1. Introduction
Melanoma is a skin tumor resulting from the malignant transformation of melanocytes. Although it accounts for less than 2% of all skin cancers, it is the key contributor to skin malignancy-related mortality, as untreated distant metastases are associated with a median survival of only 6–9 months [
1,
2].
Melanomas frequently metastasize, commencing with regional lymph node involvement, and subsequently spread to distant sites, such as other parts of the skin, lungs, liver, central nervous system, and bones [
3].
Patients with advanced metastatic or unresectable malignant melanoma are typically offered immunotherapy and targeted therapy. While chemotherapy was previously the primary treatment choice, as it does not improve survival, it is now considered appropriate only for patients who are not candidates for the aforementioned treatment modalities and for whom no suitable clinical trials are available. The aim of targeted therapies is to inhibit the mitogen-activated protein kinase (MAPK) signaling pathway components, especially in cases of its constitutive activation due to the BRAF V600E mutation [
4], which occurs in approximately 60% of melanomas [
5]. Such therapies involve intake of specific drugs, such as vemurafenib. This novel class I RAF-selective inhibitor is indicated as monotherapy in the treatment of adults with inoperable or metastatic form of melanoma with a positive BRAF V600 mutation, as it not only improves the survival rate but has also demonstrated 60% antitumor response rate in these patients [
6].
However, as melanoma can develop resistance to vemurafenib therapy within 6−7 months, it is often combined with the MEK inhibitor cobimetinib [
7]. Clinical studies have shown that this combination therapy helps counteract the mechanisms through which melanoma cells develop resistance to vemurafenib and leads to an increase in a progression-free survival from 6.2 months (with vemurafenib alone) to 9.9 months [
7].
Vemurafenib use is also associated with a range of adverse effects, which occur in 92–95% of patients and typically include erythema, maculopapular rash, photosensitivity, folliculitis, cutaneous squamous cell carcinoma, and keratoacanthoma, but may also manifest as erythema nodosum and toxic epidermal necrolysis [
8,
9,
10,
11,
12]. Clinical evidence also indicates that most of the dermatological adverse effects are caused by the inhibition of the MAPK pathway in keratinocytes, which can result in inflammatory responses, impaired keratinocyte migration, and increased keratinocyte apoptosis [
13]. While treatment discontinuation in such cases is not warranted, dose reductions or temporary therapy cessation may be required until these conditions are resolved.
More recent publications also point to a potential association between vemurafenib use and the emergence of gingival hyperplasia, with adverse impact on the patient’s quality of life owing to difficulties with mastication [
14,
15]. Given the limited data on this side effect, and even greater scarcity of reports on gingival hyperplasia in patients with metastatic melanoma receiving both vemurafenib and cobimetinib, this gap in literature has motivated the present study.
2. Case Presentation
The 59-year-old male patient in focus of this report presented at the Department of Periodontology at the Medical Faculty, University of Novi Sad, in 2019 complaining of gingival overgrowth and bleeding. The patient indicated persistent gum swelling for the preceding 6 months and associated this condition with his use of Zelboraf (vemurafenib), which was prescribed in 2018 for the treatment of malignant melanoma. The patient reported that, in 2016, malignant melanoma was diagnosed on his back and was surgically removed, without subsequent radiation treatment or chemotherapy. He attended check-ups at 3-month intervals. Initially, his condition remained stable, but after 12 months, metastases developed in the axillary lymph nodes, which were surgically removed. A few months later, metastases were found in the liver and spleen as well. Since March 2018, he has been taking Zelboraf 960 mg twice daily. Several months after commencing this treatment modality, he began to notice skin rash, followed by gum swelling, diarrhea, insomnia, nervousness, impaired vision (requiring changes to his ophthalmic prescription twice within a few months), and photosensitivity.
At the beginning of 2019, Zelboraf was discontinued for a month, leading to a marked reduction in gum enlargement and bleeding. However, the symptoms reappeared when the therapy resumed, and gradually became more pronounced.
