Submitted:
23 January 2025
Posted:
23 January 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Prostate-Specific Membrane Antigens
PSMA PET-CT for Imaging Prostate Cancer
177LU-PSMA Radiotherapy
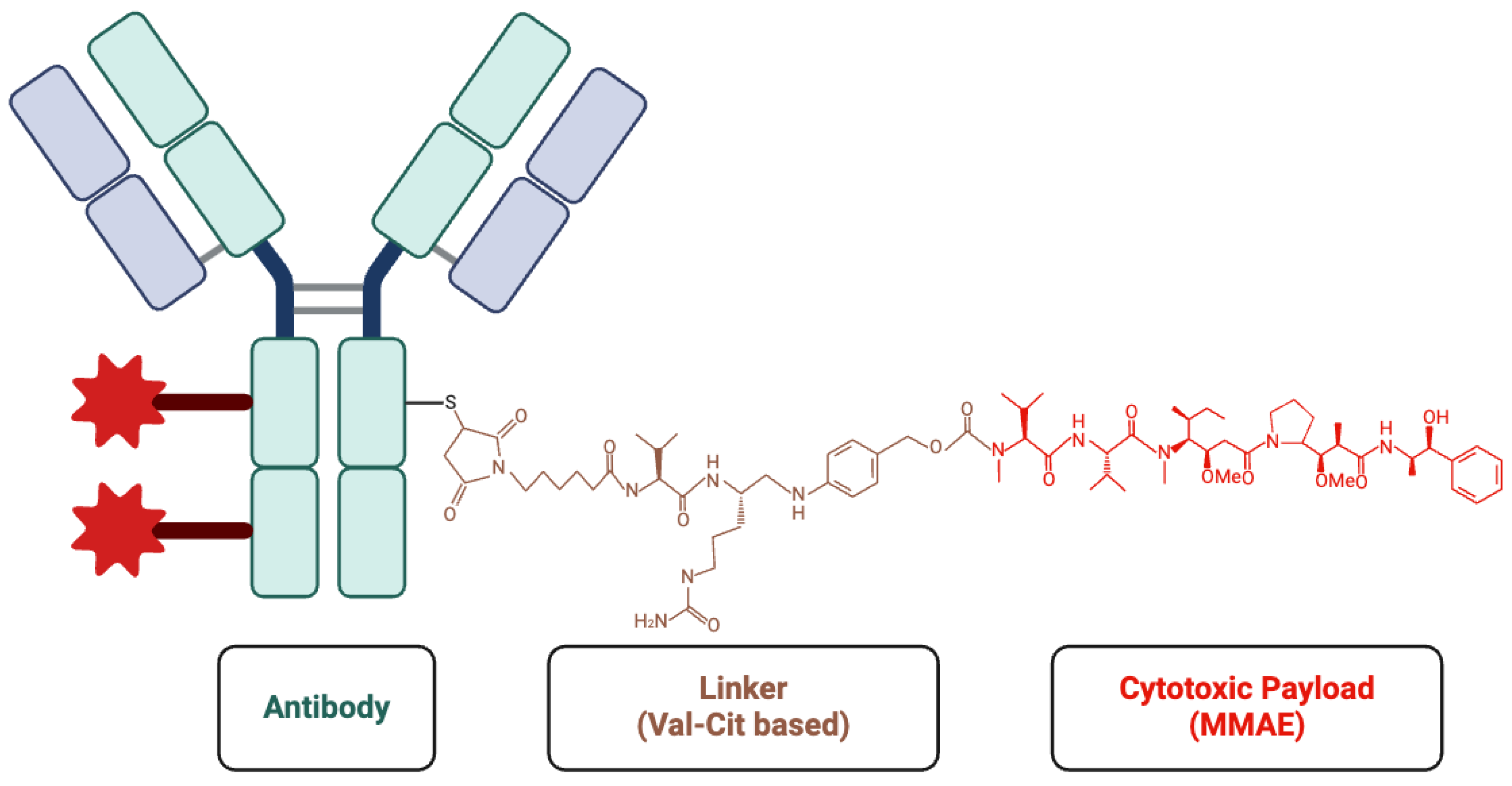
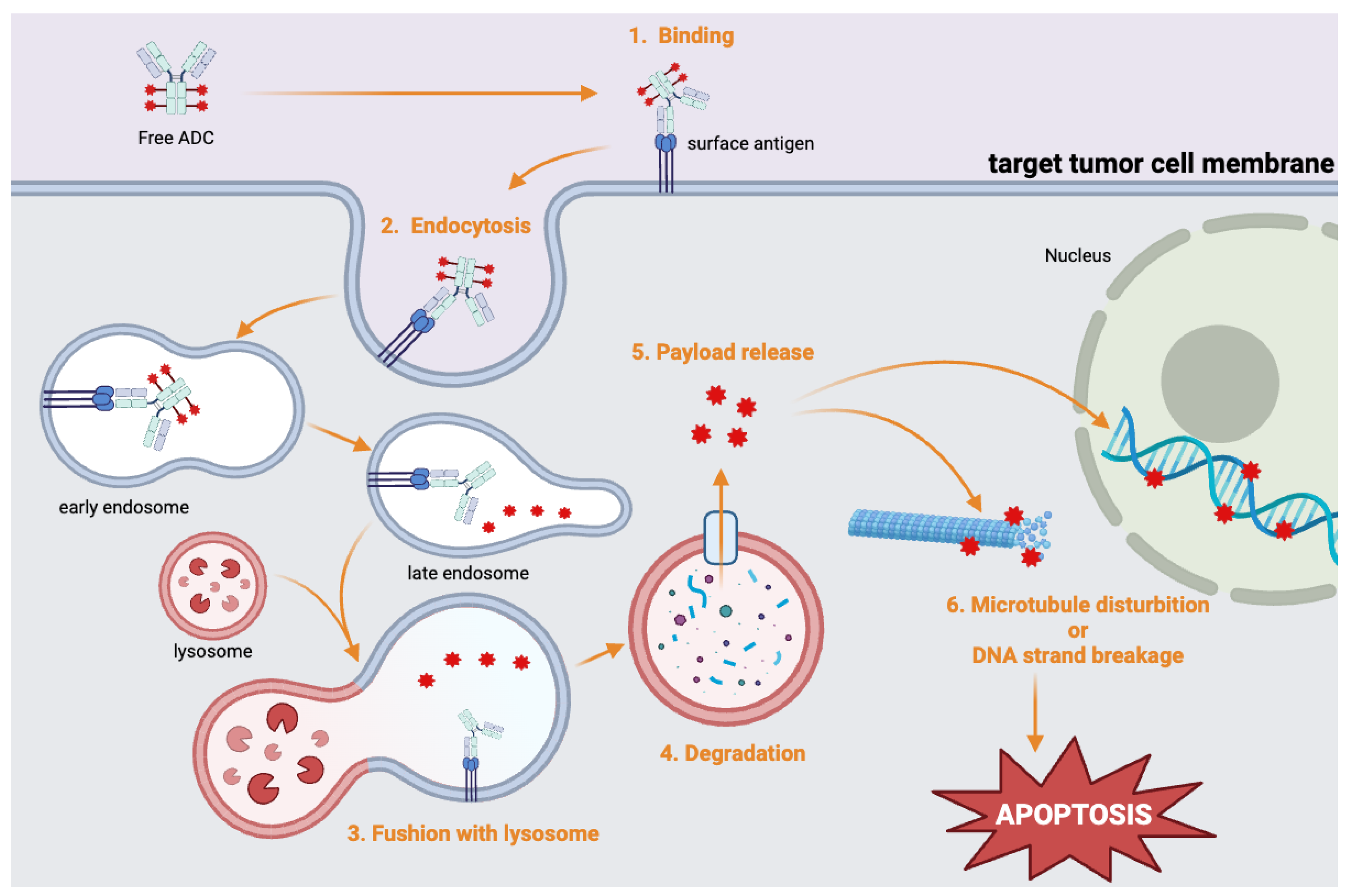
Antibody–Drug Conjugates (ADCs)
Anti-PSMA Monoclonal Antibodies
7E11
J591
5. D3
ADCs Targeting PSMA in PC
PSMA-MMAE
NCT01414283
NCT01695044
ARX517
NCT04662580
MLN2704
MEDI3726
NCT02991911
Discussion
Conclusions
References
- Rebello, R.J.; Oing, C.; Knudsen, K.E.; Loeb, S.; Johnson, D.C.; Reiter, R.E.; Gillessen, S.; Van der Kwast, T.; Bristow, R.G. Prostate cancer. Nat Rev Dis Primers 2021, 7, 9. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J Clin 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Henríquez, I.; Roach, M., 3rd; Morgan, T.M.; Bossi, A.; Gómez, J.A.; Abuchaibe, O.; Couñago, F. Current and Emerging Therapies for Metastatic Castration-Resistant Prostate Cancer (mCRPC). Biomedicines 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Francini, E.; Gray, K.P.; Shaw, G.K.; Evan, C.P.; Hamid, A.A.; Perry, C.E.; Kantoff, P.W.; Taplin, M.E.; Sweeney, C.J. Impact of new systemic therapies on overall survival of patients with metastatic castration-resistant prostate cancer in a hospital-based registry. Prostate Cancer Prostatic Dis 2019, 22, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S. Overview of prostate-specific membrane antigen. Rev Urol 2004, 6 (Suppl. 10), S13–S18. [Google Scholar] [PubMed]
- Perner, S.; Hofer, M.D.; Kim, R.; Shah, R.B.; Li, H.; Möller, P.; Hautmann, R.E.; Gschwend, J.E.; Kuefer, R.; Rubin, M.A. Prostate-specific membrane antigen expression as a predictor of prostate cancer progression. Hum Pathol 2007, 38, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Thompson, I.M.; Ankerst, D.P.; Chi, C.; Goodman, P.J.; Tangen, C.M.; Lucia, M.S.; Feng, Z.; Parnes, H.L.; Coltman, C.A., Jr. Assessing prostate cancer risk: results from the Prostate Cancer Prevention Trial. J Natl Cancer Inst 2006, 98, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Hövels, A.M.; Heesakkers, R.A.; Adang, E.M.; Jager, G.J.; Strum, S.; Hoogeveen, Y.L.; Severens, J.L.; Barentsz, J.O. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol 2008, 63, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Kahn, D.; Williams, R.D.; Seldin, D.W.; Libertino, J.A.; Hirschhorn, M.; Dreicer, R.; Weiner, G.J.; Bushnell, D.; Gulfo, J. Radioimmunoscintigraphy with 111indium labeled CYT-356 for the detection of occult prostate cancer recurrence. J Urol 1994, 152, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Adnan, A.; Basu, S. PSMA Receptor-Based PET-CT: The Basics and Current Status in Clinical and Research Applications. Diagnostics (Basel) 2023, 13. [Google Scholar] [CrossRef]
