Submitted:
02 December 2024
Posted:
03 December 2024
You are already at the latest version
Abstract
Background: Herpesviruses cause a variety of infections, ranging from localized inflammation to severe disease and malignancies. The current portfolio of anti-herpes drugs is limited, often accompanied by significant side effects, and prolonged use can lead to viral resistance. This study investigates the antiviral activity of newly synthesized compounds against cytomegalovirus (CMV) and herpes simplex virus (HSV). Methods: Saturated heterocyclic compounds with annelated or spiro rings have been synthesized using either the aza-Cope–Mannich rearrangement or the alkene metathesis reaction as the key step of the transformation. Cytotoxicity was assessed using the MTT assay, while antiviral activity was measured by changes in viral titer in the presence of the compounds. Results: Several promising lead compounds were identified. A time-of-addition assay indicated that the primary targets are likely viral DNA polymerase, with inhibition leading to a halt in viral protein synthesis. Conclusions: We identified two active compounds that inhibit both herpesviruses, likely targeting the viral replicative complex.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Tested Compounds
2.2. Cell Cultures and Viruses
2.3. Assessment of Compound Cytotoxicity
2.4. Antiviral Activity Assessment
2.5. Time-of-Addition Assay
3. Results
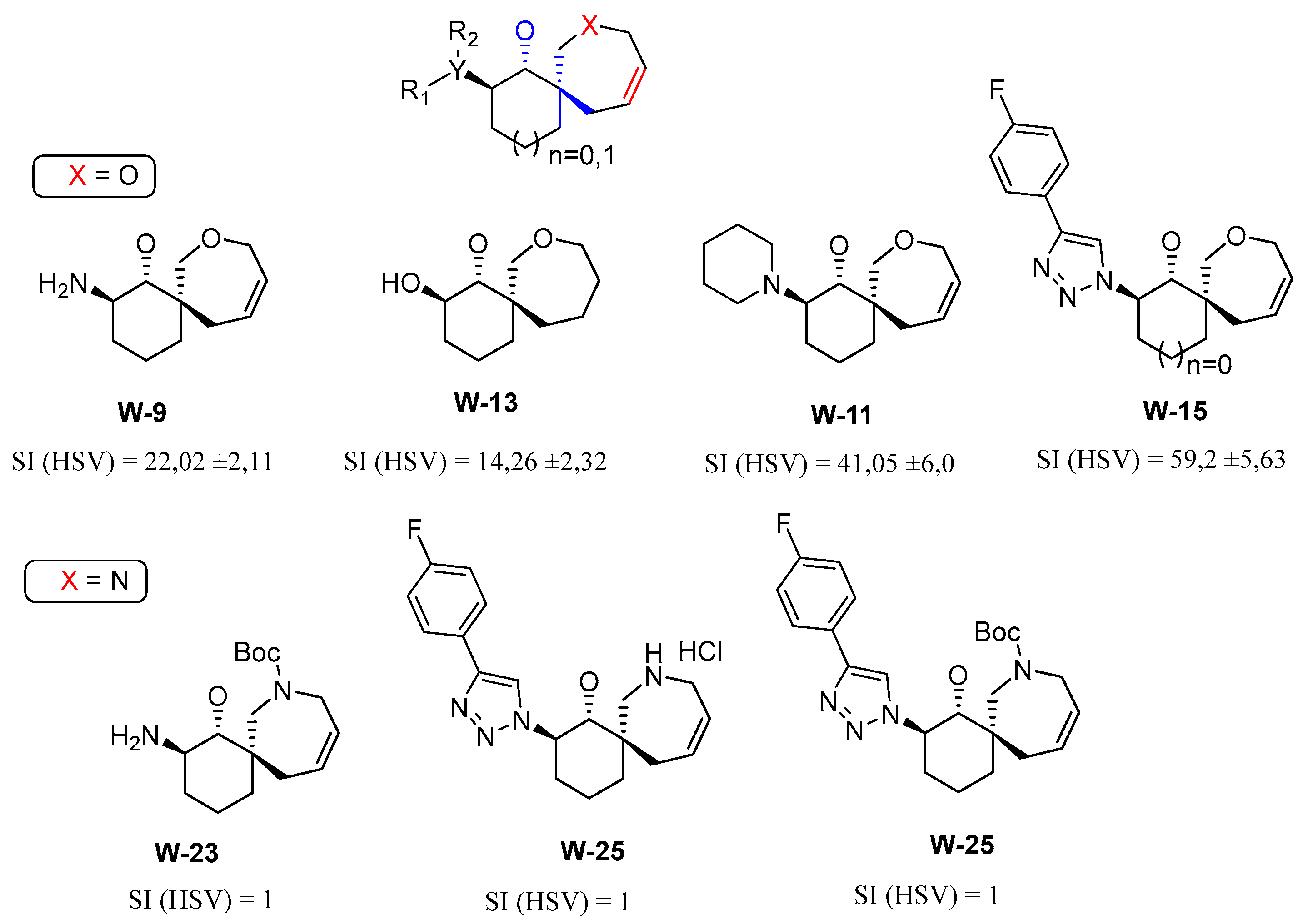
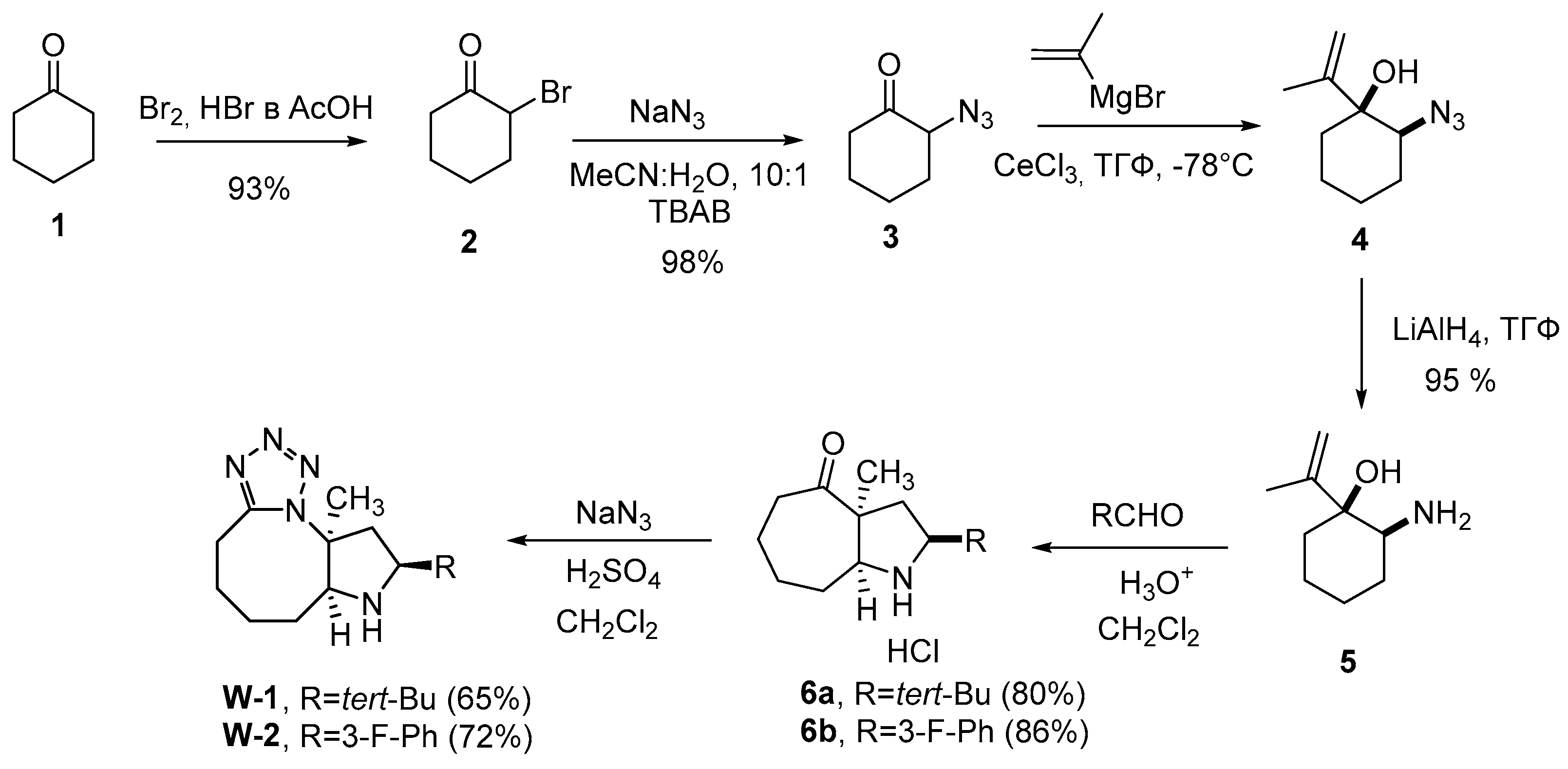
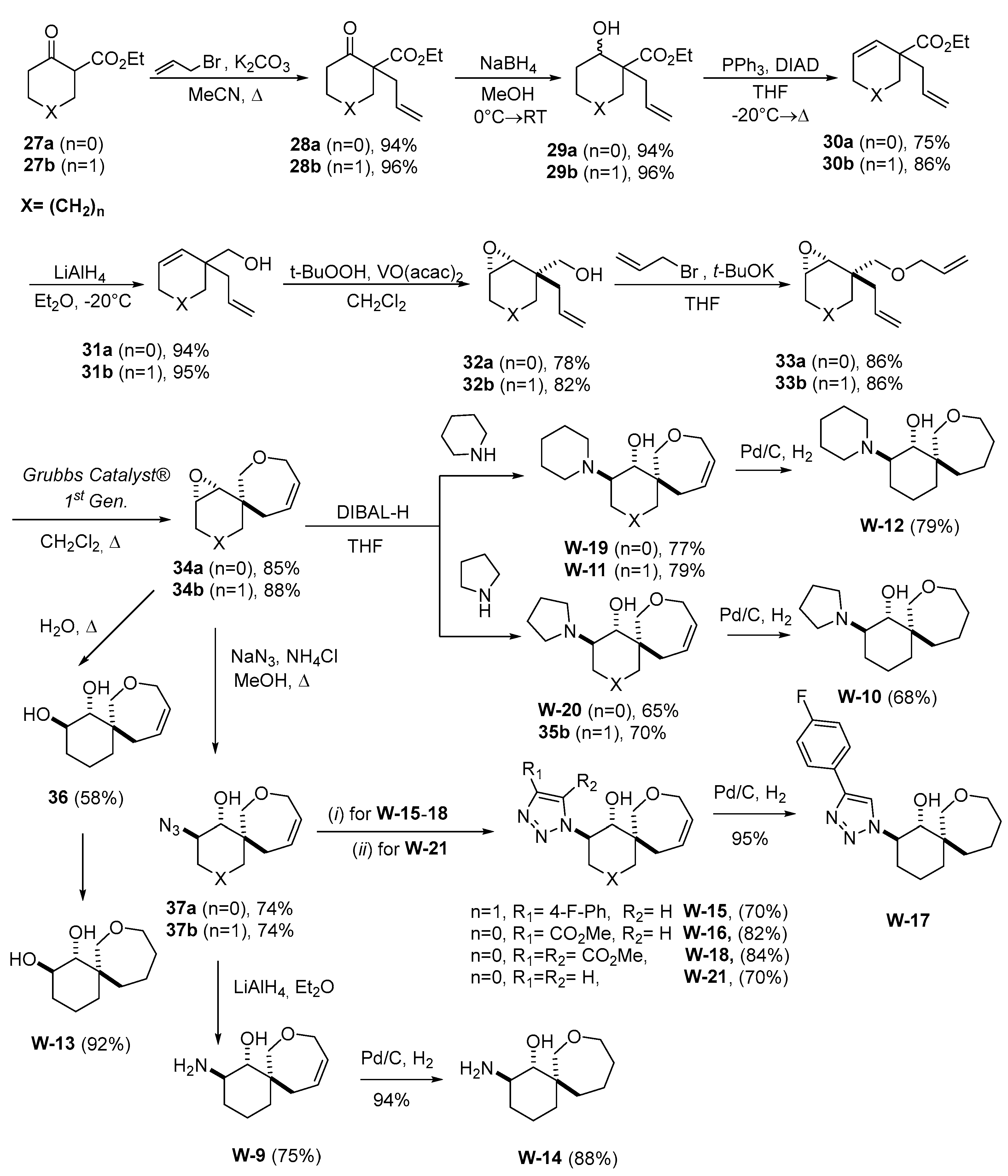
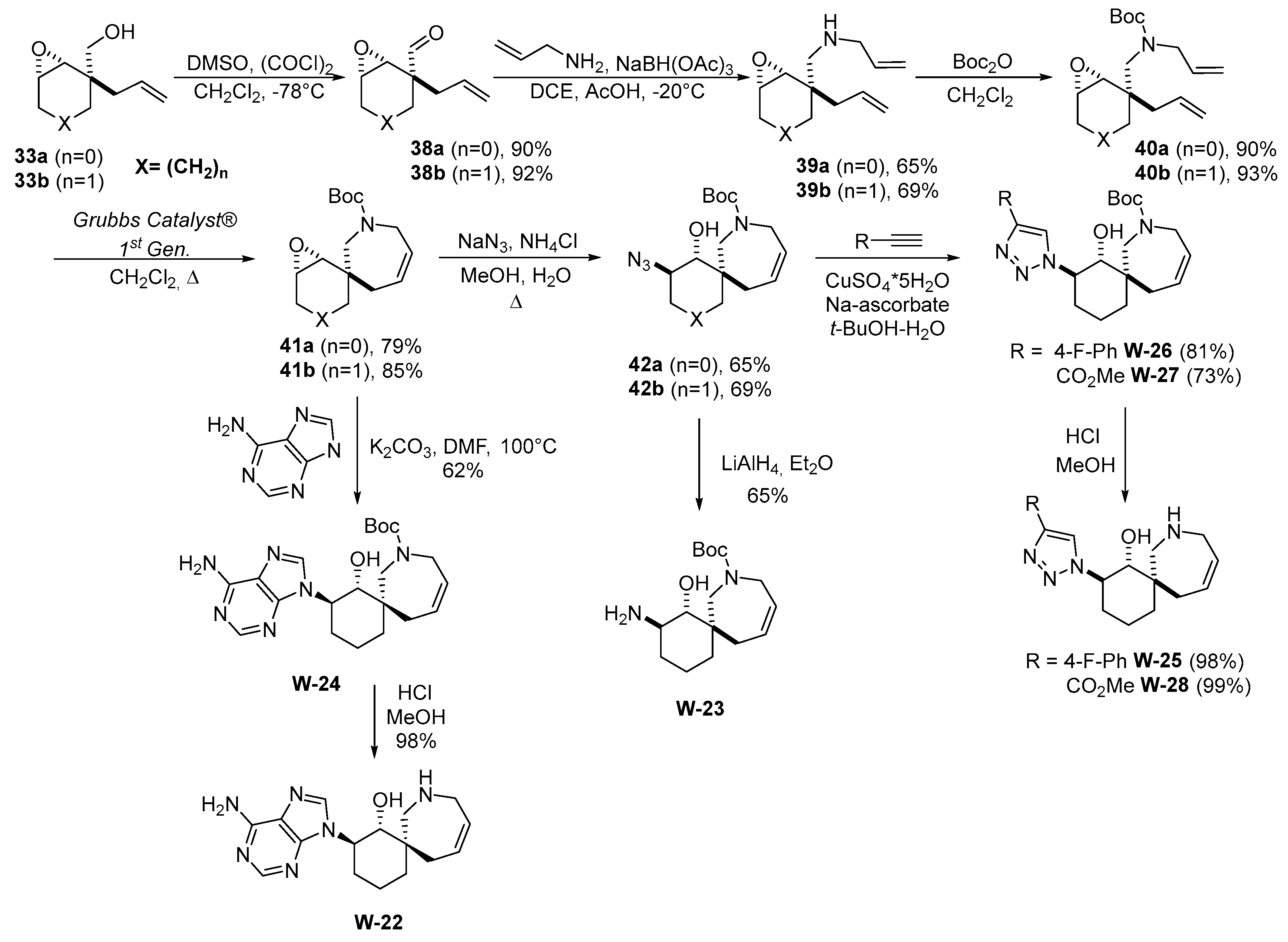
3.1. General Procedure for Synthesis of Compounds
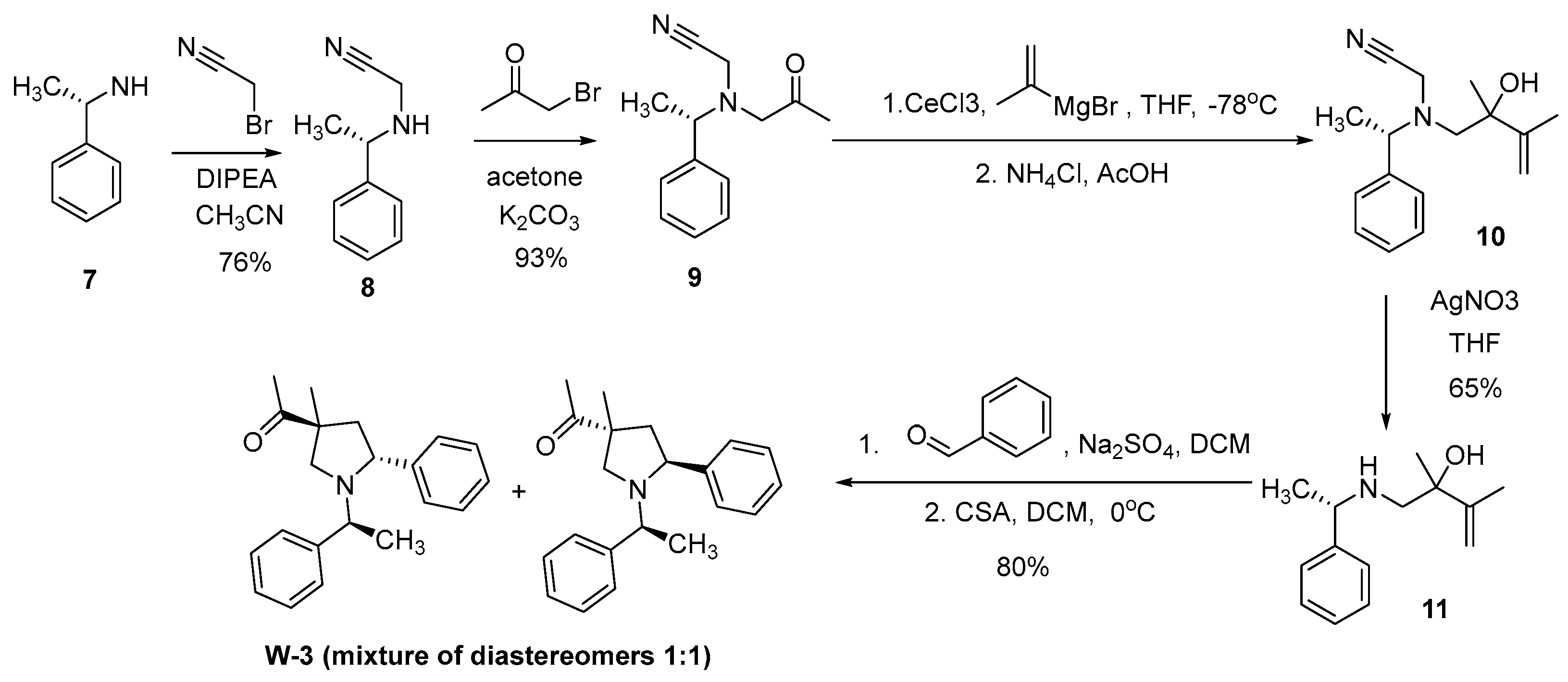
W1-W2
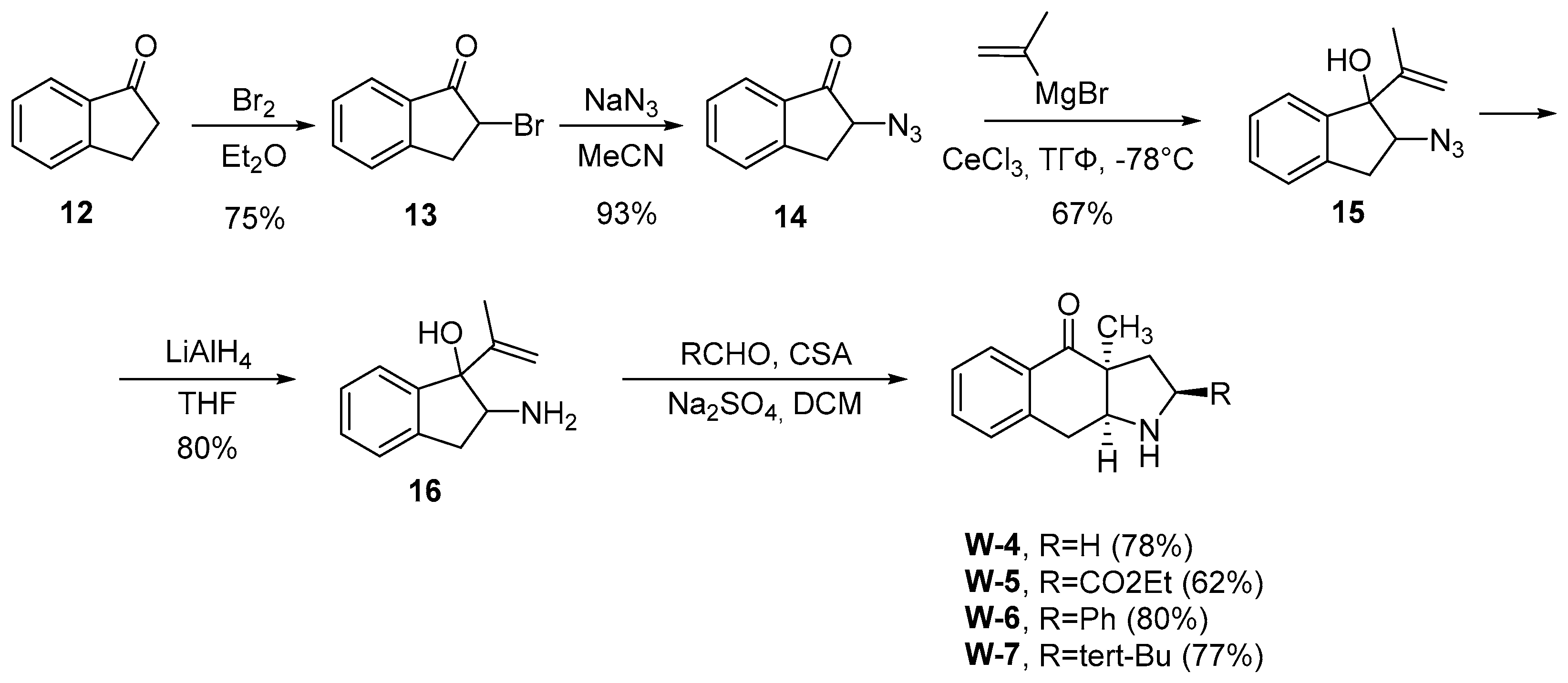
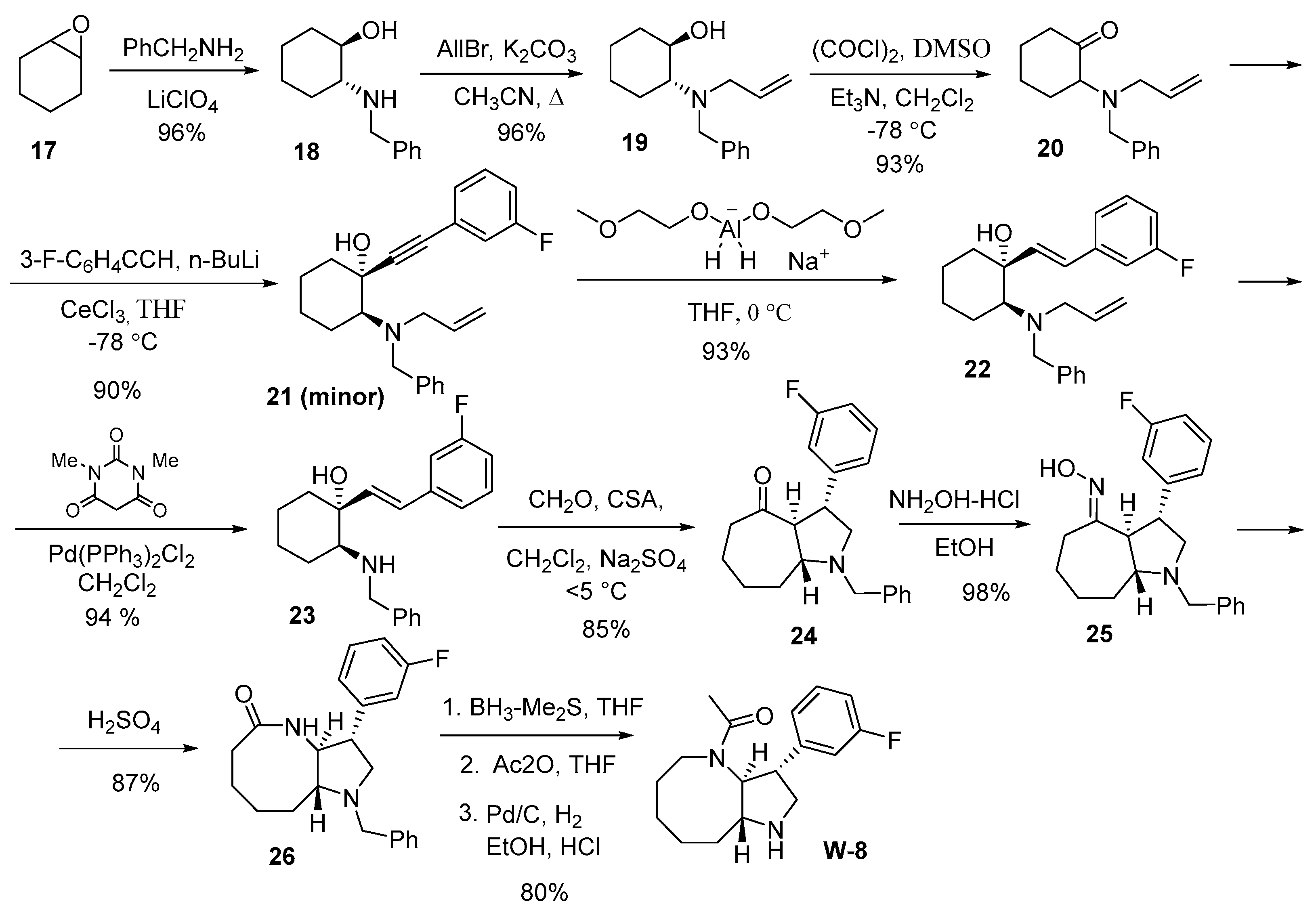
W3-W8
3.2. Cytotoxicity of Compounds on Cell Cultures
3.3. Antiviral Activity of Compounds
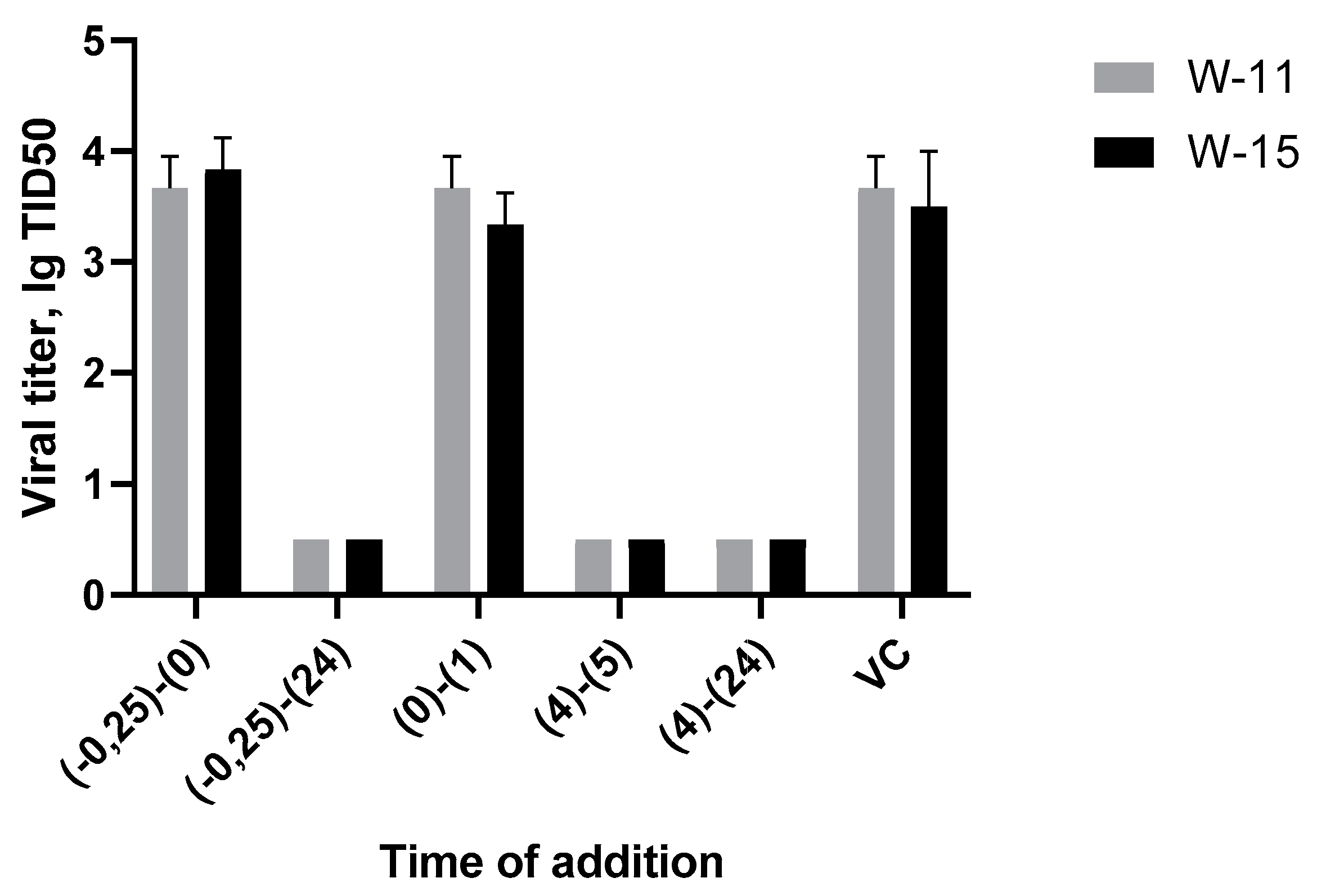
3.4. Time-of-Addition Assay Results
4. Discussion
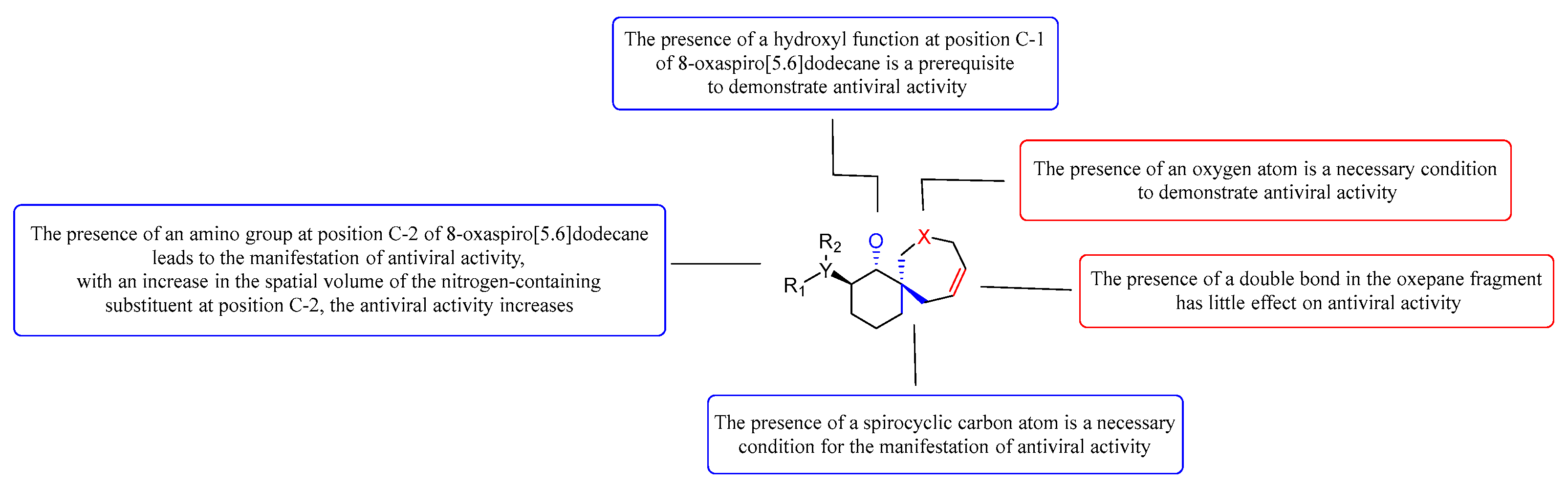
