Submitted:
03 December 2024
Posted:
04 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solubility Determination
2.3. FDA Dissolution Method
2.4. Development of the Biopredictive Dissolution Method
2.5. Dissolution Efficiency Evaluation
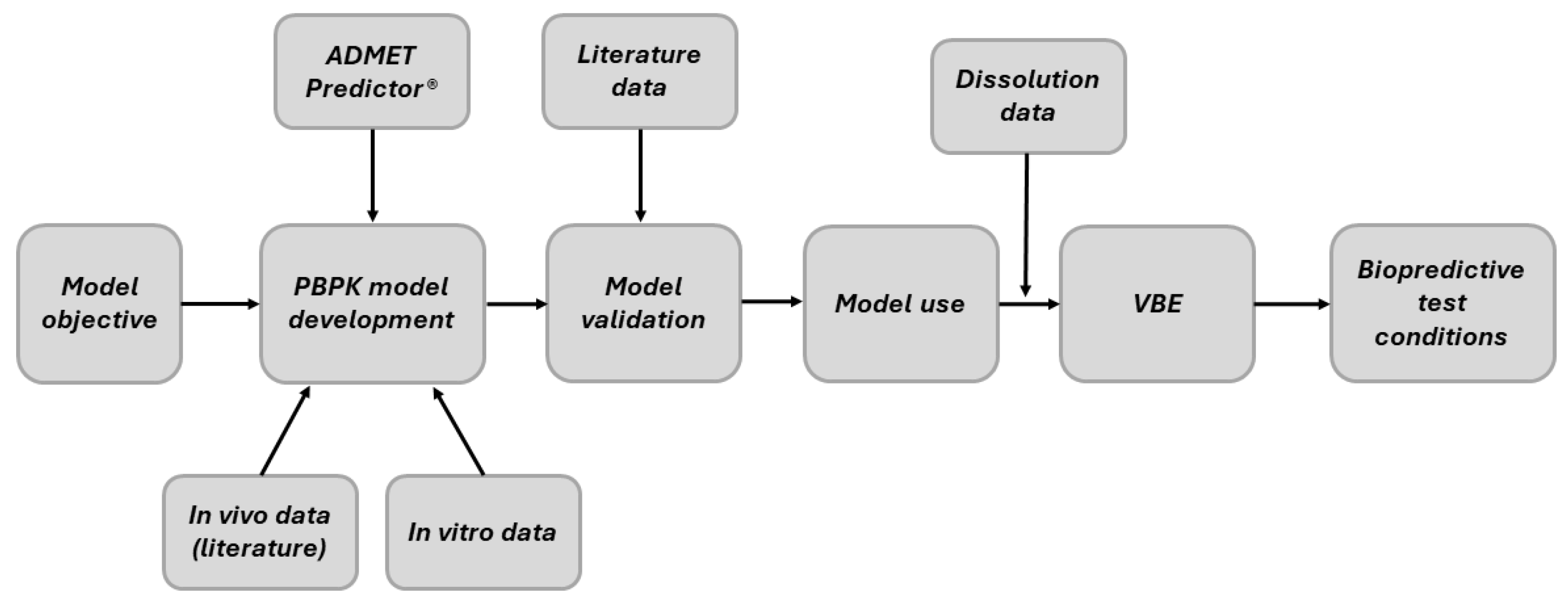
2.6. PBBM Development, Validation and Use
2.6.1. PBPK Model Development
2.6.2. PBPK Model Validation
2.6.3. Evaluation of Model Predictability
2.6.4. Model Use
2.7. Virtual Bioequivalence Studies
3. Results and Discussion
3.1. Solubility Determination
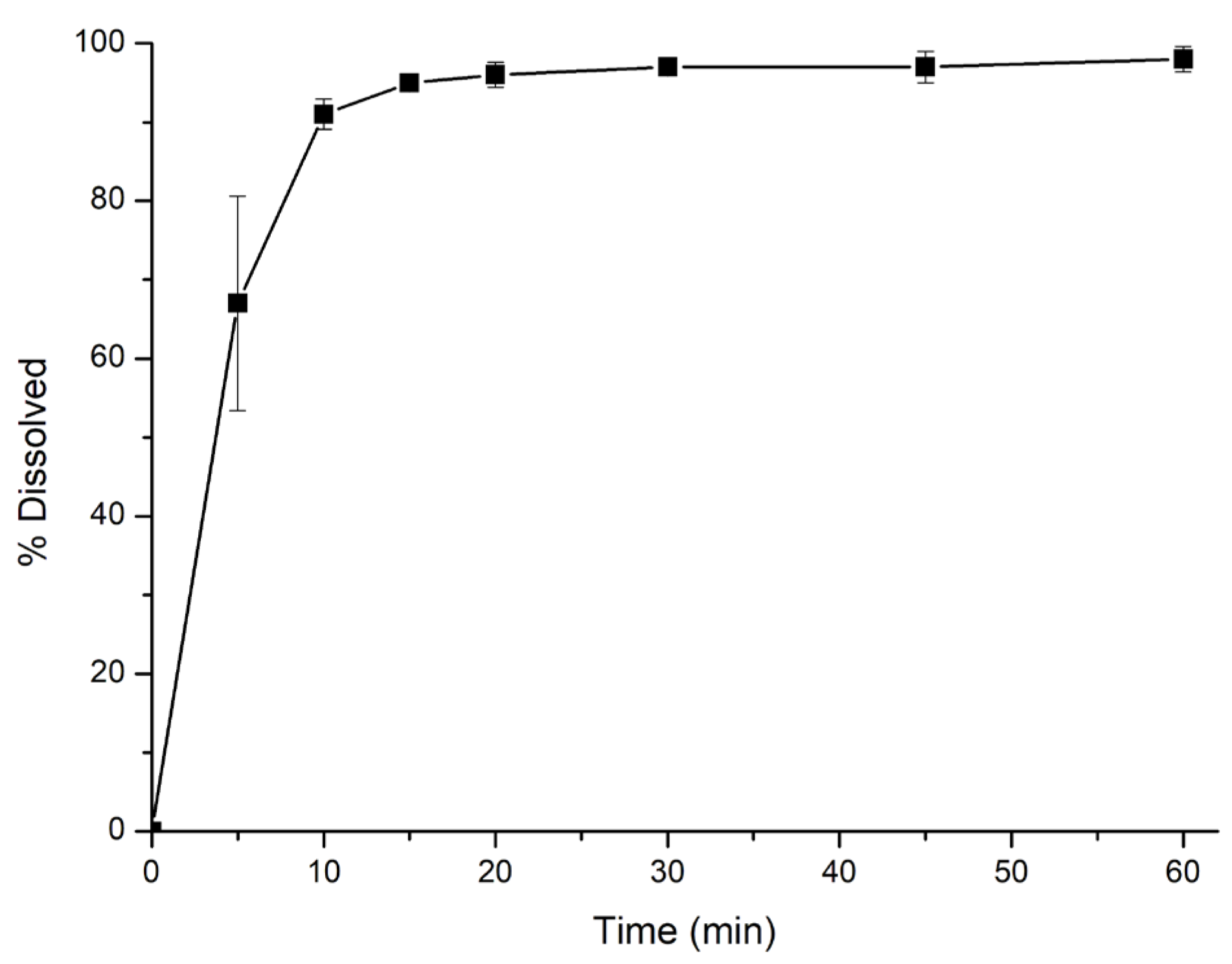
3.2. FDA Dissolution Method
3.3. Development of Biopredictive Dissolution Method and Dissolution Efficiency Evaluation
3.4. Development and Validation of the PBPK Model
3.5. PBPK Model Use and Virtual Bioequivalence Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FDA—U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Physiologically Based Pharmacokinetic Analyses - Format and Content Guidance for Industry. 2018, Available online:. Available online: https://www.fda.gov/media/101469/download (accessed on day month year).
- FDA—U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). The use of physiologically based pharmacokinetic analyses - biopharmaceutics applications for oral drug product development, manufacturing changes, and controls guidance for industry – draft guidance. 2020, Available online:. Available online: https://www.fda.gov/media/142500/download (accessed on day month year).
- EMA - European Medicines Agency Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation. Committee for Medicinal Products for Human Use (CHMP). 2019, Available online:. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-reporting-physiologically-based-pharmacokinetic-pbpk-modelling-and-simulation_en.pdf (accessed on day month year).
