Submitted:
13 December 2024
Posted:
13 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Oligonucleotide Transfection
2.3. RT-qPCR
2.4. Cytoplasmic and Nuclear Fraction Extraction
2.5. Immunoblot Analysis
2.6. Immunocytochemistry
2.7. Cell Proliferation Assay
2.8. Cell Viability
2.10. Flow Cytometry
2.11. Statistical Analysis
3. Results
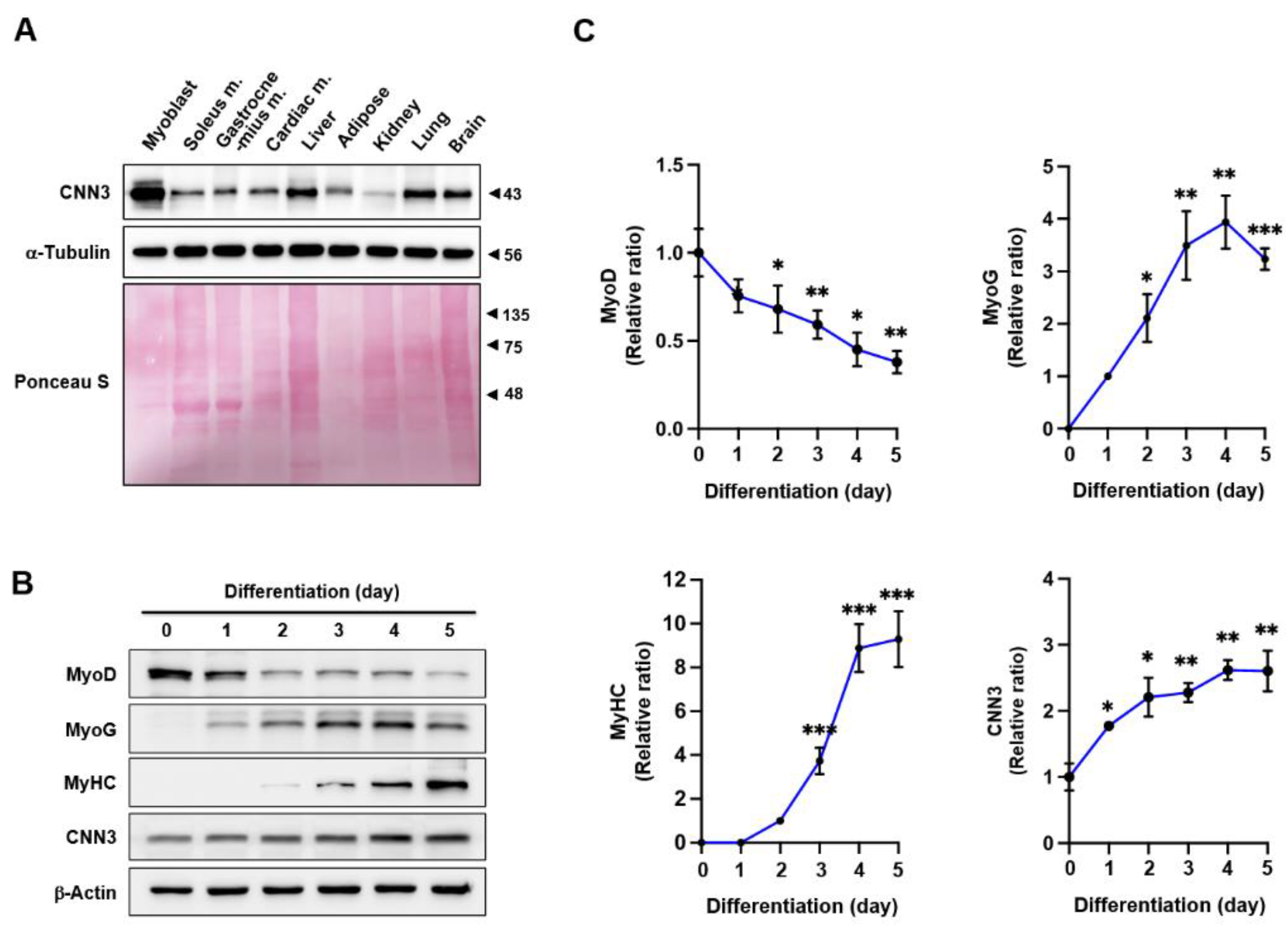
3.1. CNN3 Expression Increased During Myoblast Differentiation
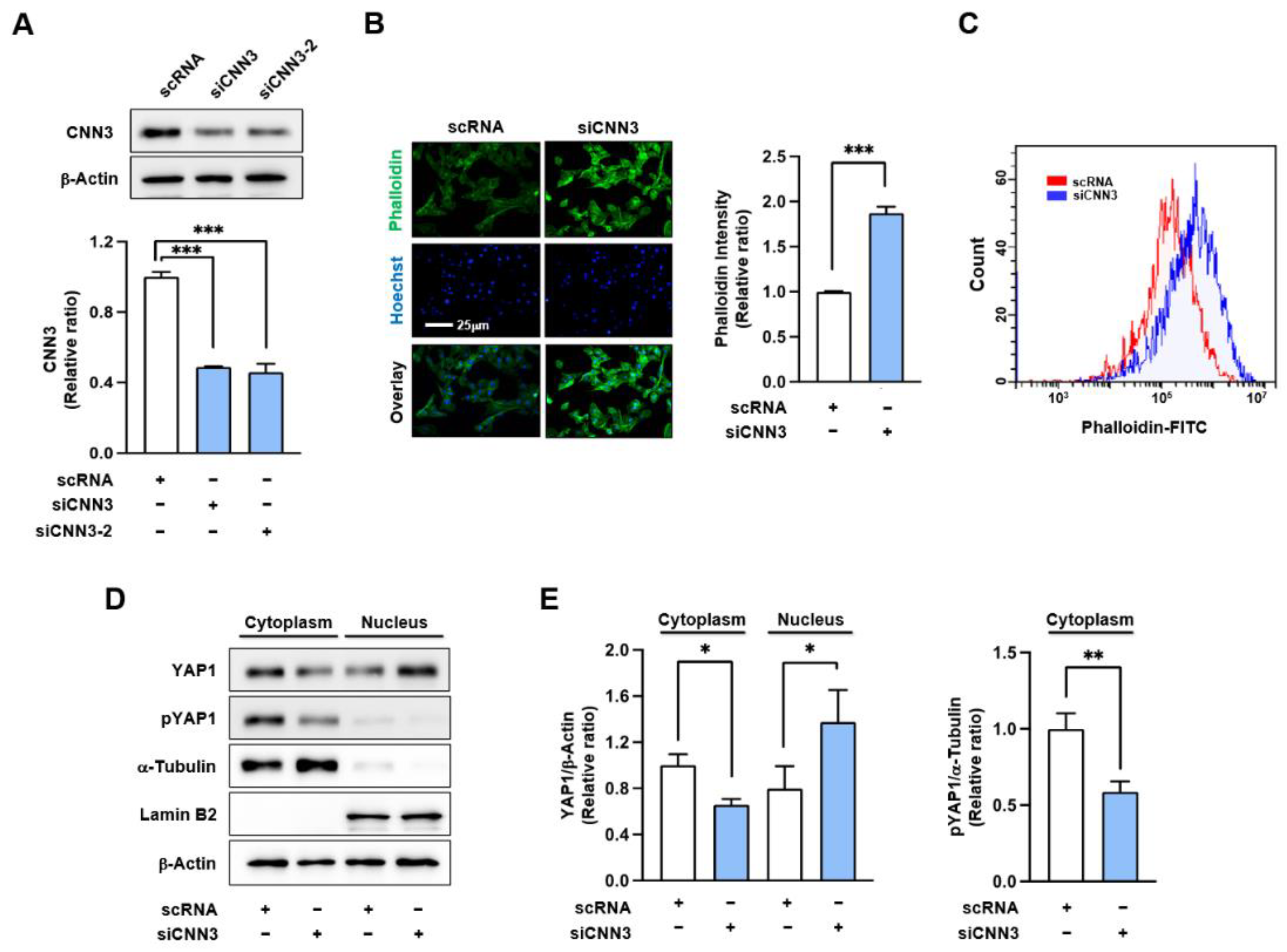
3.2. CNN3 Knockdown Enhanced F-Actin and Nuclear YAP1 Levels in Myoblasts
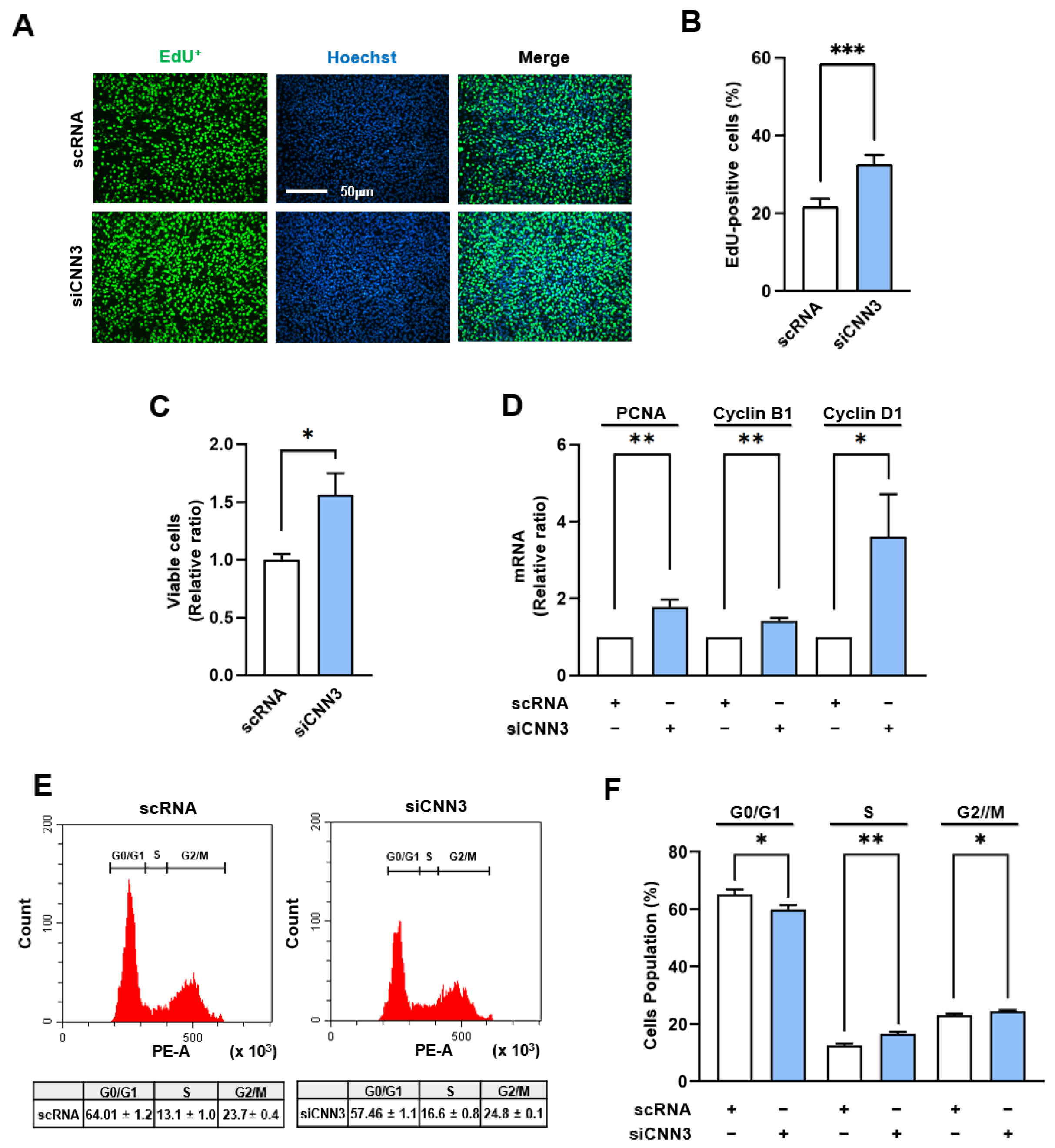
3.3. CNN3 Knockdown Increased YAP1 Target Gene Expressions and Induced Myoblast Proliferation
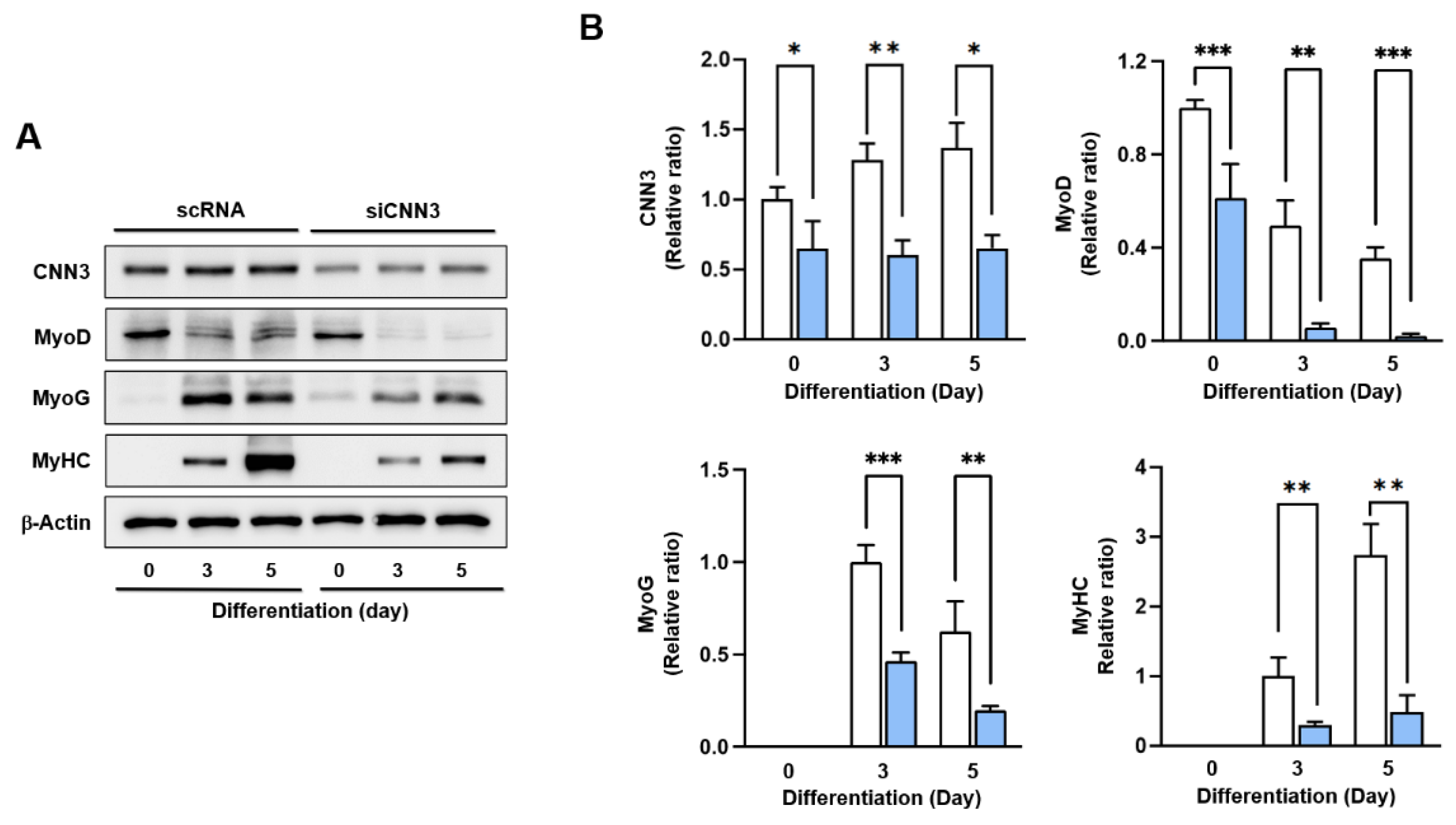
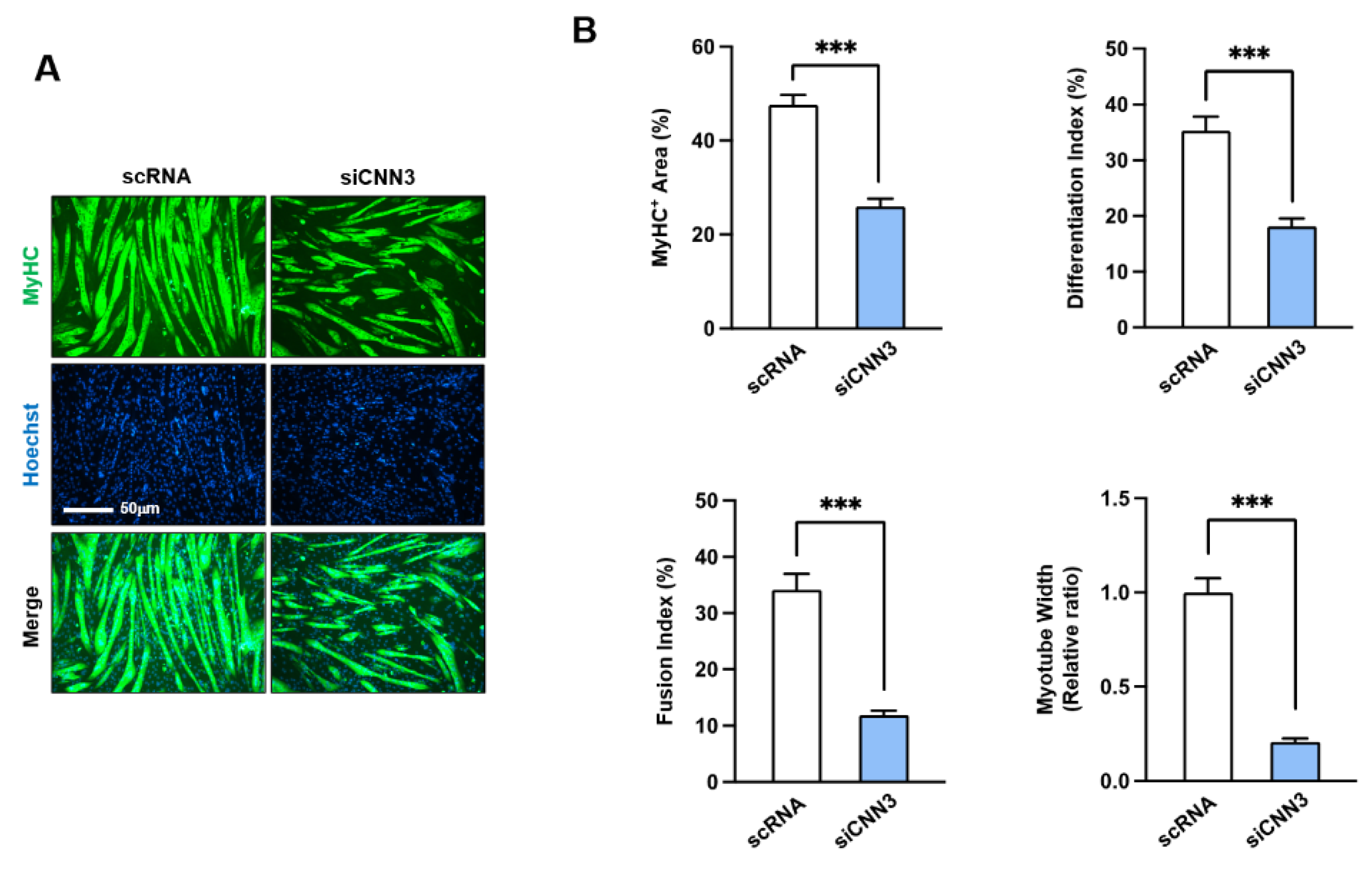
3.4. CNN3 Was Required for Myogenic Differentiation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brooks, S.V.; Guzman, S.D.; Ruiz, L.P. Skeletal muscle structure, physiology, and function. Handb Clin Neurol 2023, 195, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Dyar, K.A.; Ciciliot, S.; Blaauw, B.; Sandri, M. Mechanisms regulating skeletal muscle growth and atrophy. FEBS J 2013, 280, 4294–4314. [Google Scholar] [CrossRef] [PubMed]