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Grinde, B. Herpesviruses: Latency and reactivation—Viral strategies and host response. Journal of oral microbiology. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Asyesha, R.; Murtaz-ul-Hasan, K.; Akhtar, N. Recent understanding of the classification and life cycle of herpesvirus: A review. Science Letters. 2017, 5, 195–207. [Google Scholar]
- Murray, P.R.; Rosenthal, K.S.; Pfaller, M.A. Medical Microbiology, 5th ed.; Elsevier Mosby: St. Louis, Missouri, USA, 2005; pp. 963 p. ISBN 978-0-323-03303-9. [Google Scholar]
- Cruz-Muñoz, M.E.; Fuentes-Pananá, E.M. Beta and Gamma Human Herpesviruses: Agonistic and Antagonistic Interactions with the Host Immune System. Front. Microbiol 2018, 8, 2521. [Google Scholar] [CrossRef] [PubMed]
- Engman, M.L.; Malm, G.; Engstrom, L.; Petersson, K.; Karltorp, E.; Tear, F.K.; Uhlen, I.; Guthenberg, C.; Lewensohn-Fuchs, I. Congenital CMV infection: Prevalence in newborns and the impact on hearing deficit. Scand J Infect Dis. 2008, 40, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Nahmias, A.J.; Walls, K.W.; Stewart, J.A.; Herrmann, K.L.; Flynt, W.J. The ToRCH complex-perinatal infections associated with toxoplasma and rubella, cytomegol- and herpes simplex viruses. Pediatr Res. 1971, 5, 405–406. [Google Scholar] [CrossRef]
- Batra, P.; Batra, M.; Singh, S. Epidemiology of TORCH Infections and Understanding the Serology in Their Diagnosis. The Journal of Maternal-Fetal Medicine. 2023, 7, 25–29. [Google Scholar] [CrossRef]
- Andronova, V.L. Modern ethiotropic chemotherapy of human cytomegalovirus infection: Clinical effectiveness, molecular mechanism of action, drug resistance, new trends and prospects. Part 1. Voprosy Virusologii (Problems of Virology, Russian journal). (In Russ). 2018, 63, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Dubonosova, E.Yu.; Namazova-Baranova, L.S.; Vishneva, E.A.; Mayanskiy, N.A.; Kulichenko, T.V.; Soloshenko, M.A. Cytomegalovirus