Submitted:
15 January 2025
Posted:
16 January 2025
You are already at the latest version
Abstract
(1) Background: Gut microbial dysbiosis, leaky gut and increased transepithelial translocation of commensal bacteria have been documented in multiple sclerosis (MS). Intrathecal IgGs specific for Akkermansia muciniphila, a gut bacterium, are increased in MS patients and associated with clinical disability. Our objective here was to explore the putative involvement of intrathecal anti-A. muciniphila IgG in MS pathogenesis by characterizing patients with different anti-A. muciniphila IgG indices. (2) Methods: Serum and intrathecal IgG specific for A. muciniphila and other gut bacteria as well as routine cerebrospinal fluid (CSF) parameters were measured in 61 MS patients. Examination of these patients included immunophenotyping of CSF-infiltrating and paired circulating lymphocytes, intrathecal markers of neurodegeneration and inflammation and a detailed characterization of demographic-, clinical-, and magnetic resonance imaging (MRI) features. (3) Results: MS patients with high anti-A. muciniphila IgG index also showed higher intrathecal IgG indices against other gut bacteria. Plasma blasts, B cells and Th2 cells that might be involved in antibody production were increased in the CSF of these patients as well as blood pro-inflammatory Th17 cells. Anti-A. muciniphila IgG indices were negatively associated with blood brain barrier (BBB) permeability and circulating monocytes and positively with brain lesion load. (4) Conclusions: