Submitted:
10 December 2024
Posted:
11 December 2024
You are already at the latest version
Abstract
Background: Respiratory syncytial virus (RSV) and human metapneumovirus (hMPV) are common respiratory pathogens that cause severe illness in young children. Methods: As part of an age-de-escalation clinical development program, a phase 1 trial of mRNA-based RSV (mRNA-1345) and investigational RSV/hMPV combination (mRNA-1365) vaccines was conducted in infants and children. Participants were randomized in equal numbers to mRNA-1345 (encoding RSV preF), mRNA-1365 (encoding RSV PreF and hMPV F), or placebo, in an observer-blind study. In a stepwise fashion, children 8 to 23 months of age (Part A) received a 3-injection series of mRNA-1345 (30 μg), mRNA-1365 (30 μg), or placebo. After Data Safety Monitoring Board (DSMB) review of these data, infants 5 to 7 months of age (Part B) were similarly randomized to a 3-injection series using a dose escalation approach (starting at 15 μg). The primary study objective was to assess safety/reactogenicity; secondary objectives were evaluation of clinical RSV/hMPV infections and measurement of antibody/cell-mediated immune responses. Results: In children 8 to 23 months of age (Part A), both mRNA-1345 and mRNA-1365 were well-tolerated and induced robust RSV-A and -B neutralizing antibodies (nAbs) and preF-biased binding antibodies (bAbs). RSV-specific cellular responses assessed in a participant subset demonstrated higher type 1 T helper (Th1) than type 2 T helper (Th2) responses. No concerns were identified following active surveillance for respiratory disease through a full RSV season, and age de-escalation progressed to Part B (infants 5 to 7 months of age). Most participants (88.3%) in Part B met the criteria for “RSV-naïve,” and vaccination induced robust nAb and preF-biased bAb responses. In Part B, a protocol-defined study pause was triggered by 2 cases of severe RSV-lower respiratory tract illness (LRTI) in the 15 μg vaccine level recipients, all of whom had received 2 vaccine injections; as of October 2024 (the first RSV season post immunization), RSV-LRTI classified as severe/hospitalized was seen in 2/20, 3/20, and 1/20 of mRNA-1345 15 µg, mRNA-1365 15 µg, and placebo recipients, respectively. The respiratory illnesses in these children have resolved. Conclusions: Vaccination with mRNA-1345 (RSV vaccine) or mRNA-1365 (RSV/hMPV vaccine) increased RSV nAbs in children aged 8 to 23 months, and induced de novo nAb responses in RSV-naïve infants 5 to 7 months old. Vaccination