Submitted:
24 December 2024
Posted:
24 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Quality Parameters of Deepsea Water Concentrate
2.1.1. Physical Parameters
2.1.2. Chemical Parameters
2.1.3. Biological Parameters
2.1.4. Chemical Analysis of Deep Seawater Concentrate
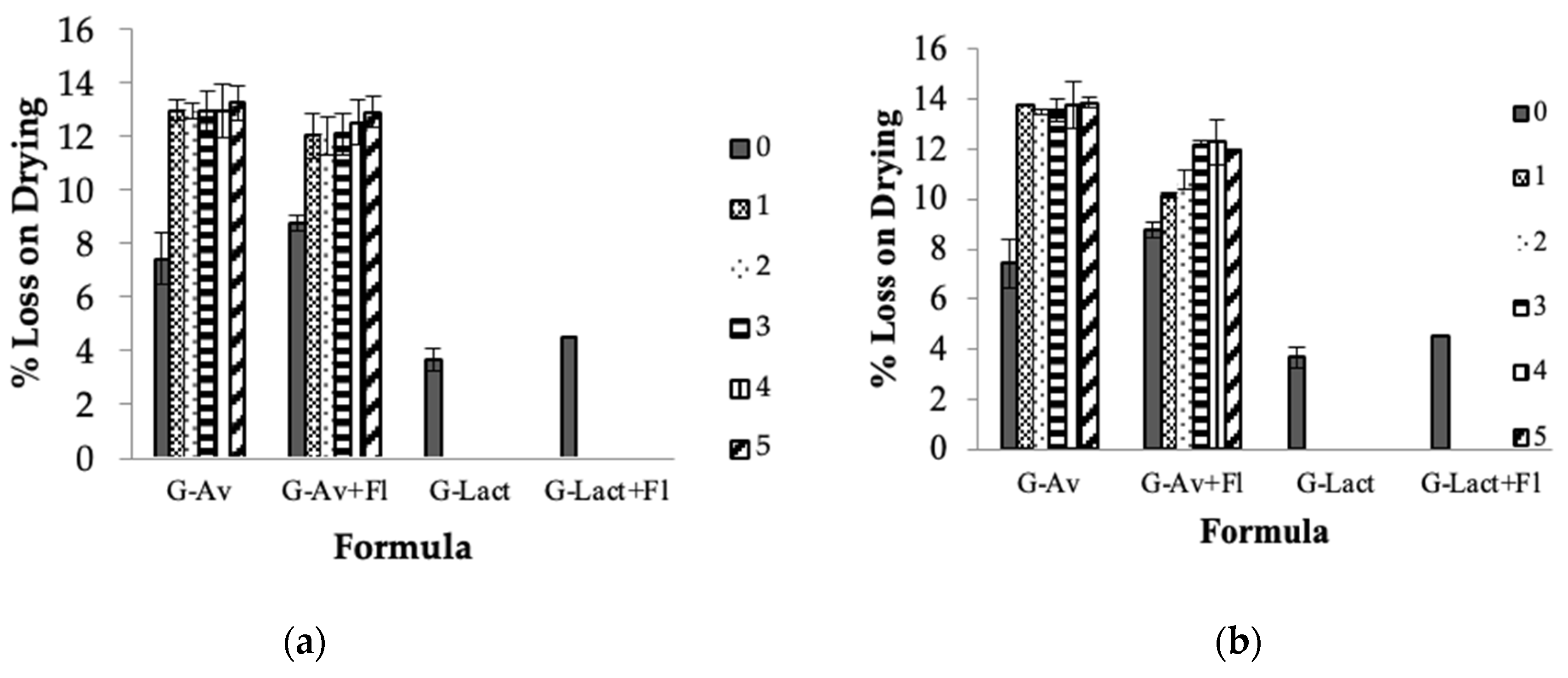
2.2. Formulations and Stability Study of Deep Seawater Granule