The differences between patients with low and high anti-A. muciniphila IgG indexes regarding BBB permeability, CSF cell infiltrates, pro-inflammatory peripheral immune cells as well as imaging features, support a role of anti-A. muciniphila immune response in MS pathogenesis.
Keywords:
1. Introduction
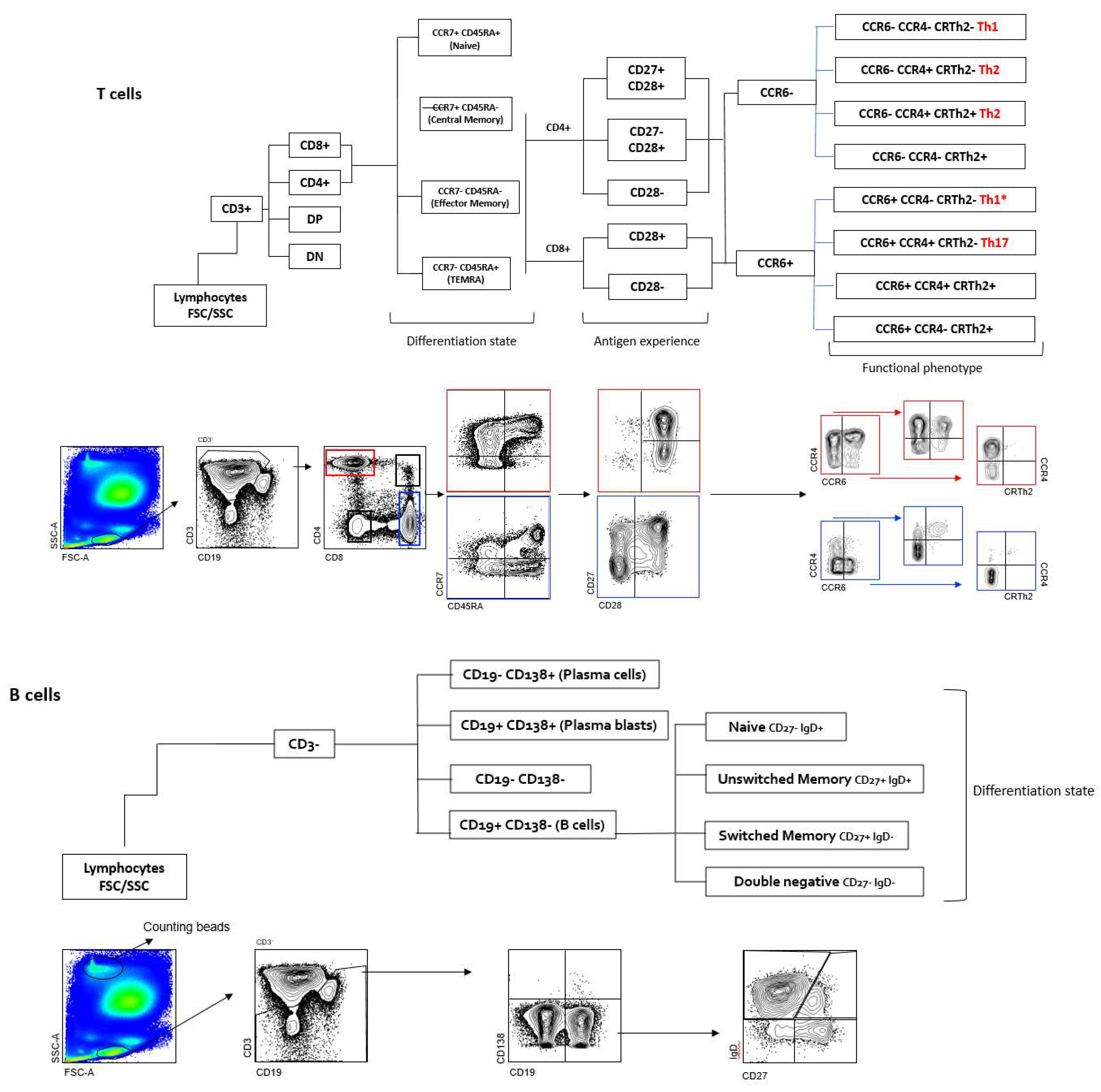
2. Materials and Methods
3. Results
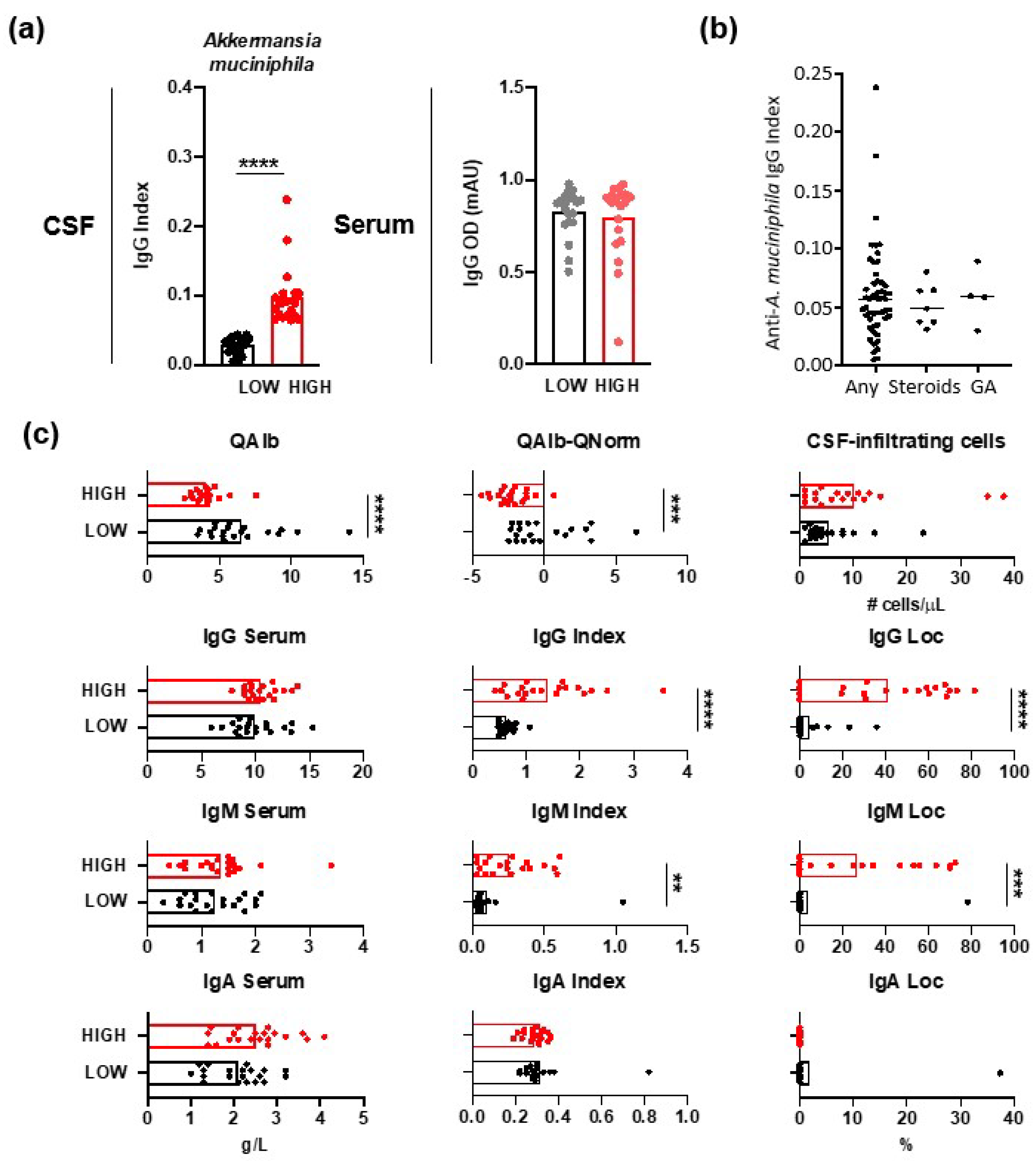
3.1. IgGs Specific for Gut Commensal Bacteria
3.2. CSF Measures in MS Patients with Different Anti-A. muciniphila IgG Index
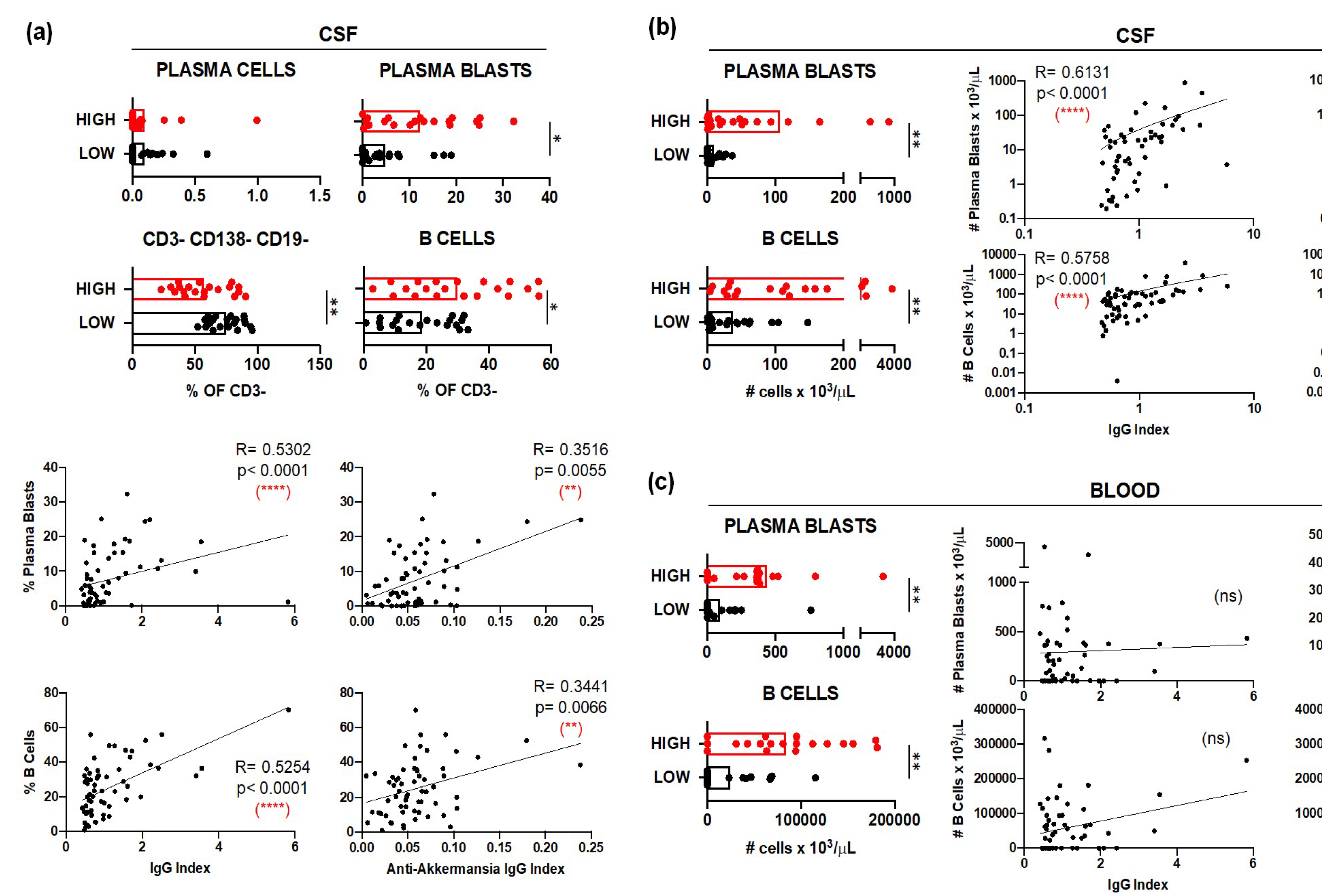
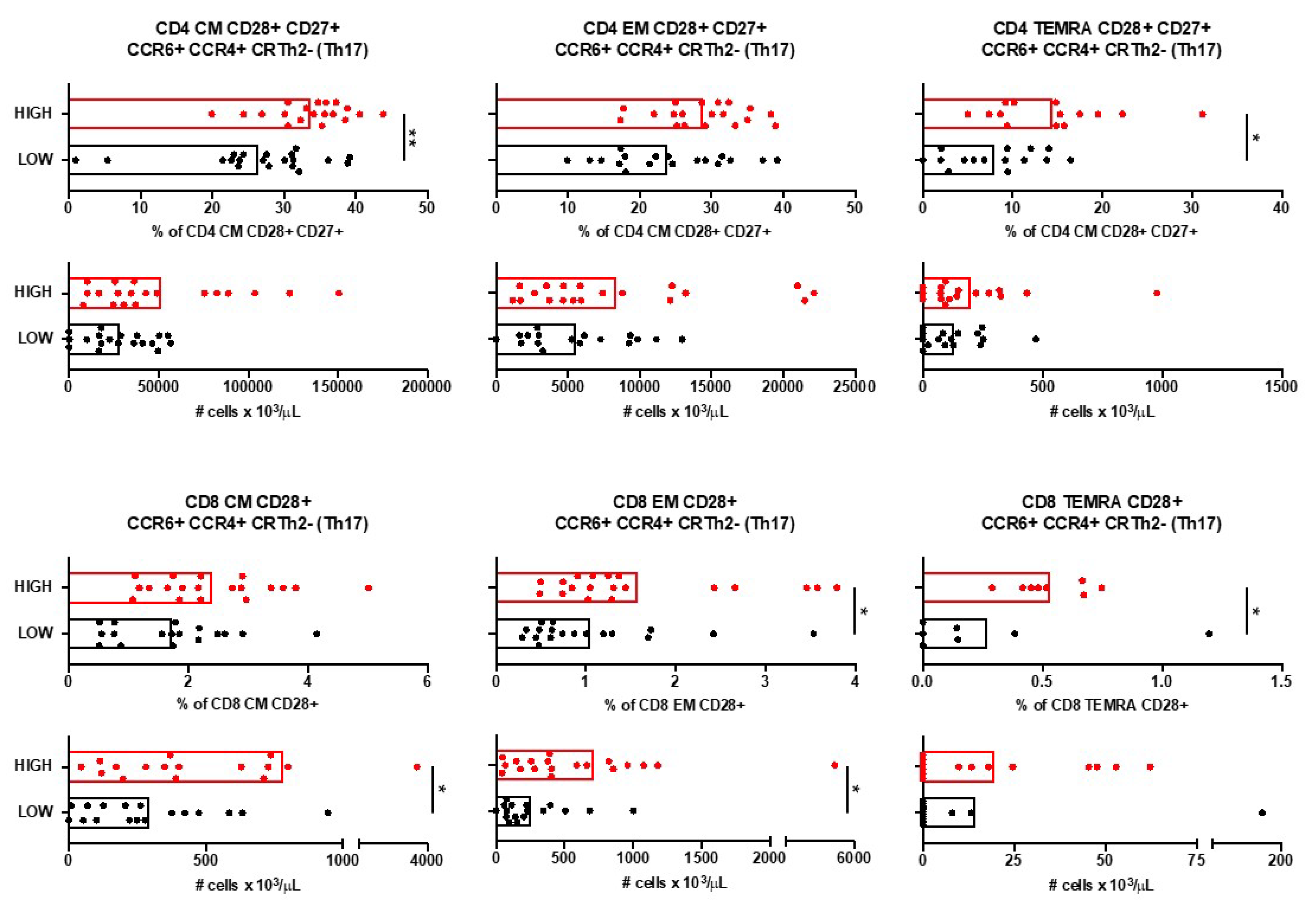
3.2.1. B Cells in MS Patients with Different Anti-A. muciniphila IgG Index
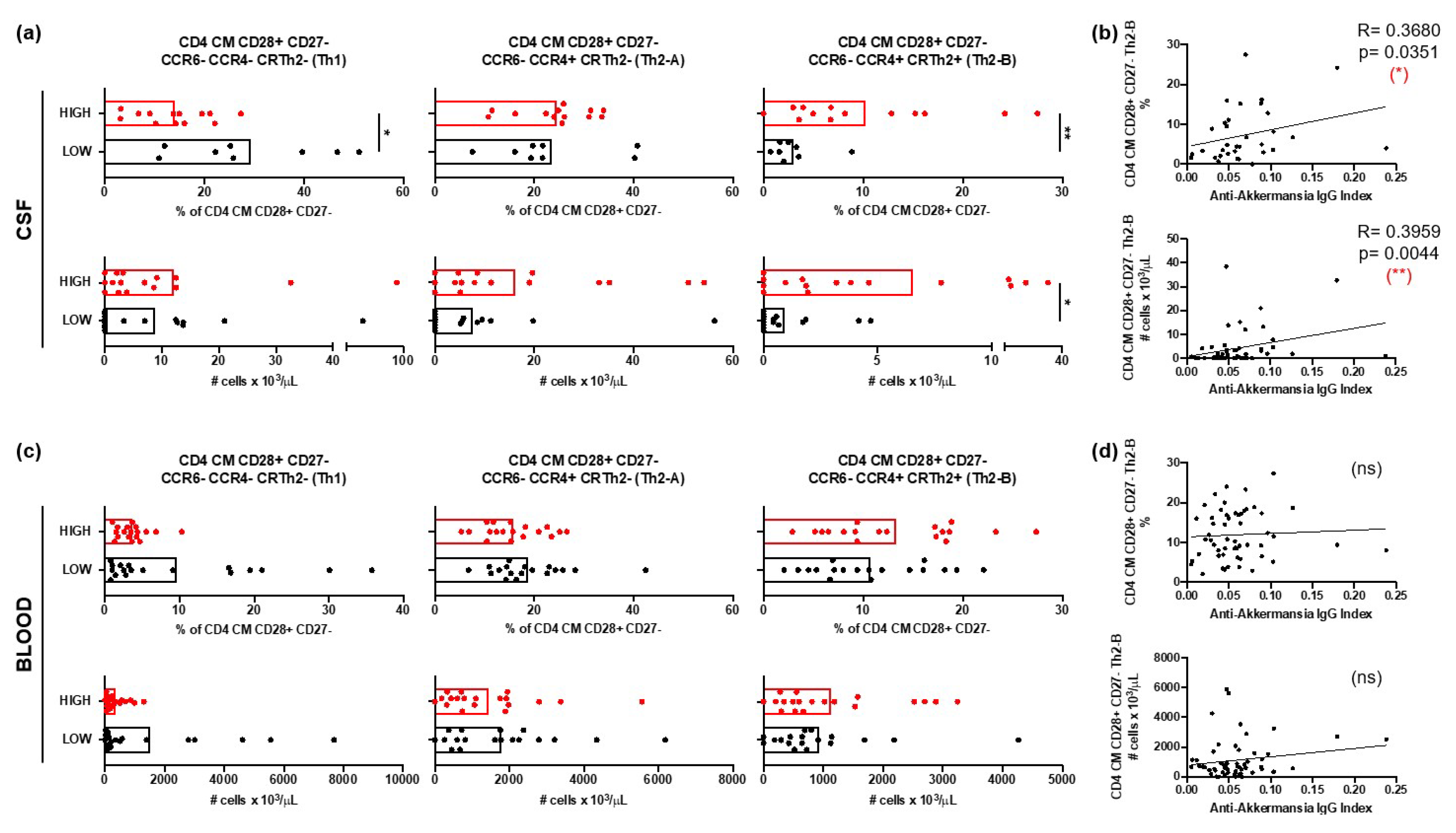
3.2.2. Th2 Cells in MS Patients with Different Anti-A. muciniphila IgG Index
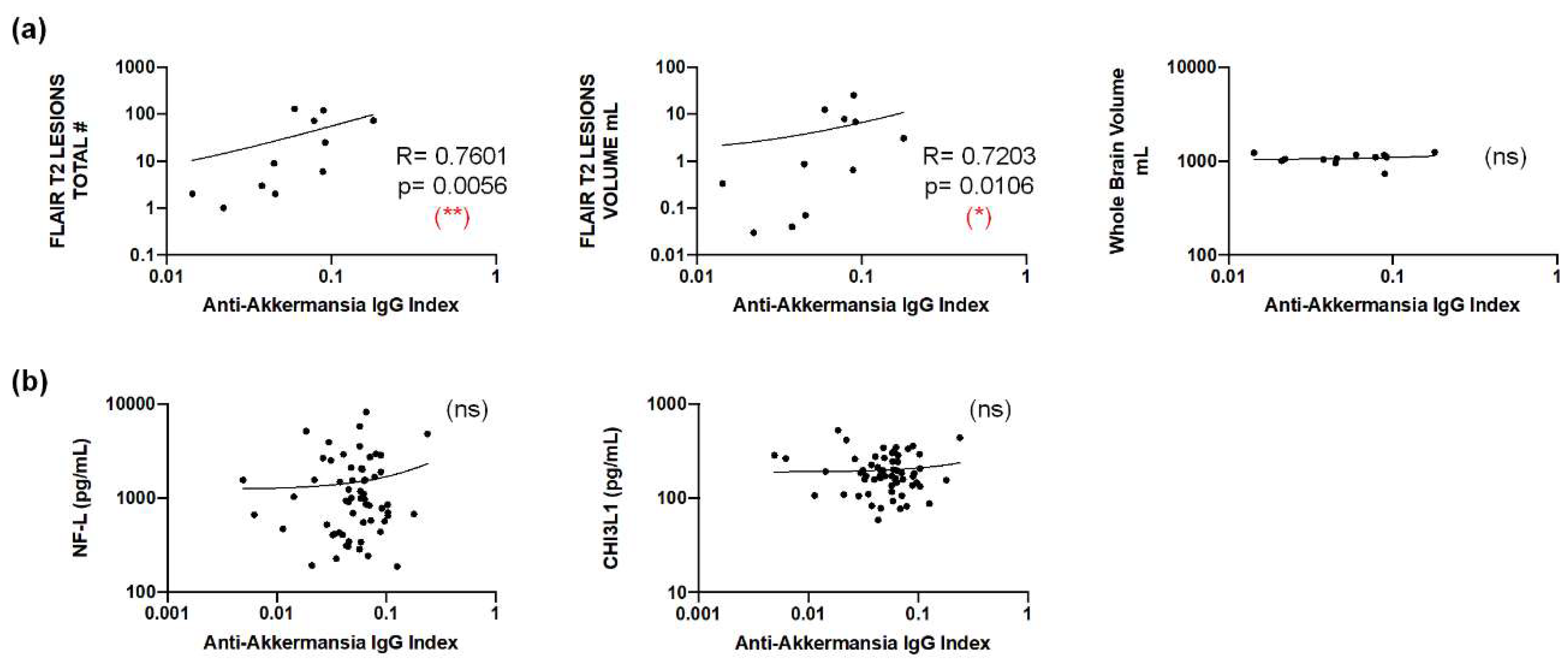
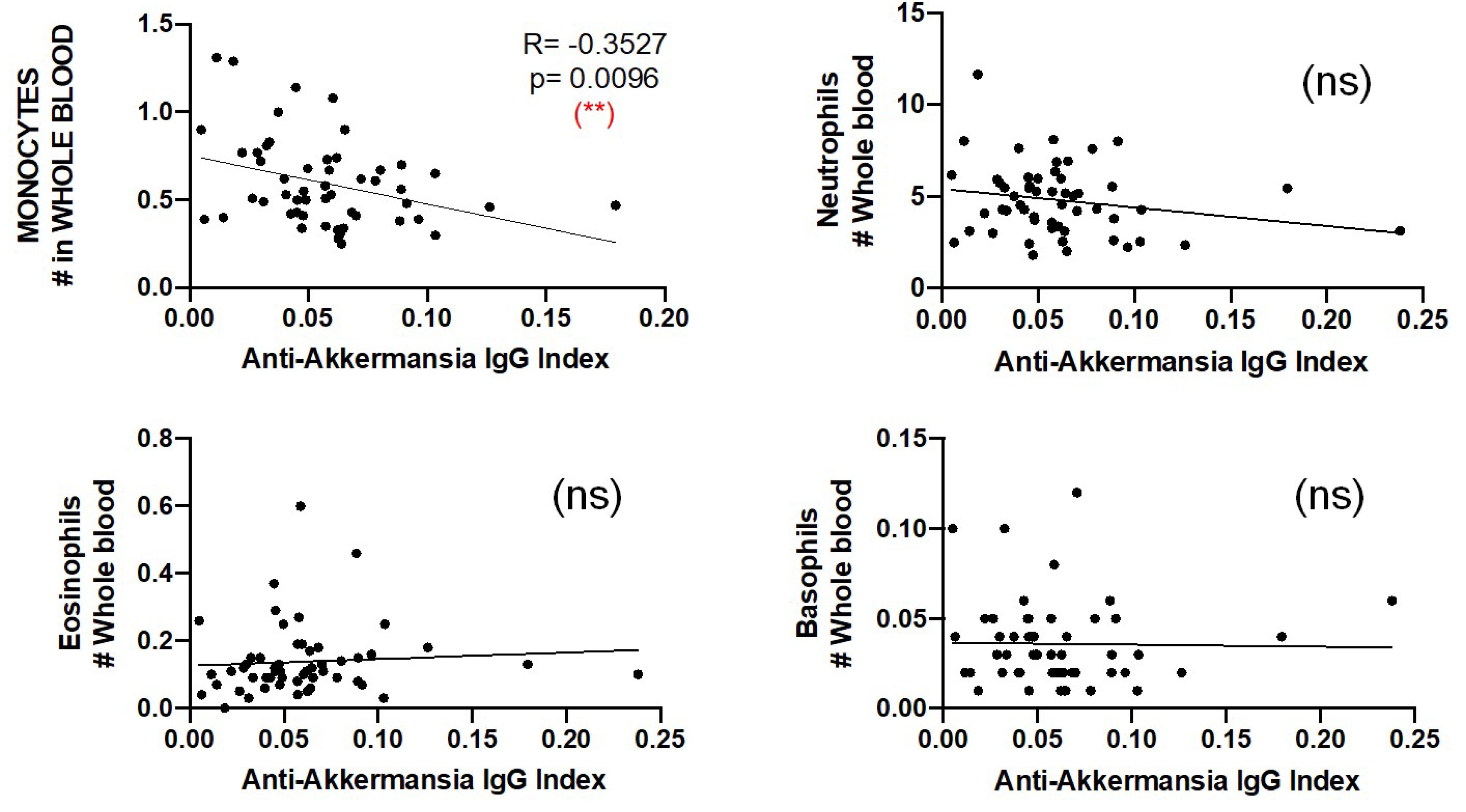
3.3. Characterization of Patients with Different Anti-A. muciniphila IgG Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sospedra, M.; Martin, R. Immunology of multiple sclerosis. Annu Rev Immunol 2005, 23, 683–747. [Google Scholar] [CrossRef] [PubMed]
- Sospedra, M. B cells in multiple sclerosis. Curr Opin Neurol 2018, 31, 256–62. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Munger, K.L.; Lunemann, J.D. The initiation and prevention of multiple sclerosis. Nat Rev Neurol 2012, 8, 602–612. [Google Scholar] [CrossRef]
- Canto, E.; Oksenberg, J.R. Multiple sclerosis genetics. Mult Scler 2018, 24, 75–79. [Google Scholar] [CrossRef]