induced a preF-biased bAb response in both age groups and induced RSV-specific Th1 responses in older children. Study dosing and enrollment are paused while immunogenicity assessments and surveillance for respiratory infections are ongoing to better understand the increase in severe/hospitalized RSV LRTI observed among vaccinated, RSV-naïve, young children. Trial registration number: NCT05743881
Keywords:
Background
Methods
Trial Design and Participants
Trial Vaccines
Study Objectives
Safety and Reactogenicity Assessments
Immunogenicity Assessments
Statistical Analysis
Results
Part A
Participants
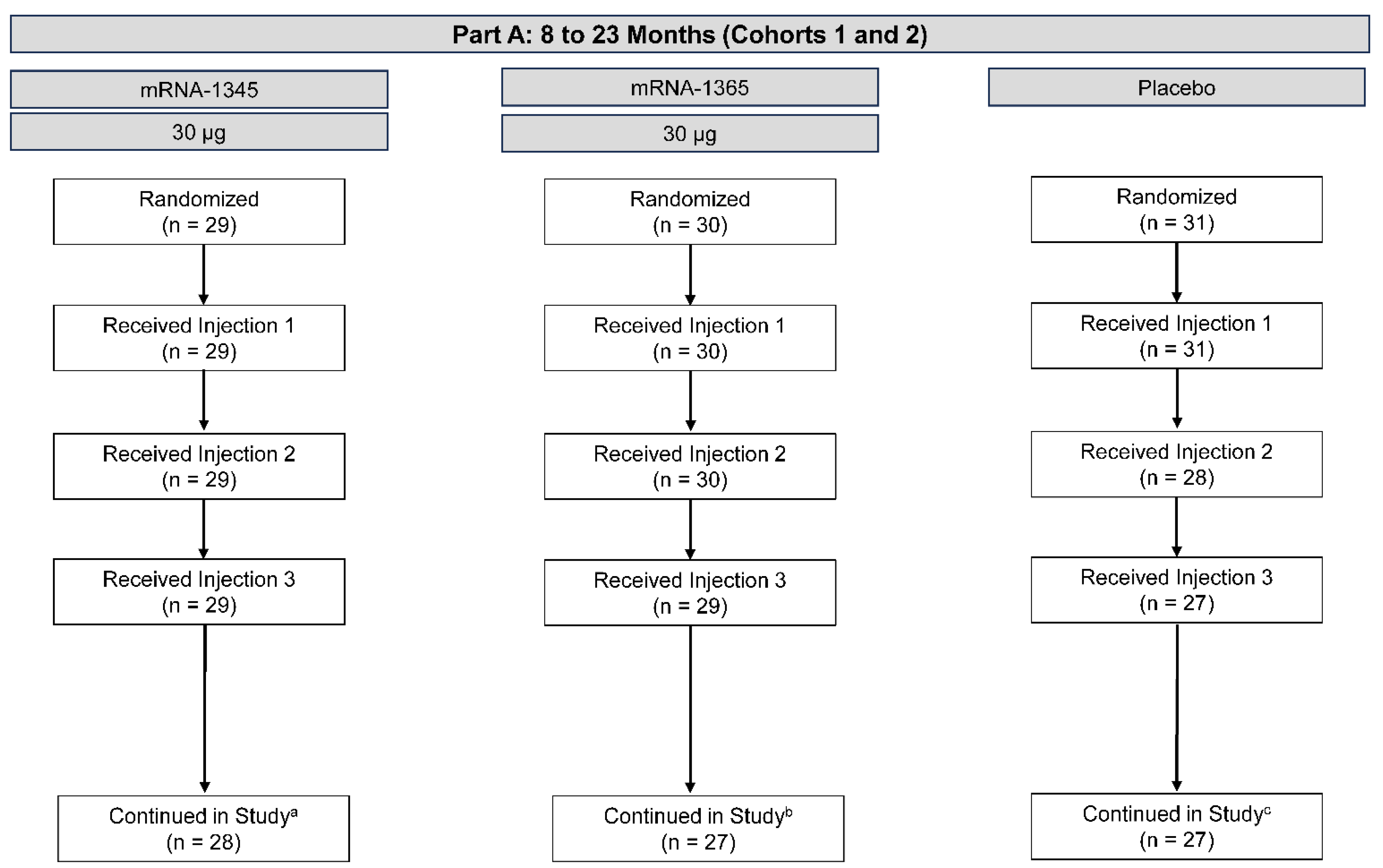
Safety
RSV Case Surveillance
| mRNA vaccines | ||||
|---|---|---|---|---|
| mRNA-1345 30 µg (n=29) |
mRNA-1365 30 µg (n=30) |
Either vaccine (n=59) |
Placebo (n=31) |
|
| RSV-naïve (n=36) | ||||
| Participants, n | 14 | 9 | 23 | 13 |
| Symptomatic RSV (all severity), n (%) | 6 (42.9) | 6 (66.7) | 12 (52.2) | 8 (61.5) |
| Severe/hospitalized RSV, n (%) | 0 | 1 (11.1)c,d | 1 (4.3) | 0 |
| RSV-experienced (n=49) | ||||
| Participants, n | 13 | 19 | 32 | 17 |
| Symptomatic RSV (all severity), n (%) | 5 (38.5) | 7 (36.8) | 12 (37.5) | 6 (35.3) |
| Severe/hospitalized RSV, n (%) | 0 | 0 | 0 | 0 |
Immunogenicity
Antibody Responses