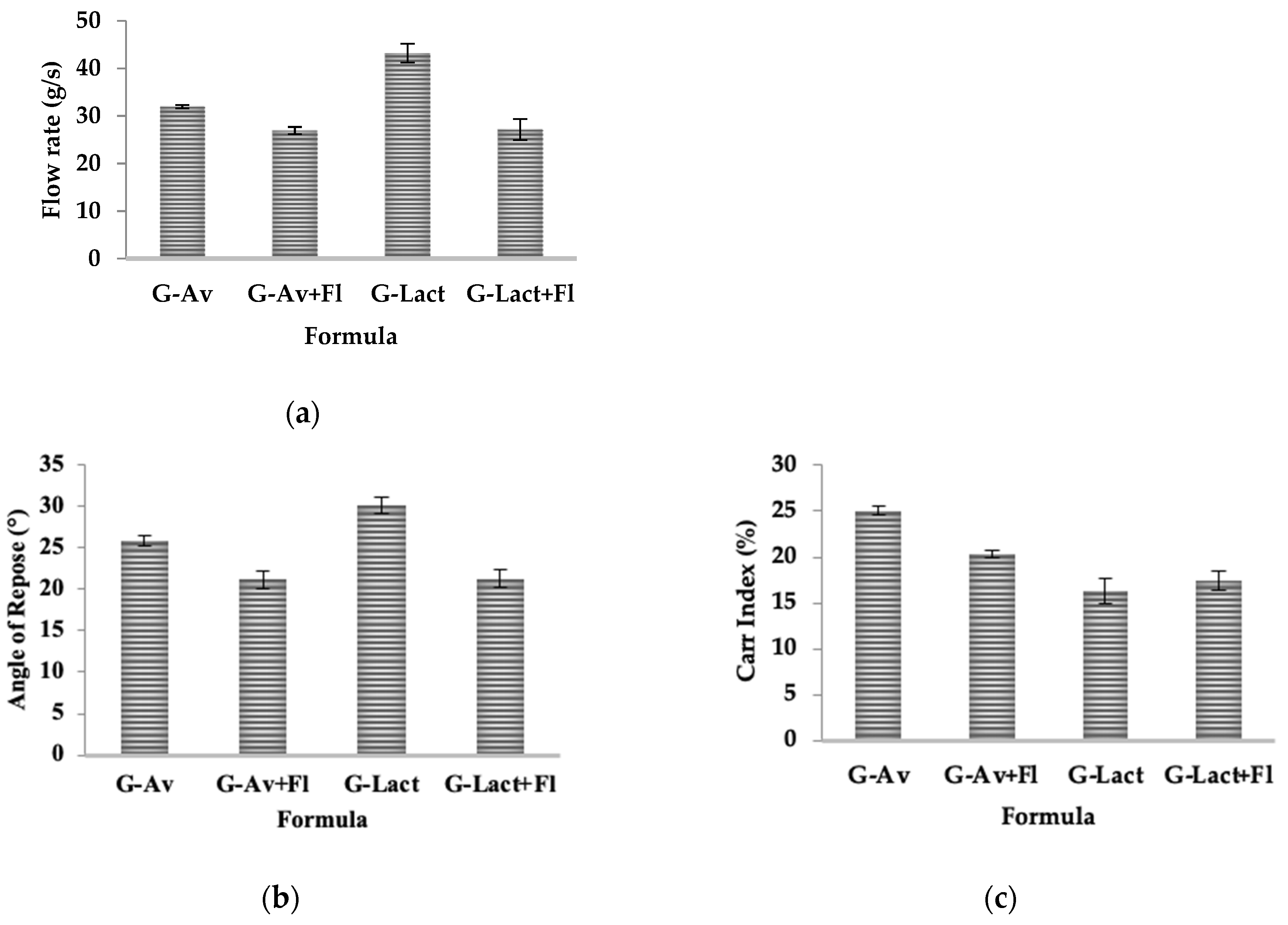
2.3. Physical Stability Study of Granules Using Instruments
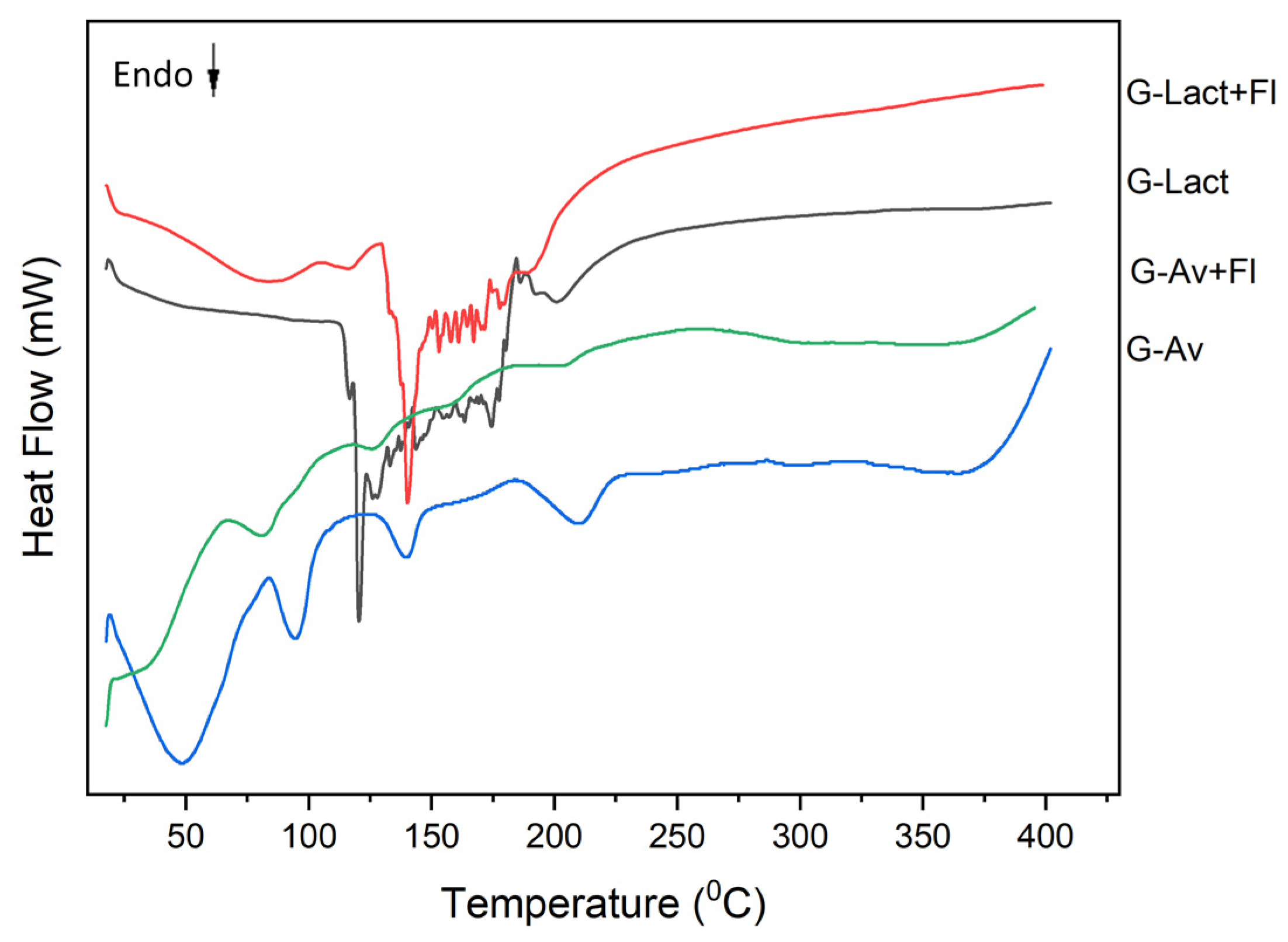
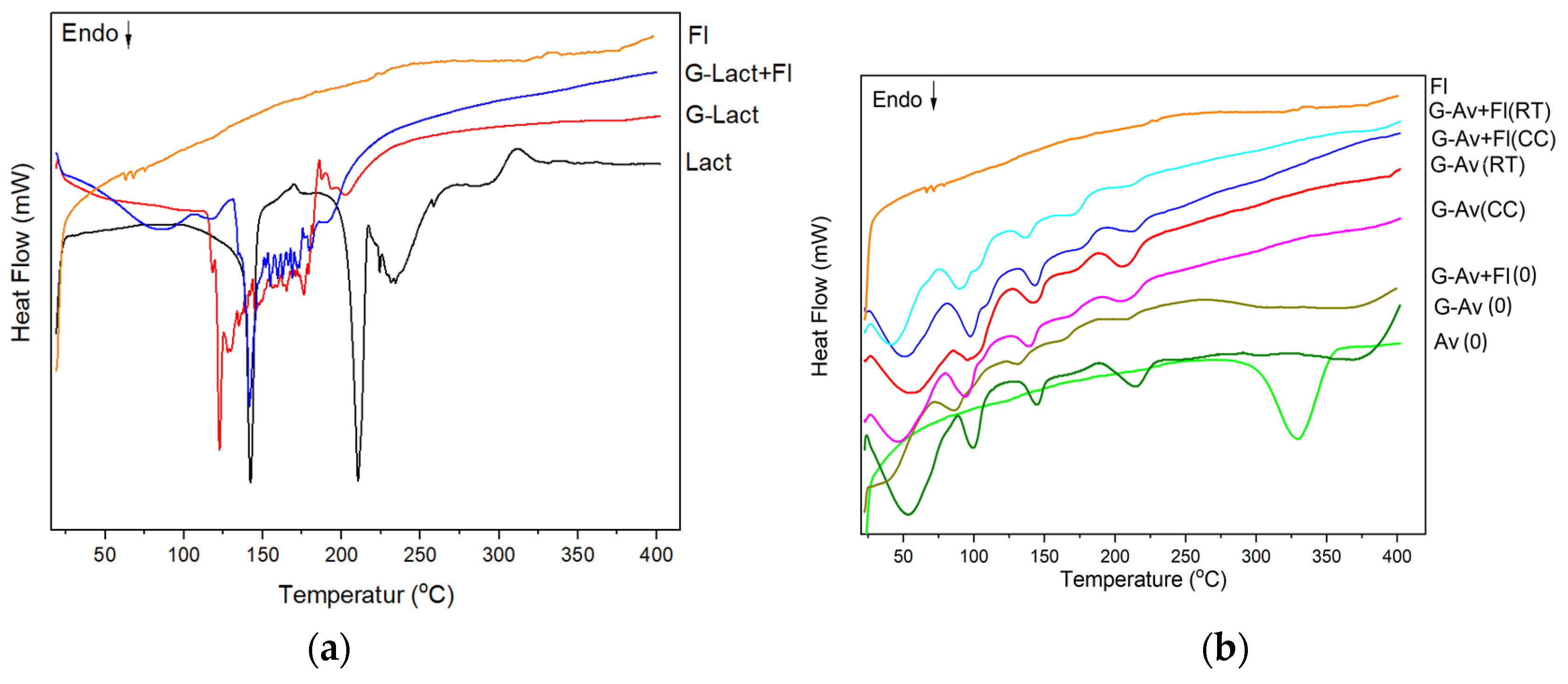
2.3.1. Thermal Analysis by DSC
