2. Case Presentation
A 30-year-old woman was referred to our institution with a 3-year history of low abdominal pain. The patient had no other associated digestive symptoms, including rectal fullness, constipation, pain on defecation, rectal bleeding or genitourinary obstruction symptoms. There were no abnormal findings at general physical and per abdominal examination. The rectal examination was also normal. During her third pregnancy a cystic pelvic mass had been diagnosed as a left ovarian cyst, with the suspicion of a dermoid origin. She underwent three consecutive surgical procedures, both laparoscopic and classical, for ovarian cyst, in different centers, without relieving symptoms. In all cases, the preoperative diagnosis was based on abdominal and transvaginal ultrasound. At the third procedure, no ovarian cyst was found, and a computed-tomography (CT) scan was recommended.
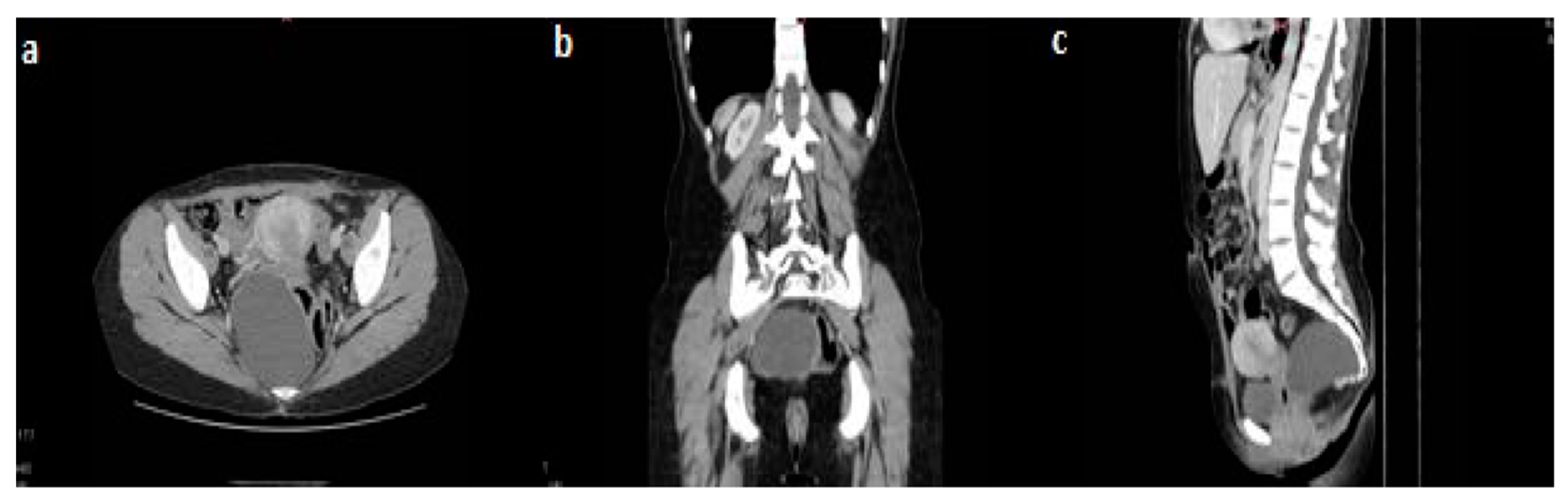
The abdominal-pelvic CT scan revealed an 88/64 mm presacral tumor with a right pararectal development and a mass effect on the rectum against which there was no cleavage plan; there was no relationship with the iliac vessels (
Figure 1). A surgical resection of the lesion was decided using an anterior approach via a Pfannenstiel incision. Intraoperatively, an 8 cm well-defined retroperitoneal presacral mass was described, which in course of the handling maneuvers teared up, eliminating an opaque, viscous liquid. A full resection of the cyst was performed. The postoperative recovery was uneventful, and the patient was discharged on the 6th post-operative day. One year after surgery the patient had no evidence of recurrence and was symptom free.
