1. Introduction
Castration-resistant prostate cancer (CRPC) is an advanced stage of prostate cancer, characterized by disease progression despite achieving castration levels of serum testosterone (≤50 ng/dL), as outlined in the 2024 EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer management. CRPC includes a heterogeneous group of patients: those without evidence of metastasis (nonmetastatic CRPC—nmCRPC) and those with metastatic disease (metastatic CRPC—mCRPC). mCRPC can be further divided into two categories: oligometastatic disease, where metastases are limited in number and location; and polymetastatic disease, characterized by widespread metastatic involvement. A specific subset, oligoprogressive mCRPC, refers to cases where disease progression occurs at a limited number of metastatic sites, presenting opportunities for localized treatment interventions.
Metastasis-directed therapy (MDT), including both stereotactic ablative radiotherapy (SABR) and surgical metastasectomy, has demonstrated promise in the management of oligometastatic prostate cancer. Substantial evidence supports MDT for patients with low-volume metastatic hormone-sensitive prostate cancer (mHSPC), as shown in the STOMP [
1] and ORIOLE [
2] trials. However, data on MDT, particularly surgical approaches, in oligoprogressive mCRPC remain sparse. This highlights the need for more robust investigations into the efficacy and safety of MDT in this specific setting.
Here, we present the first report of robotic surgery used as an MDT in the context of oligoprogressive mCRPC, followed by a discussion synthesizing recent findings on MDT, including SABR and surgical metastasectomy, in this clinical setting. The case also highlights the potential of advanced imaging and localized therapies to improve outcomes, while emphasizing gaps in current research.
2. Case Presentation
A 47-year-old male presented in 2019 with a total PSA level exceeding 2100 ng/mL. Further diagnostic evaluation confirmed adenocarcinoma of the prostate, classified as ISUP Grade 5 Gleason score 10 (5 + 5) and staged using conventional imaging, including whole-body bone scintigraphy and whole-body computed tomography (CT), as cT2aN1M1a, indicative of newly diagnosed high-volume metastatic hormone-sensitive prostate cancer (per CHAARTED criteria). The patient reported no family history of prostate cancer or other malignancies. His medical history was significant for hypertension, which was well controlled with antihypertensive therapy.
2.1. Initial Management
The patient was treated initially with maximal androgen blockade (MAB), including degarelix combined with bicalutamide, a non-steroidal antiandrogen, followed by six cycles of early docetaxel chemotherapy. With a preoperative PSA level of 0.68 ng/mL, in October 2019, he underwent a non-standardized, aggressive surgical approach comprising salvage radical retropubic prostatectomy (RRP), super-extended pelvic lymph node dissection (sePLND), and retroperitoneal lymph node dissection (RPLND). Intraoperatively, bilateral double-J ureteral stents were placed and later removed. The patient achieved undetectable PSA levels postoperatively, with castration-level testosterone maintained through continued androgen deprivation therapy (ADT): leuprolide acetate, 45 mg every six months.
2.2. Detection of Disease Progression
A rise in PSA to 0.020 ng/mL, despite castration-level testosterone, prompted restaging with a Ga-68-PSMA PET/CT scan on 11 March 2021. The scan detected radiotracer uptake in a single right inguinal lymph node with a SUVmax of 12.4, with no evidence of M1b (bone metastases) or M1c (visceral metastases) disease. The identification of this radiotracer-avid lesion, indicating new metastatic progression despite ongoing ADT, confirmed the diagnosis of oligoprogressive mCRPC. Physical examination revealed no palpable lymphadenopathy, with no other significant abnormalities detected. Diagnostic assessments were straightforward, with imaging confirming the diagnosis without significant challenges.
2.3. Preoperative Evaluation and Targeting
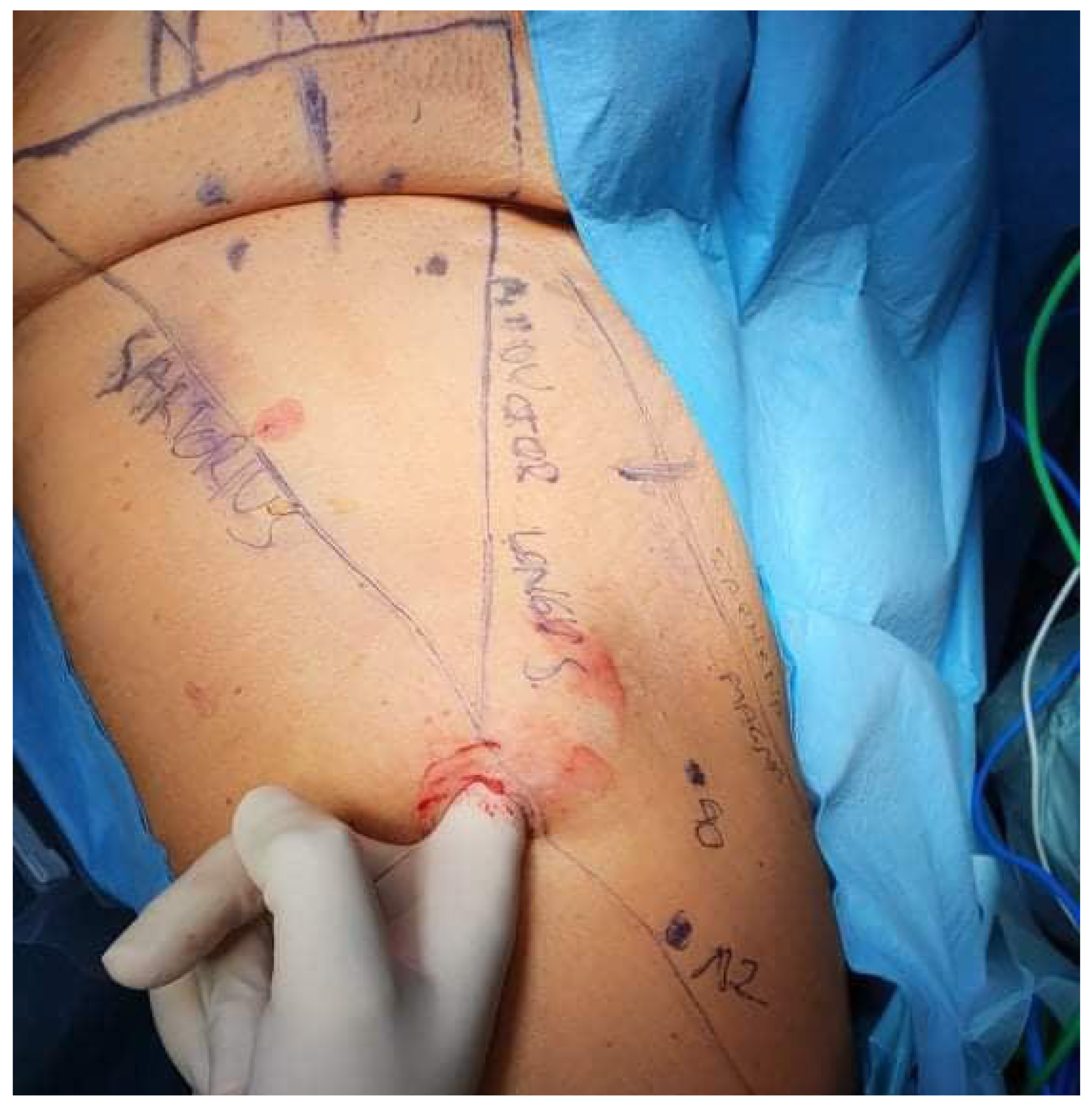
Preoperatively, detailed mapping of the targeted anatomy was performed (
Figure 1). Ultrasound was utilized to identify key anatomical landmarks, including the femoral nerve, femoral artery, femoral vein, great saphenous vein , the femoral triangle (sartorius muscle, adductor longus muscle, inguinal ligament), and four suspicious lymph nodes. These nodes were identified based on established malignancy criteria on ultrasound, including a round shape with a long-to-short axis ratio < 2:1, absent or displaced hilum, lower echogenicity than surrounding muscle tissue, and peripheral vascularity patterns observed on Doppler imaging. Preoperatively marked areas of lymph nodes identified as suspicious on ultrasound allowed for intraoperative control, ensuring that all targeted nodes were effectively removed.
