Submitted:
01 January 2025
Posted:
02 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanisms of Cancer Cell Plasticity
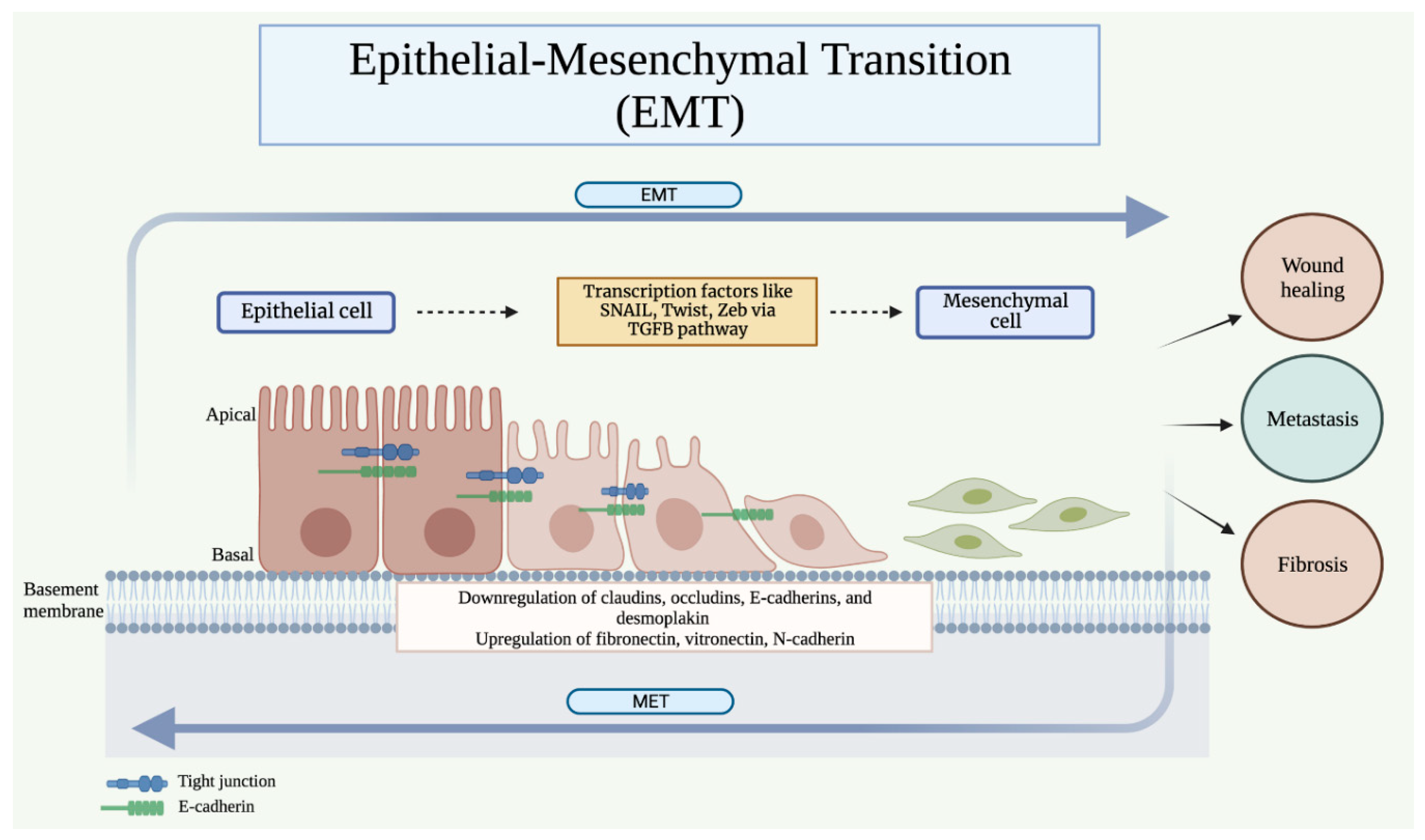
2.1. Epithelial-Mesenchymal Transition (EMT)
2.2. Dedifferentiation and Transdifferentiation
2.3. Epigenetics of Cancer Cell Plasticity
2.3.1. Epigenetic Regulation of Genes Associated with EMT
3. Cell Plasticity and Therapy Resistance
3.1. Reduced Susceptibility to Apoptosis
3.2. Immune Evasion
3.3. Epigenetic Targets
3.4. Efflux of Drugs
3.5. Alteration of Drug Targets
4. Targeting Cancer Plasticity for Therapy
4.1. Therapy Targeting the Phenomenon of EMT
4.2. Therapies Targeting Transdifferentiation
4.3. Therapies Targeting Differentiation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Grasmann, G., Mondal, A., & Leithner, K. (2021). Flexibility and Adaptation of Cancer Cells in a Heterogenous Metabolic Microenvironment. Int J Mol Sci, 22(3). [CrossRef]
- Kreuzaler, P., Panina, Y., Segal, J., & Yuneva, M. (2020). Adapt and conquer: Metabolic flexibility in cancer growth, invasion and evasion. Mol Metab, 33, 83-101. [CrossRef]
- Alsayed, R., Sheikhan, K., Alam, M. A., Buddenkotte, J., Steinhoff, M., Uddin, S., et al. (2023). Epigenetic programing of cancer stemness by transcription factors-non-coding RNAs interactions. Semin Cancer Biol, 92, 74-83. [CrossRef]
- Wagers, A. J., & Weissman, I. L. (2004). Plasticity of adult stem cells. Cell, 116(5), 639-648. [CrossRef]
- Patil, K., Khan, F. B., Akhtar, S., Ahmad, A., & Uddin, S. (2021). The plasticity of pancreatic cancer stem cells: implications in therapeutic resistance. Cancer Metastasis Rev, 40(3), 691-720. [CrossRef]
- Phi, L. T. H., Sari, I. N., Yang, Y. G., Lee, S. H., Jun, N., Kim, K. S., et al. (2018). Cancer Stem Cells (CSCs) in Drug Resistance and their Therapeutic Implications in Cancer Treatment. Stem Cells Int, 2018, 5416923. [CrossRef]
- Ahmad, A. (2013). Pathways to breast cancer recurrence. ISRN Oncol, 2013, 290568. [CrossRef]
- Marzagalli, M., Fontana, F., Raimondi, M., & Limonta, P. (2021). Cancer Stem Cells-Key Players in Tumor Relapse. Cancers (Basel), 13(3). [CrossRef]
- Li, Y., Rogoff, H. A., Keates, S., Gao, Y., Murikipudi, S., Mikule, K., et al. (2015). Suppression of cancer relapse and metastasis by inhibiting cancer stemness. Proc Natl Acad Sci U S A, 112(6), 1839-1844. [CrossRef]
- Ali, A. S., Ahmad, A., Ali, S., Bao, B., Philip, P. A., & Sarkar, F. H. (2013). The role of cancer stem cells and miRNAs in defining the complexities of brain metastasis. J Cell Physiol, 228(1), 36-42. [CrossRef]
- Steinbichler, T. B., Savic, D., Dudas, J., Kvitsaridze, I., Skvortsov, S., Riechelmann, H., et al. (2020). Cancer stem cells and their unique role in metastatic spread. Semin Cancer Biol, 60, 148-156. [CrossRef]
- Eisenberg, L. M., & Eisenberg, C. A. (2003). Stem cell plasticity, cell fusion, and transdifferentiation. Birth Defects Res C Embryo Today, 69(3), 209-218. [CrossRef]
- Filip, S., Mokry, J., English, D., & Vojacek, J. (2005). Stem cell plasticity and issues of stem cell therapy. Folia Biol (Praha), 51(6), 180-187.
- Orlic, D., Kajstura, J., Chimenti, S., Limana, F., Jakoniuk, I., Quaini, F., et al. (2001). Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proc Natl Acad Sci U S A, 98(18), 10344-10349. [CrossRef]