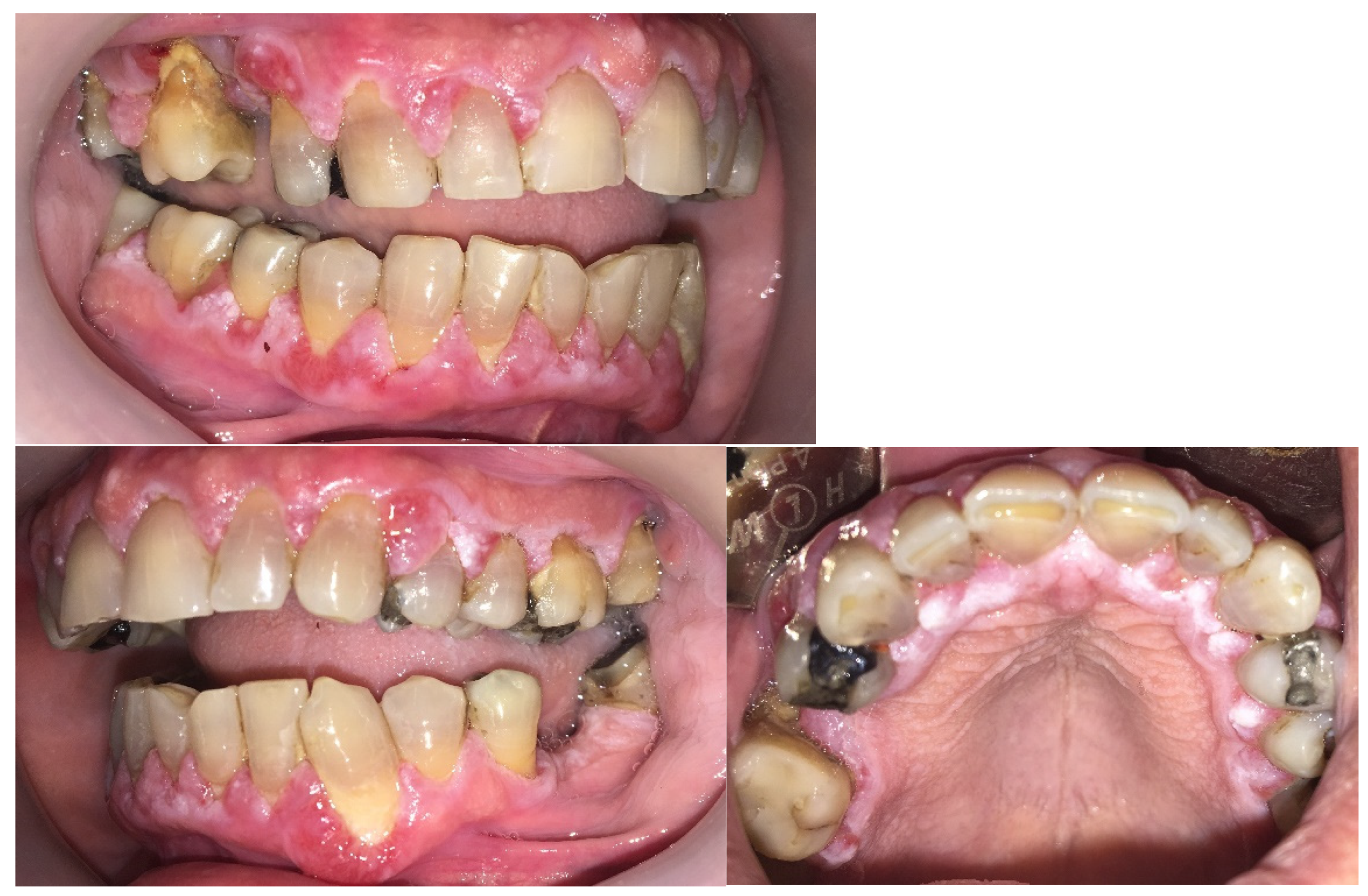
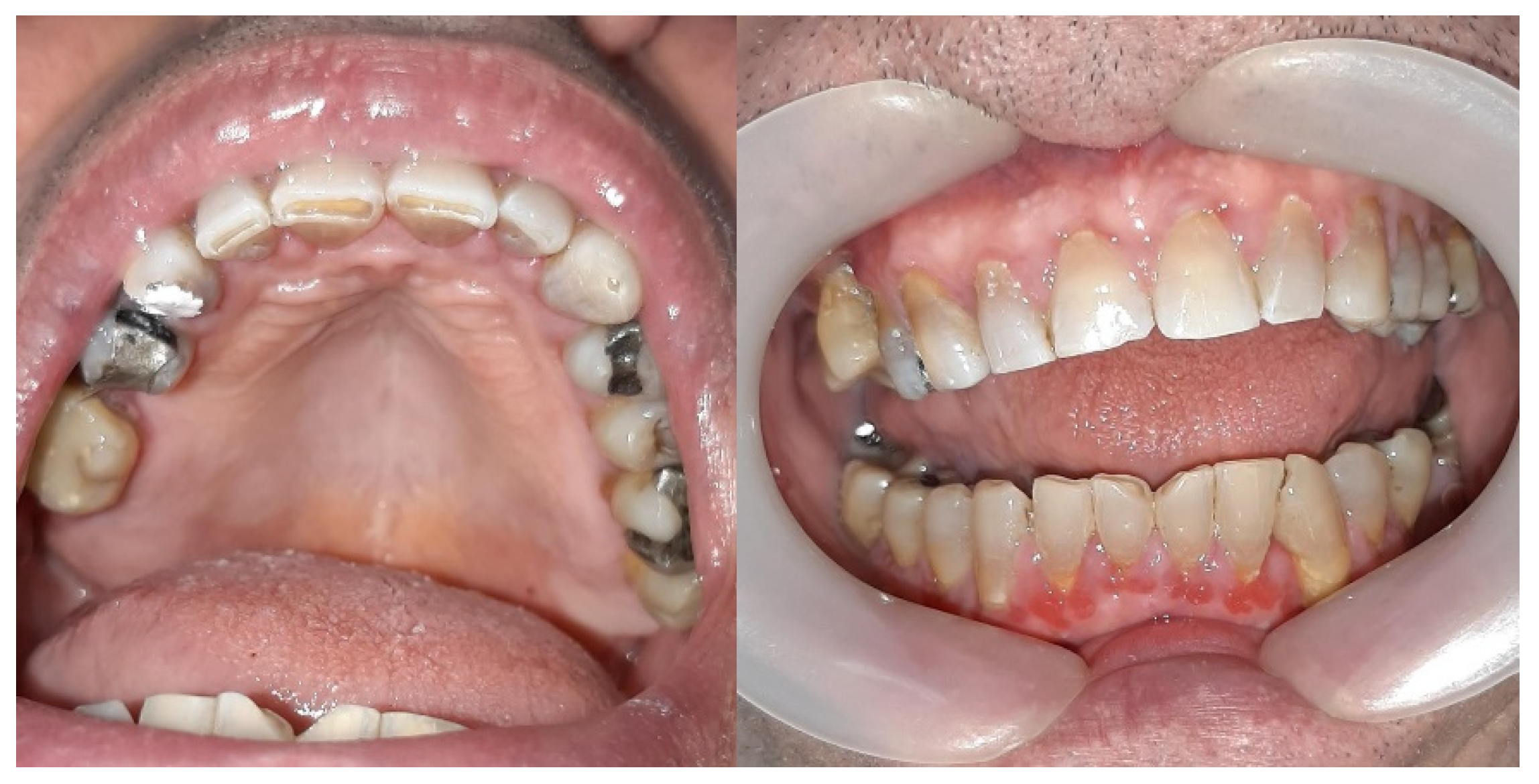
A clinical examination revealed significant gingival overgrowth around all present teeth, affecting both the vestibular and oral sides. The enlarged gums bled easily on probing and were covered with erosions and white thick plaque, which was hyperkeratotic in appearance. The patient was found to have significant supra- and subgingival tooth deposits (
Figure 1). Swabs for bacteria and fungi were negative.
To treat this condition, oral hygiene instructions were given to the patient and tooth deposits were removed, after which application of Triamcinolone 0.1% in orabase to the eroded surfaces was prescribed.
At the control examination ten days later, erosions on most of the affected surfaces were epithelized while white plaques and hyperplastic gingiva remained unchanged. Patient underwent thorough scaling and root planning and was scheduled for incisional biopsy for histopathologic examination.