2.4. Salvage Surgical Intervention
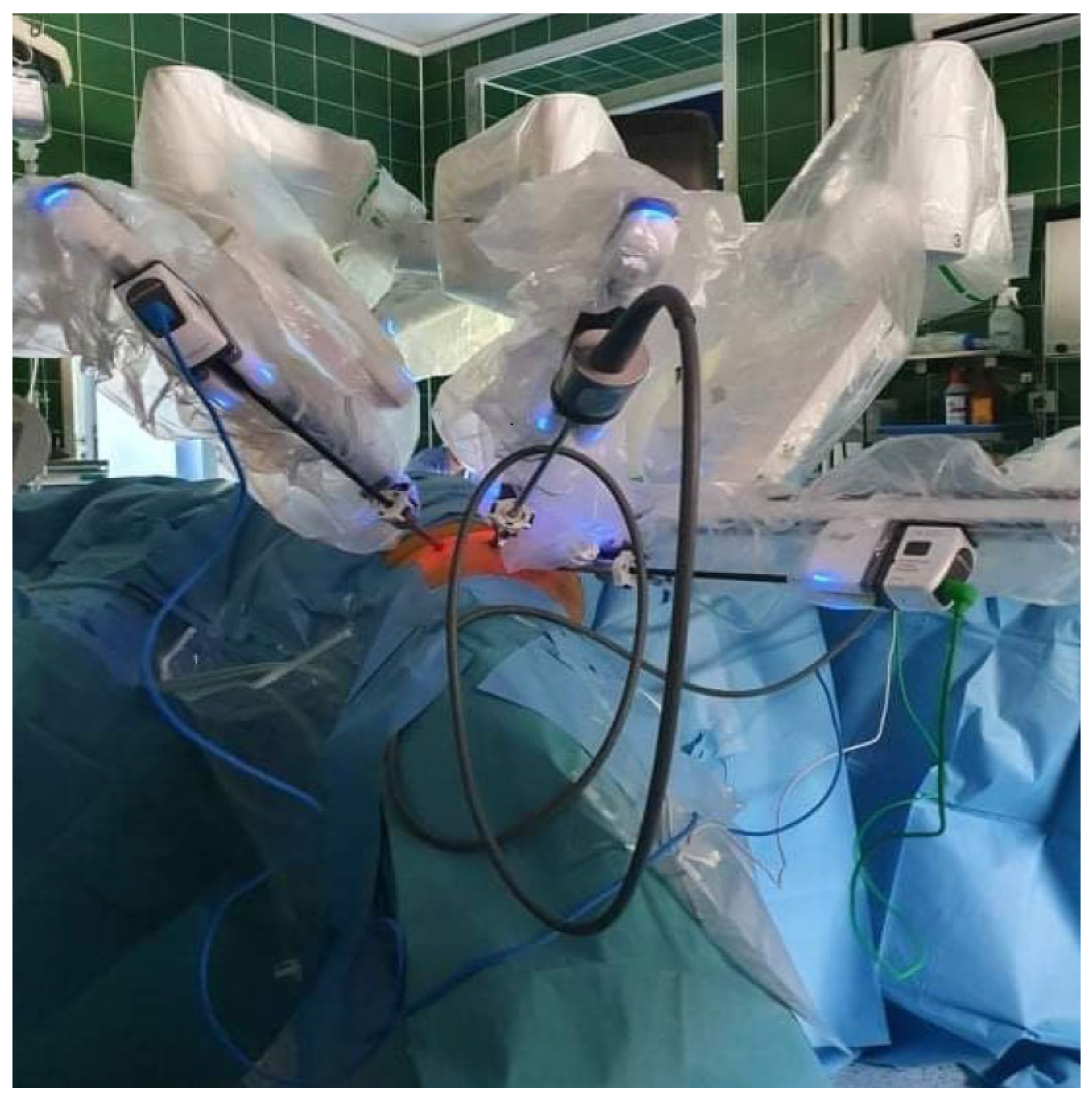
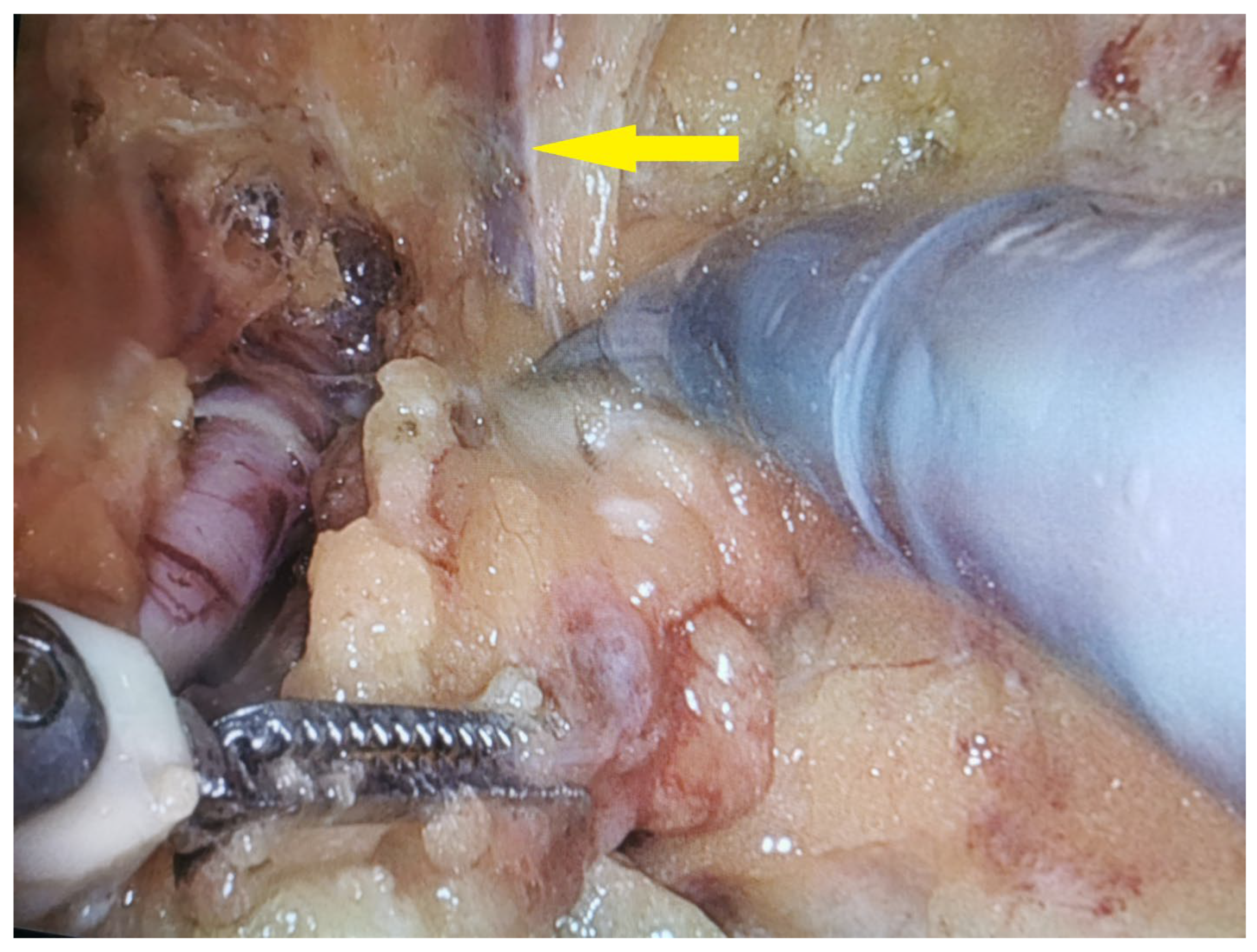
On 12 August 2021, the patient underwent a right-sided salvage robot-assisted video-endoscopic inguinal lymphadenectomy (RAVEIL) (
Figure 2) without saphenectomy (
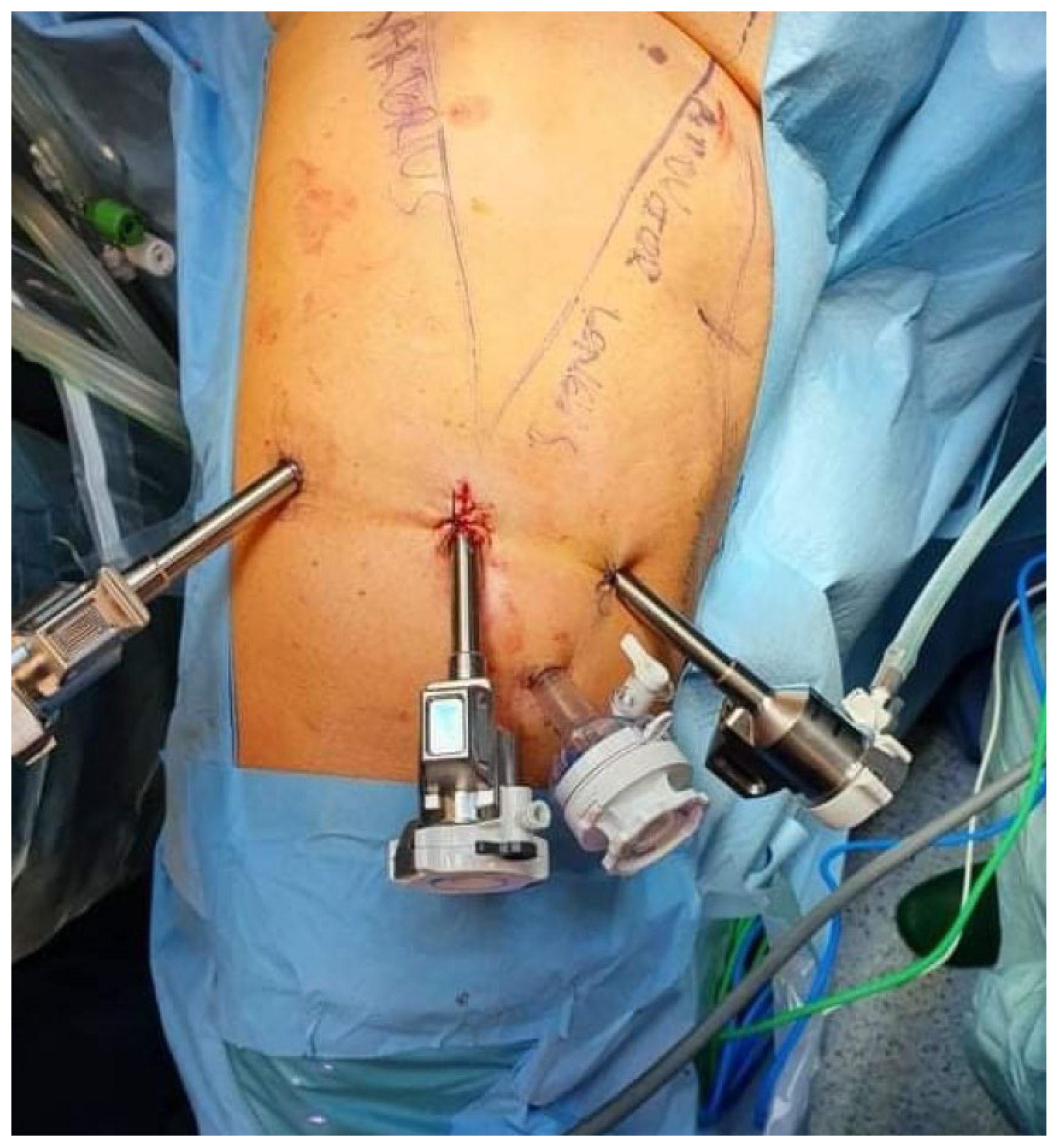
Figure 3). The trocar docking scheme is shown in
Figure 4. The procedure, performed using the da Vinci X™ Surgical System (Intuitive Surgical, Sunnyvale, CA, USA), lasted 50 min and was characterized by minimal blood loss and a hospital stay of three days (two days postoperatively). To prevent chylous drainage and lymphocele formation, two Redon drains were placed. The histopathological examination of the excised tissue included eight lymph nodes, with one node confirmed to have metastatic involvement.