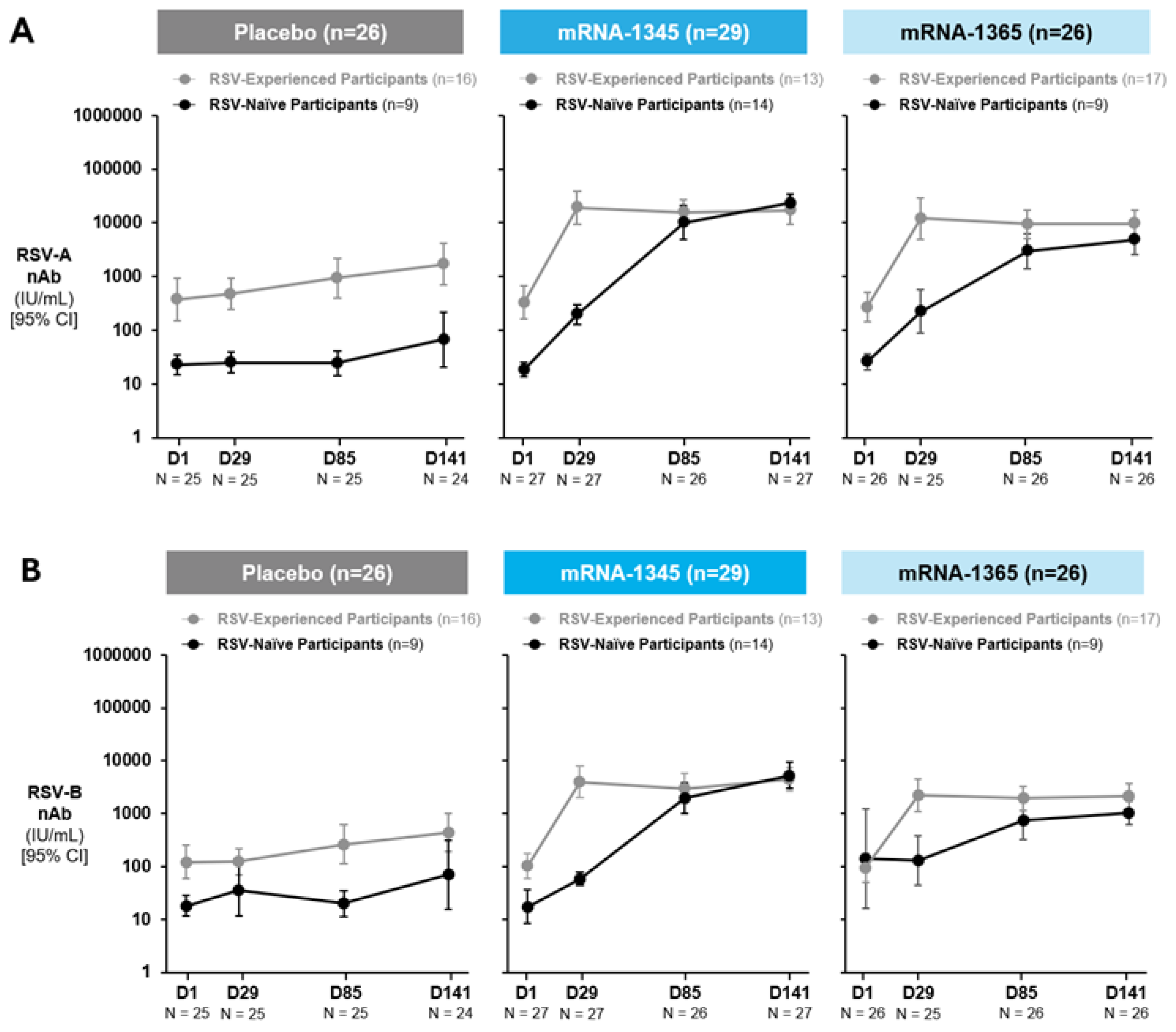
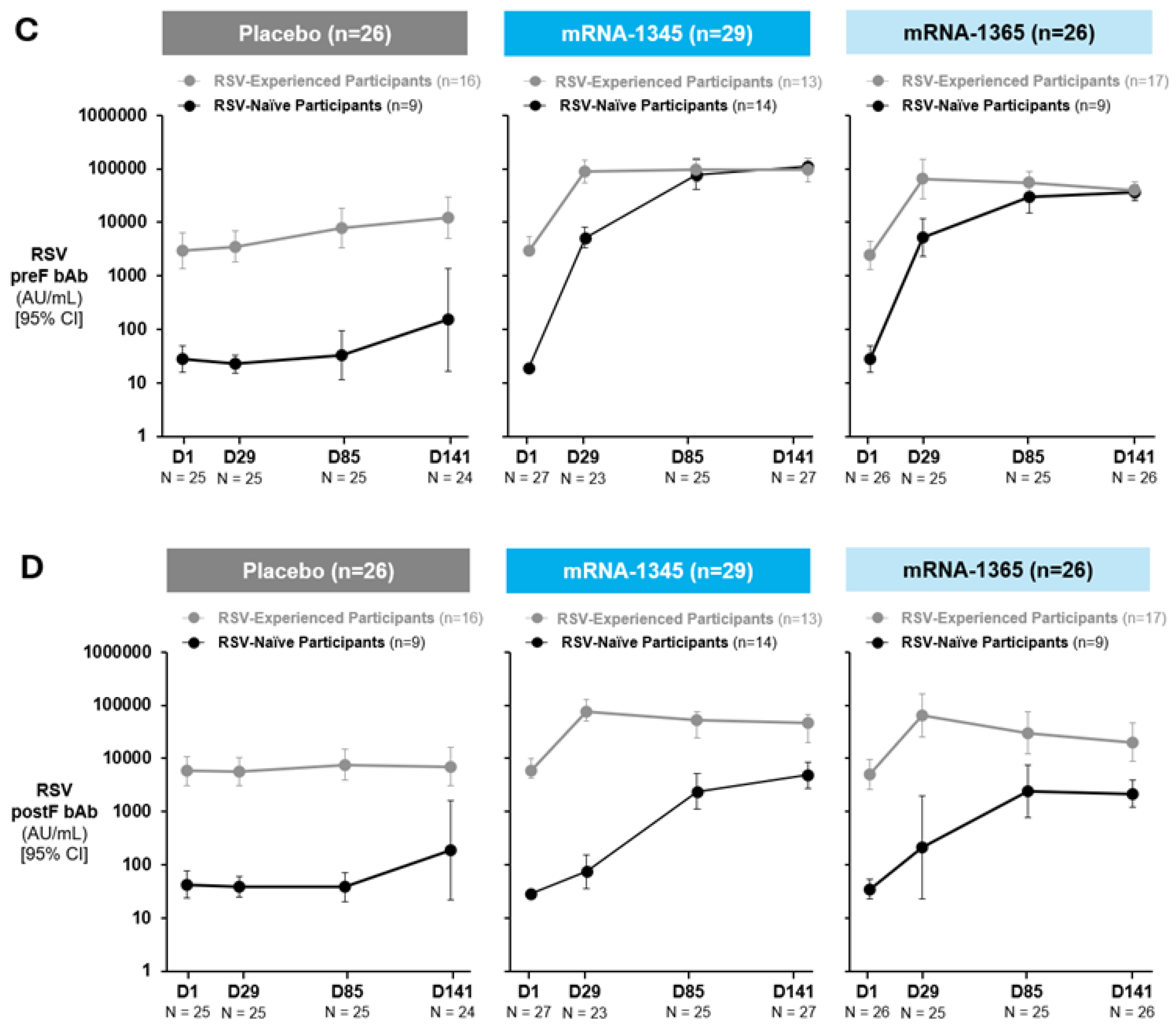
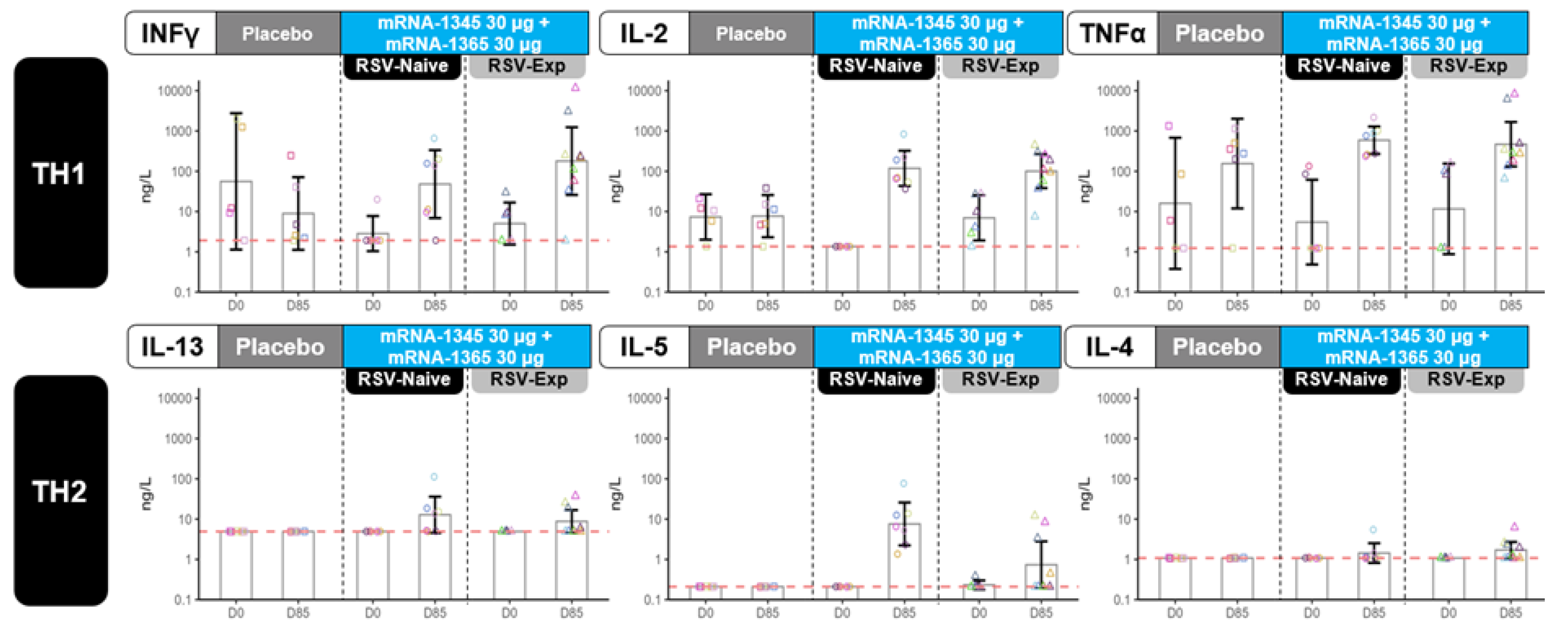
Cell-Mediated Immunogenicity
Part B
Participants
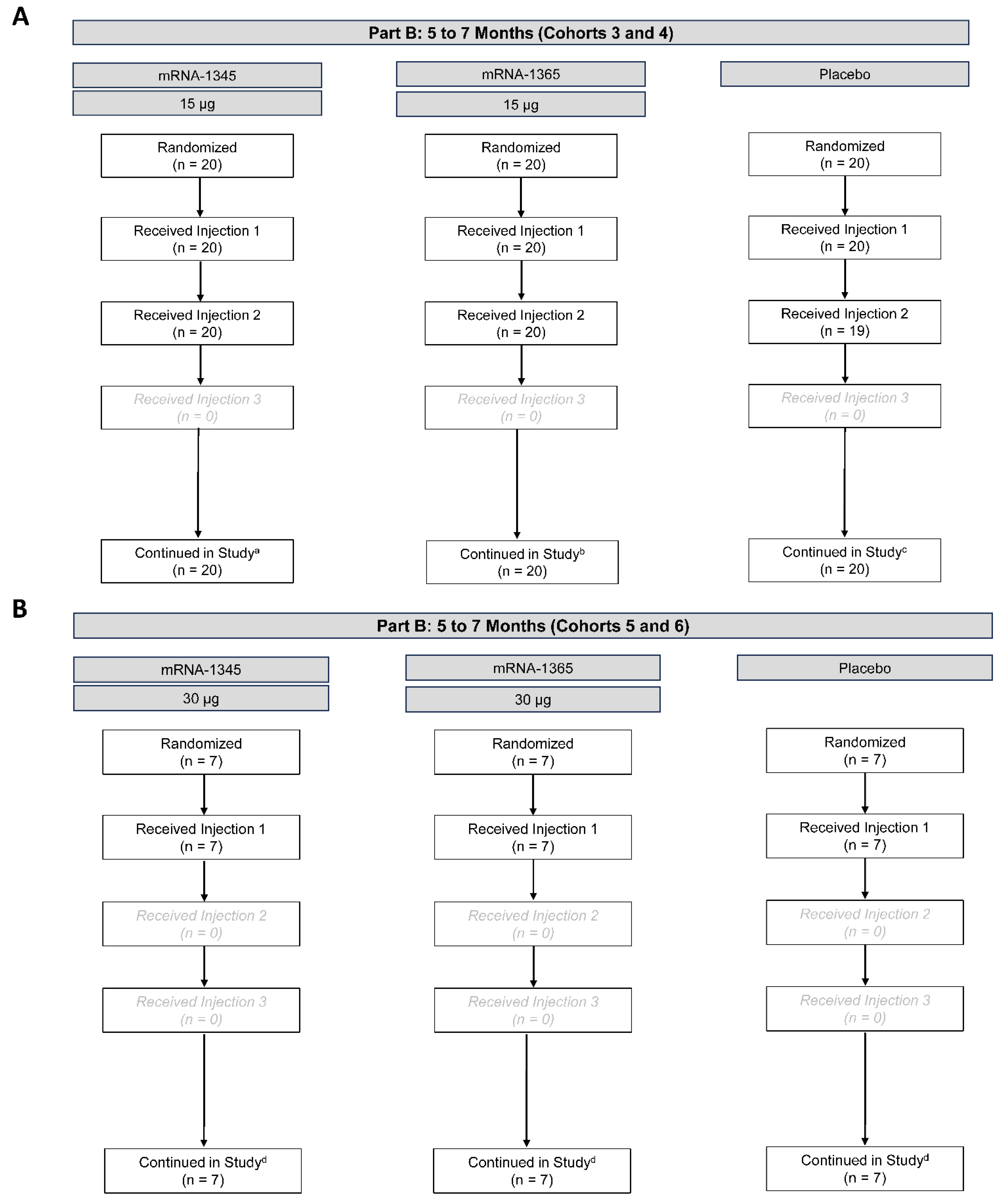
Safety
RSV Case Surveillance
| mRNA vaccines | ||||
|---|---|---|---|---|
| mRNA-1345 15 µg (n=20) |
mRNA-1365 15 µg (n=20) |
Either vaccine (n=40) |
Placebo (n=20) |
|
| RSV-naïve (n=53) | ||||
| Participants, n | 18 | 17 | 35 | 18 |
| Symptomatic RSV (all severity), n (%) | 8 (44.4) | 8 (47.1) | 16 (45.7) | 12 (66.7) |
| Severe/hospitalized RSV, n (%) | 2 (11.1) | 3 (17.6)c | 5 (14.3)c | 1 (5.6)d |
| RSV-experienced (n=7) | ||||
| Participants, n | 2 | 3 | 5 | 2 |
| Symptomatic RSV (all severity), n (%) | 0 | 1 (33.3) | 1 (20.0) | 0 |
| Severe/hospitalized RSV, n (%) | 0 | 0 | 0 | 0 |
Immunogenicity
| Neutralizing antibodies | Binding antibodies | ||||
|---|---|---|---|---|---|
| RSV-A GMT (IU/mL) |
RSV-B GMT (IU/mL) |
RSV preF IgG GMC (AU/mL) | RSV postF IgG GMC (AU/mL) | ||
| Symptomatic RSV infection between baseline and Day 85 | |||||
| mRNA-1345 15 µg | |||||
| Baseline (n=4) | 47.2 | 53.9 | 276.8 | 523.7 | |
| Day 85 (n=4) | 15135.3 | 8959.3 | 128076.7 | 8425.7 | |
| GMFRa (95% CI) (n=4) | 320.9 (9.1-11290.2) | 166.3 (4.8-5750.7) |
462.7 (20-10695.8) |