- Ghate, V.M.; Chaudhari, P.; Lewis, S.A. Physiologically based pharmacokinetic (PBPK) modelling for in vitro-in vivo extrapolation: Emphasis on the use of dissolution data. Dissolution Technologies 2019, 26, 18–27. [Google Scholar] [CrossRef]
- Bermejo, M.; Hens, B. , Dickens, J.; Mudie, D.; Paixão, P.; Tsume, Y.; Shedden K.; Amidon G.L. A mechanistic physiologically- based biopharmaceutics modeling (PBBM) approach to assess the in vivo performance of an orally administered drug product: from IVIVC to IVIVP. Pharmaceutics 2020, 12, 1–28. [Google Scholar] [CrossRef] [PubMed]
- McAllister, M.; Flanagan, T.; Cole, S.; Abend, A.; Kotzagiorgis, E.; Limberg, J.; Mead, H.; Mangas-Sanjuan, V.; Dickinson, P.A.; Moir, A.; Pepin, X.; Zhou, D.; Tistaert, C.; Dokoumetzidis, A.; Anand, O.; Le Merdy, M.; Turner, D.B.; Griffin, B.T.; Darwich, A.; Dressman, J.; Mackie, C. Developing Clinically Relevant Dissolution Specifications (CRDSs) for Oral Drug Products: Virtual Webinar Series. Pharmaceutics 2022, 14, 1010. [Google Scholar] [CrossRef] [PubMed]
- Zhang X, Lionberger RA, Davit BM, Yu LX. Utility of physiologically based absorption modeling in implementing Quality by Design in drug development. AAPS J 2011, 13, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Heimbach, T.; Suarez-Sharp, S.; Kakhi, M.; Holmstock, N.; Olivares-Morales, A.; Pepin, X.; Sjögren, E.; Tsakalozou, E.; Seo, P.; Li, M.; Zhang, X.; Lin, H.P.; Montague, T.; Mitra, A.; Morris, D.; Patel, N.; Kesisoglou, F. Dissolution and Translational Modeling Strategies Toward Establishing an In Vitro-In Vivo Link-a Workshop Summary Report. AAPS J 2019, 21, 29. [Google Scholar] [CrossRef] [PubMed]
- Gray, V.A.; Mann, J.C.; Barker, R.; Pepin, X. The case for physiologically based biopharmaceutics modelling (PBBM): what do dissolution scientists need to know? Dissolution Technologies 2020, 27, 6–19. [Google Scholar] [CrossRef]
- Jereb, R.; Opara, J.; Legen, I.; Petek, B.; Grabnar-Peklar, D. In vitro-In vivo Relationship and Bioequivalence Prediction for Modified-Release Capsules Based on a PBPK Absorption Model. AAPS PharmSciTech 2019, 21, 18. [Google Scholar] [CrossRef]
- Mitra, A.; Suarez-Sharp, S.; Pepin, X.J.H.; Flanagan, T.; Zhao, Y.; Kotzagiorgis, E.; Parrott, N.; Sharan, S.; Tistaert, C.; Heimbach, T.; Zolnik, B.; Sjögren, E.; Wu, F.; Anand, O.; Kakar, S.; Li, M.; Veerasingham, S.; Kijima, S.; Lima Santos, G.M.; Ning, B.; Raines, K.; Rullo, G.; Mandula, H.; Delvadia, P.; Dressman, J.; Dickinson, P.A.; Babiskin, A. Applications of Physiologically Based Biopharmaceutics Modeling (PBBM) to Support Drug Product Quality: A Workshop Summary Report. J Pharm Sci 2021, 110, 594–609. [Google Scholar] [CrossRef]
- Mueck, W.; Stampfuss, J.; Kubitza, D.; Becka, M. Clinical pharmacokinetic and pharmacodynamic profile of rivaroxaban. Clin Pharmacokinet. 2014, 53, 1–16. [Google Scholar] [CrossRef]
- Bayer. Package leaflet: information for the user, Approval by ANVISA, 2021. Available online: https://consultas.anvisa.gov.br/#/bulario/q/?nomeProduto=Xarelto. (accessed on day month year).