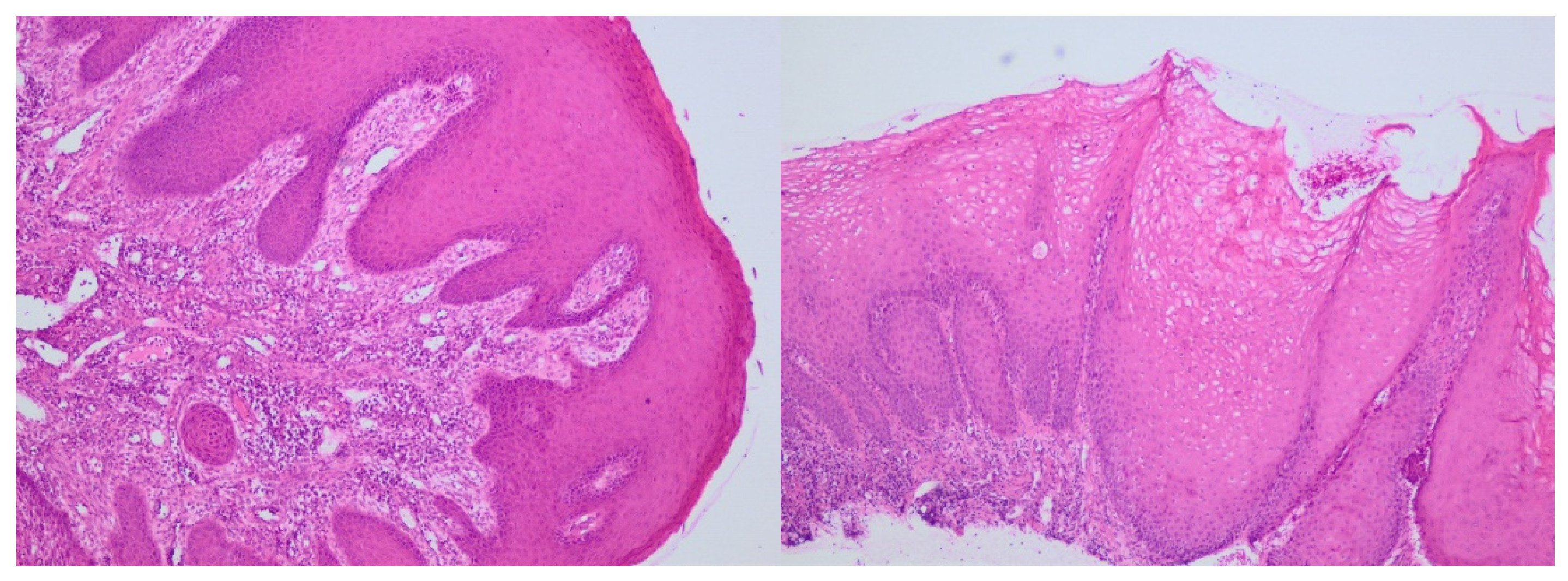
The histological report revealed stratified squamous epithelium with acanthosis, focal parakeratosis, and minor erosions. Keratinocytes exhibited vacuolated cytoplasm. In the lamina propria, there was dense infiltration of chronic inflammatory cells, including numerous granulocytes, along with dilated capillary blood vessels (
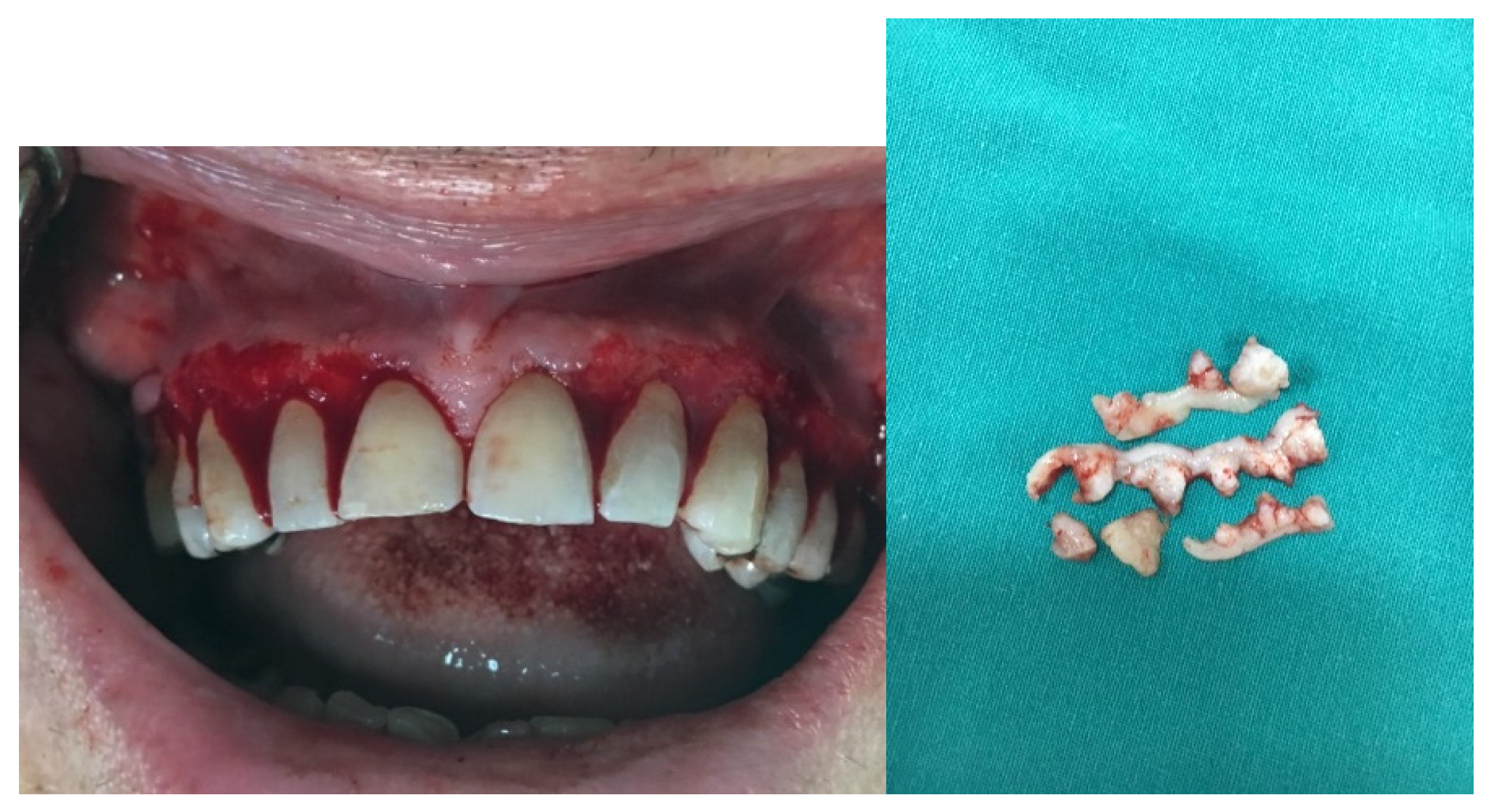
Figure 2). These findings were consistent with gingival hyperplasia, prompting surgical removal of the hyperplastic gingiva (
Figure 3).
