Submitted:
25 November 2024
Posted:
25 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Main Parameters to Be Optimized for Intraoperative Monitoring of Sensory Evoked Potentials
3. Somatosensory Evoked Potentials
4. Visual Evoked Potentials
5. Brainstem Acoustic Evoked Potentials
6. Problems in the Implementation of a Personalized Approach to Intraoperative Sensory EPs Monitoring and Their Possible Solutions
7. Conclusions
8. Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
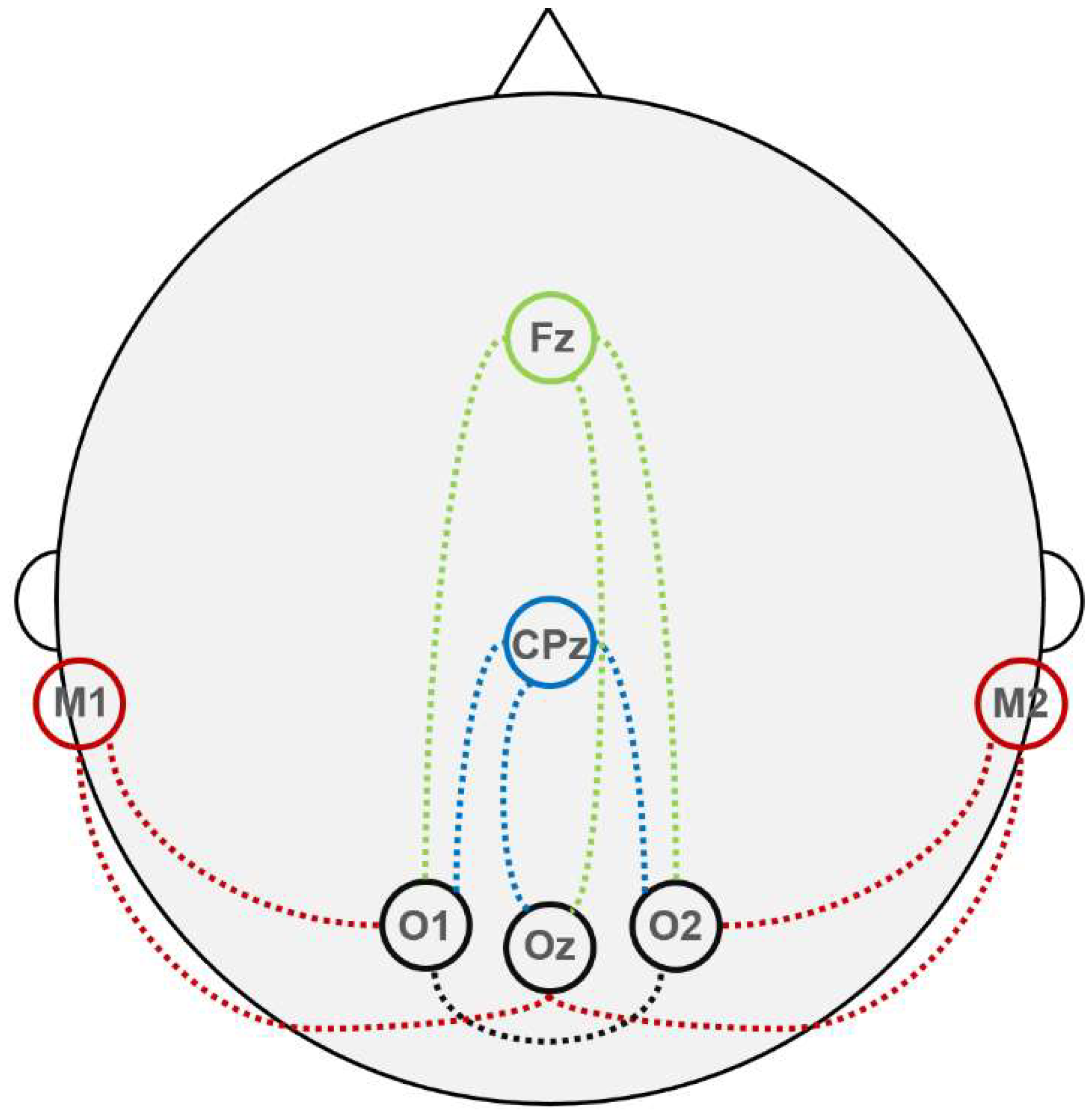
| 1 | From here on, when considering SSEPs and BAEPs, the locations of the electrodes along the fronto-occipital axis are designated in accordance with the 10–10 system [18]. For electrodes along the midline, the designations are completely consistent with this system (e.g., CPz). For electrodes located lateral to the midline, instead of numerical values encoding the side (odd numbers – left side, even – right) and distance from the midline (a higher number corresponds to a greater angular distance), the designations i and c (e.g., CPi and CPc) will be used meaning the hemisphere ipsi- and contralateral to the stimulation side, respectively. Here, they correspond to electrodes with a numerical codes of 3 or 4 (20% distance from the midline). In the VEPs section, all electrode location designations are the same as in [18]. |
References
- Toleikis, J. R.; Pace, C.; Jahangiri, F. R.; Hemmer, L. B.; Toleikis, S. C. Intraoperative somatosensory evoked potential (SEP) monitoring: an updated position statement by the American Society of Neurophysiological Monitoring. Journal of Clinical Monitoring and Computing 2024, 38, 1003–1042. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Regli, L.; Bozinov, O.; Sarnthein, J. Clinical utility and limitations of intraoperative monitoring of visual evoked potentials. PLoS ONE 2015, 10, e0120525. [Google Scholar] [CrossRef] [PubMed]
- American Clinical Neurophysiology Society. Guideline 11C: Recommended standards for intraoperative monitoring of auditory evoked potentials. Available online: https://www.acns.org/pdf/guidelines/Guideline-11C.pdf (accessed on 20 November 2024).
- Simon, M. V. Neurophysiologic intraoperative monitoring of the vestibulocochlear nerve. Journal of Clinical Neurophysiology 2011, 28, 566–581. [Google Scholar] [CrossRef] [PubMed]