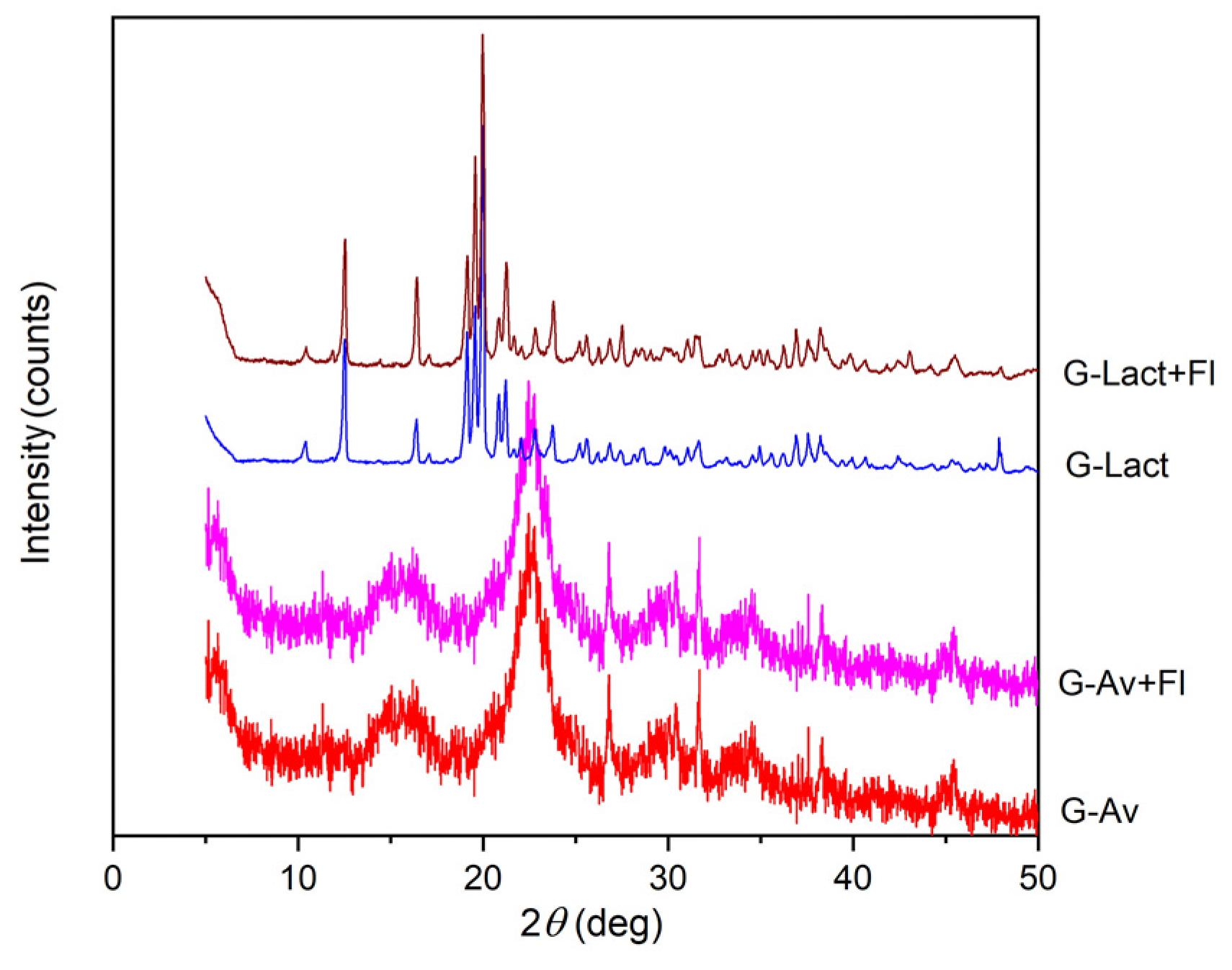
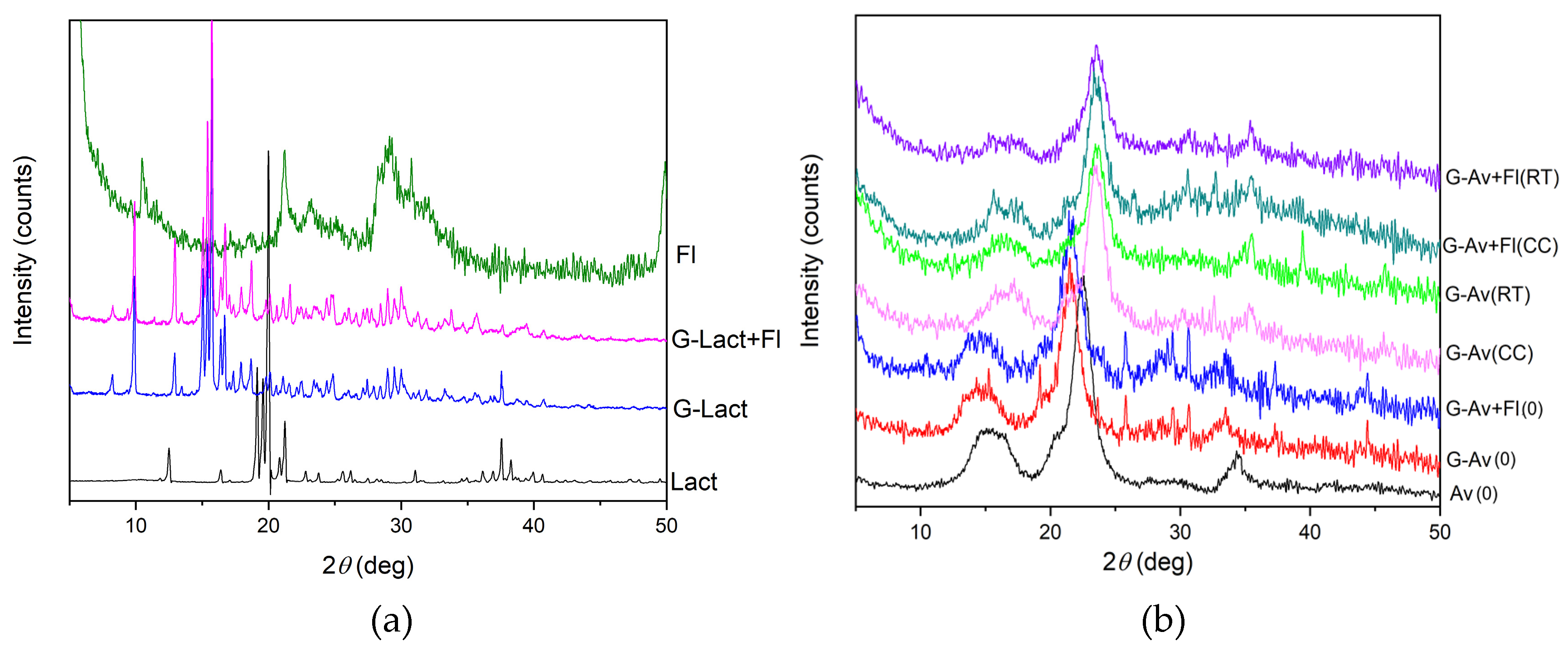
2.3.2. XRD Analysis
2.3.3. Morphology Study by Scanning Electron Microscope
3. Discussion
- Physical Parameters
- b.
