Submitted:
12 August 2025
Posted:
18 August 2025
You are already at the latest version
Abstract
Regenerative dentistry has emerged as a critical paradigm shift in oral healthcare, moving beyond symptom management to true biological reconstruction. Effective oral tissue restoration demands a shift from conventional reparative treatments to regenerative strategies capable of rebuilding complex biological structures. This review synthesizes advancements in regenerative dentistry, focusing on the synergistic interplay between bioengineering and pharmacology. Central to these next-generation therapies are dental stem cells (e.g., DPSCs, PDLSCs), bioactive signaling molecules like growth factors (BMPs, FGF-2, PDGF), and autologous blood-derived biomaterials such as Platelet-Rich Plasma (PRP) and Platelet-Rich Fibrin (PRF). These approaches leverage advanced scaffolds and delivery systems to create biomimetic microenvironments that guide cellular differentiation, angiogenesis, and matrix synthesis for dentin-pulp, periodontal, and alveolar bone regeneration. Notably, PRF provides a sustained-release, three-dimensional fibrin scaffold enriched with leukocytes and growth factors, offering enhanced therapeutic longevity and immunomodulatory benefits over earlier concentrates. Emerging tools such as nanotechnology and 3D bioprinting further expand the potential of these therapies by enabling precise, patient-specific scaffold fabrication and controlled delivery of bioactive molecules. Despite their transformative potential, significant hurdles, including a lack of protocol standardization, high costs, regulatory complexities, and variability in clinical outcomes, impede widespread adoption. Future progress requires refining biomaterials through innovations like 3D bioprinting, establishing evidence-based clinical protocols through rigorous trials, and developing scalable, off-the-shelf solutions. By integrating these advancements into mainstream dentistry, clinicians can offer patients more predictable, biologically harmonious treatments that restore both function and aesthetics while reducing long-term complications.
Keywords:
1. Introduction
2. Growth Factors in Dental Regeneration
3. Platelet-Rich Plasma in Regenerative Dentistry
3.1. Periodontal Tissue Regeneration with PRP: Defect Geometry, PRP Phenotype, and Activation Chemistry
3.2. Alveolar Ridge Preservation and Bone Augmentation with PRP: Angiogenic Indices, Mineralization Kinetics, and Implant Readiness
3.3. Oral Surgery and Implantology with PRP: Mucosal Barrier Restoration, Nociceptive Modulation, and Early Stability
4. Platelet-Rich Fibrin in Dentistry
PRF-Guided Oral Tissue Regeneration: Fibrin Architecture, Release Kinetics, and Cross-Indication Outcomes
