4. Materials and Methods
4.1. Materials
Reagents and solvents of the purest grade available for GC-MS, hydrochloric acid, trifluoroacetic acid, thionyl chloride, dry methanol, MeONa in dry MeOH (25% solution), sodium hydroxide and Amberlite IR120 were supplied by Sigma Aldrich. Silica gel chromatography was purchased from Merck (Supelco, high-purity grade, pore size 60 Å, 230-400 mesh particle size). Absolute ethanol, diisopropyl ether and hexane were purchased from Carlo Erba, potassium hydroxide was acquired from Panreac and anhydrous sodium sulphate (Na₂SO₄) from Alfa Aesar GmbH (Karlsruhe, Germany).
Deionized water was obtained using a Milli-Q Water purification system (Millipore Corporation, Bedford, MA). were purchased from Sigma-Aldrich.
Dry milled corn and wheat bran where kindly provided by Mulino Sobrino, La Morra (CN), Italy and stored in a freezer at -20°C.
4.2. Instruments
Centrifugation was performed with a 5804-R (Eppendorf s.r.l., Milan, Italy) centrifuge. Microwave (MW) assisted solid-liquid extraction was conducted with a Biotage initiator+ instrument (Uppsala, Sweden) with a maximum power of irradiation of 400W (2.45 GHz).
GC/MS analyses were carried out on a Thermo Scientific DSQII single quadrupole GC/MS system (TraceDSQII mass spectrometer, Trace GC Ultra gas chromatograph, TriPlus autosampler - ThermoFisher Scientific, Waltham, MA, USA).
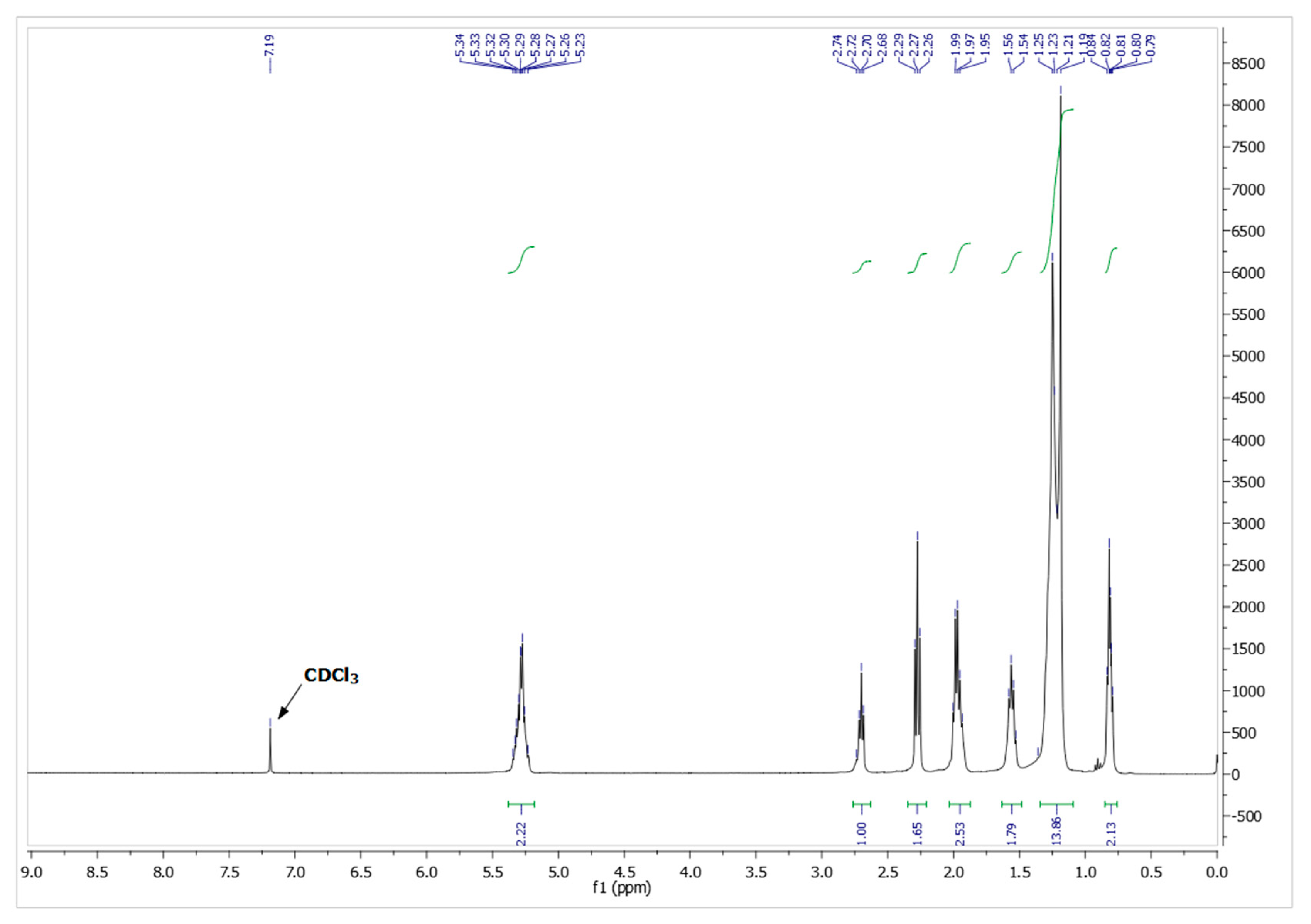
The characterization of purified FFA mixtures were performed recording the NMR spectra on a Bruker Advance III spectrometer operating at 400 MHz (Bruker Corporation, Billerica, MA, USA) setting standard pulse sequences provided in the Bruker Topspin 3.6 software. Chemical shifts of 1H NMR spectra are reported in parts per million (ppm) and the coupling constants are measured in Hertz (Hz).
4.3. Microwave-Assisted Extraction of Fatty Acids from Wheat and Corn
