Submitted:
18 December 2024
Posted:
19 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Methods
2.1. Materials
2.2. Production of Biochar
2.3. Preparation of Magnetic Biochar
2.4. Preparation of Adsorbent Beads
- Mixing: First, 4 g of sodium alginate powder was thoroughly mixed with 4 g of the various materials (including unmodified banana peel, activated carbon, and magnetic activated carbon) in a weigh boat. The resulting mixed powder was slowly added to 400 mL of deionized water while continuously stirring. The mixture was left in the beaker overnight to ensure homogeneity.
- Calcium Chloride Solution: Next, a 2 wt% solution of calcium chloride was prepared by dissolving calcium chloride in 400 mL of deionized water. This solution was maintained at room temperature on a hot plate.
- Drop-Wise Addition: While the calcium chloride solution was magnetically stirred, the various material solutions were passed through a pear-shaped separating funnel and added dropwise to the beaker.
- Gelation Process: Following the gelation process, the beads formed were subjected to gentle magnetic stirring at 350 rpm for 24 hours in their mother liquor.
- Harvesting and Drying: Finally, the beads were harvested by filtering the calcium chloride solution and washing them several times with deionized water. Subsequently, the beads were dried at room temperature in a fume cupboard.
2.5. Characterization
2.6. Adsorption Trials
2.7. Initial Concentration Effect and Isotherm Modelling
- Isotherm Modelling
2.8. Re-Use of the Beads
3. Results
3.1. Characterization Results
- 2.
- Ultimate analysis
- 3.
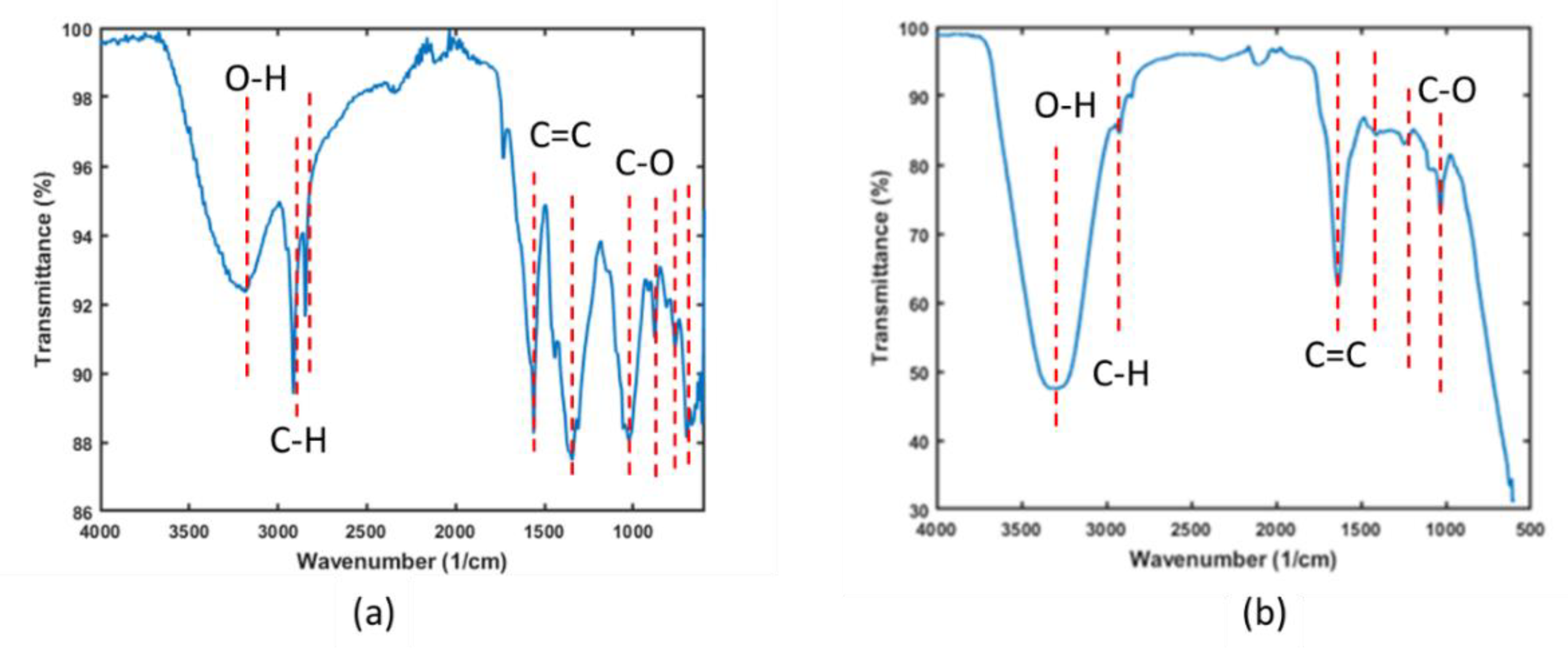
- Analysis of functional groups through FTIR
- 4.
