Submitted:
26 December 2024
Posted:
27 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
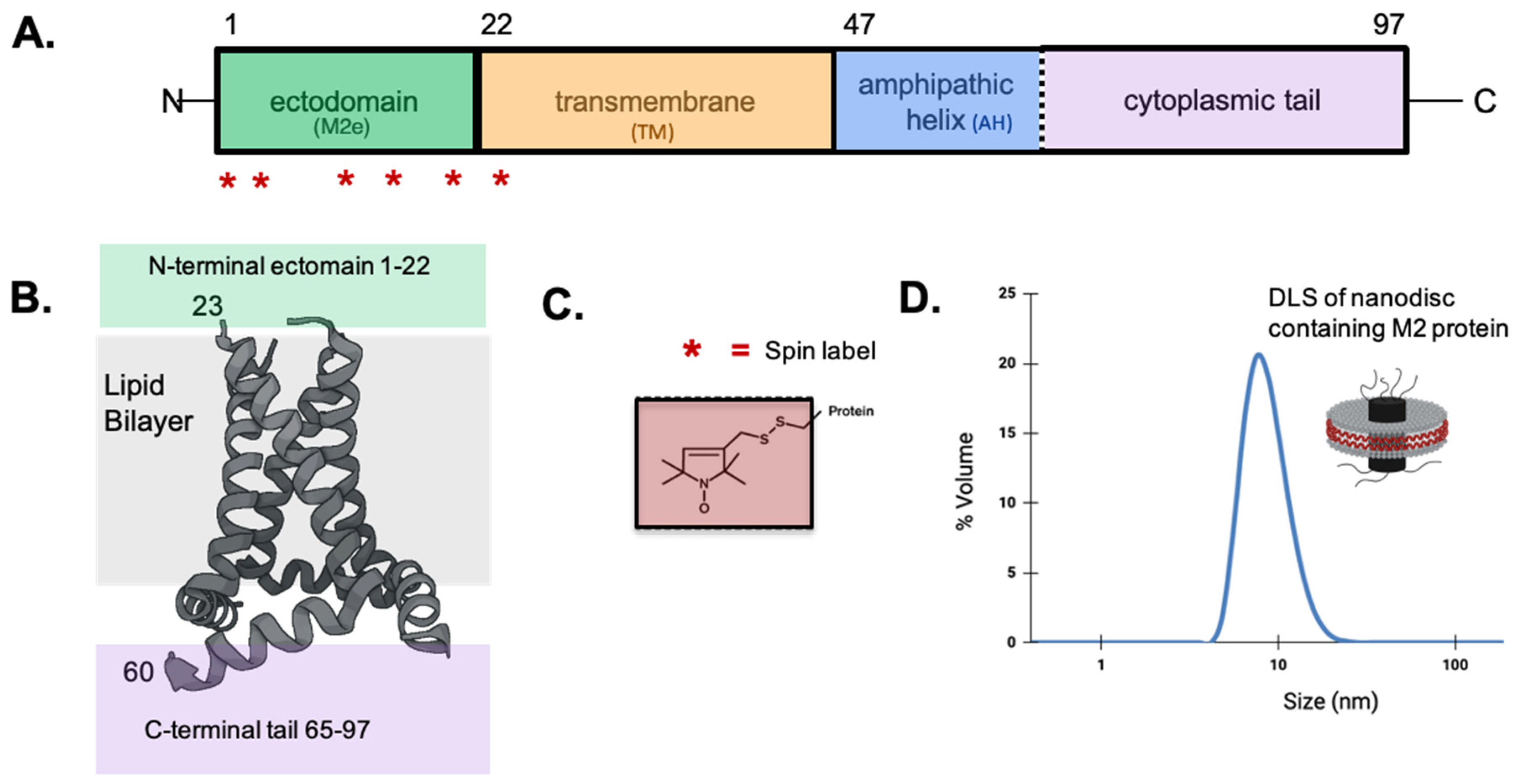
2.1. Expression, Spin Labeling, and Purification of Spin-Labeled Full Length M2 Protein
2.2. Reconstitution of Spin-Labeled M2 Protein Into Nanodiscs
2.3. Composition of Cholesterol and Drug Samples
2.4. Dynamic Light Scattering
2.5. EPR Spectroscopy and Data Analysis
3. Results
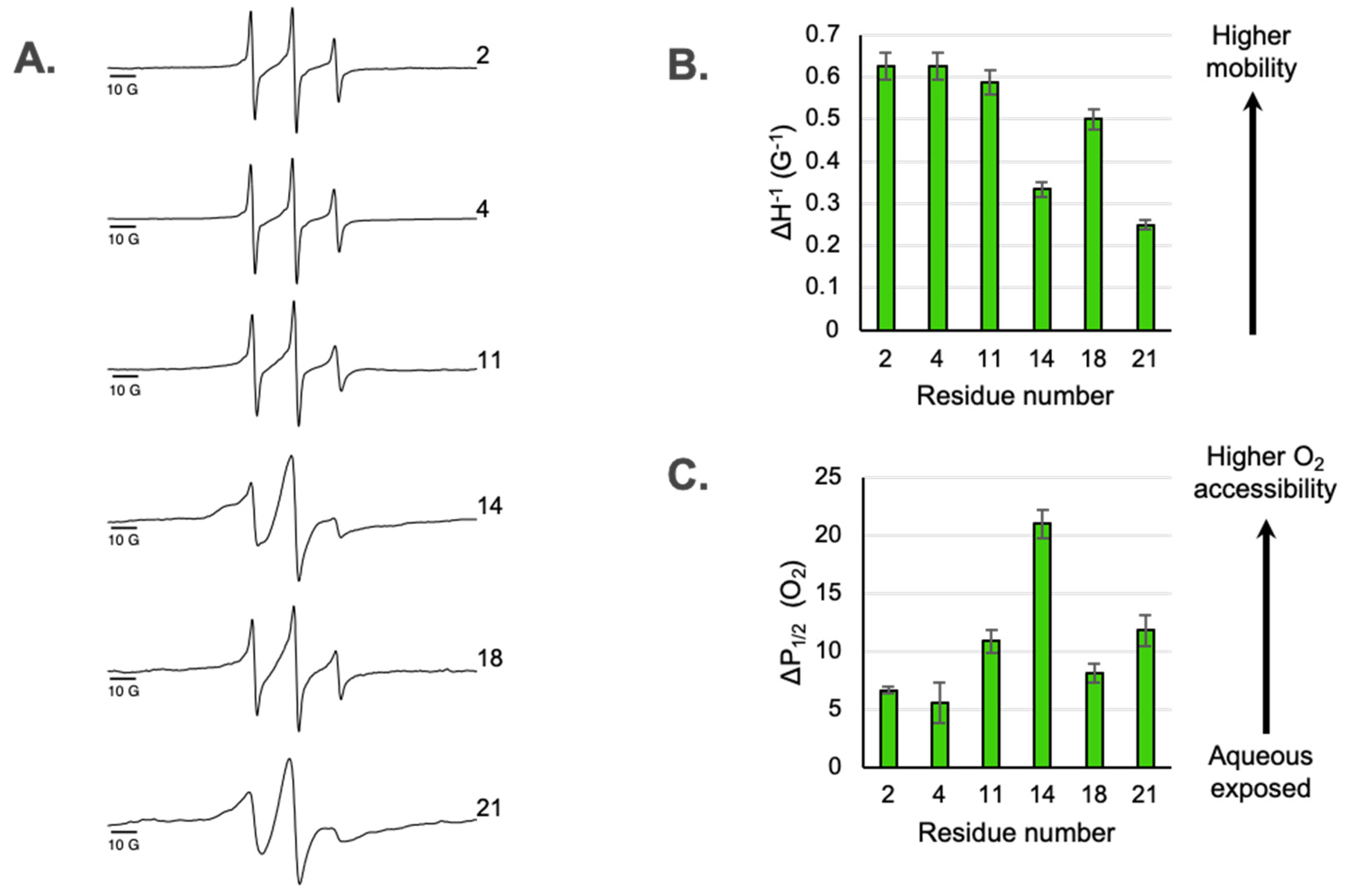
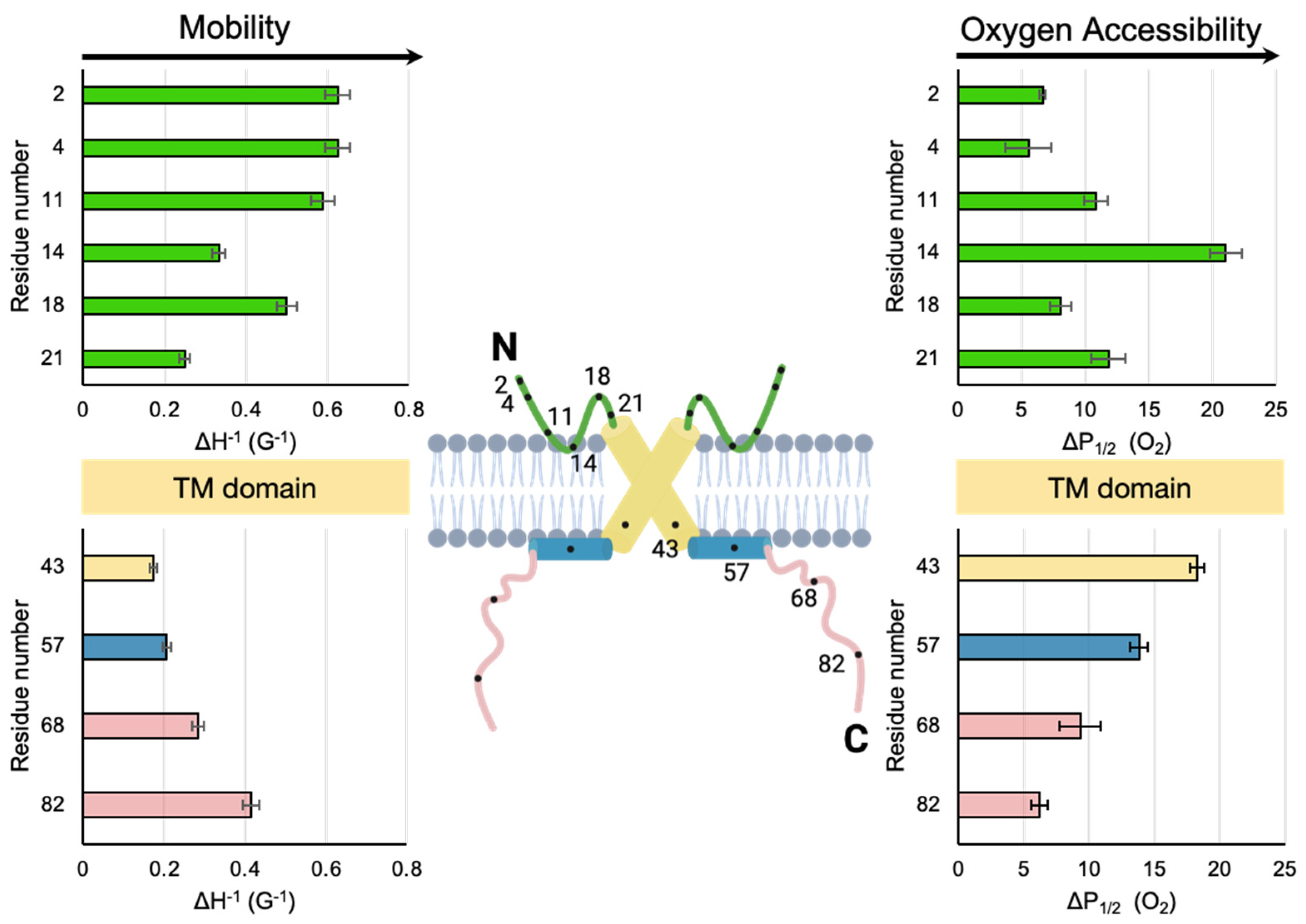
3.1. Dynamic Properties of Sites Along the Ectodomain
3.2. Accessibility to Paramagnetic Relaxation Agents Along the Ectodomain
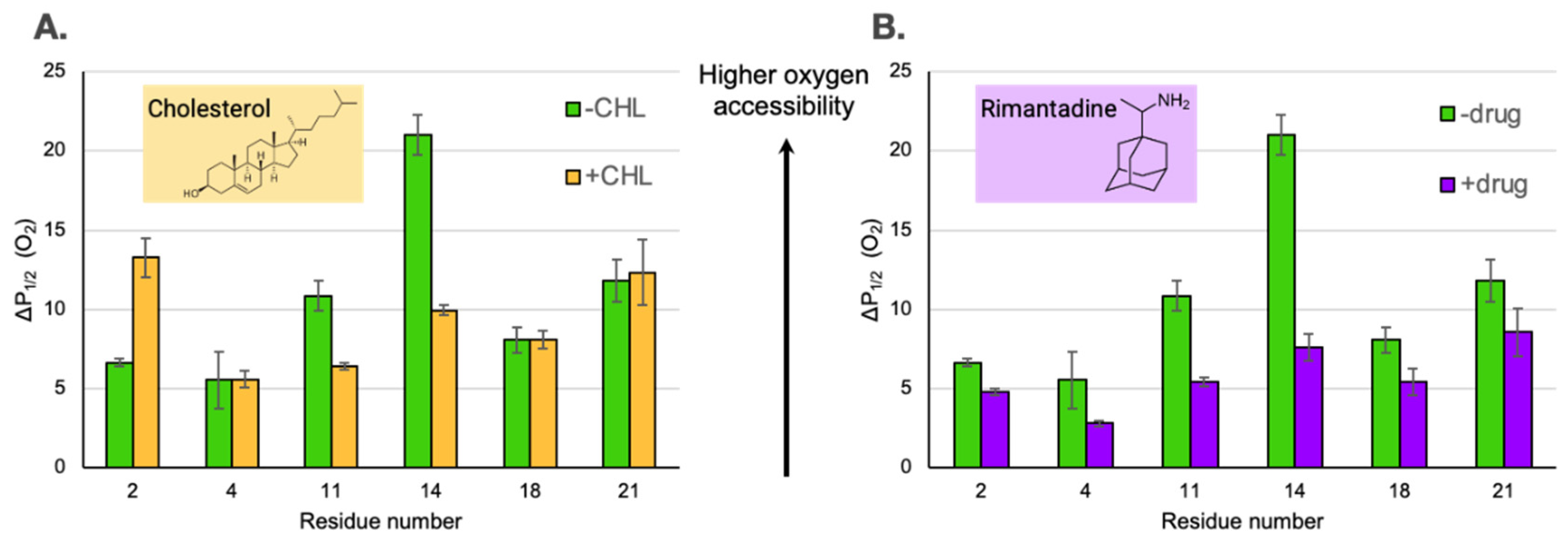
3.3. Impact of Cholesterol and Drug Upon Properties of M2e
4. Discussion
4.1. Comparison of the Two Extramembranous Domains of M2
4.2. Putative Membrane Insertion Motif Within the Ectodomain
4.3. Cholesterol and the Ectodomain
4.4. Drug Binding Impacts Ectodomain Properties
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
References
- Chan, D. I., Prenner, E. J., & Vogel, H. J. (2006). Tryptophan- and arginine-rich antimicrobial peptides: Structures and mechanisms of action. Biochimica et Biophysica Acta (BBA) - Biomembranes, 1758(9), 1184–1202. [CrossRef]
- Cho, K. J., Schepens, B., Moonens, K., Deng, L., Fiers, W., Remaut, H., & Saelens, X. (2016). Crystal Structure of the Conserved Amino Terminus of the Extracellular Domain of Matrix Protein 2 of Influenza A Virus Gripped by an Antibody. Journal of Virology, 90(1), 611–615. [CrossRef]
