Submitted:
13 January 2025
Posted:
14 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanisms of Drug-Induced Nephrotoxicity
2.1. Changes in Glomerular Hemodynamics
2.2. Tubular Cell Toxicity
2.3. Inflammation
2.4. Crystal Nephropathy
2.5. Rhabdomyolysis
2.6. Thrombotic Microangiopathy
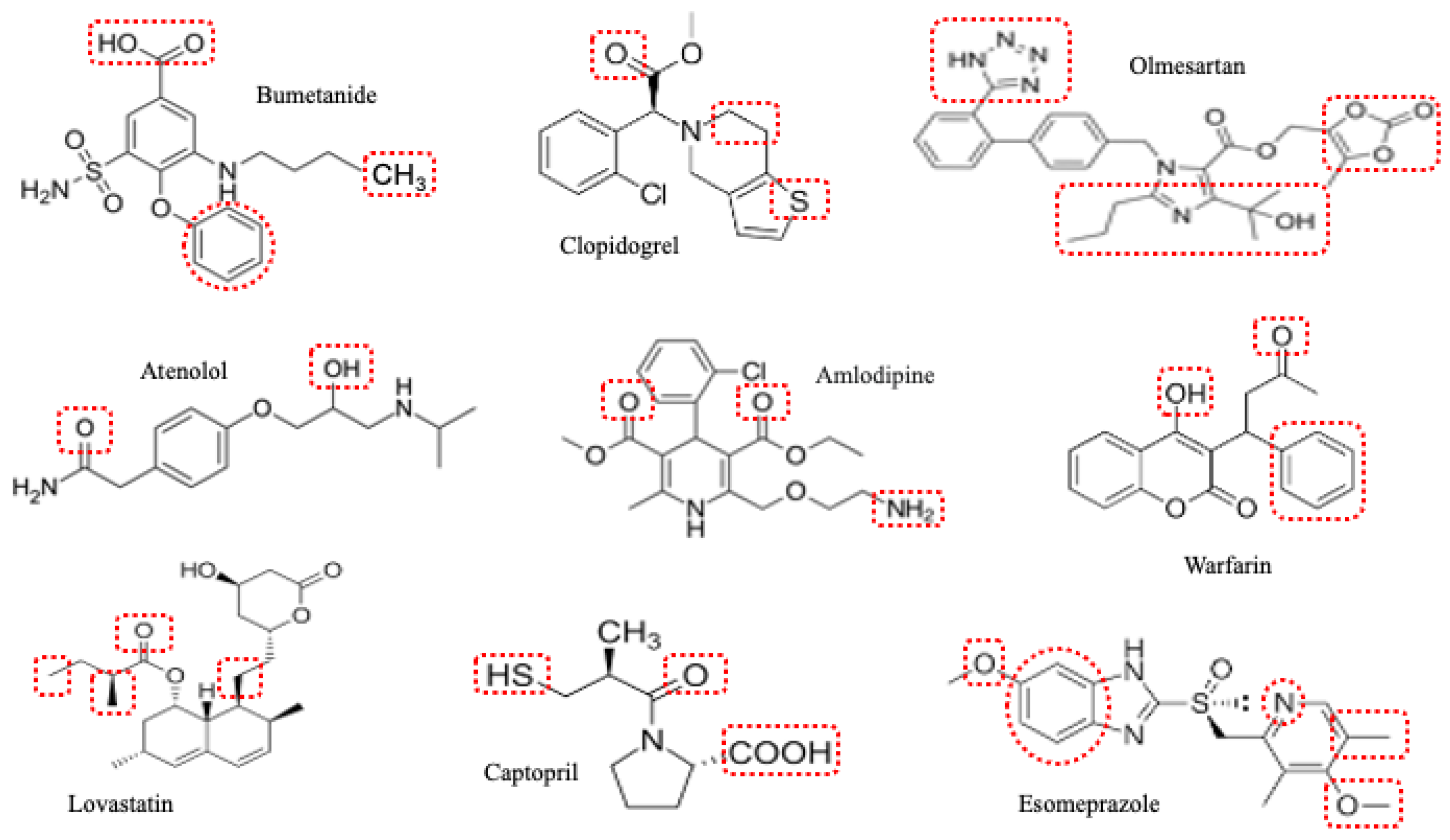
3. Renal Effects of Cardiovascular Drugs
3.1. Diuretics
3.2. Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers
3.3. Calcium Channel Blockers
3.4. Beta-Blockers
3.5. Antiplatelet Agents
3.6. Anticoagulants
3.7. Statins
3.8. Proton-Pump Inhibitors
3.9. Contrast Media
4. Role of Oxidative Stress in Drug-Induced Nephrotoxicity
- developing novel and specific biomarkers of oxidative stress and drug-induced nephrotoxicity that can reflect the degree and location of renal injury, predict the risk and outcome of drug-induced nephrotoxicity, monitor the response to treatment, and guide personalized therapy [9];
- identifying novel and effective antioxidants that can target specific sources or pathways of oxidative stress and drug-induced nephrotoxicity, modulate redox signaling, protect renal cells and tissues from oxidative damage, and preserve or restore renal function [192];
- designing personalized and precision antioxidant therapy based on individual characteristics and needs, such as genetic background, epigenetic modifications, co-morbidities, co-administered drugs, environmental factors, and oxidative stress status.
- exploring the potential synergistic or additive effects of combining antioxidant therapy with other therapeutic modalities, such beta-blockers [193].
- assessing the long-term benefits and risks of antioxidant therapy for drug-induced nephrotoxicity on various clinical outcomes, such as renal function preservation, cardiovascular protection, quality of life improvement, and survival extension.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Van Vleet, T.R.; Schnellmann, R.G. Toxic nephropathy: environmental chemicals. Seminars in nephrology 2003, 23, 500–508. [Google Scholar] [CrossRef]
- Dalal, R.; Bruss, Z.S.; Sehdev, J.S. Physiology, Renal Blood Flow and Filtration. Updated 2023 Jul 24. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482248/.
- Sanchez-Alamo, B.; Cases-Corona, C.; Fernandez-Juarez, G. Facing the Challenge of Drug-Induced Acute Interstitial Nephritis. Nephron 2023, 147, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Rennke, H.G.; Bradley, M.D. Renal Pathophysiology. The essentials. fourth ed. Baltimore, MD: Wolters Kluwer- Lippincot Williams & Wilkins. 2014. [Google Scholar]
- Petejova, N.; Martinek, A.; Zadrazil, J.; Teplan, V. Acute toxic kidney injury. Renal failure 2019, 41, 576–594. [Google Scholar] [CrossRef]
- Ghane Shahrbaf, F.; Assadi, F. Drug-induced renal disorders. Journal of renal injury prevention 2015, 4, 57–60. [Google Scholar] [CrossRef]
- Pazhayattil, G.S.; Shirali, A.C. Drug-induced impairment of renal function. International journal of nephrology and renovascular disease 2014, 7, 457–468. [Google Scholar] [CrossRef]
- Dobrek, L. A Synopsis of Current Theories on Drug-Induced Nephrotoxicity. Life (Basel, Switzerland) 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Moon, A. Drug-induced nephrotoxicity and its biomarkers. Biomolecules & therapeutics 2012, 20, 268–272. [Google Scholar] [CrossRef]
- Kaufman, D.P.; Basit, H.; Knohl, S.J. Physiology, Glomerular Filtration Rate. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Hajira Basit declares no relevant financial relationships with ineligible companies. Disclosure: Stephen Knohl declares no relevant financial relationships with ineligible companies.: StatPearls Publishing. Copyright © 2023, StatPearls Publishing LLC.; 2023.