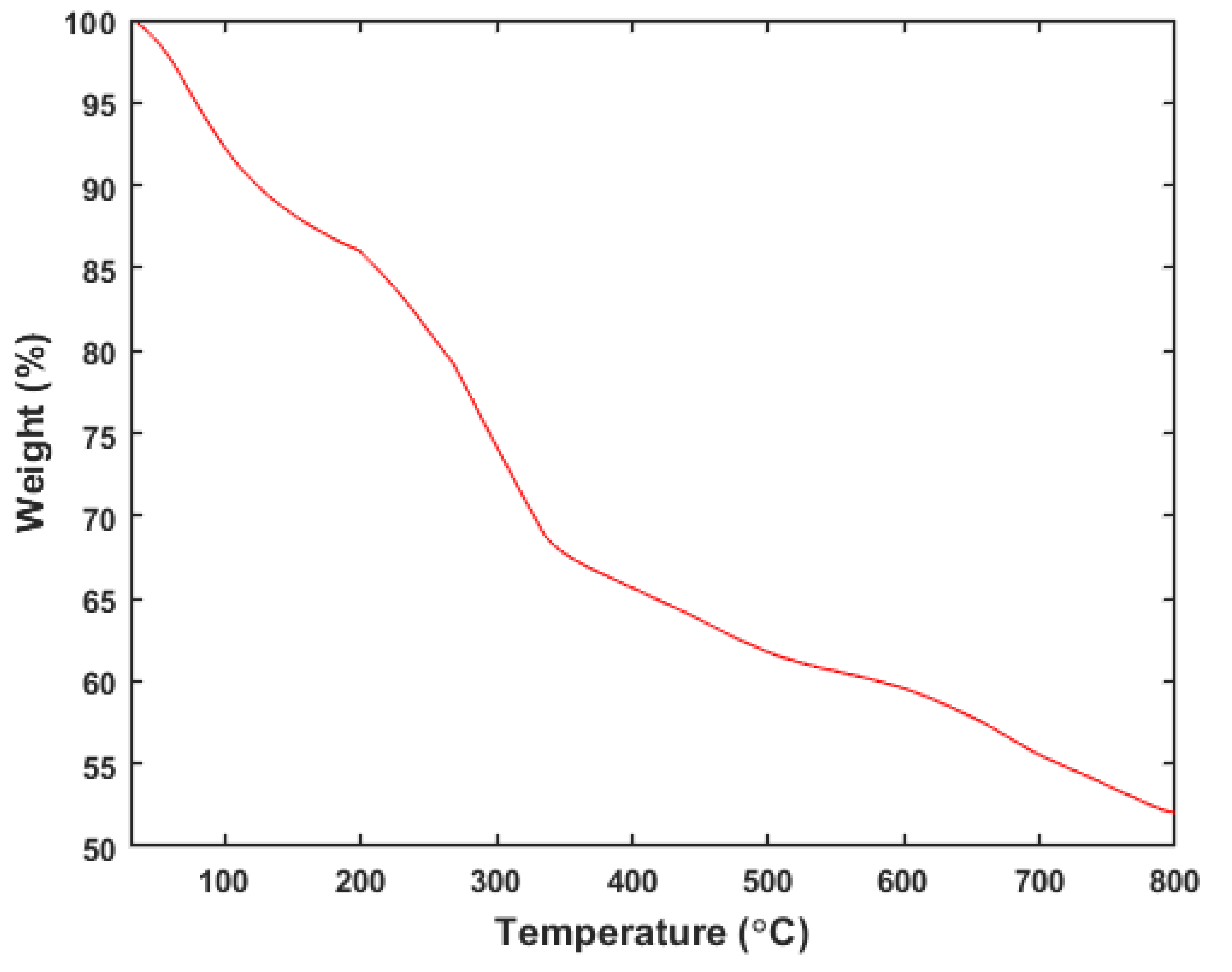
- Thermogravimetric analysis
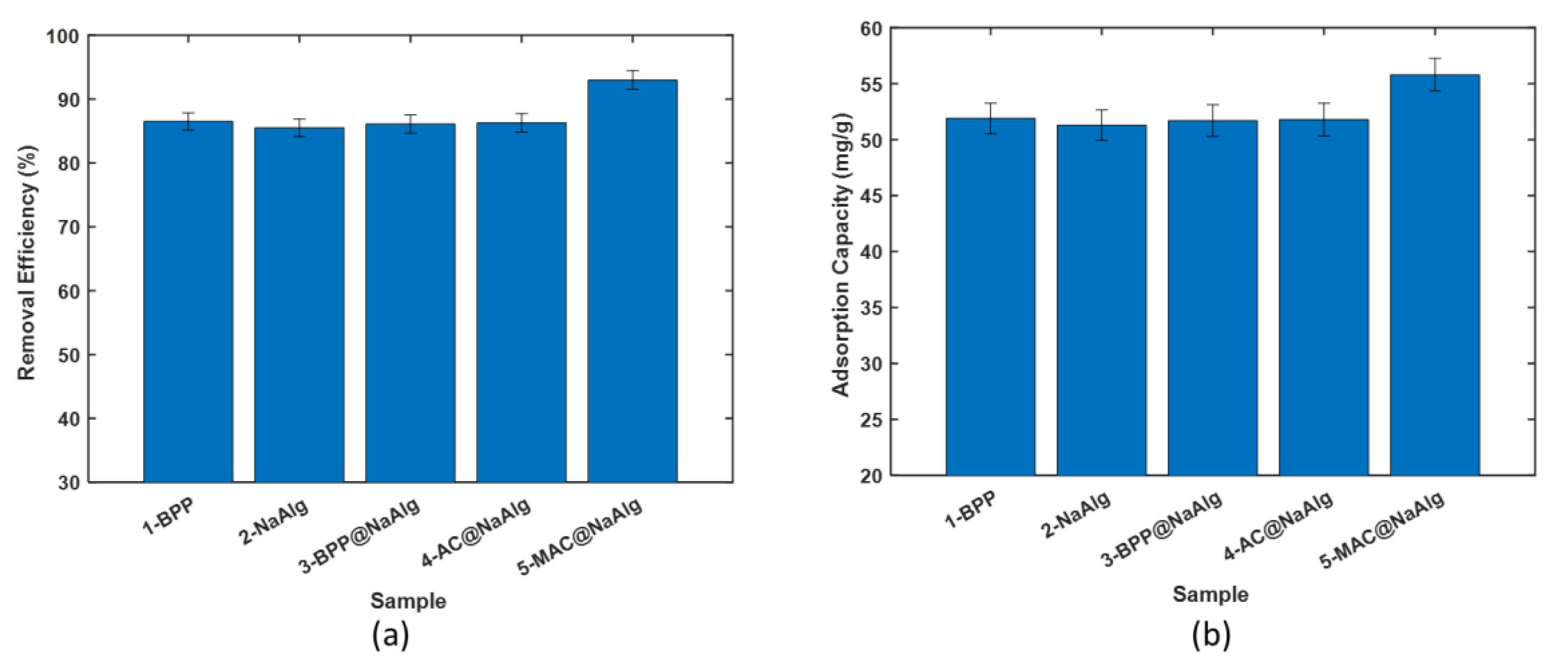
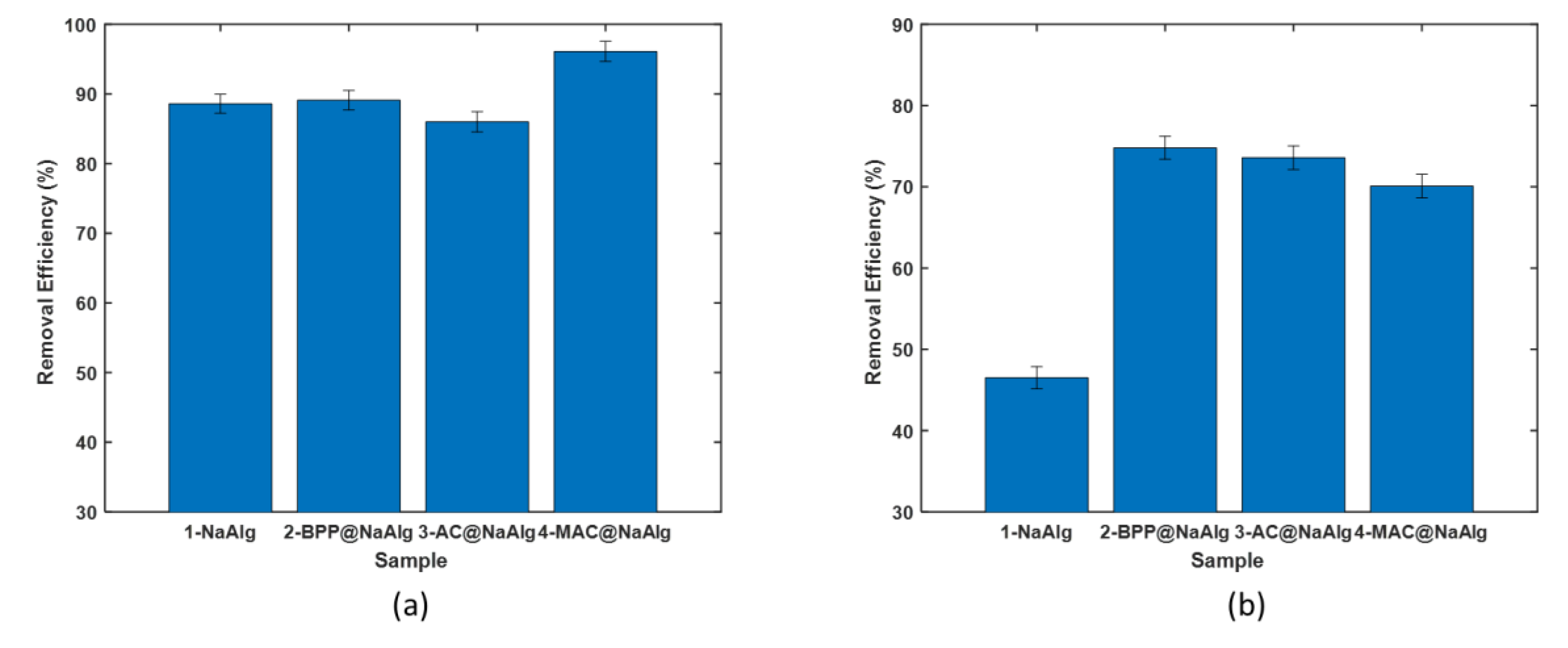
3.2. Comparison of Adsorption Performance
- 6.
- Removal of Cr(VI) with the virgin beads
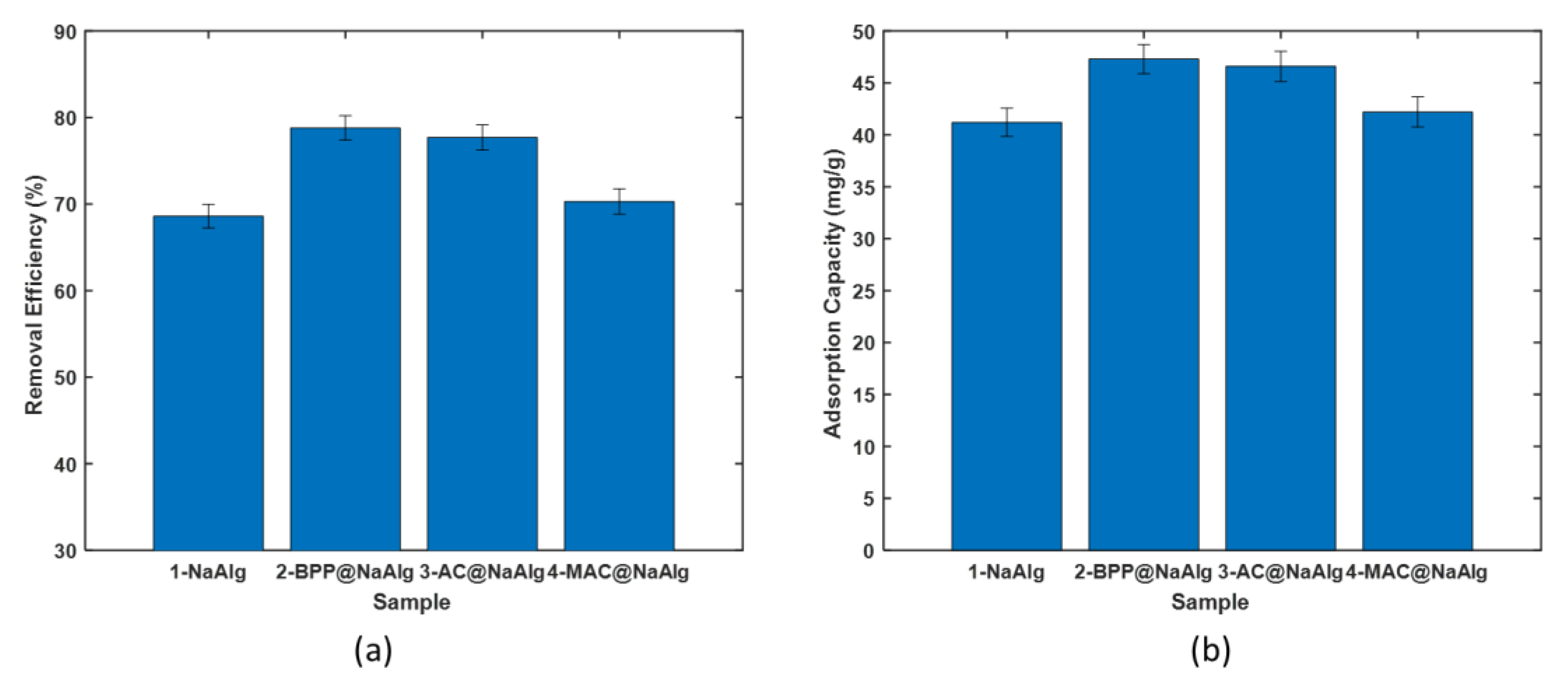