- Kubitza, D.; Becka, M.; Zuehlsdorf, M.; Mueck, W. Effect of food, an antacid, and the H2 antagonist ranitidine on the absorption of BAY 59-7939 (rivaroxaban), an oral, direct factor Xa inhibitor, in healthy subjects. J Clin Pharmacol 2006, 46, 549–58. [Google Scholar] [CrossRef]
- Stampfuss, J.; Kubitza, D.; Becka, M.; Mueck, W. The effect of food on the absorption and pharmacokinetics of rivaroxaban. Int J Clin Pharmacol Ther 2013, 51, 549–61. [Google Scholar] [CrossRef] [PubMed]
- Tsuruya, Y.; Nakanishi, T.; Komori, H.; Wang, X.; Ishiguro, N.; Kito, T.; Ikukawa, K.; Kishimoto, W.; Ito, S.; Schaefer, O.; Ebner, T.; Yamamura, N.; Kusuhara, H.; Tamai, I. Different Involvement of OAT in Renal Disposition of Oral Anticoagulants Rivaroxaban, Dabigatran, and Apixaban. J Pharm Sci 2017, 106, 2524–2534. [Google Scholar] [CrossRef] [PubMed]
- Shaik, A.N.; Lukacova, V.; Fraczkiewicz, G.; A physiologically based pharmacokinetic model of rivaroxaban: role of OAT3 and P-gp transporters in renal clearance. Conference W1230-05-39.2018 AAPS Annual meeting PharmSci 360, Washington D.C., USA, (4/11/2018). Available online: https://www.simulations-plus.com/assets/AAPS_2018-Rivaroxaban- Poster_AN-Shaik.pdf. (accessed on day month year).
- Çelebier, M.; Kaynak, M.S.; Altinöz, S.; Sahin, S. UV spectrophotometric method for determination of the dissolution profile of rivaroxaban. Dissolution Technologies 2014, 21, 56–59. [Google Scholar] [CrossRef]
- FDA—U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). FDA-recommended dissolution methods 2011. Available online: https://www.accessdata.fda.gov/scripts/cder/dissolution/dsp_getallData.cfm. (accessed on day month year).
- Willmann, S.; Becker, C.; Burghaus, R.; Coboeken, K.; Edginton, A.; Lippert, J.; Siegmund, H.U.; Thelen, K.; Mück, W. Development of a paediatric population-based model of the pharmacokinetics of rivaroxaban. Clin Pharmacokinet 2014, 53, 89–102. [Google Scholar] [CrossRef]
- Grillo, J.A.; Zhao, P.; Bullock, J.; Booth, B.P.; Lu, M.; Robie-Suh, K.; Berglund, E.G.; Pang, K.S.; Rahman, A.; Zhang, L.; Lesko, L.J.; Huang, S.M. Utility of a physiologically-based pharmacokinetic (PBPK) modeling approach to quantitatively predict a complex drug-drug-disease interaction scenario for rivaroxaban during the drug review process: implications for clinical practice. Biopharm Drug Dispos 2012. 33, 99–110. [CrossRef]
- Cheong, E.J.Y.; Teo, D.W.X.; Chua, D.X.Y.; Chan, E.C.Y. Systematic Development and Verification of a Physiologically Based Pharmacokinetic Model of Rivaroxaban. Drug Metab Dispos 2019, 2019. 47, 1291–1306. [Google Scholar] [CrossRef]
- Kubitza D, Becka M, Wensing G, Voith B, Zuehlsdorf M. Safety, pharmacodynamics, and pharmacokinetics of BAY 59-7939--an oral, direct Factor Xa inhibitor--after multiple dosing in healthy male subjects. Eur J Clin Pharmacol, 2005; 61, 873–800.
- Kubitza D, Becka M, Voith B, Zuehlsdorf M, Wensing G. Safety, pharmacodynamics, and pharmacokinetics of single doses of BAY 59-7939, an oral, direct factor Xa inhibitor. Clin Pharmacol Ther 2005, 78, 412–21. [Google Scholar] [CrossRef] [PubMed]
- FDA—U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Guidance for Industry. Extended-release oral dosage forms: development, evaluation, and application of in vitro/in vivo correlations. 1997. Available online: https://www.fda.gov/media/70939/download. (accessed on day month year).
