New England Journal of Medicine 361: 2033–2045. [CrossRef]
- Kotlarz D, Beier R, Murugan D, et al. (2012) Loss of interleukin-10 signaling and infantile inflammatory bowel disease: Implications for diagnosis and therapy. Gastroenterology 143: 347–355. [CrossRef]
- Pigneur B, Escher J, Elawad M, et al. (2013) Phenotypic characterization of very early-onset IBD due to mutations in the IL10, IL10 receptor alpha or beta gene: A survey of the GENIUS working group. Inflamm Bowel Dis 19: 2820–2828. [CrossRef]
- Asakura K, Yanai S, Nakamura S, et al. (2018) Familial Mediterranean fever mimicking Crohn disease. Medicine (United States) 97. [CrossRef]
- Baran M, Çağan Appak Y, Garipcin P, et al. (2018) The role of familial mediterranean fever gene mutation in treatment of infantile colitis with resistant perianal fistula. Arch Rheumatol 33: 473–477. [CrossRef]
- Demir A, Akyüz F, Göktürk S, et al. (2014) Small bowel mucosal damage in familial Mediterranean fever: Results of capsule endoscopy screening. Scand J Gastroenterol 49: 1414–1418. [CrossRef]
- Beşer ÖF, Çokuğraş FÇ, Kutlu T, et al. (2014) Association of familial mediterranean fever in Turkish children with inflammatory bowel disease. Turk Pediatri Ars 49: 198–202. [CrossRef]
- Fidder H, Chowers Y, Ackerman Z, et al. (2005) The familial Mediterranean fever (MEVF) gene as a modifier of Crohn’s disease. American Journal of Gastroenterology 100: 338–343. [CrossRef]
- Yun S-M, Kim S-H, Kim E-H (2019) The Molecular Mechanism of Transforming Growth Factor-β Signaling for Intestinal Fibrosis: A Mini-Review. Front Pharmacol 10. [CrossRef]
- Rizzo G, Rubbino F, Elangovan S, et al. (2023) Dysfunctional Extracellular Matrix Remodeling Supports Perianal Fistulizing Crohn′s Disease by a Mechanoregulated Activation of the Epithelial-to-Mesenchymal Transition. Cell Mol Gastroenterol Hepatol 15: 741–764. [CrossRef]
- Scharl M, Rogler G (2014) Pathophysiology of fistula formation in Crohn’s disease. World J Gastrointest Pathophysiol 5: 205. [CrossRef]
- Taxonera C, Schwartz DA, García-Olmo D (2009) Emerging treatments for complex perianal fistula in Crohn’s disease. World J Gastroenterol 15: 4263–4272. [CrossRef]
- de Groof EJ, Sahami S, Lucas C, et al. (2016) Treatment of perianal fistula in Crohn’s disease: a systematic review and meta-analysis comparing seton drainage and anti-tumour necrosis factor treatment. Colorectal Disease 18: 667–675. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).