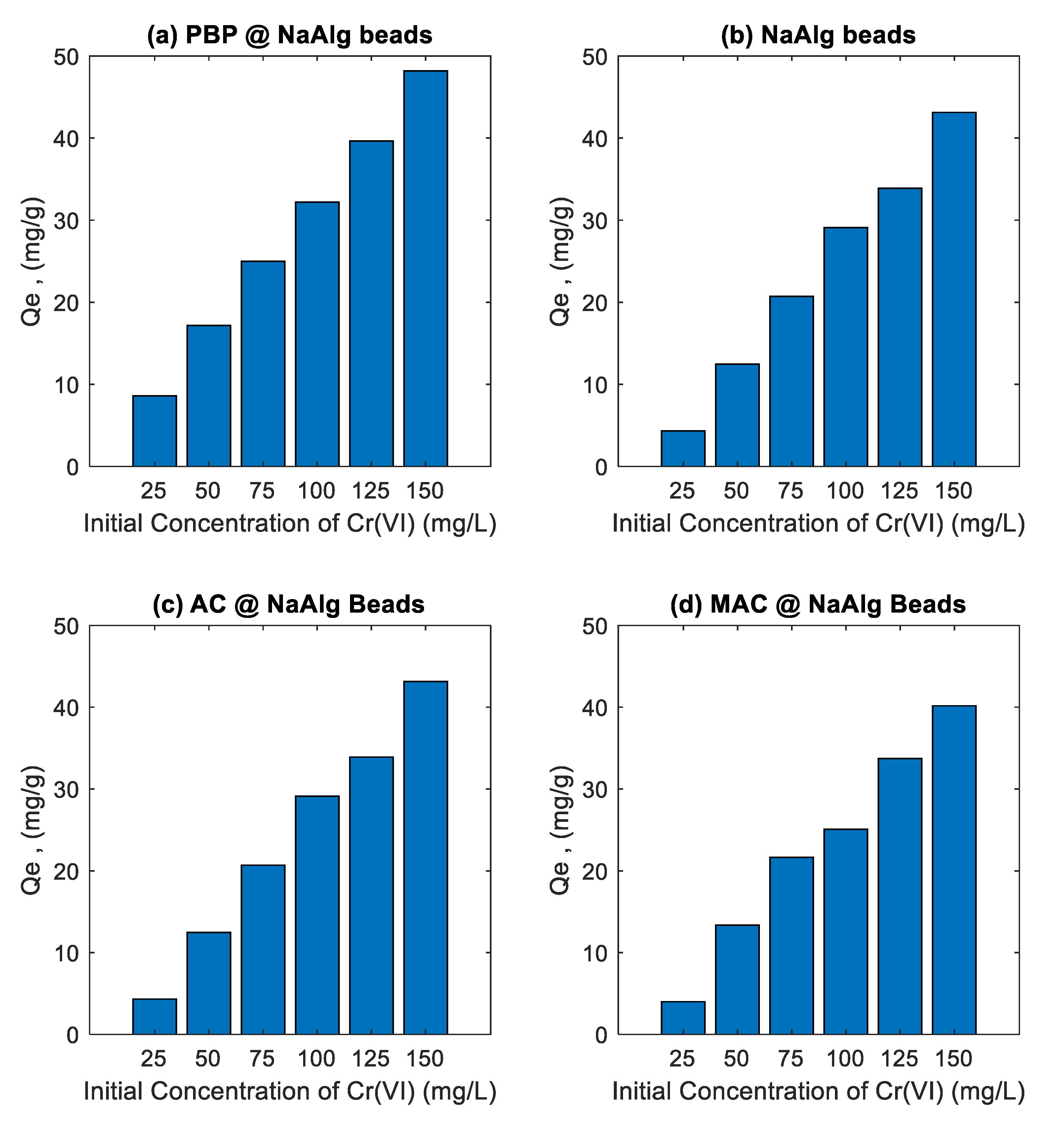
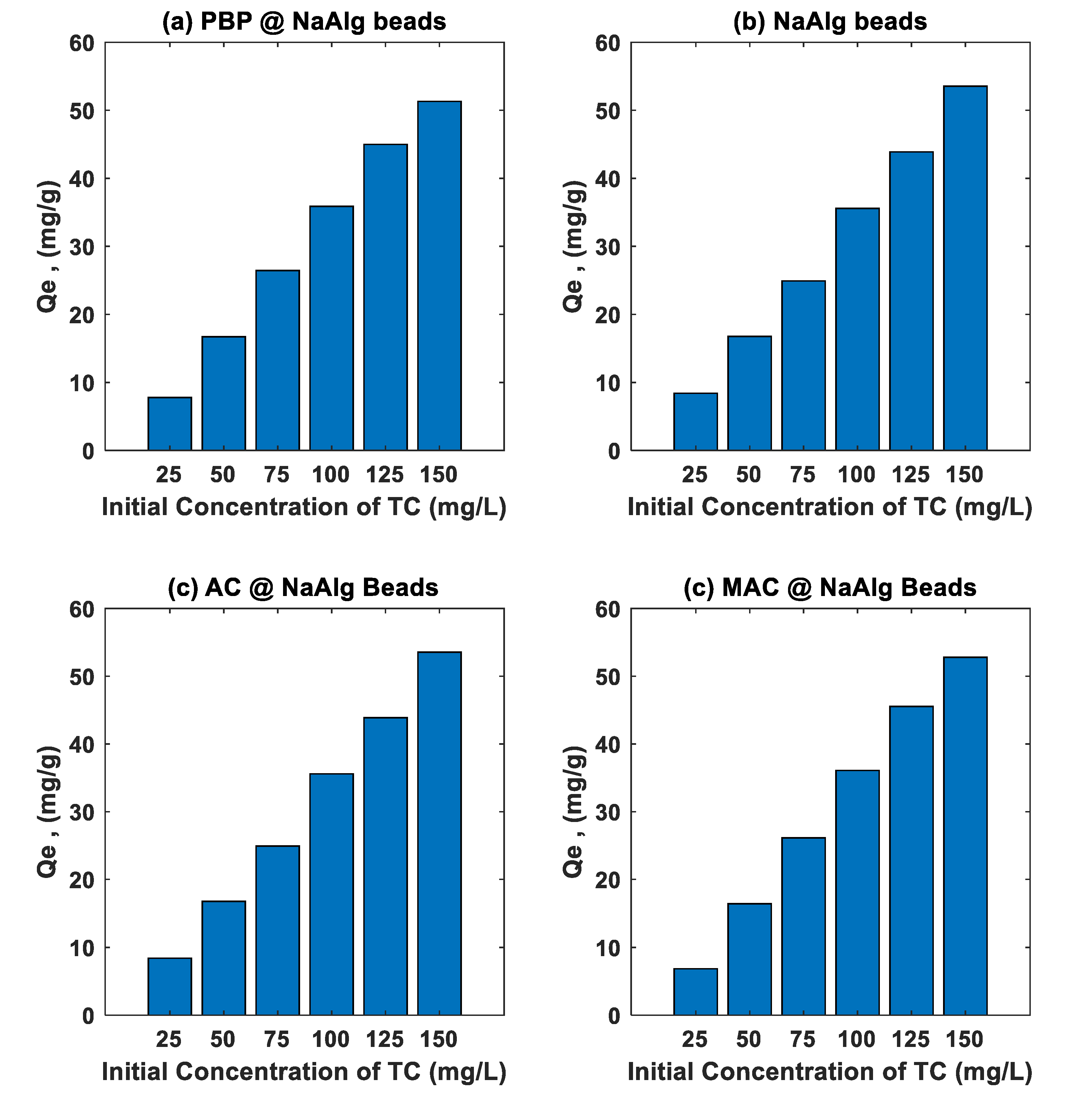
3.3. Effect of Initial Concentration Study
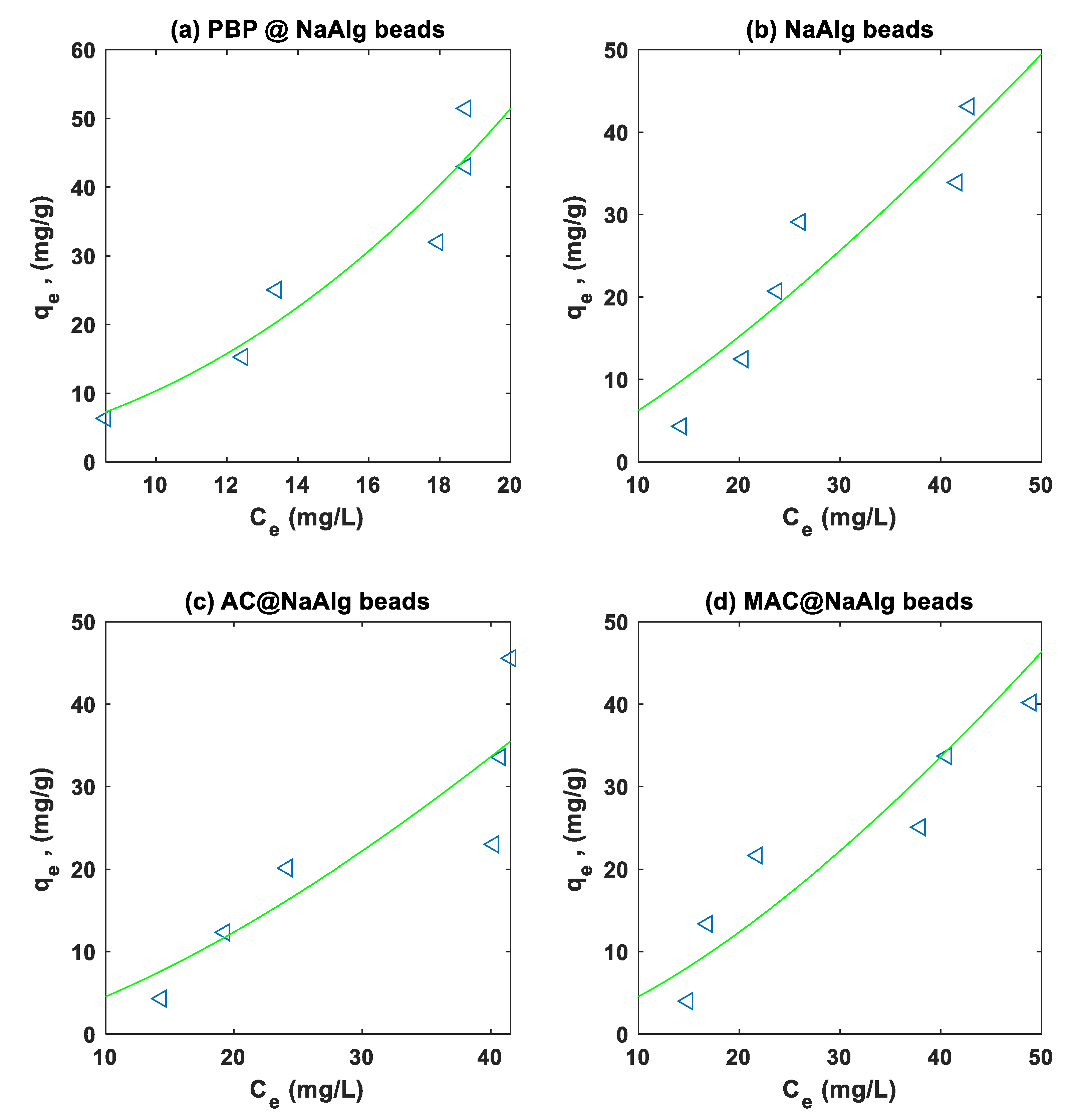
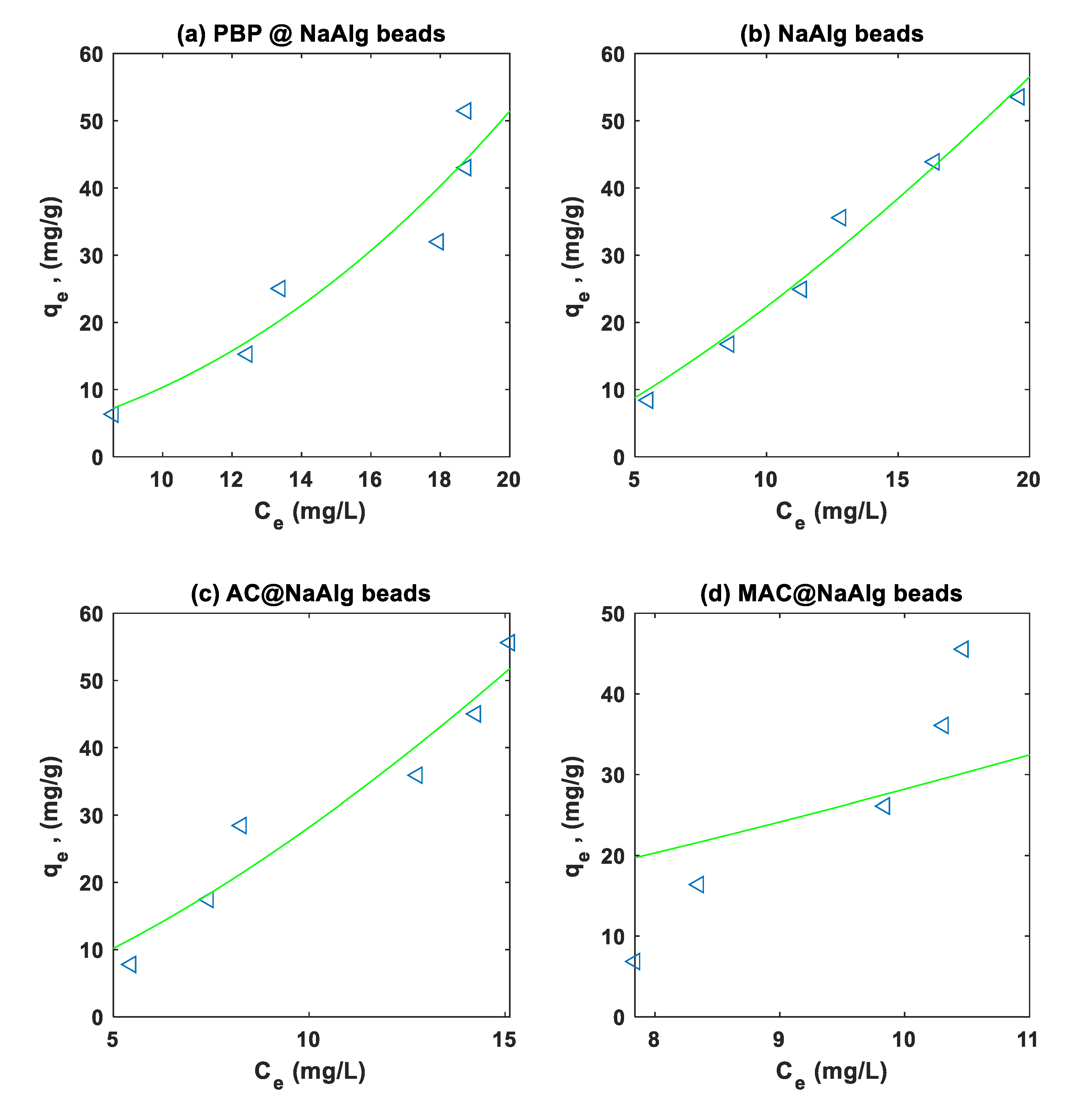
3.4. Isotherm Modelling
3.5. Reuse of the Beads for Successive Adsorption
3.6. Comparison of Adsorption with Other Materials
| Material | Removal Capacity (mg/g) | Ref. |
|---|---|---|
| Fe3O4@Alg-Ce | 14.29 | [37] |
| Chitosan beads | 3.23 | [38] |
| Polyvinyl alcohol-alginate beads | 0.17 | [39] |
| sodium alginate composite (PPS) | 83.1 | [40] |
| PBP@NaAlg | 47.3 | This study |
| AC@NaAlg | 46.6 | This study |
| MAC@ NaAlg | 42.2 | This study |
| NaAlg` | 41.2 | This study |
4. Conclusions
References
- Ahmed, M.B. , et al., Adsorptive removal of antibiotics from water and wastewater: Progress and challenges. Science of The Total Environment, 2015. 532: p. 112-126.
- Battak, N. , et al., Antibiotics usage in plant agriculture and its removal from wastewater using adsorption process: A short review. AIP Conference Proceedings, 2022. 2610(1).
- Sen, U. , et al., Removal of Antibiotics by Biochars: A Critical Review. Applied Sciences, 2023. 13(21): p. 11963.