- Sartori, R.; Romanello, V.; Sandri, M. Mechanisms of muscle atrophy and hypertrophy: Implications in health and disease. Nature communications 2021, 12, 330. [Google Scholar] [CrossRef] [PubMed]
- Chal, J.; Pourquie, O. Making muscle: Skeletal myogenesis in vivo and in vitro. Development 2017, 144, 2104–2122. [Google Scholar] [CrossRef]
- Feng, L.T.; Chen, Z.N.; Bian, H. Skeletal muscle: Molecular structure, myogenesis, biological functions, and diseases. MedComm (2020) 2024, 5, e649. [Google Scholar] [CrossRef]
- Buckingham, M.; Rigby, P.W.J. Gene Regulatory Networks and Transcriptional Mechanisms that Control Myogenesis. Dev Cell 2014, 28, 225–238. [Google Scholar] [CrossRef]
- Moujaber, O.; Stochaj, U. The Cytoskeleton as Regulator of Cell Signaling Pathways. Trends Biochem Sci 2020, 45, 96–107. [Google Scholar] [CrossRef]
- Jaalouk, D.E.; Lammerding, J. Mechanotransduction gone awry. Nat Rev Mol Cell Biol 2009, 10, 63–73. [Google Scholar] [CrossRef]
- Harris, A.R.; Jreij, P.; Fletcher, D.A. Mechanotransduction by the Actin Cytoskeleton: Converting Mechanical Stimuli into Biochemical Signals. Annu Rev Biophys 2018, 47, 617–631. [Google Scholar] [CrossRef]
- Nowak, S.J.; Nahirney, P.C.; Hadjantonakis, A.K.; Baylies, M.K. Nap1-mediated actin remodeling is essential for mammalian myoblast fusion. J Cell Sci 2009, 122, 3282–3293. [Google Scholar] [CrossRef]