- Awenat, S.; Piccardo, A.; Carvoeiras, P.; Signore, G.; Giovanella, L.; Prior, J.O.; Treglia, G. Diagnostic Role of (18)F-PSMA-1007 PET/CT in Prostate Cancer Staging: A Systematic Review. Diagnostics (Basel) 2021, 11. [Google Scholar] [CrossRef]
- Satapathy, S.; Singh, H.; Kumar, R.; Mittal, B.R. Diagnostic Accuracy of (68)Ga-PSMA PET/CT for Initial Detection in Patients With Suspected Prostate Cancer: A Systematic Review and Meta-Analysis. AJR Am J Roentgenol 2021, 216, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Jochumsen, M.R.; Bouchelouche, K. PSMA PET/CT for Primary Staging of Prostate Cancer - An Updated Overview. Seminars in Nuclear Medicine 2024, 54, 39–45. [Google Scholar] [CrossRef]
- Taneja, S.S. Imaging in the diagnosis and management of prostate cancer. Rev Urol 2004, 6, 101–113. [Google Scholar]
- Abuzallouf, S.; Dayes, I.; Lukka, H. Baseline staging of newly diagnosed prostate cancer: a summary of the literature. J Urol 2004, 171, 2122–2127. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Weber, M.; Iravani, A.; Hofman, M.S.; Calais, J.; Czernin, J.; Ilhan, H.; Saad, F.; Small, E.J.; Smith, M.R.; Perez, P.M.; Hope, T.A.; Rauscher, I.; Londhe, A.; Lopez-Gitlitz, A.; Cheng, S.; Maurer, T.; Herrmann, K.; Eiber, M.; Hadaschik, B. Prostate-Specific Membrane Antigen Ligand Positron Emission Tomography in Men with Nonmetastatic Castration-Resistant Prostate Cancer. Clin Cancer Res 2019, 25, 7448–7454. [Google Scholar] [CrossRef] [PubMed]
- Hennrich, U.; Eder, M. [(177)Lu]Lu-PSMA-617 (Pluvicto(TM)): The First FDA-Approved Radiotherapeutical for Treatment of Prostate Cancer. Pharmaceuticals (Basel) 2022, 15. [Google Scholar] [CrossRef]
- Benešová, M.; Bauder-Wüst, U.; Schäfer, M.; Klika, K.D.; Mier, W.; Haberkorn, U.; Kopka, K.; Eder, M. Linker Modification Strategies To Control the Prostate-Specific Membrane Antigen (PSMA)-Targeting and Pharmacokinetic Properties of DOTA-Conjugated PSMA Inhibitors. J Med Chem 2016, 59, 1761–1775. [Google Scholar] [CrossRef]