- Cho, K. J., Schepens, B., Seok, J. H., Kim, S., Roose, K., Lee, J.-H., … Kim, K. H. (2015). Structure of the Extracellular Domain of Matrix Protein 2 of Influenza A Virus in Complex with a Protective Monoclonal Antibody. Journal of Virology, 89(7), 3700–3711. [CrossRef]
- Dou, D., Revol, R., Östbye, H., Wang, H., & Daniels, R. (2018). Influenza A Virus Cell Entry, Replication, Virion Assembly and Movement. Frontiers in Immunology, 9, 1581. [CrossRef]
- Duong-Ly, K. C., Nanda, V., DeGrado, W. F., & Howard, K. P. (2005). The conformation of the pore region of the M2 proton channel depends on lipid bilayer environment. Protein Science, 14(4), 856–861. [CrossRef]
- Ekanayake, E. V., Fu, R., & Cross, T. A. (2016). Structural Influences: Cholesterol, Drug, and Proton Binding to Full-Length Influenza A M2 Protein. Biophysical Journal, 110(6), 1391–1399. [CrossRef]
- Frazier, A. A., Wisner, M. A., Malmberg, N. J., Victor, K. G., Fanucci, G. E., Nalefski, E. A., … Cafiso, D. S. (2002). Membrane Orientation and Position of the C2 Domain from cPLA2 by Site-Directed Spin Labeling. Biochemistry, 41(20), 6282–6292. [CrossRef]
- Gu, R.-X., Liu, L. A., & Wei, D.-Q. (2013). Structural and energetic analysis of drug inhibition of the influenza A M2 proton channel. Trends in Pharmacological Sciences, 34(10), 571–580. [CrossRef]
- Hagn, F., Nasr, M. L., & Wagner, G. (2018). Assembly of phospholipid nanodiscs of controlled size for structural studies of membrane proteins by NMR. Nature Protocols, 13(1), 79–98. [CrossRef]
