Submitted:
03 December 2024
Posted:
04 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
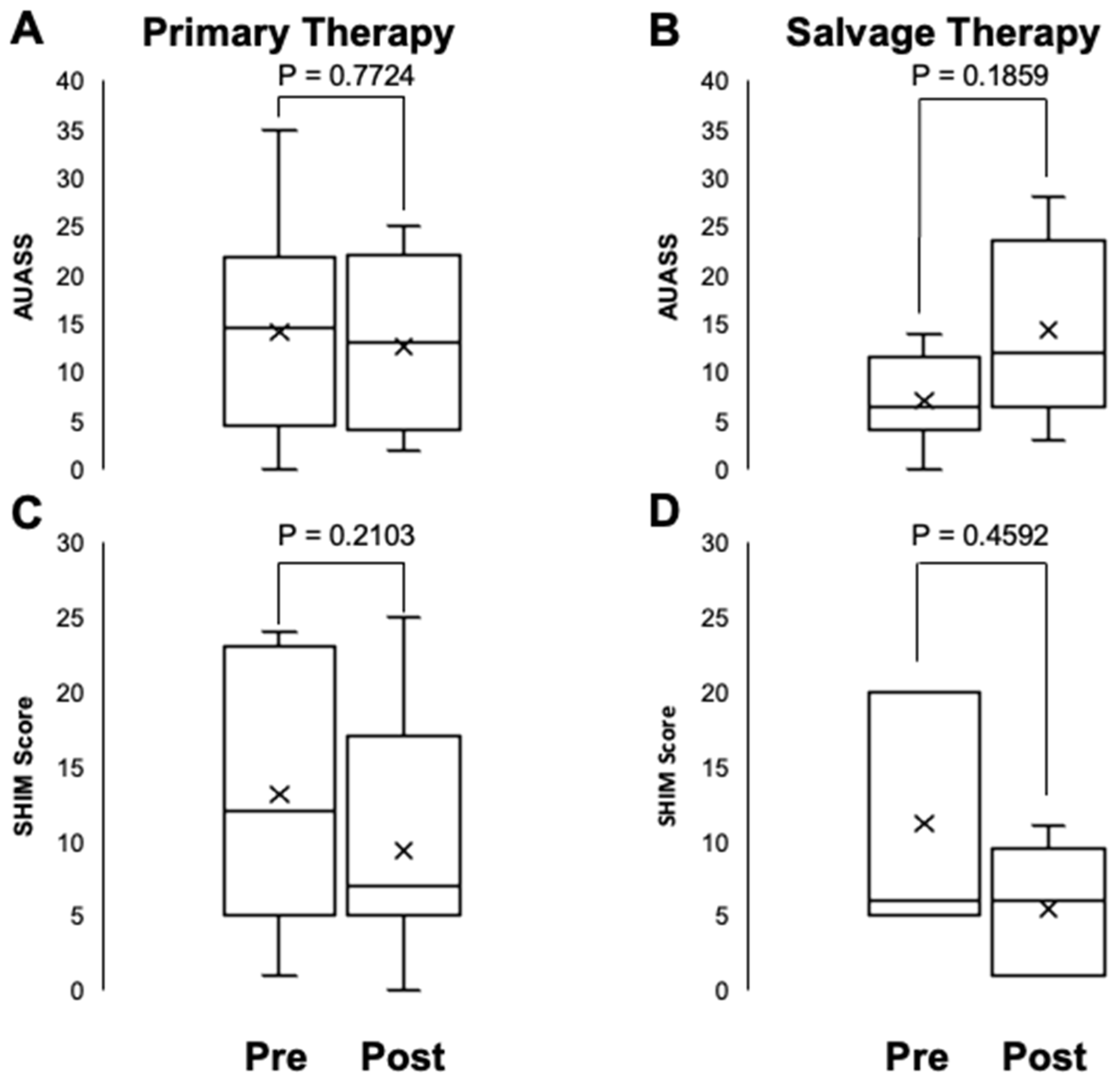
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA A Cancer J Clinicians. 2023;73(1):17-48. [CrossRef]
- Cooperberg MR, Carroll PR. Trends in Management for Patients With Localized Prostate Cancer, 1990-2013. JAMA. 2015;314(1):80. [CrossRef]
- Williams, Thomas R., Tavya G. R. Benjamin, Michael J. Schwartz, & Ardeshir R. Rastinehad. “Narrative review—focal therapy: are we ready to change the prostate cancer treatment paradigm?.” Annals of Translational Medicine [Online], 11.1 (2023): 24.