After gingivectomy, the patient was scheduled for follow-up visits at one-month intervals. Inadequate oral hygiene, tooth deposits, and slightly edematous inflamed gums with bleeding on probing were observed at each visit. Initially, there were no signs of gingival overgrowth, but in a couple of months gingiva started swelling again (
Figure 4).
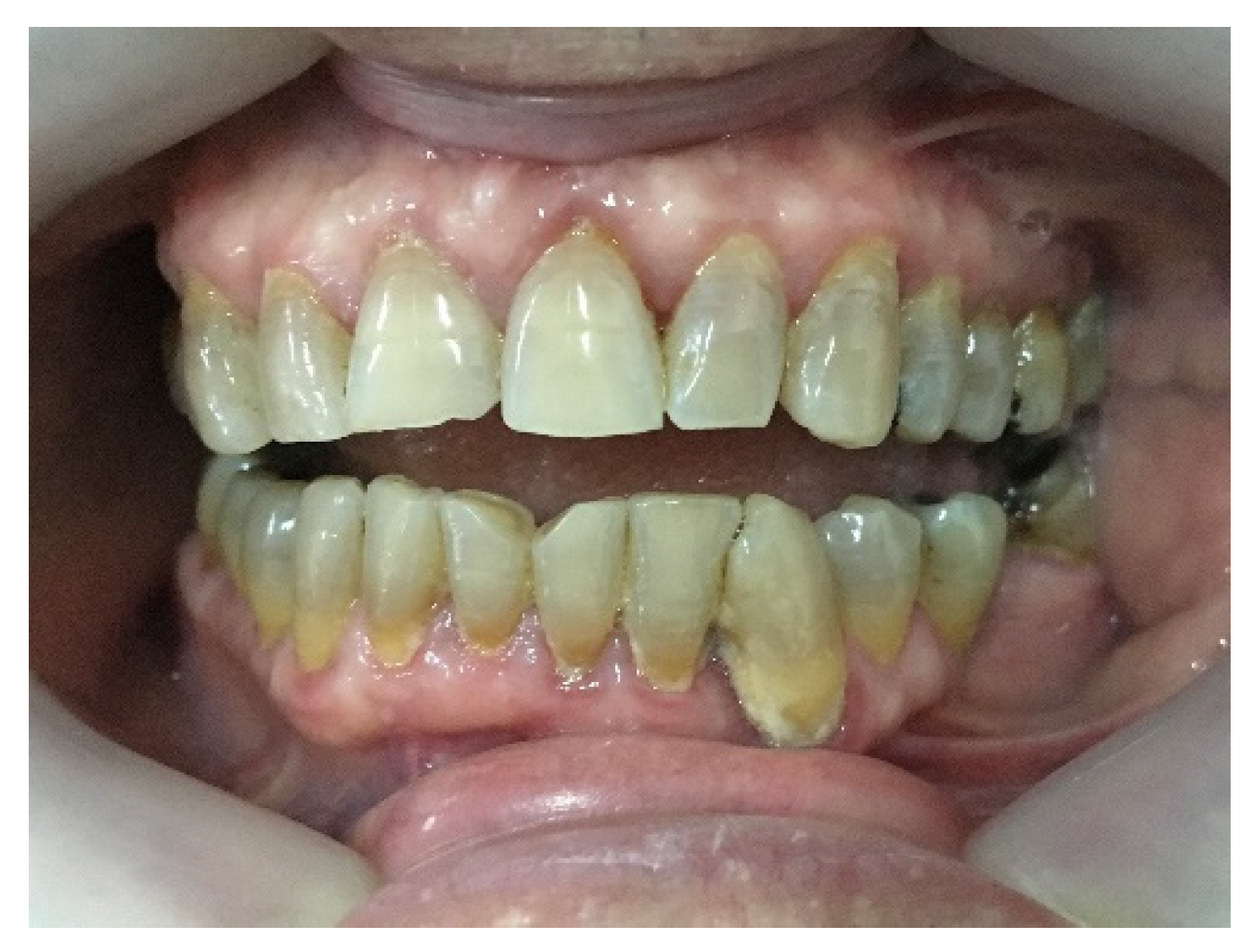
Six months after gingivectomy Zelboraf dose was reduced to 720 mg twice daily, and Cotellic (cobimetinib) was introduced at 60 mg per day, which resulted in a decrease in gingival enlargement (
Figure 5).