2.5. Postoperative Course and Complications
The postoperative course was characterized by minor (Clavien–Dindo Grade II) complications, specifically readmission due to subcutaneous cellulitis, which was successfully managed with oral antibiotics (amoxicillin + clavulanic acid), and persistent chylous drainage (lymphorrhea) lasting over two weeks, requiring the prolonged use of a surgical drain. Despite these challenges, the patient recovered fully, with no further significant issues. The patient reported satisfaction with the treatment outcomes and noted no significant impact on quality of life.
2.6. Follow-Up
Six weeks postoperatively, the patient demonstrated a PSA level that remained undetectable under continued ADT for 14 months. Adherence and tolerability were assessed through regular clinical follow-ups, with the patient reporting no major issues related to ongoing ADT. However, a subsequent rise in PSA to 0.31 ng/mL prompted periodic restaging with 68Ga-PSMA PET/CT, which identified recurrence localized to the region of the left renal hilum. The recurrence was treated with SABR using Gamma Knife™ (by Elekta AB, Stockholm, Sweden), successfully reducing the PSA level to undetectable levels once again, with no further evidence of disease progression.
Figure 5 illustrates the clinical timeline, summarizing key events from diagnosis through treatment interventions and follow-up, in accordance with CARE guidelines.
3. Discussion
Oligoprogressive mCRPC represents a critical challenge in advanced prostate cancer management. MDTs, including both SABR and surgical interventions such as RAVEIL, are increasingly explored as strategies to delay next-line systemic treatment (NEST). This discussion integrates current evidence, including promising results from the present case report, to assess the role and potential of MDT, in the management of oligoprogressive mCRPC.
3.1. Advanced Imaging and Its Role in MDT
Advanced imaging modalities such as PSMA PET/CT and dual-tracer PET/CT have revolutionized patient selection for MDT. Pan et al. conducted a prospective study utilizing dual-tracer PET/CT with ^68Ga-PSMA and ^18F-FDG tracers to improve the detection of metastatic heterogeneity in patients with nmPCa. Their findings revealed that 23% of patients exhibited discordant FDG-avid lesions, indicating the presence of metabolically active, PSMA-negative tumor sites. This advanced imaging approach significantly enhanced precision in SABR targeting, ensuring improved local control of disease while minimizing the risk of unnecessary treatments. Among the 74 patients enrolled, 66% had detectable metastases, with 60% showing a PSA-doubling time of ≤6 months. Patients who underwent SABR guided by dual-tracer PET/CT achieved a median metastasis-free survival (MFS) that was not reached during the follow-up period, compared to only 11.0 months in the ADT group (HR 4.69, 95% CI: 2.92–25.0;
p < 0.001). Additionally, an impressive 86% of patients in the SABR group experienced a PSA reduction of ≥90%, highlighting the efficacy of this imaging-guided therapeutic approach. Importantly, no grade 3 or higher toxicities were observed, with only mild grade 1–2 adverse events reported [
3]. These results underscore the clinical utility of dual-tracer PET/CT in identifying resistant tumor clones, optimizing SABR targeting, and achieving favorable outcomes, particularly in cases with discordant FDG-avid lesions.
Hahn’s work also underscored the pivotal role of advanced imaging modalities, such as PSMA PET/CT, in identifying patients most likely to benefit from MDT, including SABR. The study demonstrated that the use of PSMA PET/CT improved patient selection by detecting up to five metastases with high sensitivity and specificity, which directly impacted the effectiveness of MDT. For instance, patients whose MDT was guided by PSMA PET/CT achieved significantly longer progression-free survival (PFS) compared to those assessed with less sensitive imaging techniques, such as choline PET/CT. PSMA PET/CT-guided MDT extended PFS from a median of 9.5 months to 17.9 months, representing a notable improvement in disease management outcomes. Furthermore, PSMA PET/CT was shown to support superior localization of resistant tumor clones, enhancing the clinical utility of MDT and contributing to delayed initiation of systemic therapies [
4].
The DROP-IN study further highlighted the transformative potential of PSMA PET/CT when integrated with advanced surgical techniques. By employing a robotic-assisted DROP-IN gamma probe, the study achieved a 100% detection rate of PSMA-avid lesions in patients undergoing salvage surgery for recurrent prostate cancer. Notably, 94% of targeted tumor deposits were completely resected, with intraoperative PSMA PET/CT guidance ensuring unparalleled precision. This innovative imaging-driven approach also reduced operative times, with a median duration of 120 min, and minimized blood loss to a median of just 50 mL. The postoperative outcomes were equally impressive, with 82% of patients experiencing a PSA reduction of ≥50% within four weeks. These findings underscore the clinical utility of PSMA PET/CT not only in radiotherapy planning, but also as a critical tool for enhancing surgical precision and achieving optimal disease control in salvage scenarios [
5].
These findings collectively demonstrate the transformative impact of advanced imaging modalities in MDT, enabling the precise localization of resistant tumor clones, guiding surgical or radiotherapy interventions, and ultimately improving patient outcomes.
3.2. Evidence Supporting SABR and Its Clinical Impact in Oligoprogressive mCRPC
Key clinical trials have provided substantial evidence for MDT in CRPC. The ICE-PAC phase II trial, as detailed by Kwan et al., assessed the combination of SABR and the PD-L1 immune checkpoint inhibitor (ICI) avelumab in patients with mCRPC. The study reported a disease control rate (DCR) of 48% (95% CI: 30–67%), indicating that nearly half of the patients achieved a complete response, partial response, or stable disease for at least six months. Additionally, an objective response rate (ORR) of 31% (95% CI: 11–59%) was observed, with responses extending to 33% of non-irradiated lesions. The trial also revealed that the median radiographic progression-free survival (rPFS) was 8.4 months (95% CI: 4.5—not reached), and median overall survival (OS) was 14.1 months (95% CI: 8.9—not reached). A safety analysis found that 90% of patients experienced treatment-related adverse events (TRAEs), though only 16% had grade 3–4 TRAEs, and 10% discontinued treatment due to toxicity [
6]. These results underline the potential of combining SABR with ICI to enhance disease control in heavily pretreated mCRPC populations. Importantly, the trial demonstrated that the therapeutic effects extended beyond irradiated sites, indicating a systemic benefit and supporting the inclusion of this combination in future clinical protocols.