- Hostetter, T.H.; Rosenberg, M.E. Hemodynamic effects of glomerular permselectivity. American journal of nephrology 1990, 10 Suppl 1, 24–27. [Google Scholar] [CrossRef]
- Horl, W.H. Nonsteroidal Anti-Inflammatory Drugs and the Kidney. Pharmaceuticals (Basel) 2010, 3, 2291–2321. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.J.; Vaughan, D.E. Angiotensin-Converting Enzyme Inhibitors. 1998, 97, 1411–1420. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.M.; Trepiccione, F.; Unwin, R.J. Drug toxicity in the proximal tubule: new models, methods and mechanisms. Pediatric nephrology (Berlin, Germany) 2022, 37, 973–982. [Google Scholar] [CrossRef]
- Gai, Z.; Gui, T.; Kullak-Ublick, G.A.; Li, Y.; Visentin, M. The Role of Mitochondria in Drug-Induced Kidney Injury. Frontiers in physiology 2020, 11, 1079. [Google Scholar] [CrossRef]
- Krause, K.M.; Serio, A.W.; Kane, T.R.; Connolly, L.E. Aminoglycosides: An Overview. Cold Spring Harbor perspectives in medicine 2016, 6. [Google Scholar] [CrossRef]
- Yano, T.; Itoh, Y.; Kawamura, E.; Maeda, A.; Egashira, N.; Nishida, M.; Kurose, H.; Oishi, R. Amphotericin B-induced renal tubular cell injury is mediated by Na+ Influx through ion-permeable pores and subsequent activation of mitogen-activated protein kinases and elevation of intracellular Ca2+ concentration. Antimicrobial agents and chemotherapy 2009, 53, 1420–1426. [Google Scholar] [CrossRef] [PubMed]
- Varlam, D.E.; Siddiq, M.M.; Parton, L.A.; Rüssmann, H. Apoptosis contributes to amphotericin B-induced nephrotoxicity. Antimicrobial agents and chemotherapy 2001, 45, 679–685. [Google Scholar] [CrossRef]
- Tanji, N.; Tanji, K.; Kambham, N.; Markowitz, G.S.; Bell, A.; D'Agati V, D. Adefovir nephrotoxicity: possible role of mitochondrial DNA depletion. Human pathology 2001, 32, 734–740. [Google Scholar] [CrossRef]
- Deray, G.; Martinez, F.; Katlama, C.; Levaltier, B.; Beaufils, H.; Danis, M.; Rozenheim, M.; Baumelou, A.; Dohin, E.; Gentilini, M.; et al. Foscarnet nephrotoxicity: mechanism, incidence and prevention. American journal of nephrology 1989, 9, 316–321. [Google Scholar] [CrossRef]
- Caiazza, A.; Russo, L.; Sabbatini, M.; Russo, D. Hemodynamic and tubular changes induced by contrast media. BioMed research international 2014, 2014, 578974. [Google Scholar] [CrossRef] [PubMed]
- Imig, J.D.; Ryan, M.J. Immune and inflammatory role in renal disease. Comprehensive Physiology 2013, 3, 957–976. [Google Scholar] [CrossRef] [PubMed]
- Barreto, E.F.; Rule, A.D. Management of Drug-Associated Acute Interstitial Nephritis. Kidney360 2020, 1, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Lucas, G.N.C.; Leitão, A.C.C.; Alencar, R.L.; Xavier, R.M.F.; Daher, E.F.; Silva Junior, G.B.D. Pathophysiological aspects of nephropathy caused by non-steroidal anti-inflammatory drugs. Jornal brasileiro de nefrologia 2019, 41, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Farouk, S.S.; Rein, J.L. The Many Faces of Calcineurin Inhibitor Toxicity-What the FK? Advances in chronic kidney disease 2020, 27, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Gong, R.; Wang, P.; Dworkin, L. What we need to know about the effect of lithium on the kidney. American journal of physiology. Renal physiology 2016, 311, F1168–f1171. [Google Scholar] [CrossRef]
- Santos, M.L.C.; de Brito, B.B.; da Silva, F.A.F.; Botelho, A.; de Melo, F.F. Nephrotoxicity in cancer treatment: An overview. World journal of clinical oncology 2020, 11, 190–204. [Google Scholar] [CrossRef]
- Keen, M.U.; Aeddula, N.R. Analgesic Nephropathy. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Narothama Aeddula declares no relevant financial relationships with ineligible companies.: StatPearls Publishing. Copyright © 2023, StatPearls Publishing LLC.; 2023.