- Abduljalil K, Cain T, Humphries H, Rostami-Hodjegan A. Deciding on success criteria for predictability of pharmacokinetic parameters from in vitro studies: an analysis based on in vivo observations. Drug Metabolism and Disposition, 2014; 42.
- BRAZIL. Brazilian Health Surveillane Agency (ANVISA). Resolução RE nº1170 de 19 de abril de 2006. Guia para provas de biodisponibilidade relativa/bioequivalência de medicamentos. CFAR/GTFAR/CGMED/ANVISA. 2006. Available online: http://antigo.anvisa.gov.br/documents/10181/2718376/%281%29RE_1170_2006_COMP.pdf/52326927-c379-45b4-9a7e-9c5ecabaa16b. (accessed on day month year).
- FDA—U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Guidance for industry Statistical approaches to establishing bioequivalence. 2001. Available online: https://www.fda.gov/media/70958/download. (accessed on day month year).
- Kushwah V, Arora S, Tamás Katona M, Modhave D, Fröhlich E, Paudel A. On Absorption Modeling and Food Effect Prediction of Rivaroxaban, a BCS II Drug Orally Administered as an Immediate-Release Tablet. Pharmaceutics 2021, 13, 283. [Google Scholar] [CrossRef]
- Samant TS, Lukacova V, Schmidt S. Development and qualification of physiologically based pharmacokinetic models for drugs with atypical distribution behavior: a desipramine case study. CPT Pharmacometrics Syst Pharmacol, 2017; 6, 315–321.
- Medeiros JJS, Costa TM, Carmo MP, Nascimento DD, Lauro ENC, Oliveira CA, Duque MD, Prado LD. Efficient drug development of oseltamivir capsules based on process control, bioequivalence and PBPK modeling. Drug Dev Ind Pharm, 2022; 48, 146–157.
- Issa MG, de Souza, NV, Jou BWC, Duque MD, Ferraz HG. Development of extended-release mini-tablets containing metoprolol supported by design of experiments and physiologically based biopharmaceutics modeling. Pharmaceutics, 2022; 14, 892.
- Greenblatt DJ, Patel M, Harmatz JS, Nicholson WT, Rubino CM, Chow CR. Impaired Rivaroxaban Clearance in Mild Renal Insufficiency with Verapamil Coadministration: Potential Implications for Bleeding Risk and Dose Selection. J Clin Pharmacol 2018, 58, 533–540. [Google Scholar] [CrossRef]
- Mueck W, Lensing AW, Agnelli G, Decousus H, Prandoni P, Misselwitz F. Rivaroxaban: population pharmacokinetic analyses in patients treated for acute deep-vein thrombosis and exposure simulations in patients with atrial fibrillation treated for stroke prevention. Clin Pharmacokinet 2011, 50, 675–86. [Google Scholar] [CrossRef]
- Mitra A, Petek B, Bajc A, Velagapudi R, Legen I. Physiologically based absorption modeling to predict bioequivalence of controlled release and immediate release oral products. Eur J Pharm Biopharm 2019, 134, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Wu D, Sanghavi M, Kollipara S, Ahmed T, Saini AK, Heimbach T. Physiologically based pharmacokinetics modeling in biopharmaceutics: case studies for establishing the bioequivalence safe space for innovator and generic drugs. Pharm Res, 2023; 40, 337–357.