- Afshar-Oromieh, A.; Hetzheim, H.; Kratochwil, C.; Benesova, M.; Eder, M.; Neels, O.C.; Eisenhut, M.; Kübler, W.; Holland-Letz, T.; Giesel, F.L.; Mier, W.; Kopka, K.; Haberkorn, U. The Theranostic PSMA Ligand PSMA-617 in the Diagnosis of Prostate Cancer by PET/CT: Biodistribution in Humans, Radiation Dosimetry, and First Evaluation of Tumor Lesions. J Nucl Med 2015, 56, 1697–1705. [Google Scholar] [CrossRef]
- Alati, S.; Singh, R.; Pomper, M.G.; Rowe, S.P.; Banerjee, S.R. Preclinical Development in Radiopharmaceutical Therapy for Prostate Cancer. Semin Nucl Med 2023, 53, 663–686. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, R.; Chakraborty, S. A review of advances in the last decade on targeted cancer therapy using (177)Lu: focusing on (177)Lu produced by the direct neutron activation route. Am J Nucl Med Mol Imaging 2021, 11, 443–475. [Google Scholar] [PubMed]
- NCCN Clinical Practice Guidelines in Oncology, Prostate Cancer, Version 4. National Comprehensive Cancer Network 2024, 33.
- Hofman, M.S.; Emmett, L.; Sandhu, S.; Iravani, A.; Joshua, A.M.; Goh, J.C.; Pattison, D.A.; Tan, T.H.; Kirkwood, I.D.; Ng, S.; Francis, R.J.; Gedye, C.; Rutherford, N.K.; Weickhardt, A.; Scott, A.M.; Lee, S.T.; Kwan, E.M.; Azad, A.A.; Ramdave, S.; Redfern, A.D.; Macdonald, W.; Guminski, A.; Hsiao, E.; Chua, W.; Lin, P.; Zhang, A.Y.; McJannett, M.M.; Stockler, M.R.; Violet, J.A.; Williams, S.G.; Martin, A.J.; Davis, I.D. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet 2021, 397, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Van Wambeke, S.; Vera-Badillo, F.E.; Gyawali, B. Controlling the Control Arm in Metastatic Castration-Resistant Prostate Cancer Trials: Best Standard of Care or the Minimum Standard of Care? J Clin Oncol 2022, 40, 1518–1521. [Google Scholar] [CrossRef] [PubMed]
- Drake, P.M.; Rabuka, D. Recent Developments in ADC Technology: Preclinical Studies Signal Future Clinical Trends. BioDrugs 2017, 31, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Gogia, P.; Ashraf, H.; Bhasin, S.; Xu, Y. Antibody-Drug Conjugates: A Review of Approved Drugs and Their Clinical Level of Evidence. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.H.; Lin, Y.H.; Luo, H.L.; Sung, W.W. Antibody-drug conjugates targeting HER2 for the treatment of urothelial carcinoma: potential therapies for HER2-positive urothelial carcinoma. Front Pharmacol 2024, 15, 1326296. [Google Scholar] [CrossRef] [PubMed]
- Nagayama, A.; Ellisen, L.W.; Chabner, B.; Bardia, A. Antibody-Drug Conjugates for the Treatment of Solid Tumors: Clinical Experience and Latest Developments. Target Oncol 2017, 12, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Smith, S.W.; Ghone, S.; Tomczuk, B. Current ADC Linker Chemistry. Pharm Res 2015, 32, 3526–3540. [Google Scholar] [CrossRef] [PubMed]