- Ahmad, T. and M. Danish, Prospects of banana waste utilization in wastewater treatment: A review. Journal of Environmental Management, 2018. 206: p. 330-348.
- Nguyen, V.-T. , et al., Preliminary study of doxycycline adsorption from aqueous solution on alkaline modified biochar derived from banana peel. Environmental Engineering Research, 2024. 29(3): p. 230196-0.
- Azzam, A.B. , et al., Heterogeneous porous biochar-supported nano NiFe2O4 for efficient removal of hazardous antibiotic from pharmaceutical wastewater. Environmental Science and Pollution Research, 2023. 30(56): p. 119473-119490.
- Akpomie, K.G. and J. Conradie, Banana peel as a biosorbent for the decontamination of water pollutants. A review. Environmental Chemistry Letters, 2020. 18(4): p. 1085-1112.
- Negroiu, M. , et al., Novel Adsorbent Based on Banana Peel Waste for Removal of Heavy Metal Ions from Synthetic Solutions. Materials, 2021. 14(14): p. 3946.
- Huang, Z., R. Campbell, and C. Mangwandi, Kinetics and Thermodynamics Study on Removal of Cr(VI) from Aqueous Solutions Using Acid-Modified Banana Peel (ABP) Adsorbents. Molecules, 2024. 29(5): p. 990.
- Nadew, T.T. , et al., Synthesis of activated carbon from banana peels for dye removal of an aqueous solution in textile industries: optimization, kinetics, and isotherm aspects. Water Practice and Technology, 2023. 18(4): p. 947-966.
- Al-sareji, O.J. , et al., A Sustainable Banana Peel Activated Carbon for Removing Pharmaceutical Pollutants from Different Waters: Production, Characterization, and Application. 2024. 17(5): p. 1032.
- Azzam, A.B. , et al., Construction of porous biochar decorated with NiS for the removal of ciprofloxacin antibiotic from pharmaceutical wastewaters. Journal of Water Process Engineering, 2022. 49: p. 103006.
- Vo, T.D.H., V. T. Nguyen, and T.H. Nguyen, Removal of doxycycline antibiotics in water with adsorbents derived from banana peels. Science & Technology Development Journal: Science of the Earth & Environment, 2023. 7(2): p. 728-740.
- Ajala, O.A. , et al., Adsorptive removal of antibiotic pollutants from wastewater using biomass/biochar-based adsorbents. RSC Advances, 2023. 13(7): p. 4678-4712.
- Lam, S.S. , et al., Pyrolysis production of fruit peel biochar for potential use in treatment of palm oil mill effluent. Journal of Environmental Management, 2018. 213: p. 400-408.
- Rengga, W.D.P. , et al., Kinetic Study of Adsorption Carboxylic Acids of Used Cooking Oil Using Mesoporous Active Carbon.” IOP Conference Series: Earth and Environmental Science 2021. 700(1).
- Malakar, B., D. Das, and K. Mohanty, Evaluation of banana peel hydrolysate as alternate and cheaper growth medium for growth of microalgae Chlorella sorokiniana. Biomass Conversion and Biorefinery, 2024. 14(9): p. 10361-10371.
- Szcześniak, L., A. Rachocki, and J. Tritt-Goc, Glass transition temperature and thermal decomposition of cellulose powder. Cellulose, 2008. 15(3): p. 445-451.
- Börcsök, Z. and Z. Pásztory, The role of lignin in wood working processes using elevated temperatures: an abbreviated literature survey. European Journal of Wood and Wood Products, 2021. 79(3): p. 511-526.
- Cai, Y. , et al., Adsorption and Desorption Performance and Mechanism of Tetracycline Hydrochloride by Activated Carbon-Based Adsorbents Derived from Sugar Cane Bagasse Activated with ZnCl2. Molecules, 2019. 24(24): p. 4534.
- Long, J. , et al., Preparation of Oily Sludge-Derived Activated Carbon and Its Adsorption Performance for Tetracycline Hydrochloride. Molecules, 2024. 29(4): p. 769.
- Sağlam, S., F. N. Türk, and H. Arslanoğlu, Tetracycline (TC) removal from wastewater with activated carbon (AC) obtained from waste grape marc: activated carbon characterization and adsorption mechanism. Environmental Science and Pollution Research, 2024. 31(23): p. 33904-33923.
- Vedenyapina, M.D. , et al., Activated Carbon as Sorbents for Treatment of Pharmaceutical Wastewater (Review). Solid Fuel Chemistry, 2019. 53(6): p. 382-394.
- Bhatnagar, A. and M. Sillanpää, Utilization of agro-industrial and municipal waste materials as potential adsorbents for water treatment—A review. Chemical Engineering Journal, 2010. 157(2): p. 277-296.
- Chen, S. , et al., Removal of Cr(VI) from aqueous solution using modified corn stalks: Characteristic, equilibrium, kinetic and thermodynamic study. Chemical Engineering Journal, 2011. 168(2): p. 909-917.
- Wu, J. , et al., Upcycling tea waste particles into magnetic adsorbent materials for removal of Cr(VI) from aqueous solutions. Particuology, 2023. 80: p. 115-126.
- Mangwandi, C., H. Chen, and J. Wu, Production and optimisation of adsorbent materials from teawaste for heavy metal removal from aqueous solution: feasibility of hexavalent chromium removal – kinetics, thermodynamics and isotherm study. Water Practice and Technology, 2022. 17(9).
- Rai, M.K. , et al., Removal of hexavalent chromium Cr (VI) using activated carbon prepared from mango kernel activated with H3PO4. Resource-Efficient Technologies, 2016. 2: p. S63-S70.
- Duan, J. , et al., Enhanced adsorption of Cr(VI) from aqueous solutions by CTAB-modified schwertmannite: Adsorption performance and mechanism. Chemical Engineering Research and Design, 2024. 208: p. 464-474.
- Wang, J. and S. Wang, Removal of pharmaceuticals and personal care products (PPCPs) from wastewater: A review. Journal of Environmental Management, 2016. 182: p. 620-640.