- Chemical Parameters
- c.
- Biological Parameters
4. Materials and Methods
4.1. Deep Seawater Concentrate Production
4.2. Determination of Quality Parameters from Deepsea Water Concentrate
4.2.1. Physical Parameters
4.2.2. Chemical Parameters
4.2.3. Biological Parameters
4.3. Chemical Content Analysis of Deep Seawater Concentrates
4.4. Production of Deep Seawater Granules
4.4.1. Formulation of Deep Seawater Granules
4.4.2. Evaluation of Granules
- Organoleptic Evaluation
- b.
- Flow Properties Evaluation
- c.
- Angle of Repose
- d.
- Compressibility/Carr’s Index
- e.
- Loss on Drying (LoD)
- f.
- Content Analysis with ICP-OES
- g.
- Stability Testing
- h.
- Quality Parameter Testing of Granules Using Instruments
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Apriani, M.; Hadi, W.; Masduqi, A. Physicochemical Properties of Sea Water and Bittern in Indonesia: Quality Improvement and Potential Resources Utilization for Marine Environmental Sustainability. J. Ecol. Eng. 2018, 19, 1–10. [CrossRef]
- Sriwidodo ; Umar, A.K. ; Megawati, E. ; Butarbutar, M. ; Wathoni, N. ; Alaydrus, S. Physicochemical Characterization of Concentrated Mineral and Magnesium Isolate of Sea Water Pamekasan Madura. Int. J. Res. Pharm. Sci. 2020, 11, 2154–2157.
- Mahmoud, M.G.; El-Khir, E.A.; Ebeid, M.H.; Mohamed, L.A.; Fahmy, M.A.; Shaban, K.S. An Assessment on the Coastal Seawater Quality of the Gulf of Suez, Egypt. J. Environ. Prot. (Irvine,. Calif). 2020, 11, 34–47. [CrossRef]
- Landrigan, P.J.; Stegeman, J.J.; Fleming, L.E.; Allemand, D.; Anderson, D.M.; Backer, L.C.; Brucker-Davis, F.; Chevalier, N.; Corra, L.; Czerucka, D.; et al. Human Health and Ocean Pollution. Ann. Glob. Heal. 2020, 86, 1–64. [CrossRef]
- Barin, Juliano S; Mello, Paola A.; Mesko, Marcia F. and Duarte, Fabio A.; Flores, E.M.M. Determination of Elemental Impurities in Pharmaceutical Products and Related Matrices by ICP-Based Methods: A Review. Anal. Bioanal. Chem. 2016, 408, 4547–4566.
- Marguí, E.; Queralt, I.; de Almeida, E. X-Ray Fluorescence Spectrometry for Environmental Analysis: Basic Principles, Instrumentation, Applications and Recent Trends. Chemosphere 2022, 303, 135006. [CrossRef]
- Jadav, M.; Pal, V.; Pandit, H.; Suthar, N.; Pithadia, A.; Jha, L.L. Formulation, Optimization and Evaluation of Nutraceutical Effervescent Granules Containing Curcumin and Ascorbic Acid by Fusion Technique Using Full Factorial Design. J. Nat. Remedies 2023, 23, 1509–1516. [CrossRef]
- Guo, L.; Gu, W.; Peng, C.; Wang, W.; Jie Li, Y.; Zong, T.; Tang, Y.; Wu, Z.; Lin, Q.; Ge, M.; et al. A Comprehensive Study of Hygroscopic Properties of Calcium-and Magnesium-Containing Salts: Implication for Hygroscopicity of Mineral Dust and Sea Salt Aerosols. Atmos. Chem. Phys. 2019, 19, 2115–2133. [CrossRef]
- Zhang, H.; Gu, W.; Li, Y.J.; Tang, M. Hygroscopic Properties of Sodium and Potassium Salts as Related to Saline Mineral Dusts and Sea Salt Aerosols. J. Environ. Sci. (China) 2020, 95, 65–72. [CrossRef]
- Anbarasan, A.; Nataraj, J.; Shanmukhan, N.; Radhakrishnan, A. Effect of Hygroscopicity on Pharmaceutical Ingredients, Methods to Determine and Overcome: An Overview. Available online www.jocpr.com J. Chem. Pharm. Res. 2018, 10, 61–67.
- Ng, L.H.; Kim, J.; Ling, U.; Hadinoto, K. Formulation Strategies to Improve the Stability and Handling of Oral Solid Dosage Forms of Highly Hygroscopic Pharmaceuticals and Nutraceuticals. 2022.
- El Mabrouki, H.; Kaukhova, I.E. Formulation and Development of Aqueous Film Coating for Moisture Protection of Hygroscopic Herniaria Glabra L. Tablets. Turkish J. Pharm. Sci. 2022, 19, 153–160. [CrossRef]
- Weyh, C.; Krüger, K.; Peeling, P.; Castell, L. The Role of Minerals in the Optimal Functioning of the Immune System. Nutrients 2022, 14, 1–15. [CrossRef]
- Petersen, S. Marine Mineral Resources. Encycl. Mar. Geosci. 2015, 1–10. [CrossRef]
- Breik, L.; Tatucu-Babet, O.A.; Ridley, E.J. Micronutrient Intake from Enteral Nutrition in Critically Ill Adults: A Systematic Review of Randomised Controlled Trials. Aust. Crit. Care 2022, 35, 564–574. [CrossRef]
- 2016; 16. Indonesian Ministry of Environment and Forestry Sea Water Quality Monitoring; Indonesian Ministry of Environment and Forestry, 2016.