- Lin L, Wong H. Predicting Oral Drug Absorption: Mini Review on Physiologically-Based Pharmacokinetic Models. Pharmaceutics 2017, 9, 41. [Google Scholar] [CrossRef] [PubMed]
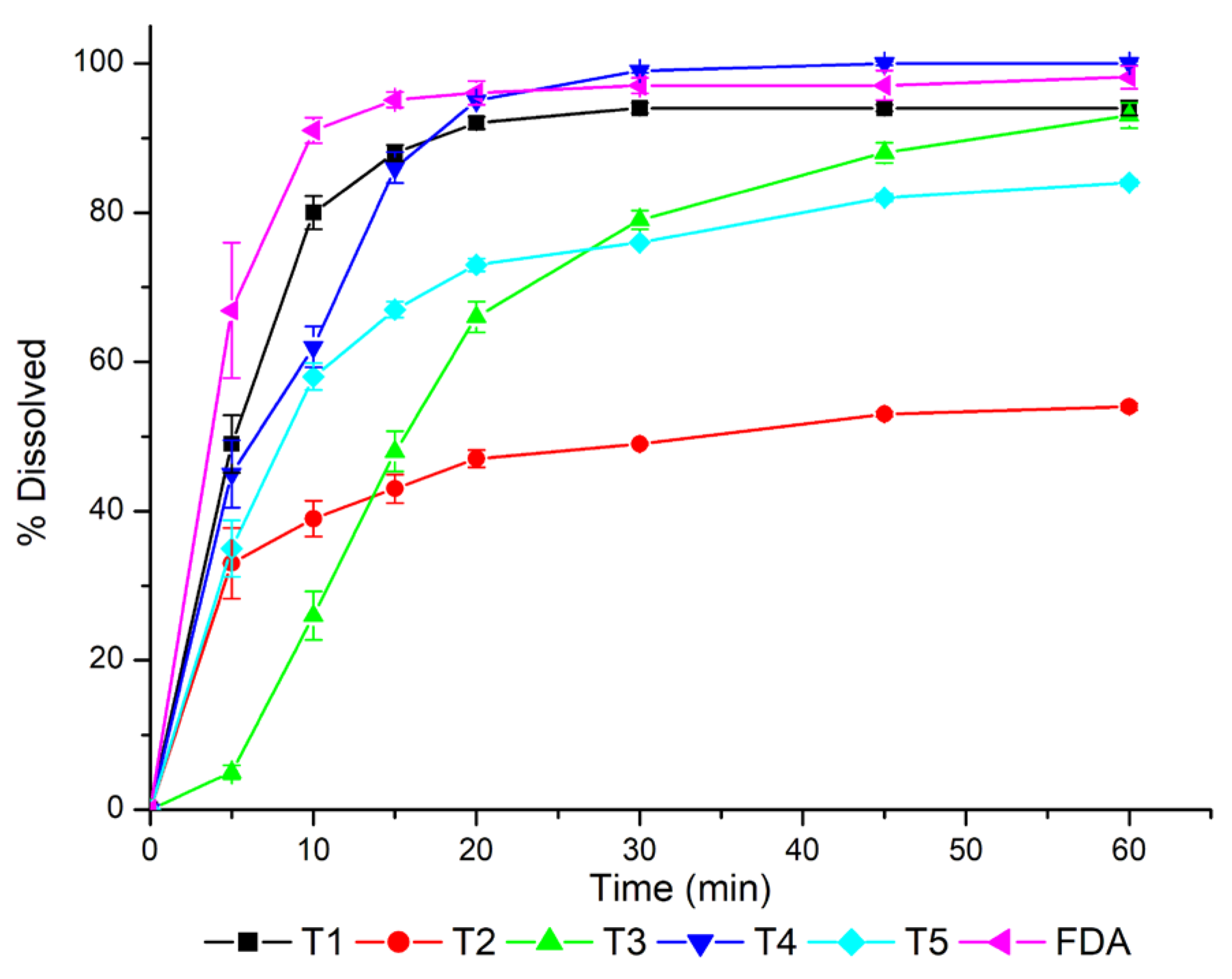
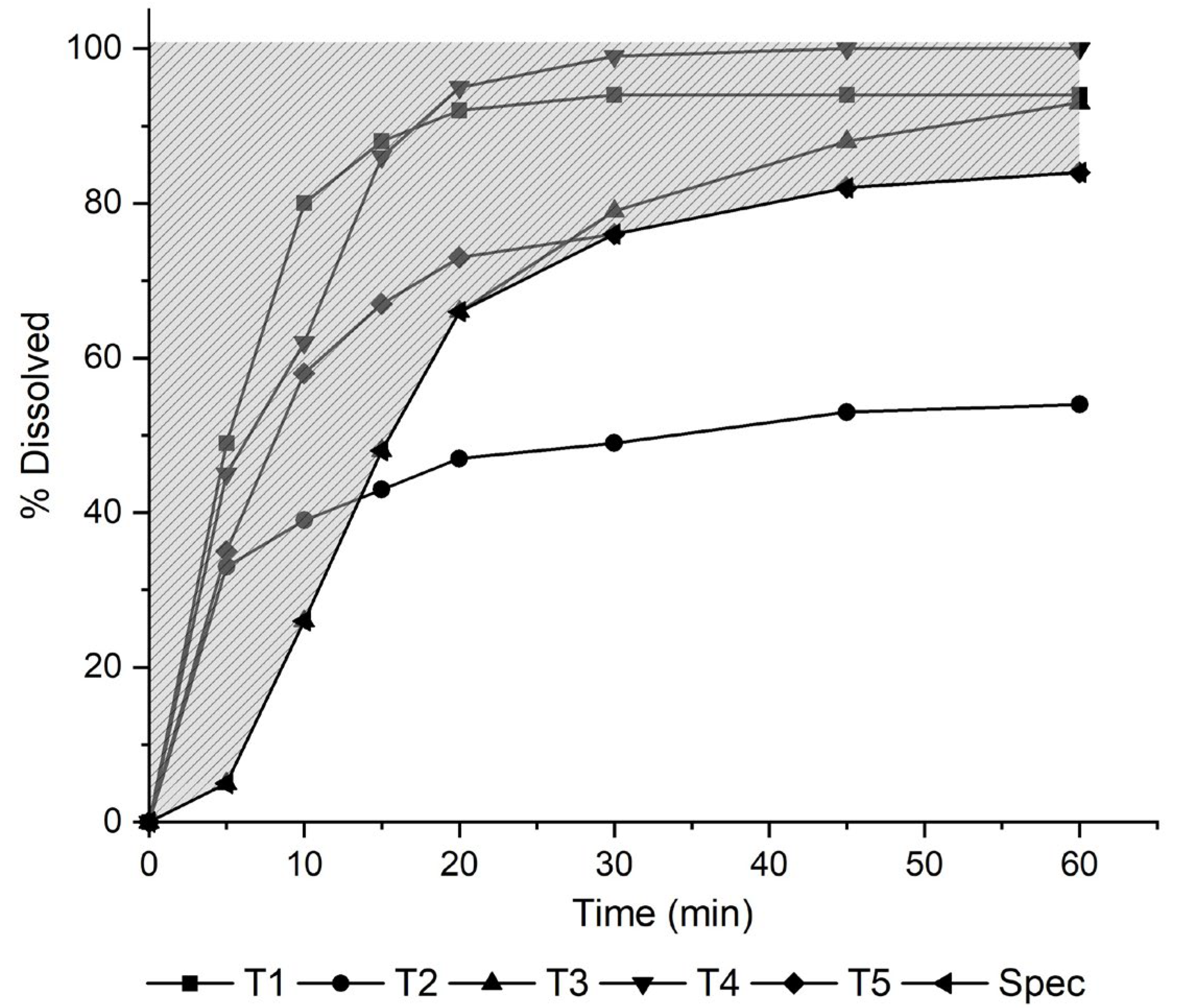
| Run | Apparatus | Rotation speed (rpm) | Volume (mL) | SDS (%) | SDS (M) |
|---|---|---|---|---|---|
| T1 | 2 | 75 | 900 | 0.25 | 0.00867 |
| T2 | 2 | 75 | 900 | 0.10 | 0.00347 |
| T3 | 2 | 50 | 900 | 0.25 | 0.00867 |
| T4 | 2 | 100 | 900 | 0.25 | 0.00867 |
| T5 | 2 | 60 | 900 | 0.15 | 0.00520 |
| Parameters | Input vales | References |
|---|---|---|
| Molecular weight | 435.89 g/mol | ADMET Predictor® |
| logP | 1.5 | [17] |
| Solubility | 0.006 mg/mL (pH 1.2, pH 4.5 and pH 6.8) | Experimental data |
| FaSSIF solubility | 0.02 mg/mL | [20] |
| FeSSIF solubility | 0.08 mg/mL | [20] |
| pKa | 10.87 | ADMET Predictor® |
| Mean particle radius | 7.79 µm | Optimized data |
| Fup 1 | 5.1% | [17] |
| B/P 2 | 0.716 | [21] |
| Peff 3 | 3.02 x 10-4 cm/s | [22] |
| Kidney OAT3 influx transporter | Vmax 4 = 4.62 x 10-7 mg/s Km 5 = 0.479 mgL |
Optimized data |
| Kidney Pgp efflux transporter | Vmax 4 = 2.13 x 10-5 mg/s Km 5 = 10.03 mg/L |