- Ong, S.; Leonardo, M.; Chengodu, T.; Bagguley, D.; Lawrentschuk, N. Irreversible Electroporation for Prostate Cancer. Life 2021, 11, 490. Author 1, A.B. (University, City, State, Country); Author 2, C. (Institute, City, State, Country). Personal communication, 2012. [CrossRef]
- Baskaran, Rengarajan, et al. “Clinical Development of Photodynamic Agents and Therapeutic Applications.” Biomaterials Research, vol. 22, no. 1, 26 Sept. 2018, Author 1, A.B. Title of Thesis. Level of Thesis, Degree-Granting University, Location of University, Date of Completion. [CrossRef]
- Jung JH, Risk MC, Goldfarb R, Reddy B, Coles B, Dahm P. Primary cryotherapy for localised or locally advanced prostate cancer. Cochrane Database Syst Rev. 2018 May 30;5(5):CD005010. [CrossRef] [PubMed] [PubMed Central]
- van Luijtelaar, A., Greenwood, B.M., Ahmed, H.U. et al. Focal laser ablation as clinical treatment of prostate cancer: report from a Delphi consensus project. World J Urol 37, 2147–2153 (2019). [CrossRef]
- Philippou Y, Parker RA, Volanis D, Gnanapragasam VJ. Comparative Oncologic and Toxicity Outcomes of Salvage Radical Prostatectomy Versus Nonsurgical Therapies for Radiorecurrent Prostate Cancer: A Meta-Regression Analysis. Eur Urol Focus. 2016 Jun;2(2):158-171. [CrossRef] [PubMed]
- Valle LF, Lehrer EJ, Markovic D, et al. A Systematic Review and Meta-analysis of Local Salvage Therapies After Radiotherapy for Prostate Cancer (MASTER). European Urology. 2021;80(3):280-292. [CrossRef]
- Hopstaken JS, Bomers JGR, Sedelaar MJP, Valerio M, Fütterer JJ, Rovers MM. An Updated Systematic Review on Focal Therapy in Localized Prostate Cancer: What Has Changed over the Past 5 Years? European Urology. 2022;81(1):5-33. [CrossRef]
- Dickinson, L., Arya, M., Afzal, N., Cathcart, P., Charman, S. C., Cornaby, A., Hindley, R. G., Lewi, H., McCartan, N., Moore, C. M., Nathan, S., Ogden, C., Persad, R., van der Meulen, J., Weir, S., Emberton, M., & Ahmed, H. U. (2016). Medium-term Outcomes after Whole-gland High-intensity Focused Ultrasound for the Treatment of Nonmetastatic Prostate Cancer from a Multicentre Registry Cohort. European urology, 70(4), 668–674. [CrossRef]
- Kaufmann, B. Kaufmann, B., Raess, E., Schmid, F.A., Bieri, U., Scherer, T.P., Elleisy, M., Donati, O.F., Rupp, N.J., Moch, H., Gorin, M.A., Mortezavi, A. and Eberli, D. (2024), Focal therapy with high-intensity focused ultrasound for prostate cancer: 3-year outcomes from a prospective trial. BJU Int, 133: 413-424. [CrossRef]
- Ghai, S.; Finelli, A.; Corr, K.; Lajkosz, K.; McCluskey, S.; Chan, R.; Gertner, M.; van der Kwast, T.H.; Incze, P.F.; Zlotta, A.R.; et al. MRI-Guided Focused Ultrasound Focal Therapy for Intermediate-Risk Prostate Cancer: Final Results from a 2-Year Phase II Clinical Trial. Radiology 2024, 310. [Google Scholar] [CrossRef] [PubMed]
- Nagaraj Y, Falkenbach F, Veleva V, Pose RM, Ekrutt J, Abrams-Pompe R, et al. MP73-02 FOCAL THERAPY - 7 YEARS EXPERIENCE WITH FOCAL HIGH INTENSITY FOCUSED ULTRASOUND IN 164 PATIENTS WITH PROSTATE CANCER: SINGLE CENTER RESULTS. Journal of Urology. 2023 Apr 1;209(Supplement 4):e1034. [CrossRef]
- Hill CR, Ter Haar GR. High intensity focused ultrasound—potential for cancer treatment. The British Journal of Radiology. 1995;68(816):1296-1303. [CrossRef]
- Sundaram K, Chang S, Penson D, Arora S. Therapeutic Ultrasound and Prostate Cancer. Semin intervent Radiol. 2017;34(02):187-200. [CrossRef]
- Taylor, J.; Patel, S.; Gaitonde, K.; Greene, K.; Liao, J.C.; McWilliams, G.; Sawyer, M.; Schroeck, F.; Alrabaa, A.; Saffati, G.; et al. The Management of Non-Muscle-Invasive Bladder Cancer in a Veteran Patient Population: Issues and Recommendations. Current Oncology 2024, 31, 6686–6698. [Google Scholar] [CrossRef] [PubMed]
- Schmid FA, Schindele D, Mortezavi A, Spitznagel T, Sulser T, Schostak M, Eberli D. Prospective multicentre study using high intensity focused ultrasound (HIFU) for the focal treatment of prostate cancer: Safety outcomes and complications. Urol Oncol 2020; 38(4):225–230.