5. Stem Cell-Based Therapies in Dental Regeneration
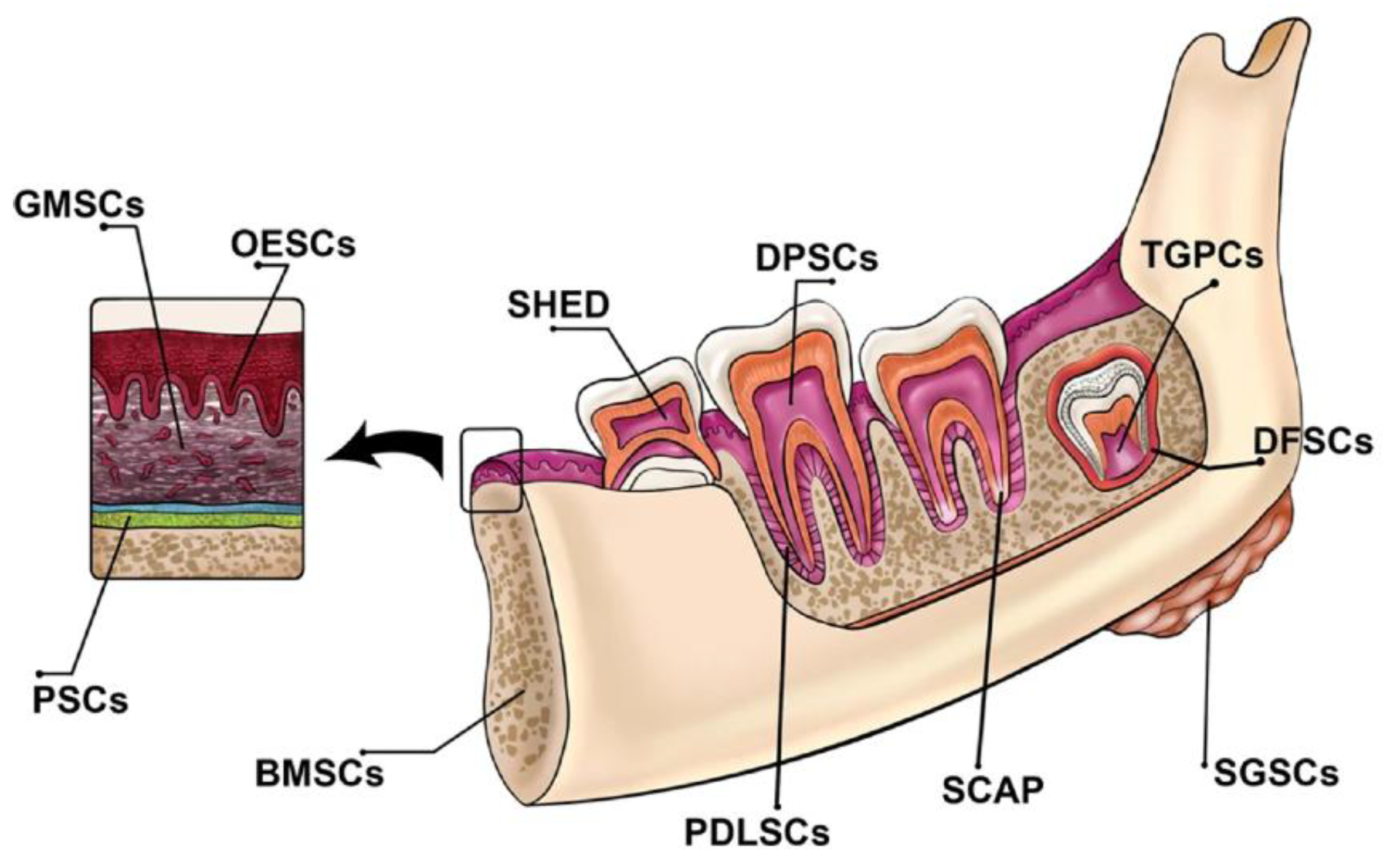
5.1. Type of Dental Stem Cell and Their Clinical Application in Dentistry
5.1.1. Dental Pulp Stem Cells (DPSCs)
5.1.2. Dental Follicle Progenitor Cells (DFPCs)
5.1.3. Stem Cells from Human Exfoliated Deciduous Teeth (SHEDs)
5.1.4. Stem Cells from Apical Papilla (SCAP)
5.1.5. Tooth Germ Progenitor Cells (TGPCs)
5.1.6. Periodontal Ligament Stem Cells (PDLSCs)
5.1.7. Gingival Mesenchymal Stem Cells (GMSCs)
5.1.8. Oral Mucosa-Derived Stem Cells (OMSCs)
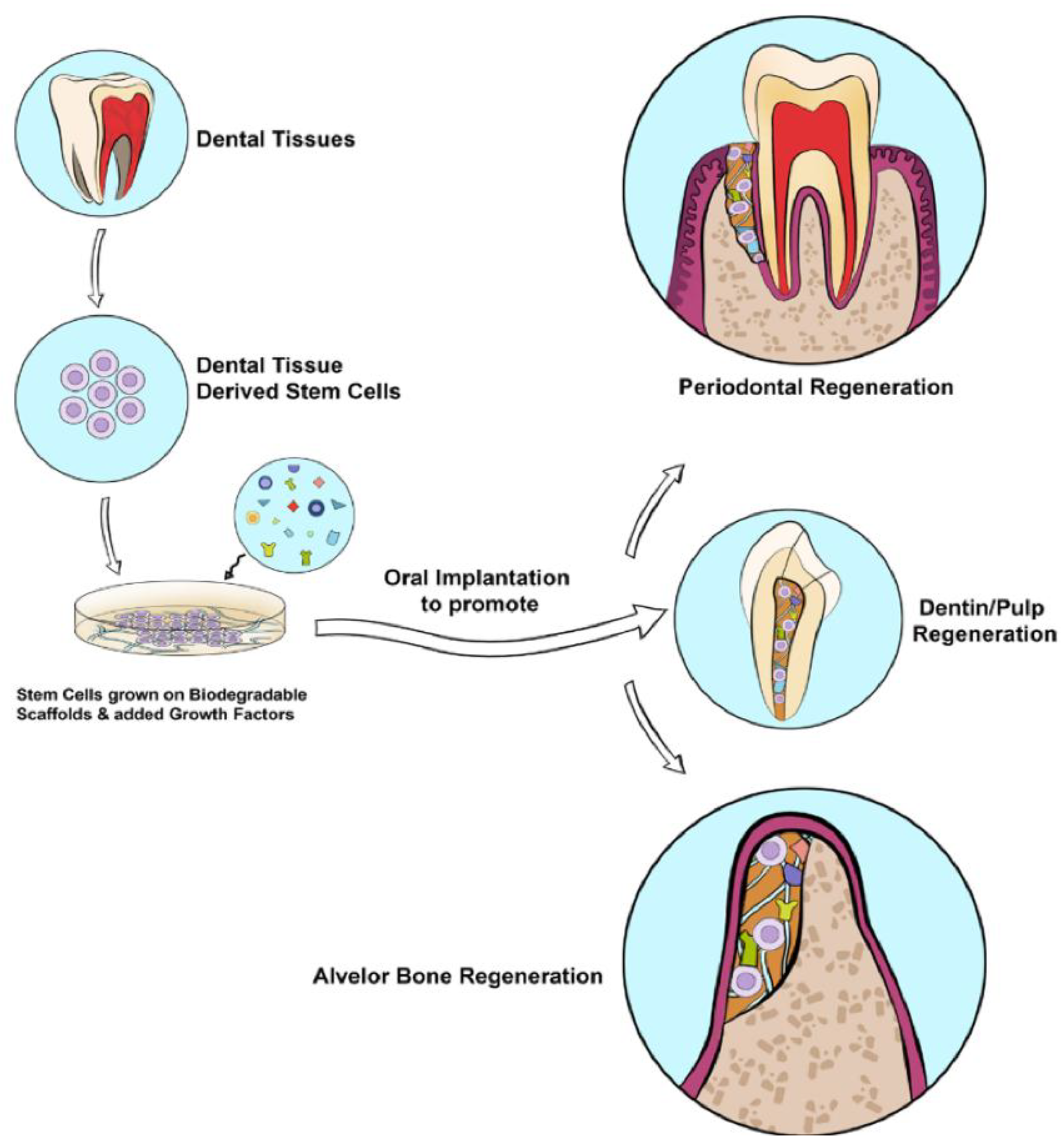
5.2. The potential of stem cell-based therapies for tissue regeneration in dentistry.
5.2.1. Regeneration of the Dentin-Pulp Complex
5.2.2. Periodontal and Alveolar Bone Regeneration
5.2.3. Nerve Regeneration in the Oral Cavity
6. Challenges and Future Directions
7. Conclusion
8. Unresolved Questions
Authors' contributions
Financial declaration
Ethics approval
Consent to participate
Consent for publication
Availability of Data and Material
Acknowledgments
Competing Interests
Abbreviations
| Abbreviation | Full Name |
| PRP | Platelet-Rich Plasma |
| PRF | Platelet-Rich Fibrin |
| BMPs | Bone Morphogenetic Proteins |
| FGF-2 | Fibroblast Growth Factor-2 |
| PDGF | Platelet-Derived Growth Factor |
| VEGF | Vascular Endothelial Growth Factor |
| TGF-β | Transforming Growth Factor-beta |
| MSCs | Mesenchymal Stem Cells |
| DPSCs | Dental Pulp Stem Cells |
| SCAP | Stem Cells from Apical Papilla |
| SHED | Stem Cells from Human Exfoliated Deciduous Teeth |
| PDLSCs | Periodontal Ligament Stem Cells |
| GMSCs | Gingival Mesenchymal Stem Cells |
| OMSCs | Oral Mucosa-Derived Stem Cells |
| NGF | Nerve Growth Factor |
| BDNF | Brain-Derived Neurotrophic Factor |
| G-CSF | Granulocyte Colony-Stimulating Factor |
| EMD | Enamel Matrix Derivative |
| BRONJ | Bisphosphonate-Related Osteonecrosis of the Jaw |
| MTA | Mineral Trioxide Aggregate |
References
- Morita, K.; Wang, J.; Okamoto, K.; Iwata, T. The next generation of regenerative dentistry: From tooth development biology to periodontal tissue, dental pulp, and whole tooth reconstruction in the clinical setting. Regen Ther 2025, 28, 333–344. [Google Scholar] [CrossRef]
- Alqahtani, A.M. Guided Tissue and Bone Regeneration Membranes: A Review of Biomaterials and Techniques for Periodontal Treatments. Polymers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Cho, Y.D.; Kim, K.H.; Lee, Y.M.; Ku, Y.; Seol, Y.J. Periodontal Wound Healing and Tissue Regeneration: A Narrative Review. Pharmaceuticals (Basel) 2021, 14. [Google Scholar] [CrossRef]