The treatment protocol remained unchanged until the last follow-up appointment, approximately three years after the initial visit. At this stage, gingiva condition was stable. Due to poor oral hygiene, gingiva remained inflamed and edematous, but was no longer hyperplastic and hyperkeratotic in appearance. Unfortunately, the patient soon passed away due to the underlying disease.
3. Discussion
Mangold et al. [
14] were the first to document gingival hyperplasia in a patient undergoing vemurafenib treatment for malignant melanoma. Similar to our patient, their study subject experienced a resolution of the gingival hyperplasia during 1-month temporary vemurafenib withdrawal. The authors attributed this adverse outcome to the drug’s cutaneous side-effect profile that, according to Rinderknecht et al. [
16], overlaps with the dermatological manifestations of genetic disorders associated with activating germline RAS mutations (RASopathies), such as cardiofaciocutaneous syndrome (CFC), Costello syndrome (CS), Noonan syndrome, and hereditary gingival fibromatosis [
17,
18]. Accordingly, they proposed that presence of germline RASopathies could be seen as a risk factor for the side effects associated with RAS/MAPK pathway activation. Mangold et al. [
14] thus concluded that gingival hyperplasia observed in their patient might represent an additional RASopathic adverse effect of vemurafenib therapy.
In another report on gingival hyperplasia, Salman et al. [
15] also ascribed it to vemurafenib use. However, they proposed an alternative mechanism for this association, hypothesizing that it involves increased PI3K–mTOR pathway activation, which could occur during the course of treatment, given that secondary resistance to vemurafenib develops in nearly all patients after about 6 months of therapy. Salman et al. supported this argument by highlighting that gingival hyperplasia is associated with tuberous sclerosis complex and Cowden syndrome, in which mutations leading to mTOR pathway activation are well established [
19]. This hypothesis was further substantiated by the emergence of gingival hyperplasia later in the treatment regimen, as was the case in our patient.
In addition to gingival hyperplasia, our patient exhibited whitish changes on the gingiva, which we initially suspected to be hyperkeratosis, as similar lesions have been linked to vemurafenib therapy by other authors. However, we did not obtain pathohistological confirmation of hyperkeratosis. The whitish lesions resembled those seen in white sponge nevus, a rare autosomal dominant condition characterized by extensive amounts of white, soft, and thick plaques on the oral mucosa [
20]. Despite these similarities, the observed changes were confined solely to the gingiva and the patient had no family history of that condition. While the histopathological findings did not conclusively match the typical presentation of this condition, the clinical and histopathological features were most similar to a white sponge nevus-like lesion.
Clinical examination confirmed the presence of periodontal disease in our patient. The treatment protocol involved scaling and root planing, along with gingivectomy, which yielded favorable results. However, despite the initial improvements, during follow-up visits, persistent inflammation of the gingiva was observed and was attributed to suboptimal oral hygiene.
Shepard et al. [
21] reported significant improvement in gingival appearance following intensive periodontal treatment—scaling and root planing, extraction of teeth with considerable bone loss, and rigorous maintenance of oral hygiene—in a patient with metastatic melanoma undergoing vemurafenib therapy. The authors concluded that the marked improvement in gingival clinical characteristics following the periodontal intervention was likely due to the inflammatory processes associated with periodontal disease and their interaction with vemurafenib, which is an immune-modifying drug.