- Oh, S. H., Witek, R. P., Bae, S. H., Zheng, D., Jung, Y., Piscaglia, A. C., et al. (2007). Bone marrow-derived hepatic oval cells differentiate into hepatocytes in 2-acetylaminofluorene/partial hepatectomy-induced liver regeneration. Gastroenterology, 132(3), 1077-1087. [CrossRef]
- Marjanovic, N. D., Weinberg, R. A., & Chaffer, C. L. (2013). Cell plasticity and heterogeneity in cancer. Clin Chem, 59(1), 168-179. [CrossRef]
- Thankamony, A. P., Saxena, K., Murali, R., Jolly, M. K., & Nair, R. (2020). Cancer Stem Cell Plasticity - A Deadly Deal. Front Mol Biosci, 7, 79. [CrossRef]
- van der Schaft, D. W., Hillen, F., Pauwels, P., Kirschmann, D. A., Castermans, K., Egbrink, M. G., et al. (2005). Tumor cell plasticity in Ewing sarcoma, an alternative circulatory system stimulated by hypoxia. Cancer Res, 65(24), 11520-11528. [CrossRef]
- Liu, S., Cong, Y., Wang, D., Sun, Y., Deng, L., Liu, Y., et al. (2014). Breast cancer stem cells transition between epithelial and mesenchymal states reflective of their normal counterparts. Stem Cell Reports, 2(1), 78-91. [CrossRef]
- Qin, S., Jiang, J., Lu, Y., Nice, E. C., Huang, C., Zhang, J., et al. (2020). Emerging role of tumor cell plasticity in modifying therapeutic response. Signal Transduct Target Ther, 5(1), 228. [CrossRef]
- Shi, Z. D., Pang, K., Wu, Z. X., Dong, Y., Hao, L., Qin, J. X., et al. (2023). Tumor cell plasticity in targeted therapy-induced resistance: mechanisms and new strategies. Signal Transduct Target Ther, 8(1), 113. [CrossRef]
- Zhang, L., & Goodrich, D. W. (2022). RB1, Cancer Lineage Plasticity, and Therapeutic Resistance. Annual Review of Cancer Biology, 6(1), 201-221. [CrossRef]
- Kobayashi, S., Yamada-Okabe, H., Suzuki, M., Natori, O., Kato, A., Matsubara, K., et al. (2012). LGR5-positive colon cancer stem cells interconvert with drug-resistant LGR5-negative cells and are capable of tumor reconstitution. Stem Cells, 30(12), 2631-2644. [CrossRef]
- Wang, L., Saci, A., Szabo, P. M., Chasalow, S. D., Castillo-Martin, M., Domingo-Domenech, J., et al. (2018). EMT- and stroma-related gene expression and resistance to PD-1 blockade in urothelial cancer. Nat Commun, 9(1), 3503. [CrossRef]
- Shen, S., & Clairambault, J. (2020). Cell plasticity in cancer cell populations. F1000Res, 9. [CrossRef]
- Brabletz, S., Schuhwerk, H., Brabletz, T., & Stemmler, M. P. (2021). Dynamic EMT: a multi-tool for tumor progression. EMBO J, 40(18), e108647. [CrossRef]
- Xiong, S., Feng, Y., & Cheng, L. (2019). Cellular Reprogramming as a Therapeutic Target in Cancer. Trends Cell Biol, 29(8), 623-634. [CrossRef]
- Ahmad, A., Sarkar, S. H., Bitar, B., Ali, S., Aboukameel, A., Sethi, S., et al. (2012). Garcinol regulates EMT and Wnt signaling pathways in vitro and in vivo, leading to anticancer activity against breast cancer cells. Mol Cancer Ther, 11(10), 2193-2201. [CrossRef]
- Nieto, M. A., Huang, R. Y., Jackson, R. A., & Thiery, J. P. (2016). Emt: 2016. Cell, 166(1), 21-45. [CrossRef]
- Pei, D., Shu, X., Gassama-Diagne, A., & Thiery, J. P. (2019). Mesenchymal-epithelial transition in development and reprogramming. Nat Cell Biol, 21(1), 44-53. [CrossRef]