- Han, A. X., De Jong, S. P. J., & Russell, C. A. (2023). Co-evolution of immunity and seasonal influenza viruses. Nature Reviews Microbiology, 21(12), 805–817. [CrossRef]
- Hayden, F. G., Asher, J., Cowling, B. J., Hurt, A. C., Ikematsu, H., Kuhlbusch, K., … Monto, A. S. (2022). Reducing Influenza Virus Transmission: The Potential Value of Antiviral Treatment. Clinical Infectious Diseases, 74(3), 532–540. [CrossRef]
- Herneisen, A. L., Sahu, I. D., McCarrick, R. M., Feix, J. B., Lorigan, G. A., & Howard, K. P. (2017). A Budding-Defective M2 Mutant Exhibits Reduced Membrane Interaction, Insensitivity to Cholesterol, and Perturbed Interdomain Coupling. Biochemistry, 56(44), 5955–5963. [CrossRef]
- Hu, F., Luo, W., Cady, S. D., & Hong, M. (2011). Conformational plasticity of the influenza A M2 transmembrane helix in lipid bilayers under varying pH, drug binding, and membrane thickness. Biochimica et Biophysica Acta (BBA) - Biomembranes, 1808(1), 415–423. [CrossRef]
- Huang, S., Green, B., Thompson, M., Chen, R., Thomaston, J., DeGrado, W. F., & Howard, K. P. (2015). C-terminal juxtamembrane region of full-length M2 protein forms a membrane surface associated amphipathic helix. Protein Science, 24(3), 426–429. [CrossRef]
- Jalily, P. H., Duncan, M. C., Fedida, D., Wang, J., & Tietjen, I. (2020). Put a cork in it: Plugging the M2 viral ion channel to sink influenza. Antiviral Research, 178, 104780. [CrossRef]
- Kandeil, A., Patton, C., Jones, J. C., Jeevan, T., Harrington, W. N., Trifkovic, S., … Webby, R. J. (2023). Rapid evolution of A(H5N1) influenza viruses after intercontinental spread to North America. Nature Communications, 14(1), 3082. [CrossRef]