- Zhang, T.; Zaal, K.J.; Sheridan, J.; Mehta, A.; Gundersen, G.G.; Ralston, E. Microtubule plus-end binding protein EB1 is necessary for muscle cell differentiation, elongation and fusion. J Cell Sci 2009, 122, 1401–1409. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.T.; Won, Y.H.; Kwon, T.W.; Lee, W. Twinfilin-1 is an essential regulator of myogenic differentiation through the modulation of YAP in C2C12 myoblasts. Biochem Bioph Res Co 2022, 599, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.T.; Min, K.H.; Kim, D.; Park, S.Y.; Lee, W. CFL2 is an essential mediator for myogenic differentiation in C2C12 myoblasts. Biochem Bioph Res Co 2020, 533, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Guerin, C.M.; Kramer, S.G. Cytoskeletal remodeling during myotube assembly and guidance: Coordinating the actin and microtubule networks. Communicative & integrative biology 2009, 2, 452–457. [Google Scholar]
- Nguyen, M.T.; Dash, R.; Jeong, K.; Lee, W. Role of Actin-Binding Proteins in Skeletal Myogenesis. Cells 2023, 12. [Google Scholar] [CrossRef]
- Vakhrusheva, A.V.; Murashko, A.V.; Trifonova, E.S.; Efremov, Y.M.; Timashev, P.S.; Sokolova, O.S. Role of actin-binding proteins in the regulation of cellular mechanics. Eur J Cell Biol 2022, 101, 151241. [Google Scholar] [CrossRef]