- Perazella, M.A.; Herlitz, L.C. The Crystalline Nephropathies. Kidney international reports 2021, 6, 2942–2957. [Google Scholar] [CrossRef]
- Mulay, S.R.; Shi, C.; Ma, X.; Anders, H.J. Novel Insights into Crystal-Induced Kidney Injury. Kidney diseases (Basel, Switzerland) 2018, 4, 49–57. [Google Scholar] [CrossRef]
- Genc, G.; Ozkaya, O.; Acikgöz, Y.; Yapici, O.; Bek, K.; Gülnar Sensoy, S.; Ozyürek, E. Acute renal failure with acyclovir treatment in a child with leukemia. Drug and chemical toxicology 2010, 33, 217–219. [Google Scholar] [CrossRef]
- Izzedine, H.; Launay-Vacher, V.; Deray, G. Antiviral drug-induced nephrotoxicity. American journal of kidney diseases: the official journal of the National Kidney Foundation 2005, 45, 804–817. [Google Scholar] [CrossRef]
- Derebail, V.K.; McGregor, J.G.; Colindres, R.E.; Singh, H.K.; Kshirsagar, A.V. The Case: Acute kidney injury in a patient with P. carinii pneumonia. Kidney international 2009, 75, 865–866. [Google Scholar] [CrossRef]
- Perazella, M.A. Crystal-induced acute renal failure. The American journal of medicine 1999, 106, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Hamed, K.M.; Dighriri, I.M.; Baomar, A.F.; Alharthy, B.T.; Alenazi, F.E.; Alali, G.H.; Alenazy, R.H.; Alhumaidi, N.T.; Alhulayfi, D.H.; Alotaibi, Y.B.; et al. Overview of Methotrexate Toxicity: A Comprehensive Literature Review. Cureus 2022, 14, e29518. [Google Scholar] [CrossRef] [PubMed]
- Yarlagadda, S.G.; Perazella, M.A. Drug-induced crystal nephropathy: an update. Expert opinion on drug safety 2008, 7, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Hebert, J.F.; Burfeind, K.G.; Malinoski, D.; Hutchens, M.P. Molecular Mechanisms of Rhabdomyolysis-Induced Kidney Injury: From Bench to Bedside. Kidney international reports 2023, 8, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Marcoff, L.; Thompson, P.D. The Role of Coenzyme Q10 in Statin-Associated Myopathy: A Systematic Review. Journal of the American College of Cardiology 2007, 49, 2231–2237. [Google Scholar] [CrossRef] [PubMed]
- Mollazadeh, H.; Tavana, E.; Fanni, G.; Bo, S.; Banach, M.; Pirro, M.; von Haehling, S.; Jamialahmadi, T.; Sahebkar, A. Effects of statins on mitochondrial pathways. Journal of cachexia, sarcopenia and muscle 2021, 12, 237–251. [Google Scholar] [CrossRef]
- Daras, M.; Kakkouras, L.; Tuchman, A.J.; Koppel, B.S. Rhabdomyolysis and hyperthermia after cocaine abuse: a variant of the neuroleptic malignant syndrome? Acta neurologica Scandinavica 1995, 92, 161–165. [Google Scholar] [CrossRef]
- Fernández-Cuadros, M.E.; Goizueta-San-Martin, G.; Varas-de-Dios, B.; Casique-Bocanegra, L.O.; Manrique-de-Lara-Cadiñanos, P.; Albaladejo-Florin, M.J.; Algarra-López, R.; Pérez-Moro, O.S. Colchicine-Induced Rhabdomyolysis: Clinical, Biochemical, and Neurophysiological Features and Review of the Literature. Clinical medicine insights. Arthritis and musculoskeletal disorders 2019, 12, 1179544119849883. [Google Scholar] [CrossRef]
- Packard, K.; Price, P.; Hanson, A. Antipsychotic use and the risk of rhabdomyolysis. Journal of pharmacy practice 2014, 27, 501–512. [Google Scholar] [CrossRef]
- Khosla, U.; Ruel, K.S.; Hunt, D.P. Antihistamine-induced rhabdomyolysis. Southern medical journal 2003, 96, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- Comelli, I.; Lippi, G.; Magnacavallo, A.; Cervellin, G. Mefloquine-associated rhabdomyolysis. The American journal of emergency medicine 2016, 34, 2250.e2255–2250.e2256. [Google Scholar] [CrossRef] [PubMed]
- Blain, P.G.; Lane, R.J.; Bateman, D.N.; Rawlins, M.D. Opiate-induced rhabdomyolysis. Human toxicology 1985, 4, 71–74. [Google Scholar] [CrossRef]
- Arnold, D.M.; Patriquin, C.J.; Nazy, I. Thrombotic microangiopathies: a general approach to diagnosis and management. CMAJ: Canadian Medical Association journal = journal de l'Association medicale canadienne 2017, 189, E153–e159. [Google Scholar] [CrossRef] [PubMed]
- Mazzierli, T.; Allegretta, F.; Maffini, E.; Allinovi, M. Drug-induced thrombotic microangiopathy: An updated review of causative drugs, pathophysiology, and management. Frontiers in pharmacology 2022, 13, 1088031. [Google Scholar] [CrossRef] [PubMed]
- Zakarija, A.; Kwaan, H.C.; Moake, J.L.; Bandarenko, N.; Pandey, D.K.; McKoy, J.M.; Yarnold, P.R.; Raisch, D.W.; Winters, J.L.; Raife, T.J.; et al. Ticlopidine- and clopidogrel-associated thrombotic thrombocytopenic purpura (TTP): review of clinical, laboratory, epidemiological, and pharmacovigilance findings (1989-2008). Kidney international. Supplement, 2009; S20–S24. [Google Scholar] [CrossRef]
- Zakarija, A.; Bennett, C. Drug-induced thrombotic microangiopathy. Seminars in thrombosis and hemostasis 2005, 31, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Page, E.E.; Little, D.J.; Vesely, S.K.; George, J.N. Quinine-Induced Thrombotic Microangiopathy: A Report of 19 Patients. American journal of kidney diseases: the official journal of the National Kidney Foundation 2017, 70, 686–695. [Google Scholar] [CrossRef] [PubMed]
- Daviet, F.; Rouby, F.; Poullin, P.; Moussi-Francès, J.; Sallée, M.; Burtey, S.; Mancini, J.; Duffaud, F.; Sabatier, R.; Pourroy, B.; et al. Thrombotic microangiopathy associated with gemcitabine use: Presentation and outcome in a national French retrospective cohort. British journal of clinical pharmacology 2019, 85, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Hilburg, R.; Geara, A.S.; Qiu, M.K.; Palmer, M.B.; Chiang, E.Y.; Burger, R.A.; Hogan, J.J. Bevacizumab-associated thrombotic microangiopathy treated with eculizumab: A case series and systematic review of the literature. Clinical nephrology 2021, 96, 51–59. [Google Scholar] [CrossRef]
- Noronha, V.; Punatar, S.; Joshi, A.; Desphande, R.V.; Prabhash, K. Sunitinib-induced thrombotic microangiopathy. Journal of cancer research and therapeutics 2016, 12, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Arumugham, V.B.; Shahin, M.H. Therapeutic Uses of Diuretic Agents. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Mohamed Shahin declares no relevant financial relationships with ineligible companies.: StatPearls Publishing. Copyright © 2023, StatPearls Publishing LLC.; 2023.