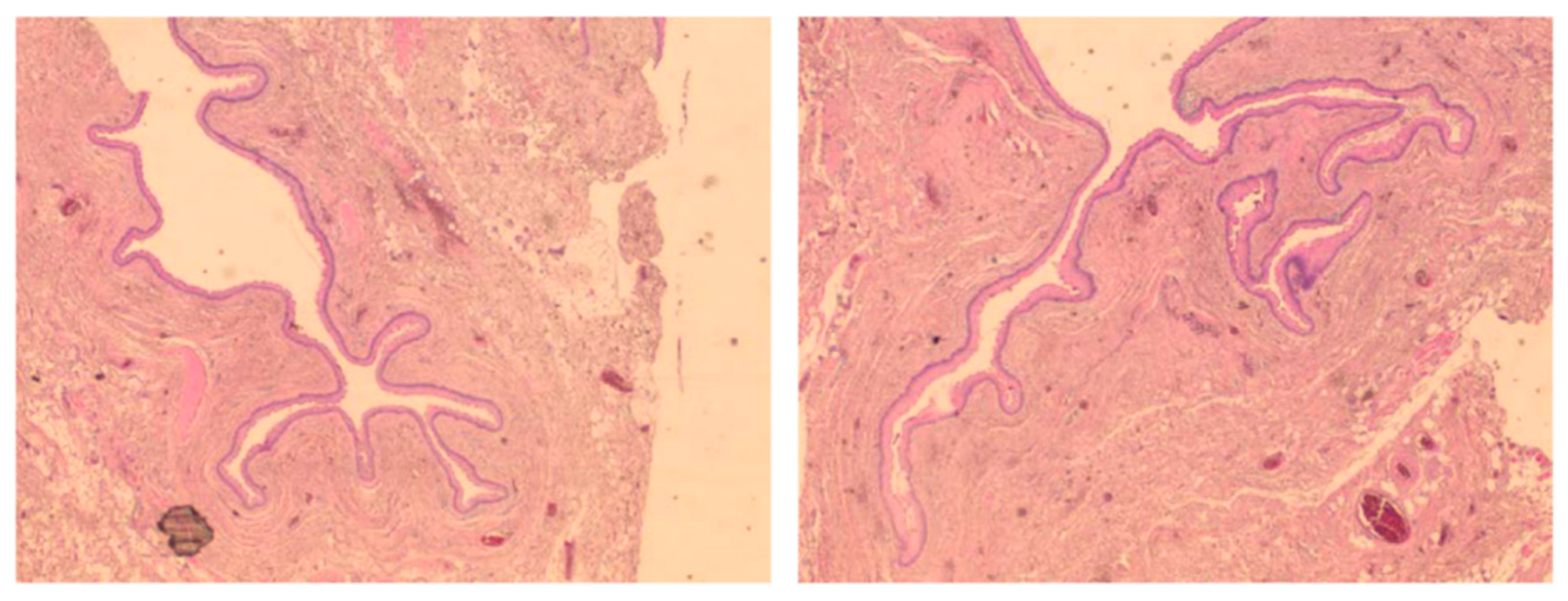
Cytological examination of the fluid revealed a benign smear, and histopathology revealed an 8/3/1 cm cyst lined by non-keratinized stratified squamous epithelium without granular layer, fibrous connective tissue walls with reduced inflammatory lymphoplasmacytic infiltration and hemorrhage areas (
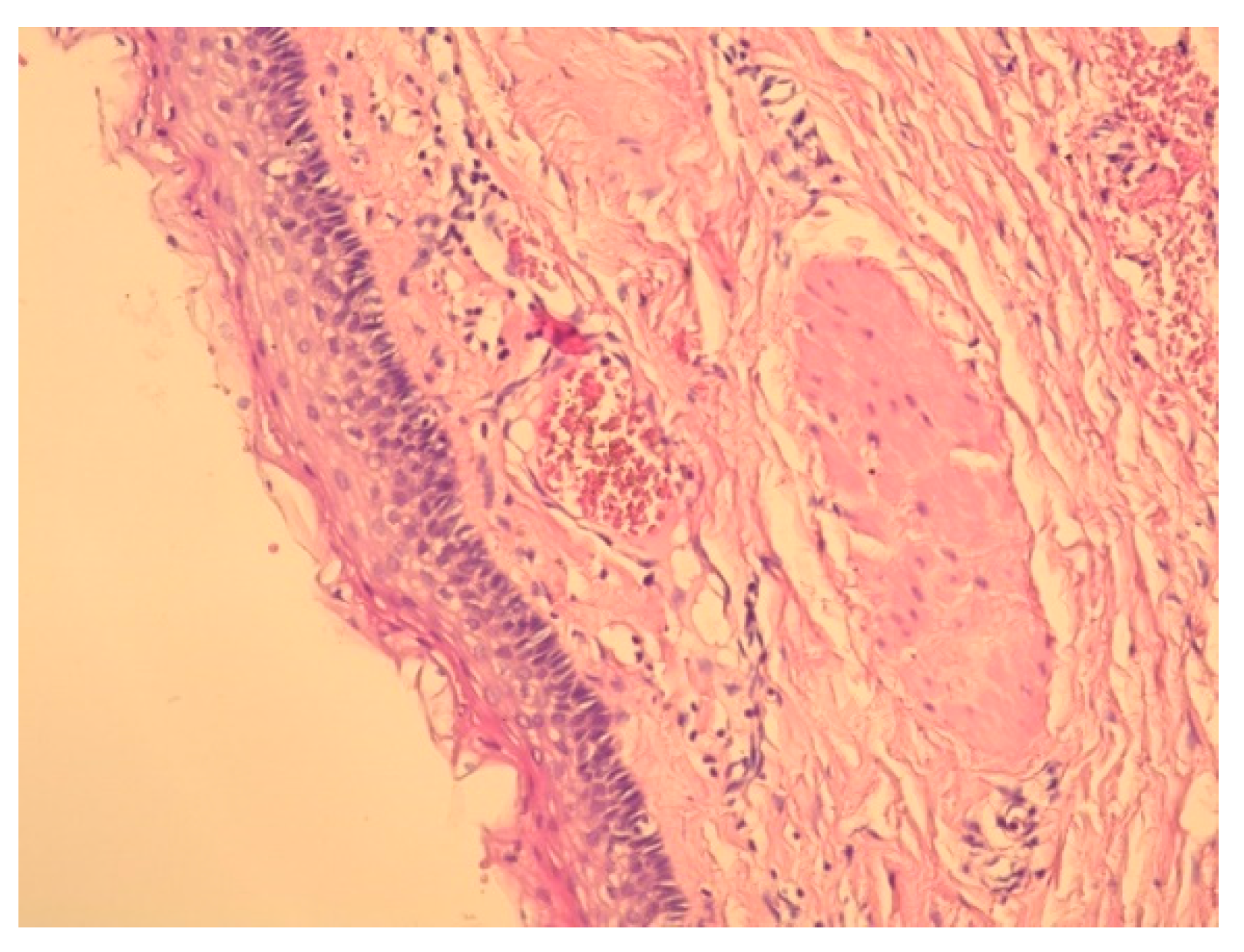
Figure 2). Smooth muscle fibers were described at the periphery (