- Kalluri, R., & Weinberg, R. A. (2009). The basics of epithelial-mesenchymal transition. J Clin Invest, 119(6), 1420-1428. [CrossRef]
- Huang, Y., Hong, W., & Wei, X. (2022). The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J Hematol Oncol, 15(1), 129. [CrossRef]
- Pearson, G. W. (2019). Control of Invasion by Epithelial-to-Mesenchymal Transition Programs during Metastasis. J Clin Med, 8(5). [CrossRef]
- Lamouille, S., Xu, J., & Derynck, R. (2014). Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol, 15(3), 178-196. [CrossRef]
- Huang, R. Y., Guilford, P., & Thiery, J. P. (2012). Early events in cell adhesion and polarity during epithelial-mesenchymal transition. J Cell Sci, 125(Pt 19), 4417-4422. [CrossRef]
- Peinado, H., Olmeda, D., & Cano, A. (2007). Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? Nat Rev Cancer, 7(6), 415-428. [CrossRef]
- Ahmad, A., Maitah, M. Y., Ginnebaugh, K. R., Li, Y., Bao, B., Gadgeel, S. M., et al. (2013). Inhibition of Hedgehog signaling sensitizes NSCLC cells to standard therapies through modulation of EMT-regulating miRNAs. J Hematol Oncol, 6(1), 77. [CrossRef]
- Kalluri, R. (2009). EMT: when epithelial cells decide to become mesenchymal-like cells. J Clin Invest, 119(6), 1417-1419. [CrossRef]
- Marconi, G. D., Fonticoli, L., Rajan, T. S., Pierdomenico, S. D., Trubiani, O., Pizzicannella, J., et al. (2021). Epithelial-Mesenchymal Transition (EMT): The Type-2 EMT in Wound Healing, Tissue Regeneration and Organ Fibrosis. Cells, 10(7). [CrossRef]
- Mittal, V. (2018). Epithelial Mesenchymal Transition in Tumor Metastasis. Annu Rev Pathol, 13, 395-412. [CrossRef]
- Beerling, E., Seinstra, D., de Wit, E., Kester, L., van der Velden, D., Maynard, C., et al. (2016). Plasticity between Epithelial and Mesenchymal States Unlinks EMT from Metastasis-Enhancing Stem Cell Capacity. Cell Rep, 14(10), 2281-2288. [CrossRef]
- Pastushenko, I., Mauri, F., Song, Y., de Cock, F., Meeusen, B., Swedlund, B., et al. (2021). Fat1 deletion promotes hybrid EMT state, tumour stemness and metastasis. Nature, 589(7842), 448-455. [CrossRef]
- Huang, M. S., Fu, L. H., Yan, H. C., Cheng, L. Y., Ru, H. M., Mo, S., et al. (2022). Proteomics and liquid biopsy characterization of human EMT-related metastasis in colorectal cancer. Front Oncol, 12, 790096. [CrossRef]
- Fantozzi, A., Gruber, D. C., Pisarsky, L., Heck, C., Kunita, A., Yilmaz, M., et al. (2014). VEGF-mediated angiogenesis links EMT-induced cancer stemness to tumor initiation. Cancer Res, 74(5), 1566-1575. [CrossRef]
- Yu, M., Bardia, A., Wittner, B. S., Stott, S. L., Smas, M. E., Ting, D. T., et al. (2013). Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science, 339(6119), 580-584. [CrossRef]
- Mani, S. A., Guo, W., Liao, M. J., Eaton, E. N., Ayyanan, A., Zhou, A. Y., et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 133(4), 704-715. [CrossRef]
- Yang, J., Mani, S. A., Donaher, J. L., Ramaswamy, S., Itzykson, R. A., Come, C., et al. (2004). Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell, 117(7), 927-939. [CrossRef]
- Zubair, H., & Ahmad, A. (2017). Chapter 1 - Cancer Metastasis: An Introduction. In A. Ahmad (Ed.), Introduction to Cancer Metastasis (pp. 3-12): Academic Press.