- Freedman, S.N.; Shahi, S.K.; Mangalam, A.K. The “Gut Feeling”: Breaking Down the Role of Gut Microbiome in Multiple Sclerosis. Neurotherapeutics 2018, 15, 109–125. [Google Scholar] [CrossRef]
- Cekanaviciute, E.; Yoo, B.B.; Runia, T.F.; Debelius, J.W.; Singh, S.; Nelson, C.A.; Kanner, R.; Bencosme, Y.; Lee, Y.K.; Hauser, S.L.; Crabtree-Hartman, E.; Sand, I.K.; Gacias, M.; Zhu, Y.; Casaccia, P.; Cree, B.A.C.; Knight, R.; Mazmanian, S.K.; Baranzini, S.E. Gut bacteria from multiple sclerosis patients modulate human T cells and exacerbate symptoms in mouse models. Proc Natl Acad Sci U S A 2017, 114, 10713–10718. [Google Scholar] [CrossRef]
- Berer, K.; Gerdes, L.A.; Cekanaviciute, E.; Jia, X.; Xiao, L.; Xia, Z.; Liu, C.; Klotz, L.; Stauffer, U.; Baranzini, S.E.; Kumpfel, T.; Holdfeld, R.; Krishnamoorthy, G.; Wekerle, H. Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice. Proc Natl Acad Sci U S A 2017, 114, 10719–10724. [Google Scholar] [CrossRef]
- Mu, Q.; Kirby, J.; Reilly, C.M.; Luo, X.M. Leaky Gut As a Danger Signal for Autoimmune Diseases. Front Immunol 2017, 8, 598. [Google Scholar] [CrossRef]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; deRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; Rudensky, A.Y. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef]
- Haghikia, A.; Jorg, S.; Duscha, A.; Berg, J.; Manzel, A.; Waschbisch, A.; Hammer, A.; Lee, D.H.; May, C.; Wilck, N.; Balogh, A.; Ostermann, A.I.; Schebb, N.H.; Akkad, D.A.; Grohme, D.A.; Kleinewietfeld, M.; Kempa, S.; Thöne, J.; Demir, S.; Müller, D.N.; Gold, R.; Linker, R.A. Dietary