- Zhachko, N.I.; Nespriadko-Monborgne, T.S.; Skrypnyk, I.L.; Zhachko, M.S. IMPROVING DENTAL HEALTH - IS IMPROVING QUALITY OF LIFE. Wiad Lek 2021, 74, 722–725. [Google Scholar] [CrossRef]
- Tatullo, M.; Zavan, B.; Piattelli, A. Critical Overview on Regenerative Medicine: New Insights into the Role of Stem Cells and Innovative Biomaterials. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; El Moshy, S.; Radwan, I.A.; Rady, D.; El-Rashidy, A.A.; Abbass, M.M.S.; Dörfer, C.E. Stem Cells From Dental Pulp, Periodontal Tissues, and Other Oral Sources: Biological Concepts and Regenerative Potential. J Periodontal Res 2025. [Google Scholar] [CrossRef]
- Okpala, O.E.; Rondevaldova, J.; Kokoska, L. Anti-inflammatory drugs as potential antimicrobial agents: a review. Front Pharmacol 2025, 16, 1557333. [Google Scholar] [CrossRef]
- Hunsberger, J.G.; Pandya, P.; Mulligan, M.K.; Marotta, D.; Moroni, L.; Shusteff, M.; Brogan, G.; Brovold, M.; Yoo, J.; Koffler, J.; et al. Review of Disruptive Technologies in 3D Bioprinting. Current Stem Cell Reports 2025, 11, 5. [Google Scholar] [CrossRef]
- Alsuraifi, A.; Mouzan, M.M.; Ali, A.A.A.; Algzaare, A.; Aqeel, Z.; Ezzat, D.; Ayad, A. Revolutionizing Tooth Regeneration: Innovations from Stem Cells to Tissue Engineering. Regenerative Engineering and Translational Medicine 2025. [Google Scholar] [CrossRef]
- Ezzat, D.; Azab, A.; Kamel, I.S.; Abdelmonem, M.; Ibrahim, M.A.; Ayad, A.; Soomro, R.; Wagdy, M.; Eldebawy, M. Phytomedicine and green nanotechnology: enhancing glass ionomer cements for sustainable dental restorations: a comprehensive review. Beni-Suef University Journal of Basic and Applied Sciences 2025, 14, 48. [Google Scholar] [CrossRef]
- Ezzat, D.; Sheta, M.S.; Kenawy, E.-R.; Eid, M.A.; Elkafrawy, H. Synthesis, characterization and evaluation of experimental dental composite resin modified by grapefruit seed extract-mediated TiO₂ nanoparticles: green approach. Odontology 2025, 113, 1148–1164. [Google Scholar] [CrossRef]
- Alsuraifi, A.; Sulaiman, Z.M.; Mohammed, N.A.R.; Mohammed, J.; Ali, S.K.; Abdualihamaid, Y.H.; Husam, F.; Ayad, A. Explore the most recent developments and upcoming outlooks in the field of dental nanomaterials. Beni-Suef University Journal of Basic and Applied Sciences.
- Muskan; Gupta, D. ; Negi, N.P. 3D bioprinting: Printing the future and recent advances. Bioprinting 2022, 27, e00211. [Google Scholar] [CrossRef]
- Belugali Nataraj, N.; Yarden, Y. Growth Factors. 2021.