16.1 (0.2-1337.7) |
|
| mRNA-1365 15 µg | |||||
| Baseline (n=8) | 79.5 | 70.5 | 292.6 | 283.0 | |
| Day 85 (n=6) | 4436.8 | 3851.6 | 49322.8 | 4474.0 | |
| GMFRa (95% CI) (n=6) | 46.4 (2.6-830.8) |
51.1 (3.8-686.0) |
157.2 (7.8-3163.8) |
13.1 (0.9-192) |
|
| Placebo | |||||
| Baseline (n=8) | 66.7 | 92.1 | 261.5 | 228.8 | |
| Day 85 (n=6) | 2007.0 | 557.0 | 8061.2 | 11289.8 | |
| GMFRa (95% CI) (n=6) | 32.1 (15.2-67.8) |
6.3 (2.3-17.4) |
34.7 (16.0-75.4) |
47.2 (20.0-111.2) |
|
| No symptomatic RSV infection between baseline and Day 85 | |||||
| mRNA-1345 15 µg | |||||
| Baseline (n=16) | 104.5 | 80.3 | 500.2 | 429.8 | |
| Day 85 (n=16) | 3762.3 | 2102.0 | 31376.6 | 1796.2 | |
| GMFRa (95% CI) (n=16) | 36.0 (19.5-66.3) |
26.2 (15.4-44.5) |
62.7 (35.9-109.5) |
4.2 (2.2-7.9) |
|
| mRNA-1365 15 µg | |||||
| Baseline (n=12) | 91.6 | 62.3 | 129.7 | 144.0 | |
| Day 85 (n=11) | 2043.7 | 712.3b | 23076.1 | 1655.3 | |
| GMFRa (95% CI) (n=11) | 20.5 (4.3-97.8) |
9.5 (2.4-37.4)b |
148.3 (27.3-806.9) |
9.9 (2.6-38.6) |
|
| Placebo | |||||
| Baseline (n=12) | 170.9 | 123.4 | 546.5 | 794.9 | |
| Day 85 (n=11) | 181.6 | 116.7 | 501.0 | 581.0 | |
| GMFRa (95% CI) (n=11) | 1 (0.3-2.9) | 0.9 (0.4-2.4) | 0.9 (0.1-5,5) | 0.7 (0.1-3.6) | |
Discussion
Data Availability