- Tran, H.N., S. -J. You, and H.-P. Chao, Effect of pyrolysis temperatures and times on the adsorption of cadmium onto orange peel derived biochar. Waste Management & Research, 2016. 34(2): p. 129-138.
- Yu, F. , et al., Adsorptive removal of antibiotics from aqueous solution using carbon materials. Chemosphere, 2016. 153: p. 365-385.
- Gao, Y. , et al., Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. Journal of Colloid and Interface Science, 2012. 368(1): p. 540-546.
- Barbooti, M.M. and S. H. Zahraw. REMOVAL OF TETRACYCLINE FROM WATER BY ADSORPTION ON WATER TREATMENT RESIDUES. 2020.
- Başakçılardan-Kabakcı, S. , et al., Adsorption and Precipitation of Tetracycline with Struvite. Water Environment Research, 2007. 79(13): p. 2551-2556.
- Goyal, S. and R. Auchat, ADSORPTIVE REMOVAL OF TETRACYCLINE ANTIBIOTIC FROM AQUEOUS SOLUTION USING MAGNETIC NANOCOMPOSITES AS ADSORBENT. Journal of Advenced Scientific Research, 2022. 13(2).
- Gopalakannan, V. and N. Viswanathan, Synthesis of magnetic alginate hybrid beads for efficient chromium (VI) removal. International Journal of Biological Macromolecules, 2015. 72: p. 862-867.
- Du, X. , et al., Removal of Chromium(VI) by Chitosan Beads Modified with Sodium Dodecyl Sulfate (SDS). Applied Sciences, 2020. 10(14): p. 4745.
- Lee Te Chuan, et al., Polyvinyl Alcohol-Alginate Adsorbent Beads for Chromium (VI) Removal. international Journal of Engineering & Technology, 2018. 7 (20).
- Li, Y. , et al., Removal of Cr(VI) by polyaniline embedded polyvinyl alcohol/sodium alginate beads − Extension from water treatment to soil remediation. Journal of Hazardous Materials, 2022. 426: p. 127809.
- Abed, M.F. and A.A.H. Faisal, Calcium/iron-layered double hydroxides-sodium alginate for removal of tetracycline antibiotic from aqueous solution. Alexandria Engineering Journal, 2023. 63: p. 127-142.
- Ranjbari, S. , et al., Efficient tetracycline adsorptive removal using tricaprylmethylammonium chloride conjugated chitosan hydrogel beads: Mechanism, kinetic, isotherms and thermodynamic study. International Journal of Biological Macromolecules, 2020. 155: p. 421-429.
- Jaiyeola, O.O., H. Annath, and C. Mangwandi, Synthesis and evaluation of a new CeO2@starch nanocomposite particles for efficient removal of toxic Cr(VI) ions. Energy Nexus, 2023. 12: p. 100244.
- Göçenoğlu Sarıkaya, A. and B. Osman, Tetracycline adsorption via dye-attached polymeric microbeads. Cumhuriyet Science Journal, 2021. 42(3): p. 638-648.
- Liao, Q. , et al., Strong adsorption properties and mechanism of action with regard to tetracycline adsorption of double-network polyvinyl alcohol-copper alginate gel beads. Journal of Hazardous Materials, 2022. 422: p. 126863.
| Material | Element Composition (%) | |||
|---|---|---|---|---|
| C | H | N | S | |
| Banana Peel Powder | 39.4 | 6.4 | 1.1 | <0.3 |
| AC@NaAlg Beads | 24.7 | 4.5 | <0.3 | <0.3 |
| MAC@NaAlg Beads | 19.9 | 3.8 | <0.3 | <0.3 |
| Adsorbate | Model | Parameter | Bead Type | |||
|---|---|---|---|---|---|---|
| NaAlg | PBP@NaAlg | AC@NaAlg | MAC@NaAlg | |||
| Cr(VI) |
Langmuir |
6.740e-05 | 0.0184 | 1.4529e-05 | 0.0113 | |
| 1.232e+04 | 130.8496 | 6.3920e+04 | 155.5158 | |||
| 0.7693 | 0.9785 | 0.8265 | 0.9619 | |||
| 6.6740 | 2.3926 | 9.1146 | 2.8778 | |||
|
Freundlich |
0.0501 | 3.4473 | 0.0722 | 2.2068 | ||
| 0.4321 | 1.2988 | 0.5772 | 1.1745 | |||
| 0.8983 | 0.9835 | 0.9855 | 0.9620 | |||
| 6.0251 | 2.0927 | 2.6307 | 2.8723 | |||
|
TC |
Langmuir |
8.108e-05 | 2.0569e-04 | 1.6807e-05 | 3.6901e-05 | |
| 3.174e+04 | 1.3676e+04 | 6.9141e+04 | 8.7443e+04 | |||
| 0.9315 | 0.8300 | 0.8938 | 0.6215 | |||
| 4.9718 | 7.6993 | 7.3638 | 12.1199 | |||
|
Freundlich |
0.8087 | 0.1667 | 0.2842 | 0.004 | ||
| 0.7067 | 0.4690 | 0.7165 | 0.2018 | |||
| 0.9972 | 0.9947 | 0.9498 | 0.9430 | |||
| 1.1586 | 1.4203 | 5.0605 | 3.7811 | |||
| Material | Removal Capacity (mg/g) | Ref. |
|---|---|---|
| Calcium/iron-layered double hydroxides | 10.393 | [41] |
| Ionic liquid-impregnated chitosan hydrogel beads | 22.42 | [42] |
| CeO2@starch nano composite particles | 48.54 | [43] |
| Dye-attached polymeric microbeads | 9.63 | [44] |
| Double-network polyvinyl alcohol-copper alginate gel beads | 231.43 | [45] |
| PBP@NaAlg | 51.3 | This study |
| AC@NaAlg | 51.8 | This study |
| MAC@ NaAlg | 55.8 | This study |
| NaAlg | 51.9 | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