- Takahashi, K.; Nadal-Ginard, B. Molecular cloning and sequence analysis of smooth muscle calponin. J Biol Chem 1991, 266, 13284–13288. [Google Scholar] [CrossRef]
- Shibukawa, Y.; Yamazaki, N.; Kumasawa, K.; Daimon, E.; Tajiri, M.; Okada, Y.; Ikawa, M.; Wada, Y. Calponin 3 regulates actin cytoskeleton rearrangement in trophoblastic cell fusion. Mol Biol Cell 2010, 21, 3973–3984. [Google Scholar] [CrossRef]
- Daimon, E.; Shibukawa, Y.; Wada, Y. Calponin 3 regulates stress fiber formation in dermal fibroblasts during wound healing. Arch Dermatol Res 2013, 305, 571–584. [Google Scholar] [CrossRef]
- Rami, G.; Caillard, O.; Medina, I.; Pellegrino, C.; Fattoum, A.; Ben-Ari, Y.; Ferhat, L. Change in the shape and density of dendritic spines caused by overexpression of acidic calponin in cultured hippocampal neurons. Hippocampus 2006, 16, 183–197. [Google Scholar] [CrossRef]
- Shibukawa, Y.; Yamazaki, N.; Daimon, E.; Wada, Y. Rock-dependent calponin 3 phosphorylation regulates myoblast fusion. Exp Cell Res 2013, 319, 633–648. [Google Scholar] [CrossRef] [PubMed]