- Kim, G., Raymond, H. E., Herneisen, A. L., Wong-Rolle, A., & Howard, K. P. (2019). The distal cytoplasmic tail of the influenza A M2 protein dynamically extends from the membrane. Biochimica et Biophysica Acta (BBA) - Biomembranes, 1861(8), 1421–1427. [CrossRef]
- Kim, S. S., Upshur, M. A., Saotome, K., Sahu, I. D., McCarrick, R. M., Feix, J. B., … Howard, K. P. (2015). Cholesterol-Dependent Conformational Exchange of the C-Terminal Domain of the Influenza A M2 Protein. Biochemistry, 54(49), 7157–7167. [CrossRef]
- Klug, C. S., & Feix, J. B. (2008). Methods and Applications of Site-Directed Spin Labeling EPR Spectroscopy. In Methods in Cell Biology (Vol. 84, pp. 617–658). Elsevier. [CrossRef]
- Kumar, G., & Sakharam, K. A. (2024). Tackling Influenza A virus by M2 ion channel blockers: Latest progress and limitations. European Journal of Medicinal Chemistry, 267, 116172. [CrossRef]
- Kwon, B., & Hong, M. (2016). The Influenza M2 Ectodomain Regulates the Conformational Equilibria of the Transmembrane Proton Channel: Insights from Solid-State Nuclear Magnetic Resonance. Biochemistry, 55(38), 5387–5397. [CrossRef]
- Kyaw, A., Roepke, K., Arthur, T., & Howard, K. P. (2023). Conformation of influenza AM2 membrane protein in nanodiscs and liposomes. Biochimica et Biophysica Acta (BBA) - Biomembranes, 1865(5), 184152. [CrossRef]
- Leiding, T., Wang, J., Martinsson, J., DeGrado, W. F., & Årsköld, S. P. (2010). Proton and cation transport activity of the M2 proton channel from influenza A virus. Proceedings of the National Academy of Sciences, 107(35), 15409–15414. [CrossRef]