Author Contributions
Funding
Acknowledgments
References
- Li, Y. , et al., Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet, 2022. 399(10340): p. 2047-2064.
- Wang, X. , et al., Global burden of acute lower respiratory infection associated with human metapneumovirus in children under 5 years in 2018: a systematic review and modelling study. Lancet Glob Health, 2021. 9(1): p. e33-e43.
- Prevention, C.f.D.C.a. Clinical Overview of RSV. 2024 [cited 2024 ]; Available from: https://www.cdc.gov/rsv/hcp/clinical-overview/index.html. 25 November.
- Centers for Disease Control and Prevention. Human Metapneumovirus: About Human Metapneumovirus, /: 2024 [cited 2024 November 25]; Available from: https, 11 April 2024.
- Ren, L. , et al., Viral infections of the lower respiratory tract. Curr Infect Dis Rep, 2012. 14(3): p. 284-91.
- Deshmukh, H. , et al., Impact of Viral Lower Respiratory Tract Infection (LRTI) in Early Childhood (0-2 Years) on Lung Growth and Development and Lifelong Trajectories of Pulmonary Health: A National Institutes of Health (NIH) Workshop Summary. Pediatr Pulmonol, 2024: p. e27357.
- Tregoning, J.S. and J. Schwarze, Respiratory viral infections in infants: causes, clinical symptoms, virology, and immunology. Clin Microbiol Rev, 2010. 23(1): p. 74-98.