Fatty Acids Directly Impact Central Nervous System Autoimmunity via the Small Intestine. Immunity 2015, 43, 817–829. [Google Scholar] [CrossRef]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; Young, V.B.; Henrissat, B.; Wlimes, P.; Stappenbeck, T.S.; Nunez, G.; Martens, E.C. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353.e21. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Douek, D.C. Microbial translocation across the GI tract. Annu Rev Immunol 2012, 30, 149–173. [Google Scholar] [CrossRef] [PubMed]
- Varatharaj, A.; Galea, I. The blood-brain barrier in systemic inflammation. Brain Behav Immun 2017, 60, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Erny, D.; Hrabe de Angelis, A.L.; Jaitin, D.; Wieghofer, P.; Staszewski, O.; David, E.; Keren-Shaul, H.; Mahlakoiv, T.; Jakobshagen, K.; Buch, T.; Schwierzeck, V.; Utermöhlen, O.; Chun, E.; Garrett, W.S.; McCoy, K.D.; Diefenbach, A.; Staeheli, P.; Stecher, B.; Amit, I.; Prinz, M. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci 2015, 18, 965–977. [Google Scholar] [CrossRef]
- Rothhammer, V.; Mascanfroni, I.D.; Bunse, L.; Takenaka, M.C.; Kenison, J.E.; Mayo, L.; Chao, C.C.; Patel, B.; Yan, R.; Blain, M.; Alvarez, J.I.; Kébir, H.; Anandasabapathy, N.; Izquierdo, G.; Jung, S.; Obholzer, N.; Pochet, N.; Clish, C.B.; Prinz, M.; Prat, A.; Antel, J.; Quintana, F.J. Type I interferons and microbial metabolites of tryptophan modulate astrocyte activity and central nervous system inflammation via the aryl hydrocarbon receptor. Nat Med 2016, 22, 586–597. [Google Scholar] [CrossRef]