- Waight, A.B.; Bargsten, K.; Doronina, S.; Steinmetz, M.O.; Sussman, D.; Prota, A.E. Structural Basis of Microtubule Destabilization by Potent Auristatin Anti-Mitotics. PLoS One 2016, 11, e0160890. [Google Scholar] [CrossRef]
- Voigt, W.; Matsui, S.; Yin, M.B.; Burhans, W.C.; Minderman, H.; Rustum, Y.M. Topoisomerase-I inhibitor SN-38 can induce DNA damage and chromosomal aberrations independent from DNA synthesis. Anticancer Res 1998, 18, 3499–3505. [Google Scholar]
- Miyahira, A.K.; Pienta, K.J.; Morris, M.J.; Bander, N.H.; Baum, R.P.; Fendler, W.P.; Goeckeler, W.; Gorin, M.A.; Hennekes, H.; Pomper, M.G.; Sartor, O.; Tagawa, S.T.; Williams, S.; Soule, H.R. Meeting report from the Prostate Cancer Foundation PSMA-directed radionuclide scientific working group. Prostate 2018, 78, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Wittrup, K.D.; Thurber, G.M.; Schmidt, M.M.; Rhoden, J.J. Practical theoretic guidance for the design of tumor-targeting agents. Methods Enzymol 2012, 503, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.M.; Tannock, I.F. The distribution of the therapeutic monoclonal antibodies cetuximab and trastuzumab within solid tumors. BMC Cancer 2010, 10, 255. [Google Scholar] [CrossRef] [PubMed]
- Troyer, J.K.; Feng, Q.; Beckett, M.L.; Wright, G.L., Jr. Biochemical characterization and mapping of the 7E11-C5.3 epitope of the prostate-specific membrane antigen. Urol Oncol 1995, 1, 29–37. [Google Scholar] [CrossRef]
- Barren, R.J., 3rd; Holmes, E.H.; Boynton, A.L.; Misrock, S.L.; Murphy, G.P. Monoclonal antibody 7E11.C5 staining of viable LNCaP cells. Prostate 1997, 30, 65–68. [Google Scholar] [CrossRef]
- Aloysius, H.; Hu, L. Targeted prodrug approaches for hormone refractory prostate cancer. Med Res Rev 2015, 35, 554–585. [Google Scholar] [CrossRef] [PubMed]
- Tagawa, S.T.; Thomas, C.; Sartor, A.O.; Sun, M.; Stangl-Kremser, J.; Bissassar, M.; Vallabhajosula, S.; Huicochea Castellanos, S.; Nauseef, J.T.; Sternberg, C.N.; Molina, A.; Ballman, K.; Nanus, D.M.; Osborne, J.R.; Bander, N.H. Prostate-Specific Membrane Antigen-Targeting Alpha Emitter via Antibody Delivery for Metastatic Castration-Resistant Prostate Cancer: A Phase I Dose-Escalation Study of (225)Ac-J591. J Clin Oncol 2024, 42, 842–851. [Google Scholar] [CrossRef]
