Submitted:
18 November 2024
Posted:
19 November 2024
You are already at the latest version
Abstract
The study focused on developing transungual patches for the targeted treatment of onychomycosis, a common fungal infection of the nail. By incorporating Sertaconazole nitrate into patches using the Self-Microemulsifying Drug Delivery System (SMEDDS) technique, the aim was to overcome challenges related to drug penetration across the nail plate while enhancing solubility and permeability. Various patch formulations were prepared and extensively evaluated for key parameters such as thickness, weight variation, drug content, folding endurance, moisture content, moisture uptake, and in vitro drug release. The release data were analyzed using different kinetic models to understand the drug release mechanism. Selected formulations underwent accelerated stability testing per ICH guidelines, demonstrating stability under accelerated conditions. The study successfully addressed the challenges of transungual drug delivery, with the developed patches showing promising characteristics and stable release profiles. The application of kinetic models provided valuable insights, and the formulations passed stability testing, indicating their potential as effective and stable transungual drug delivery systems for the treatment of onychomycosis.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Emulsion Stability Testing
- Freeze-Thaw Cycles: Emulsions underwent three freeze-thaw cycles, alternating between -5°C for 24 hours and 35°C for the next 24 hours, and were checked for precipitation, phase separation, or turbidity.
- Heat-Cool Cycles: Emulsions were subjected to three heat-cool cycles by heating at 40°C for 6 hours, followed by cooling in an ice bath and maintaining at room temperature for 18 hours. Visual inspections were done for signs of instability.
- Centrifugation: Emulsions were centrifuged at 1000 rpm for 15 minutes and assessed for phase separation or turbidity.
2.2. Dispersibility Test
- Grade A: Forms a clear or bluish nano-emulsion within 1 minute.
- Grade B: Forms a slightly less clear bluish-white emulsion within 1 minute.
- Grade C: Forms a fine milky emulsion within 2 minutes.