Dry milled corn was initially used to carry out preliminary extractions combining microwave irradiation with the use of organic solvents. For the purpose, a variety of organic solvents have been tested. Specifically, 1g of milled corn was suspended in 10 mL of either n-hexane, dichloromethane, ethanol or isopropanol and subjected to microwave extraction. The procedure was carried out for 10 min, setting the temperature at the boiling point of the solvent: n-hexane (70°C), dichloromethane (40°C), isopropanol (82°C) and ethanol (78°C). The suspension was then filtered over a sintered glass filter and the solvent evaporated in vacuo. The thus obtained oily residue was transesterified with MeONa in dry MeOH (25% solution) under inert atmosphere. The reaction was monitored through TLC and quenched with Amberlite IR120 upon complete consumption of the starting material. The acidic resin was removed through filtration and the solvent dried under reduced pressure. The fatty acids methyl esters (FAME) were thus characterized by GC-MS without further purification. The extraction procedure afforded the fatty acids mixture as FAME, but for subsequent conjugation with hydrophilic substances, pure free fatty acids (FFAs) were required. Due to the formation of by-products during the hydrolysis of FAME, the conditions were modified to directly obtain FFAs, bypassing the intermediate steps.
Thus, the extraction and hydrolysis were conducted simultaneously under the following conditions: dry milled corn (1 g) was suspended in 10 mL of 1M NaOH solution and heated in a microwave at 100°C for 10 minutes, followed by filtration through a Büchner funnel. The aqueous solution was extracted with n-hexane (3 x 50 mL) to remove the unsaponifiable fraction. The organic layers were collected, washed with water, and allowed to equilibrate before separation. The combined aqueous fractions were acidified with 6N HCl to pH 1 to release the free fatty acids, which were then extracted with n-hexane (3 x 100 mL). The organic phases were washed with water, with NaCl added to clear any remaining emulsion, and allowed to settle before separation. After drying with anhydrous Na2SO4, the solvent was filtered and evaporated under reduced pressure at 35°C. The crude product was purified by flash chromatography using n-hexane and diisopropyl ether (60/40 v/v) with the addition of 0.2% trifluoroacetic acid. After solvent removal, the oily residue was dried in vacuo to constant weight.