- Holland, J.P.; Divilov, V.; Bander, N.H.; Smith-Jones, P.M.; Larson, S.M.; Lewis, J.S. 89Zr-DFO-J591 for immunoPET of prostate-specific membrane antigen expression in vivo. J Nucl Med 2010, 51, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Nováková, Z.; Foss, C.A.; Copeland, B.T.; Morath, V.; Baranová, P.; Havlínová, B.; Skerra, A.; Pomper, M.G.; Barinka, C. Novel Monoclonal Antibodies Recognizing Human Prostate-Specific Membrane Antigen (PSMA) as Research and Theranostic Tools. Prostate 2017, 77, 749–764. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.R.; Kumar, V.; Lisok, A.; Plyku, D.; Nováková, Z.; Brummet, M.; Wharram, B.; Barinka, C.; Hobbs, R.; Pomper, M.G. Evaluation of (111)In-DOTA-5D3, a Surrogate SPECT Imaging Agent for Radioimmunotherapy of Prostate-Specific Membrane Antigen. J Nucl Med 2019, 60, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Petrylak, D.P.; Kantoff, P.; Vogelzang, N.J.; Mega, A.; Fleming, M.T.; Stephenson, J.J., Jr.; Frank, R.; Shore, N.D.; Dreicer, R.; McClay, E.F.; Berry, W.R.; Agarwal, M.; DiPippo, V.A.; Rotshteyn, Y.; Stambler, N.; Olson, W.C.; Morris, S.A.; Israel, R.J. Phase 1 study of PSMA ADC, an antibody-drug conjugate targeting prostate-specific membrane antigen, in chemotherapy-refractory prostate cancer. Prostate 2019, 79, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Petrylak, D.P.; Vogelzang, N.J.; Chatta, K.; Fleming, M.T.; Smith, D.C.; Appleman, L.J.; Hussain, A.; Modiano, M.; Singh, P.; Tagawa, S.T.; Gore, I.; McClay, E.F.; Mega, A.E.; Sartor, A.O.; Somer, B.; Wadlow, R.; Shore, N.D.; Olson, W.C.; Stambler, N.; DiPippo, V.A.; Israel, R.J. PSMA ADC monotherapy in patients with progressive metastatic castration-resistant prostate cancer following abiraterone and/or enzalutamide: Efficacy and safety in open-label single-arm phase 2 study. Prostate 2020, 80, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Doehn, C.; Jocham, D. Technology evaluation: MLN-591, Cornell University/BZL Biologics/ImmunoGen/Millennium. Curr Opin Mol Ther 2002, 4, 606–613. [Google Scholar] [PubMed]
- Milowsky, M.I.; Galsky, M.D.; Morris, M.J.; Crona, D.J.; George, D.J.; Dreicer, R.; Tse, K.; Petruck, J.; Webb, I.J.; Bander, N.H.; Nanus, D.M.; Scher, H.I. Phase 1/2 multiple ascending dose trial of the prostate-specific membrane antigen-targeted antibody drug conjugate MLN2704 in metastatic castration-resistant prostate cancer. Urol Oncol 2016, 34, 530.e15–530.e21. [Google Scholar] [CrossRef]