Optimized data |
| Kp 6 | Lung (0.48) Adipose (0.37) Muscle (0.77) Spleen (0.79) Heart (0.83) Brain (1.44) Skin (0.84) Reproductive organs (0.78) Red marrow (1.36) Yellow marrow (0.37) Rest of the body (0.79) Kidney (0.78) Liver (1.03) |
Predicted values using GastroPlus® |
| Absorption scale factors (ASF) – fasted state | Duodenum (1.836) Jejunum 1 (1.836) Jejunum 2 (1.836) Ileum 1 (1.836) Ileum 2 (1.836) Ileum 3 (1.836) Caecum (0.023) Ascendent colon (0.241) |
Predicted values using GastroPlus® |
| Absorption scale factors (ASF) – fed state | Duodenum (2.673) Jejunum 1 (2.658) Jejunum 2 (2.629) Ileum 1 (2.592) Ileum 2 (2.568) Ileum 3 (2.505) Caecum (0.622) Ascendent colon (1.206) |
Predicted values using GastroPlus® |
| Solubility media | Solubility (mg/mL) | CV (%) | D/S ratio (mL) |
|---|---|---|---|
| 0.1M HCl | 0.006 | 3.7 | 3333.00 |
| Acetate buffer pH 4.5 | 0.006 | 1.2 | 3333.00 |
| Phosphate buffer pH 6.8 | 0.006 | 1.3 | 3333.00 |
| Acetate buffer pH 4.5 + 0.1% SDS | 0.046 | 5.0 | 434.78 |
| Acetate buffer pH 4.5 + 0.2% SDS | 0.088 | 2.0 | 227.27 |
| Acetate buffer pH 4.5 + 0.4% SDS | 0.101 | 3.1 | 198.02 |