- Fridman, J.; John, E. R.; Bergelson, M.; Kaiser, J. B.; Baird, H. W. Application of digital filtering and automatic peak detection to brain stem auditory evoked potential. Electroencephalography and Clinical Neurophysiology 1982, 53, 405–416. [Google Scholar] [CrossRef]
- MacDonald, D. B. Individually optimizing posterior tibial somatosensory evoked potential P37 scalp derivations for intraoperative monitoring. Journal of Clinical Neurophysiology 2001, 18, 364–371. [Google Scholar] [CrossRef]
- Scherg, M.; Berg, P.; Nakasato, N.; Beniczky, S. Taking the EEG back into the brain: the power of multiple discrete sources. Frontiers in Neurology 2019, 10, 855. [Google Scholar] [CrossRef]
- Martin, W. H.; Stecker, M. M. ASNM Position Statement: Intraoperative monitoring of auditory evoked p otentials. Journal of Clinical Monitoring and Computing 2007, 22, 75–85. [Google Scholar] [CrossRef]
- Odom, J. V.; Bach, M.; Brigell, M.; Holder, G. E.; McCulloch, D. L.; Mizota, A.; Tormene, A. P. ISCEV standard for clinical visual evoked potentials: (2016 update). Documenta Ophthalmologica 2016, 133, 1–9. [Google Scholar] [CrossRef]
- Joo, B.-E.; Park, S.-K.; Cho, K.-R.; Kong, D.-S.; Seo, D.-W.; Park, K. Real-time intraoperative monitoring of brainstem auditory evoked potentials during microvascular decompression for hemifacial spasm. Journal of Neurosurgery 2016, 125, 1061–1067. [Google Scholar] [CrossRef]
- Dimakopoulos, V.; Selmin, G.; Regli, L.; Sarnthein, J. Optimization of signal-to-noise ratio in short-duration SEP recordings by variation of stimulation rate. Clinical Neurophysiology 2023, 150, 89–97. [Google Scholar] [CrossRef]
- Stecker, M. M. Generalized averaging and noise levels in evoked responses. Computers in Biology and Medicine 2000, 30, 247–265. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, D. B.; Zayed, Z. A.; Stigsby, B. Tibial somatosensory evoked potential intraoperative monitoring: Recommendations based on signal to noise ratio analysis of popliteal fossa, optimized P37, standard P37, and P31 potentials. Clinical Neurophysiology 2005, 116, 1858–1869. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, D. B.; Al-Zayed, Z.; Stigsby, B.; Al-Homoud, I. Median somatosensory evoked potential intraoperative monitoring: Recommendations based on signal-to-noise ratio analysis. Clinical Neurophysiology 2009, 120, 315–328. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, D. B.; Dong, C.; Quatrale, R.; Sala, F.; Skinner, S.; Soto, F.; Szelényi, A. Recommendations of the International Society of Intraoperative Neurophysiology for intraoperative somatosensory evoked potentials. Clinical Neurophysiology 2019, 130, 161–179. [Google Scholar] [CrossRef]