- Cho, S.; Zammarchi, F.; Williams, D.G.; Havenith, C.E.G.; Monks, N.R.; Tyrer, P.; D'Hooge, F.; Fleming, R.; Vashisht, K.; Dimasi, N.; Bertelli, F.; Corbett, S.; Adams, L.; Reinert, H.W.; Dissanayake, S.; Britten, C.E.; King, W.; Dacosta, K.; Tammali, R.; Schifferli, K.; Strout, P.; Korade, M., 3rd; Masson Hinrichs, M.J.; Chivers, S.; Corey, E.; Liu, H.; Kim, S.; Bander, N.H.; Howard, P.W.; Hartley, J.A.; Coats, S.; Tice, D.A.; Herbst, R.; van Berkel, P.H. Antitumor Activity of MEDI3726 (ADCT-401), a Pyrrolobenzodiazepine Antibody-Drug Conjugate Targeting PSMA, in Preclinical Models of Prostate Cancer. Mol Cancer Ther 2018, 17, 2176–2186. [Google Scholar] [CrossRef] [PubMed]
- Adair, J.R.; Howard, P.W.; Hartley, J.A.; Williams, D.G.; Chester, K.A. Antibody-drug conjugates - a perfect synergy. Expert Opin Biol Ther 2012, 12, 1191–1206. [Google Scholar] [CrossRef]
- de Bono, J.S.; Fleming, M.T.; Wang, J.S.; Cathomas, R.; Miralles, M.S.; Bothos, J.; Hinrichs, M.J.; Zhang, Q.; He, P.; Williams, M.; Rosenbaum, A.I.; Liang, M.; Vashisht, K.; Cho, S.; Martinez, P.; Petrylak, D.P. Phase I Study of MEDI3726: A Prostate-Specific Membrane Antigen-Targeted Antibody-Drug Conjugate, in Patients with mCRPC after Failure of Abiraterone or Enzalutamide. Clin Cancer Res 2021, 27, 3602–3609. [Google Scholar] [CrossRef] [PubMed]
- de Bono, J.S.; Scher, H.I.; Montgomery, R.B.; Parker, C.; Miller, M.C.; Tissing, H.; Doyle, G.V.; Terstappen, L.W.; Pienta, K.J.; Raghavan, D. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 2008, 14, 6302–6309. [Google Scholar] [CrossRef]
- Zhang, D.; Yu, S.F.; Khojasteh, S.C.; Ma, Y.; Pillow, T.H.; Sadowsky, J.D.; Su, D.; Kozak, K.R.; Xu, K.; Polson, A.G.; Dragovich, P.S.; Hop, C. Intratumoral Payload Concentration Correlates with the Activity of Antibody-Drug Conjugates. Mol Cancer Ther 2018, 17, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Su, D.; Zhang, D. Linker Design Impacts Antibody-Drug Conjugate Pharmacokinetics and Efficacy via Modulating the Stability and Payload Release Efficiency. Front Pharmacol 2021, 12, 687926. [Google Scholar] [CrossRef] [PubMed]
- Johann, F.; Wöll, S.; Gieseler, H. “Negative” Impact: The Role of Payload Charge in the Physicochemical Stability of Auristatin Antibody–Drug Conjugates. Journal of Pharmaceutical Sciences 2024, 113, 2433–2442. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, H.; Gou, L.; Li, W.; Wang, Y. Antibody-drug conjugates: Recent advances in payloads. Acta Pharm Sin B 2023, 13, 4025–4059. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