- Liao, S. Y., Fritzsching, K. J., & Hong, M. (2013). Conformational analysis of the full-length M2 protein of the influenza A virus using solid-state NMR. Protein Science, 22(11), 1623–1638. [CrossRef]
- Martyna, A., Bahsoun, B., Madsen, J. J., Jackson, F. St. J. S., Badham, M. D., Voth, G. A., & Rossman, J. S. (2020). Cholesterol Alters the Orientation and Activity of the Influenza Virus M2 Amphipathic Helix in the Membrane. The Journal of Physical Chemistry B, 124(31), 6738–6747. [CrossRef]
- Matthys, A., & Saelens, X. (2024). Promises and challenges of single-domain antibodies to control influenza. Antiviral Research, 222, 105807. [CrossRef]
- Mezhenskaya, D., Isakova-Sivak, I., & Rudenko, L. (2019). M2e-based universal influenza vaccines: A historical overview and new approaches to development. Journal of Biomedical Science, 26(1), 76. [CrossRef]
- Nguyen, Phuong A., Cinque S. Soto, Alexei Polishchuk, Gregory A. Caputo, Chad D. Tatko, Chunlong Ma, Yuki Ohigashi, Lawrence H. Pinto, William F. DeGrado, and Kathleen P. Howard. (2008) pH-Induced Conformational Change of the Influenza M2 Protein C-Terminal Domain. Biochemistry 47(38), 9934–36. [CrossRef]
- Ohigashi, Y., Ma, C., Jing, X., Balannick, V., Pinto, L. H., & Lamb, R. A. (2009). An amantadine-sensitive chimeric BM2 ion channel of influenza B virus has implications for the mechanism of drug inhibition. Proceedings of the National Academy of Sciences, 106(44), 18775–18779. [CrossRef]