Infection in Adolescents of Russian Federation: Results of Cross-Sectional Population Analysis of Seroprevalence. Pediatric pharmacology. (In Russ). 2021, 18, 451–459. [Google Scholar] [CrossRef]
- Hofstee, M.I.; Cevirgel, A.; de Zeeuw-Brouwer, ML.; et al. Cytomegalovirus and Epstein–Barr virus co-infected young and middle-aged adults can have an aging-related T-cell phenotype. Sci Rep. 2023, 13, 10912. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Jana, S.; Patra, S.; Haldar, P.K.; Bhowmik, R.; Mandal, A.; et al. Chapter 20 - Insights into the mechanism of action of antiviral drugs. In How Synthetic Drugs Work; Kazmi, I., Karmakar, S., Shaharyar, Md. A., Afzal, M., Al-Abbasi., *!!! REPLACE !!!*, F.A., *!!! REPLACE !!!*, Eds.; Academic Press: Cambridge, Massachusetts, USA, 2023; pp. 447–475. ISBN 9780323998550. [Google Scholar] [CrossRef]
- Britt, W.J.; Prichard, M.N. New therapies for human cytomegalovirus infections. Antiviral Res Epub 2018 Sep 15. 2018, 159, 153–174. [Google Scholar] [CrossRef] [PubMed]
- Demin, M.V.; Tikhomirov, D.S.; Biderman, B.V.; Glinshchikova, O.A.; Drokov, M.Yu.; Sudarikov, A.B.; Tupoleva, T.A.; Filatov, F.P. Mutations in the cytomegalovirus UL97 gene associated with ganciclovir resistance in recipients of allogeneic hematopoietic stem cell transplants. Kliniceskaa Mikrobiologia i Antimikrobnaa Himioterapia (In Russ.). 2019, 21, 352–357. [Google Scholar] [CrossRef]
- Obshcherossiyskaya obshchestvennaya organizatsiya sodeystviya razvitiyu neonatologii «Rossiyskoye obshchestvo neonatologov» [All-Russian public organization for promoting the development of neonatology “Russian Society of Neonatologists”], Obshchestvennaya organizatsiya «Rossiyskaya assotsiatsiya spetsialistov perinatal'noy meditsiny» [Public organization "Russian Association of Perinatal Medicine Specialists"]. Clinical recommendations Vrozhdennaya tsitomegalovirusnaya infektsiya [Congenital cytomegalovirus infection]. Moscow, 2023. 62p.