- Camara-Lemarroy, C.R.; Silva, C.; Greenfield, J.; Liu, W.Q.; Metz, L.M.; Yong, V.W. Biomarkers of intestinal barrier function in multiple sclerosis are associated with disease activity. Mult Scler 2020, 26, 1340–1350. [Google Scholar] [CrossRef]
- Buscarinu, M.C.; Fornasiero, A.; Romano, S.; Ferraldeschi, M.; Mechelli, R.; Renie, R.; Ferraldeschi, M.; Mechelli, R.; Reniè, R.; Morena, E.; Romano, C.; Pellicciari, G.; Landi, A.C.; Salvetti, M.; Ristori, G. The Contribution of Gut Barrier Changes to Multiple Sclerosis Pathophysiology. Front Immunol 2019, 10, 1916. [Google Scholar] [CrossRef]
- Teixeira, B.; Bittencourt, V.C.; Ferreira, T.B.; Kasahara, T.M.; Barros, P.O.; Alvarenga, R.; Hygino, J.; Andrade, R.M.; Andrade, A.F.; Bento, C.A. Low sensitivity to glucocorticoid inhibition of in vitro Th17-related cytokine production in multiple sclerosis patients is related to elevated plasma lipopolysaccharide levels. Clin Immunol 2013, 148, 209–218. [Google Scholar] [CrossRef]
- Jangi, S.; Gandhi, R.; Cox, L.M.; Li, N.; von Glehn, F.; Yan, R.; Patel, B.; Mazzola, M.A.; Liu, S.; Glank, B.L.; Cook, S.; Tankou, S.; Stuart, F.; Melo, K.; Nejad, P.; Smith, K.; Topçuolu, B.D.; Holden, J.; Kivisäkk, P.; Chitnis, T.; De Jager, P.L.; Quintana, F.J.; Gerber, G.K.; Bry, L.; Weiner, H.L. Alterations of the human gut microbiome in multiple sclerosis. Nat Commun 2016, 7, 12015. [Google Scholar] [CrossRef]