- MacDonald, D. B. Monitoring somatosensory evoked potentials. In Elsevier eBooks; 2020; pp 35–51. [CrossRef]
- Dietrich, C.; Blume, K. R.; Franz, M.; Huonker, R.; Carl, M.; Preißler, S.; Hofmann, G. O.; Miltner, W. H. R.; Weiss, T. Dermatomal organization of SI leg representation in humans: Revising the somatosensory homunculus. Cerebral Cortex 2017, 27, 4564–4569. [Google Scholar] [CrossRef]
- Nuwer, M. R.; Comi, G.; Emerson, R.; Fuglsang-Frederiksen, A.; Guérit, J.-M.; Hinrichs, H.; Ikeda, A.; Luccas, F. J. C.; Rappelsburger, P. IFCN standards for digital recording of clinical EEG. Electroencephalography and Clinical Neurophysiology 1998, 106, 259–261. [Google Scholar] [CrossRef]
- Hanson, C.; Lolis, A. M.; Beric, A. SEP montage variability comparison during intraoperative neurophysiologic monitoring. Frontiers in Neurology 2016, 7, 105. [Google Scholar] [CrossRef]
- De Weerd, J. P. C.; Kap, J. I. Spectro-temporal representations and time-varying spectra of evoked potentials. Biological Cybernetics 1981, 41, 101–117. [Google Scholar] [CrossRef]
- Allison, D. W.; Silverstein, J. W.; Thirumalai, S. S.; D’Amico, R. S. Misconceptions in IONM Part III: Stimulation repetition rate effects on intraoperative somatosensory evoked potential amplitude and latency. The Neurodiagnostic Journal 2022, 62, 239–250. [Google Scholar] [CrossRef]
- Houlden, D. A.; Turgeon, C. A.; Polis, T.; Sinclair, J.; Coupland, S.; Bourque, P.; Corsten, M.; Kassam, A. Intraoperative flash VEPs are reproducible in the presence of low amplitude EEG. Journal of Clinical Monitoring and Computing 2013, 28, 275–285. [Google Scholar] [CrossRef]
- Shcherbuk A. Yu.; Shcherbuk Yu. A.; Pyanzin S. Yu. Intraoperative monitoring of visual evoked potentials as a component of a comprehensive neurophysiological supply of transnasal endoscopic interventions for tumors of chiasmatic-sellar region. Rossiiskii neirokhirurgicheskii zhurnal im. prof. A.L. Polenova = Russian Journal of Neurosurgery n. a. prof. A.L. Polenov 2011, 3(4), 37–43. (In Russ.).