- Wei, X.; Yang, M.; Yue, L.; Huang, D.; Zhou, X.; Wang, X.; Zhang, Q.; Qiu, L.; Huang, Z.; Wang, H.; et al. Expert consensus on regenerative endodontic procedures. Int J Oral Sci 2022, 14, 55. [Google Scholar] [CrossRef]
- Yan, H.; De Deus, G.; Kristoffersen, I.M.; Wiig, E.; Reseland, J.E.; Johnsen, G.F.; Silva, E.J.N.L.; Haugen, H.J. Regenerative Endodontics by Cell Homing: A Review of Recent Clinical trials. Journal of Endodontics 2023, 49, 4–17. [Google Scholar] [CrossRef]
- Goldring, M.B.; Goldring, S.R. Cytokines and cell growth control. Crit Rev Eukaryot Gene Expr 1991, 1, 301–326. [Google Scholar]
- Cicciù, M. Growth Factor Applied to Oral and Regenerative Surgery. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Liu, M.; Goldman, G.; MacDougall, M.; Chen, S. BMP Signaling Pathway in Dentin Development and Diseases. Cells 2022, 11. [Google Scholar] [CrossRef]
- Lissenberg-Thunnissen, S.N.; de Gorter, D.J.; Sier, C.F.; Schipper, I.B. Use and efficacy of bone morphogenetic proteins in fracture healing. Int Orthop 2011, 35, 1271–1280. [Google Scholar] [CrossRef]
- Kurogoushi, R.; Hasegawa, T.; Akazawa, Y.; Iwata, K.; Sugimoto, A.; Yamaguchi-Ueda, K.; Miyazaki, A.; Narwidina, A.; Kawarabayashi, K.; Kitamura, T.; et al. Fibroblast growth factor 2 suppresses the expression of C-C motif chemokine 11 through the c-Jun N-terminal kinase pathway in human dental pulp-derived mesenchymal stem cells. Exp Ther Med 2021, 22, 1356. [Google Scholar] [CrossRef]
- Farooq, M.; Khan, A.W.; Kim, M.S.; Choi, S. The Role of Fibroblast Growth Factor (FGF) Signaling in Tissue Repair and Regeneration. Cells 2021, 10. [Google Scholar] [CrossRef]
- Novais, A.; Chatzopoulou, E.; Chaussain, C.; Gorin, C. The Potential of FGF-2 in Craniofacial Bone Tissue Engineering: A Review. Cells 2021, 10. [Google Scholar] [CrossRef]
- Shimabukuro, Y.; Ueda, M.; Ozasa, M.; Anzai, J.; Takedachi, M.; Yanagita, M.; Ito, M.; Hashikawa, T.; Yamada, S.; Murakami, S. Fibroblast growth factor-2 regulates the cell function of human dental pulp cells. J Endod 2009, 35, 1529–1535. [Google Scholar] [CrossRef]
- Andrae, J.; Gallini, R.; Betsholtz, C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev 2008, 22, 1276–1312. [Google Scholar] [CrossRef]
- Chiu, C.-H.; Chen, P.; Chen, A.C.-Y.; Chan, Y.-S.; Hsu, K.-Y.; Lei, K.F. Time-Dependent Cytokine-Release of Platelet-Rich Plasma in 3-Chamber Co-Culture Device and Conventional Culture Well. Applied Sciences 2021, 11, 6947. [Google Scholar] [CrossRef]
- Schär, M.O.; Diaz-Romero, J.; Kohl, S.; Zumstein, M.A.; Nesic, D. Platelet-rich concentrates differentially release growth factors and induce cell migration in vitro. Clin Orthop Relat Res 2015, 473, 1635–1643. [Google Scholar] [CrossRef]
- Roh, Y.H.; Kim, W.; Park, K.U.; Oh, J.H. Cytokine-release kinetics of platelet-rich plasma according to various activation protocols. Bone Joint Res 2016, 5, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Tai, H.; Tanabe, K.; Suzuki, H.; Sato, T.; Kawase, T.; Saito, Y.; Wolff, L.F.; Yoshiex, H. Platelet-rich plasma combined with a porous hydroxyapatite graft for the treatment of intrabony periodontal defects in humans: a comparative controlled clinical study. J Periodontol 2005, 76, 890–898. [Google Scholar] [CrossRef]
- Hou, X.; Yuan, J.; Aisaiti, A.; Liu, Y.; Zhao, J. The effect of platelet-rich plasma on clinical outcomes of the surgical treatment of periodontal intrabony defects: A systematic review and meta-analysis. BMC Oral Health 2016, 16, 71. [Google Scholar] [CrossRef]
- Ouyang, X.Y.; Qiao, J. Effect of platelet-rich plasma in the treatment of periodontal intrabony defects in humans. Chin Med J (Engl) 2006, 119, 1511–1521. [Google Scholar] [CrossRef]
- Döri, F.; Huszár, T.; Nikolidakis, D.; Arweiler, N.B.; Gera, I.; Sculean, A. Effect of platelet-rich plasma on the healing of intrabony defects treated with an anorganic bovine bone mineral and expanded polytetrafluoroethylene membranes. J Periodontol 2007, 78, 983–990. [Google Scholar] [CrossRef]
- Christgau, M.; Moder, D.; Wagner, J.; Glässl, M.; Hiller, K.A.; Wenzel, A.; Schmalz, G. Influence of autologous platelet concentrate on healing in intra-bony defects following guided tissue regeneration therapy: a randomized prospective clinical split-mouth study. J Clin Periodontol 2006, 33, 908–921. [Google Scholar] [CrossRef]
- Demir, B.; Sengün, D.; Berberoğlu, A. Clinical evaluation of platelet-rich plasma and bioactive glass in the treatment of intra-bony defects. J Clin Periodontol 2007, 34, 709–715. [Google Scholar] [CrossRef]
- Ozdemir, B.; Okte, E. Treatment of intrabony defects with beta-tricalciumphosphate alone and in combination with platelet-rich plasma. J Biomed Mater Res B Appl Biomater 2012, 100, 976–983. [Google Scholar] [CrossRef]
- Kaushick, B.T.; Jayakumar, N.D.; Padmalatha, O.; Varghese, S. Treatment of human periodontal infrabony defects with hydroxyapatite + β tricalcium phosphate bone graft alone and in combination with platelet rich plasma: a randomized clinical trial. Indian J Dent Res 2011, 22, 505–510. [Google Scholar] [CrossRef]