- Vallino, A.; Dos Santos, A.; Mathe, C.V.; Garcia, A.; Morille, J.; Dugast, E.; Shah, S.P.; Héry-Arnaud, G.; Guilloux, C.A.; Gleeson, P.J.; Monteiro, R.C.; Soulillou, J.P.; Harb, J.; Bigot-Corbel, E.; Michel, L.; Wiertlewski, S.; Nicot, A.B.; Laplaud, D.A.; Berthelot, L. Gut bacteria Akkermansia elicit a specific IgG response in CSF of patients with MS. Neurol Neuroimmunol Neuroinflamm 2020, 7, e688. [Google Scholar] [CrossRef]
- Planas, R.; Santos, R.; Tomas-Ojer, P.; Cruciani, C.; Lutterotti, A.; Faigle, W.; Schaeren6wiemers, N.; Espejo, C.; Eixarch, H.; Pinilla, C.; Martin, R.; Sospedra, M. GDP-l-fucose synthase is a CD4(+) T cell-specific autoantigen in DRB3*02:02 patients with multiple sclerosis. Sci Transl Med 2018, 10, eaat4301. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jelcic, I.; Muhlenbruch, L.; Haunerdinger, V.; Toussaint, N.C.; Zhao, Y.; et al. HLA-DR15 Molecules Jointly Shape an Autoreactive T Cell Repertoire in Multiple Sclerosis. Cell 2020, 183, 1264–1281. [Google Scholar] [CrossRef] [PubMed]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; Lublin, F.D.; Montalban, X.; O’Connor, P.; Sandberg-Wolheim, M.; Thompson, A.J.; Waubant, E.; Winshenker, B.; Wollinsky, J.S. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 2011, 69, 292–302. [Google Scholar] [CrossRef]
- Puthenparampil, M.; Tomas-Ojer, P.; Hornemann, T.; Lutterotti, A.; Jelcic, I.; Ziegler, M.; Hulsmeier, A.J.; Cruciani, C.; Faigle, W.; Martin, R.; Sospedra, M. Altered CSF Albumin Quotient Links Peripheral Inflammation and Brain Damage in MS. Neurol Neuroimmunol Neuroinflamm 2021, 8, e951. [Google Scholar] [CrossRef] [PubMed]