| Run | Time (min) | % Dissolved | Geometric ratio (%) and 90% CI | |
|---|---|---|---|---|
| Cmax | AUC0-t | |||
| T1 - Fasted | 30 | 94 | 100.6 | 100.3 |
| 45 | 94 | 92.96 – 108.85 | 90.64 – 111.03 | |
| 60 | 94 | |||
| T2 - Fasted | 30 | 49 | 67.34 | 69.21 |
| 45 | 53 | 63.26 – 71.67 | 63.79 – 75.09 | |
| 60 | 54 | |||
| T3 - Fasted | 30 | 79 | 93.46 | 93.46 |
| 45 | 88 | 87.47 – 99.87 | 85.60 – 102.05 | |
| 60 | 93 | |||
| T4 - Fasted | 30 | 99 | 100.0 | 99.77 |
| 45 | 100 | 93.62 – 106.85 | 90.90 – 109.5 | |
| 60 | 100 | |||
| T5 - Fasted | 30 | 76 | 93.83 | 94.14 |
| 45 | 82 | 86.72 – 101.53 | 84.56 – 104.79 | |
| 60 | 84 | |||
| T1 - Fed | 30 | 94 | 97.66 | 97.35 |
| 45 | 94 | 93.23 – 102.29 | 89.72 – 105.61 | |
| 60 | 94 | |||
| T2 - Fed | 30 | 49 | 65.40 | 65.40 |
| 45 | 53 | 60.70 – 70.50 | 58.20 – 73.40 | |
| 60 | 54 | |||
| T3 - Fed | 30 | 79 | 96.03 | 95.74 |
| 45 | 88 | 90.90 – 101.43 | 87.39 – 104.89 | |
| 60 | 93 | |||
| T4 - Fed | 30 | 99 | 101.40 | 101.80 |
| 45 | 100 | 95.84 – 107.36 | 93.68 – 110.61 | |
| 60 | 100 | |||
| T5 - Fed | 30 | 76 | 91.46 | 91.77 |
| 45 | 82 | 85.89 – 97.38 | 83.32 – 101.06 | |
| 60 | 84 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