- Kasimir-Bauer, S., Hoffmann, O., Wallwiener, D., Kimmig, R., & Fehm, T. (2012). Expression of stem cell and epithelial-mesenchymal transition markers in primary breast cancer patients with circulating tumor cells. Breast Cancer Res, 14(1), R15. [CrossRef]
- Mego, M., Mani, S. A., Lee, B. N., Li, C., Evans, K. W., Cohen, E. N., et al. (2012). Expression of epithelial-mesenchymal transition-inducing transcription factors in primary breast cancer: The effect of neoadjuvant therapy. Int J Cancer, 130(4), 808-816. [CrossRef]
- Khoo, B. L., Lee, S. C., Kumar, P., Tan, T. Z., Warkiani, M. E., Ow, S. G., et al. (2015). Short-term expansion of breast circulating cancer cells predicts response to anti-cancer therapy. Oncotarget, 6(17), 15578-15593. [CrossRef]
- Barriere, G., Fici, P., Gallerani, G., Fabbri, F., Zoli, W., & Rigaud, M. (2014). Circulating tumor cells and epithelial, mesenchymal and stemness markers: characterization of cell subpopulations. Ann Transl Med, 2(11), 109. [CrossRef]
- Shibue, T., Brooks, M. W., Inan, M. F., Reinhardt, F., & Weinberg, R. A. (2012). The outgrowth of micrometastases is enabled by the formation of filopodium-like protrusions. Cancer Discov, 2(8), 706-721. [CrossRef]
- Malta, T. M., Sokolov, A., Gentles, A. J., Burzykowski, T., Poisson, L., Weinstein, J. N., et al. (2018). Machine Learning Identifies Stemness Features Associated with Oncogenic Dedifferentiation. Cell, 173(2), 338-354 e315. [CrossRef]
- Moon, J. H., Kwon, S., Jun, E. K., Kim, A., Whang, K. Y., Kim, H., et al. (2011). Nanog-induced dedifferentiation of p53-deficient mouse astrocytes into brain cancer stem-like cells. Biochem Biophys Res Commun, 412(1), 175-181. [CrossRef]
- Nakano, M., Kikushige, Y., Miyawaki, K., Kunisaki, Y., Mizuno, S., Takenaka, K., et al. (2019). Dedifferentiation process driven by TGF-beta signaling enhances stem cell properties in human colorectal cancer. Oncogene, 38(6), 780-793. [CrossRef]
- Flavahan, W. A., Gaskell, E., & Bernstein, B. E. (2017). Epigenetic plasticity and the hallmarks of cancer. Science, 357(6348). [CrossRef]
- Nishiyama, A., & Nakanishi, M. (2021). Navigating the DNA methylation landscape of cancer. Trends Genet, 37(11), 1012-1027. [CrossRef]
- Ku, S. C., Liu, H. L., Su, C. Y., Yeh, I. J., Yen, M. C., Anuraga, G., et al. (2022). Comprehensive analysis of prognostic significance of cadherin (CDH) gene family in breast cancer. Aging (Albany NY), 14(20), 8498-8567. [CrossRef]
- Tur, M. K., Daramola, A. K., Gattenlohner, S., Herling, M., Chetty, S., & Barth, S. (2017). Restoration of DAP Kinase Tumor Suppressor Function: A Therapeutic Strategy to Selectively Induce Apoptosis in Cancer Cells Using Immunokinase Fusion Proteins. Biomedicines, 5(4). [CrossRef]
- Li, C., Xiong, W., Liu, X., Xiao, W., Guo, Y., Tan, J., et al. (2019). Hypomethylation at non-CpG/CpG sites in the promoter of HIF-1alpha gene combined with enhanced H3K9Ac modification contribute to maintain higher HIF-1alpha expression in breast cancer. Oncogenesis, 8(4), 26. [CrossRef]
- Stone, A., Cowley, M. J., Valdes-Mora, F., McCloy, R. A., Sergio, C. M., Gallego-Ortega, D., et al. (2013). BCL-2 hypermethylation is a potential biomarker of sensitivity to antimitotic chemotherapy in endocrine-resistant breast cancer. Mol Cancer Ther, 12(9), 1874-1885. [CrossRef]
- Flavahan, W. A., Drier, Y., Liau, B. B., Gillespie, S. M., Venteicher, A. S., Stemmer-Rachamimov, A. O., et al. (2016). Insulator dysfunction and oncogene activation in IDH mutant gliomas. Nature, 529(7584), 110-114. [CrossRef]
- Pradhan, N., Parbin, S., Kar, S., Das, L., Kirtana, R., Suma Seshadri, G., et al. (2019). Epigenetic silencing of genes enhanced by collective role of reactive oxygen species and MAPK signaling downstream ERK/Snail axis: Ectopic application of hydrogen peroxide repress CDH1 gene by enhanced DNA methyltransferase activity in human breast cancer. Biochim Biophys Acta Mol Basis Dis, 1865(6), 1651-1665. [CrossRef]
- Hu, Y., Zheng, Y., Dai, M., Wang, X., Wu, J., Yu, B., et al. (2019). G9a and histone deacetylases are crucial for Snail2-mediated E-cadherin repression and metastasis in hepatocellular carcinoma. Cancer Sci, 110(11), 3442-3452. [CrossRef]