- Park, E. K., Castrucci, M. R., Portner, A., & Kawaoka, Y. (1998). The M2 Ectodomain Is Important for Its Incorporation into Influenza A Virions. Journal of Virology, 72(3), 2449–2455. [CrossRef]
- Paules, C., & Subbarao, K. (2017). Influenza. The Lancet, 390(10095), 697–708. [CrossRef]
- Paulino, J., Pang, X., Hung, I., Zhou, H.-X., & Cross, T. A. (2019). Influenza A M2 Channel Clustering at High Protein/Lipid Ratios: Viral Budding Implications. Biophysical Journal, 116(6), 1075–1084. [CrossRef]
- Pinto, L. H., Dieckmann, G. R., Gandhi, C. S., Papworth, C. G., Braman, J., Shaughnessy, M. A., … DeGrado, W. F. (1997). A functionally defined model for the M 2 proton channel of influenza A virus suggests a mechanism for its ion selectivity. Proceedings of the National Academy of Sciences, 94(21), 11301–11306. [CrossRef]
- Pinto, L. H., & Lamb, R. A. (2006). The M2 Proton Channels of Influenza A and B Viruses. Journal of Biological Chemistry, 281(14), 8997–9000. [CrossRef]
- Rossman, J. S., & Lamb, R. A. (2011). Influenza virus assembly and budding. Virology, 411(2), 229–236. [CrossRef]
- Schepens, B., De Vlieger, D., & Saelens, X. (2018). Vaccine options for influenza: Thinking small. Current Opinion in Immunology, 53, 22–29. [CrossRef]
- Shuck, K., Lamb, R. A., & Pinto, L. H. (2000). Analysis of the Pore Structure of the Influenza A Virus M 2 Ion Channel by the Substituted-Cysteine Accessibility Method. Journal of Virology, 74(17), 7755–7761. [CrossRef]