- Grade D: Forms a dull, greyish-white emulsion with a slightly oily appearance, taking longer than 2 minutes to emulsify.
- Grade E: Shows poor emulsification with large oil globules on the surface.
2.3. Preparation of Micro Emulsion Concentrate
2.4. Selection of Self Microemulsifying Drug Delivery System
2.5. Characterization of SMEDDS
2.5.1. Drug Content
2.5.2. Particle size Distribution
2.5.3. Zeta Potential
2.6. Formulation of Transungual Patches
2.6.1. Formulation of Backing Membrane
2.6.2. Formulation of Plain Drug Matrix
2.6.3. Drug in Transcutol
2.6.4. Drug in Self Microemulsifying Drug Delivery System
2.7. Evaluation of Transungual Patches
2.7.1. Appearance
2.7.2. Folding Endurance
2.7.3. Percentage Moisture Loss
2.7.4. Percentage of Moisture Uptake
2.7.5. Thickness and Weight Variation
2.7.6. Drug Content
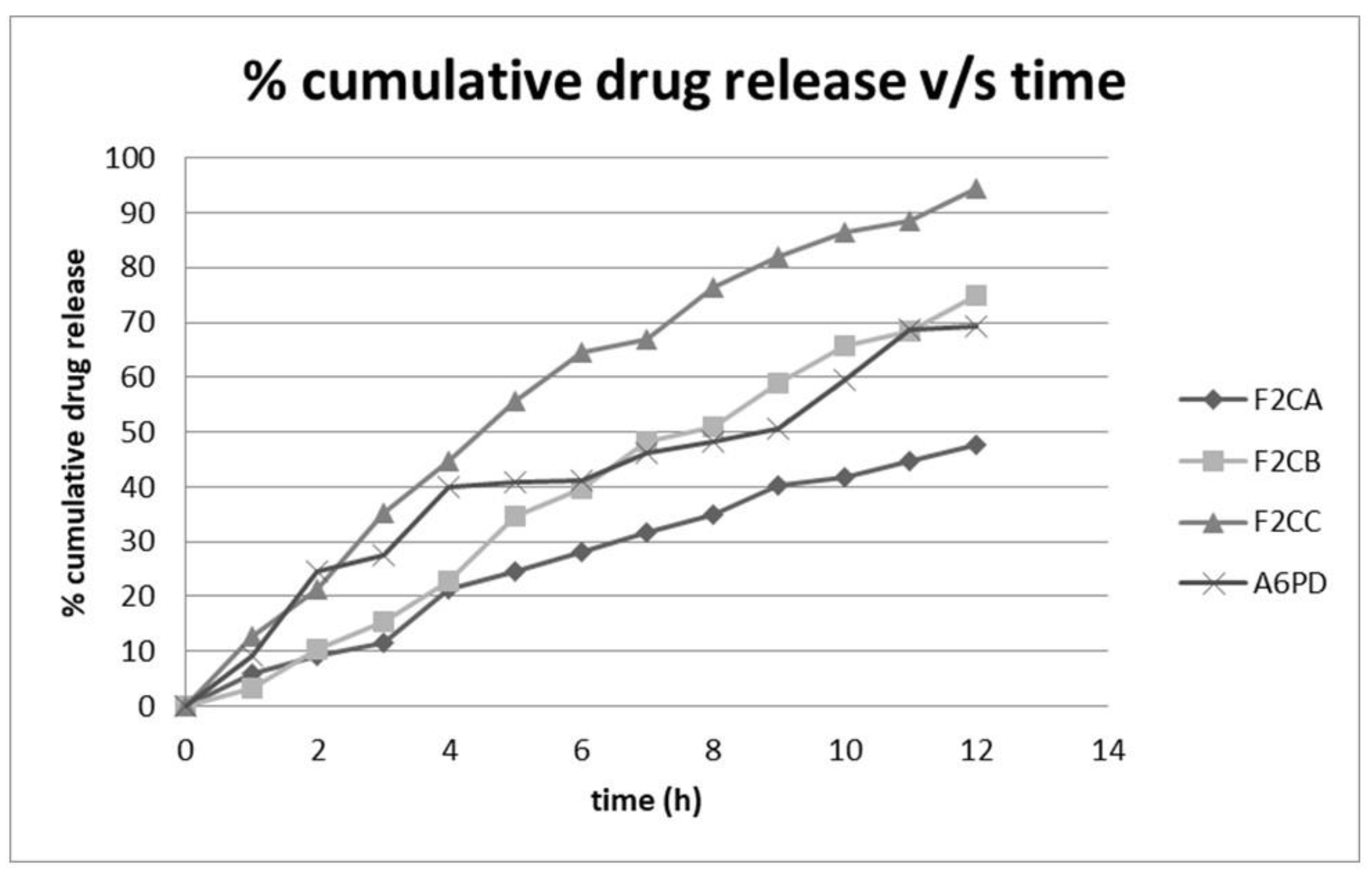
2.7.7. In-Vitro Diffusion Study
2.7.8. Kinetic Analysis
Results
3. Discussion
4. Conclusion
References
- Zaias, N. The nail in health and disease. 2nd edition. Appleton and Lange, Connecticut; 1990:1-255.
- Chabasse D, Baran R, De Chauvin MF. Onychomycosis 1: epidemiology and etiology. J Mycol Med. 2000; 10:177-190.
- Baran R, Hay R, Tosti A and Haneke E. A new classification of onychomycosis. Br J Dermatol. 1998; 139:567-571.
- Baran R, Kaoukhov A. Topical antifungal drugs for the treatment of onychomycosis: an overview of current strategies for monotherapy and combination therapy. J Eur Acad Dermatol Venereol. 2005; 19:21-29.
- Barot B, Parejiya P, Patel H, Mehta D, Shelat P. Microemulsion-based antifungal gel delivery to nail for the treatment of onychomicosis: formulation, optimization, and efficacy studies. AAPS PharmSciTech. 2012 March; 13(1):184-192.
- Bhowmik D, Kumar K. Recent trends in dermal and transdermal drug delivery systems: current and future prospects. Pharma Innov. 2013; 2(6).
- Bseiso E, Nasr M, Sammour O. Novel nail penetration enhancer containing vesicles nPEVs for the treatment of onychomicosis. J Drug Deliv. 2: 2016; 23(8), 2016.
- Chein, YW. Transdermal drug delivery and delivery system. Novel Drug Delivery System. 2nd ed. New York: M. Dekker; 2009; 50(2): 301-380.
- Chen Y, Quan P, Liu X, Wang M. Fang L. Review on novel chemical permeation enhancers for transdermal drug delivery. Asian J Pharm Sci. 2014; 9:51-64.