- Douros, K. and M.L. Everard, Time to Say Goodbye to Bronchiolitis, Viral Wheeze, Reactive Airways Disease, Wheeze Bronchitis and All That. Front Pediatr, 2020. 8: p. 218.
- Centers for Disease Control and Prevention. Healthcare Providers: RSV Prevention Information, /: 2023 [cited 2024 July 18]; Available from: https, 28 September 2023.
- Centers for Disease Control and Prevention. Human Metapneumovirus (HMPV) About, /: [cited 2024 July 18]; Available from: https, 11 April 2024.
- National Foundation for Infectious Diseases. Respiratory Syncytial Virus (RSV), /: 2024 July 2]; Available from: https, 24 June 2024.
- Kampmann, B., D. Radley, and I. Munjal, Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. Reply. N Engl J Med, 2023. 389(11): p. 1053-1055.
- Centers for Disease Control and Prevention. Healthcare Providers: RSV Vaccination for Pregnant People, /: 2023 [cited 2024 June 17]; Available from: https, 29 September 2023.
- UK Health Security Agency. Complete routine immunisation schedule from 1 September 2024, U: 5]; Available from, 1 September 1294.
- Jones, J.M. , et al., Use of Nirsevimab for the Prevention of Respiratory Syncytial Virus Disease Among Infants and Young Children: Recommendations of the Advisory Committee on Immunization Practices - United States, 2023. MMWR Morb Mortal Wkly Rep, 2023. 72(34): p. 920-925.
- Wolf, D.G. , et al., High seroprevalence of human metapneumovirus among young children in Israel. J Infect Dis, 2003. 188(12): p. 1865-7.
- van den Hoogen, B.G. , et al., A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat Med, 2001. 7(6): p. 719-24.
- Lu, G. , et al., Large-scale seroprevalence analysis of human metapneumovirus and human respiratory syncytial virus infections in Beijing, China. Virol J, 2011. 8: p. 62.
- Andeweg, S.P. , et al., Population-based serology reveals risk factors for RSV infection in children younger than 5 years. Sci Rep, 2021. 11(1): p. 8953.
- Crank, M.C. , et al., A proof of concept for structure-based vaccine design targeting RSV in humans. Science, 2019. 365(6452): p. 505-509.
- McLellan, J.S. , et al., Structure-based design of a fusion glycoprotein vaccine for respiratory syncytial virus. Science, 2013. 342(6158): p. 592-8.
- Graham, B.S., M. S.A. Gilman, and J.S. McLellan, Structure-Based Vaccine Antigen Design. Annu Rev Med, 2019. 70: p. 91-104.
- Package Insert - MRESVIA. 2024 [cited 2024 ]; Available from: https://www.fda.gov/media/179005/download?attachment. 5 December.
- Wilson, E. , et al., Efficacy and Safety of an mRNA-Based RSV PreF Vaccine in Older Adults. N Engl J Med, 2023. 389(24): p. 2233-2244.
- Goswami, J. , et al., Humoral Immunogenicity of mRNA-1345 RSV Vaccine in Older Adults. J Infect Dis, 2024.
- Das, R. Update on Moderna’s RSV Vaccine, mRESVIA (mRNA-1345), in Adults ≥60 Years of Age. 2024 [cited 2024 ]; Available from: https://www.cdc.gov/acip/downloads/slides-2024-06-26-28/04-RSV-Adult-Das-508.pdf. 5 December.
- Shaw, C.A. , et al., Safety and Immunogenicity of an mRNA-Based RSV Vaccine Including a 12-Month Booster in a Phase I Clinical Trial in Healthy Older Adults. J Infect Dis, 2024.
- Paris, R. , et al., T-Cell Responses Following Vaccination With an mRNA Respiratory Syncytial Virus Vaccine, mRNA-1345, in Older Adults, in International Federation of Ageing (IFA). 2023: Bangkok, Thailand.
- Anderson, E.J. , et al., Evaluation of mRNA-1273 vaccine in children 6 months to 5 years of age. N Engl J Med, 2022. 387(18): p. 1673-1687.
- Creech, C.B. , et al., Evaluation of mRNA-1273 Covid-19 Vaccine in Children 6 to 11 Years of Age. N Engl J Med, 2022. 386(21): p. 2011-2023.
- Ali, K. , et al., Evaluation of mRNA-1273 SARS-CoV-2 Vaccine in Adolescents. N Engl J Med, 2021. 385(24): p. 2241-2251.
- Munoz, F.M. , et al., Evaluation of BNT162b2 Covid-19 Vaccine in Children Younger than 5 Years of Age. N Engl J Med, 2023. 388(7): p. 621-634.
- Polack, F.P. , et al., Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med, 2020. 383(27): p. 2603-2615.