The focus was then directed to wheat bran, which offers significant potential for contributing to circular economy efforts. The aforementioned procedure was thus repeated on wheat bran and to overcome issues of viscosity of the media, which significantly hindered handling, the procedure was modified accordingly. This involved changing the media composition and utilizing a centrifuge instead of a Büchner funnel, enhancing manageability and efficiency.
In addition to these modifications, we switched from measuring the solvent volume from millilitres (mL) to grams (g) for improved accuracy.
Thus, a suspension of 1g of wheat bran in absolute ethanol was prepared and an aqueous solution of 20M KOH was added. The heterogeneous mixture was then heated in a microwave reactor at 78°C with magnetic stirring. Two extraction media were tested, differing in water content: SA, which included an additional 5% water, and SB, which contained no extra water. Specifically, SA was prepared by mixing absolute ethanol, 20M KOH, and water in the ratios of 15.3g ethanol, 1.8g KOH, and 0.9g water for a total of 18g. For smaller batches, 5.1g ethanol, 0.6g KOH, and 0.3g water were combined to yield 6g of SA. SB was prepared using 16.2g of ethanol and 1.8g of 20M KOH for 18g of solution, while the 6g batch consisted of 5.4g ethanol and 0.6g KOH.
The total quantity of extraction media was determined based on the number of cycles. A single 30-minute extraction was performed using 18g of the ethanolic solution, while for a three-cycle process, 6g of solvent was used for each 10-minute extraction. After each cycle, the solvent was replaced with fresh 6g aliquots, reaching a total of 18g over three cycles. Additionally, a validation experiment was carried out using two consecutive 15-minute extraction cycles.