- Basseri, S., Perlis, N. & Ghai, S. Focal therapy for prostate cancer. Abdom Radiol (2024). [CrossRef]
- Crouzet S, Blana A, Murat FJ, et al. Salvage high-intensity focused ultrasound ( HIFU ) for locally recurrent prostate cancer after failed radiation therapy: Multi-institutional analysis of 418 patients. BJU International. 2017;119(6):896-904. [CrossRef]
- He Y, Tan P, He M, et al. The primary treatment of prostate cancer with high-intensity focused ultrasound: A systematic review and meta-analysis. Medicine (Baltimore). 2020;99(41):e22610. [CrossRef]
- Autran-Gomez AM, Scarpa RM, Chin J. High-Intensity Focused Ultrasound and Cryotherapy as Salvage Treatment in Local Radio-Recurrent Prostate Cancer. Urol Int. 2012;89(4):373-379. [CrossRef]
- Hernandez KM, Venkat A, Elbers DC, et al. Prostate cancer patient stratification by molecular signatures in the Veterans Precision Oncology Data Commons. Cold Spring Harb Mol Case Stud. 2023;9(4):a006298. [CrossRef]
| Primary Therapy | Salvage Therapy | |
|---|---|---|
| No. Patients (N) | 31 | 12 |
| Median age (IQR) | 70 (64-74) | 71 (67-73) |
| Median cc prostate volume (IQR) | 33 (24-50) | 22 (16-32) |
| Median ng/ml PSA (IQR) | 6.2 (4.3-9.0) | 4.9 (4.8-8.8) |
| Median ng/ml/cc PSA density (IQR) | 0.19 (0.09-0.29) | 0.22 (0.15-0.51) |
| No. clinical stage (%) | ||
| T1c | 23 (74.2) | 10 (83.3) |
| T2a | 1 (3.2) | 0 (0.0) |
| T2b | 2 (6.5) | 1 (8.3) |
| T2c | 5 (16.1) | 1 (9.3) |