- Ebner, N.; Anker, S.D.; von Haehling, S. Recent developments in the field of cachexia, sarcopenia, and muscle wasting: Highlights from the 12th Cachexia Conference. J Cachexia Sarcopenia Muscle 2020, 11, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, E.J.; Koncarevic, A.; Giresi, P.G.; Jackman, R.W.; Kandarian, S.C. Transcriptional profile of a myotube starvation model of atrophy. J Appl Physiol (1985) 2005, 98, 1396–1406. [Google Scholar] [CrossRef] [PubMed]
- Screen, M.; Raheem, O.; Holmlund-Hampf, J.; Jonson, P.H.; Huovinen, S.; Hackman, P.; Udd, B. Gene expression profiling in tibial muscular dystrophy reveals unfolded protein response and altered autophagy. PLoS ONE 2014, 9, e90819. [Google Scholar] [CrossRef]
- Provenzano, P.P.; Keely, P.J. Mechanical signaling through the cytoskeleton regulates cell proliferation by coordinated focal adhesion and Rho GTPase signaling. J Cell Sci 2011, 124, 1195–1205. [Google Scholar] [CrossRef]
- Watt, K.I.; Goodman, C.A.; Hornberger, T.A.; Gregorevic, P. The Hippo Signaling Pathway in the Regulation of Skeletal Muscle Mass and Function. Exercise and Sport Sciences Reviews 2018, 46, 92–96. [Google Scholar] [CrossRef]
- Fischer, M.; Rikeit, P.; Knaus, P.; Coirault, C. YAP-Mediated Mechanotransduction in Skeletal Muscle. Front Physiol 2016, 7, 41. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Qin, S.; Li, C.; Lu, H.; Feng, Y.; Guo, T.; Han, Y.; Zhang, Y.; Tang, Z. Biology of Hippo signaling pathway: Skeletal muscle development and beyond. Journal of Integrative Agriculture 2024, 23, 1825–1838. [Google Scholar] [CrossRef]