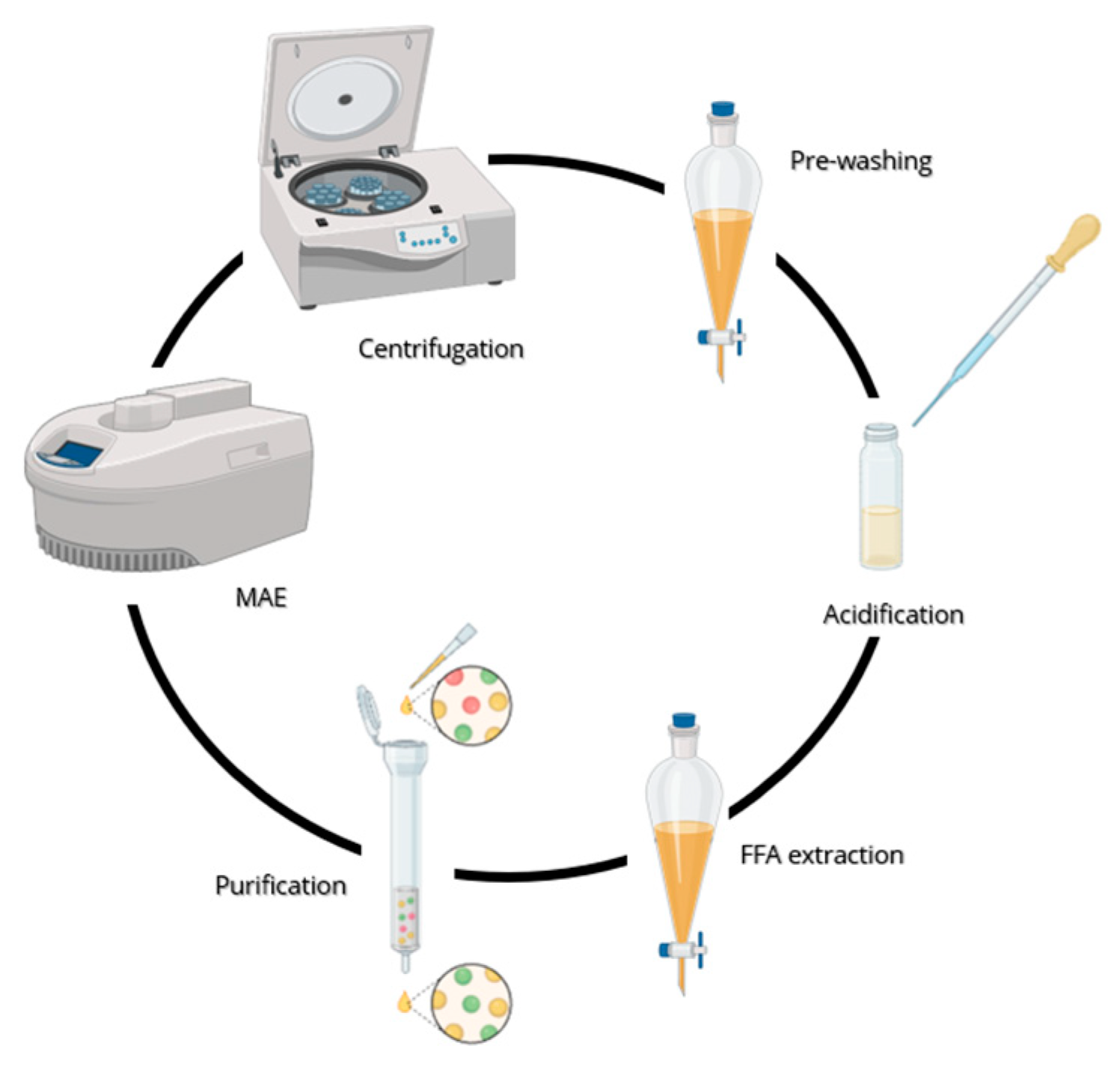
Once the solid-liquid extraction was completed, the ethanolic solution was collected, diluted in 20 mL of deionized water and centrifuged (4500g) for 20 min at room temperature (25°C) to allow the wheat residue to settle. The solvent was thus removed and replaced with fresh water (20 mL) before another centrifuge cycle. Overall, the process was repeated 4 times. The aqueous solution was then subjected to hexane washes (3 x 50 mL) in order to remove the lipophile impurities. Hexane was then collected and extracted with water (2 x 100 mL). The aqueous fractions were thus gathered all together and acidified to pH=1 with HCl 6N to induce the protonation of the extracted fatty acids. Subsequently, hexane extractions were performed (3 x 100 mL) and the collected organic phase was then washed with water (3 x 100 mL). Hexane was thus treated with dry Na
2SO
4, filtered and evaporated under reduced pressure. The oily residue was let dry
in vacuo to constant weight and the crude product purified through silica gel chromatography (60:40, hexane: diisopropyl ether (v/v) + 0.2% trifluoroacetic acid). A schematic representation of the extraction of the vegetable oil from wheat bran is provided in
Figure 4.
4.4. Optimization of Liquid-Liquid Extraction
In the framework of circular economy principles, the primary objective was to minimize the use of organic solvents. Therefore, the liquid-liquid extraction following microwave-assisted extraction was optimized. To achieve this, the experiment involving the suspension of 1g of wheat bran in SB for a single 30-minute extraction cycle was replicated using the revised methodology. This procedure was repeated three times to ensure data comparability.