- Oh, S.W.; Han, S.Y. Loop Diuretics in Clinical Practice. Electrolyte & blood pressure: E & BP 2015, 13, 17–21. [Google Scholar] [CrossRef]
- Ho, K.M.; Power, B.M. Benefits and risks of furosemide in acute kidney injury. Anaesthesia 2010, 65, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Hegde, A. Diuretics in Acute Kidney Injury. Indian journal of critical care medicine: peer-reviewed, official publication of Indian Society of Critical Care Medicine 2020, 24, S98–s99. [Google Scholar] [CrossRef] [PubMed]
- Miltiadous, G.; Mikhailidis, D.P.; Elisaf, M. Acid-base and electrolyte abnormalities observed in patients receiving cardiovascular drugs. Journal of cardiovascular pharmacology and therapeutics 2003, 8, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Liu, H.; Qi, W.; Jiang, H.; Li, Y.; Wu, X.; Sun, H.; Gross, K.; Salvi, R. Ototoxic effects and mechanisms of loop diuretics. Journal of otology 2016, 11, 145–156. [Google Scholar] [CrossRef]
- Kondo, C.S.; Macchionne, M.; Nakagawa, N.K.; de Carvalho, C.R.; King, M.; Saldiva, P.H.; Lorenzi-Filho, G. Effects of intravenous furosemide on mucociliary transport and rheological properties of patients under mechanical ventilation. Critical care (London, England) 2002, 6, 81–87. [Google Scholar] [CrossRef]
- Mullens, W.; Abrahams, Z.; Francis, G.S.; Sokos, G.; Taylor, D.O.; Starling, R.C.; Young, J.B.; Tang, W.H.W. Importance of venous congestion for worsening of renal function in advanced decompensated heart failure. J Am Coll Cardiol 2009, 53, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Damman, K.; van Deursen, V.M.; Navis, G.; Voors, A.A.; van Veldhuisen, D.J.; Hillege, H.L. Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol 2009, 53, 582–588. [Google Scholar] [CrossRef]
- Mullens, W.; Abrahams, Z.; Skouri, H.N.; Francis, G.S.; Taylor, D.O.; Starling, R.C.; Paganini, E.; Tang, W.H. Elevated intra-abdominal pressure in acute decompensated heart failure: a potential contributor to worsening renal function? J Am Coll Cardiol 2008, 51, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Jessup, M.; Costanzo, M.R. The cardiorenal syndrome: do we need a change of strategy or a change of tactics? J Am Coll Cardiol 2009, 53, 597–599. [Google Scholar] [CrossRef] [PubMed]
- Palazzuoli, A.; Ruocco, G.; Ronco, C.; McCullough, P.A. Loop diuretics in acute heart failure: beyond the decongestive relief for the kidney. Critical care (London, England) 2015, 19, 296. [Google Scholar] [CrossRef]
- Hasselblad, V.; Gattis Stough, W.; Shah, M.R.; Lokhnygina, Y.; O'Connor, C.M.; Califf, R.M.; Adams, K.F., Jr. Relation between dose of loop diuretics and outcomes in a heart failure population: results of the ESCAPE trial. European journal of heart failure 2007, 9, 1064–1069. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, W.; Ren, H.; Chen, X.; Xie, J.; Chen, N. Diuretics associated acute kidney injury: clinical and pathological analysis. Renal failure 2014, 36, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Milliner, D.S.; Wooldridge, T.D.; Sethi, S. Triamterene crystalline nephropathy. American journal of kidney diseases: the official journal of the National Kidney Foundation 2014, 63, 148–152. [Google Scholar] [CrossRef]
- Zhang, Y.; He, D.; Zhang, W.; Xing, Y.; Guo, Y.; Wang, F.; Jia, J.; Yan, T.; Liu, Y.; Lin, S. ACE Inhibitor Benefit to Kidney and Cardiovascular Outcomes for Patients with Non-Dialysis Chronic Kidney Disease Stages 3-5: A Network Meta-Analysis of Randomised Clinical Trials. Drugs 2020, 80, 797–811. [Google Scholar] [CrossRef]
- Baltatzi, M.; Savopoulos, C.; Hatzitolios, A. Role of angiotensin converting enzyme inhibitors and angiotensin receptor blockers in hypertension of chronic kidney disease and renoprotection. Study results. Hippokratia 2011, 15, 27–32. [Google Scholar] [PubMed]
- Schoolwerth, A.C.; Sica, D.A.; Ballermann, B.J.; Wilcox, C.S. Renal Considerations in Angiotensin Converting Enzyme Inhibitor Therapy. 104 2001, 104, 1985–1991. [Google Scholar] [CrossRef]
- Knight, E.L.; Glynn, R.J.; McIntyre, K.M.; Mogun, H.; Avorn, J. Predictors of decreased renal function in patients with heart failure during angiotensin-converting enzyme inhibitor therapy: Results from the Studies of Left Ventricular Dysfunction (SOLVD). American Heart Journal 1999, 138, 849–855. [Google Scholar] [CrossRef]
- Weinfeld, M.S.; Chertow, G.M.; Stevenson, L.W. Aggravated renal dysfunction during intensive therapy for advanced chronic heart failure. American Heart Journal 1999, 138, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Muslih, A.I. Reduction of mean arterial pressure and proteinuria by the effect of ACEIs (Lisinopril) in Kurdish hypertensive patients in Hawler City. Global journal of health science 2012, 4, 14–19. [Google Scholar] [CrossRef]
- Swartz, S.L. The role of prostaglandins in mediating the effects of angiotensin converting enzyme inhibitors and other antihypertensive drugs. Cardiovascular drugs and therapy 1987, 1, 39–43. [Google Scholar] [CrossRef]
- Banas, J.S., Jr. Effects of inhibitors of angiotensin-converting enzyme on regional hemodynamics. The American journal of cardiology 1992, 69, 40c–45c. [Google Scholar] [CrossRef]
- Lant, A.F. Evolution of diuretics and ACE inhibitors, their renal and antihypertensive actions--parallels and contrasts. British journal of clinical pharmacology 1987, 23 Suppl 1, 27s–41s. [Google Scholar] [CrossRef]
- Mandal, A.K.; Markert, R.J.; Saklayen, M.G.; Mankus, R.A.; Yokokawa, K. Diuretics potentiate angiotensin converting enzyme inhibitor-induced acute renal failure. Clinical nephrology 1994, 42, 170–174. [Google Scholar]
- Packer, M.; Lee, W.H.; Medina, N.; Yushak, M.; Kessler, P.D. Functional Renal Insufficiency During Long-Term Therapy with Captopril and Enalapril in Severe Chronic Heart Failure. Annals of Internal Medicine 1987, 106, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Wynckel, A.; Ebikili, B.; Melin, J.-P.; Randoux, C.; Lavaud, S.; Chanard, J. Long-term follow-up of acute renal failure caused by angiotensin converting enzyme inhibitors. American Journal of Hypertension 1998, 11, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Main, J. Atherosclerotic renal artery stenosis, ACE inhibitors, and avoiding cardiovascular death. Heart (British Cardiac Society) 2005, 91, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Schoolwerth, A.C.; Sica, D.A.; Ballermann, B.J.; Wilcox, C.S. Renal considerations in angiotensin converting enzyme inhibitor therapy: a statement for healthcare professionals from the Council on the Kidney in Cardiovascular Disease and the Council for High Blood Pressure Research of the American Heart Association. Circulation 2001, 104, 1985–1991. [Google Scholar] [CrossRef]