Two years into vemurafenib therapy, our patient started taking Cotellic (cobimetinib). This combined therapy aimed at inhibiting the signaling pathway of the V600E-mutated BRAF protein (vemurafenib), as well as the MEK enzyme, which is involved in the signaling pathway of both V600E- and V600K-mutated BRAF proteins (cobimetinib). The patient reported a complete resolution of gingival hyperplasia within two weeks of initiating Cotellic. Although a higher incidence of side effects compared to monotherapy is associated with this treatment protocol, Larkin et al. suggested that it reduces the incidence of keratoacanthomas and cutaneous squamous-cell carcinoma, alopecia, and arthralgias [
22]. Long-term studies are nonetheless needed to evaluate the risks and toxicities associated with each agent when used in combination.
4. Conclusions
Although BRAF and MEK inhibitors are effective in treating melanoma, the oral side effects associated with their use, such as gingival hyperplasia, are often overlooked when assessing their suitability for individual patients. While the nature and intensity of these adverse reactions can differ considerably, given their potential to undermine patients’ quality of life, leading to decreased adherence to treatment protocols, there is an evident need for a multidisciplinary strategy to prevent and mitigate potential complications.
Author Contributions
Conceptualization and methodology, T.V. and M.D.; resources, J.M. and B.R.; data curation, I.G..; writing—original draft preparation, T.V.; writing—review and editing, M.D.; visualization, J.M. and I.G.; supervision, M.D and N.V. All authors have read and agreed to the published version of the manuscript
Funding
This case report received no external funding.
Institutional Review Board Statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration and approved by the Ethics Committee of the Clinic for Dentistry of Vojvodina in Novi Sad, Serbia (protocol code: 01-13/20-2024; date of approval: 25 November 2024.).
Informed Consent Statement
has been obtained from the patient to publish this paper.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- American Cancer Society. Cancer Facts & Figures 2014. Atlanta: American Cancer Society; 2014.
- Grahovac, J.; Srdić-Rajić, T.; Santibañez, J.F.; Pavlović, M.; Čavić, M.; Radulović, S. Telmisartan induces melanoma cell apoptosis and synergizes with vemurafenib in vitro by altering cell bioenergetics. Cancer Biol Med. 2019, 16, 247–63. [Google Scholar] [CrossRef] [PubMed]
- Inić, I.; Inić, M.; Inić, Z.; Zegarac, M.; Martinović, A.; Šašić, M.; Bracanović, M.; Pupić, G. Braf v600e mutations in metastatic melanoma—case report. Acta Medica Medianae. 2014, 53, 39–41. [Google Scholar] [CrossRef]
- Dean, L. Vemurafenib Therapy and BRAF and NRAS Genotype. 2017 Aug 15. In: Pratt VM, Scott SA, Pirmohamed M, et al., editors. Medical Genetics Summaries [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2012-. doi:ncbi.nlm.nih.gov/books.
- Davies, H.; Bignell, GR.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.; Bottomley, W.; Davis, N.; Dicks, E.; Ewing, R.; Floyd, Y.; Gray, K.; Hall, S.; Hawes, R.; Hughes, J.; Kosmidou, V.; Menzies, A.; Mould, C.; Parker, A, Stevens; Watt, S, Hooper; Wilson, R.; Jayatilake, H.; Gusterson, B.A.; Cooper, C.; Shipley, J.; Hargrave, D.; Pritchard-Jones, K.; Maitland, N.; Chenevix-Trench, G.; Riggins, G.J.; Bigner, D.D.; Palmieri, G.; Cossu, A.; Flanagan, A.; Nicholson, A.; Ho, J.W.; Leung, S.Y.; Yuen, S.T.; Weber, B.L.; Seigler, H.F.; Darrow, T.L.; Paterson, H.; Marais, R.; Marshall, C.J.; Wooster, R.; Stratton, M.R.; Futreal, P.A. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef]
- Boussemart, L.; Routier, E.; Mateus, C.; Opletalova, K.; Sebille, G.; Kamsu-Kom, N.; Thomas, M.; Vagner, S.; Favre, M.; Tomasic, G.; Wechsler, J.; Lacroix, L.; Robert, C. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013, 24, 1697–1703. [Google Scholar] [CrossRef] [PubMed]