Initially, the volume of water used to dilute the ethanolic solution and wash the wheat bran residue upon centrifugation was reduced from 80 mL to 40 mL: three centrifugations were performed using 20 mL at first and 10 mL for the last two rounds (1 x 20 mL + 2 x 10 mL). Similarly, the volume of hexane for subsequent washes was decreased from 50 mL to 30 mL (3 x 30 mL), and the final water wash volume was reduced from 100 mL to 50 mL (3 x 50 mL). After acidifying the aqueous phase with HCl (4 mL), the extraction was carried out using 50 mL of hexane (3 x 50 mL), while water was reduced to 50 mL (3 x 50 mL). The crude product was purified through silica gel chromatography using a mixture of hexane and diisopropyl ether (60:40, hexane:diisopropyl ether (v/v)) with the addition of 0.2% trifluoroacetic acid and the oily residue was then let dry in vacuo to constant weight.
Moreover, in order to further reduce solvent consume and streamline the process, a final column chromatography replaced the pre-washes before acidification, making the workup less time-consuming without compromising the quality of the extraction.
4.5. Esterification of the Free Fatty Acids Mixture
Aiming to determine the composition of the isolated products, the free fatty acids were treated with thionyl chloride for the conversion into methyl esters fatty acids (FAME). The overall reaction can be summarized as follows:
Referring to the literature [
18], the oily residue was dissolved in dry MeOH in a round bottomed flask filled with nitrogen and placed in an ice-bath at 0°C. Thionyl chloride (1.5 eq.) was then added in a dropwise manner under stirring. In the end, the flask was sealed and the mixture let react for 1h. The progression of the reaction was monitored through TLC. Upon completion, the solvent was evaporated
in vacuo, and the product used without any further purification.
4.6. Determination of Corn Kernels and Wheat Bran Moisture Content
Dry milled corn kernels and wheat bran (500 mg) were placed in an oven heated at 100°C till constant weight, aiming to determine their humidity content.
Given the initial weight of the samples (W
i) and their weight after the thermal treatment (W
f), the moisture content was calculated as follows:
The yield of each extraction process was determined taking into account the moisture content of the sample to ensure accurate determination of the dry matter extracted. The assays were performed in triplicate and the results are expressed as the mean ± standard deviation.
4.7. Oil Extraction Yield
The oil extraction yield for the overall process was calculated as the ratio of the grams of oil obtained to the weight of corn or wheat suspended in the solvent, expressed as a percentage:
Specifically, for the preliminary extractions with corn the amount of oil in the numerator refers to the product obtained after the liquid-liquid extraction, since the mixture was analysed without further purification. In contrast, the yield for wheat was calculated after the silica gel chromatography purification and taking into account the moisture content.
4.8. Solutions Preparation
In the context of the optimization of extraction procedures, the preparation of two different extraction media, SA and SB, was undertaken. These media differed in their water content to assess their efficiency in extracting free fatty acids from wheat bran.
For the preparation of SA, the mixture included absolute ethanol, a 20M KOH solution, and an additional 5% of water. Specifically, to prepare 18 grams of SA, 15.3 grams of absolute ethanol were combined with 1.8 grams of 20M KOH and 0.9 grams of water. For the samples subjected to three MW extraction, 6 grams of SA were prepared for each cycle by mixing 5.1 grams of absolute ethanol, 0.6 grams of 20M KOH, and 0.3 grams of water.
Conversely, the preparation of SB did not include any additional water, relying solely on absolute ethanol and the 20M KOH solution. To prepare 18 grams of SB, 16.2 grams of absolute ethanol were mixed with 1.8 grams of 20M KOH. Similarly, for 6 grams, SB was prepared using 5.4 grams of absolute ethanol and 0.6 grams of 20M KOH.
The control experiment was then conducted preparing 6 grams of the extraction media and using it for two 15-minute extraction cycles.
SA and SB were freshly prepared for each use and utilized immediately thereafter.
20M KOH was freshly prepared in a volumetric flask daily in 5 mL aliquots to minimize waste and was utilized throughout the day. If there was a delay between preparations, the solution was stored in the refrigerator (4°C) for a few hours to maintain stability.