The MEDCARE phase II trial, led by Kato Rans et al., showcased the significant role of SABR in delaying systemic progression and improving patient outcomes in cases of oligoprogressive mCRPC. Specifically, the trial reported a median next-line systemic treatment-free survival (NEST-FS) of 17 months (95% CI: 9–25 months), with a 2-year NEST-FS rate of 35%. Importantly, patients who received SABR to all lesions identified on baseline PSMA PET/CT had a significantly longer NEST-FS of 31 months compared to 13 months in those with untreated lesions (
p = 0.002). The study also observed a 2-year local control rate of 95%, highlighting SABR’s efficacy in managing treated lesions. PSA levels decreased in 65% of patients, with 45% achieving a ≥50% reduction. Toxicity was minimal, with no grade 3 or higher adverse events reported, and early grade 2 gastrointestinal/genitourinary toxicities occurred in only 15% of patients [
7]. These findings emphasize SABR’s capacity not only to delay systemic therapies, but also to enhance quality of life by maintaining effective disease control with low toxicity, making it a viable strategy for managing oligoprogressive mCRPC.
Zhang et al. highlighted that SABR markedly enhanced the median PFS in patients with oligoprogressive disease. The study found that PSA PFS reached a median of 9.2 months, with 39.5% of patients remaining free from progression at 1 year and 20.8% at 2 years. SABR also provided sustained local control, with local PFS rates of 84.4% at 1 year and 75.3% at 2 years. Additionally, the treatment delayed systemic disease progression, as distant PFS was observed at 17.6% and 5.0% at 1 and 2 years, respectively [
8]. These outcomes underscore the efficacy of SABR in maintaining localized control and postponing the advancement of systemic disease in patients with limited metastatic progression.
In the ARTO phase II randomized trial, Francolini et al. demonstrated that the combination of SABR with abiraterone acetate resulted in significantly improved disease control compared to systemic therapy alone. The study reported a median PFS of 10 months (95% CI, 8–17 months) in the treatment group, highlighting the enhanced efficacy of SABR combined with abiraterone in managing oligo-recurrent prostate cancer. Moreover, the median OS for the cohort was an impressive 114 months (95% CI, 85–114 months), showcasing the long-term benefits of this approach [
9]. An updated analysis of the ARTO trial, based on 157 patients and a median follow-up of 27 months, demonstrated a positive trend favoring SABR in combination with abiraterone acetate for overall survival (OS). Among the 157 patients randomized 1:1, 29 deaths were recorded (9 in the SABR plus abiraterone arm versus 20 in the abiraterone-alone arm). Although median OS was not reached in either treatment group, the experimental arm showed a non-significant trend towards better OS (
p = 0.07). This underscores the promising role of SABR in enhancing outcomes when combined with systemic therapy in oligometastatic CRPC, though a longer follow-up is necessary to determine the definitive impact of this combined approach [
10]. These findings underscore the potential of SABR and abiraterone acetate as a synergistic strategy for achieving superior disease control and potentially improving survival in patients with limited metastatic progression.
Onal et al. demonstrated the efficacy of SABR in oligoprogressive mCRPC through a retrospective analysis of 67 patients with 133 lesions treated with SBRT based on 68Ga-PSMA-PET/CT imaging. At a median follow-up of 17.5 months, the 2-year overall survival (OS) rate was 86.9%, with a median PFS of 16.6 months (95% CI, 11.8–21.4 months) and a median time to systemic treatment change of 16.4 months. Notably, a PSA response (≥25% decrease) was observed in 73.1% of patients, and a PSA nadir was achieved in 52.9%. Among the 37 patients who progressed after SBRT, 48.6% experienced oligometastatic relapse, while 35.1% exhibited diffuse progression, highlighting SABR's ability to contain disease progression in most cases [
11].
Le Guevelou et al. further demonstrated SABR’s benefits in delaying the need for systemic therapies in CRPC. SABR, when combined with androgen receptor pathway inhibitors, extended PFS with a hazard ratio of 0.35 (p < 0.001) and prolonged next-line systemic treatment-free survival (NEST-FS) to a median of two to three years, depending on progression type. Moreover, the treatment was associated with a favorable safety profile, as grade 3 toxicities were observed in less than 5% of cases [
12]. These findings collectively underscore SABR's dual role in prolonging the effectiveness of systemic therapies and ensuring long-term disease stability in oligoprogressive mCRPC, emphasizing its integration into treatment protocols for selected patients.
Deek et al. demonstrated that SABR significantly delayed key clinical endpoints in patients with oligoprogressive mCRPC. SABR-treated patients achieved a median PSA failure time of 9.7 months (95% CI, 6.8–13.3 months) compared to 4.2 months (95% CI, 3.3–9.5 months) in patients receiving systemic therapy alone (p = 0.066). The median time to next intervention (TTNI) was 14.9 months (95% CI, 10.9–20.3 months) for SABR patients, compared to 8.8 months (95% CI, 7.5–11.2 months) for systemic therapy alone (p = 0.03), and the median distant metastasis-free survival (DMFS) was 12.7 months versus 8.9 months (p = 0.05). Two-year overall survival (OS) was also higher in the SABR cohort (90.3%) compared to systemic therapy alone (76.8%) [
13].
Similarly, Eule et al. validated the utility of SABR in prolonging treatment efficacy in a cohort of 32 patients, where the median time to next systemic therapy (TTNST) was 10.1 months (95% CI, 7.5–14.8 months) and increased to 11.3 months when all progressive lesions were treated (p = 0.24). PSA responses were notable, with 40.6% achieving a ≥50% reduction at three months post-treatment. The median OS in the SABR cohort reported by Eule et al. was 40.3 months, demonstrating the extended benefits of SABR [
14]. These findings collectively highlight SABR’s role in deferring systemic therapies, maintaining disease control, and serving as a cornerstone in managing oligoprogressive mCRPC, while ensuring high tolerability and minimal toxicity.
3.3. Evidence Supporting Surgical Interventions and Their Clinical Impact in CRPC
Although surgical interventions are less studied, their potential in oligoprogressive mCRPC is notable. Surgical metastasectomy could complement SABR by addressing isolated metastatic lesions resistant to systemic therapies. Soma et al. presented a case of repeat oligoprogressive mCRPC where a solitary lung metastasis was successfully treated with thoracoscopic pulmonary metastasectomy. The patient achieved undetectable PSA levels for nine months post surgery, highlighting the potential efficacy of metastasectomy in selected patients [
15].