- Kodama, K.; Goto, T. Neurophysiology of the visual system: basics and intraoperative neurophysiology techniques. In Elsevier eBooks; 2020; pp 53–64. [CrossRef]
- Vijayan, S.; Ching, S.; Purdon, P. L.; Brown, E. N.; Kopell, N. J. Thalamocortical Mechanisms for the Anteriorization of Alpha Rhythms during Propofol-Induced Unconsciousness. Journal of Neuroscience 2013, 33, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Blain-Moraes, S.; Tarnal, V.; Vanini, G.; Alexander, A.; Rosen, D.; Shortal, B.; Janke, E.; Mashour, G. A. Neurophysiological correlates of sevoflurane-induced unconsciousness. Anesthesiology 2014, 122, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Kodama, K.; Goto, T.; Sato, A.; Sakai, K.; Tanaka, Y.; Hongo, K. Standard and limitation of intraoperative monitoring of the visual evoked potential. Acta Neurochirurgica 2010, 152, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Sato, A. Interpretation of the causes of instability of flash visual evoked potentials in intraoperative monitoring and proposal of a recording method for reliable functional monitoring of visual evoked potentials using a light-emitting device. Journal of Neurosurgery 2016, 125, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, I.; Park, H.; Kotsugi, M.; Yokoyama, S.; Omoto, K.; Myochin, K.; Takeshima, Y.; Matsuda, R.; Nishimura, F.; Yamada, S.; Takatani, T.; Nakase, H. Diagnostic impact of monitoring visual evoked potentials to prevent visual complications during endovascular treatment for intracranial aneurysm. Frontiers in Neurology 2022, 13, 761263. [Google Scholar] [CrossRef]
- Sasaki, T.; Itakura, T.; Suzuki, K.; Kasuya, H.; Munakata, R.; Muramatsu, H.; Ichikawa, T.; Sato, T.; Endo, Y.; Sakuma, J.; Matsumoto, M. Intraoperative monitoring of visual evoked potential: introduction of a clinically useful method. Journal of Neurosurgery 2010, 112, 273–284. [Google Scholar] [CrossRef]
- Gutzwiller, E. M.; Cabrilo, I.; Radovanovic, I.; Schaller, K.; Boëx, C. Intraoperative monitoring with visual evoked potentials for brain surgeries. Journal of Neurosurgery 2018, 1–7. [Google Scholar] [CrossRef]
- Levin, E. A.; Kilchukov, M. G.; Glushaeva, A. A. Intraoperative monitoring of visual evoked potentials: experience of 240 operations. Russian Journal of Neurosurgery 2024, 26, 57–71. [Google Scholar] [CrossRef]
- San-Juan, D.; Cortés, M. E.; Tena-Suck, M.; Garduño, A. J. O.; Pizano, J. A. L.; Domínguez, J. V.; Gónzalez-Aragón, M. F.; Gómez-Amador, J. L. Neurophysiological intraoperative monitoring during an optic nerve schwannoma removal. Journal of Clinical Monitoring and Computing 2016, 31, 1059–1064. [Google Scholar] [CrossRef]
- Uribe, A. A.; Mendel, E.; Peters, Z. A.; Shneker, B. F.; Abdel-Rasoul, M.; Bergese, S. D. Comparison of visual evoked potential monitoring during spine surgeries under total intravenous anesthesia versus balanced general anesthesia. Clinical Neurophysiology 2017, 128, 2006–2013. [Google Scholar] [CrossRef]
- Seubert, C. N.; Herman, M. Auditory-Evoked potentials. In Monitoring the Nervous System for Anesthesiologists and Other Health Care Professionals; Koht, A., Sloan, T., Toleikis, J., Eds.; Springer, 2017; pp 35–49. [CrossRef]
- Zamel, K. M. Brainstem auditory evoked potential monitoring. In Cambridge University Press eBooks; 2010; pp 90–100. [CrossRef]
- Greve, T.; Beyer, F.; Szelényi, A. Intraoperative Erb’s Point-Vertex recording increases brainstem auditory evoked potential wave V amplitude. Clinical Neurophysiology 2019, 131, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Hammerschlag, P. E.; Berg, H. M.; Prichep, L. S.; John, E. R.; Cohen, N. L.; Ransohoff, J. Real-Time Monitoring of Brainstem Auditory Evoked Response (BAER) during Cerebellopontine Angle (CPA) Surgery. Otolaryngology 1986, 95, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Nuwer, M. R. Introduction, history, and staffing for intraoperative monitoring. In Intraoperative Neurophysiologic Monitoring; Galloway, G. M., Nuwer, M. R., Lopez, J. R., Zamel, K. M..; Cambridge University Press, 2010; pp 1–9. [CrossRef]
- Clinical practice guidelines for intraoperative neurophysiological monitoring: 2020 update. Annals of Clinical Neurophysiology 2021, 23, 35–45. [CrossRef]
- Commission on Accreditation of Allied Health Education Programs. Standards and guidelines for the accreditation of educational programs in intraoperative neurophysiologic monitoring. Available online: https://cdn.prod.website-files.com/5f466098572bfe97f28d59df/614b674da83bc04a1fab718a_IONM_Standards_Approve3192021.pdf (accessed on 18 November 2024).