- Romero, S., Musleh, M., Bustamante, M., Stambuk, J., Pisano, R., Lanzarini, E., et al. (2018). Polymorphisms in TWIST1 and ZEB1 Are Associated with Prognosis of Gastric Cancer Patients. Anticancer Res, 38(7), 3871-3877. [CrossRef]
- Paco, A., Leitao-Castro, J., & Freitas, R. (2021). Epigenetic Regulation of CDH1 Is Altered after HOXB7-Silencing in MDA-MB-468 Triple-Negative Breast Cancer Cells. Genes (Basel), 12(10). [CrossRef]
- Sengupta, D., Deb, M., Rath, S. K., Kar, S., Parbin, S., Pradhan, N., et al. (2016). DNA methylation and not H3K4 trimethylation dictates the expression status of miR-152 gene which inhibits migration of breast cancer cells via DNMT1/CDH1 loop. Exp Cell Res, 346(2), 176-187. [CrossRef]
- Sengupta, D., Deb, M., & Patra, S. K. (2018). Antagonistic activities of miR-148a and DNMT1: Ectopic expression of miR-148a impairs DNMT1 mRNA and dwindle cell proliferation and survival. Gene, 660, 68-79. [CrossRef]
- Fang, X. Q., Lee, M., Lim, W. J., Lee, S., Lim, C. H., & Lim, J. H. (2022). PGC1alpha Cooperates with FOXA1 to Regulate Epithelial Mesenchymal Transition through the TCF4-TWIST1. Int J Mol Sci, 23(15). [CrossRef]
- Baylin, S. B. (2012). The cancer epigenome: its origins, contributions to tumorigenesis, and translational implications. Proc Am Thorac Soc, 9(2), 64-65. [CrossRef]
- Kar, S., Niharika, Roy, A., & Patra, S. K. (2023). Overexpression of SOX2 Gene by Histone Modifications: SOX2 Enhances Human Prostate and Breast Cancer Progression by Prevention of Apoptosis and Enhancing Cell Proliferation. Oncology, 101(9), 591-608. [CrossRef]
- Niharika, Roy, A., Mishra, J., Chakraborty, S., Singh, S. P., & Patra, S. K. (2023). Epigenetic regulation of pluripotency inducer genes NANOG and SOX2 in human prostate cancer. Prog Mol Biol Transl Sci, 197, 241-260. [CrossRef]
- Wang, X., Jin, J., Wan, F., Zhao, L., Chu, H., Chen, C., et al. (2019). AMPK Promotes SPOP-Mediated NANOG Degradation to Regulate Prostate Cancer Cell Stemness. Dev Cell, 48(3), 345-360 e347. [CrossRef]
- De Angelis, M. L., Francescangeli, F., La Torre, F., & Zeuner, A. (2019). Stem Cell Plasticity and Dormancy in the Development of Cancer Therapy Resistance. Front Oncol, 9, 626. [CrossRef]
- Wiley, C. D., & Campisi, J. (2021). The metabolic roots of senescence: mechanisms and opportunities for intervention. Nat Metab, 3(10), 1290-1301. [CrossRef]
- Adhikari, S., Bhattacharya, A., Adhikary, S., Singh, V., Gadad, S. S., Roy, S., et al. (2022). The paradigm of drug resistance in cancer: an epigenetic perspective. Biosci Rep, 42(4). [CrossRef]
- Toh, T. B., Lim, J. J., & Chow, E. K. (2017). Epigenetics in cancer stem cells. Mol Cancer, 16(1), 29. [CrossRef]
- Ahmad, A., Li, Y., Bao, B., Kong, D., & Sarkar, F. H. (2014). Epigenetic regulation of miRNA-cancer stem cells nexus by nutraceuticals. Mol Nutr Food Res, 58(1), 79-86. [CrossRef]
- Choi, M., Kipps, T., & Kurzrock, R. (2016). ATM Mutations in Cancer: Therapeutic Implications. Mol Cancer Ther, 15(8), 1781-1791. [CrossRef]
- Vasiliou, V., Vasiliou, K., & Nebert, D. W. (2009). Human ATP-binding cassette (ABC) transporter family. Hum Genomics, 3(3), 281-290. [CrossRef]
- Moitra, K. (2015). Overcoming Multidrug Resistance in Cancer Stem Cells. Biomed Res Int, 2015, 635745. [CrossRef]
- Honoki, K., Fujii, H., Kubo, A., Kido, A., Mori, T., Tanaka, Y., et al. (2010). Possible involvement of stem-like populations with elevated ALDH1 in sarcomas for chemotherapeutic drug resistance. Oncol Rep, 24(2), 501-505. [CrossRef]
- Peitzsch, C., Cojoc, M., Hein, L., Kurth, I., Mabert, K., Trautmann, F., et al. (2016). An Epigenetic Reprogramming Strategy to Resensitize Radioresistant Prostate Cancer Cells. Cancer Res, 76(9), 2637-2651. [CrossRef]
- Fernald, K., & Kurokawa, M. (2013). Evading apoptosis in cancer. Trends Cell Biol, 23(12), 620-633. [CrossRef]
- Taniai, M., Grambihler, A., Higuchi, H., Werneburg, N., Bronk, S. F., Farrugia, D. J., et al. (2004). Mcl-1 mediates tumor necrosis factor-related apoptosis-inducing ligand resistance in human cholangiocarcinoma cells. Cancer Res, 64(10), 3517-3524. [CrossRef]