- Brodie, T.; Rothaeusler, K.; Sospedra, M. OMIP-033: A comprehensive single step staining protocol for human T- and B-cell subsets. Cytometry A 2016, 89, 629–632. [Google Scholar] [CrossRef]
- Kruger, J.; Opfer, R.; Gessert, N.; Ostwaldt, A.C.; Manogaran, P.; Kitzler, H.H.; Schlaefer, A.; Schippling, S. Fully automated longitudinal segmentation of new or enlarged multiple sclerosis lesions using 3D convolutional neural networks. Neuroimage Clin 2020, 28, 102445. [Google Scholar] [CrossRef]
- Schippling, S.; Ostwaldt, A.C.; Suppa, P.; Spies, L.; Manogaran, P.; Gocke, C.; Huppertz, H.J.; Opfer, R. Global and regional annual brain volume loss rates in physiological aging. J Neurol 2017, 264, 520–528. [Google Scholar] [CrossRef]
- Bergman, J.; Dring, A.; Zetterberg, H.; Blennow, K.; Norgren, N.; Gilthorpe, J.; et al. Neurofilament light in CSF and serum is a sensitive marker for axonal white matter injury in MS. Neurol Neuroimmunol Neuroinflamm 2016, 3, e271. [Google Scholar] [CrossRef]
- Bonneh-Barkay, D.; Bissel, S.J.; Kofler, J.; Starkey, A.; Wang, G.; Wiley, C.A. Astrocyte and macrophage regulation of YKL-40 expression and cellular response in neuroinflammation. Brain Pathol 2012, 22, 530–546. [Google Scholar] [CrossRef]
- Watts, T.H. TNF/TNFR family members in costimulation of T cell responses. Annu Rev Immunol 2005, 23, 23–68. [Google Scholar] [CrossRef]