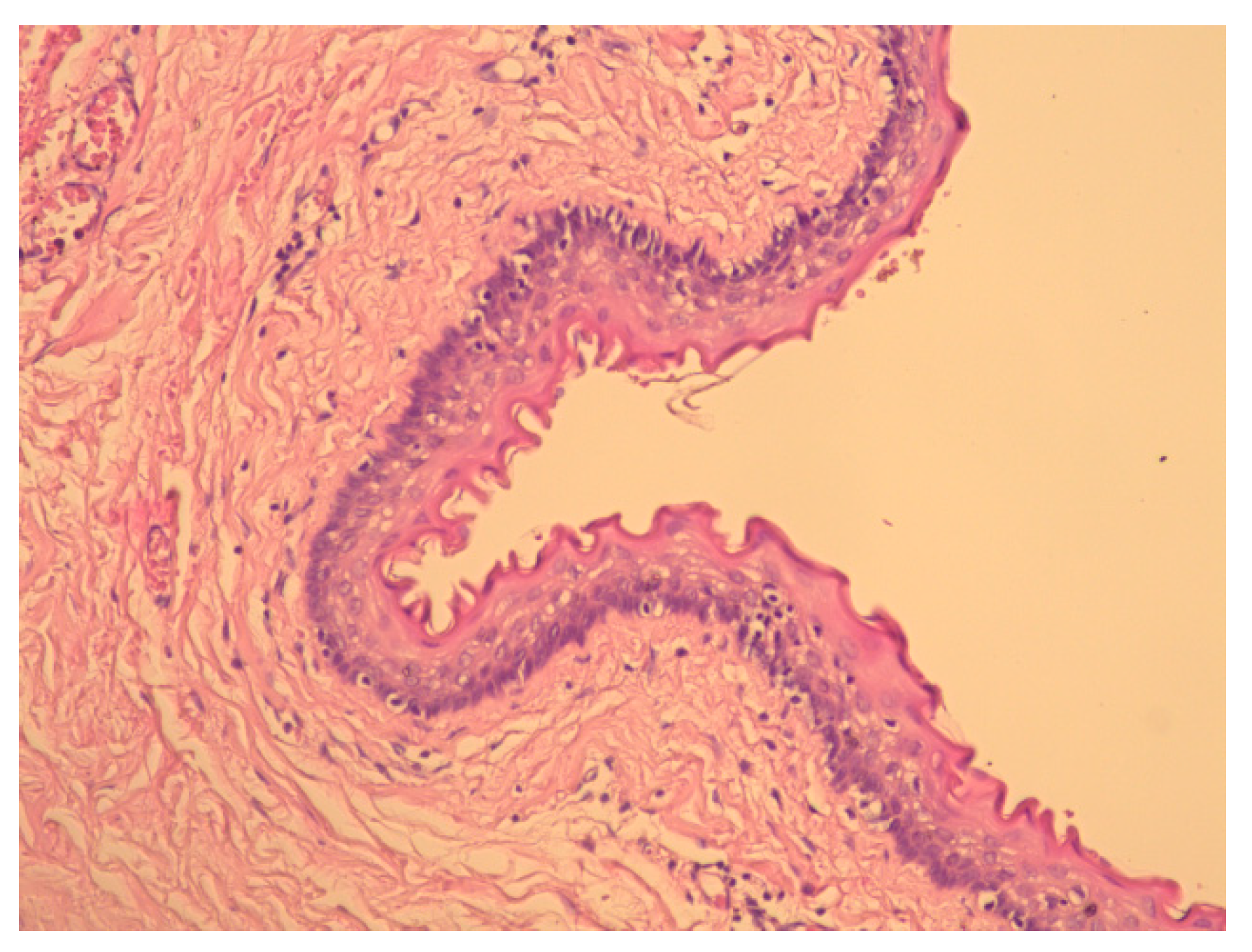
Figure 3 and
Figure 4). The final diagnosis was “Retrorectal cystic hamartoma (Tailgut cyst)”.
4. Discussion
The tailgut cyst is a rare congenital anomaly typically located in retrorectal (presacral) space. This virtual anatomical space is bordered anteriorly by the posterior rectal wall, posteriorly by the sacrum, superiorly by the peritoneal reflection, and inferiorly by the levator ani and coccygeus muscles. Laterally, its boundaries include the ureters, iliac vessels, sacral nerve roots, and lateral rectal stalks. The rectosacral fascia further divides this space into two compartments: a superior and an inferior section. These compartments contain loose connective tissue, the middle sacral, iliolumbar, and middle hemorrhoidal vessels, along with branches of the sympathetic and parasympathetic nervous systems, as well as lymphatic structures [
1,
5].