- Republic of Indonesia Government Regulations Implementation of Environmental Protection and Management.
- Minister of Health of the Republic of Indonesia Regulation of the Minister of Health of the Republic of Indonesia No 492/Menkes/PER/IV/2010 Concerning Drinking Water Quality Requirements.
- Madeira, D.; Andrade, J.; Leal, M.C.; Ferreira, V.; Rocha, R.J.; Rosa, R.; Calado, R. Synergistic Effects of Ocean Warming and Cyanide Poisoning in an Ornamental Tropical Reef Fish. Front. Mar. Sci. 2020, 7.
- Dumka, N. J.; Kingdom, A. Total Hydrocarbon Concentrations (THC) in Surface Water, Sediments and Biota from Otamiri River, Rivers State, Nigeria. IJCS 2018, 6, 2743–2748.
- Mascarenhas, V.; Keck, T. Marine Optics and Ocean Color Remote Sensing. In Proceedings of the YOUMARES 8; 2018; pp. 41–54.
- Alaydrus, S.; Umar, A.; Sriwidodo, S.; Diantini, A.; Wathoni, N.; Amalia, R. Characterization and Acute Oral Toxicity of Concentrated Minerals of Pamekasan Madura Seawater. J. Adv. Pharm. Technol. Res. 2021, 12, 305–309. [CrossRef]
- Debelius, B.; Gómez-Parra, A.; Forja, J.M. Oxygen Solubility in Evaporated Seawater as a Function of Temperature and Salinity. Hydrobiologia 2009, 632, 157–165. [CrossRef]
- Rajendiran, T.; Sabarathinam, C.; Panda, B.; Elumalai, V. Influence of Dissolved Oxygen, Water Level and Temperature on Dissolved Organic Carbon in Coastal Groundwater. Hydrology 2023, 10. [CrossRef]
- O’Connor Šraj, L.; Almeida, M.I.G.S.; McKelvie, I.D.; Kolev, S.D. Determination of Trace Levels of Ammonia in Marine Waters Using a Simple Environmentally-Friendly Ammonia (SEA) Analyser. Mar. Chem. 2017, 194, 133–145. [CrossRef]
- Wu, X.; Hou, L.; Lin, X.; Xie, Z. Application of Novel Nanomaterials for Chemo- and Biosensing of Algal Toxins in Shellfish and Water; Elsevier Inc., 2019; ISBN 9780128144978.
- Banerjee, P.; Garai, P.; Saha, N.C.; Saha, S.; Sharma, P.; Maiti, A.K. A Critical Review on the Effect of Nitrate Pollution in Aquatic Invertebrates and Fish. Water. Air. Soil Pollut. 2023, 234, 1–14. [CrossRef]
- Ciji, A.; Akhtar, M.S. Nitrite Implications and Its Management Strategies in Aquaculture: A Review. Rev. Aquac. 2020, 12, 878–908. [CrossRef]
- Hofer, H.T.B.; Ng, P.C.; Witeof, A.E.; Mahon, S.B.; Brenner, M.; Boss, G.R.; Bebarta, V.S. A Review on Ingested Cyanide: Risks, Clinical Presentation, Diagnostics, and Treatment Challenges. J Med Toxicol 2019, 15, 128–133.
- WHO Hydrogen Sulfide in Drinking-Water.
- Dumka, N.J. Total Hydrocarbon Concentrations (THC) in Surface Water, Sediments and Biota from Otamiri River, Rivers State, Nigeria. IJCS 2018, 6, 2743–2748.
- Mainali, K. Phenolic Compounds Contaminants in Water: A Glance. Cur Trends Civ. Struct Eng 2020, 4.
- Abd El-Gawad, H.S. Aquatic Environmental Monitoring and Removal Efficiency of Detergents. Water Sci. 2014, 28, 51–64. [CrossRef]
- Khozanah; Yogaswara, D.; Wulandari, I. Oil and Grease (OG) Content in Seawater and Sediment Of The Jakarta Bay and Its Surrounding. IOP Conf.Ser Earth Environ. Sci. 2015, 789.
- Gruber, J.S.; Ercumen, A.; Colford Jr, J.M. Coliform Bacteria as Indicators of Diarrheal Risk in Household Drinking Water: Systematic Review and Metaanalysis. PLoS One 2014, 9.
- WHO Guidelines for Drinking-Water Quality; 4th ed. ; World Health Organization: Geneva, 2011; 36. WHO Guidelines for Drinking-Water Quality; 4th ed.; World Health Organization: Geneva, 2011.
- Moriya, T. Analysis of Trace Impurities in Pharmaceutical Products Using Polarized EDXRF Spectrometer NEX CG. Rigaku J. 2013, 29, 19–21.
- Nölte, J. ICP Emission Spectrometry: A Practical Guide; 2nd ed.; Wiley-VCH: Weinheim, 2021.
- Singh, V.K. ; Kawai, J. ; Tripathi, D.K. X-Ray Fluorescence in Biological Sciences: Principles, Instrumentation, and Applications; Wiley Publisher: Hoboken, 2022.
- Na, C.; Park, H.; Jho, E.H. Utilization of Waste Bittern from Saltern as a Source for Magnesium and an Absorbent for Carbon Dioxide Capture. Env. Sci Pollut Res. 2017, 24, 22980–22989.