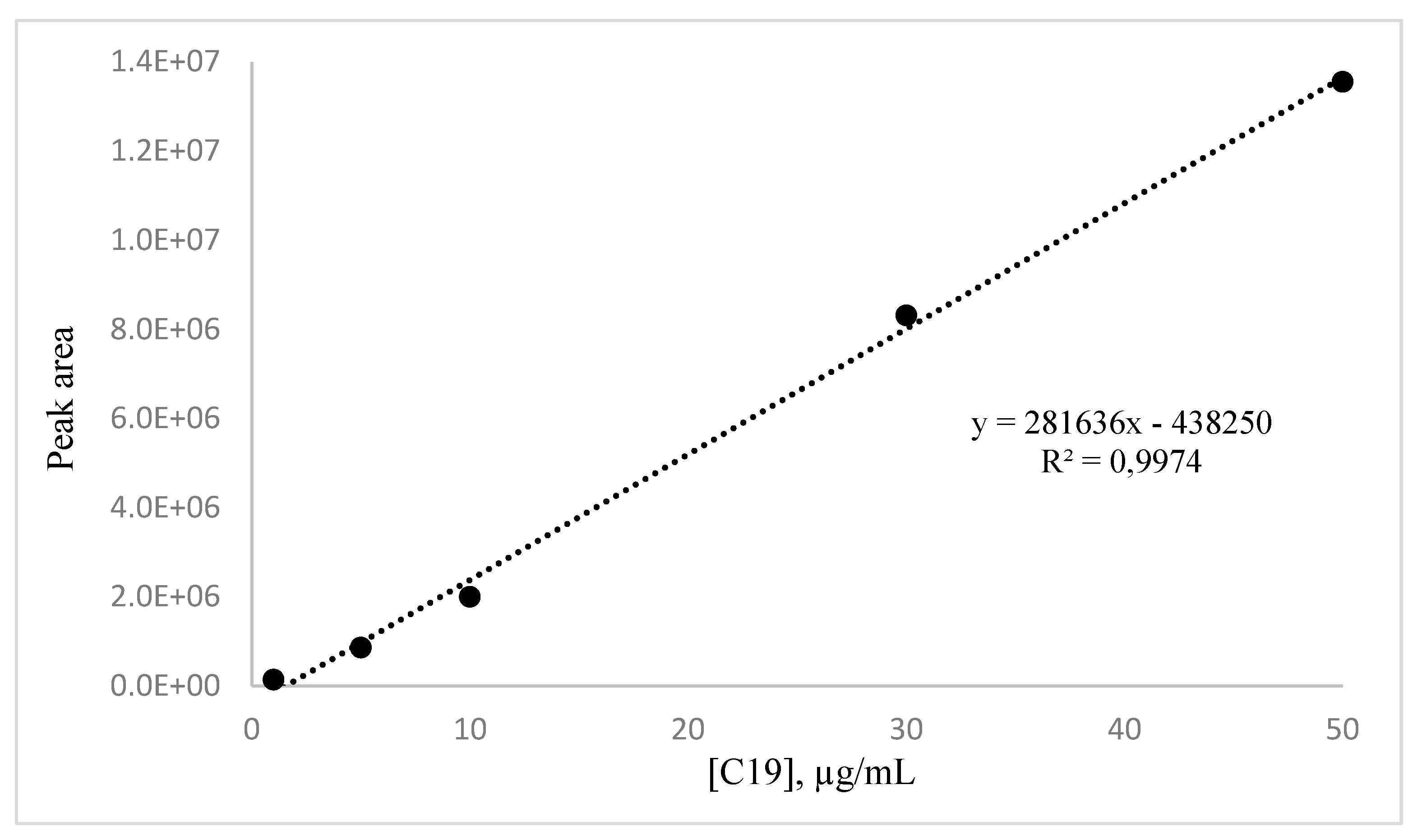
The calibration curve of nonadecanoic acid (C19) was built through GC-MS. In order to detect the standard, it was at first esterified as described in paragraph 4.5.