- Noor, A. and L.R. Krilov, A Historical Perspective on Respiratory Syncytial Virus Prevention: A Journey Spanning Over Half a Century From the Setback of an Inactive Vaccine Candidate to the Success of Passive Immunization Strategy. J Pediatric Infect Dis Soc, 2024. 13(Supplement_2): p. S103-s109.
- Mejias, A. and O. Ramilo, RSV Prevention Within Reach for Older Infants and Toddlers: The Role of Active Immunization. J Pediatric Infect Dis Soc, 2024. 13(Supplement_2): p. S125-s130.
- Murphy, B.R. and E.E. Walsh, Formalin-inactivated respiratory syncytial virus vaccine induces antibodies to the fusion glycoprotein that are deficient in fusion-inhibiting activity. J Clin Microbiol, 1988. 26(8): p. 1595-7.
- Stephens, L.M. , et al., Prefusion F-Based Polyanhydride Nanovaccine Induces Both Humoral and Cell-Mediated Immunity Resulting in Long-Lasting Protection against Respiratory Syncytial Virus. J Immunol, 2021. 206(9): p. 2122-2134.
- Acosta, P.L., M. T. Caballero, and F.P. Polack, Brief history and characterization of enhanced respiratory syncytial virus disease. Clin Vaccine Immunol, 2016. 23(3): p. 189-195.
- Giersing, B.K. , et al., Report from the World Health Organization's Product Development for Vaccines Advisory Committee (PDVAC) meeting, Geneva, 7-9th Sep 2015. Vaccine, 2016. 34(26): p. 2865-2869.
- Snape, M.D. , et al., Phase 1 Safety and Immunogenicity Results of Two Investigational mRNA Vaccines, mRNA-1345, a Respiratory Syncytial Virus Vaccine, and mRNA-1653, a Human Metapneumovirus and Parainfluenza Virus Type 3 Combination Vaccine in Seropositive Young Children. Open Forum Infect Dis, 2023. 10: p. ofad500.2266.
- August, A. , et al. Safety and immunogenicity of an mRNA-based human metapneumovirus and parainfluenza virus type 3 combined vaccine in healthy adults, 2022. [Google Scholar]
- Schnyder Ghamloush, S. , et al., Safety and Immunogenicity of an mRNA-Based hMPV/PIV3 Combination Vaccine in Seropositive Children. Pediatrics, 2024. 153(6).
- Shaw, C.A. , et al., Design and preclinical assessment of mRNA-1345 prefusion F glycoprotein-encoding mRNA vaccine 1 for respiratory syncytial virus. Submitted., 2024.
- World Health Organization. Guidelines on the quality, safety and efficacy of respiratory syncytial virus vaccines, /: 2024 ]; Available from: https, 29 April 2024.
- United States Department of Health and Human Services. Respiratory Syncytial Virus Infection: Developing Antiviral Drugs for Prophylaxis and Treatment Guidance for Industry, /: 2024 ]; Available from: https, 5 December 2024.
- European Medicines Agency. Guideline on the clinical evaluation of medicinal products indicated for the prophylaxis or treatment of respiratory syncytial virus (RSV) disease, /: 2024 ]; Available from: https, 5 December 2024.
- Centers for Disease Control and Prevention. RSV Immunization Guidance for Infants and Young Children, /: 2024 ]; Available from: https, 2 December 2024.
- Li, Z. , et al., Efficacy, immunogenicity and safety of COVID-19 vaccines in older adults: a systematic review and meta-analysis. Frontiers in Immunology, 2022. 13.
- Giersing, B.K. , et al., Meeting report: WHO consultation on Respiratory Syncytial Virus (RSV) vaccine development, Geneva, 25-. Vaccine, 2019. 37(50): p. 7355-7362. 26 April.
- Vaccines and Related Biological Products Advisory Committee Meeting. FDA Briefing Document: Development of Vaccines for Prevention of RSV Disease in RSV-Naive Infants, /: 2024 ]; Available from: https://public4.pagefreezer.com/content/FDA/01-02-2023T10:30/https, 3 December 2024.
- DiPiazza, A.T. , et al., COVID-19 vaccine mRNA-1273 elicits a protective immune profile in mice that is not associated with vaccine-enhanced disease upon SARS-CoV-2 challenge. Immunity, 2021. 54(8): p. 1869-1882 e6.
- Jackson, L.A. , et al., An mRNA Vaccine against SARS-CoV-2 - Preliminary Report. N Engl J Med, 2020. 383(20): p. 1920-1931.
- Mateus, J. , et al., Low-dose mRNA-1273 COVID-19 vaccine generates durable memory enhanced by cross-reactive T cells. Science, 2021. 374(6566): p. eabj9853.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