- Kudo, Y.; Yasuda, M.; Matsusaka, S. Effect of Particle Size Distribution on Flowability of Granulated Lactose. Adv. Powder Technol. 2020, 31, 121–127. [CrossRef]
- Juarez-Enriquez, E.; Olivas, G.I.; Zamudio-Flores, P.B.; Ortega-Rivas, E.; Perez-Vega, S.; Sepulveda, D.R. Effect of Water Content on the Flowability of Hygroscopic Powders. J. Food Eng. 2017, 205, 12–17. [CrossRef]
- Shah, D.S.; Moravkar, K.K.; Jha, D.K.; Lonkar, V.; Amin, P.D.; Chalikwar, S.S. A Concise Summary of Powder Processing Methodologies for Flow Enhancement. Heliyon 2023, 9, e16498. [CrossRef]
- Berkenkemper, S.; Klinken, S.; Kleinebudde, P. Investigating Compressibility Descriptors for Binary Mixtures of Different Deformation Behavior. Powder Technol. 2023, 424, 118571. [CrossRef]
- Baumgartner, A.; Planinšek, O. Effect of Process Parameters in High Shear Granulation on Characteristics of a Novel Co-Processed Mesoporous Silica Material. Eur. J. Pharm. Sci. 2023, 188. [CrossRef]
- Tomita ph Florite. 2018.
- Rowe, C.R.; Sheskey, J.P.; Owen, C.S. Hanbook of Pharmaceutical Excipients Sixth Edition; Sixth Edit.; RPS Publishing, 2009.
- Nofrerias, I.; Nardi, A.; Suñé-Pou, M.; Suñé-Negre, J.M.; García-Montoya, E.; Pérez-Lozano, P.; Ticó, J.R.; Miñarro, M. Comparison between Microcrystalline Celluloses of Different Grades Made by Four Manufacturers Using the SeDeM Diagram Expert System as a Pharmaceutical Characterization Tool. Powder Technol. 2019, 342, 780–788. [CrossRef]
- Almoselhy, R.I.M. Applications of Differential Scanning Calorimetry (DSC) in Oils and Fats Research. A Review. Am. Res. J. Agric. 2020, 6, 1–9. [CrossRef]
- Bonnaud, P.A.; Ji, Q.; Van Vliet, K.J. Effects of Elevated Temperature on the Structure and Properties of Calcium-Silicate-Hydrate Gels: The Role of Confined Water. Soft Matter 2013, 9, 6418–6429. [CrossRef]
- Rowe, R. C., Sheskey, P. J., & Quinn, M.E. Handbook Pharmaceutical Excepients (6th Ed.); by the Pharmaceutical Press, 2009.
- Rafiu, R. Khulna University of Engineering and Technology Department of Materials Science and Engineering Course Title Materials Characterization Sessional X-Ray Diffraction ( XRD ) Analysis Name : Rifat Rafiu. 2024, 0–7. [CrossRef]
- Ali, A.; Chiang, Y.W.; Santos, R.M. X-Ray Diffraction Techniques for Mineral Characterization: A Review for Engineers of the Fundamentals, Applications, and Research Directions. Minerals 2022, 12. [CrossRef]
- Ulbrich, M.; Bültena, M.; Braun, B.; Meißner, K.; Bussert, R.; Flöter, E. Specific Modification of Granular Potato Starch by Means of Partial Debranching Using Pullulanase. Starch/Staerke 2023, 2300154, 1–14. [CrossRef]
- El-Naggar, N.E.A.; El-Malkey, S.E.; Abu-Saied, M.A.; Mohammed, A.B.A. Exploration of a Novel and Efficient Source for Production of Bacterial Nanocellulose, Bioprocess Optimization and Characterization. Sci. Rep. 2022, 12, 1–22. [CrossRef]
- Velarde, L.; Nabavi, M.S.; Escalera, E.; Antti, M.L.; Akhtar, F. Adsorption of Heavy Metals on Natural Zeolites: A Review. Chemosphere 2023, 328, 138508. [CrossRef]
- Marguí, E.; Queralt, I. Sample Preparation for X-Ray Fluorescence Analysis. Encycl. Anal. Chem. 2024, 1–29. [CrossRef]
- Icp, S.; Emission, O. Agilent 700 Series ICP Optical Emission Spectrometers User’s Guide 2 Agilent 700 Series ICP Optical Emission Spectrometers User’s Guide Notices Manual Part Number Instrument Manufacturing. 2018.
- Abdelfattah, D.S.E.; Fouad, M.A.; Elmeshad, A.N.; El-Nabarawi, M.A.; Elhabal, S.F. Formulation and Stability Evaluation of Anti-Obesity Nutraceuitcal Blend of White Kidney Bean Extract (Phaseolus Vulgaris L.), Propolis Ethanolic Extract and Crpic3. Int. J. Appl. Pharm. 2024, 16, 225–235. [CrossRef]
- In Republic of Indonesia Ministry of Health Indonesian Pharmacopoeia Edition V; Ministry of Health of the Republic of Indonesia: Jakarta, 2014; 60. Republic of Indonesia Ministry of Health Indonesian Pharmacopoeia Edition V; Ministry of Health of the Republic of Indonesia: Jakarta, 2014.
- Kholodenko, A.L. Applications of Contact Geometry and Topology in Physics. Appl. Contact Geom. Topol. Phys. 2013, 02, 1–475. [CrossRef]
- Leyva-Porras, C.; Cruz-Alcantar, P.; Espinosa-Sol, V.; Saavedra-Leos, M.Z. Application of Differential Scanning Calorimetry (DSC) and Modulated Differential Scanning. Polymers (Basel). 2019, 12, 1–21.
- Ermrich, M.; Opper, D. X-Ray Powder Diffraction for the Analyst; 2011; ISBN 6754-6762.
- Mohammed; A. Abdullah Scanning Electron Microscopy (SEM): A Review. Proc. 2018 Int. Conf. Hydraul. Pneum. - HERVEX 2018, 77–85.