- Zarrouk, K.; Zhu, X.; Pham, V.D.; Goyette, N.; Piret, J.; Shi, R.; Boivin, G. Impact of Amino Acid Substitutions in Region II and Helix K of Herpes Simplex Virus 1 and Human Cytomegalovirus DNA Polymerases on Resistance to Foscarnet. Antimicrob Agents Chemother 2021, 65, e0039021. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zarrouk, K.; Piret, J.; Boivin, G. Herpesvirus DNA polymerases: Structures, functions and inhibitors. Virus Res 2017, 234, 177–192. [Google Scholar] [CrossRef] [PubMed]
- James, Ch.; Harfouche, M.; Welton, N.; Turner, K.; Abu-Raddad, L.; Gottlieb, S.; Looker, K. Herpes simplex virus: Global infection prevalence and incidence estimates, 2016. Bulletin of the World Health Organization 2020, 98, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Andronova, V.L. Modern ethiotropic chemotherapy of herpesvirus infections: Advances, new trends and perspectives. Alphaherpesvirinae (part I). Voprosy Virusologii (Problems of Virology, Russian journal) (In Russ). 2018, 63, 106–114. [Google Scholar] [CrossRef]
- Belen’kii, L.I. Part 13. Seven-membered Heterocyclic Rings and their Fused Derivatives. In Comprehensive Heterocyclic Chemistry III; Katritzky, A.R., Scriven, E.F.V., Ramsden, Ch.A., Taylor, R.J.K., Eds.; Elsevier Science, Ltd.: Amsterdam, Netherlands, 2008; pp. 45–95. ISBN 978-008044992-0. [Google Scholar] [CrossRef]
- Chiacchio, M.A.; Legnani, L.; Chiacchio, U.; Iannazzo, D. Chapter 16. Recent Advances on the Synthesis of Azepane-Based Compounds. In More Synthetic Approaches to Nonaromatic Nitrogen Heterocycles. Recent Advances on the Synthesis of Azepane-Based Compounds, Phillips, A.M.M.M.F.; John Wiley & Sons, Ltd: Oxford, England, 2022; pp. 529–558. ISBN 9781119757153. [Google Scholar] [CrossRef]
- Belov, D.S.; Ratmanova, N.K.; Andreev, I.A.; Kurkin, A.V. Synthesis of Bicyclic Proline Derivatives by the Aza-Cope–Mannich Reaction: Formal Synthesis of (±)-Acetylaranotin. Chem. Eur. J. 2015, 21, 4141–4147. [Google Scholar] [CrossRef] [PubMed]
- Overman, L.E.; Jacobsen, E.J.; Doedens, R.J. Synthesis Applications of Cationic Aza-Cope Rearrangements. Part 13. Stereoselective Synthesis of Cis- and Trans-3a-Aryl-4-oxodecahydrocyclohepta[b]pyrroles. J. Org. Chem. 1983, 48, 3393–3400. [Google Scholar] [CrossRef]
- Overman, L.E.; Mendelson, L.T.; Jacobsen, E.J. Synthesis Applications of Aza-Cope Rearrangements. 12. Applications of Cationic Aza-Cope Rearrangements for Alkaloid Synthesis. Stereoselective Preparation of Cis-3a-Aryloctahydroindoles and a New Short Route to Amaryllidaceae Alkaloids. J. Org. Chem. 1983, 105, 6629–6637. [Google Scholar] [CrossRef]
- Overman, L. E.; Sugai, S. Total Synthesis of (-)-Crinine. Use of Tandem Cationic Aza-Cope Rearrangement/Mannich Cyclizations for The Synthesis of Enantiomerically Pure Amaryllidaceae Alkaloids. Helv. Chim. Acta. 1985, 68, 745–749. [Google Scholar] [CrossRef]
- Guibe, F.; Grierson, D. S.; Husson, H.-P. 2-Cyano Δ3piperideine VII1: The condensation of 2-cyano Δ3piperideine with sodium dimethylmalonate catalyzed by ZnCl2 or zero valent palladium and platinum complexes. Tetrahedron Lett. 1982, 23, 5055–5058. [Google Scholar] [CrossRef]
- Jacobsen, E.J.; Levin, J.; Overman, L.E. Synthesis applications of cationic aza-Cope rearrangements. Part 18. Scope and mechanism of tandem cationic aza-Cope rearrangement-Mannich cyclization reactions. J. Am. Chem. Soc. 1988, 110, 4329–4336. [Google Scholar] [CrossRef]
- Belov, D.S.; Lukyanenko, E.R.; Kurkin, A.V.; Yurovskaya, M.A. Synthesis of (3RS,3aSR,8aSR)-3-phenyloctahydrocyclohepta[b]pyrrol-4(1H)-one via the aza-Cope–Mannich rearrangement. Tetrahedron Letters 2011, 67, 9214–9218. [Google Scholar] [CrossRef]
- Belov, D.S.; Lukyanenko, E.R.; Kurkin, A.V.; Yurovskaya, M.A. Highly stereoselective and scalable synthesis of trans-fused octahydrocyclohepta[b]pyrrol-4(1H)-ones via the aza-Cope–Mannich rearrangement in racemic and enantiopure forms. J. Org. Chem. 2012, 77, 10125–10134. [Google Scholar] [CrossRef] [PubMed]
- Wuts, P.G.M.; Greene, T.W. Protective Groups in Organic Synthesis, 4th ed.; Wiley-Blackwell: Hoboken, New Jersey, USA, 2006; pp. 1110 P. ISBN 978-0-471-69754-1. [Google Scholar]