- Manzano, J.L.; Layos, L.; Bugés, C.; de Los Llanos Gil, M.; Vila, L.; Martínez-Balibrea, E.; Martínez-Cardús, A. Resistant mechanisms to BRAF inhibitors in melanoma. Ann Transl Med. 2016, 4, 237. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K.T.; Puzanov, I.; Kim, K.B.; Ribas, A.; McArthur, G.A.; Sosman, J.A.; O’Dwyer, P.J.; Lee, R.J.; Grippo, J.F.; Nolop, K.; Chapman, P.B. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med 2010, 363, 809–819. [Google Scholar] [CrossRef]
- Robert, C.; Arnault, J.P.; Mateus, C. RAF inhibition and induction of cutaneous squamous cell carcinoma. Curr Opin Oncol. 2011, 23, 177–82. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Soria, J.C.; Spatz, A.; Le Cesne, A.; Malka, D.; Pautier, P.; Wechsler, J.; Lhomme, C.; Escudier, B.; Boige, V.; Armand, J.P.; Le Chevalier, T. Cutaneous side-effects of kinase inhibitors and blocking antibodies. Lancet Oncol. 2005, 6, 491–500. [Google Scholar] [CrossRef]
- McArthur, G.A.; Chapman, P.B.; Robert, C.; Larkin, J.; Haanen, J.B.; Dummer, R.; Ribas, A.; Hogg, D.; Hamid, O.; Ascierto, P.A.; Garbe, C.; Testori, A.; Maio, M.; Lorigan, P.; Lebbé, C.; Jouary, T.; Schadendorf, D.; O’Day, S.J.; Kirkwood, J.M.; Eggermont, A.M.; Dréno, B.; Sosman, J.A.; Flaherty, K.T.; Yin, M.; Caro, I; Cheng, S.; Trunzer, K.; Hauschild, A. Safety and efficacy of Vemurafenib in BRAFV600E and BRAFV600K mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomized, open-label study. Lancet Oncol 2014, 15, 323–332. [Google Scholar] [CrossRef]
- Todorovic, V.; Martinovic, D. Skin toxicity of targeted therapy: vemurafenib,first experiences from Montenegro. Sanamed. 2015, 10, 109–14. [Google Scholar] [CrossRef]
- Sinha, R.; Edmonds, K.; Newton-Bishop, J.A.; Gore, M.E.; Larkin, J.; Fearfield, L. Cutaneous adverse events associated with vemurafenib in patients with metastatic melanoma: Practical advice on diagnosis, prevention and management of the main treatment-related skin toxicities. Br. J. Dermatol. 2012, 167, 987–994. [Google Scholar] [CrossRef]
- Mangold, A.R.; Bryce, A.; Sekulic, A. Vemurafenib-associated gingival hyperplasia in patient with metastatic melanoma. J Am Acad Dermatol. 2014, 71, 205–6. [Google Scholar] [CrossRef] [PubMed]
- Salman, A.; Tekin, B.; Koca, S.; Akgul Babacan, N.; Yumuk, P.F.; Ergun, T. Another adverse effect of vemurafenib: Gingival hyperplasia. J Dermatol. 2016, 43, 706–7. [Google Scholar] [CrossRef]
- Rinderknecht, J.D; Goldinger, S.M.; Rozati, S.; Kamarashev, J.; Kerl, K.; French, L.E.; Dummer, R.; Belloni, B. RASopathic Skin Eruptions during Vemurafenib Therapy. PLoS ONE. 2013, 8(3), e58721. [Google Scholar] [CrossRef]
- Siegel, D.H.; Mann, J.A.; Krol, A.L.; Rauen, K.A. Dermatological phenotype in Costello syndrome: consequences of Ras dysregulation in development. Br J Dermatol. 2012, 166, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Siegel, D.H.; McKenzie, J.; Frieden, I.J; Rauen, K.A. Dermatological findings in 61 mutation-positive individuals with cardiofaciocutaneous syndrome. Br J Dermatol. 2011, 164, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Sparling, J.D.; Hong, C.H.; Brahim, J.S.; Moss, J.; Darling, T.N. Oral findings in 58 adults with tuberous sclerosis complex. J Am Acad Dermatol. 2007, 56, 786–90. [Google Scholar] [CrossRef]
- Elfatoiki, F.Z.; Capatas, S.; Skali, H.D.; Hali, F.; Attar, H.; Chiheb, S. Oral White Sponge Nevus: An Exceptional Differential Diagnosis in Childhood. Case Rep Dermatol Med. 2020, 9296768. [Google Scholar] [CrossRef] [PubMed]
- Shephard, M.K.; Lloyd-Lavery, A. Resolution of severe oral mucosal changes related to vemurafenib therapy with intensive periodontal treatment. Br J Dermatol. 2019, 181, 637–45. [Google Scholar] [CrossRef] [PubMed]
- Larkin, J.; Ascierto, P.A.; Dréno, B.; Atkinson, V.; Liszkay, G.; Maio., M.; Mandalà, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; de la Cruz-Merino, L.; Dutriaux, C.; Garbe, C.; Sovak, M.A.; Chang, I.; Choong, N.; Hack, S.P.; McArthur, G.A.; Ribas, A. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med 2014, 371, 1867–1876. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).