- Sica, D.A. Angiotensin-Converting Enzyme Inhibitors' Side Effects—Physiologic and Non-Physiologic Considerations. J Clin Hypertens (Greenwich) 2007, 7 (Suppl 8), 17–23. [Google Scholar] [CrossRef]
- Ponticelli, C.; Cucchiari, D. Renin-angiotensin system inhibitors in kidney transplantation: a benefit-risk assessment. Journal of Nephrology 2017, 30, 155–157. [Google Scholar] [CrossRef]
- Calvo Barbado, D.M.; Saiz Fernández, L.C.; Leache Alegría, L.; Celaya Lecea, M.C.; Gutiérrez-Valencia, M. Acute Kidney Injury associated with "Triple whammy" combination: a protocol for a systematic review. F1000Res 2022, 11, 496. [Google Scholar] [CrossRef]
- Leete, J.; Wang, C.; López-Hernández, F.J.; Layton, A.T. Determining risk factors for triple whammy acute kidney injury. Mathematical Biosciences 2022, 347, 108809. [Google Scholar] [CrossRef] [PubMed]
- Loboz, K.K.; Shenfield, G.M. Drug combinations and impaired renal function -- the 'triple whammy'. British journal of clinical pharmacology 2005, 59, 239–243. [Google Scholar] [CrossRef]
- Patil, V.P.; Salunke, B.G. Fluid Overload and Acute Kidney Injury. Indian journal of critical care medicine: peer-reviewed, official publication of Indian Society of Critical Care Medicine 2020, 24, S94–s97. [Google Scholar] [CrossRef] [PubMed]
- Busse, L.W.; Ostermann, M. Vasopressor Therapy and Blood Pressure Management in the Setting of Acute Kidney Injury. Seminars in nephrology 2019, 39, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Mushiyakh, Y.; Dangaria, H.; Qavi, S.; Ali, N.; Pannone, J.; Tompkins, D. Treatment and pathogenesis of acute hyperkalemia. Journal of community hospital internal medicine perspectives 2011, 1. [Google Scholar] [CrossRef]
- Bianchi, S.; Aucella, F.; De Nicola, L.; Genovesi, S.; Paoletti, E.; Regolisti, G. Management of hyperkalemia in patients with kidney disease: a position paper endorsed by the Italian Society of Nephrology. J Nephrol 2019, 32, 499–516. [Google Scholar] [CrossRef] [PubMed]
- Griffin, K.A.; Bidani, A.K. Potential risks of calcium channel blockers in chronic kidney disease. Curr Cardiol Rep 2008, 10, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Caiati, C.; Argentiero, A.; Favale, S.; Lepera, M.E. Cardiorenal Syndrome Triggered by Slowly Progressive Drugs Toxicity-Induced Renal Failure along with Minimal Mitral Disease: A Case Report. Endocrine, metabolic & immune disorders drug targets 2022, 22, 970–977. [Google Scholar] [CrossRef]
- Griffin, K.A.; Abu-Amarah, I.; Picken, M.; Bidani, A.K. Renoprotection by ACE Inhibition or Aldosterone Blockade Is Blood Pressure–Dependent. Hypertension 2003, 41, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Mishima, E.; Maruyama, K.; Nakazawa, T.; Abe, T.; Ito, S. Acute Kidney Injury from Excessive Potentiation of Calcium-channel Blocker via Synergistic CYP3A4 Inhibition by Clarithromycin Plus Voriconazole. Internal medicine (Tokyo, Japan) 2017, 56, 1687–1690. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; Fleet, J.L.; Bailey, D.G.; McArthur, E.; Wald, R.; Rehman, F.; Garg, A.X. Calcium-channel blocker-clarithromycin drug interactions and acute kidney injury. Jama 2013, 310, 2544–2553. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, R. Beta-blockers and renal function. Drugs 1982, 23, 195–206. [Google Scholar] [CrossRef]
- Zech, P.; Pozet, N.; Labeeuw, M.; Laville, M.; Hadj-Aissa, A.; Arkouche, W.; Poncet, J.F. Acute renal effects of beta-blockers. American journal of nephrology 1986, 6 Suppl 2, 15–19. [Google Scholar] [CrossRef]
- Sullivan, J.M.; Adams, D.F.; Hollenberg, N.K. beta-adrenergic blockade in essential hypertension: reduced renin release despite renal vasoconstriction. Circulation Research 1976, 39, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Epstein, M.; Oster, J.R. Beta blockers and renal function: a reappraisal. Journal of clinical hypertension 1985, 1, 85–99. [Google Scholar] [PubMed]
- Bakris, G.L.; Hart, P.; Ritz, E. Beta blockers in the management of chronic kidney disease. Kidney international 2006, 70, 1905–1913. [Google Scholar] [CrossRef]
- Kalaitzidis, R.; Bakris, G. Should nephrologists use beta-blockers? A perspective. Nephrology Dialysis Transplantation 2009, 24, 701–702. [Google Scholar] [CrossRef]
- Hall, M.E.; Rocco, M.V.; Morgan, T.M.; Hamilton, C.A.; Jordan, J.H.; Edwards, M.S.; Hall, J.E.; Hundley, W.G. Beta-Blocker Use Is Associated with Higher Renal Tissue Oxygenation in Hypertensive Patients Suspected of Renal Artery Stenosis. Cardiorenal medicine 2016, 6, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Borchard, U. Pharmacokinetics of beta-adrenoceptor blocking agents: clinical significance of hepatic and/or renal clearance. Clinical physiology and biochemistry 1990, 8 Suppl 2, 28–34. [Google Scholar]
- Capodanno, D.; Angiolillo, D.J. Antithrombotic Therapy in Patients With Chronic Kidney Disease. Circulation 2012, 125, 2649–2661. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.H.; Liou, H.H.; Huang, Y.C.; Lee, T.S.; Chen, M.; Fang, Y.W. Hazardous Effect of Low-Dose Aspirin in Patients with Predialysis Advanced Chronic Kidney Disease Assessed by Machine Learning Method Feature Selection. Healthcare (Basel, Switzerland) 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Arteriosclerosis, thrombosis, and vascular biology 2011, 31, 986–1000. [Google Scholar] [CrossRef]
- Crofford, L.J. COX-1 and COX-2 tissue expression: implications and predictions. The Journal of rheumatology. Supplement 1997, 49, 15–19. [Google Scholar]
- Shibata, K.; Akagi, Y.; Nozawa, N.; Shimomura, H.; Aoyama, T. Influence of nonsteroidal anti-inflammatory drugs on aspirin's antiplatelet effects and suggestion of the most suitable time for administration of both agents without resulting in interaction. Journal of pharmaceutical health care and sciences 2017, 3, 9. [Google Scholar] [CrossRef]
- Bjorkman, D.J. The effect of aspirin and nonsteroidal anti-inflammatory drugs on prostaglandins. The American journal of medicine 1998, 105, 8S–12S. [Google Scholar] [CrossRef]
- Packer, M. Interaction of prostaglandins and angiotensin II in the modulation of renal function in congestive heart failure. Circulation 1988, 77, I64–73. [Google Scholar] [PubMed]
- Kramer, H.J.; Stinnesbeck, B.; Klautke, G.; Kipnowski, J.; Klingmueller, D.; Glaenzer, K.; Duesing, R. Interaction of renal prostaglandins with the renin-angiotensin and renal adrenergic nervous systems in healthy subjects during dietary changes in sodium intake. Clinical science (London, England: 1979) 1985, 68, 387–393. [Google Scholar] [CrossRef]