- Fernandes, G.; Yang, S. Application of platelet-rich plasma with stem cells in bone and periodontal tissue engineering. Bone Res 2016, 4, 16036. [Google Scholar] [CrossRef]
- Bacevich, B.M.; Smith, R.D.J.; Reihl, A.M.; Mazzocca, A.D.; Hutchinson, I.D. Advances with Platelet-Rich Plasma for Bone Healing. Biologics 2024, 18, 29–59. [Google Scholar] [CrossRef]
- Kargarpour, Z.; Nasirzade, J.; Panahipour, L.; Miron, R.J.; Gruber, R. Liquid PRF Reduces the Inflammatory Response and Osteoclastogenesis in Murine Macrophages. Front Immunol 2021, 12, 636427. [Google Scholar] [CrossRef]
- Nibali, L.; Sultan, D.; Arena, C.; Pelekos, G.; Lin, G.H.; Tonetti, M. Periodontal infrabony defects: Systematic review of healing by defect morphology following regenerative surgery. J Clin Periodontol 2021, 48, 100–113. [Google Scholar] [CrossRef]
- Pradeep, A.R.; Pai, S.; Garg, G.; Devi, P.; Shetty, S.K. A randomized clinical trial of autologous platelet-rich plasma in the treatment of mandibular degree II furcation defects. J Clin Periodontol 2009, 36, 581–588. [Google Scholar] [CrossRef]
- Varshney, S.; Dwivedi, A.; Dwivedi, V. Comparing efficacies of autologous platelet concentrate preparations as mono-therapeutic agents in intra-bony defects through systematic review and meta-analysis. J Oral Biol Craniofac Res 2023, 13, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Dohan Ehrenfest, D.M.; Andia, I.; Zumstein, M.A.; Zhang, C.Q.; Pinto, N.R.; Bielecki, T. Classification of platelet concentrates (Platelet-Rich Plasma-PRP, Platelet-Rich Fibrin-PRF) for topical and infiltrative use in orthopedic and sports medicine: current consensus, clinical implications and perspectives. Muscles Ligaments Tendons J 2014, 4, 3–9. [Google Scholar] [CrossRef]
- DeLong, J.M.; Russell, R.P.; Mazzocca, A.D. Platelet-rich plasma: the PAW classification system. Arthroscopy 2012, 28, 998–1009. [Google Scholar] [CrossRef]
- Song, D.; Shujaat, S.; Huang, Y.; Van Dessel, J.; Politis, C.; Lambrichts, I.; Jacobs, R. Effect of platelet-rich and platelet-poor plasma on 3D bone-to-implant contact: a preclinical micro-CT study. Int J Implant Dent 2021, 7, 11. [Google Scholar] [CrossRef]
- Caponio, V.C.A.; Baca-González, L.; González-Serrano, J.; Torres, J.; López-Pintor, R.M. Effect of the use of platelet concentrates on new bone formation in alveolar ridge preservation: a systematic review, meta-analysis, and trial sequential analysis. Clin Oral Investig 2023, 27, 4131–4146. [Google Scholar] [CrossRef] [PubMed]
- Ucer, C.; Khan, R.S. Extraction Socket Augmentation with Autologous Platelet-Rich Fibrin (PRF): The Rationale for Socket Augmentation. Dent J (Basel) 2023, 11. [Google Scholar] [CrossRef]
- Del Fabbro, M.; Taschieri, S.; Corbella, S. Efficacy of Different Materials for Maxillary Sinus Floor Augmentation With Lateral Approach. A Systematic Review. Clin Implant Dent Relat Res 2025, 27, e70053. [Google Scholar] [CrossRef]
- Malcangi, G.; Patano, A.; Palmieri, G.; Di Pede, C.; Latini, G.; Inchingolo, A.D.; Hazballa, D.; de Ruvo, E.; Garofoli, G.; Inchingolo, F.; et al. Maxillary Sinus Augmentation Using Autologous Platelet Concentrates (Platelet-Rich Plasma, Platelet-Rich Fibrin, and Concentrated Growth Factor) Combined with Bone Graft: A Systematic Review. Cells 2023, 12. [Google Scholar] [CrossRef]
- Albanese, A.; Licata, M.E.; Polizzi, B.; Campisi, G. Platelet-rich plasma (PRP) in dental and oral surgery: from the wound healing to bone regeneration. Immun Ageing 2013, 10, 23. [Google Scholar] [CrossRef]
- Qu, C.; Luo, F.; Hong, G.; Wan, Q. Effects of platelet concentrates on implant stability and marginal bone loss: a systematic review and meta-analysis. BMC Oral Health 2021, 21, 579. [Google Scholar] [CrossRef]
- Stähli, A.; Strauss, F.J.; Gruber, R. The use of platelet-rich plasma to enhance the outcomes of implant therapy: A systematic review. Clin Oral Implants Res. 2018, 29, 20–36. [Google Scholar] [CrossRef]
- Patel, H.; Pundkar, A.; Shrivastava, S.; Chandanwale, R.; Jaiswal, A.M. A Comprehensive Review on Platelet-Rich Plasma Activation: A Key Player in Accelerating Skin Wound Healing. Cureus 2023, 15, e48943. [Google Scholar] [CrossRef]
- Pineda-Cortel, M.R.B.; Suarez, C.G.; Cabrera, J.-T.; Daya, M.; Bonifacio, R.B.L.; Vergara, R.C.; et al. Complexity of Platelet-Rich Plasma: Mechanism of Action, Growth Factor Utilization and Variation in Preparation. Plasmatology 2024, 18. [Google Scholar] [CrossRef]
- Anitua, E.; Troya, M.; Alkhraisat, M.H. Immunoregulatory role of platelet derivatives in the macrophage-mediated immune response. Front Immunol 2024, 15, 1399130. [Google Scholar] [CrossRef]
- Everts, P.; Onishi, K.; Jayaram, P.; Lana, J.F.; Mautner, K. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Lim, L.; Bui, H.; Farrelly, O.; Yang, J.; Li, L.; Enis, D.; Ma, W.; Chen, M.; Oliver, G.; Welsh, J.D.; et al. Hemostasis stimulates lymphangiogenesis through release and activation of VEGFC. Blood 2019, 134, 1764–1775. [Google Scholar] [CrossRef]