A wide variety of masses can arise within retrorectal space. Differential diagnosis includes primary tumors (of neurogenic, osteogenic, or congenital origin), metastatic lesions, and inflammatory processes. Among congenital lesions, developmental cysts (epidermoid, dermoid, enteric duplication cysts, and tailgut cysts), chordomas, teratomas, and anterior sacral meningoceles are notable. Epidermoid and dermoid cysts share the presence of stratified squamous epithelium, but dermoid cysts also contain dermal components such as sweat glands, hair follicles, and tooth buds [
6].
The distinction between tailgut cysts and rectal duplication cysts lies in the cyst wall composition: tailgut cysts exhibit disorganized smooth muscle fibers without neural plexuses, whereas rectal duplication cysts have two layers of smooth muscle accompanied by nerve plexuses. Tailgut cysts can feature multiple epithelial types, sometimes within the same cyst, including stratified squamous, transitional, stratified columnar, mucinous or ciliated columnar, ciliated pseudostratified columnar, and gastric epithelium. They are typically uni- or multilocular with thin walls, and their contents range from clear fluid to dense mucus [
6,
7,
8].
The prevailing hypothesis for the development of tailgut cysts suggests that these lesions originate from vestigial remnants of the embryonic hindgut. During early embryogenesis, the embryo possesses a true tail until approximately the 35th day of gestation, extending caudally beyond the future site of the anus. This primitive hindgut extends into the embryonic tail, giving rise to the terms "postanal gut" and "tailgut." Normally, this structure undergoes complete involution by the 56th day of gestation. However, incomplete involution may result in the persistence of tailgut remnants, which are hypothesized to contribute to the formation of tailgut cysts [
5,
7].
The estimated incidence of this condition is 1 in 40,000 individuals. While cases have been reported in neonates, tailgut cysts predominantly affect women in the third to sixth decades of life, with a female-to-male ratio of 3:1. No specific risk factors have been identified in the literature [
5,
9].
The clinical presentation of tailgut cysts is often nonspecific, leading to delayed or incidental diagnoses. According to a study by Singer et al., an average of 4.7 invasive procedures or surgeries is typically required to achieve an accurate diagnosis and appropriate treatment of a retrorectal mass [
7]. Many patients remain asymptomatic for extended periods, with symptoms usually emerging only when complications occur. These complications include infection, fistula formation, bleeding, or malignant transformation, with infection being the most common (40–50%). Rectal bleeding is more characteristic of rectal duplication cysts [
10].
Symptoms typically arise due to mass effect on adjacent structures and may include lower abdominal pain, back pain, rectal fullness, constipation, painful defecation, altered stool patterns, genitourinary obstruction, dysuria, pollakiuria, or right-sided sciatica [
1,
2,
3,
4]. In rare cases, it is found during labor, where a tailgut cyst may obstruct the birth canal, necessitating a transition to cesarean delivery [
5]. On digital rectal examination, these lesions are often identified as extrinsic masses with a fluctuant consistency, further aiding in clinical suspicion [
5].
Advancements in imaging techniques have significantly improved the diagnostic accuracy for this rare lesion. Transrectal ultrasound (TRUS) is particularly useful in patients presenting with rectal bleeding, providing valuable information regarding the location, size, content, and potential local invasion of the lesion, while also aiding in the exclusion of other differential diagnoses [
3,
8]. Computed tomography (CT) characterizes tailgut cysts as well-circumscribed, uni- or multilocular masses with liquid or soft-tissue density. In cases of infection, diffuse wall thickening may be evident. Features such as intra-cystic proliferation, septations, irregular or nodular thickening, involvement of adjacent structures, or lymphadenopathy on CT may raise suspicion for malignant transformation [
8]. The preferred imaging modality for retrorectal masses is magnetic resonance imaging (MRI), which offers superior anatomical detail, excellent soft tissue contrast, and the advantage of multiplanar reconstruction [
1,
5,
7]. On MRI, retrorectal tumors typically exhibit low signal intensity on T1-weighted images and high signal intensity on T2-weighted images, although these characteristics can vary with the composition of the cyst. For instance, increased protein concentration (e.g., from bleeding) can produce hyperintense signals on T1-weighted images; mucinous fluid, depending on its protein content and viscosity, may appear hypointense to hyperintense on both T1- and T2-weighted images. Malignant degeneration is suggested by consistently low signal intensity on both T1- and T2-weighted images, while septations generally appear hypointense on T2-weighted images [
5,
11,
12]. Aflalo-Hazan et al. observed that most tailgut cysts demonstrate high signal intensity relative to muscle tissue on MRI [
13]. Kim et al. [
14] further described a characteristic MRI finding of tailgut cysts as multilocular masses with internal septations on T2-weighted images. These imaging features aid in distinguishing tailgut cysts from other retrorectal lesions.