- Wu, D. W., Lee, M. C., Hsu, N. Y., Wu, T. C., Wu, J. Y., Wang, Y. C., et al. (2015). FHIT loss confers cisplatin resistance in lung cancer via the AKT/NF-kappaB/Slug-mediated PUMA reduction. Oncogene, 34(29), 3882-3883. [CrossRef]
- Lu, M., Marsters, S., Ye, X., Luis, E., Gonzalez, L., & Ashkenazi, A. (2014). E-cadherin couples death receptors to the cytoskeleton to regulate apoptosis. Mol Cell, 54(6), 987-998. [CrossRef]
- Rhodes, A., & Hillen, T. (2016). Mathematical Modeling of the Role of Survivin on Dedifferentiation and Radioresistance in Cancer. Bull Math Biol, 78(6), 1162-1188. [CrossRef]
- Chakravarti, A., Zhai, G. G., Zhang, M., Malhotra, R., Latham, D. E., Delaney, M. A., et al. (2004). Survivin enhances radiation resistance in primary human glioblastoma cells via caspase-independent mechanisms. Oncogene, 23(45), 7494-7506. [CrossRef]
- Chen, X., Duan, N., Zhang, C., & Zhang, W. (2016). Survivin and Tumorigenesis: Molecular Mechanisms and Therapeutic Strategies. J Cancer, 7(3), 314-323. [CrossRef]
- Ahmad, A., Sakr, W. A., & Rahman, K. M. (2012). Novel targets for detection of cancer and their modulation by chemopreventive natural compounds. Front Biosci (Elite Ed), 4(1), 410-425. [CrossRef]
- Rahman, K. M., Banerjee, S., Ali, S., Ahmad, A., Wang, Z., Kong, D., et al. (2009). 3,3'-Diindolylmethane enhances taxotere-induced apoptosis in hormone-refractory prostate cancer cells through survivin down-regulation. Cancer Res, 69(10), 4468-4475. [CrossRef]
- Chen, L., Gibbons, D. L., Goswami, S., Cortez, M. A., Ahn, Y. H., Byers, L. A., et al. (2014). Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression. Nat Commun, 5, 5241. [CrossRef]
- Vathiotis, I. A., Gomatou, G., Stravopodis, D. J., & Syrigos, N. (2021). Programmed Death-Ligand 1 as a Regulator of Tumor Progression and Metastasis. Int J Mol Sci, 22(10). [CrossRef]
- Shibue, T., & Weinberg, R. A. (2017). EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat Rev Clin Oncol, 14(10), 611-629. [CrossRef]
- Mehta, A., Kim, Y. J., Robert, L., Tsoi, J., Comin-Anduix, B., Berent-Maoz, B., et al. (2018). Immunotherapy Resistance by Inflammation-Induced Dedifferentiation. Cancer Discov, 8(8), 935-943. [CrossRef]
- Fetsch, P. A., Marincola, F. M., Filie, A., Hijazi, Y. M., Kleiner, D. E., & Abati, A. (1999). Melanoma-associated antigen recognized by T cells (MART-1): the advent of a preferred immunocytochemical antibody for the diagnosis of metastatic malignant melanoma with fine-needle aspiration. Cancer, 87(1), 37-42.
- Bakker, A. B., Schreurs, M. W., de Boer, A. J., Kawakami, Y., Rosenberg, S. A., Adema, G. J., et al. (1994). Melanocyte lineage-specific antigen gp100 is recognized by melanoma-derived tumor-infiltrating lymphocytes. J Exp Med, 179(3), 1005-1009. [CrossRef]
- Boiko, A. D., Razorenova, O. V., van de Rijn, M., Swetter, S. M., Johnson, D. L., Ly, D. P., et al. (2010). Human melanoma-initiating cells express neural crest nerve growth factor receptor CD271. Nature, 466(7302), 133-137. [CrossRef]
- Liu, J., Xiao, Q., Xiao, J., Niu, C., Li, Y., Zhang, X., et al. (2022). Wnt/beta-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct Target Ther, 7(1), 3. [CrossRef]
- Kahn, M. (2014). Can we safely target the WNT pathway? Nat Rev Drug Discov, 13(7), 513-532. [CrossRef]
- Katoh, M., & Katoh, M. (2022). WNT signaling and cancer stemness. Essays Biochem, 66(4), 319-331. [CrossRef]
- Teeuwssen, M., & Fodde, R. (2019). Wnt Signaling in Ovarian Cancer Stemness, EMT, and Therapy Resistance. J Clin Med, 8(10). [CrossRef]
- Schmitt, C. A., Fridman, J. S., Yang, M., Lee, S., Baranov, E., Hoffman, R. M., et al. (2002). A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell, 109(3), 335-346. [CrossRef]
- Zhan, T., Ambrosi, G., Wandmacher, A. M., Rauscher, B., Betge, J., Rindtorff, N., et al. (2019). MEK inhibitors activate Wnt signalling and induce stem cell plasticity in colorectal cancer. Nat Commun, 10(1), 2197. [CrossRef]