- Liao, B.; Tian, Y.; Guan, M.; Han, W.; Yi, W.; Li, K.; Yang, X.; Niu, Y.; Zhang, B.; Teng, P.; et al. Exosomes derived from platelet-rich plasma alleviate synovial inflammation by enhancing synovial lymphatic function. Journal of Nanobiotechnology 2025, 23, 522. [Google Scholar] [CrossRef]
- Jiang, J.; Cong, X.; Alageel, S.; Dornseifer, U.; Schilling, A.F.; Hadjipanayi, E.; et al. In Vitro Comparison of Lymphangiogenic Potential of Hypoxia Preconditioned Serum (HPS) and Platelet-Rich Plasma (PRP). International Journal of Molecular Sciences 2023, 24, 1961. [Google Scholar] [CrossRef]
- Dutta, S.R.; Passi, D.; Singh, P.; Sharma, S.; Singh, M.; Srivastava, D. A randomized comparative prospective study of platelet-rich plasma, platelet-rich fibrin, and hydroxyapatite as a graft material for mandibular third molar extraction socket healing. Natl J Maxillofac Surg 2016, 7, 45–51. [Google Scholar] [CrossRef]
- Albanese, A.; Licata, M.E.; Polizzi, B.; Campisi, G. Platelet-rich plasma (PRP) in dental and oral surgery: from the wound healing to bone regeneration. Immunity & Ageing 2013, 10, 23. [Google Scholar] [CrossRef]
- Hanif, M.; Sheikh, M.A. Efficacy of platelet rich plasma (PRP) on mouth opening and pain after surgical extraction of mandibular third molars. Journal of Oral Medicine and Oral Surgery 2020, 7, 9. [Google Scholar] [CrossRef]
- Samani, M.K.; Saberi, B.V.; Ali Tabatabaei, S.M.; Moghadam, M.G. The clinical evaluation of platelet-rich plasma on free gingival graft's donor site wound healing. Eur J Dent 2017, 11, 447–454. [Google Scholar] [CrossRef]
- Barone, S.; Zaffino, P.; Salviati, M.; Destito, M.; Antonelli, A.; Bennardo, F.; Cevidanes, L.; Spadea, M.F.; Giudice, A. Automated pipeline for linear and volumetric assessment of facial swelling after third molar surgery. BMC Oral Health 2024, 24, 1404. [Google Scholar] [CrossRef]
- Shah, S.A.; Singh, B.P.; Rao, J.; Kumar, L.; Singh, M.; Singh, P.K. Biological and esthetic outcome of immediate dental implant with the adjunct pretreatment of immediate implants with platelet-rich plasma or photofunctionalization: A randomized controlled trial. J Indian Prosthodont Soc 2021, 21, 348–355. [Google Scholar] [CrossRef]
- Attia, S.; Narberhaus, C.; Schaaf, H.; Streckbein, P.; Pons-Kühnemann, J.; Schmitt, C.; Neukam, F.W.; Howaldt, H.P.; Böttger, S. Long-Term Influence of Platelet-Rich Plasma (PRP) on Dental Implants after Maxillary Augmentation: Retrospective Clinical and Radiological Outcomes of a Randomized Controlled Clinical Trial. J Clin Med 2020, 9. [Google Scholar] [CrossRef]
- Barone, S.; Bennardo, F.; Salviati, M.; Antonelli, A.; Giudice, A. Evaluation of the usefulness of platelet-rich fibrin (PRF) in mandibular third molar surgery with 3D facial swelling analysis: a split-mouth randomized clinical trial. Head & Face Medicine 2025, 21, 8. [Google Scholar]
- Bai, M.Y.; Wang, C.W.; Wang, J.Y.; Lin, M.F.; Chan, W.P. Three-dimensional structure and cytokine distribution of platelet-rich fibrin. Clinics (Sao Paulo) 2017, 72, 116–124. [Google Scholar] [CrossRef]
- Wang, X.; Fok, M.R.; Pelekos, G.; Jin, L.; Tonetti, M.S. In Vitro and Ex Vivo Kinetic Release Profile of Growth Factors and Cytokines from Leucocyte- and Platelet-Rich Fibrin (L-PRF) Preparations. Cells 2022, 11. [Google Scholar] [CrossRef]
- Ghanaati, S.; Booms, P.; Orlowska, A.; Kubesch, A.; Lorenz, J.; Rutkowski, J.; Landes, C.; Sader, R.; Kirkpatrick, C.; Choukroun, J. Advanced platelet-rich fibrin: a new concept for cell-based tissue engineering by means of inflammatory cells. J Oral Implantol 2014, 40, 679–689. [Google Scholar] [CrossRef]
- Dohan Ehrenfest, D.M.; Bielecki, T.; Jimbo, R.; Barbé, G.; Del Corso, M.; Inchingolo, F.; Sammartino, G. Do the fibrin architecture and leukocyte content influence the growth factor release of platelet concentrates? An evidence-based answer comparing a pure platelet-rich plasma (P-PRP) gel and a leukocyte- and platelet-rich fibrin (L-PRF). Curr Pharm Biotechnol 2012, 13, 1145–1152. [Google Scholar] [CrossRef]
- Jia, K.; You, J.; Zhu, Y.; Li, M.; Chen, S.; Ren, S.; et al. Platelet-rich fibrin as an autologous biomaterial for bone regeneration: mechanisms, applications, optimization. Front Bioeng Biotechnol. 2024, 12, 1286035. [Google Scholar] [CrossRef]
- Miron, R.J.; Fujioka-Kobayashi, M.; Sculean, A.; Zhang, Y. Optimization of platelet-rich fibrin. Periodontol 2000 2024, 94, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Bains, V.K.; Mahendra, J.; Mittal, M.; Bedi, M.; Mahendra, L. Technical considerations in obtaining platelet rich fibrin for clinical and periodontal research. J Oral Biol Craniofac Res 2023, 13, 714–719. [Google Scholar] [CrossRef]
- Masuki, H.; Isobe, K.; Kawabata, H.; Tsujino, T.; Yamaguchi, S.; Watanabe, T.; Sato, A.; Aizawa, H.; Mourão, C.F.; Kawase, T. Acute cytotoxic effects of silica microparticles used for coating of plastic blood-collection tubes on human periosteal cells. Odontology 2020, 108, 545–552. [Google Scholar] [CrossRef]