- Kim, G.H. Renal effects of prostaglandins and cyclooxygenase-2 inhibitors. Electrolyte & blood pressure: E & BP 2008, 6, 35–41. [Google Scholar] [CrossRef]
- Culpepper, R.M.; Andreoli, T.E. Interactions among prostaglandin E2, antidiuretic hormone, and cyclic adenosine monophosphate in modulating Cl- absorption in single mouse medullary thick ascending limbs of Henle. The Journal of clinical investigation 1983, 71, 1588–1601. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, P.; Bhojani, K.; Joshi, V.R. NSAIDs and kidney. The Journal of the Association of Physicians of India 2004, 52, 632–640. [Google Scholar] [PubMed]
- Burukoglu, D.; Baycu, C.; Taplamacioglu, F.; Sahin, E.; Bektur, E. Effects of nonsteroidal anti-inflammatory meloxicam on stomach, kidney, and liver of rats. Toxicology and industrial health 2016, 32, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Drożdżal, S.; Lechowicz, K.; Szostak, B.; Rosik, J.; Kotfis, K.; Machoy-Mokrzyńska, A.; Białecka, M.; Ciechanowski, K.; Gawrońska-Szklarz, B. Kidney damage from nonsteroidal anti-inflammatory drugs-Myth or truth? Review of selected literature. Pharmacology research & perspectives 2021, 9, e00817. [Google Scholar] [CrossRef]
- Dreischulte, T.; Morales, D.R.; Bell, S.; Guthrie, B. Combined use of nonsteroidal anti-inflammatory drugs with diuretics and/or renin-angiotensin system inhibitors in the community increases the risk of acute kidney injury. Kidney international 2015, 88, 396–403. [Google Scholar] [CrossRef] [PubMed]
- McLeish, K.R.; Senitzer, D.; Gohara, A.F. Acute interstitial nephritis in a patient with aspirin hypersensitivity. Clinical immunology and immunopathology 1979, 14, 64–69. [Google Scholar] [CrossRef]
- Dixit, M.P.; Nguyen, C.; Carson, T.; Guedes, B.; Dixit, N.M.; Bell, J.M.; Wang, Y. Non-steroidal anti-inflammatory drugs-associated acute interstitial nephritis with granular tubular basement membrane deposits. Pediatric nephrology (Berlin, Germany) 2008, 23, 145–148. [Google Scholar] [CrossRef]
- Ravnskov, U. Glomerular, tubular and interstitial nephritis associated with non-steroidal antiinflammatory drugs. Evidence of a common mechanism. British journal of clinical pharmacology 1999, 47, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Harirforoosh, S.; Asghar, W.; Jamali, F. Adverse effects of nonsteroidal antiinflammatory drugs: an update of gastrointestinal, cardiovascular and renal complications. Journal of pharmacy & pharmaceutical sciences: a publication of the Canadian Society for Pharmaceutical Sciences, Societe canadienne des sciences pharmaceutiques 2013, 16, 821–847. [Google Scholar] [CrossRef]
- Leung, S.J.; Cisu, T.; Grob, B.M. Bilateral Ureteral Obstruction Secondary to Papillary Necrosis From Non-Steroidal Anti-Inflammatory Drug Use in an Adult Patient. Cureus 2021, 13, e16926. [Google Scholar] [CrossRef] [PubMed]
- Samuel, G.; Atanda, A.C.; Onyemeh, A.; Awan, A.; Ajiboye, O. A Unique Case of Drug Interaction between Ticagrelor and Statin Leading to Acute Renal Failure. Cureus 2017, 9, e1633. [Google Scholar] [CrossRef] [PubMed]
- Park, I.S.; Lee, S.B.; Song, S.H.; Seong, E.Y.; Kim, I.Y.; Rhee, H.; Kim, M.J.; Lee, D.W. Ticagrelor-induced acute kidney injury can increase serum concentration of statin and lead to concurrence of rhabdomyolysis. Anatolian journal of cardiology 2018, 19, 225–226. [Google Scholar] [CrossRef]
- Tada, K.; Ito, K.; Hamauchi, A.; Takahashi, K.; Watanabe, R.; Uchida, A.; Abe, Y.; Yasuno, T.; Miyake, K.; Sasatomi, Y.; et al. Clopidogrel-induced Thrombotic Microangiopathy in a Patient with Hypocomplementemia. Internal medicine (Tokyo, Japan) 2016, 55, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Etta, P.K.; Gowrishankar, S. Clopidogrel Induced Thrombotic Microangiopathy Successfully Treated with Conservative Approach. Indian journal of nephrology 2020, 30, 209–210. [Google Scholar] [CrossRef]
- Afiari, A.; Drekolias, D.; Jacob, J. Anticoagulant-Related Nephropathy: A Common, Under-Diagnosed Clinical Entity. Cureus 2022, 14, e22038. [Google Scholar] [CrossRef] [PubMed]
- Zakrocka, I.; Załuska, W. Anticoagulant-related nephropathy: Focus on novel agents. A review. Advances in clinical and experimental medicine: official organ Wroclaw Medical University 2022, 31, 165–173. [Google Scholar] [CrossRef]
- Brodsky, S.; Eikelboom, J.; Hebert, L.A. Anticoagulant-Related Nephropathy. Journal of the American Society of Nephrology: JASN 2018, 29, 2787–2793. [Google Scholar] [CrossRef]
- Oliver, T.; Salman, L.A.; Ciaudelli, B.; Cohen, D.A. Anticoagulation-Related Nephropathy: The Most Common Diagnosis You've Never Heard Of. The American journal of medicine 2019, 132, e631–e633. [Google Scholar] [CrossRef]
- Brodsky, S.V.; Satoskar, A.; Chen, J.; Nadasdy, G.; Eagen, J.W.; Hamirani, M.; Hebert, L.; Calomeni, E.; Nadasdy, T. Acute kidney injury during warfarin therapy associated with obstructive tubular red blood cell casts: a report of 9 cases. American journal of kidney diseases: the official journal of the National Kidney Foundation 2009, 54, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, S.V.; Mhaskar, N.S.; Thiruveedi, S.; Dhingra, R.; Reuben, S.C.; Calomeni, E.; Ivanov, I.; Satoskar, A.; Hemminger, J.; Nadasdy, G.; et al. Acute kidney injury aggravated by treatment initiation with apixaban: Another twist of anticoagulant-related nephropathy. Kidney research and clinical practice 2017, 36, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Mezue, K.; Ram, P.; Egbuche, O.; Menezes, R.G.; Lerma, E.; Rangaswami, J. Anticoagulation-related nephropathy for the internist: a concise review. American journal of cardiovascular disease 2020, 10, 301–305. [Google Scholar]
- Mach, F.; Ray, K.K.; Wiklund, O.; Corsini, A.; Catapano, A.L.; Bruckert, E.; De Backer, G.; Hegele, R.A.; Hovingh, G.K.; Jacobson, T.A.; et al. Adverse effects of statin therapy: perception vs. the evidence - focus on glucose homeostasis, cognitive, renal and hepatic function, haemorrhagic stroke and cataract. Eur Heart J 2018, 39, 2526–2539. [Google Scholar] [CrossRef]
- Ward, N.C.; Watts, G.F.; Eckel, R.H. Statin Toxicity. Circ Res 2019, 124, 328–350. [Google Scholar] [CrossRef]
- Wolfe, S.M. Dangers of rosuvastatin identified before and after FDA approval. The Lancet 2004, 363, 2189–2190. [Google Scholar] [CrossRef] [PubMed]
- van Zyl-Smit, R.; Firth, J.C.; Duffield, M.; Marais, A.D. Renal tubular toxicity of HMG-CoA reductase inhibitors. Nephrol Dial Transplant 2004, 19, 3176–3179. [Google Scholar] [CrossRef]
- Londrino, F.; Zattera, T.; Falqui, V.; Corbani, V.; Cavallini, M.; Stefanini, T.; Chiappini, N.; Ardini, M.; Martina, V.; Rombolà, G. Rosuvastatin-induced acute interstitial nephritis. Case Rep Nephrol Urol 2013, 3, 87–90. [Google Scholar] [CrossRef]