- Jimeno, A., Gordon, M., Chugh, R., Messersmith, W., Mendelson, D., Dupont, J., et al. (2017). A First-in-Human Phase I Study of the Anticancer Stem Cell Agent Ipafricept (OMP-54F28), a Decoy Receptor for Wnt Ligands, in Patients with Advanced Solid Tumors. Clin Cancer Res, 23(24), 7490-7497. [CrossRef]
- Feng, Y., Liu, X., & Pauklin, S. (2021). 3D chromatin architecture and epigenetic regulation in cancer stem cells. Protein Cell, 12(6), 440-454. [CrossRef]
- Mohan, D. R., Borges, K. S., Finco, I., LaPensee, C. R., Rege, J., Little, D. W., et al. (2023). Abstract 1501: Epigenetic dedifferentiation as a therapeutic strategy in adrenal cancer. Cancer Research, 83(7_Supplement), 1501-1501. [CrossRef]
- Yamada, Y., Haga, H., & Yamada, Y. (2014). Concise review: dedifferentiation meets cancer development: proof of concept for epigenetic cancer. Stem Cells Transl Med, 3(10), 1182-1187. [CrossRef]
- Scaffidi, P., & Misteli, T. (2010). Cancer epigenetics: from disruption of differentiation programs to the emergence of cancer stem cells. Cold Spring Harb Symp Quant Biol, 75, 251-258. [CrossRef]
- Del Vecchio, C. A., Feng, Y., Sokol, E. S., Tillman, E. J., Sanduja, S., Reinhardt, F., et al. (2014). De-differentiation confers multidrug resistance via noncanonical PERK-Nrf2 signaling. PLoS Biol, 12(9), e1001945. [CrossRef]
- Saxena, M., Stephens, M. A., Pathak, H., & Rangarajan, A. (2011). Transcription factors that mediate epithelial-mesenchymal transition lead to multidrug resistance by upregulating ABC transporters. Cell Death Dis, 2(7), e179. [CrossRef]
- Fung, S. W., Cheung, P. F., Yip, C. W., Ng, L. W., Cheung, T. T., Chong, C. C., et al. (2019). The ATP-binding cassette transporter ABCF1 is a hepatic oncofetal protein that promotes chemoresistance, EMT and cancer stemness in hepatocellular carcinoma. Cancer Lett, 457, 98-109. [CrossRef]
- Debeb, B. G., Lacerda, L., Xu, W., Larson, R., Solley, T., Atkinson, R., et al. (2012). Histone deacetylase inhibitors stimulate dedifferentiation of human breast cancer cells through WNT/beta-catenin signaling. Stem Cells, 30(11), 2366-2377. [CrossRef]
- Morris, J. C., Tan, A. R., Olencki, T. E., Shapiro, G. I., Dezube, B. J., Reiss, M., et al. (2014). Phase I study of GC1008 (fresolimumab): a human anti-transforming growth factor-beta (TGFbeta) monoclonal antibody in patients with advanced malignant melanoma or renal cell carcinoma. PLoS One, 9(3), e90353. [CrossRef]
- Birchmeier, C., Birchmeier, W., Gherardi, E., & Vande Woude, G. F. (2003). Met, metastasis, motility and more. Nat Rev Mol Cell Biol, 4(12), 915-925. [CrossRef]
- Douillard, J. Y., Ostoros, G., Cobo, M., Ciuleanu, T., McCormack, R., Webster, A., et al. (2014). First-line gefitinib in Caucasian EGFR mutation-positive NSCLC patients: a phase-IV, open-label, single-arm study. Br J Cancer, 110(1), 55-62. [CrossRef]
- Beasley, G. M., Riboh, J. C., Augustine, C. K., Zager, J. S., Hochwald, S. N., Grobmyer, S. R., et al. (2011). Prospective multicenter phase II trial of systemic ADH-1 in combination with melphalan via isolated limb infusion in patients with advanced extremity melanoma. J Clin Oncol, 29(9), 1210-1215. [CrossRef]
- Singh, M., Yelle, N., Venugopal, C., & Singh, S. K. (2018). EMT: Mechanisms and therapeutic implications. Pharmacol Ther, 182, 80-94. [CrossRef]
- Elaskalani, O., Razak, N. B., Falasca, M., & Metharom, P. (2017). Epithelial-mesenchymal transition as a therapeutic target for overcoming chemoresistance in pancreatic cancer. World J Gastrointest Oncol, 9(1), 37-41. [CrossRef]
- Reid, A., & Tursun, B. (2018). Transdifferentiation: do transition states lie on the path of development? Curr Opin Syst Biol, 11, 18-23. [CrossRef]
- Cieslar-Pobuda, A., Knoflach, V., Ringh, M. V., Stark, J., Likus, W., Siemianowicz, K., et al. (2017). Transdifferentiation and reprogramming: Overview of the processes, their similarities and differences. Biochim Biophys Acta Mol Cell Res, 1864(7), 1359-1369. [CrossRef]
- Liu, Z., Wang, L., Welch, J. D., Ma, H., Zhou, Y., Vaseghi, H. R., et al. (2017). Single-cell transcriptomics reconstructs fate conversion from fibroblast to cardiomyocyte. Nature, 551(7678), 100-104. [CrossRef]