- Grundy, B.L. Monitoring of sensory evoked potentials during neurosurgical operations: methods and applications. Neurosurgery 1982, 11, 556–575. [Google Scholar] [CrossRef]
- Fried, S. J.; Smith, D. M.; Legatt, A. D. Median nerve somatosensory evoked potential monitoring during carotid endarterectomy. Journal of Clinical Neurophysiology 2014, 31, 55–57. [Google Scholar] [CrossRef]
- Rajan, S.; Simon, M. V.; Nair, D. Intraoperative visual evoked potentials: there is more to it than meets the eye. Journal of Neurology and Neuroscience 2016, 7, 106. [Google Scholar] [CrossRef]
- Burns, P. B.; Rohrich, R. J.; Chung, K. C. The levels of evidence and their role in Evidence-Based Medicine. Plastic & Reconstructive Surgery 2011, 128, 305–310. [Google Scholar] [CrossRef]
- Jin, S.-H.; Chung, C. K.; Kim, J. E.; Choi, Y. D. A new measure for monitoring intraoperative somatosensory evoked potentials. Journal of Korean Neurosurgical Society 2014, 56. [Google Scholar] [CrossRef]
- Park, S.-K.; Lee, H.S.; Cho, K.R.; Park, K. Recent advances in intraoperative brainstem auditory evoked potential monitoring during microvascular decompression surgery for hemifacial spasm. Life 2023, 13, 1789. [Google Scholar] [CrossRef]
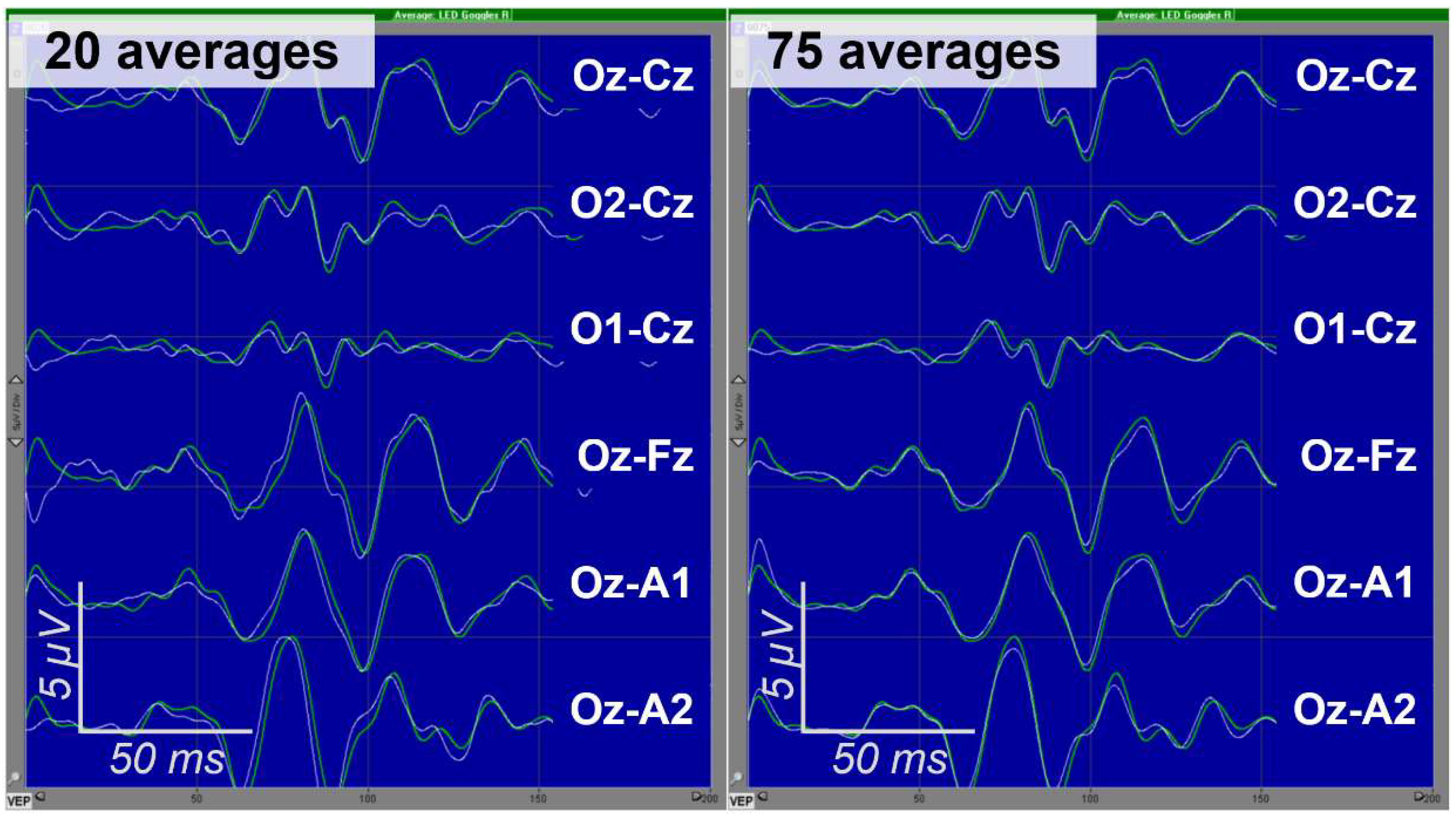