A controversy arises regarding the necessity of a preoperative biopsy. Most of the physicians agree that the disadvantages outweigh the advantages, this procedure being usually discouraged [
15,
16]. The risks that may occur include biopsy tract or peritoneal dissemination, bleeding, infection. Some authors support the necessity of a preoperative biopsy for malignant suspected lesions, evoking the possibility of neoadjuvant therapy for certain pathological subtypes before surgery, which may improve the final result [
5,
7]. Hjermstad et al. presented a 2% rate of malignancy in a series of 53 tailgut cysts, but it is assumed that this rate is much higher, Mayo Clinic Group reporting a 13% rate in a paper published in 2010 [
17,
18]. The most common malignant degenerations of tailgut cysts are adenocarcinomas, carcinoids and sarcomas [
7,
8].
Complete surgical resection is the treatment of choice for tailgut cysts [
1,
8]. Over the past years different approaches have been described in the literature: the anterior approach (transabdominal), the posterior approaches, and a combination of these two. The posterior approaches include many surgical techniques, like inter-sphincteric, trans-sphincteric, para-sacrococcygeal, trans-sacral, trans-sacrococcygeal, trans-anorectal or trans-vaginal. The transabdominal approach offers a direct image of pelvic structures, the ureters and iliac vessels. The advantage of the posterior approach is the access to the distal component of the tumor, but the lack of control over pelvic structures and the possibility of injuring the pelvic nerves is higher [
1]. The surgical technique selection is usually based on several factors, such as the size, location, morphology of the lesion, possible adherences, infection or malignancy. It is thought that the anterior approach should be carried out for lesions that are above S3 or the sacral promontory and the posterior approaches should be reserved for masses that are below the above mentioned point [
1,
7,
16]. For the posterior approaches the patient should be placed in a jack knife, lithotomy or lateral position and all the efforts be made to not injure and to preserve the sacral nerve roots. If this is not an option, the bladder and the bowel are thought to keep a normal function even with unilateral preservation of S2-S4 nerve roots. The posterior approach may be a good choice, if the proximal extent of the mass can be palpated at the digital rectal examination. The combined method sums up the advantages of the two approaches, offering a proper view of the anatomy, the lesion’s extent and the capacity of a better vascular control, being used for large masses that are located above and below the S3 vertebrae or tailgut cysts with a diameter greater than 4-5 cm [
4]. Laparoscopic excision was also performed by some authors with promising results. Transanal endoscopic microsurgery excisions may also become a feasible option. The advantages of these modern techniques include a good exposure of the presacral space, a reduced surgical trauma and an easier recovery. The disadvantages are represented by the high costs, being carried out only in centers of expertise. The patient must be well informed about the limitations and the risks of surgery, explaining the possibility of a stoma if necessary [
1,
5,
16].
Postoperatively, long-term complications could appear, which includes pelvic floor dysfunction, delayed wound healing, sexual dysfunction and a risk of recurrence that may reach 16% [
9,
12]. There has not been established a standard follow-up, but an annual digital exam and a CT scan in the first and the fifth year after surgery should be performed [
4].