- Dupont, S. Role of YAP/TAZ in cell-matrix adhesion-mediated signalling and mechanotransduction. Exp Cell Res 2016, 343, 42–53. [Google Scholar] [CrossRef]
- Panciera, T.; Azzolin, L.; Cordenonsi, M.; Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat Rev Mol Cell Biol 2017, 18, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Deato, M.D.E.; Marr, M.T.; Sottero, T.; Inouye, C.; Hu, P.; Tjian, R. MyoD targets TAF3/TRF3 to activate myogenin transcription. Molecular cell 2008, 32, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Shibukawa, Y.; Yamazaki, N.; Daimon, E.; Wada, Y. Rock-dependent calponin 3 phosphorylation regulates myoblast fusion. Experimental cell research 2013, 319, 633–648. [Google Scholar] [CrossRef]
- Flemming, A.; Huang, Q.Q.; Jin, J.P.; Jumaa, H.; Herzog, S. A Conditional Knockout Mouse Model Reveals That Calponin-3 Is Dispensable for Early B Cell Development. PLoS ONE 2015, 10, e0128385. [Google Scholar] [CrossRef]
- Nair, V.A.; Al-Khayyal, N.A.; Sivaperumal, S.; Abdel-Rahman, W.M. Calponin 3 promotes invasion and drug resistance of colon cancer cells. World J Gastrointest Oncol 2019, 11, 971–982. [Google Scholar] [CrossRef]