- Chouhan P, Saini T. Hydration of nail plate: a novel screening model for transungual drug permeation enhancers. Int J Pharm. 2012; 426(1-2):179-182.
- Chouhan P, Saini T. D-optimal Design and development of microemulsion based transungual drug delivery formulation of ciclopirox olamine for the treatment of onychomicosis. Indian J Pharm Sci. 2016; 78(4):498-511.
- Chouhan P, Saini TR. Hydroxypropyl-ß-cyclodextrin: A novel transungual permeation enhancer for development of topical drug delivery system for onychomycosis. J Drug Deliv. 1: 2014, 2014.
- Cohen P, Scher R. Topical and surgical treatment of onychomycosis. J Am Acad Dermatol. 1994; 31:74-77.
- Dash S, Murthy P, Nath L, Chowdhary P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Poloniae Pharmaceutica Drug Research. 2010; 67(3):217-223.
- Deepthi BV, Poornachandra Rao K, Chennapa G, Naik MK, Chandrashekara KT, Sreenivasa MY. Antifungal attributes of Lactobacillus plantarum MYS6 against fumonisin producing Fusarium proliferatum associated with poultry feeds. PLoS One. 2016 Jun 10;11(6):e0155122.
- Higuchi, T. Mechanism of sustained- action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. Journal of Pharmaceutical Sciences. 1963; 52:114-1149.
- Jha S, Dey S, Karki R. Microemulsions- potential carrier for improved drug delivery.Asian Journal of Biomedical and Pharmaceutical Sciences. 2011; 1(1):5-9.
- Kansagra H, Subrata M, Asif K. Formulation and evaluation of transdermal patch of sertaconazole nitrate. International research Journal of Pharmacy. 2012; 3(11):109-113.
| Formulation code | S: CoS ratio (Smix) | Oil: Smix ratio | Amount of Sertaconazole nitrate added (mg) | Total volume of mixture (ml) |
|---|---|---|---|---|
| A1 | 1:1 | 1:4 | 80 | 2 |
| A2 | 1:1 | 1:2:3 | 80 | 2 |
| A3 | 1:2 | 1:4 | 80 | 2 |
| A4 | 1:2 | 1:2:3 | 80 | 2 |
| Formulation code | Backing membrane (Ethyl cellulose) | Dibutyl phthalate | Propylene glycol | Propylene glycol Glycerine |
|---|---|---|---|---|
| A1 | 600 | 30% | - | - |
| A2 | 600 | - | 30% | - |
| A3 | 600 | - | - | 30% |
| A4 | 600 | 15 | - | 15% |
| A5 | 600 | - | 15% | 15% |
| A6 | 600 | 15% | 15% | - |
| Formulation code Reservoir |
Plasticizer | |||
|---|---|---|---|---|
| Dibutylpthalate | Propylene glycol | Glycerine | ||
| A1P | 1% | 30% | - | - |
| A2P | 1% | - | 30% | - |
| A3P | 1% | - | - | 30% |
| A4P | 1% | 15% | - | 15% |
| A5P | 1% | - | 15% | 15% |
| A6P | 1% | 15% | 15% | - |
| Formulation code Reservoir |
Plasticizer | |||
|---|---|---|---|---|
| Dibutylpthalate | Propylene glycol | glycerine | ||
| F1C | 2% | 30% | - | - |
| F2C | 2% | - | 30% | - |
| F3C | 2% | - | - | 30% |
| F4C | 2% | 15% | - | 15% |
| F5C | 2% | - | 15% | 15% |
| F6C | 2% | 15% | 15% | - |
| Formulation code | Penetration enhancer | SMEDDS system | Sertaconazole nitrate |
|---|---|---|---|
| F2CA | - | - | 80mg |
| F2CB | 2ml | - | 80mg |
| F3CC | - | 2ml | 80mg |
| A6PD | - | - | 80mg |
| Formulation code | S:CoS ratio (Smix) | Oil:Smix ratio | Freeze Thawing | Heat cool cycle | centrifugation |
|---|---|---|---|---|---|
| A1 | 1:1 | 1:4 | |||
| A2 | 1:1 | 1:2:3 | |||
| A3 | 1:2 | 1:4 | |||
| A4 | 1:2 | 1:2:3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