Nozaki et al. further highlighted the potential of surgical interventions in oligoprogressive mCRPC by demonstrating the efficacy of lymphadenectomy guided by 11C-choline PET/CT imaging. Among 12 patients treated, 7 underwent salvage lymphadenectomy for progressive lymph node metastases, achieving a median PSA reduction of 86% (range: 23–100%) post surgery.. The median PFS after lymphadenectomy was 8.5 months (range: 2.8–25.3 months), and the median time to initiation of systemic therapy was delayed to 9.3 months (range: 2.8–25.3 months). Notably, one patient achieved complete remission lasting 25.3 months, highlighting the potential durability of surgical outcomes in well-selected patients. These findings underscore the role of advanced imaging in patient selection and the ability of surgery to provide substantial disease control, even in cases resistant to systemic therapy [
16].
The ongoing MEDCARE phase III trial builds upon the findings of the phase II study by rigorously evaluating the integration of surgical interventions, including metastasectomy, alongside other progression-directed therapies in oligoprogressive mCRPC. While the MEDCARE phase II trial primarily focused on the feasibility and efficacy of SABR, it also highlighted the potential role of surgical approaches, such as metastasectomy, in managing oligoprogressive disease. Although no patients underwent metastasectomy in phase II, the trial underscored the importance of addressing all progressive lesions to achieve optimal clinical outcomes, paving the way for further research [
4]. The phase III trial, a randomized multicentric study with a planned enrollment of 246 patients stratified by the number and location of progressive lesions, aims to provide critical insights into the role of metastasectomy in enhancing disease control, improving quality of life (QoL), and delaying systemic therapies. With OS as the primary endpoint and secondary outcomes including NEST-FS, local control rates, and treatment-related toxicity, this study seeks to validate the utility of surgical strategies within multimodal treatment protocols, offering a comprehensive framework for progression-directed therapy in mCRPC [
17].
Pelvic exenteration surgery (PES) has demonstrated substantial potential for symptom management in locally advanced and symptomatic CRPC. In a study involving 103 patients, PES achieved symptom-free survival in 93.2% of cases, with a median symptom-free duration of 27.9 months. Additionally, the one-year and three-year symptom-free survival rates were 89.2% and 64.1%, respectively. The procedure reduced critical symptoms such as urinary obstruction (62.1% preoperatively to 1.6% postoperatively), gross hematuria (41.7% to 0%), and opioid analgesic use (22.3% to 2.9%). The median overall survival reached 33.6 months, with 92.2% survival at one year and 43.7% at three years. Grade 3 and 4 complications occurred in 11.6% and 8.1% of patients, respectively, underscoring the importance of careful patient selection [
18]. These findings reinforce the complementary role of surgery in managing localized progression, particularly when systemic therapies fail, offering durable symptom control and extended intervals before systemic treatment initiation.
3.4. Combination Therapies: MDT with Systemic Agents
Combining MDT with systemic therapies has shown promise. The ICE-PAC trial highlighted the role of SABR in enhancing immunotherapy’s efficacy. Specifically, the combination of SABR and avelumab achieved a significant improvement in rPFS, reaching 8.4 months compared to 3.8 months for immunotherapy alone (
p = 0.016). A subgroup analysis indicated a notable benefit for PD-L1-positive tumors, underscoring the potential of biomarker-driven treatment strategies [
3]. Recent analyses suggest that MDT combined with next-generation androgen receptor inhibitors (ARIs), such as enzalutamide or apalutamide, improves outcomes in oligoprogressive mCRPC. Patients treated with SABR and ARIs showed a longer median time to progression (TTP) of 14.5 months compared to 8.3 months for ARIs alone (
p = 0.03), while achieving PSA reductions of ≥50% in 35% of cases within six months [
14]. Another promising combination involves MDT paired with radiopharmaceutical agents such as 177Lu-PSMA-617. This approach prolonged biochemical PFS to 12.1 months versus 7.4 months for 177Lu-PSMA-617 monotherapy (
p = 0.045), and reduced skeletal-related events to 10% compared to 22% in the radiopharmaceutical-only group. These findings support the integration of radiopharmaceuticals with SABR for enhanced systemic and localized disease control [
12]. MDT also shows promise in amplifying the effects of chemotherapy. A prospective cohort evaluating SABR combined with docetaxel demonstrated a median overall survival (OS) of 42.6 months compared to 29.8 months for docetaxel monotherapy (HR 0.65,
p = 0.008). This combination was particularly effective in patients with low-volume metastatic disease, highlighting the additive effects of systemic and localized therapies [
13]. Zhang et al. explored SABR in combination with PARP inhibitors in patients with homologous recombination repair mutations. Their findings revealed a significant benefit for patients with BRCA1/2 mutations, with median PFS extended to 13.2 months compared to 6.8 months for PARP inhibitors alone, and median time to systemic progression reaching 16.4 months versus 9.1 months for systemic therapy alone (
p < 0.01). These results highlight the importance of genetic biomarkers in optimizing MDT strategies [
8]. These findings collectively emphasize the versatility of MDT, particularly SABR, in combination with systemic therapies. Whether enhancing immune responses, targeting genetic vulnerabilities, or augmenting traditional chemotherapy and radiopharmaceuticals, MDT offers a tailored approach that maximizes outcomes in oligoprogressive mCRPC.
3.5. The Role of Multidisciplinary Teams in Managing Oligoprogressive mCRPC
The optimal management of oligoprogressive mCRPC requires a comprehensive multidisciplinary team (MuDT) involving urologists, oncologists, radiologists, nuclear medicine specialists, and imaging experts. Belda-Ferre et al. reported that 62.8% of advanced prostate cancer cases presented at MuDT meetings required the collaboration of at least two specialties, emphasizing the complexity of care for these patients. Among surveyed physicians, 90.7% agreed that MuDTs improved clinical decision-making and patient satisfaction. However, only 60.5% of decisions made during MuDT meetings were binding, with variability in adherence across institutions, highlighting areas for improvement in integration and execution. Training in MuDT practices was found to be limited, with only 46.5% of respondents considering themselves adequately trained, although 37.2% reported that MuDT meetings significantly improved decision-making efficiency [
19].
Zhu et al. demonstrated that dynamic MuDT discussions at key treatment milestones for metastatic CRPC significantly improved OS. Patients participating in MuDT meetings had a median OS of 39.7 months compared to 27.0 months for non-participants (HR: 0.542,
p < 0.001). This survival benefit persisted across subgroups, including those undergoing first-line therapy (median OS: not reached vs. 27.0 months,
p < 0.001) and multi-line therapy (median OS: 36.7 months vs. 25.6 months,
p = 0.044). Moreover, MuDT involvement correlated with higher rates of advanced diagnostic utilization, with 38.4% of MuDT patients receiving multi-line therapies compared to 21.8% of non-MuDT participants (
p < 0.001), underscoring the importance of MuDTs in coordinating complex treatment strategies [
20].