- Maddala, R.; Mongan, M.; Xia, Y.; Rao, P.V. Calponin-3 deficiency augments contractile activity, plasticity, fibrogenic response and Yap/Taz transcriptional activation in lens epithelial cells and explants. Sci Rep 2020, 10, 1295. [Google Scholar] [CrossRef]
- Aragona, M.; Panciera, T.; Manfrin, A.; Giulitti, S.; Michielin, F.; Elvassore, N.; Dupont, S.; Piccolo, S. A mechanical checkpoint controls multicellular growth through YAP/TAZ regulation by actin-processing factors. Cell 2013, 154, 1047–1059. [Google Scholar] [CrossRef]
- Mosaddad, S.A.; Salari, Y.; Amookhteh, S.; Soufdoost, R.S.; Seifalian, A.; Bonakdar, S.; Safaeinejad, F.; Moghaddam, M.M.; Tebyanian, H. Response to Mechanical Cues by Interplay of YAP/TAZ Transcription Factors and Key Mechanical Checkpoints of the Cell: A Comprehensive Review. Cell Physiol Biochem 2021, 55, 33–60. [Google Scholar] [CrossRef]
- Yang, C.; Zhu, S.; Feng, W.; Chen, X. Calponin 3 suppresses proliferation, migration and invasion of non-small cell lung cancer cells. Oncol Lett 2021, 22, 634. [Google Scholar] [CrossRef]
- Junghans, D.; Herzog, S. Cnn3 regulates neural tube morphogenesis and neuronal stem cell properties. FEBS J 2018, 285, 325–338. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