A stock solution of methyl nonadecanoate (1 mg/mL) was prepared by dissolving the standard in analytical-grade DCM and stored at -20°C. For the linearity study, the stock solution was appropriately diluted with DCM to achieve concentrations ranging from 1 to 50 µg/mL (3.3-167.5 µM).
4.9. GC-MS for Fatty Acids Analysis
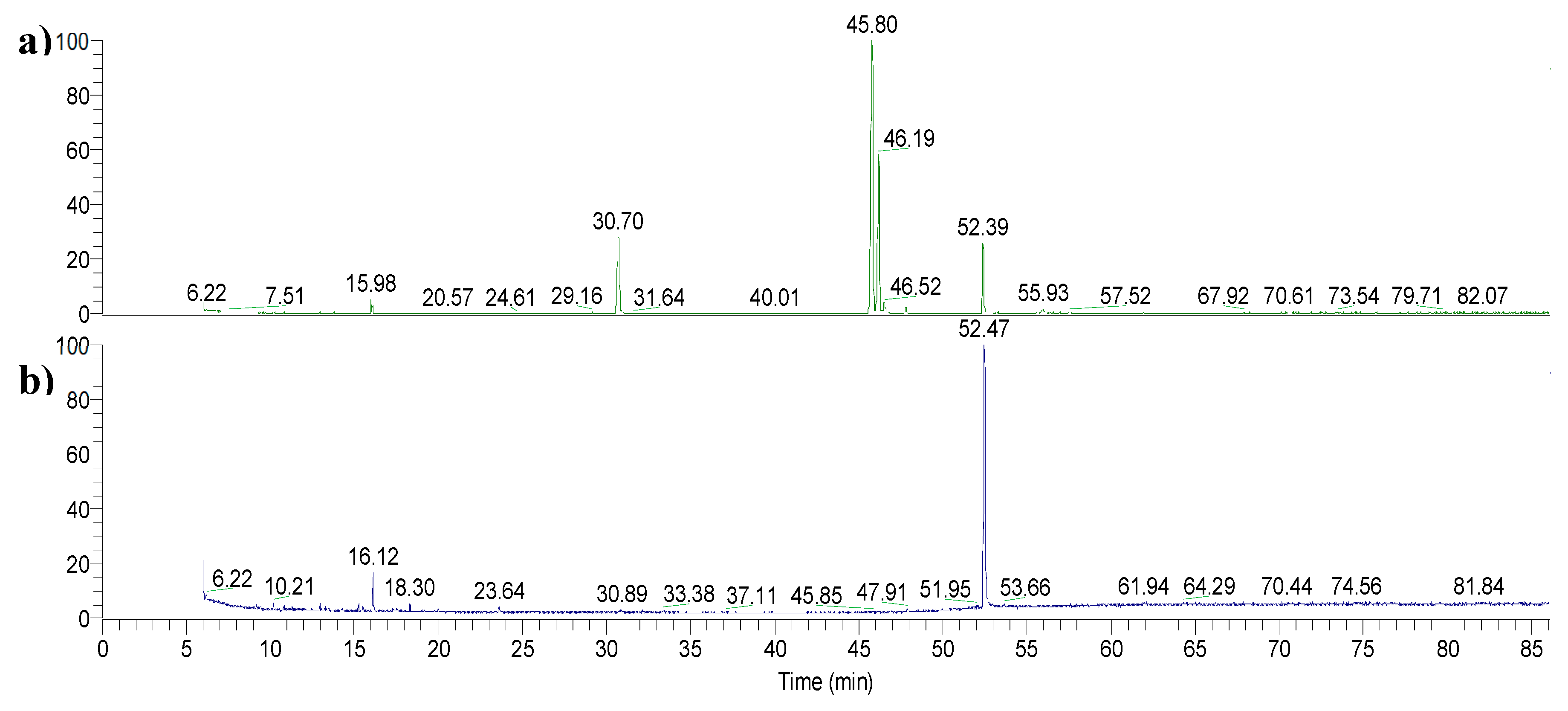
The esterified fatty acids were dissolved in analytical-grade dichloromethane (DCM) and appropriately diluted to ensure optimal conditions for GC-MS analysis. Chromatography was performed on a Rxi-5Sil MS capillary column (30 m length x 0.25 mm ID x 0.25 µm film thickness, Restek, Milan, Italy) with helium (>99.99%) as the carrier gas at a constant flow rate of 1.0 mL/min. An injection volume of 1 µL was employed, with the injector temperature set at 250°C and operated in split mode, with a split flow of 10 mL/min. The oven temperature program started at 45°C (isothermal for 4 min), then increased to 175°C at a rate of 13°C/min, and finally to 215°C (isothermal for 35 min) at a rate of 4°C/min. The mass transfer line temperature was set at 250°C and the total GC runtime was 86 minutes. Mass spectra were acquired using an electron ionization system (EI, Electron Impact mode) with ionization energy of 70 eV, a source temperature of 250°C and a spectral acquisition in Full Scan mode, positive polarity, over a mass range of 35-650 Da with a scan rate of 804 amu/s.
Chromatogram acquisition, mass spectral peak detection, and waveform processing were carried out using Xcalibur MS Software Version 2.1 (Thermo Scientific Inc.). Chemical structures were assigned to chromatographic peaks by comparing the mass spectra with the databases from the NIST Mass Spectral Library (NIST 08) and the Wiley Registry of Mass Spectral Data (8th Edition).
FAMEs were identified by extracting their molecular ions from the total ion current, and the peaks at their corresponding retention times were integrated.
4.10. Wheat Oil Composition