- Wu, Q.; Yu, S.; Wang, Y.; Zhang, X. Effect of thermal manipulation on the biological and mechanical characteristics of horizontal platelet rich fibrin membranes. BMC Oral Health 2023, 23, 956. [Google Scholar] [CrossRef]
- Castro, A.B.; Andrade, C.; Li, X.; Pinto, N.; Teughels, W.; Quirynen, M. Impact of g force and timing on the characteristics of platelet-rich fibrin matrices. Scientific Reports 2021, 11, 6038. [Google Scholar] [CrossRef]
- Simões-Pedro, M.; Tróia, P.; Dos Santos, N.B.M.; Completo, A.M.G.; Castilho, R.M.; de Oliveira Fernandes, G.V. Tensile Strength Essay Comparing Three Different Platelet-Rich Fibrin Membranes (L-PRF, A-PRF, and A-PRF+): A Mechanical and Structural In Vitro Evaluation. Polymers (Basel) 2022, 14. [Google Scholar] [CrossRef]
- Silva, F.; Chauca-Bajaña, L.; Caponio, V.C.A.; Cueva, K.A.S.; Velasquez-Ron, B.; Padín-Iruegas, M.E.; Almeida, L.L.; Lorenzo-Pouso, A.I.; Suárez-Peñaranda, J.M.; Pérez-Sayáns, M. Regeneration of periodontal intrabony defects using platelet-rich fibrin (PRF): a systematic review and network meta-analysis. Odontology 2024, 112, 1047–1068. [Google Scholar] [CrossRef]
- Yavuz, A.; Güngörmek, H.S.; Kuru, L.; Doğan, B. Treatment of multiple adjacent gingival recessions using leucocyte- and platelet-rich fibrin with coronally advanced flap: a 12-month split-mouth controlled randomized clinical trial. Clin Oral Investig 2024, 28, 291. [Google Scholar] [CrossRef]
- Saleh, M.H.A.; Sabri, H.; Di Pietro, N.; Comuzzi, L.; Geurs, N.C.; Bou Semaan, L.; Piattelli, A. Clinical Indications and Outcomes of Sinus Floor Augmentation With Bone Substitutes: An Evidence-Based Review. Clin Implant Dent Relat Res 2025, 27, e13400. [Google Scholar] [CrossRef]
- Saleh, W.; Abdelhaleem, M.; Elmeadawy, S. Assessing the effectiveness of advanced platelet rich fibrin in treating gingival recession: a systematic review and meta-analysis. BMC Oral Health 2024, 24, 1400. [Google Scholar] [CrossRef]
- Ghanaati, S.; Śmieszek-Wilczewska, J.; Al-Maawi, S.; Neff, P.; Zadeh, H.H.; Sader, R.; et al. Solid PRF Serves as Basis for Guided Open Wound Healing of the Ridge after Tooth Extraction by Accelerating the Wound Healing Time Course-A Prospective Parallel Arm Randomized Controlled Single Blind Trial. Bioengineering (Basel) 2022, 9. [Google Scholar] [CrossRef]
- Aldommari, E.A.; Omair, A.; Qasem, T. Titanium-prepared platelet-rich fibrin enhances alveolar ridge preservation: a randomized controlled clinical and radiographic study. Scientific Reports 2025, 15, 24065. [Google Scholar] [CrossRef]
- Babich, O.; Lugassy, E.; Babich, M.; Abayov, P.; Haimov, E.; Juodzbalys, G. The Use of Platelet-Rich Fibrin in Sinus Floor Augmentation Surgery: a Systematic Review. J Oral Maxillofac Res 2024, 15, e1. [Google Scholar] [CrossRef]
- Sabeti, M.; Ghobrial, D.; Zanjir, M.; da Costa, B.R.; Young, Y.; Azarpazhooh, A. Treatment outcomes of regenerative endodontic therapy in immature permanent teeth with pulpal necrosis: A systematic review and network meta-analysis. Int Endod J 2024, 57, 238–255. [Google Scholar] [CrossRef]
- Niemczyk, W.; Janik, K.; Niemczyk, S.; Żurek, J.; Lynch, E.; Parker, S.; Cronshaw, M.; Skaba, D.; Wiench, R. Use of platelet-rich plasma (PRP) and injectable platelet-rich fibrin (i-PRF) in oral lichen planus treatment: a systematic review of randomized controlled trials. BMC Oral Health 2025, 25, 832. [Google Scholar] [CrossRef]
- Silva, F.F.V.e.; Chauca-Bajaña, L.; Caponio, V.C.A.; Cueva, K.A.S.; Velasquez-Ron, B.; Padín-Iruegas, M.E.; Almeida, L.L.; Lorenzo-Pouso, A.I.; Suárez-Peñaranda, J.M.; Pérez-Sayáns, M. Regeneration of periodontal intrabony defects using platelet-rich fibrin (PRF): a systematic review and network meta-analysis. Odontology 2024, 112, 1047–1068. [Google Scholar] [CrossRef]
- Kolios, G.; Moodley, Y. Introduction to stem cells and regenerative medicine. Respiration 2013, 85, 3–10. [Google Scholar] [CrossRef]
- Yu, L.; Zeng, L.; Zhang, Z.; Zhu, G.; Xu, Z.; Xia, J.; Weng, J.; Li, J.; Pathak, J.L. Cannabidiol Rescues TNF-α-Inhibited Proliferation, Migration, and Osteogenic/Odontogenic Differentiation of Dental Pulp Stem Cells. Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Soudi, A.; Yazdanian, M.; Ranjbar, R.; Tebyanian, H.; Yazdanian, A.; Tahmasebi, E.; Keshvad, A.; Seifalian, A. Role and application of stem cells in dental regeneration: A comprehensive overview. Excli j 2021, 20, 454–489. [Google Scholar] [CrossRef]
- Azab, A.; Alam-Eldein, A.; Aboutaleb, F. Qualitative and Radiographic Assessment of PEEK Bar Versus Titanium Bar Fabricated by CAD-CAM in Mandibular Hybrid Prosthesis: a randomized controlled clinical trial. Ain Shams Dental Journal 2024, 35, 334–343. [Google Scholar] [CrossRef]
- Azab, A.; El-Sheikh, A.; Abd-Allah, S. Stress analysis of short implants with different diameters in maxillary bilateral distal extension bases. Tanta Dental Journal 2020, 17, 73. [Google Scholar] [CrossRef]