- Subczynski, W K, J S Hyde, and A Kusumi. (1989) “Oxygen Permeability of Phosphatidylcholine--Cholesterol Membranes.” Proceedings of the National Academy of Sciences 86, (12) 4474–78. [CrossRef]
- Subczynski, W. K., Widomska, J., & Feix, J. B. (2009). Physical properties of lipid bilayers from EPR spin labeling and their influence on chemical reactions in a membrane environment. Free Radical Biology and Medicine, 46(6), 707–718. [CrossRef]
- Thomaston, J. L., Nguyen, P. A., Brown, E. C., Upshur, M. A., Wang, J., DeGrado, W. F., & Howard, K. P. (2013). Detection of drug-induced conformational change of a transmembrane protein in lipid bilayers using site-directed spin labeling. Protein Science, 22(1), 65–73. [CrossRef]
- Townsend, J. A., Sanders, H. M., Rolland, A. D., Park, C. K., Horton, N. C., Prell, J. S., … Marty, M. T. (2021). Influenza AM2 Channel Oligomerization Is Sensitive to Its Chemical Environment. Analytical Chemistry, 93(48), 16273–16281. [CrossRef]
- Yi, M., Cross, T. A., & Zhou, H.-X. (2009). Conformational heterogeneity of the M2 proton channel and a structural model for channel activation. Proceedings of the National Academy of Sciences, 106(32), 13311–13316. [CrossRef]
- Yin, H., Jiang, N., Shi, W., Chi, X., Liu, S., Chen, J.-L., & Wang, S. (2021). Development and Effects of Influenza Antiviral Drugs. Molecules, 26(4), 810. [CrossRef]
- Yu, C., Ding, W., Zhu, L., Zhou, Y., Dong, Y., Li, L., … Wang, J. (2023). Screening and characterization of inhibitory vNAR targeting nanodisc-assembled influenza M2 proteins. iScience, 26(1), 105736. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