- Annigeri, R.A.; Mani, R.M. Acute interstitial nephritis due to statin and its class effect. Indian journal of nephrology 2015, 25, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Ezad, S.; Cheema, H.; Collins, N. Statin-induced rhabdomyolysis: a complication of a commonly overlooked drug interaction. Oxford medical case reports 2018, 2018, omx104. [Google Scholar] [CrossRef]
- Botshekan, S.; Yalameha, B. Are statins toxic or safe for kidney diseases? An updated mini-review study. Journal of Nephropathology 2020, 9, e38–e38. [Google Scholar] [CrossRef]
- Liu, A.; Wu, Q.; Guo, J.; Ares, I.; Rodríguez, J.-L.; Martínez-Larrañaga, M.-R.; Yuan, Z.; Anadón, A.; Wang, X.; Martínez, M.-A. Statins: Adverse reactions, oxidative stress and metabolic interactions. Pharmacology & Therapeutics 2019, 195, 54–84. [Google Scholar] [CrossRef]
- McMurray, J.J.; Dunselman, P.; Wedel, H.; Cleland, J.G.; Lindberg, M.; Hjalmarson, A.; Kjekshus, J.; Waagstein, F.; Apetrei, E.; Barrios, V.; et al. Coenzyme Q10, rosuvastatin, and clinical outcomes in heart failure: a pre-specified substudy of CORONA (controlled rosuvastatin multinational study in heart failure). J Am Coll Cardiol 2010, 56, 1196–1204. [Google Scholar] [CrossRef] [PubMed]
- Deichmann, R.; Lavie, C.; Andrews, S. Coenzyme q10 and statin-induced mitochondrial dysfunction. The Ochsner journal 2010, 10, 16–21. [Google Scholar]
- Liu, A.; Wu, Q.; Guo, J.; Ares, I.; Rodríguez, J.L.; Martínez-Larrañaga, M.R.; Yuan, Z.; Anadón, A.; Wang, X.; Martínez, M.A. Statins: Adverse reactions, oxidative stress and metabolic interactions. Pharmacol Ther 2019, 195, 54–84. [Google Scholar] [CrossRef]
- Sirvent, P.; Mercier, J.; Vassort, G.; Lacampagne, A. Simvastatin triggers mitochondria-induced Ca2+ signaling alteration in skeletal muscle. Biochemical and biophysical research communications 2005, 329, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, Y.; Ghorbanihaghjo, A.; Naghi-Zadeh, M.; Yagin, N.L. Oxidative stress as a possible mechanism of statin-induced myopathy. Inflammopharmacology 2018, 26, 667–674. [Google Scholar] [CrossRef]
- Verdoodt, A.; Honore, P.M.; Jacobs, R.; De Waele, E.; Van Gorp, V.; De Regt, J.; Spapen, H.D. Do Statins Induce or Protect from Acute Kidney Injury and Chronic Kidney Disease: An Update Review in 2018. J Transl Int Med 2018, 6, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Chitralli, D.; Raheja, R.; Br, K. Clinical Rhabdomyolysis With Acute Kidney Injury Secondary to High-Intensity Rosuvastatin Use: A Case Report. Cureus 2020, 12, e10932. [Google Scholar] [CrossRef] [PubMed]
- Morschel, C.F.; Mafra, D.; Eduardo, J.C.C. The relationship between proton pump inhibitors and renal disease. Jornal brasileiro de nefrologia 2018, 40, 301–306. [Google Scholar] [CrossRef]
- Moledina, D.G.; Perazella, M.A. PPIs and kidney disease: from AIN to CKD. J Nephrol 2016, 29, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Van Laecke, S.; Van Biesen, W.; Vanholder, R. Hypomagnesaemia, the kidney and the vessels. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 2012, 27, 4003–4010. [Google Scholar] [CrossRef] [PubMed]
- Malavade, P.; Hiremath, S. Proton Pump Inhibitors: More Indigestion than Relief? Indian journal of nephrology 2017, 27, 249–257. [Google Scholar] [CrossRef]
- Leonard, J.; Marshall, J.K.; Moayyedi, P. Systematic review of the risk of enteric infection in patients taking acid suppression. The American journal of gastroenterology 2007, 102, 2047–2056. [Google Scholar] [CrossRef] [PubMed]
- Arrich, J.; Sodeck, G.H.; Sengolge, G.; Konnaris, C.; Mullner, M.; Laggner, A.N.; Domanovits, H. Clostridium difficile causing acute renal failure: case presentation and review. World journal of gastroenterology 2005, 11, 1245–1247. [Google Scholar] [CrossRef]
- Fontecha-Barriuso, M.; Martín-Sanchez, D.; Martinez-Moreno, J.M.; Cardenas-Villacres, D.; Carrasco, S.; Sanchez-Niño, M.D.; Ruiz-Ortega, M.; Ortiz, A.; Sanz, A.B. Molecular pathways driving omeprazole nephrotoxicity. Redox biology 2020, 32, 101464. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Gong, X. Research Progress on the Potential Mechanisms of Acute Kidney Injury and Chronic Kidney Disease Induced by Proton Pump Inhibitors. Integrative Medicine in Nephrology and Andrology 2023, 10. [Google Scholar] [CrossRef]
- Caiati, C.; Scardapane, A.; Iacovelli, F.; Pollice, P.; Achille, T.I.; Favale, S.; Lepera, M.E. Coronary Flow and Reserve by Enhanced Transthoracic Doppler Trumps Coronary Anatomy by Computed Tomography in Assessing Coronary Artery Stenosis. Diagnostics (Basel) 2021, 11, 245. [Google Scholar] [CrossRef] [PubMed]
- McCullough, P.A.; Soman, S.S. Contrast-induced nephropathy. Crit Care Clin 2005, 21, 261–280. [Google Scholar] [CrossRef] [PubMed]
- Azzalini, L.; Spagnoli, V.; Ly, H.Q. Contrast-Induced Nephropathy: From Pathophysiology to Preventive Strategies. The Canadian journal of cardiology 2016, 32, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Morcos, S.K.; Dawson, P.; Pearson, J.D.; Jeremy, J.Y.; Davenport, A.P.; Yates, M.S.; Tirone, P.; Cipolla, P.; de Haën, C.; Muschick, P.; et al. The haemodynamic effects of iodinated water soluble radiographic contrast media: a review. European Journal of Radiology 1998, 29, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Tumlin, J.; Stacul, F.; Adam, A.; Becker, C.R.; Davidson, C.; Lameire, N.; McCullough, P.A. Pathophysiology of contrast-induced nephropathy. The American journal of cardiology 2006, 98, 14k–20k. [Google Scholar] [CrossRef]
- Persson, P.B.; Hansell, P.; Liss, P. Pathophysiology of contrast medium-induced nephropathy. Kidney international 2005, 68, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ren, K. The Mechanism of Contrast-Induced Acute Kidney Injury and Its Association with Diabetes Mellitus. Contrast media & molecular imaging 2020, 2020, 3295176. [Google Scholar] [CrossRef]
- Seeliger, E.; Sendeski, M.; Rihal, C.S.; Persson, P.B. Contrast-induced kidney injury: mechanisms, risk factors, and prevention. European heart journal 2012, 33, 2007–2015. [Google Scholar] [CrossRef] [PubMed]
- McCullough, P.A. Contrast-induced acute kidney injury. J Am Coll Cardiol 2008, 51, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Modi, K.; Padala, S.A.; Gupta, M. Contrast-Induced Nephropathy. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Sandeep Padala declares no relevant financial relationships with ineligible companies. Disclosure: Mohit Gupta declares no relevant financial relationships with ineligible companies.: StatPearls Publishing. Copyright © 2023, StatPearls Publishing LLC.; 2023.