| Parameters | Unit | Samples | Drinking Water Quality Standards | Seawater Quality Standards | Reference(s) | |||
| Grow up | Market Product | Marine Life | Port Facilities | Marine Tourism | ||||
| Color | Pt/Co Scale | 333 | 570 | 15 | - | - | 30 | [16,17] |
| Odor | Slightly Odorous | Slightly Odorous | Odorless | Natural | Odorless | Odorless | [16,17] | |
| Total Suspended Solids (TSS) | m/L | < 7.7 | < 7.7 | 40 | Coral = 20 Mangrove = 80 Seagrass = 20 |
80 | 20 | [16,17] |
| Parameters | Unit | Samples | Drinking Water Quality Standards | Seawater Quality Standards | Reference(s) | |||
| Grow up | Market Products | Marine Life | Port Facilities | Marine Tourism | ||||
| pH | Pt/Co Scale | 5.3 | 6.6 | 6–9 | 7–8.5 | 6.5–8.5 | 7–8.5 | [16,17] |
| Salinity | % | 311 | 283 | 33–34 | Natural Coral = 33–34 Mangrove = 33–34 Seagrass = 33–34 |
Natural | Natural | [16] |
| Dissolved Oxygen (DO) | mg/L | 2.1 | 3.1 | 6 | > 5 | - | > 5 | [16,17] |
| Biological Oxygen Demand (BOD) | mg/L | 304 | - | 6 | 20 | - | 10 | [16,17] |
| Ammonia | mg/L | 0.77 | 1.2 | 1.5 | 0.3 | 0.3 | - | [16,18] |
| Phosphate | mg/L | 0.096 | 0.23 | 0.2 | 0.015 | 0.015 | [16,17] | |
| Nitrate | mg/L | 0.084 | 0.097 | 10 | 0.008 | - | 0.008 | [16,17] |
| Nitrite | mg/L | 0.0047 | < 0.004 | 0.06 | 0.06 | 0.06 | 0.06 | [16,17] |
| Cyanide | mg/L | 0.011 | 0.005 | 0.02 | 0.5 | - | - | [16,17] |
| Sulfide | mg/L | 0.011 | 0.015 | 0.05 | 0.01 | 0.03 | 0 | [16,19] |
| Total Hydrocarbons | mg/L | < 0.79 | - | 10 | - | 1 | - | [16,20] |
| Total Phenolic Compounds | mg/L | 0.0016 | - | 0.002 | 0.002 | 0.002 | 0 | [16,17] |
| Surfactants | MBAS | 1.8 | 2.9 | 0.2 | 1 | 1 | 0.001 | [16,17] |
| Oil and fat | mg/L | <0,11 | - | 1 | 1 | 5 | 1 | [16,17] |
| Parameters | Quality Standard Unit | Sample | Drinking Water Quality Standards | Seawater Quality Standards | Reference(s) | |||
| Grow up | Market Products | Marine Life | Port Facilities | Marine Tourism | ||||
| Fecal Coliform | Total amount per 100 ml sample (CFU) | < 10 | < 10 | 0 | - | 200 | [16,17] | |
| Total Coliform | Total amount per 100 ml sample (CFU) | 14 | <10 | 0 | 1.000 | 1000 | 1000 | [16,17] |
| Essential Minerals | ||||
| Elements | Samples (mg/l) | Market Product (mg/l) | ||
| XRF | ICP-OES | XRF | ICP-OES | |
| Mg | 138000 | 61817.1466 | 53200 | 1656.2590 |
| Cl | 626000 | * | 160000 | * |
| K | 82300 | 72888.7852 | 21500 | 172141.0630 |
| Ca | 8370 | 527.9098 | 0 | 94.4760 |
| On | 0 | 350.7511 | 0 | 643.0545 |
| B | 0 | 110.1492 | 0 | 154.7290 |
| Cr | 0 | < 0.0001 | 2.56 | < 0.0001 |
| With | 18.0 | < 0.0001 | 7.46 | < 0.0001 |
| Fe | 26.1 | < 0.0001 | 0 | 0.0095 |
| Mn | 0 | < 0.0001 | 0 | 4.3815 |
| Zn | 0 | < 0.0001 | 0 | 1.0785 |
| Toxic Minerals | ||||
| To the | 0 | < 0.0001 | 0 | 6.4350 |
| In | 0 | * | 0 | * |
| Tl | 0 | < 0.0001 | 0 | < 0.0001 |
| As | 0 | < 0.0001 | 0 | < 0.0001 |
| Hg | 0 | < 0.0001 | 0 | < 0.0001 |
| Cd | 0 | < 0.0001 | 0 | < 0.0001 |
| Pb | 0 | < 0.0001 | 0 | < 0.0001 |
| Non-essential and Non-toxic Minerals | ||||
| S | 79000 | * | 20700 | * |
| Br | 19100 | * | 3010 | * |
| Yes | 5340 | * | 1870 | * |
| Rb | 790 | * | 49.9 | * |
| Sn | 325 | <0.0001 | 78.6 | <0.0001 |
| Ta | 31.2 | * | 11.4 | * |
| In | 26 | <0.0001 | 0 | <0.0001 |
| No | Formula | Filler | Desiccant | Binder (%) |
|---|---|---|---|---|
| 1 | G-AV | Avicel® PH 102 | - | Amylum 10 |
| 2 | G-Av- Fl | Avicel® PH 102 | Floritis® | Amylum 10 |
| 3 | G-Lact | Lactose | - | Amylum 10 |
| 4 | G-Lact-Fl | Lactose | Floritis® | Amylum 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