3.6. Limitations and Future Directions
The surgical management of oligoprogressive mCRPC as a modality of MDT faces several critical limitations. First, as mentioned above, the existing evidence base for surgical interventions is significantly weaker than for SABR. While SABR is supported by prospective trials demonstrating consistent outcomes in delaying disease progression, most surgical studies are retrospective or based on small, single-center cohorts, which limits their reliability and generalizability. Additionally, robotic-assisted surgical techniques, such as RAVEIL, though promising, lack robust comparative data against SABR or other MDT modalities. However, as demonstrated in this case report, RAVEIL shows potential for advancing the surgical management of oligoprogressive mCRPC by achieving excellent local control, prolonged biochemical progression-free survival, and minimal complications. These findings highlight the need for further studies to validate its efficacy and establish its role within MDT strategies. A further limitation lies in the higher perioperative risks and resource requirements associated with surgery. Unlike SABR, which is non-invasive and widely accessible, surgical approaches require specialized expertise, advanced equipment, and longer recovery times. These logistical and clinical challenges hinder the broader adoption and integration of surgery into standard MDT protocols for CRPC. Finally, the lack of standardization in reporting outcomes for surgical interventions complicates comparisons across studies. Definitions of key concepts such as “oligoprogression” and the reporting of endpoints such as PFS or systemic treatment-free survival (STFS) vary widely, making meta-analyses and evidence synthesis difficult. This case report underscores the importance of detailed and standardized outcome reporting in surgical studies, providing a valuable foundation for future research.
To address these gaps, future research should prioritize the development of high-quality, prospective randomized controlled trials (RCTs) directly comparing surgical interventions with SABR in oligoprogressive mCRPC. These trials should emphasize robust endpoints, such as PFS, OS, QoL, and systemic treatment-free intervals, while also incorporating cost-effectiveness analyses to evaluate broader implications. Advanced imaging techniques, such as PSMA PET/CT, should be systematically integrated into surgical research to optimize patient selection and enhance procedural precision. Prospective studies should assess how imaging-guided approaches can improve outcomes by identifying metastatic lesions that may benefit more from surgical resection than from SABR. Collaborative multicenter studies will be essential to overcome challenges associated with the rarity of oligoprogressive mCRPC. International networks pooling resources and expertise can enable larger patient cohorts and more statistically robust analyses. Additionally, biomarker-driven research should identify specific patient subgroups most likely to benefit from surgical interventions, facilitating personalized treatment approaches. Finally, the integration of surgery into multimodal treatment regimens offers potential for enhanced outcomes. Combining surgery with systemic therapies such as androgen receptor inhibitors (ARIs), immunotherapies, or radiopharmaceuticals like 177Lu-PSMA-617 could provide synergistic effects and improve long-term disease control. Future research should also investigate the optimal timing of surgical interventions within the treatment continuum, particularly in relation to systemic therapies and radiotherapy.
4. Conclusions
Building on the evidence supporting MDTs in oligoprogressive mCRPC, salvage RAVEIL emerges as a promising surgical approach within this therapeutic framework. By utilizing advanced imaging modalities such as 68Ga-PSMA PET/CT for precise targeting, this approach enables localized treatment while reducing the reliance on systemic therapies. In our case report, RAVEIL demonstrated durable disease control with minimal morbidity, emphasizing its practicality and safety in clinical practice.
These findings support the inclusion of RAVEIL within a multidisciplinary framework for mCRPC management, particularly for patients with isolated metastatic progression. Future prospective studies are needed to confirm its efficacy and long-term outcomes compared to non-invasive MDT modalities such as SABR. Moreover, expanding the application of robotic-assisted surgical techniques to other metastatic sites could further enhance their role in MDT strategies for mCRPC. Enhancing collaboration among specialties and further advancements in imaging and surgical techniques will be essential for optimizing RAVEIL and other robotic-assisted approaches in personalized oncology care.
Author Contributions
Conceptualization: R.B.D.; Methodology: R.B.D.; Software: R.B.D.; Validation: R.B.D. and A.A.A.; Formal Analysis: R.B.D.; Investigation: R.B.D.; Resources: R.B.D. and M.L.; Data Curation: R.B.D.; Writing—Original Draft Preparation: R.B.D.; Writing—Review and Editing: R.B.D.; Visualization: R.B.D.; Supervision: A.A.A.; Project Administration: R.B.D.; Funding Acquisition: R.B.D. All authors have read and agreed to the published version of the manuscript.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Institutional Review Board Statement
Ethical review and approval were waived for this study due to its retrospective design and data collection. The study did not involve any prospective interventions or procedures requiring ethical approval, in line with applicable ethical standards.
Informed Consent Statement
Informed consent for publication and surgical procedure was obtained from all subjects involved in the study for the performed medical procedure. The data presented in the manuscript have been fully anonymized, and no details that could lead to patient identification are included. Additionally, all images used in the manuscript have been reviewed to ensure they do not contain identifiable patient information.
Conflicts of Interest
The authors have no relevant financial or non-financial interests to disclose.