- Garro-Helion, F.; Merzouk, A.; Guibe, F. Mild and selective palladium(0)-catalyzed deallylation of allylic amines. Allylamine and diallylamine as very convenient ammonia equivalents for the synthesis of primary amines. J. Org. Chem. 1993, 58, 6109–6113. [Google Scholar] [CrossRef]
- Schiffers, I.; Bolm, C. Synthesis and resolution of racemic trans-2-(N-benzyl)amino-1-cyclohexanol: Enantiomer separation by sequential use of (R)- and (S)-mandelic acid. Org. Synth. 2008, 85, 106–117. [Google Scholar] [CrossRef]
- Mancuso, A. J.; Huang, S. L.; Swern, D. J. Oxidation of long-chain and related alcohols to carbonyls by dimethyl sulfoxide "activated" by oxalyl chloride. Org. Chem. 1978, 43, 2480–2482. [Google Scholar] [CrossRef]
- Iusupov, I.R.; Lukyanenko, E.R.; Altieri, A.; Kurkin, A.V. Design and Synthesis of Fsp3-Enriched Spirocyclic-Based Biological Screening Compound Arrays via DOS Strategies and Their NNMT Inhibition Profiling. ChemMedChem. 2022, 17, e202200394. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.A.M.; Teixeira, L.H.P.; Menezes, C.M.S.; Sant′Anna, C.M.R.; Conceição, M.; Ramos, K.V.; Neto, F.R.A.; Barreiro, E.J. Studies on diastereoselective reduction of cyclic β-ketoesters with boron hydrides. Part 4: The reductive profile of functionalized cyclohexanone derivatives. Tetrahedron 2004, 60, 2745–2755. [Google Scholar] [CrossRef]
- Iusupov, I.R.; Lyssenko, K.A.; Altieri, A.; Kurkin, A.V. (1RS,2RS,6RS)-2-(6-Amino-9H-purin-9-yl)-8-azaspiro[5.6]dodec-10-en-1-ol Dihydrochloride. Molbank 2022, 2022, M1495. [Google Scholar] [CrossRef]
- Morten, M.; Frederik, D. Recent Fascinating Aspects of the CuAAC Click Reaction. Trends in Chemistry 2020, 2, 569–584. [Google Scholar] [CrossRef]
- Osolodkin, D.I.; Kozlovskaya, L.I.; Iusupov, I.R.; Kurkin, A.V.; Shustova, E.Y.; Orlov, A.A.; Khvatov, E.V.; Mutnykh, E.S.; Kurashova, S.S.; Vetrova, A.N.; Yatsenko, D.O.; Goryashchenko, A.S.; Ivanov, V.N.; Lukyanenko, E.R.; Karpova, E.V.; Stepanova, D.A.; Volok, S.; Sotskova, E.; Tamara, K.; Dzagurova, G.; Karganova, G.; Alexander, N.; Lukashev, V.P.; Ishmukhametov, A.A. Phenotypic assessment of antiviral activity for spiro-annulated oxepanes and azepenes. Chem Biol Drug Des. 2024, 103, e14553. [Google Scholar] [CrossRef] [PubMed]
| Compound | CC50, µg/ml | |
|---|---|---|
| Vero | MRC-5 | |
| W-1 | 146,0 | 79,9 |
| W-2 | 214,8 | 75,1 |
| W-3 | 90,74 | 13,3 |
| W-4 | 294,2 | 119 |
| W-5 | 473,4 | 106,8 |
| W-6 | 251,7 | 83,7 |
| W-7 | 262,4 | 114,9 |
| W-8 | 417,3 | 134,7 |
| W-9 | 473,5 | 152,8 |
| W-10 | 299,4 | 108,9 |
| W-11 | 513,1 | 83,3 |
| W-12 | 467,8 | 80 |
| W-13 | 570,2 | 90,9 |
| W-14 | 346,7 | 96,5 |
| W-15 | 749,5 | 606,3 |
| W-16 | 543,1 | 1346 |
| W-17 | 200,2 | 816,6 |
| W-18 | 596,2 | 1055 |
| W-19 | 723,9 | 1074 |
| W-20 | 331,3 | 726,7 |
| W-21 | 290,9 | 1152 |
| W-22 | 379,2 | 620 |
| W-23 | 111,6 | 142,2 |
| W-24 | 140 | 537,2 |
| W-25 | 185 | 118,7 |
| W-26 | 327,2 | 292,5 |
| W-27 | 224,1 | 238,5 |
| W-28 | 295,7 | 238,1 |
| Compound | CMV | HSV-1 | ||
| IC50, µg/ml | SI | IC50, µg/ml | SI | |
| W-1 | 15,9±1.3 | 5,03 | >146,0 | 1 |
| W-2 | 17,7±2.3 | 4,24 | >214,8 | 1 |
| W-3 | 0,97±0.03 | 13,7 | >90,74 | 1 |
| W-4 | 22,95±5.6 | 5,19 | >294,2 | 1 |
| W-5 | 45,7±8.9 | 2,34 | >473,4 | 1 |
| W-6 | 165,5±12.8 | 0,51 | >251,7 | 1 |
| W-7 | 22,67±3.4 | 5,07 | >262,4 | 1 |
| W-8 | 82,7±7.6 | 1,63 | >417,3 | 1 |
| W-9 | 93,3±10.1 | 1,64 | 19,9±2.2 | 22,02 |
| W-10 | 3,04±0.9 | 44,75 | >299,4 | 1 |
| W-11 | 0,97±0.01 | 97,2 | 12,5±3.2 | 41,05 |
| W-12 | >80 | 1 | >467,8 | 1 |
| W-13 | 82,9±22 | 1,1 | 40±5.1 | 14,25 |
| W-14 | 27,1±3.5 | 3,56 | >346,7 | 1 |
| W-15 | 47,3±5.9 | 12,82 | 12,8±2.6 | 59,2 |
| W-16 | >1346 | 1 | 381 ± | 1,4 |
| W-17 | >816.6 | 1 | >200,2 | 1 |
| W-18 | >1055 | 1 | >596,2 | 1 |
| W-19 | >1074 | 1 | >723,9 | 1 |
| W-20 | >726.7 | 1 | >331,3 | 1 |
| W-21 | >1152 | 1 | >290,9 | 1 |
| W-22 | >620 | 1 | >379,2 | 1 |
| W-23 | >142.2 | 1 | >111,6 | 1 |
| W-24 | >537.2 | 1 | >140 | 1 |
| W-25 | >118.7 | 1 | >185 | 1 |
| W-26 | >292.5 | 1 | >327,2 | 1 |
| W-27 | >238.5 | 1 | >224,1 | 1 |
| W-28 | >238.1 | 1 | >295,7 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