- Jarriault, S., Schwab, Y., & Greenwald, I. (2008). A Caenorhabditis elegans model for epithelial-neuronal transdifferentiation. Proc Natl Acad Sci U S A, 105(10), 3790-3795. [CrossRef]
- Szabo, E., Rampalli, S., Risueno, R. M., Schnerch, A., Mitchell, R., Fiebig-Comyn, A., et al. (2010). Direct conversion of human fibroblasts to multilineage blood progenitors. Nature, 468(7323), 521-526. [CrossRef]
- Jonsson, J., Carlsson, L., Edlund, T., & Edlund, H. (1994). Insulin-promoter-factor 1 is required for pancreas development in mice. Nature, 371(6498), 606-609. [CrossRef]
- Ber, I., Shternhall, K., Perl, S., Ohanuna, Z., Goldberg, I., Barshack, I., et al. (2003). Functional, persistent, and extended liver to pancreas transdifferentiation. J Biol Chem, 278(34), 31950-31957. [CrossRef]
- Huang, Z., Wu, T., Liu, A. Y., & Ouyang, G. (2015). Differentiation and transdifferentiation potentials of cancer stem cells. Oncotarget, 6(37), 39550-39563. [CrossRef]
- Wang, R., Chadalavada, K., Wilshire, J., Kowalik, U., Hovinga, K. E., Geber, A., et al. (2010). Glioblastoma stem-like cells give rise to tumour endothelium. Nature, 468(7325), 829-833. [CrossRef]
- Barcellos-de-Souza, P., Gori, V., Bambi, F., & Chiarugi, P. (2013). Tumor microenvironment: bone marrow-mesenchymal stem cells as key players. Biochim Biophys Acta, 1836(2), 321-335. [CrossRef]
- de The, H. (2018). Differentiation therapy revisited. Nat Rev Cancer, 18(2), 117-127. [CrossRef]
- Ishay-Ronen, D., Diepenbruck, M., Kalathur, R. K. R., Sugiyama, N., Tiede, S., Ivanek, R., et al. (2019). Gain Fat-Lose Metastasis: Converting Invasive Breast Cancer Cells into Adipocytes Inhibits Cancer Metastasis. Cancer Cell, 35(1), 17-32 e16. [CrossRef]
- Ishay-Ronen, D., & Christofori, G. (2019). Targeting Cancer Cell Metastasis by Converting Cancer Cells into Fat. Cancer Res, 79(21), 5471-5475. [CrossRef]
- Bruna, A., Greenwood, W., Le Quesne, J., Teschendorff, A., Miranda-Saavedra, D., Rueda, O. M., et al. (2012). TGFbeta induces the formation of tumour-initiating cells in claudinlow breast cancer. Nat Commun, 3, 1055. [CrossRef]
- Blazquez-Medela, A. M., Jumabay, M., & Bostrom, K. I. (2019). Beyond the bone: Bone morphogenetic protein signaling in adipose tissue. Obes Rev, 20(5), 648-658. [CrossRef]
- Ma, X., Wang, D., Zhao, W., & Xu, L. (2018). Deciphering the Roles of PPARgamma in Adipocytes via Dynamic Change of Transcription Complex. Front Endocrinol (Lausanne), 9, 473. [CrossRef]
- Shan, T., Liu, W., & Kuang, S. (2013). Fatty acid binding protein 4 expression marks a population of adipocyte progenitors in white and brown adipose tissues. FASEB J, 27(1), 277-287. [CrossRef]
- Liu, Y., El-Naggar, S., Darling, D. S., Higashi, Y., & Dean, D. C. (2008). Zeb1 links epithelial-mesenchymal transition and cellular senescence. Development, 135(3), 579-588. [CrossRef]
- Flynn, P. J., Miller, W. J., Weisdorf, D. J., Arthur, D. C., Brunning, R., & Branda, R. F. (1983). Retinoic acid treatment of acute promyelocytic leukemia: in vitro and in vivo observations. Blood, 62(6), 1211-1217.
- Leiva, M., Moretti, S., Soilihi, H., Pallavicini, I., Peres, L., Mercurio, C., et al. (2012). Valproic acid induces differentiation and transient tumor regression, but spares leukemia-initiating activity in mouse models of APL. Leukemia, 26(7), 1630-1637. [CrossRef]
- Takahashi, N., Nobusue, H., Shimizu, T., Sugihara, E., Yamaguchi-Iwai, S., Onishi, N., et al. (2019). ROCK Inhibition Induces Terminal Adipocyte Differentiation and Suppresses Tumorigenesis in Chemoresistant Osteosarcoma Cells. Cancer Res, 79(12), 3088-3099. [CrossRef]
- Hirozane, T., Masuda, M., Sugano, T., Sekita, T., Goto, N., Aoyama, T., et al. (2021). Direct conversion of osteosarcoma to adipocytes by targeting TNIK. JCI Insight, 6(3). [CrossRef]
- Nemec, S., & Kilian, K. A. (2021). Materials control of the epigenetics underlying cell plasticity. Nature Reviews Materials, 6(1), 69-83. [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