References
- Ost, P.; Reynders, D.; Decaestecker, K.; Fonteyne, V.; Lumen, N.; De Bruycker, A.; Lambert, B.; Delrue, L.; Bultijnck, R.; Claeys, T.; et al. Surveillance or Metastasis-Directed Therapy for Oligometastatic Prostate Cancer Recurrence: A Prospective, Randomized, Multicenter Phase II Trial. J. Clin. Oncol. 2018, 36, 446–453. [CrossRef]
- Phillips, R.; Shi, W.Y.; Deek, M.; Radwan, N.; Lim, S.J.; Antonarakis, E.S.; Rowe, S.P.; Ross, A.E.; Gorin, M.A.; Deville, C.; et al. Outcomes of Observation vs Stereotactic Ablative Radiation for Oligometastatic Prostate Cancer: The ORIOLE Phase 2 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 650–659. [CrossRef]
- Pan, J.; Wei, Y.; Zhang, T.; Liu, C.; Hu, X.; Zhao, J.; Gan, H.; Liu, W.; Zhu, B.; Wu, J.; et al. Stereotactic radiotherapy for lesions detected via 68Ga-prostate-specific membrane antigen and 18F-fluorodeoxyglucose PET/CT in patients with nonmetastatic prostate cancer with early PSA progression on androgen deprivation therapy: A prospective single-center study. Eur. Urol. Oncol. 2022, 5, 420–427. [CrossRef]
- Hahn, A.W.; Aparicio, A.; Jadvar, H.; Poon, D.M.C. MDT perspective: Innovative applications of stereotactic body radiation therapy in metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis. 2024. [CrossRef]
- de Barros, H.A.; van Oosterom, M.N.; Donswijk, M.L.; Hendrikx, J.J.; Vis, A.N.; Maurer, T.; van Leeuwen, F.W.; van der Poel, H.G.; van Leeuwen, P.J. Robot-assisted PSMA-radioguided salvage surgery in recurrent prostate cancer using a DROP-IN gamma probe: The first prospective feasibility study. Eur. Urol. 2022, 82, 97–105. [CrossRef]
- Kwan, E.M.; Spain, L.; Anton, A.; Gan, C.L.; Garrett, L.; Chang, D.; Liow, E.; Bennett, C.; Zheng, T.; Yu, J.; et al. Avelumab Combined with Stereotactic Ablative Body Radiotherapy in Metastatic Castration-Resistant Prostate Cancer: The Phase 2 ICE-PAC Clinical Trial. Eur. Urol. 2021. [CrossRef]
- Rans, K.; Joniau, S.; Berghen, C.; Goffin, K.; Dumez, H.; Haustermans, K.; De Meerleer, G. Progression-directed therapy in oligoprogressive castration-resistant prostate cancer: Final results from the prospective, single-arm, phase 2 MEDCARE trial. Eur. Urol. Oncol. 2024, 7, 1441–1450. [CrossRef]
- Zhang, H.; Orme, J.J.; Abraha, F.; Stish, B.J.; Lowe, V.J.; Lucien, F.; Tryggestad, E.J.; Bold, M.S.; Pagliaro, L.C.; Choo, C.R.; et al. Phase II Evaluation of 8Stereotactic Ablative Radiotherapy (SABR) and Immunity in 11C-Choline-PET/CT–Identified Oligometastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2021, 27, 6376–6383. [CrossRef]
- Francolini, G.; Garlatti, P.; Di Cataldo, V.; Triggiani, L.; Simoni, N.; Detti, B.; Lorenzetti, V.; Colombo, F.; Morelli, V.; Ganovelli, M.; et al. Pattern of recurrence after stereotactic body radiotherapy for para-aortic oligo-recurrent prostate cancer: A multicentric analysis. Radiother. Oncol. 2023, 128, 1423–1428. [CrossRef]
- Francolini, G.; Di Cataldo, V.; Garlatti, P.; Detti, B.; Caini, S.; Bruni, A.; Ingrosso, G.; D’Angelillo, R.M.; Alitto, A.R.; Augugliaro, M.; et al. ARTO trial: Overall survival analysis from a randomized phase II trial testing the benefit of adding stereotactic body radiotherapy to abiraterone acetate in patients with oligometastatic castrate-resistant prostate cancer. J. Clin. Oncol. 2024, 42 (Suppl. 6), 132. [CrossRef]
- Onal, C.; Ozyigit, G.; Oymak, E.; Guler, O.C.; Tilki, B.; Hurmuz, P.; Akyol, F. Stereotactic radiotherapy to oligoprogressive lesions detected with 68Ga-PSMA-PET/CT in castration-resistant prostate cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 3612–3621. [CrossRef]
- Le Guevelou, J.; Cuccia, F.; Flippot, R.; Ferrera, G.; Terlizzi, M.; Zilli, T.; De Crevoisier, R.; Hannoun-Levi, J.M.; Supiot, S.; Sargos, P.; et al. The current landscape of stereotactic body radiation therapy for metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis. 2024. [CrossRef]
- Deek, M.P.; Taparra, K.; Phillips, R.; Velho, P.I.; Gao, R.W.; Deville, C.; Song, D.Y.; Greco, S.; Carducci, M.; Eisenberger, M.; et al. Metastasis-directed therapy prolongs efficacy of systemic therapy and improves clinical outcomes in oligoprogressive castration-resistant prostate cancer. Eur. Urol. Oncol. 2021, 4, 447–455. [CrossRef]
- Eule, C.J.; Candelario, N.; Nath, S.K.; Robin, T.P. Time to Next Systemic Therapy After Stereotactic Body Radiation Therapy for Oligoprogressive Metastatic Castrate-Resistant Prostate Cancer. Adv. Radiat. Oncol. 2024, 9, 101655. [CrossRef]
- Soma, T.; Yoshida, S.; Wakejima, R.; Taguchi, T.; Fukuda, S.; Tanaka, H.; Yokoyama, M.; Ohashi, K.; Okubo, K.; Fujii, Y. A case of repeat oligoprogressive castration-resistant prostate cancer treated with pulmonary metastasectomy. IJU Case Rep. 2023, 6, 216–218. [CrossRef]
- Nozaki, T.; Shimizu, T.; Kawai, N.; Fukushima, T.; Yokoyama, K.; Nonomura, K.; et al. The efficacy 4 of salvage lymphadenectomy guided by 11C-choline PET/CT in patients with oligoprogressive castration-resistant prostate cancer. Prostate Cancer Prostatic Dis. 2023, 26, 567–574.
- Kato, R.; Goffin, K.; Joniau, S.; Daugaard, G.; den Hartog, J.; Van Wynsberge, L.; De Meerleer, G. The impact of progression-directed therapy on survival in metastatic castration-refractory prostate cancer: MEDCARE phase 3 trial. BJU Int. 2025, 135, 63–70. [CrossRef]
- Heidenreich, A.; Bludau, M.; Bruns, C.; Nestler, T.; Porres, D.; Pfister, D.J.K.P. Pelvic exenteration surgery in patients with locally advanced castration-naïve and castration-resistant symptomatic prostate cancer. BJU Int. 2020, 126, 651–659. [CrossRef]
- Belda-Ferre, M.; Garcia-Segui, A.; Pacheco-Bru, J.J.; Valencia-Guadalajara, V.J.; Verdú-Verdú, L.P.; Sánchez-Cano, E.; Chillón-Sempere, F.S.; Vázquez-Mazón, F. Multicenter study on multidisciplinary committees in advanced prostate cancer. Actas Urol. Esp. 2022, 46, 106–113. [CrossRef]
- Zhu, S.; Chen, J.; Ni, Y.; Zhang, H.; Liu, Z.; Shen, P.; Sun, G.; Liang, J.; Zhang, X.; Wang, Z.; et al. Dynamic multidisciplinary team discussions can improve the prognosis of metastatic castration-resistant prostate cancer patients. Prostate 2021, 81, 721–727. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).