- Iftikhar, H.; Saleem, M.; Kaji, A. Metformin-associated Severe Lactic Acidosis in the Setting of Acute Kidney Injury. Cureus 2019, 11, e3897. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative medicine and cellular longevity 2017, 2017, 8416763. [Google Scholar] [CrossRef]
- Deavall, D.G.; Martin, E.A.; Horner, J.M.; Roberts, R. Drug-induced oxidative stress and toxicity. Journal of toxicology 2012, 2012, 645460. [Google Scholar] [CrossRef] [PubMed]
- Tomsa, A.M.; Alexa, A.L.; Junie, M.L.; Rachisan, A.L.; Ciumarnean, L. Oxidative stress as a potential target in acute kidney injury. PeerJ 2019, 7, e8046. [Google Scholar] [CrossRef]
- Tirichen, H.; Yaigoub, H.; Xu, W.; Wu, C.; Li, R.; Li, Y. Mitochondrial Reactive Oxygen Species and Their Contribution in Chronic Kidney Disease Progression Through Oxidative Stress. Frontiers in physiology 2021, 12, 627837. [Google Scholar] [CrossRef] [PubMed]
- Che, R.; Yuan, Y.; Huang, S.; Zhang, A. Mitochondrial dysfunction in the pathophysiology of renal diseases. American journal of physiology. Renal physiology 2014, 306, F367–378. [Google Scholar] [CrossRef] [PubMed]
- Daenen, K.; Andries, A.; Mekahli, D.; Van Schepdael, A.; Jouret, F.; Bammens, B. Oxidative stress in chronic kidney disease. Pediatric nephrology (Berlin, Germany) 2019, 34, 975–991. [Google Scholar] [CrossRef]
- Ho, H.J.; Shirakawa, H. Oxidative Stress and Mitochondrial Dysfunction in Chronic Kidney Disease. Cells 2022, 12. [Google Scholar] [CrossRef]
- Podkowińska, A.; Formanowicz, D. Chronic Kidney Disease as Oxidative Stress- and Inflammatory-Mediated Cardiovascular Disease. Antioxidants (Basel, Switzerland) 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Moledina, D.G.; Parikh, C.R. Differentiating Acute Interstitial Nephritis from Acute Tubular Injury: A Challenge for Clinicians. Nephron 2019, 143, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Hosohata, K. Role of Oxidative Stress in Drug-Induced Kidney Injury. International journal of molecular sciences 2016, 17. [Google Scholar] [CrossRef]
- Arany, I.; Safirstein, R.L. Cisplatin nephrotoxicity. Seminars in nephrology 2003, 23, 460–464. [Google Scholar] [CrossRef]
- Santos, N.A.; Catão, C.S.; Martins, N.M.; Curti, C.; Bianchi, M.L.; Santos, A.C. Cisplatin-induced nephrotoxicity is associated with oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Archives of toxicology 2007, 81, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Novoa, J.M.; Quiros, Y.; Vicente, L.; Morales, A.I.; Lopez-Hernandez, F.J. New insights into the mechanism of aminoglycoside nephrotoxicity: an integrative point of view. Kidney international 2011, 79, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Kusirisin, P.; Chattipakorn, S.C.; Chattipakorn, N. Contrast-induced nephropathy and oxidative stress: mechanistic insights for better interventional approaches. Journal of translational medicine 2020, 18, 400. [Google Scholar] [CrossRef] [PubMed]
- Manda, P.; Srinivasa Rao, P.; Bitla, A.R.; Vinapamula, K.S.; Jeyaseelan, L.; Rajasekhar, D.; Vishnubhotla, S. Study of contrast-induced oxidative stress in nondiabetic patients undergoing coronary angiography. Saudi journal of kidney diseases and transplantation: an official publication of the Saudi Center for Organ Transplantation, Saudi Arabia 2019, 30, 45–52. [Google Scholar] [PubMed]
- Hizoh, I.; Haller, C. Radiocontrast-induced renal tubular cell apoptosis: hypertonic versus oxidative stress. Investigative radiology 2002, 37, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, M.M.; Bernardo, D.R.D.; de Bragança, A.C.; Massola Shimizu, M.H.; Seguro, A.C.; Volpini, R.A.; Canale, D. Treatment with β-blocker nebivolol ameliorates oxidative stress and endothelial dysfunction in tenofovir-induced nephrotoxicity in rats. Frontiers in medicine 2022, 9, 953749. [Google Scholar] [CrossRef] [PubMed]
- Gokturk, H.; Ulusu, N.N.; Gok, M.; Tuncay, E.; Can, B.; Turan, B. Long-term treatment with a beta-blocker timolol attenuates renal-damage in diabetic rats via enhancing kidney antioxidant-defense system. Molecular and cellular biochemistry 2014, 395, 177–186. [Google Scholar] [CrossRef]
- Coats, A.; Jain, S. Protective effects of nebivolol from oxidative stress to prevent hypertension-related target organ damage. Journal of Human Hypertension 2017, 31, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Murakami, M.; Miura, D.; Yunoki, K.; Enko, K.; Tanaka, M.; Saito, Y.; Nishii, N.; Miyoshi, T.; Yoshida, M.; et al. Beta-Blockers and Oxidative Stress in Patients with Heart Failure. Pharmaceuticals (Basel, Switzerland) 2011, 4, 1088–1100. [Google Scholar] [CrossRef] [PubMed]
- Caiati, C.; Stanca, A.; Lepera, M.E. Free Radicals and Obesity-Related Chronic Inflammation Contrasted by Antioxidants: A New Perspective in Coronary Artery Disease. 2023, 13, 712. [Google Scholar]
- Ayza, M.A.; Zewdie, K.A.; Yigzaw, E.F.; Ayele, S.G.; Tesfaye, B.A.; Tafere, G.G.; Abrha, M.G. Potential Protective Effects of Antioxidants against Cyclophosphamide-Induced Nephrotoxicity. International journal of nephrology 2022, 2022, 5096825. [Google Scholar] [CrossRef]
- Ranasinghe, R.; Mathai, M.; Zulli, A. Cytoprotective remedies for ameliorating nephrotoxicity induced by renal oxidative stress. Life Sciences 2023, 318, 121466. [Google Scholar] [CrossRef] [PubMed]
- Tylicki, L.; Rutkowski, B.; Hörl, W.H. Antioxidants: a possible role in kidney protection. Kidney & blood pressure research 2003, 26, 303–314. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