- Morgan, A.J.; Symon, F.A.; Berry, M.A.; Pavord, I.D.; Corrigan, C.J.; Wardlaw, A.J. IL-4-expressing bronchoalveolar T cells from asthmatic and healthy subjects preferentially express CCR 3 and CCR 4. J Allergy Clin Immunol 2005, 116, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Cosmi, L.; Annunziato, F.; Galli, M.I.G.; Maggi, R.M.E.; Nagata, K.; Romagnani, S. CRTH2 is the most reliable marker for the detection of circulating human type 2 Th and type 2 T cytotoxic cells in health and disease. Eur J Immunol 2000, 30, 2972–2979. [Google Scholar] [CrossRef] [PubMed]
- Tesmer, L.A.; Lundy, S.K.; Sarkar, S.; Fox, D.A. Th17 cells in human disease. Immunol Rev 2008, 223, 87–113. [Google Scholar] [CrossRef] [PubMed]
- Akdis, C.A. Does the epithelial barrier hypothesis explain the increase in allergy, autoimmunity and other chronic conditions? Nat Rev Immunol 2021, 21, 739–751. [Google Scholar] [CrossRef]
| All | Anti-A. mucniniphila IgG Index Low | Anti-A. mucniniphila IgG Index High | p1 | |
|---|---|---|---|---|
| Number of patients | 61 | 20 | 20 | |
| Female/male ratio | 2.05 | 1.2 | 3 | 0.32 |
| Age at CSF puncture (years) | 36.2 ± 10.1 | 36.9 ± 11.6 | 35.8 ± 9.3 | 0.89 |
| Age at disease onset (years) | 33.7 ± 9.2 | 35.0 ± 10.8 | 32.2 ± 9.4 | 0.54 |
| Disease duration (Months) | 31.9 ± 58.8 | 29.9 ± 60.1 | 42.4 ± 72.9 | 0.89 |
| RIS /CIS (%) | 19.6 | 30.0 | 10.0 | 0.23 |
| RRMS (%) | 73.7 | 60.0 | 85.0 | 0.15 |
| PMS (%) | 6.5 | 10.0 | 5.0 | 0.90 |
| CSF OCB Type II (%) | 81.9 | 70.0 | 100.0 | 0.02 * |
| HLA DR15 (%) | 44.2 | 25.0 | 55.0 | 0.10 |
| QAlb-QNorm>0 (%) | 19.6 | 35.0 | 5.0 | 0.04 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