- Haworth, S.; Shungin, D.; Kwak, S.Y.; Kim, H.Y.; West, N.X.; Thomas, S.J.; Franks, P.W.; Timpson, N.J.; Shin, M.J.; Johansson, I. Tooth loss is a complex measure of oral disease: Determinants and methodological considerations. Community Dent Oral Epidemiol 2018, 46, 555–562. [Google Scholar] [CrossRef]
- Marian, D.; Toro, G.; D'Amico, G.; Trotta, M.C.; D'Amico, M.; Petre, A.; Lile, I.; Hermenean, A.; Fratila, A. Challenges and Innovations in Alveolar Bone Regeneration: A Narrative Review on Materials, Techniques, Clinical Outcomes, and Future Directions. Medicina (Kaunas) 2024, 61. [Google Scholar] [CrossRef]
- Mosaddad, S.A.; Rasoolzade, B.; Namanloo, R.A.; Azarpira, N.; Dortaj, H. Stem cells and common biomaterials in dentistry: a review study. J Mater Sci Mater Med 2022, 33, 55. [Google Scholar] [CrossRef]
- Ohlsson, E.; Galler, K.; Widbiller, M. A Compilation of Study Models for Dental Pulp Regeneration. International Journal of Molecular Sciences 2022, 23, 14361. [Google Scholar] [CrossRef]
- Gomez-Sosa, J.F.; Diaz-Solano, D.; Wittig, O.; Cardier, J.E. Dental Pulp Regeneration Induced by Allogenic Mesenchymal Stromal Cell Transplantation in a Mature Tooth: A Case Report. J Endod 2022, 48, 736–740. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, Y.; Zhang, Y.; Zhao, Z.; Xue, T.; Wang, J.; Li, M.; Zhao, S.; Zhang, H.; Ding, Y. 3D bioprinting advanced biomaterials for craniofacial and dental tissue engineering – A review. Materials & Design 2024, 241, 112886. [Google Scholar] [CrossRef]
- Esdaille, C.J.; Washington, K.S.; Laurencin, C.T. Regenerative engineering: a review of recent advances and future directions. Regen Med 2021, 16, 495–512. [Google Scholar] [CrossRef]
| Device | Spins and speeds reported | Activator | Platelet count reporting | Notes for reproducibility | Ref |
| Heraeus Labofuge 300 | 2400 rpm 10 min plus 3600 rpm 16 min | Sodium alginate 0.1 g | Baseline 257 ± 46 ×10³ μL reported in prior study | Transparent on device and spin, activation unusual for periodontology | [29] |
| SmartPReP | 2400 rpm 10 min plus 3600 rpm 15 min | 10 percent CaCl₂ plus 1000 U topical thrombin | Not recorded | Classic buffy-coat kit plus exogenous activation | [30] |
| Universal 16R | 1220 rpm 15 min plus 3600 rpm 15 min | 10 percent CaCl₂ plus 100 U mL bovine thrombin | Baseline 189 ± 37, post 680 ± 103 ×10³ μL | Reports pre and post counts for transparency | [31] |
| Curasan PRP kit | 1220 rpm 15 min plus 3600 rpm 15 min | 10 percent CaCl₂ plus 100 U mL thrombin | Post 2519.6 ± 834.3 ×10³ μL in referenced prior | High post counts with CaCl₂ plus thrombin | [32] |
| Spectra cell separator | Not recorded | 0.5 mL of 10 percent CaCl₂ | Baseline 273 ± 56, post 2134 ± 782 ×10³ μL | Early RCT with cell separator and CaCl₂ activation | [33] |
| Heraeus Christ Medifuge | 3000 rpm 10 min plus 3600 rpm 10 min | 0.3 mL of 0.025 M CaCl₂ mixed with surgical blood | Baseline 189 ± 37, post 680 ± 103 ×10³ μL, as reported by Hou 2016 | Details on CaCl₂ concentration included. Counts are identical to Ouyang 2006 in Hou’s table and are attributed here exactly as reported by the secondary source | [34] |
| Curasan PRP kit | 2400 rpm 10 min plus 3600 rpm 15 min | Not recorded | Baseline 290 ± 86, post 1075 ± 636 ×10³ μL | Spins specified, activation not reported | [35] |
| Not reported | 5000 rpm 10 min plus 2000 rpm 10 min | 10 percent CaCl₂ in saline | Baseline 200, post 1250 ×10³ μL | Unusual first spin speed, still reports counts | [36] |
| Morphology variable or contrast | Quantitative association with outcome | Outcome type | Notes for translation | Ref |
| More residual walls increase odds of radiographic bone gain | OR 3.43, 95% CI 1.09 to 10.85 | Radiographic bone gain at 12 months | greater containment may improve scaffold retention and early signal retention | [40] |
| More residual walls increase odds of CAL gain | OR 1.42, 95% CI 1.14 to 1.77 | CAL gain at 12 months | Associations occurred irrespective of biomaterials used as reported by the review | |
| Three-wall vs one-wall defects | −1.18 mm difference favoring 3-wall for bone gain | Radiographic bone gain | From one study within the review’s synthesis | |
| Initial depth as continuous predictor | OR 1.32, 95% CI 1.19 to 1.47, deeper defects associated with more bone gain | Radiographic bone gain at 12 months | Continuous-model meta-analysis reported by the review | |
| Initial depth ≥ 4 mm vs < 4 mm | 0.75 mm more bone gain for deeper defects | Radiographic bone gain at 12 months | Threshold at 4 mm per the categorical meta-analysis |
| Approach | Key Components | Target Tissue | Status | Challenges |
| Stem Cell Therapy | DPSCs, PDLSCs | Pulp, bone, PDL | Preclinical/clinical | Standardization |
| Growth Factor Delivery | BMPs, FGF, VEGF | Pulp, bone | Approved (some) | Short half-life |
| Scaffold-Based Regeneration | Collagen, Chitosan | Pulp, alveolar bone | In trials | Integration, vascularization |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