The composition of wheat oil extracts was analysed using GC-MS to determine the area percentage and relative concentration of each fatty acid component. The relative abundance of each fatty acid methyl ester (FAME) was calculated by integrating the area under each FAME peak in the GC chromatogram. Specifically, the relative abundance was determined by dividing the area of each FAME peak by the total area of all FAME peaks and then multiplying by 100, yielding the percentage of each fatty acid relative to the overall mixture.
4.11. C19 Standard Calibration Curve
Solutions of methyl nonadecanoate acid with concentrations ranging from 1 to 50 µg/mL (3.3-167.5 µM) were prepared and injected in the gas chromatography system. Linearity was evaluated across five concentration levels, with three replicates run for each concentration. Calibration curves were generated using linear regression analysis.
4.12. Precision
The precision of the method for methyl nonadecanoate was evaluated by examining the repeatability of the data. Five standard solutions, each representing a concentration within the range of the calibration curve, were analyzed in triplicate. The relative standard deviations (RSDs) of the peak areas at each concentration level were calculated, along with the overall average RSD, to assess precision. The results are expressed as the mean ± standard deviation (SD).
4.13. Internal Standard Recovery
The optimized MAE conditions were applied to monitor the overall efficiency of the extraction process. For the purpose, nonadecanoic acid (C19) (4 mg) was employed to spike the extraction medium in which 1g of wheat bran was suspended in 16.2g of absolute ethanol and 1.8g of 20M KOH solution. The sealed flask was subjected to microwave extraction for 10 minutes at 78°C, followed by a simplified work-up, omitting the pre-washing steps before acidification as described in paragraph 4.4. The mixture of free fatty acids (FFAs) was subsequently purified, esterified, and analysed via GC-MS. All experiments were performed in triplicate to ensure repeatability and accuracy.
The internal standard signal was identified by extracting its ion current and integrating the corresponding peak. The peak area for each batch was determined and applied to the calibration curve equation to quantify the reference compound. Recovery (%) was calculated using the following formula:
The data collected are expressed as the mean recovery (%) with the corresponding RSD.