| No ISUP Grade Group (%) | ||
| 1 | 2 (6.5) | 2 (16.6) |
| 2 | 11 (35.5) | 2 (16.6) |
| 3 | 7 (22.6) | 2 (16.6) |
| 4 | 6 (19.4) | 0 (0.0) |
| 5 | 5 (16.1) | 6 (50.0) |
| Entry Biopsy | ||
| Median No. cores take (IQR) | 12 (12-12) | 12 (12-12) |
| Median pos cores (any Ca) (IQR) | 4 (2-6) | 4 (2-7) |
| Median max Ca Core % (IQR) | 50 (20-65) | 43 (24-65) |
| No. bilateral Ca on biopsy (%) | 11 (35.5) | 5 (41.7) |
| No NCCN risk group (%) | ||
| Low | 2 (6.5) | 2 (16.6) |
| Intermediate favorable | 8 (25.8) | 2 (16.6) |
| Intermediate unfavorable | 10 (32.2) | 1 (8.3) |
| High | 11 (35.5) | 7 (58.3) |
| Prior therapy (%) | ||
| Radiation | 0 (0.0) | 12 (100.0) |
| Hormonal Therapy | 0 (0.0) | 1 (8.3) |
| Median CCI (IQR) | 7 (5-8) | 5 (4-6) |
| Median BMI (IQR) | 27 (22-31) | 33 (30-34) |
| Prior abdominal surgery (%) | 9 (29.0) | 2 (16.6) |
| Primary Therapy | Salvage Therapy | |
|---|---|---|
| Median months follow-up (IQR) | 23 (15-34) | 25 (9-39) |
| Median PSA nadir (IQR) | 0.16 (0.06-2.96) | 0.12 (0.01-0.71) |
| Median time to PSA nadir, months (IQR) | 6 (3-9) | 3 (3-9) |
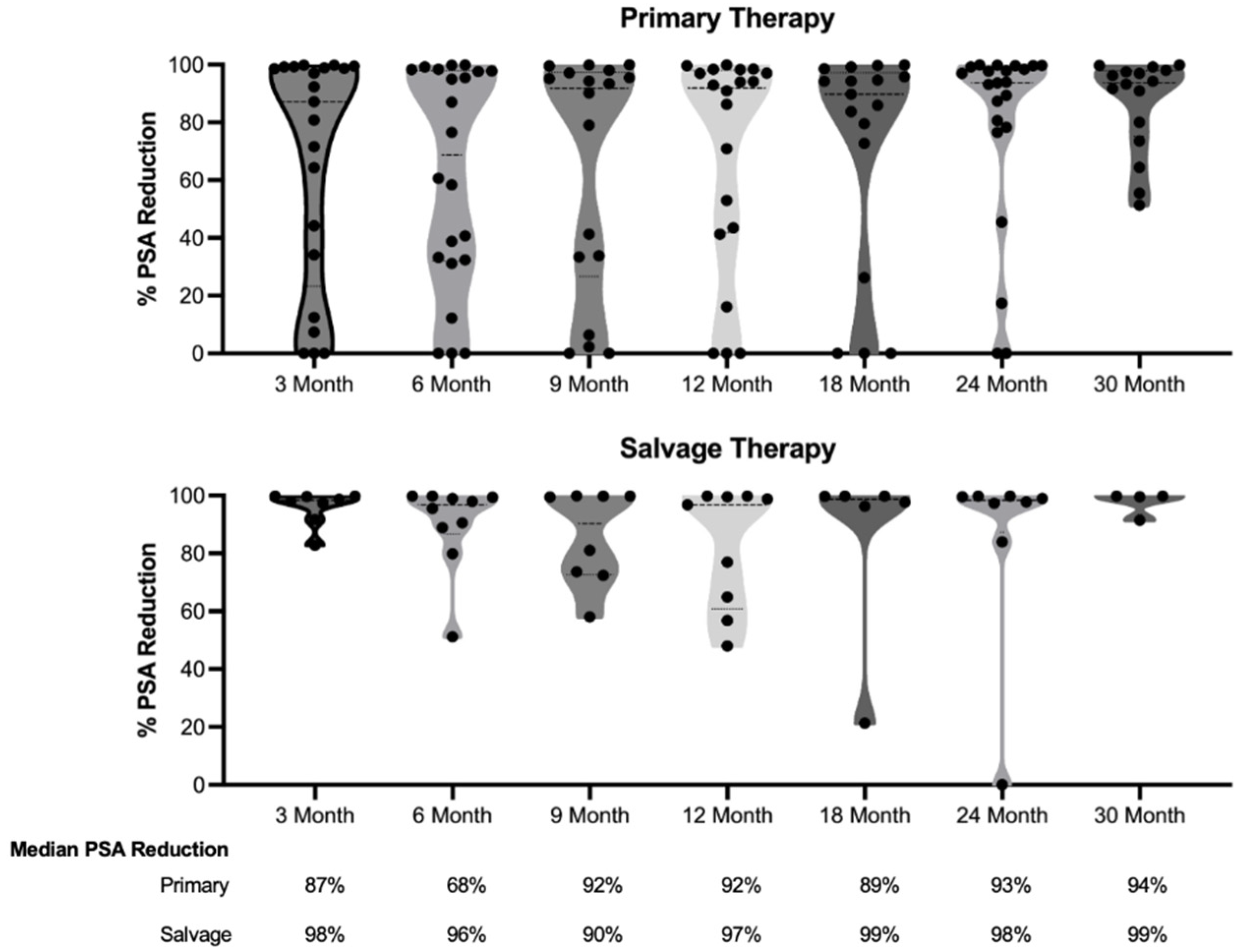
| Median % PSA decrease (IQR) | 96 (55-99) | 98 (93-99) |
| Local Recurrence (%) | 5 (16.1) | 2 (16.6) |
| Post-HIFU Treatment (%) | ||
| Radiation | 2 (6.5) | 0 (0) |
| Hormonal | 1 (3.2) | 2 (16.6) |
| HIFU | 1 (3.2) | 0 (0) |
| Radical Prostatectomy | 1 (3.2) | 0 (0) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