| Recording of EPs during clinical neurophysiological diagnostics | Intraoperative monitoring of EPs | |
|---|---|---|
| Task | Identification of existing sensory impairments | Detection of emerging new sensory dysfunctions |
| Dysfunction detection method | Comparison with population standart | Comparison with the same patient’s records obtained at the beginning of the operation |
| Conditions | Patient is conscious; external interference is minimized; electromyographic interference may be significant |
Patient is under general anesthesia; external interference (electrocoagulation, mechanical, etc.) is usual; electromyographic interference can be reduced by muscle relaxation |
| Registration parameters | Same as for the normative population, same for all patients | Changes during monitoring are undesirable, but differences between patients are acceptable |
| Time limits | Nosignificant time limitations | It is necessary to update records as often as possible |
| Publication | Locations of recording electrodes | Filter passband | Number of averages | Stimulation rate |
|---|---|---|---|---|
| Kodama et al., 2010 [27] * | P7/O1/Oz/O2/P8 – (A1+A2) | 10 – 1000 Hz | 40 – 100 | 1/sec |
| Sasaki et al., 2010 [30] | O1 – M1, O2 – M2 | 20 – 500 Hz | 100 | 1/sec |
| Scherbuk et al., 2011 [23] | O1/O2/Oz – Fz | 2 – 100 Hz | 200 | 1/sec |
| Houlden et al., 2013 [22] | Oz – Fz | 1–5 – 100 Hz | 150 | 1.41/sec |
| Luo et al., 2015 [2] | O1/Oz/O2 – Fz/Cz/(A1+A2) | 5 – 100 Hz | 100 – 200 | ND |
| San-Juan et al., 2016 [33] | O1–O2, O1–Oz, O2–Oz, Oz–Fz | 2 – 100 Hz | 250 | 4.1/sec |
| Sato, 2016 [28] * | P7/PO7/O1/Oz/O2/PO8/P8 – (A1+A2) | 0.1 – 200 Hz | 100 | 1/sec |
| Uribe et al., 2017 [34] | O1/Oz/O2 – M1/M2/Fz | 20 – 500 Hz | 100 | 1.1/sec |
| Gutzwiller et al. 2018 [31] | O1/Oz/O2 – CPz | 2 – 400 Hz | 50 | 0.9/sec |
| Kodama, Goto, 2020 [24] * | P7/O1/Oz/O2/P8 – (A1+A2) | 10 – 1000 Hz | 40 – 100 | 1/sec |
| Nakagawa et al., 2022 [29] | O1/Oz/O2 – (A1+A2) | ND | 100 | 1/sec |
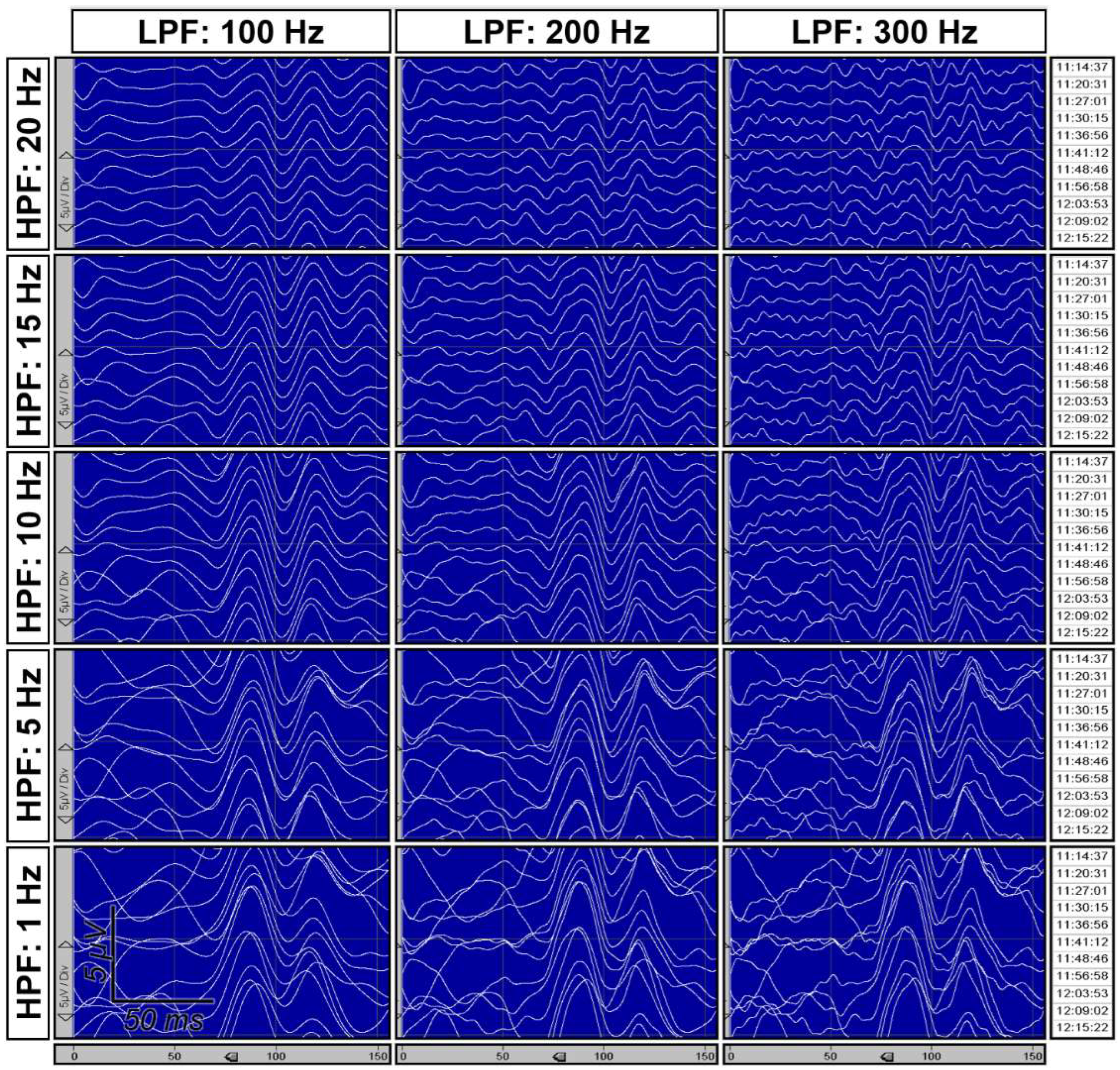
| High-pass filter, Hz | The number of eyes for which VEPs were recorded using the specified frequency filters, n | ||||||
|---|---|---|---|---|---|---|---|
| Low-pass filter, Hz | Total | ||||||
| 100 | 150 | 200 | 250 | 300 | 350-400 | ||
| 5 | 1 | 6 | 29 | 1 | 0 | 0 | 37 |
| 10 | 2 | 47 | 196 | 13 | 26 | 2 | 286 |
| 15 | 2 | 4 | 74 | 6 | 7 | 1 | 94 |
| 20 | 0 | 2 | 1 | 0 | 0 | 1 | 4 |
| Total | 5 | 59 | 300 | 20 | 33 | 4 | 421 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




